Articles
- Page Path
- HOME > J Pathol Transl Med > Volume 49(3); 2015 > Article
-
Review
Pathology-MRI Correlation of Hepatocarcinogenesis: Recent Update - Jimi Huh1, Kyung Won Kim,1,2, Jihun Kim2,3, Eunsil Yu2,3
-
Journal of Pathology and Translational Medicine 2015;49(3):218-229.
DOI: https://doi.org/10.4132/jptm.2015.04.15
Published online: May 15, 2015
1Department of Radiology, Asan Medical Center, University of Ulsan College of Medicine, Seoul, Korea
2Asan Liver Center, Asan Medical Center, University of Ulsan College of Medicine, Seoul, Korea
3Department of Pathology, Asan Medical Center, University of Ulsan College of Medicine, Seoul, Korea
- Corresponding Author Kyung Won Kim, M.D., Ph.D. Department of Radiology, Asan Medical Center, University of Ulsan College of Medicine, 88 Olympic-ro 43-gil, Songpa-gu, Seoul 138-736, Korea Tel: +82-2-3010-4377 Fax: +82-2-3010-8724 E-mail: medimash@gmail.com
© 2015 The Korean Society of Pathologists/The Korean Society for Cytopathology
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/3.0/) which permits unrestricted noncommercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
Abstract
- Understanding the important alterations during hepatocarcinogenesis as well as the characteristic magnetic resonance imaging (MRI) and histopathological features will be helpful for managing patients with chronic liver disease and hepatocellular carcinoma. Recent advances in MRI techniques, such as fat/iron quantification, diffusion-weighted images, and gadoxetic acid-enhanced MRI, have greatly enhanced our understanding of hepatocarcinogenesis.
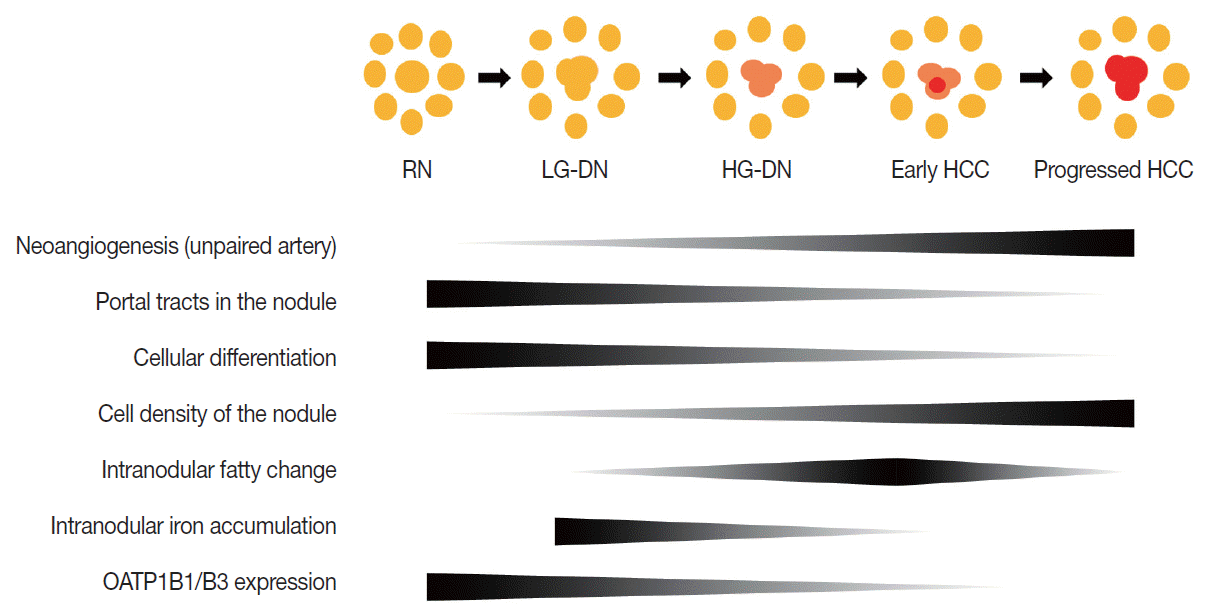
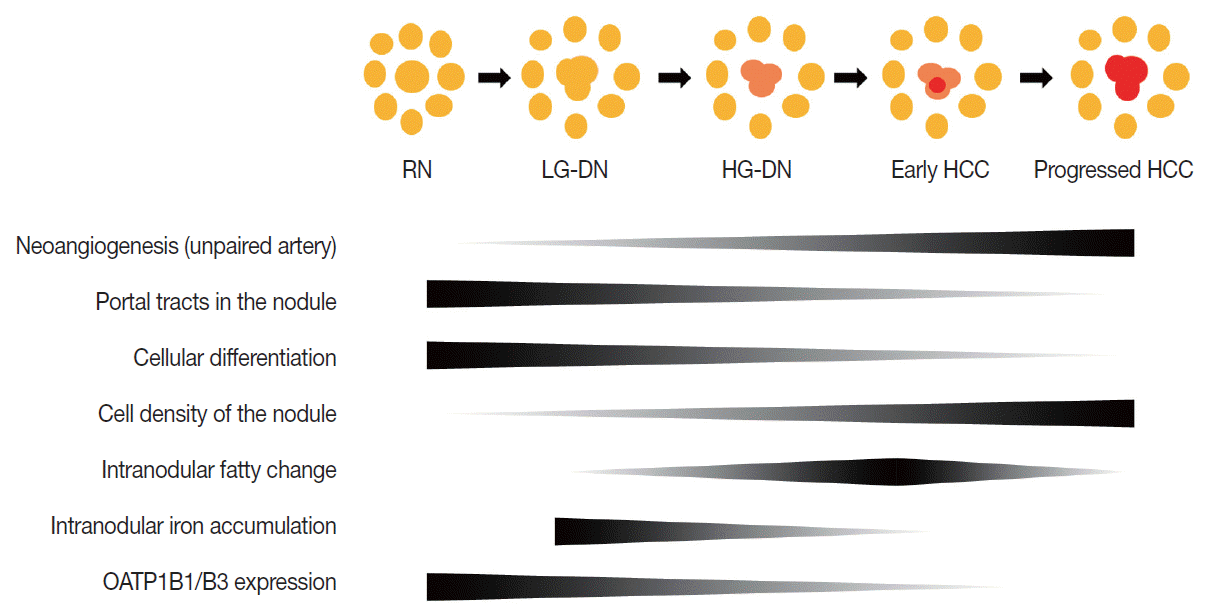
- Multistep hepatocarcinogenesis is characterized by progressive dedifferentiation of phenotypically abnormal nodular lesions in the liver and the emergence of successively more advanced precancerous, early cancerous, and overtly cancerous lesions (Fig. 1) [7,8]. Chronic inflammation causes repeated injury to liver cells and regeneration of injured tissue, both of which promote accumulation of genetic and epigenetic alterations [9-12]. These alterations begin from the early preneoplastic phase and progress parallel to the evolution of hepatic fibrosis or cirrhosis [13,14].
- This linear, repetitive, and successive process of the expansion and development of less differentiated, aberrant clonal populations of hepatocytes causes their evolution over time [15]. These dedifferentiated clonal populations eventually completely replace those of the more differentiated environment. Repetitive clonal growth and expansion ultimately generates nodules of the malignant phenotype. The dedifferentiated evolution process occurs in serial order as a biologic continuum in vivo; however, these processes are divided into discrete steps for clinical use [16].
- In 1995, after the stepwise development of HCC from regenerating nodules of liver cirrhosis was proposed, the International Working Party (IWP) of the World Congress of Gastroenterology defined regenerative nodules (RNs), low-grade dysplastic nodules (DNs), high-grade DNs, and HCC as steps from regeneration to cancer [17,18]. In addition, the IWP defined small HCCs as tumors less than 2 cm in diameter. However, there had been debate regarding the definition of small HCC, as these tumors can be divided into early HCCs, i.e., an early stage of hepatocarcinogenesis, and progressed HCCs, i.e., advanced HCC less than 2 cm in diameter. In addition, the accurate pathological definition of early HCCs differentiated from DNs was not established until recently. Finally, in 2009 the International Consensus Group for Hepatocellular Neoplasia (ICGHN) reached a consensus regarding the pathological criteria of early HCCs, as discussed below [19]. These international efforts have contributed greatly to the standardization of the nomenclature regarding nodules during hepatocarcinogenesis.
- Regenerative nodules
- A RN, also known as a cirrhotic nodule, is a well-defined nodular region surrounded by fibrotic/scar tissue, which has emerged from the regenerative process of the injured liver tissue. In RNs, the cells are histologically normal and lack clonal features [18]. A RN is considered a benign hepatocellular nodule.
- Dysplastic nodules
- DNs are small nodules which are in the precancerous stage between benign RNs and malignant HCC. DNs usually differ from the surrounding liver parenchyma in color, texture, and cellular change [18]. DNs are subdivided into low-grade DN and high-grade DN.
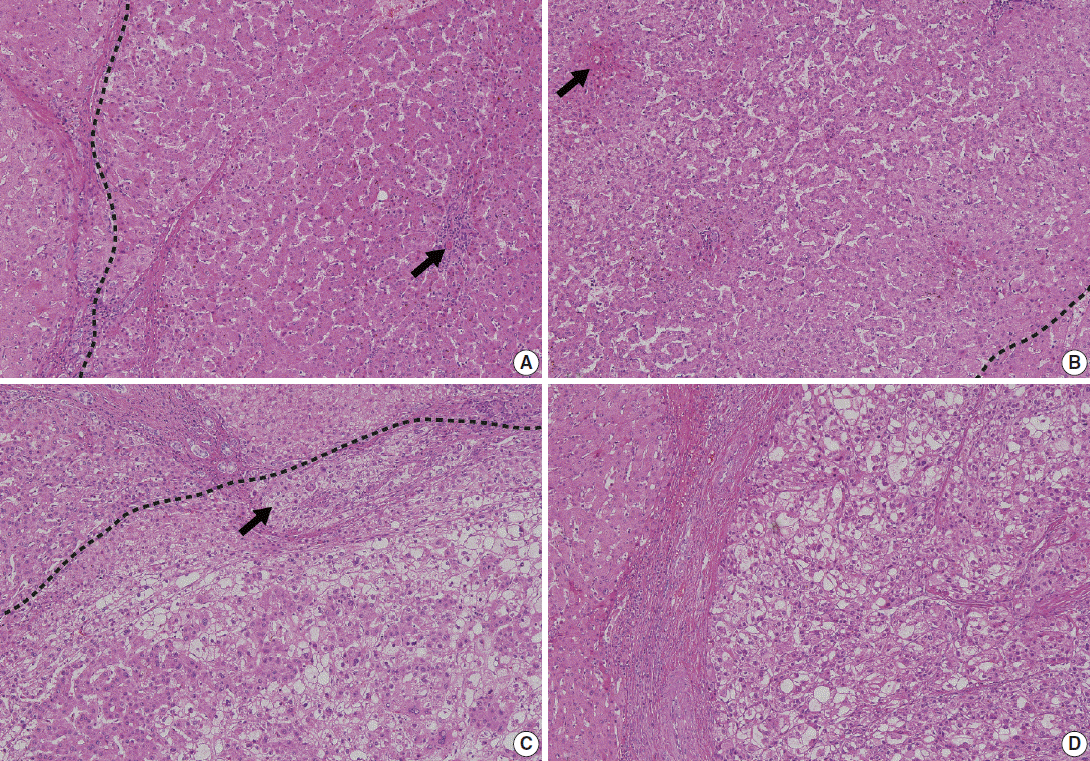
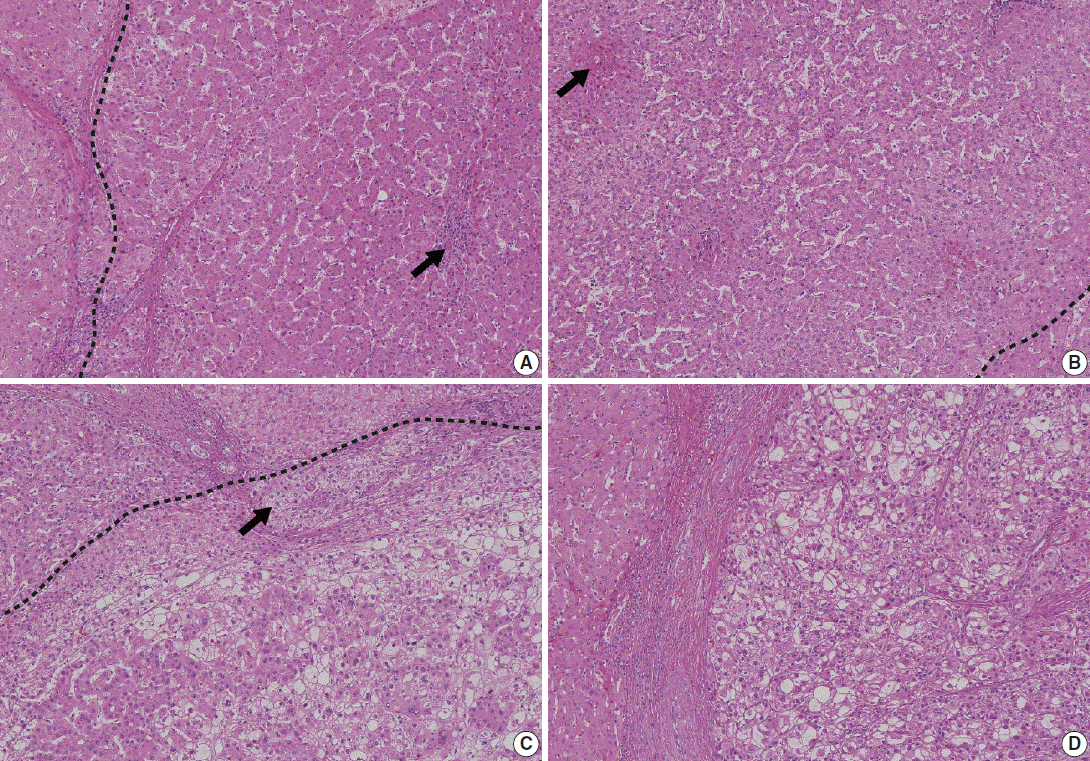
- Low-grade DNs show features suggestive of a clonal cell population without significant architectural or cellular atypia (Fig. 2A). Occasionally, low-grade DNs demonstrate large cell change, although they do not show small cell change. Low-grade DNs can show a mild increase in cell density with a monotonous pattern. A nodule-in-nodule pattern is not present in low-grade DNs [18,20].
- In contrast, high-grade DNs show cytological and architectural atypia, although those changes are insufficient for a diagnosis of malignancy [18]. Small cell change is the most frequently seen cytological atypia. The architectural atypia observed in high-grade DNs includes the thick cell plates up to three cells thick and/or pseudoglandular structures (Fig. 2B). The cell density of these lesions is usually 1.3–2 times greater than that of the adjacent cirrhotic parenchyma [19].
- Early HCC
- In 2009, the ICGHN made a consensus for the definition of early HCC [19] Early HCCs are considered ‘‘in situ carcinoma’’ showing well-differentiated proliferation [18,20]. Early HCCs are referred to as ‘small HCC of the vaguely nodular type.’ The most important histologic feature that distinguishes early HCCs from high-grade DNs is stromal invasion, defined as the presence of tumor cells invading the portal tracts or fibrous septa (Fig. 2C). Early HCCs also show various combinations of the following histologic features: “(1) increased cell density more than two times that of the surrounding liver parenchyma and with an increased nuclear/cytoplasm ratio and irregular, thin trabecular pattern; (2) varying numbers of portal tracts within the nodule (intratumoral portal tracts); (3) a pseudoglandular pattern; (4) diffuse fatty change; and (5) various numbers of unpaired arteries [19].”
- There may be confusion regarding the terminology of early HCC and small HCC. By the definition of the IWP consensus in 1995, small HCC is a tumor less than 2 cm, whereas early HCC refers to well-differentiated HCC of the vaguely nodular type during the hepatocarcinogenesis process. The question has remained for small HCCs with moderate or poor differentiation. After much debate, such tumors are now termed “small progressed HCC” or “small HCC of the distinctively nodular type.” In Table 1, the terminology and histologic characteristics of these small, hepatocellular lesions are summarized.
- Progressed HCC
- Progressed HCCs are overtly malignant hepatocellular lesions. Small lesions with a maximum diameter less than 2 cm usually have distinctly nodular macroscopic features [19], while large lesions greater than 2 cm in diameter, which are termed “large HCCs,” have variable macroscopic features. Although three common macroscopic patterns of large HCCs have been proposed [21], six macroscopic patterns, which were proposed by the Korean Liver Cancer Study Group, are more widely accepted in Korea [22]. Briefly, expanding nodular pattern is defined by one single, dominant mass and is the most common type. In these tumors, mosaic architecture is frequently observed, characterized by the multiple sub-nodules separated by fibrous septations and foci of hemorrhage or necrosis [23]. Nodular and perinodular extension type and infiltrative pattern show a dominant mass with infiltration into adjacent hepatic parenchyma along <50% of tumor border in the former and ≥50% in the latter. Those tumors are usually associated with poorly differentiated cancer cells that spread into the surrounding sinusoids and cell plates [17,24]. Cirrhotomimetic pattern represents widespread infiltration by innumerable small nodules that frequently replace the whole liver [21]. In addition, multinodular confluent type and pedunculated type have also been described [22]. These progressed HCCs with both small and large lesions frequently show macroscopic vascular invasion with involvement of portal veins and less frequently hepatic veins. Sometimes, bile duct invasion may be seen macroscopically.
- On histology, the main hallmark of HCC is that the malignant cells resemble the normal liver, both in architecture and cytology. Progressed HCCs can show different degrees of hepatocellular differentiation ranging from well-differentiated to poorly differentiated. Based on the degree of nuclear anaplasia, the Edmondson and Steiner system was proposed and is widely used to divide HCC into four grades from I to IV [17,25]. The common histological patterns include (1) trabecular pattern, where tumor cells with hepatocellular differentiation are arranged in plates of various thickness, separated by sinusoid vascular spaces (Fig. 2D); (2) pseudoglandular pattern, where gland-like dilatation of the canaliculi are present between tumor cells forming the lumen; (3) compact pattern, which is composed of thick trabeculae compressed into a compact mass; (4) scirrhous pattern, where desmoplastic stroma divides tumor cell masses into smaller nests; and (5) fibrolamellar pattern, which shows characteristic lamellar collagen bundles running between large glassy tumor cells.
- RECENT ADVANCES IN MAGNETIC RESONANCE IMAGING TECHNIQUES
- Recently, the diagnosis of hepatocellular carcinoma has become primarily based on imaging [26]. In clinical practice, computed tomography (CT) is the most widely used imaging modality to diagnose HCC. With the dramatic technical advances in MRI, it is currently more useful than other imaging modalities for diagnosing HCC because it provides better soft-tissue contrast and information regarding cellularity, tissue components, and hemodynamic changes of the hepatocellular nodules. In the recent past, the spatial resolution and scan speed were generally better on CT than on MRI [27,28]; however, recently developed MRI units have fast scanning with high spatial resolution, comparable to CT scanning. Therefore, the use of MRI for the diagnosis of HCC has greatly increased.
- Nonradiologist physicians may not be familiar with MRI sequences and the information that can be obtained from those sequences. Indeed, there are many sequences of liver MRI which may cause confusion. In addition, the interpretation of MRI largely depends on which contrast agent is used. We summarize the commonly used sequences in Table 2 and illustrate the representative images of such sequences in Fig. 3.
- T2-weighted images
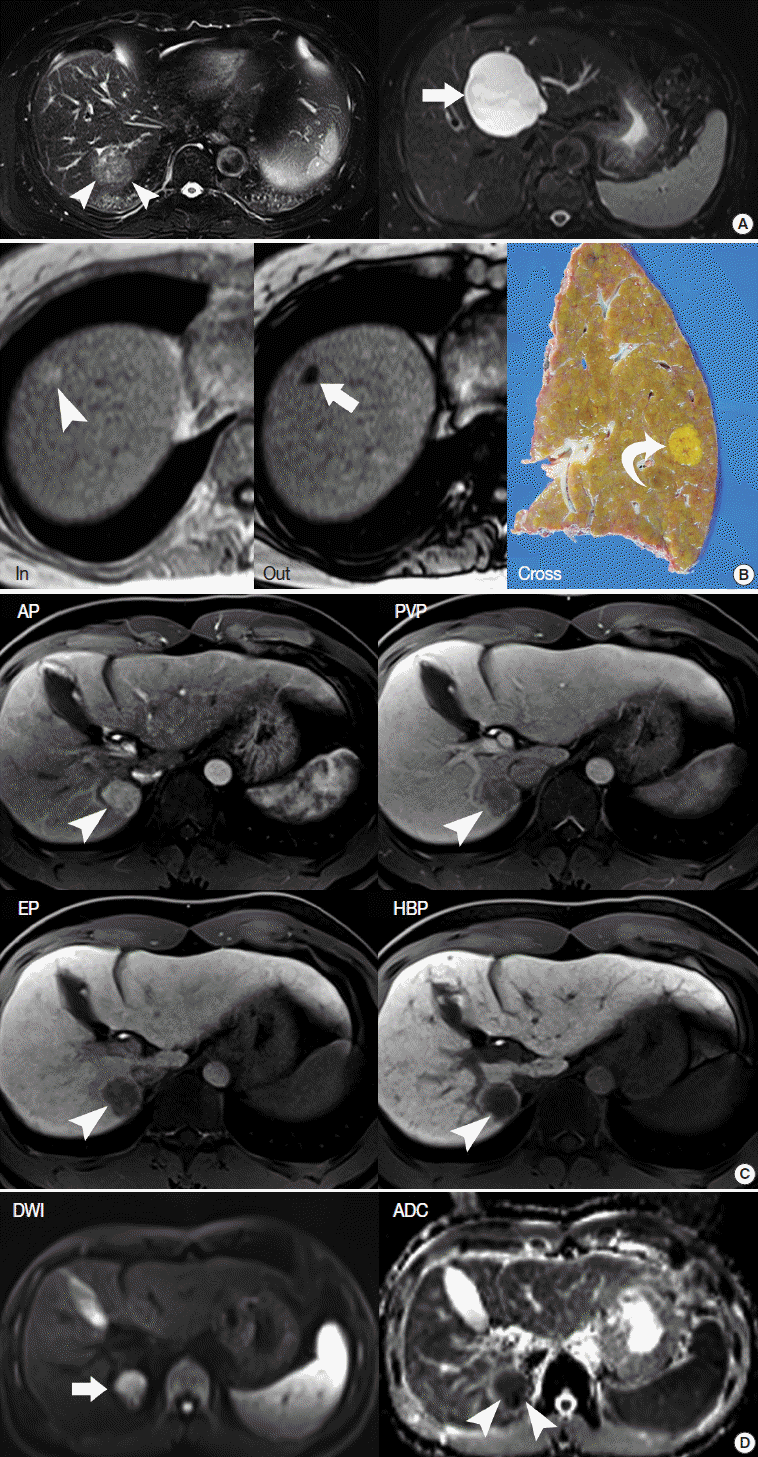
- T2-weighted imaging (T2-WI) highlights the differences in the T2 relaxation time of tissues. Sequences for T2-WI are the basic components of MRI, and are quite helpful for the differential diagnosis of liver tumors. For example, hepatic cysts and hemangiomas show very high T2 hyperintensity, while HCCs show intermediately high T2 hyperintensity (Fig. 3A) [29].
- For routine liver MRI, two kinds of T2-WI sequences are used: sequences moderately weighting T2, including fast spinecho or turbo spin-echo, and sequences exaggerating T2 of tissues, including single-shot fast spin-echo or half-Fourier acquisition single-shot turbo spin-echo [29].
- T1-weighted, dual gradient-echo sequence (in-phase and oppposed-phase)
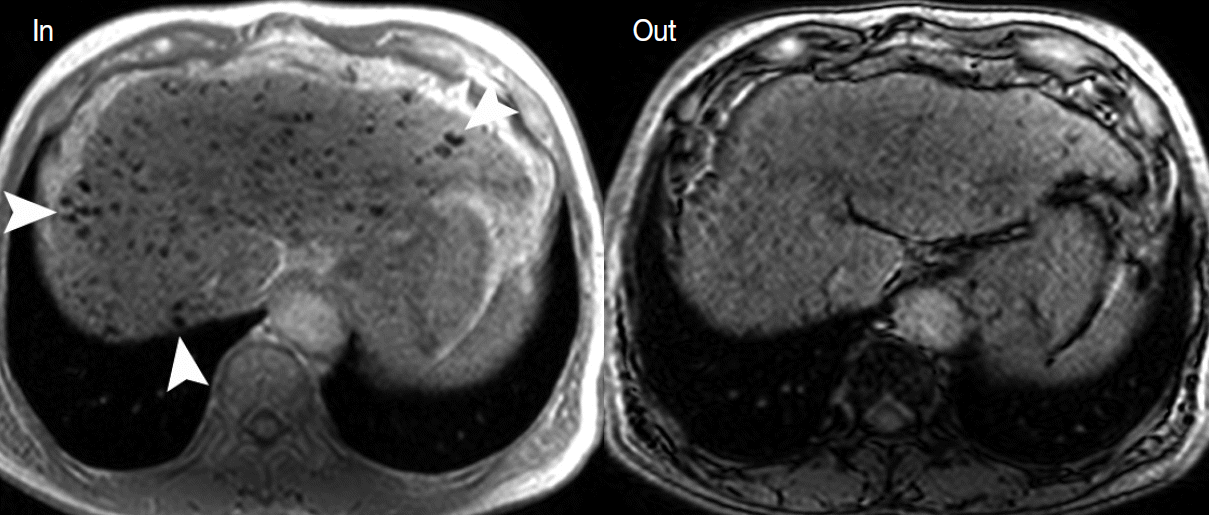
- A T1-weighted, dual gradient-echo sequence is composed of an in-phase image and an opposed-phase image. These images are routine components of the liver MRI protocol, as they can evaluate the fat content of tissues [30]. They are primarily used to identify diffuse or focal hepatic steatosis or to detect the fatty component of liver tumors, such as fat-containing, dysplatic nodules or HCC. When comparing the signal intensity (SI) between in-phase and opposed-phase images, the fatty component of tissue shows a signal drop in the opposed-phase (Fig. 3B).
- This sequence is also helpful for detecting the iron component of tissues using the dual-echo approach. In contrast to the fatty component, the iron component shows a signal drop on inphase images compared to opposed-phase images. Therefore, the pathological conditions, such as hemochromatosis or hemosiderosis, can be identified [7]. It is also helpful to detect iron-containing liver tumors, such as siderotic nodules or iron-containing hepatocellular lesions.
- Multiphasic dynamic imaging
- In liver MRI, contrast agents have crucial roles in the detection and characterization of HCC, based on evaluation of the hemodynamic pattern of liver tumors and liver parenchyma. Multiphasic dynamic imaging consists of repeated acquisition of T1-weighted images (T1-WIs) after contrast agent injection and is composed of the arterial phase, portal-venous phase, and three minutes of the equilibrium phase (Fig. 3C). Enhancement in the arterial phase and washout in the portal or equilibrium phase is regarded as a characteristic feature of HCC on both dynamic CT and dynamic MRI [31].
- Hepatobiliary phase images
- The hepatobiliary phase refers to a 20-minute delayed phase following injection of gadoxetic acid (Fig. 3C). Therefore, the hepatobiliary phase is only performed when gadoxetic acid is used as the contrast agent. Gadoxetic acid is a dual-function agent allowing both dynamic imaging and hepatobiliary-phase imaging. It has characteristics like extracellular contrast agents and can be used in dynamic MRI within three minutes after contrast agent injection. In the hepatobiliary phase, gadoxetic acid is taken up by cells that express the OATP receptor. Normal hepatocytes usually express abundant OATP receptors, while HCCs or liver metastases lack OATP receptors. This results in improved lesion-to-liver contrast because normal liver parenchyma shows high SI while liver tumors show dark SI in the hepatobiliary phase of gadoxetic-acid–enhanced MRI [32].
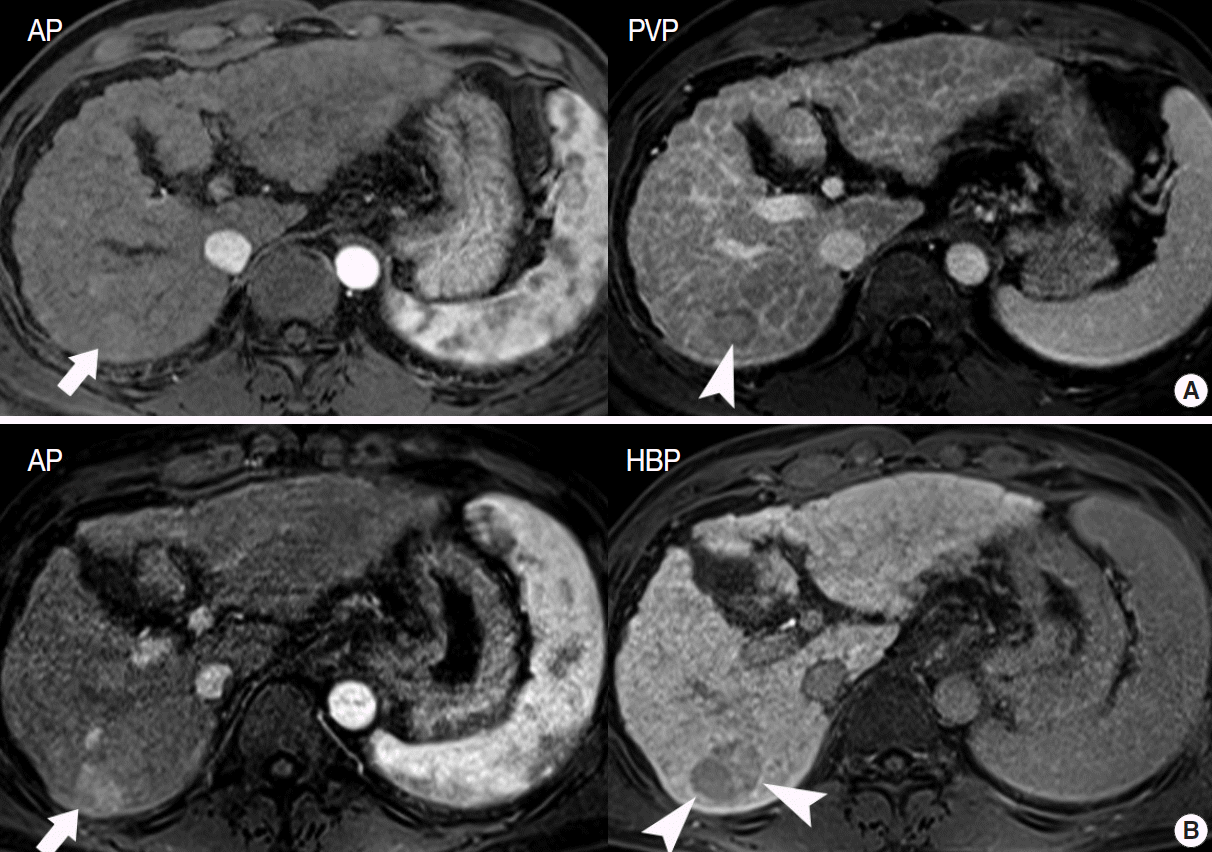
- Gadoxetic acid is excreted into the biliary system. In the hepatobiliary phase it is possible to observe the biliary excretion of gadoxetic acid, which thus allows functional biliary imaging. In liver tissue in which the bile duct is obstructed, biliary excretion is not observed. Regarding liver tumor evaluation, the hepatobiliary phase is sometimes helpful for obtaining a differential diagnosis. For example, focal nodular hyperplasia (FNH) has a functioning, hepatocyte-expressing OATP receptor, although it has no biliary excretion system. Therefore, gadoxetic acid is retained for a long time within an FNH nodule. FNH shows high SI in the hepatobiliary phase, which is helpful for differentiating FNH from HCC [8].
- Diffusion-weighted imaging
- Diffusion-weighted imaging is a sequence to map the diffusion process of molecules, primarily water molecules, in biological tissues. Diffusion is a random motion of water molecules. In tissues or cells, diffusion of water molecules is restricted due to the interactions of molecules [33]. The degree of diffusion restriction varies across tissues, thus reflecting the microscopic details of biological tissues. The apparent diffusion coefficient (ADC) is a quantitative parameter of diffusion restriction calculated from diffusion-weighted images. The ADC map, together with diffusion-weighted images, is quite helpful for visual assessment and quantitative analysis of a liver tumor (Fig. 3D).
- The diffusion of water molecules is highly restricted in dense cellular tissue such as HCC, compared to that of normal liver parenchyma, whereas necrosis or nonviable tumor tissues cause increased membranous permeability allowing free diffusion of water molecules [34]. Therefore, diffusion-weighted images are helpful for detecting highly cellular tumors and for differentiating malignant lesions from benign lesions.
MULTISTEP HEPATOCARCINOGENESIS: HISTOLOGIC CHANGE
- During multistep hepatocarcinogenesis, the important changes in hepatocellular nodules are neo-angiogenesis, cellular differentiation and density, fat or iron accumulation, and biliary drainage dysfunction (Fig. 1). Recent advances in MR technology allow the evaluation of these pathological and functional changes [6,35].
- Alteration of hepatic vasculature
- During hepatocarcinogenesis, hemodynamic change occurs in hepatocellular nodules and can be visualized by radiologic imaging and histopathology [36]. The blood flow of RNs is supplied from both the portal vein (75%–80%) and the hepatic artery (20%–25%), as with normal liver parenchyma. As the hepatocellular nodules evolve from low-grade DN through high-grade DN to early HCC, both the intranodular portal blood supply and the hepatic arterial blood supply tend to decrease [37]. The majority of these nodules are still hypovascular on dynamic MRI.9 As the grade of HCCs increases, i.e., Edmondson grade II or greater, the intranodular portal supply definitely decreases, whereas the intranodular arterial supply tends to progressively increase. This increased intranodular arterial input is due to neoangiogenesis of the unpaired artery in the lesion [10]. Therefore, the majority of advanced HCCs show typical hemodynamic features of arterial enhancement and portal/delayed washout (Fig. 3C).
- Cellular change in hepatocellular nodules
- As hepatocellular nodules evolve from DNs to HCCs, several cellular changes occur, including small cell change, increased cell density, and nodule-in-nodule growth. These cellular changes can be detected in T2-WI and diffusion-weighted images [35].
- First, cell density can be evaluated by diffusion-weighted imaging and T2-WI. Low-grade DNs show a mild increase in cell density, and high-grade DNs show a moderate increase in cell density, i.e., as much as twice that of the surrounding liver parenchyma [11]. However, these cellular changes of DNs are usually insufficient to show overt high SI on diffusion-weighted images and T2-WI. In contrast, in HCCs, an increase in cell density is usually sufficiently detectable on diffusion-weighted images or T2-WIs [38]. Small cell change is frequently observed in HCC and may also contribute to diffusion restriction of water molecules in the malignant cells seen on diffusion-weighted images (Fig. 3D).
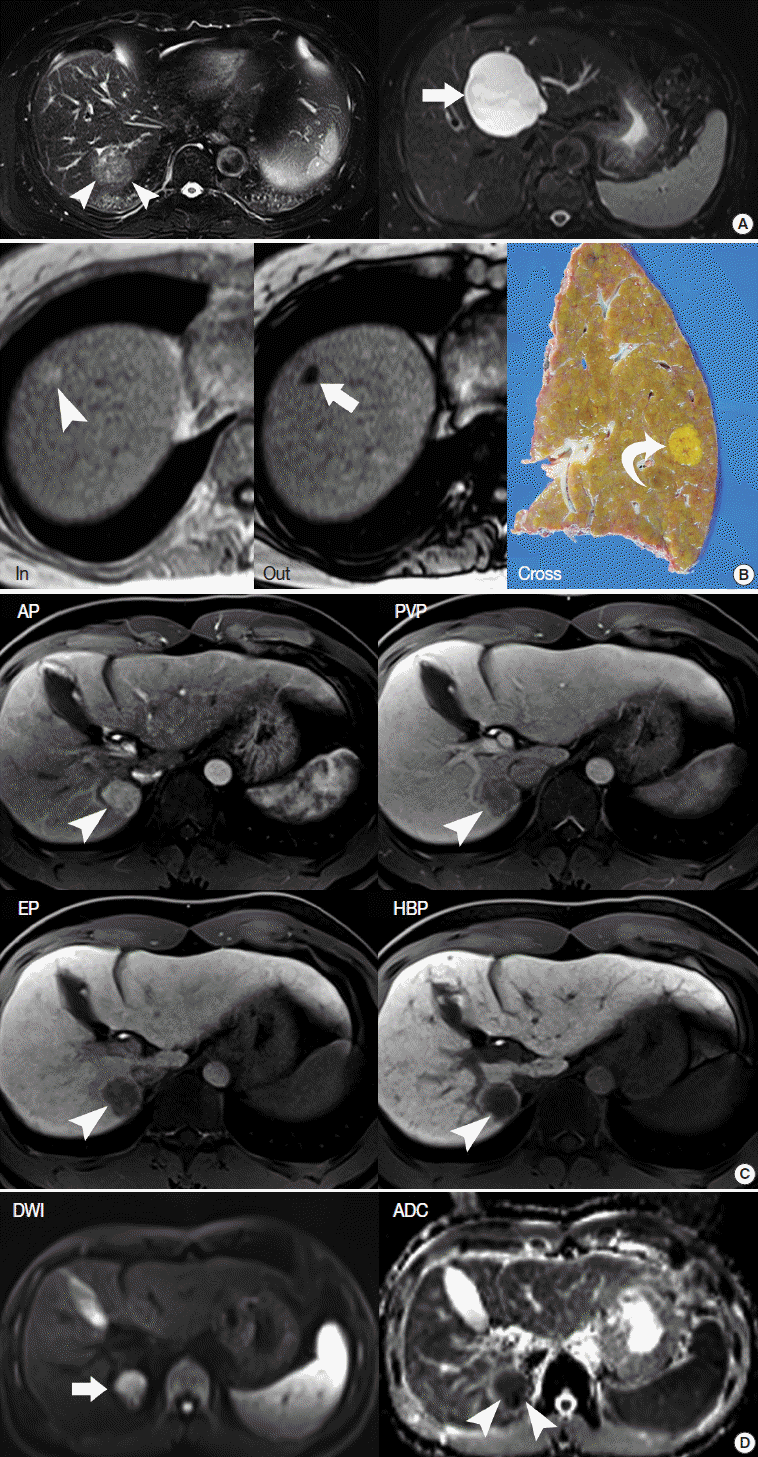
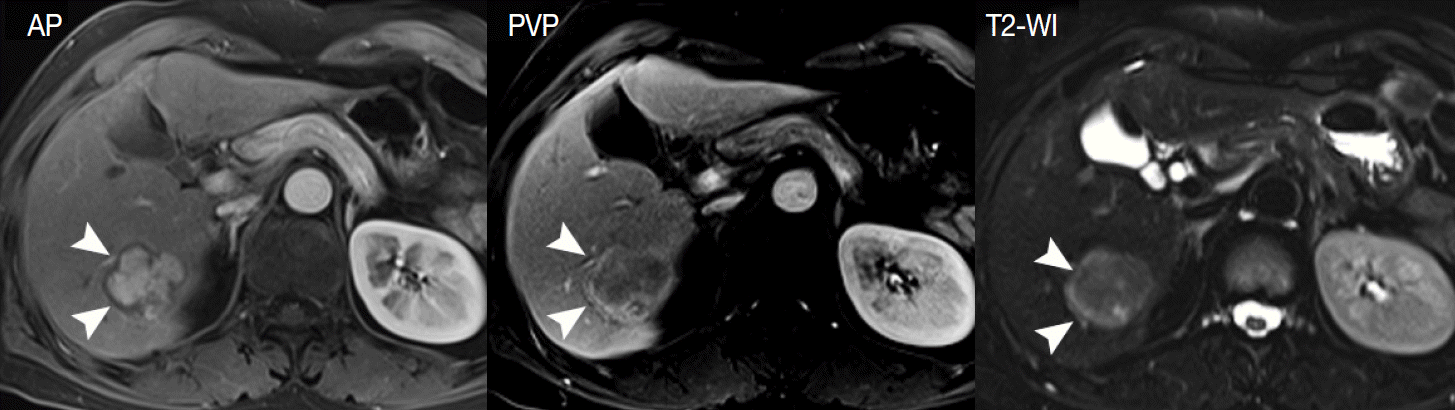
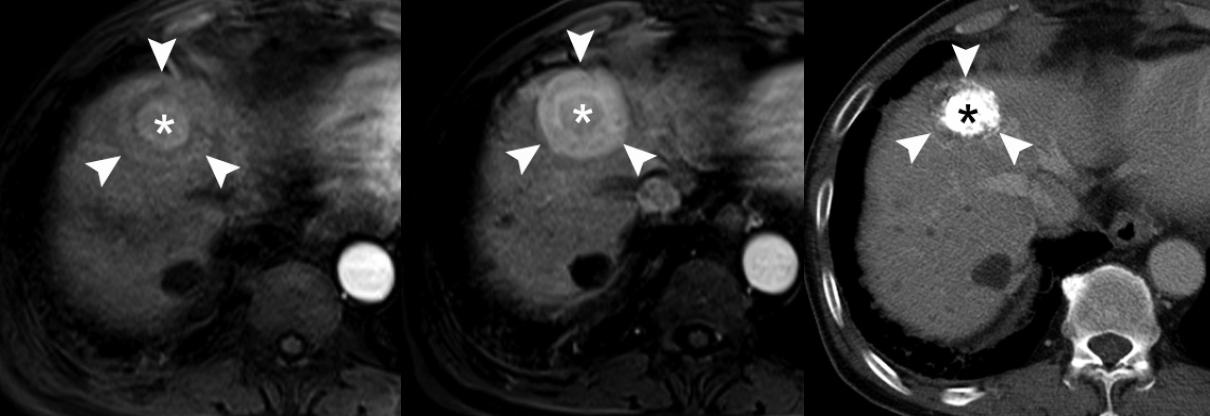
- Secondly, the nodule-in-nodule pattern is one of the typical histologic and imaging features of HCC, although it is uncommon. Usually, cirrhotic nodules and low-grade DNs are seen as distinct single nodules, while some high-grade DNs contain sub-nodules which are usually HCC foci, i.e., clones of malignant-transformed cells from DNs during hepatocarcinogenesis. The nodule-in-nodule pattern can be depicted by either T2-WI or dynamic MRI [39]. If the sub-nodule within the DN shows arterial enhancement, it is easily detected in the arterial phase of dynamic MRI (Fig. 4).
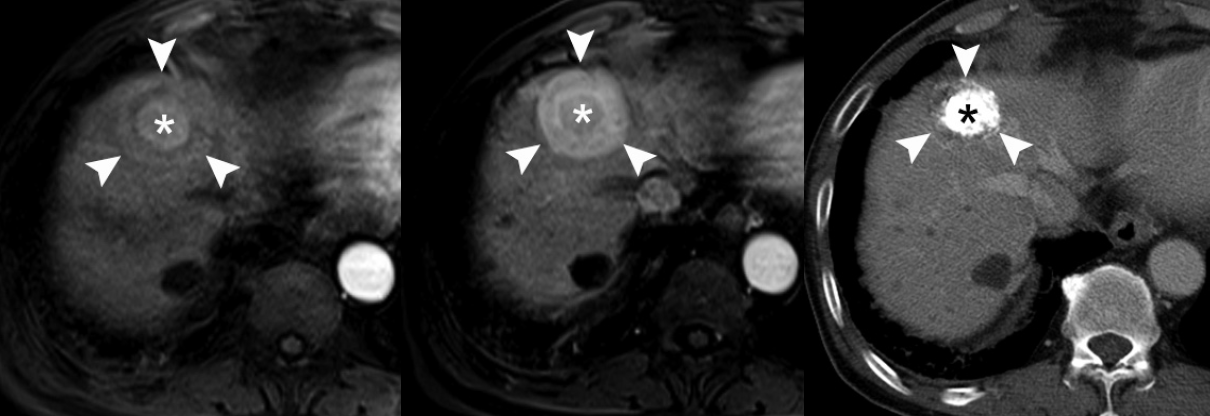
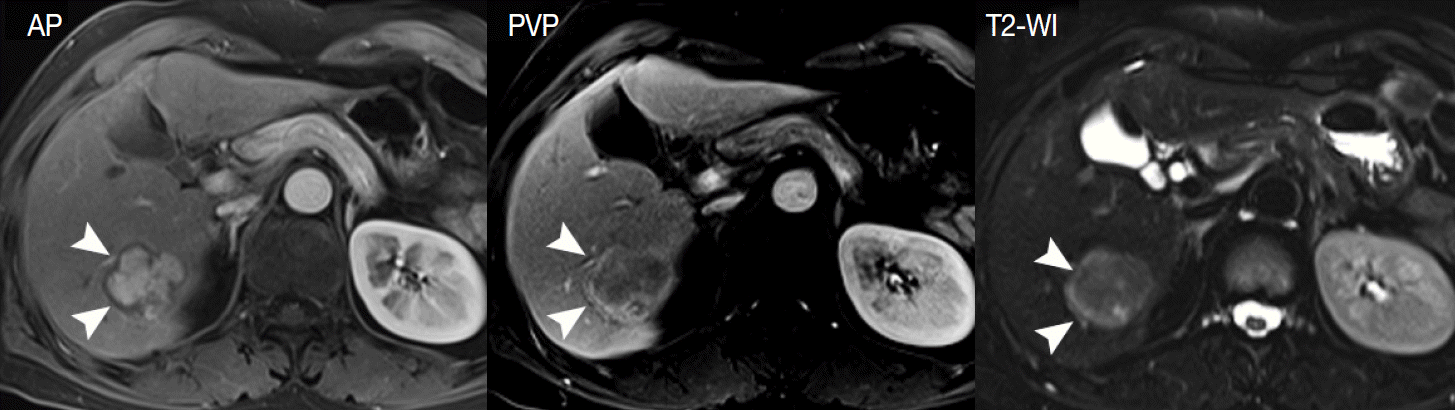
- Thirdly, the tumor capsule is a characteristic feature of progressed HCCs [39]. It is usually seen as a thin rim of low SI on noncontrast T1-WI. On contrast-enhanced MRI, the tumor capsule can be progressively enhanced following injection of gadolinium contrast agent, as the tumor capsule is generally composed of fibrous tissue containing compressed vessels (Fig. 5) [12]. On T2-WI, the tumor capsule may show variable SI as either low SI or high SI. Although the tumor capsule is regarded as a characteristic finding of progressed HCCs, it may be present in large DNs and may not be seen in small HCCs. For the diagnosis of HCC, the presence of a tumor capsule is helpful, although not essential.
- Alterations of the fat/iron composition in hepatocellular nodules
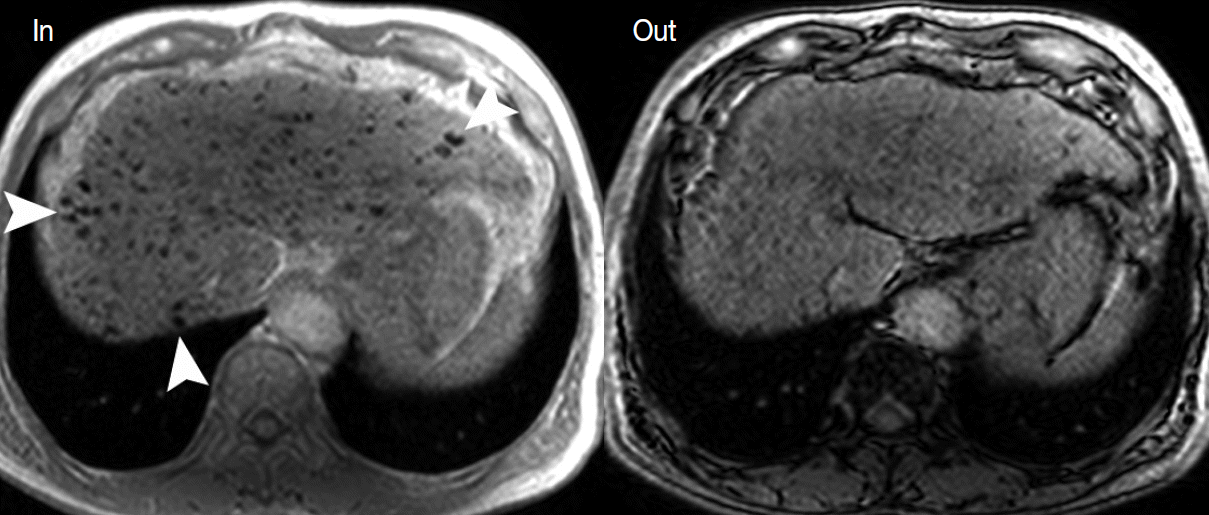
- During hepatocarcinogenesis, iron may accumulate in low-grade DNs and in some high-grade DNs. These nodules containing an iron deposit are referred to as ‘siderotic nodules’ (Fig. 6). In hepatocytes of siderotic nodules, the iron-transporter system, such as the transferrin transporter, are upregulated [14]. However, as these nodules progress into HCCs, the iron-transportation system also alters, and iron utilization increases, thus causing iron deficiency in HCCs [15]. Therefore, with progression to HCC, iron usually decreases.
- These alterations of the fat composition in DNs and HCCs are often helpful for characterizing hepatocellular nodules on MRI. On T2-WI or T2*-WI, the iron component is seen as dark SI. On T1-weighted, dual gradient-echo images, the iron component shows a signal drop in in-phase images compared to that of opposed-phase images. Iron-containing, hepatocellular nodules are usually DNs and are unlikely to be HCCs. The development of an iron-deficient sub-nodule within a siderotic nodule indicates HCC foci arising from the DN. Likewise, if a siderotic nodule is seen to be iron-free on a follow-up study, it may suggest its transformation into HCC [17].
- Fat accumulation within hepatocellular nodules is also altered during hepatocarcinogenesis. In low-grade DNs, high-grade DNs, and early HCCs, fat can accumulate focally or diffusely in the nodules. The intralesional fat accumulation peaks in the early HCC stage and regresses according to the increasing size and grade of HCCs. Usually, poorly differentiated HCCs or large HCCs >3 cm rarely show intralesional fat accumulation [13]. The fat component can be evaluated on T1-weighted, dual gradientecho images. The fatty component of tissue shows a signal drop in opposed-phase images compared to in-phase images (Fig. 3B).
MAGNETIC RESONANCE IMAGING OF ALTERATIONS DURING HEPATOCARCINOGENESIS
- Regenerative nodules
- On MRI, most RNs are imperceptible and isointense on unenhanced T1-WIs and T2-WIs. Some of these nodules may be differentiated by unusual imaging features, such as hyperintensity on T1-WIs and hypointensity on T2-WIs. When enhanced with contrast material, most RNs demonstrate a similar degree of enhancement with that of the adjacent parenchyma, although occasionally mild, delayed-phase hypoenhancement or hepatobiliary phase hyperintensity may be seen [27,40].
- Dysplastic nodules
- Common imaging findings of DNs are hyperintensity on T1- WIs and isointensity or hypointensity on T2-WIs [27,41]. The T1 hyperintensity can be explained by the fact that DNs accumulate copper or iron, which are paramagnetic materials leading to T1 hyperintensity depending on the concentration. Such intranodular copper or iron accumulation may lead to T2 hypointensity due to the T2-shortening effects. Some DNs, especially high-grade DNs, may show fatty change manifested as T1 hyperintensity on MRI.
- After contrast agent injection, most DNs are usually isointense to the liver parenchyma in the hepatic arterial, portal venous, and hepatobiliary phases. As some DNs show washout or capsular enhancement in the delayed phase and hypointensity in the hepatobiliary phase, these findings may be good predictors of pre-malignancy [27].
- Early HCC
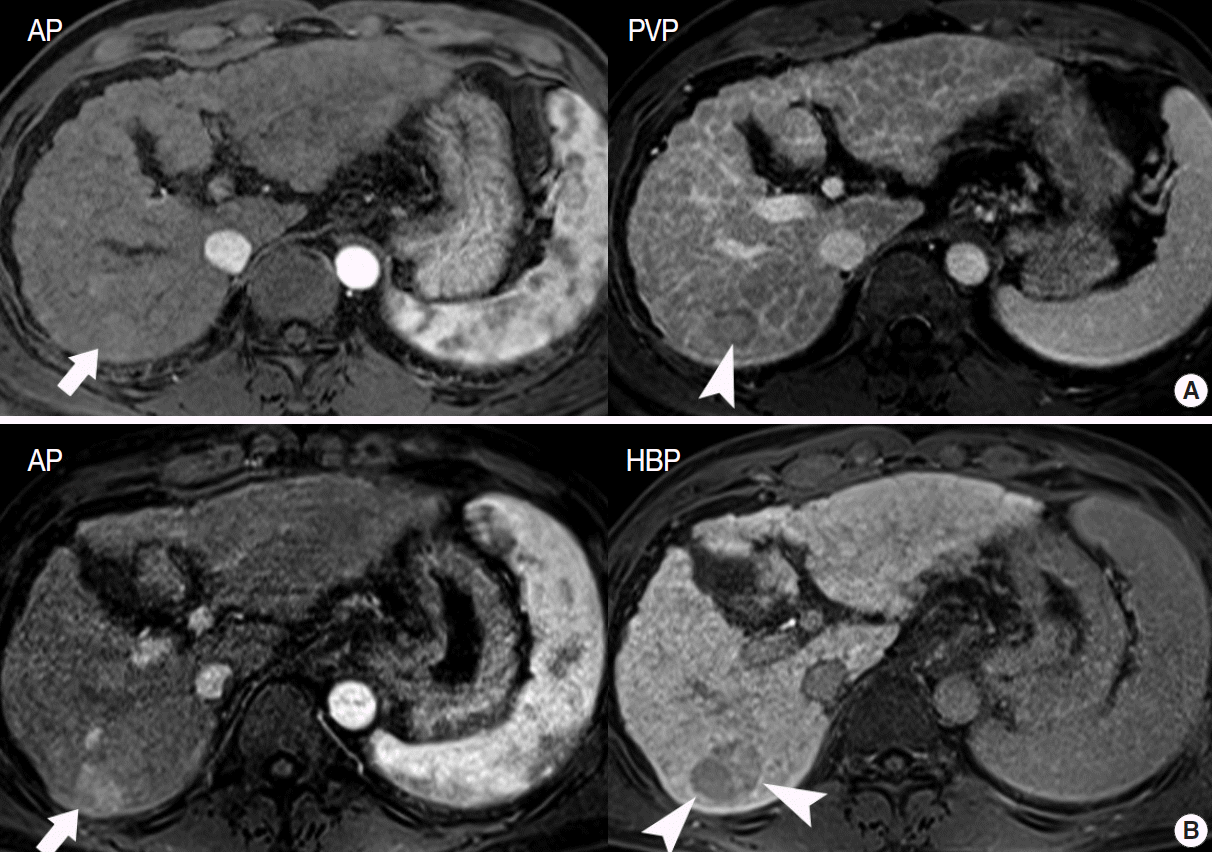
- Most early HCCs are either isointense or hyperintense on T1- WIs and are mostly isointense on T2-WIs [42]. In addition, most early HCCs are hypovascular, and only 5% of the early HCCs show arterial hypervascularity on dynamic MRI. These characteristics are nonspecific, suggesting that it is difficult to accurately diagnose early HCC using conventional MRI.
- In particular, differentiating early HCC from high-grade DN on MRI is quite challenging. There are a few differential points. First, high SI on diffusion-weighted images almost always indicates early HCC. DNs are almost never hyperintense on T2-WI and diffusion-weighted images [17]. Second, nodule hypointensity on the hepatobiliary phase of gadoxetic-acid-enhanced MRI favors early HCC rather than high-grade DN, as a substantial number of such hypointense nodules show hypervascularity during the follow-up period (Fig. 7) [43].
- Progressed HCC
- The typical MRI findings of HCC, which have been extensively investigated in numerous research studies, mostly apply to progressed HCC rather than early HCC. The hallmarks of HCC are arterial hypervascularity and washout on the portalvenous phase and/or equilibrium phase of multiphasic dynamic CT or MRI [4,37]. This finding is due to the predominant vascular supply to HCCs from unpaired hepatic arteries and the absence of the portal vein supply. These hemodynamic changes are apparent in the progressed HCC rather than the early HCC. Therefore, it is very helpful to differentiate small progressed HCC from early HCC. In addition, the nodule margin seen on MRI is also helpful to differentiate small, progressed HCC, because it is more distinctly nodular than early HCC with its indistinct margin. Indeed, the tumor capsule is a characteristic feature of progressed HCCs and is seen as a persistent enhancing rim at the periphery of the tumor [39].
- Progressed HCCs can present as infiltrative masses as well as expansile nodules/masses. In infiltrative masses, the tumor cells may infiltrate through the tumor capsule into the surrounding parenchyma [17]. These infiltrative HCCs frequently show vascular invasion or biliary invasion, thus leading to a poor patient prognosis.
- Vascular invasion is common in progressed HCCs, including portal vein invasion or hepatic vein invasion. As vascular invasion is rarely observed in other types of liver malignancies, this is a helpful characteristic for diagnosing HCC. Vascular invasion generally indicates a poor prognosis for the patients, as it is a route of tumor spread through the liver and of systemic metastasis [26].
MAGNETIC RESONANCE IMAGING FINDINGS OF HEPATOCARCINOGENESIS
- The incidence of small HCCs has recently been increasing due to a nationwide surveillance program and advances in imaging techniques. It is difficult to correctly diagnose such small HCCs during histological and radiological examinations. Understanding the important alterations during hepatocarcinogenesis as well as the characteristic MRI and histopathological features will be helpful for managing patients with chronic liver disease and HCC. There is no doubt that special MRI techniques, such as fat/iron quantification, diffusion-weighted images, and gadoxetic-acid-enhanced MRI, have greatly enhanced our understanding of hepatocarcinogenesis.
CONCLUSION
Acknowledgments
- 1. Song P, Tang W, Tamura S, et al. The management of hepatocellular carcinoma in Asia: a guideline combining quantitative and qualitative evaluation. Biosci Trends 2010; 4: 283-7. PubMed
- 2. Kudo M, Han KH, Kokudo N, et al. Liver Cancer Working Group report. Jpn J Clin Oncol 2010; 40 Suppl 1: i19-27. Article
- 3. Lee SH, Choi HC, Jeong SH, et al. Hepatocellular carcinoma in older adults: clinical features, treatments, and survival. J Am Geriatr Soc 2011; 59: 241-50. ArticlePubMed
- 4. Kudo M. Multistep human hepatocarcinogenesis: correlation of imaging with pathology. J Gastroenterol 2009; 44 Suppl 19: 112-8. ArticlePubMedPDF
- 5. Yoo HJ, Lee JM, Lee MW, et al. Hepatocellular carcinoma in cirrhotic liver: double-contrast-enhanced, high-resolution 3.0T-MR imaging with pathologic correlation. Invest Radiol 2008; 43: 538-46. ArticlePubMed
- 6. Choi BI, Lee JM. Advancement in HCC imaging: diagnosis, staging and treatment efficacy assessments: imaging diagnosis and staging of hepatocellular carcinoma. J Hepatobiliary Pancreat Sci 2010; 17: 369-73. PubMed
- 7. Henninger B, Zoller H, Rauch S, et al. Automated two-point dixon screening for the evaluation of hepatic steatosis and siderosis: comparison with R2*-relaxometry and chemical shift-based sequences. Eur Radiol 2015; 25: 1356-65. ArticlePubMedPDF
- 8. Suh CH, Kim KW, Kim GY, Shin YM, Kim PN, Park SH. The diagnostic value of Gd-EOB-DTPA-MRI for the diagnosis of focal nodular hyperplasia: a systematic review and meta-analysis. Eur Radiol 2015; 25: 950-60. ArticlePubMedPDF
- 9. Krinsky G. Imaging of dysplastic nodules and small hepatocellular carcinomas: experience with explanted livers. Intervirology 2004; 47: 191-8. ArticlePubMedPDF
- 10. Fournier LS, Cuenod CA, de Bazelaire C, et al. Early modifications of hepatic perfusion measured by functional CT in a rat model of hepatocellular carcinoma using a blood pool contrast agent. Eur Radiol 2004; 14: 2125-33. ArticlePubMedPDF
- 11. Roncalli M, Terracciano L, Di Tommaso L, et al. Liver precancerous lesions and hepatocellular carcinoma: the histology report. Dig Liver Dis 2011; 43 Suppl 4: S361-72. Article
- 12. Ishigami K, Yoshimitsu K, Nishihara Y, et al. Hepatocellular carcinoma with a pseudocapsule on gadolinium-enhanced MR images: correlation with histopathologic findings. Radiology 2009; 250: 435-43. ArticlePubMed
- 13. Kutami R, Nakashima Y, Nakashima O, Shiota K, Kojiro M. Pathomorphologic study on the mechanism of fatty change in small hepatocellular carcinoma of humans. J Hepatol 2000; 33: 282-9. ArticlePubMed
- 14. Pascale RM, De Miglio MR, Muroni MR, et al. Transferrin and transferrin receptor gene expression and iron uptake in hepatocellular carcinoma in the rat. Hepatology 1998; 27: 452-61. ArticlePubMed
- 15. Holmstrom P, Gåfvels M, Eriksson LC, et al. Expression of iron regulatory genes in a rat model of hepatocellular carcinoma. Liver Int 2006; 26: 976-85. ArticlePubMed
- 16. Libbrecht L, Craninx M, Nevens F, Desmet V, Roskams T. Predictive value of liver cell dysplasia for development of hepatocellular carcinoma in patients with non-cirrhotic and cirrhotic chronic viral hepatitis. Histopathology 2001; 39: 66-73. ArticlePubMedPDF
- 17. Choi JY, Lee JM, Sirlin CB. CT and MR imaging diagnosis and staging of hepatocellular carcinoma: part I. Development, growth, and spread: key pathologic and imaging aspects. Radiology 2014; 272: 635-54. ArticlePubMed
- 18. International Working Party. Terminology of nodular hepatocellular lesions. Hepatology 1995; 22: 983-93. ArticlePubMed
- 19. International Consensus Group for Hepatocellular Neoplasia; The International Consensus Group for Hepatocellular Neoplasia. Pathologic diagnosis of early hepatocellular carcinoma: a report of the International Consensus Group for Hepatocellular Neoplasia. Hepatology 2009; 49: 658-64. ArticlePubMed
- 20. Park YN. Update on precursor and early lesions of hepatocellular carcinomas. Arch Pathol Lab Med 2011; 135: 704-15. ArticlePubMedPDF
- 21. Okuda K, Peters RL, Simson IW. Gross anatomic features of hepatocellular carcinoma from three disparate geographic areas: proposal of new classification. Cancer 1984; 54: 2165-73. ArticlePubMed
- 22. Korean Liver Cancer Study Group. Pathology for prediction of liver. General rules for the study of primary liver cancer. Seoul: Korean Liver Cancer Study Group, 2004; 39-40.
- 23. Choi BI, Lee GK, Kim ST, Han MC. Mosaic pattern of encapsulated hepatocellular carcinoma: correlation of magnetic resonance imaging and pathology. Gastrointest Radiol 1990; 15: 238-40. ArticlePubMedPDF
- 24. Demirjian A, Peng P, Geschwind JF, et al. Infiltrating hepatocellular carcinoma: seeing the tree through the forest. J Gastrointest Surg 2011; 15: 2089-97. ArticlePubMedPMCPDF
- 25. Edmondson HA, Steiner PE. Primary carcinoma of the liver: a study of 100 cases among 48,900 necropsies. Cancer 1954; 7: 462-503. ArticlePubMed
- 26. Bruix J, Sherman M; American Association for the Study of Liver Diseases. Management of hepatocellular carcinoma: an update. Hepatology 2011; 53: 1020-2. ArticlePubMedPMC
- 27. Lee JM, Choi BI. Hepatocellular nodules in liver cirrhosis: MR evaluation. Abdom Imaging 2011; 36: 282-9. ArticlePubMedPDF
- 28. Zech CJ, Reiser MF, Herrmann KA. Imaging of hepatocellular carcinoma by computed tomography and magnetic resonance imaging: state of the art. Dig Dis 2009; 27: 114-24. ArticlePubMedPDF
- 29. Bitar R, Leung G, Perng R, et al. MR pulse sequences: what every radiologist wants to know but is afraid to ask. Radiographics 2006; 26: 513-37. ArticlePubMed
- 30. Kang BK, Yu ES, Lee SS, et al. Hepatic fat quantification: a prospective comparison of magnetic resonance spectroscopy and analysis methods for chemical-shift gradient echo magnetic resonance imaging with histologic assessment as the reference standard. Invest Radiol 2012; 47: 368-75. PubMed
- 31. Ayyappan AP, Jhaveri KS. CT and MRI of hepatocellular carcinoma: an update. Expert Rev Anticancer Ther 2010; 10: 507-19. ArticlePubMed
- 32. Sun HY, Lee JM, Shin CI, et al. Gadoxetic acid-enhanced magnetic resonance imaging for differentiating small hepatocellular carcinomas (< or =2 cm in diameter) from arterial enhancing pseudolesions: special emphasis on hepatobiliary phase imaging. Invest Radiol 2010; 45: 96-103. PubMed
- 33. Taouli B, Ehman RL, Reeder SB. Advanced MRI methods for assessment of chronic liver disease. AJR Am J Roentgenol 2009; 193: 14-27. ArticlePubMedPMC
- 34. Taouli B, Koh DM. Diffusion-weighted MR imaging of the liver. Radiology 2010; 254: 47-66. ArticlePubMed
- 35. Bartolozzi C, Battaglia V, Bozzi E. HCC diagnosis with liver-specific MRI: close to histopathology. Dig Dis 2009; 27: 125-30. ArticlePubMedPDF
- 36. Hayashi M, Matsui O, Ueda K, et al. Correlation between the blood supply and grade of malignancy of hepatocellular nodules associated with liver cirrhosis: evaluation by CT during intraarterial injection of contrast medium. AJR Am J Roentgenol 1999; 172: 969-76. ArticlePubMed
- 37. Choi BI. Hepatocarcinogenesis in liver cirrhosis: imaging diagnosis. J Korean Med Sci 1998; 13: 103-16. ArticlePubMedPMC
- 38. Han JK, Eun HW, Kim SH. Radiologic findings of dysplastic nodule. Korean J Hepatol 2008; 14: 231-4. ArticlePubMed
- 39. Hanna RF, Aguirre DA, Kased N, Emery SC, Peterson MR, Sirlin CB. Cirrhosis-associated hepatocellular nodules: correlation of histopathologic and MR imaging features. Radiographics 2008; 28: 747-69. ArticlePubMed
- 40. Trevisani F, Cantarini MC, Wands JR, Bernardi M. Recent advances in the natural history of hepatocellular carcinoma. Carcinogenesis 2008; 29: 1299-305. ArticlePubMed
- 41. Aravalli RN, Cressman EN, Steer CJ. Cellular and molecular mechanisms of hepatocellular carcinoma: an update. Arch Toxicol 2013; 87: 227-47. ArticlePubMedPDF
- 42. Thorgeirsson SS, Grisham JW. Molecular pathogenesis of human hepatocellular carcinoma. Nat Genet 2002; 31: 339-46. ArticlePubMedPDF
- 43. Ichikawa T, Sano K, Morisaka H. Diagnosis of pathologically early HCC with EOB-MRI: experiences and current consensus. Liver Cancer 2014; 3: 97-107. ArticlePubMedPMCPDF
REFERENCES
Figure & Data
References
Citations

- Update on Hepatocellular Carcinoma Imaging Features Associated With Histology, Subtype, and Prognosis Along With Changes to LI-RADS in 2024
Sergio P. Klimkowski, Ann Shi, Omar Altabbakh, Janio Szklaruk, AnuradhaS. Shenoy-Bhangle, Gauruv S. Likhari, Khaled M. Elsayes
Seminars in Roentgenology.2025; 60(1): 6. CrossRef - Gadoxetic acid in hepatocellular carcinoma and liver metastases: pearls and pitfalls
H.M. Kwok, C.M. Chau, H.C.H. Lee, T. Wong, H.F. Chan, W.H. Luk, W.T.A. Yung, L.F. Cheng, K.F.J. Ma
Clinical Radiology.2023; 78(10): 715. CrossRef - Multi-phasic magnetic resonance imaging of hemodynamic interchanges in hepatocarcinogenesis
Ahmed Mahmoud Elzeneini, Mohsen Ahmed Abdelmohsen, Mohamed Ibrahim Yousef
Egyptian Journal of Radiology and Nuclear Medicine.2023;[Epub] CrossRef - Risk Factors for Hypervascularization in Hepatobiliary Phase Hypointense Nodules without Arterial Phase Hyperenhancement: A Systematic Review and Meta-analysis
Tae-Hyung Kim, Sungmin Woo, Sangwon Han, Chong Hyun Suh, Richard Kinh Gian Do, Jeong Min Lee
Academic Radiology.2022; 29(2): 198. CrossRef - Comparison of IDEAL-IQ and IVIM-DWI for Differentiating between Alpha Fetoprotein-Negative Hepatocellular Carcinoma and Focal Nodular Hyperplasia
Shaopeng Li, Peng Wang, Jun Qiu, Yiju Xie, Dawei Yin, Kexue Deng
Oncologie.2022; 24(3): 527. CrossRef - Hepatocarcinogenesis
Alice Fung, Krishna P. Shanbhogue, Myles T. Taffel, Brian T. Brinkerhoff, Neil D. Theise
Magnetic Resonance Imaging Clinics of North America.2021; 29(3): 359. CrossRef - Pathologic, Molecular, and Prognostic Radiologic Features of Hepatocellular Carcinoma
Kathryn J. Fowler, Adam Burgoyne, Tyler J. Fraum, Mojgan Hosseini, Shintaro Ichikawa, Sooah Kim, Azusa Kitao, Jeong Min Lee, Valérie Paradis, Bachir Taouli, Neil D. Theise, Valérie Vilgrain, Jin Wang, Claude B. Sirlin, Victoria Chernyak
RadioGraphics.2021; 41(6): 1611. CrossRef - Update on Hepatocellular Carcinoma: a Brief Review from Pathologist Standpoint
Nese Karadag Soylu
Journal of Gastrointestinal Cancer.2020; 51(4): 1176. CrossRef - Gadoxetate-enhanced dynamic contrast-enhanced MRI for evaluation of liver function and liver fibrosis in preclinical trials
Jimi Huh, Su Jung Ham, Young Chul Cho, Bumwoo Park, Bohyun Kim, Chul-Woong Woo, Yoonseok Choi, Dong-Cheol Woo, Kyung Won Kim
BMC Medical Imaging.2019;[Epub] CrossRef - Non-Hypervascular Hypointense Nodules at Gadoxetic Acid MRI: Hepatocellular Carcinoma Risk Assessment with Emphasis on the Role of Diffusion-Weighted Imaging
Chiara Briani, Marco Di Pietropaolo, Massimo Marignani, Francesco Carbonetti, Paola Begini, Vincenzo David, Elsa Iannicelli
Journal of Gastrointestinal Cancer.2018; 49(3): 302. CrossRef
 PubReader
PubReader ePub Link
ePub Link-
 Cite this Article
Cite this Article
- Cite this Article
-
- Close
- Download Citation
- Close
- Figure







Fig. 1.
Fig. 2.
Fig. 3.
Fig. 4.
Fig. 5.
Fig. 6.
Fig. 7.
| Terminology | Feature | Histologic characteristic |
|---|---|---|
| Regenerative nodules | Gross | Well-defined rounded regions of the cirrhotic parenchyma surrounded by scar tissue |
| Microscopic | Phenotypically normal cells | |
| Dysplastic nodules | Gross | Distinctly nodular lesions which differ from adjacent cirrhotic parenchyma with regard to size, color, texture, and degree of bulging at the cut surface |
| Microscopic | Divided into low-grade DNs and high-grade DNs | |
| - Low-grade DN: a clonal cell population without significant architectural atypia and with a mild increase in cell density | ||
| - High-grade DN: a clonal cell population with cytological and architectural atypia although not sufficient for a diagnosis of malignancy | ||
| Early HCCs (=small HCCs of the vaguely nodular type) | Gross | Vaguely nodular lesions with indistinct margins |
| Microscopic | Well-differentiated histology of HCC with increased cell density and an irregular, thin-trabecular pattern; the stromal invasion is the most helpful feature to differentiate early HCC from high-grade DN | |
| Progressed HCCs (=small HCCs of the distinctly nodular type) | Gross | Distinctly nodular lesions |
| Microscopic | Well (G1) to moderately (G2) differentiated histology of HCC |
| MRI sequences | Information |
|---|---|
| T2-weighted images | Helpful for differential diagnosis |
| Hepatic cysts and hemangiomas show very high T2 hyperintensity, while HCCs show intermediately high T2 hyperintensity | |
| Tl-weighted dual gradient-echo images (in-phase and opposed-phase) | Helpful for detecting the intralesional fat component |
| Multiphasic dynamic imaging (arterial phase, portal-venous phase, and three-minute equilibrium phase) | Evaluate the hemodynamic pattern of hepatic tumors |
| HCCs typically show arterial enhancement and delayed washout | |
| Hepatobiliary phase imaging (i.e., 20-minute delayed image) | When gadoxetic acid is used as a contrast agent, the hepatobiliary phase is used to improve the detection and characterization of hepatocellular nodules |
| Diffusion-weighted imaging | Helpful for the detection and characterization of hepatocellular nodules |
DN, dysplastic nodule; HCC, hepatocellular carcinoma.
MRI, magnetic resonance imaging; HCC, hepatocellular carcinoma.

 E-submission
E-submission