Articles
- Page Path
- HOME > J Pathol Transl Med > Volume 51(4); 2017 > Article
-
Brief Case Report
Bronchial Washing Cytology of Pulmonary Langerhans Cell Histiocytosis: A Case Report - Taeyeong Kim, Hyeong Ju Kwon, Minseob Eom, Sang Wook Kim, Min Hi Sin, Soon-Hee Jung
-
Journal of Pathology and Translational Medicine 2017;51(4):444-447.
DOI: https://doi.org/10.4132/jptm.2017.02.15
Published online: July 14, 2017
Department of Pathology, Yonsei University Wonju College of Medicine, Wonju, Korea
- Corresponding Author Soon-Hee Jung, MD, PhD Department of Pathology, Yonsei University Wonju College of Medicine, 20 Ilsan-ro, Wonju 26426, Korea Tel: +82-33-741-1551, Fax: +82-33-731-6590, E-mail: 'soonheej@yonsei.ac.kr'
• Received: September 2, 2016 • Revised: February 3, 2017 • Accepted: February 15, 2017
© 2017 The Korean Society of Pathologists/The Korean Society for Cytopathology
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/4.0) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
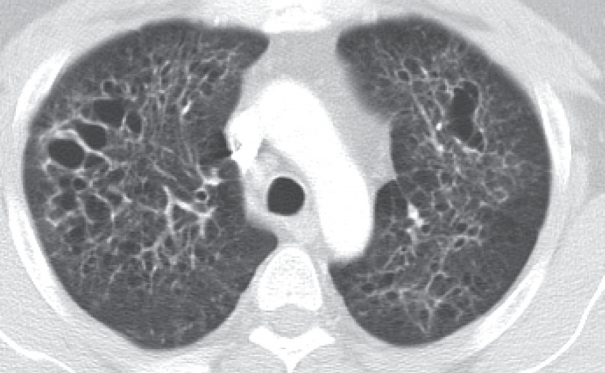
- The 41-year-old male patient visited our hospital due to a cough that had persisted for 7 months. He had a history of 20 pack-year smoking and diabetes mellitus. Physical examination of the chest revealed fine crackle of both upper lung fields. There was no skin rash or palpable lymph node. The chest computed tomography (CT) showed multiple, irregularly-shaped cysts and centrilobular nodules of variable sizes in both lungs (Fig. 1).
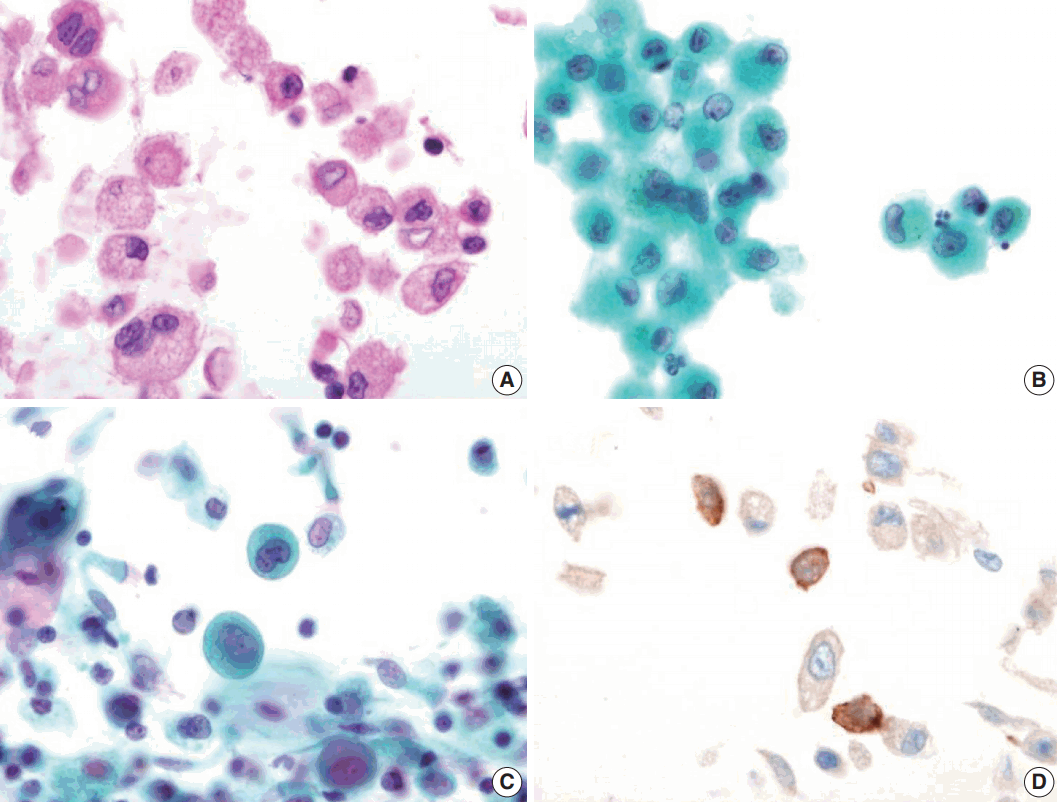
- Bronchial washing cytology, BAL, and wedge resection of the lung were performed. The BAL slides revealed a fraction rate of 59.6% monocytes and 9.4% eosinophils with about 6.5% LCs. The bronchial washing slides showed scattered non-cohesive large cells with abundant, granular, and eosinophilic cytoplasm. These cells had grooved or convoluted nuclei with fine chromatin, delicate nuclear membranes, and prominent nucleoli. Immunohistochemical staining for CD1a was positive in these cells (Fig. 2).
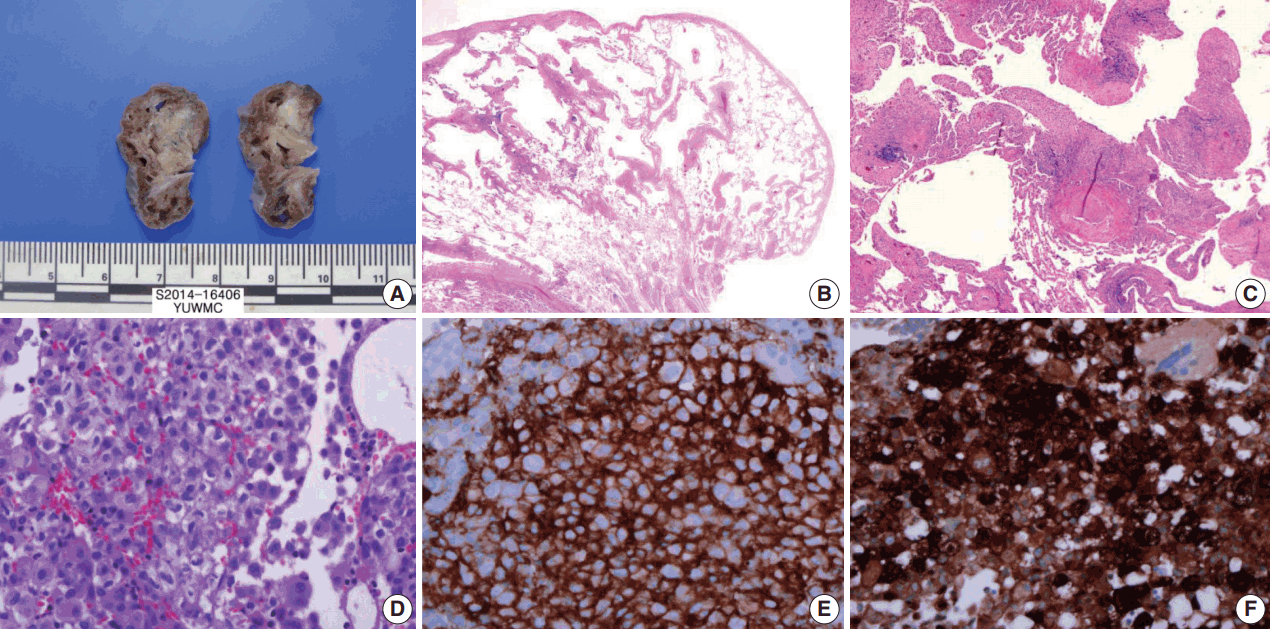
- The cut surface of the wedge-resected lung specimen revealed numerous cystic spaces with whitish gray stellate fibrous scars. Microscopic examination indicated multiple cystic spaces of variable sizes with diffuse alveolar wall thickening, cellular infiltration, and fibrosis. The infiltrated cells had eosinophilic cytoplasm and grooved or infolded nuclei. These cells were proven to be LCs based on immunohistochemical staining, showing strong positive reaction for CD1a and S-100 protein (Fig. 3).
- Bronchial washing cytology was performed again during follow-up. Some cells with the features described above were detected on the bronchial washing cytology. Therefore, we used the same approach to determine that the PLCH was still present.
CASE REPORT
- When patients with ongoing PLCH present at a hospital, non-or less-invasive pulmonological approaches, such as chest CT, pulmonary function test, and fiberoptic bronchoscopy, are usually performed. The cytological specimens obtained from fiberoptic bronchoscopy are then sent to the pathology department, and these cytological specimens can be challenging for pathologists to diagnose. The cytological diagnosis of PLCH with bronchial washing can be difficult because it is a rare disease with unfamiliar features. Therefore, in this report, we focused on bronchial washing cytology findings of PLCH.
- Some reports have presented cytological findings of Langerhans cell histiocytosis (LCH) that have arisen in various organs. Lee et al. [5] reported a case of LCH in the mandible diagnosed with fine needle aspiration cytology and indicated that the smear revealed high cellularity, and that the LCs had abundant vacuolated cytoplasm with round or occasionally folded nuclei. The LC nuclei showed a fine granular chromatin pattern with prominent nucleoli. Ha et al. [6] reported a case of LCH in lymph nodes using aspiration cytology similar to the case reported by Lee et al. [5]. In their study, the LCs had wrinkled or grooved nuclei with thin nuclear membranes and a delicate chromatin pattern. Multinucleated cells were also identified. In another report that applied fine needle aspiration LCH cytology, many of the cytological findings were similar, but the nucleoli were absent [7].
- Another cytological method applied fiberoptic bronchoscopy for diagnosing PLCH is BAL fluid analysis. Takizawa et al. [3] conducted BAL fluid analysis in PLCH and found a high mean cell fraction rate for macrophages (75.2%) and a variable eosinophil fraction rate (1% to 16%). The LCs revealed chromatin-poor nuclei with well-developed nucleoli. Previously described cytological findings with quantitative analysis of PLCH in BAL fluid are summarized in Table 1. In two studies with CD1a-positive cells in PLCH, BAL fluid obtained from patients with PLCH revealed more than 5% CD1a-positive cells [8,9]. In the present case, BAL fluid cytology showed similar fraction rates and cytological features. The percentage of LCs was about 6.5%. Based on these results, the 5% cut-off value of CD1a-positive cells or LCs could be postulated in BAL fluid cytology to make a PLCH diagnosis.
- In this report, we described bronchial washing cytology findings of PLCH. These findings are similar to other studies describing aspiration cytology and BAL. A common finding in all of the previous reports indicates that grooved or infolded nuclei with abundant cytoplasm could be used as diagnostic features for LCs. The difference in the present case compared with other reports could be that prominent nucleoli were observed only in some cells. The Birbeck granules are known as diagnostic LCH structures on electron microscopy (EM). However, EM study requires additional time and costs. We did not perform EM to detect Birbeck granules since other authors have suggested that it is not essential for diagnosing LCH [10].
- In conclusion, the possibility of LCH can be considered when bronchial washing cytology is mainly composed of Langerhans cells and eosinophils. In addition, more than 5% CD1a-positive cells in BAL fluid could be helpful for diagnosing PLCH. However, to date, there is no quantitative guideline for diagnosing PLCH. Therefore, additional cytological studies of PLCH, including quantitative analysis, are required to further identify and improve diagnostic approaches.
DISCUSSION
-
Conflicts of Interest
No potential conflict of interest relevant to this article was reported.
Notes
Fig. 1.Chest computed tomography findings. There are multiple, variable-sized, irregularly-shaped cysts and centrilobular nodules in both lungs.
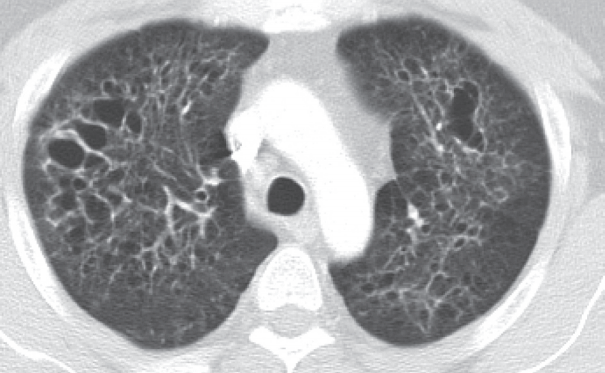

Fig. 2.Bronchial washing cytology. Bronchial washing cytology reveals many Langerhans cells. (A–C) The cells have abundant pale granular cytoplasm with a convoluted irregular nucleus and fine chromatin pattern (A, cell block; B, Thin prep; C, conventional smear). (D) Immunohistochemical staining for CD1a is positive in the Langerhans cells.
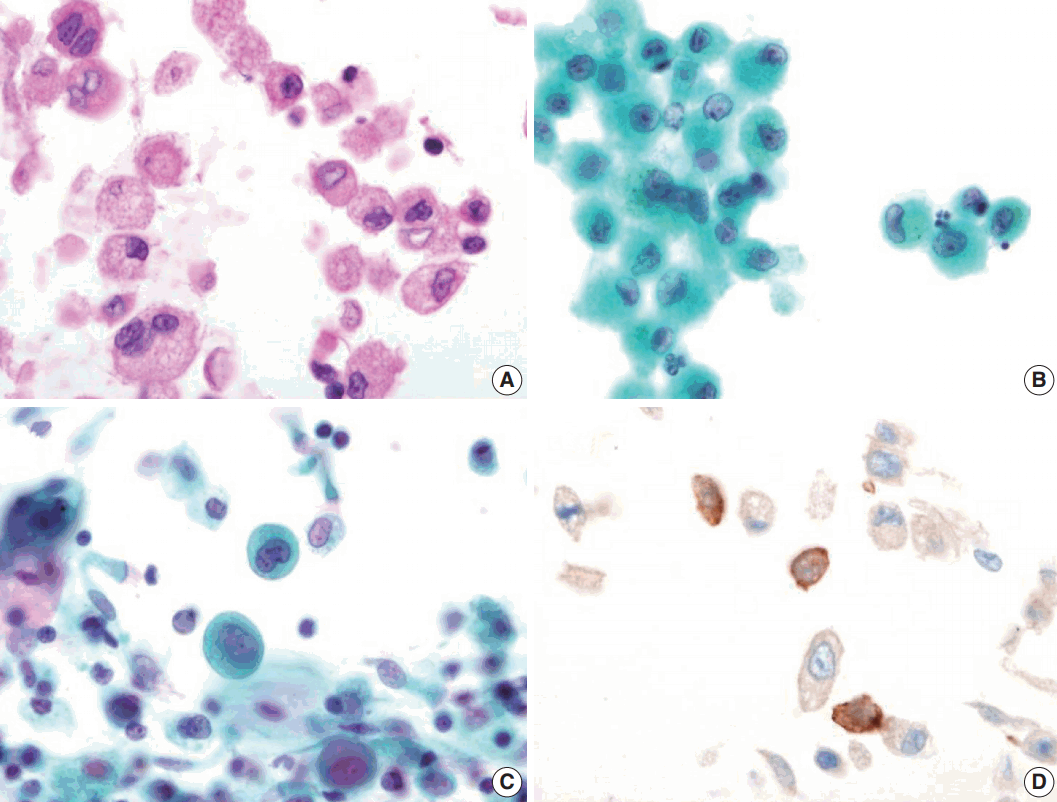

Fig. 3.Wedge resected lung via video-assisted thoracoscopic surgery. (A) The cut surface of the wedge-resected lung shows multiple numerous cystic spaces with white gray stellate fibrous scars. (B, C) There are multiple cystic spaces with diffuse thickening, cellular infiltration, and fibrous tissue. (D) The infiltrated cells have pale eosinophilic cytoplasm with grooved or infolded nuclei. (E, F) Immunohistochemical staining for CD1a and S-100 protein is positive in the proliferating cells.
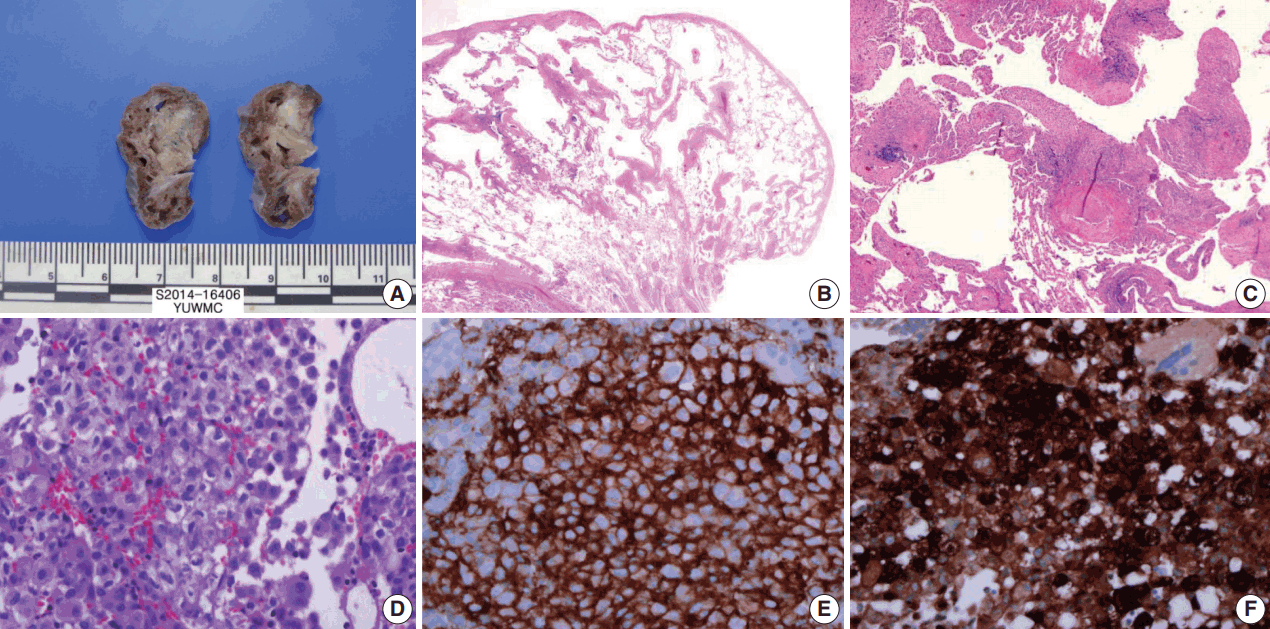

Table 1.Summary of previously reported cytological findings of pulmonary Langerhans cell histiocytosis in bronchoalveolar lavage fluid
| Study | Cytological finding | Langerhans cells (%) |
|---|---|---|
| Takizawa et al. [3] | Clear and velvety cytoplasm | 8.0 ± 1.3 |
| Oval or kidney-shaped, vesicular nuclei with irregular shapes | ||
| Nucleoli | ||
| Frequent groove and indentations | ||
| Sharma and Dey [4] | Nuclear grooving | 16.5 (CD1a-positive cells) |
| Bi-nucleation | ||
| Multinucleation | ||
| This case | Pale and granular cytoplasm | 6.5 |
| Indistinct cell border | ||
| Occasional binucleation or multinucleation | ||
| Grooved or convoluted nuclei | ||
| Prominent nucleoli |
- 1. Tazi A. Adult pulmonary Langerhans’ cell histiocytosis. Eur Respir J 2006; 27: 1272–85. ArticlePubMed
- 2. Yousem SA, Colby TV, Chen YY, Chen WG, Weiss LM. Pulmonary Langerhans' cell histiocytosis: molecular analysis of clonality. Am J Surg Pathol 2001; 25: 630–6. ArticlePubMed
- 3. Takizawa Y, Taniuchi N, Ghazizadeh M, et al. Bronchoalveolar lavage fluid analysis provides diagnostic information on pulmonary Langerhans cell histiocytosis. J Nippon Med Sch 2009; 76: 84–92. ArticlePubMed
- 4. Sharma S, Dey P. Childhood pulmonary langerhans cell histiocytosis in bronchoalveolar lavage: a case report along with review of literature. Diagn Cytopathol 2016; 44: 1102–6. ArticlePubMed
- 5. Lee SR, Suh JH, Cha HJ, Kim YM, Choi HJ. Fine needle aspiration cytology of Langerhans cell histiocytosis of mandible: a case report. Korean J Pathol 2010; 44: 106–9. Article
- 6. Ha SY, Kim MJ, Kim GY, Cho HY, Chung DH, Kim NR. Fine needle aspiration cytology of Langerhans cell histiocytosis in a lymph node: a case report. Korean J Cytopathol 2007; 18: 87–91. Article
- 7. Kumar N, Sayed S, Vinayak S. Diagnosis of Langerhans cell histiocytosis on fine needle aspiration cytology: a case report and review of the cytology literature. Patholog Res Int 2011; 2011: 439518.ArticlePubMedPMCPDF
- 8. Auerswald U, Barth J, Magnussen H. Value of CD-1-positive cells in bronchoalveolar lavage fluid for the diagnosis of pulmonary histiocytosis X. Lung 1991; 169: 305–9. ArticlePubMed
- 9. Refabert L, Rambaud C, Mamou-Mani T, Scheinmann P, de Blic J. Cd1a-positive cells in bronchoalveolarlavage samples from children with Langerhans cell histiocytosis. J Pediatr 1996; 129: 913–5. ArticlePubMed
- 10. Kilpatrick SE. Fine needle aspiration biopsy of Langerhans cell histiocytosis of bone: are ancillary studies necessary for a “definitive diagnosis”? Acta Cytol 1998; 42: 820–3. PubMed
References
Figure & Data
References
Citations
Citations to this article as recorded by 

 PubReader
PubReader ePub Link
ePub Link-
 Cite this Article
Cite this Article
- Cite this Article
-
- Close
- Download Citation
- Close
- Figure

 E-submission
E-submission