Articles
- Page Path
- HOME > J Pathol Transl Med > Volume 49(2); 2015 > Article
-
Review
Utility of Transmission Electron Microscopy in Small Round Cell Tumors - Na Rae Kim, Seung Yeon Ha, Hyun Yee Cho
-
Journal of Pathology and Translational Medicine 2015;49(2):93-101.
DOI: https://doi.org/10.4132/jptm.2015.01.30
Published online: March 12, 2015
Department of Pathology, Gachon University Gil Medical Center, Incheon, Korea
- Corresponding Author: Hyun Yee Cho, M.D. Department of Pathology, Gachon University Gil Medical Center, 21 Namdong-daero 774beon-gil, Namdong-gu, Incheon 405-760, Korea Tel: +82-32-460-3865 Fax: +82-32-460-2394 E-mail: 'hicho@gilhospital.com'
• Received: August 31, 2014 • Revised: January 28, 2015 • Accepted: January 30, 2015
© 2015 The Korean Society of Pathologists/The Korean Society for Cytopathology
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/3.0/) which permits unrestricted noncommercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
Abstract
- Small round cell tumors (SRCTs) are a heterogeneous group of neoplasms composed of small, primitive, and undifferentiated cells sharing similar histology under light microscopy. SRCTs include Ewing sarcoma/peripheral neuroectodermal tumor family tumors, neuroblastoma, desmoplastic SRCT, rhabdomyosarcoma, poorly differentiated round cell synovial sarcoma, mesenchymal chondrosarcoma, small cell osteosarcoma, small cell malignant peripheral nerve sheath tumor, and small cell schwannoma. Non-Hodgkin’s malignant lymphoma, myeloid sarcoma, malignant melanoma, and gastrointestinal stromal tumor may also present as SRCT. The current shift towards immunohistochemistry and cytogenetic molecular techniques for SRCT may be inappropriate because of antigenic overlapping or inconclusive molecular results due to the lack of differentiation of primitive cells and unavailable genetic service or limited moleculocytogenetic experience. Although usage has declined, electron microscopy (EM) remains very useful and shows salient features for the diagnosis of SRCTs. Although EM is not always required, it provides reliability and validity in the diagnosis of SRCT. Here, the ultrastructural characteristics of SRCTs are reviewed and we suggest that EM would be utilized as one of the reliable modalities for the diagnosis of undifferentiated and poorly differentiated SRCTs.
- In this review, we explain SRCTs with following subdivisions; first, SRCTs are mainly comprised of tumors with unknown histogenesis. In this group, ES/PNET, neuroblastoma, DSRCT, and malignant rhabdoid tumor (MRT) are included. Second, certain types of soft tissue neoplasms can rarely appear as SRCTs. The third group is hematologic malignancies, including nonHodgkin’s lymphoma and leukemia [7,16]. Others include epithelioid gastrointestinal stromal tumor (GIST), small cell or rhabdoid type melanomas [8,15,32]. Among these groups, certain type of SRCTs can be diagnosed under light microscopy and immunohistochemistry by their unique histologic features such as GIST or small cell liposarcoma, which are not discussed here.
ULTRASTRUCTURAL FINDINGS OF SMALL ROUND CELL TUMORS
- ES/PNET, neuroblastoma, DSRCT, and MRT are categorized as tumors of unknown histogenesis [8,31]. ES and PNET that present primarily in bone and soft tissues have been reported and previously studied as separate entities. However, there has been a recognition of a shared cytogenetic abnormality between the two and they are now regarded as different manifestations of the same entity at the genetic level (i.e., ES/PNET family tumors including Askin tumor) [12].
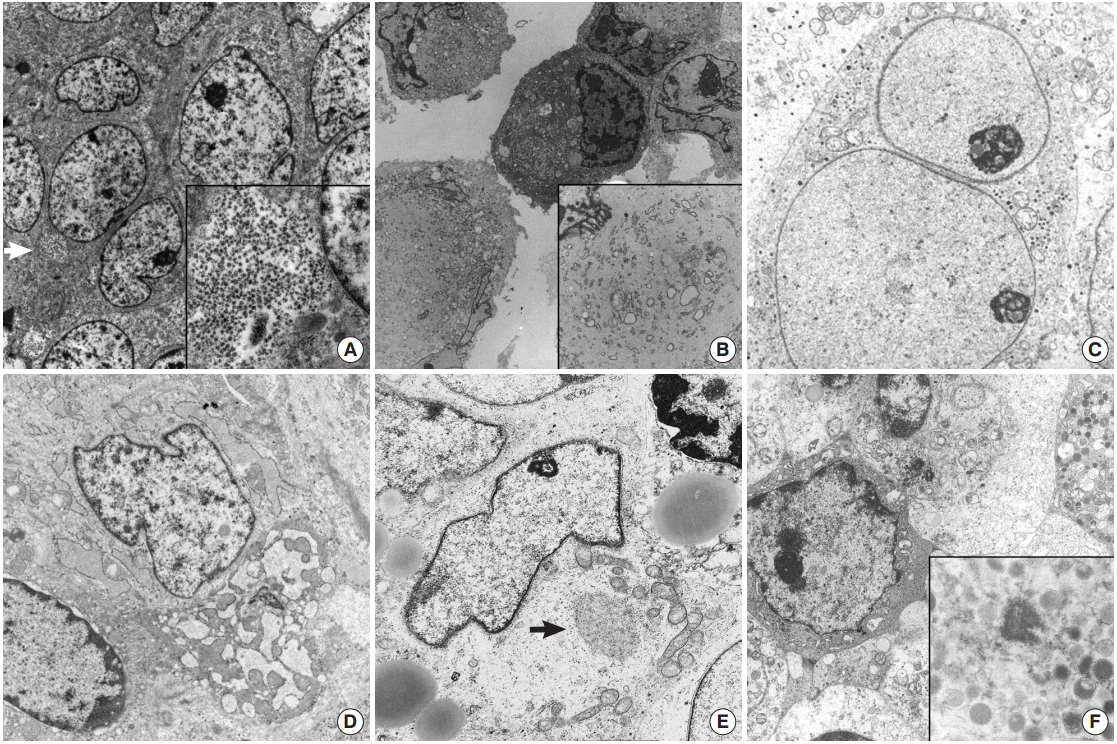
- ES/PNET family tumors show diverse heterogeneous histologic features, including large cell atypical types or adamantinoma-like variant ES/PNET, which have shared moleculogenetic abnormalities. ES/PNET family tumors show varying degrees of neuroectodermal differentiation. The ultrastructural findings of ES/PNET show undifferentiated mesenchymal cells arising from a mesenchymal stem cell and differentiating to neural cells [12,33]. Ultrastructural findings of this neural differentiation include intermediate filaments, microtubules, or neurosecretory granules and processes [12]. Small amounts of glycogen can be seen in other types of tumors, but in ES, glycogen deposits usually form lakes or pools (Fig. 1A). The tumors show undifferentiated primitive cells with no discernible characteristics except for abundant glycogen pools. Rich glycogen within the cytoplasm is a notable and distinctive finding for ES/PNET, but is not specific [33]; MRT may show plentiful glycogen particles and no other specialized structures, mimicking ES/PNET (Fig. 1B) [11]. MRT of soft tissue show large paranuclear whorls of intermediate filaments, with entrapped organelles and lipid droplets [34]. These paranuclear cytoplasmic whorls of intermediate filaments are shared by DSRCT of the abdominal wall [5,21,22,35].
- DSRCT is a rare malignant pediatric tumor that usually affects the abdominal cavity [22]. When DSRCT is presented in visceral organs, its diagnosis poses significant difficulty. In such circumstances, DSRCT showing multidirectional differentiation may add diagnostic confusion [5,23,35]. Neural differentiation characteristics such as neurosecretory granules, microtubules, dendritic interdigitating processes, or epithelial differentiation such as cell junctions or intracytoplasmic lumen can also be identified in DSRCT. Confirmation of neurosecretory granules, formerly known as dense core granules, requires careful examination because they may be easily confused with primary lysosomes, which are larger than the former. DSRCT have mitochondria, lipid aggregates, and a few groups of paranuclear intermediate filaments [21]. The multidirectional differentiation is concordant with t(11;22)(p13;q12), resulting in a chimeric EWS/WT1 transcript [23]. Presence of paranuclear whorls of intermediate filaments corresponds to the dot-like immunostaining with desmin or vimentin, together with the absence of specialized ultrastructural structures [21]. Among pediatric SRCTs, a tumor showing only neural differentiation is a neuroblastoma, the most frequently encountered extracranial pediatric solid tumor [36,37]. It originates from neural crest cells of the sympathetic nervous system and EM is very helpful in its diagnosis. These neoplasms have neurosecretory granules (50–200 nm in diameter) with membranebound dense core granules, cytoplasmic processes containing parallel arranged microtubules of 20 nm in diameter, and primitive cell junctions (Fig. 1C). Cell junctions are not demonstrated in neuroblastoma.
SRCT of unknown histogenesis
- Soft tissue tumors are classified according to morphology and histogenesis. As up to 30% of soft tissue tumors show chromosomal alterations, integration of molecular study in the diagnosis of soft tissue tumors has been recommended. However, accumulating data on these molecular alterations sometimes further complicates the molecular diagnoses of some soft tissue tumors. Certain types of soft tissue tumors can present as SRCTs including monophasic synovial sarcoma [8], small cell osteosarcoma [18], mesenchymal chondrosarcoma [38,39], small cell variant of melanoma [15,40], solid variant of alveolar rhabdomyosarcoma [41], round cell liposarcoma [42], small cell schwannoma, and small cell malignant peripheral nerve sheath tumor (MPNST) [9,36]. ES/PNET family tumors are too undifferentiated to have characterized ultrastructural findings, and ultrastructural morphology is hardly distinguished from the undifferentiated cells of small osteosarcoma and mesenchymal chondrosarcoma of soft tissue [43-46]. In primitive cells differentiating to chondrocytes in mesenchymal chondrosarcoma, some thin cytoplasmic projections to the extracellular spaces are the only distinguishing finding [13,46-48]. The chondrocyte-like tumor cells usually found in conventional myxoid chondrosarcoma are not found in most undifferentiated forms of chondrosarcomas [48]. The chondrocytes of well differentiated forms are ovoid to fusiform shaped ones with an eccentric nucleus and cytoplasm having abundant mitochondria, conspicuous endoplasmic reticulum with dilated cisternae of rough endoplasmic reticulums (RERs), cytoplasmic glycogen, lipid droplets, and numerous thin cytoplasmic projections such as microvilli into the extracellular area, which is filled with glycosaminoglycan granules, and collagen fibrils [49]. The higher the grade of chondrosarcoma, the lower the number of organelles, along with less prominent cytoplasmic projections into the extracellular spaces, bizarre nuclei, and more prominent nucleoli [50,51]. More immature cells correspond to small undifferentiated cells mimicking fibroblasts. Cells of mesenchymal chondrosarcoma have rounded nuclei, scanty cytoplasm, and few organelles and glycogen, like ES/PNET [13]. The intercellular matrix is very scanty and does not contain collagen fibrils. The search for more differentiated foci and correlation with the light microscopic findings is rewarding in such cases. These well-formed microvilli are found in low to intermediate grade chondrosarcoma, and are well observed even in clear cell chondrosarcomas, which lack other organelles, but have abundant glycogen particles [51]. Small cell osteosarcoma should be differentiated from other more common SRCTs. Chondroblasts showing features similar to those of chondroblastoma or chondrosarcoma can be seen in osteosarcoma [18]. There are rare osteoblastic or multinucleated giant cells with a large amount of RERs without ruffled borders. The tumor cells of a rare variant of osteosarcoma are a primitive form of osteoblasts, and the critical point of its ultrastructural diagnosis is to search for extracellular osteoid, extracellular matrix containing collagen fibers with deposition of hydroxyapatite crystals, showing correlation with light microscopy. The tumor cells have round shaped nuclei with occasional infolding and a more moderate amount of cytoplasm than those of ES/PNET. They also have a considerable number of organelles, most of which are RERs and ribosomes (Fig. 1D). Although tumor cells in a small cell osteosarcoma resemble those of ES, osteoid will provide an ultrastructural distinguishing point as it does in the light microscopic diagnosis [17,52].
- In pediatric age, a solid variant of alveolar rhabdomyosarcoma rarely presents as SRCT [41]. Like other tumors, the EM features of a solid variant of alveolar rhabdomyosarcoma depend on the degree of differentiation of rhabdomyoblasts [53]. Contrary to much more differentiated rhabdomyosarcoma, the primitive small cells are monomorphous round to elongated in shape and are surrounded by external lamina with a moderate amount of cytoplasm filled with glycogen and other organelles and have no easily detectable muscular features such as thick myosin-ribosome complexes or dense plaques. A careful search for thick myosin filament-ribosome complexes defined as 15-nm filaments intimately associated with numerous free ribosomes, the earliest evidence of rhabdomyoblast, can confirm the diagnosis of a rare solid variant of rhabdomyosarcoma (Fig. 1E) [41]. Ultrastructural evidence of skeletal differentiation such as specific myofilament arrays, focal density, Z-band material, and pinocytotic vesicles may not be detectable in solid variants of alveolar rhabdomyosarcoma [53]. Caution is needed because such neoplastic rhabdomyoblastic differentiation may be found in other SRCTs, such as malignant melanomas or schwannian Triton tumors.
- MPNST can present with small cell components, and it may cause confusion in small biopsy samples [54]. For example, benign peripheral nerve sheath tumor such as schwannoma, may show small cell components [55]. The presence of small cell changes of peripheral nerve sheath tumors is mainly regarded as malignant transformation; however, it sometimes implies differentiation toward immature neural cells [9]. Distinguishing poorly differentiated small cell MPNST from ES/PNET is not easy, at least on histologic and immunohistochemical grounds alone because MPNST has neither a reliable immunohistochemical marker nor specific moleculogenetic changes. Therefore, EM is often useful in detecting elongated interdigitating cytoplasmic processes, pinocytotic vesicles, and fragments of basal lamina [56]. In particular, the presence of basal lamina around tumor cells under EM confirms bidirectional differentiation into melanocytes and nerve sheath tumor. In such cases, basal lamina can be found in epithelial, myoepithelial, endothelial, Schwann or perineurial cells, or muscle cells, but cannot be seen in neurons, glial, hematolymphoid cells, and myofibroblastic cells [57]. Therefore, misinterpretation of the basal lamina of these surrounding reactive cells for the lamina of tumor cells should be avoided.
- Small cell variant of poorly differentiated monophasic synovial sarcoma, one of the poorly differentiated monophasic synovial sarcoma, should be distinguished from other SRCTs [8,58]. Monophasic synovial sarcoma showing epithelial membrane antigen–negative and cytokeratin-negative poorly differentiated one, requires either molecular cytogenetic study to evaluate the specific translocation t(X;18)(p11;q11) or EM examination [59]. Ultrastructurally, differentiation of synovial sarcoma ranges from unclassifiable spindle cells to fully differentiated epithelioid cells [60]. The spindle to fusiform-shaped tumor cells of synovial sarcoma has a high N/C ratio and non-descript cytoplasm devoid of schwannian differentiation, fibroblastic and leiomyosarcomatous differentiation, continuous basal lamina, long spacing collagen, and long intervening processes. Tapering bipolar processes joined by occasional rudimentary cell junctions or desmosomes, or infrequent tonofibrils must be distinguished from MPNST [60].
SRCT categorized as a soft tissue neoplasm
- The tumor cells of some SRCTs, particularly the solid variant of alveolar rhabdomyosarcomas, resemble lymphoblastic leukemic cells or non-Hodgkin’s lymphoma [61,62]. Conversely, dense infiltrates of relatively small undifferentiated tumor cells of uncertain origin, which mimic undifferentiated sarcoma, may be the first presentation of hematologic and lymphoid malignancies [63]. In particular, myeloid sarcomas with no evidence of circulating blasts may manifest as undifferentiated tumors with small round cell morphology, which frequently show aberrant immunophenotypes for myeloperoxidase, terminal deoxynucleotidyl transferase, CD68 (KP1), lysozyme, CD117, or CD34 [64]. Ultrastructural demonstration of myeloid granules of various morphology and stage are crucial in such cases [65-68]. Elongated Auer bodies as well as round or rod-like primary or azurophilic granules are useful ultrastructural findings. Primitive myeloid blasts have little cytoplasm but show greatly enlarged nucleoli with coarse granular nucleoplasm and thin peripheral heterochromatin (Fig. 1F) [16]. Myeloid tumors show morphological forms ranging from myeloblasts through promyelocytes to monocytes [68]. This is not the case for lymphoid malignancy [19,65,66] and subtyping of lymphoma has been attempted. EM cannot determine the line of differentiation in lymphoid malignancy, but it plays a role in excluding lymphoid malignancy. Abundant ribosomes and polysomes with an absence of cell junctions or basal lamina are the most common features of lymphoid malignancy.
SRCT of hematologic malignancies
- As previously mentioned in the SRCT of unknown histogenesis, MRTs showing rhabdoid morphology under light microscopy have been reported in every location of the body including the brain, liver, soft tissues, lung, skin, heart, and kidney, in which the tumor was originally described [11,69-72]. Recent immunohistochemistry reveals that the tumor cells have nuclear immunoreactivity for integrase interactor 1 (INI-1), which is critical for the diagnosis of this highly aggressive tumor, and show paranuclear bundles of intermediate filaments under ultrastructural examination [70]. Cell junctions are not observed in MRT. These paranuclear whorls of intermediate filaments are seen in other tumors of rhabdoid appearance; a rhabdoid variant of malignant melanoma also shows prominent nuclei and cytoplasmic accumulation of whorls of intermediate filaments, which compress the nuclei and a few organelles entrapped in the cytoplasm and melanosomes. As stated above, DSRCT also shares these ultrastructural findings. The so called rhabdoid appearance under light microscopy mimicking renal MRT (i.e., large cells with eccentrically located nuclei and abundant, eosinophilic cytoplasm) refers to tumors showing diffuse organoid sheets of round or polygonal shaped cells with paranuclear whorls of intermediate filaments. Tumors showing rhabdoid appearance are found in various tumors including pleomorphic fibrosarcoma with rhabdoid appearance, rhabdoid tumors of the CNS such as atypical teratoid/rhabdoid tumor, the CNS counterpart of MRT, rhabdoid meningioma, astroblastoma with rhabdoid appearance or rhabdoid choroid plexus carcinoma [70,71]. Extrarenal rhabdoid tumors are a heterogeneous group of neoplasms and it is also described as poorly differentiated neoplasms with rhabdoid features for these undifferentiated tumors [72]. Ultrastructural features shared with other tumors with rhabdoid features may not provide a diagnostic clue other than loss of INI-1 nuclear staining, which is critical for the diagnosis of this highly aggressive tumor.
- Under light microscopy, small cell (Merkel-like) melanomas appearing in adolescents and children are composed of small undifferentiated cells of melanocytic-lineage [15]. Ultrastructural examination is required to determine if the cells are pre-melanosomes or melanosomes of various stages and differentiating them from the reactive cells is important [73]. Pathologists should search meticulously for melanosome granules of various stages.
Other tumors presented with SRCTs
- In summary, the current shift in SRCT diagnosis is towards immunohistochemistry and cytogenetic molecular techniques, but sometimes these techniques are inadequate. Even with careful traditional analyses, the correct diagnosis remains elusive. Immunohistochemistry has limitations in sensitivity and specificity, while moleculocytogenetic study has proven to be useful in characterization of SRCTs; however, it has also become evident that some genetic abnormalities are not tumor specific, and even demonstrate results not correlating with the histologic morphology. As stated above, light microscopy should remain as the main diagnostic tool. As EM has several overlapping features (Table 1), it may not be able to identify a line of differentiation if the tumor cells are cytologically undifferentiated. However, EM should be included in the standard diagnostic processes for SRCTs, as it can provide both validity and reliability in its specificity.
- Here, we emphasize that pathologists need to understand the appropriate applications and pitfalls as well as the benefits of EM, which permits optimal diagnosis of SRCTs.
CONCLUSION
Acknowledgments
Fig. 1.(A) Ewing sarcoma shows polygonal shaped cells having large round cells and a small amount of cytoplasm with mainly glycogen pools (arrow) (×7,500). Inset indicates abundant cytoplasmic glycogen particles (×10,000). (B) Tumor cells of malignant rhabdoid tumor show paranuclear whorls packed with intermediate filaments intermingled with cytoplasmic organelles (×2,000). Inset shows paranuclear whorls filled with dense intermediate filaments, dilated rough endoplasmic reticulum (RER) cisternae and degenerated mitochondria (×8,000). (C) Neuroblastoma. Round shaped tumor cells have neurosecretory dense core granules in the cytoplasm (×8,000). (D) Small cell osteosarcoma shows ovoid shaped tumor cells containing a moderate amount of mitochondria and ribosomes with dilated RERs (×v8,500). (E) Alveolar rhabdomyosarcoma, solid variant. Closely apposed round to ovoid shaped cells have irregular shaped nuclei and a moderate amount of cytoplasm containing lipid vacuoles, mitochondria, and Golgi apparatuses (×3,500). Arrow indicates myosin-ribosome complexes (×12,000). (F) Myeloid sarcoma demonstrates cytoplasmic myeloid granules, measuring 200 nm on average (×1,500). Inset shows membrane bound myeloid granules (×6,000).
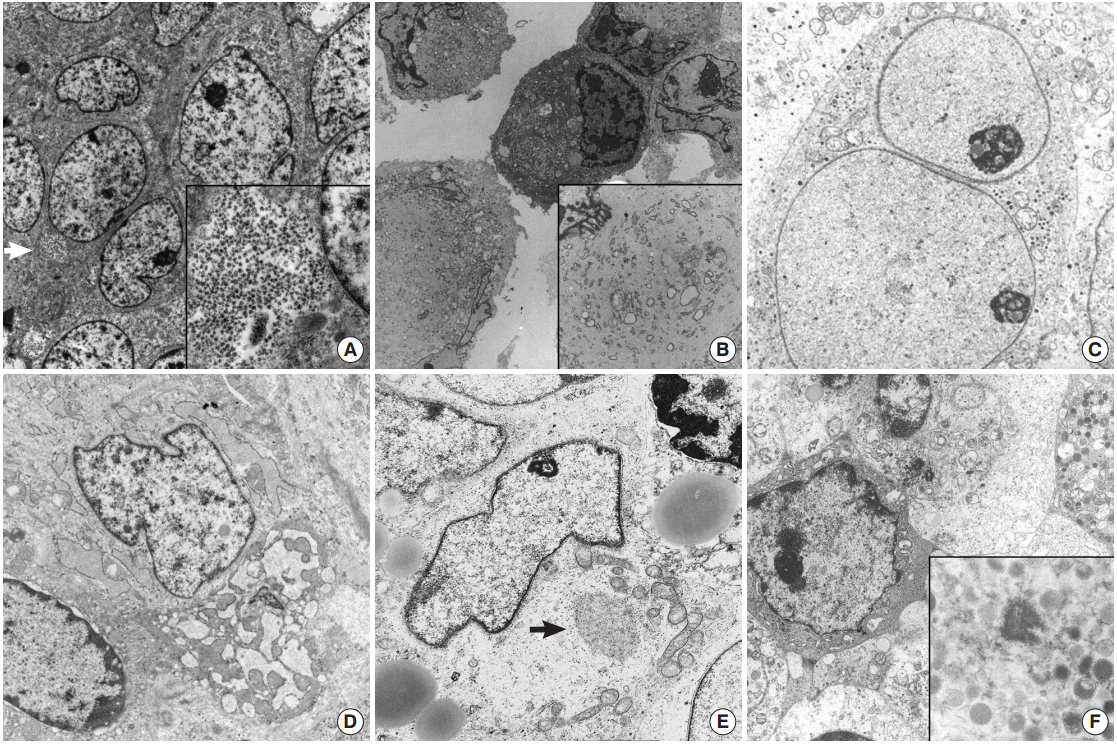

Table 1.Disease entities appearing as small round cell tumors along with ultrastructural differential characteristic findings
- 1. Bahrami A, Truong LD, Ro JY. Undifferentiated tumor: true identity by immunohistochemistry. Arch Pathol Lab Med 2008; 132: 326–48. ArticlePubMed
- 2. Rajwanshi A, Srinivas R, Upasana G. Malignant small round cell tumors. J Cytol 2009; 26: 1–10. ArticlePubMedPMC
- 3. Ponce-Castañeda MV, García-Chéquer AJ, Eguía Aguilar P, et al. Detection of common chromosomal translocations in small round blue cell pediatric tumors. Arch Med Res 2014; 45: 143–51. ArticlePubMed
- 4. Peydró-Olaya A, Llombart-Bosch A, Carda-Batalla C, Lopez-Guerrero JA. Electron microscopy and other ancillary techniques in the diagnosis of small round cell tumors. Semin Diagn Pathol 2003; 20: 25–45. PubMed
- 5. de Alava E, Ladanyi M, Rosai J, Gerald WL. Detection of chimeric transcripts in desmoplastic small round cell tumor and related developmental tumors by reverse transcriptase polymerase chain reaction: a specific diagnostic assay. Am J Pathol 1995; 147: 1584–91. PubMedPMC
- 6. Rossouw DJ, Cinti S, Dickersin GR. Liposarcoma: an ultrastructural study of 15 cases. Am J Clin Pathol 1986; 85: 649–67. ArticlePubMed
- 7. Alameda F, Corominas JM, Barranco C, et al. Primitive round cell liposarcoma of the omentum: diagnostic value of ultrastructural study. Ultrastruct Pathol 2003; 27: 433–7. ArticlePubMed
- 8. d’Amore ES, Ninfo V. Soft tissue small round cell tumors: morphological parameters. Semin Diagn Pathol 1996; 13: 184–203. PubMed
- 9. Roncaroli F, Betts C, Dorji T, Calbucci F, Eusebi V. Schwannoma with small cell component. Virchows Arch 2003; 443: 586–8. ArticlePubMed
- 10. Masjosthusmann K, Bielack SS, Köhler G, et al. Concomitant Ewing sarcoma and acute lymphoblastic leukemia in a 5-year-old girl. Pediatr Blood Cancer 2005; 45: 846–9. ArticlePubMed
- 11. Wagner LM, Garrett JK, Ballard ET, et al. Malignantrhabdoid tumor mimicking hepatoblastoma: a case report and literature review. Pediatr Dev Pathol 2007; 10: 409–15. ArticlePubMed
- 12. Franchi A, Pasquinelli G, Cenacchi G, et al. Immunohistochemical and ultrastructural investigation of neural differentiation in Ewing sarcoma/PNET of bone and soft tissues. Ultrastruct Pathol 2001; 25: 219–25. ArticlePubMed
- 13. Kurotaki H, Tateoka H, Takeuchi M, Yagihashi S, Kamata Y, Nagai K. Primary mesenchymal chondrosarcoma of the lung: a case report with immunohistochemical and ultrastructural studies. Acta Pathol Jpn 1992; 42: 364–71. PubMed
- 14. Steiner GC, Forest M, Vacher-Lavenu MC. Ultrastructure of lowgrade intraosseous osteosarcoma of bone: a comparative study with fibrous dysplasia and parosteal osteosarcoma. Ultrastruct Pathol 2006; 30: 293–9. ArticlePubMed
- 15. Barnhill RL. Childhood melanoma. Semin Diagn Pathol 1998; 15: 189–94. PubMed
- 16. Terzakis JA, Taskin M. Bone marrow leukemias and lymphoproliferative disorders: scanning electron microscope diagnosis. Ultrastruct Pathol 2002; 26: 143–52. ArticlePubMed
- 17. Salhany KE, Feldman M, Kahn MJ, et al. Hepatosplenic gammadelta T-cell lymphoma: ultrastructural, immunophenotypic, and functional evidence for cytotoxic T lymphocyte differentiation. Hum Pathol 1997; 28: 674–85. ArticlePubMed
- 18. Park SH, Kim I. Small cell osteogenic sarcoma of the ribs: cytological, immunohistochemical, and ultrastructural study with literature review. Ultrastruct Pathol 1999; 23: 133–40. ArticlePubMed
- 19. Mierau GW, Weeks DA, Hicks MJ. Role of electron microscopy and other special techniques in the diagnosis of childhood round cell tumors. Hum Pathol 1998; 29: 1347–55. ArticlePubMed
- 20. D’Cruze L, Dutta R, Rao S, R A, Varadarajan S, Kuruvilla S. The role of immunohistochemistry in the analysis of the spectrum of small round cell tumours at a tertiary care centre. J Clin Diagn Res 2013; 7: 1377–82. ArticlePubMedPMC
- 21. Ordóñez NG. Desmoplastic smallround cell tumor: II: an ultrastructural and immunohistochemical study with emphasis on new immunohistochemical markers. Am J Surg Pathol 1998; 22: 1314–27. ArticlePubMed
- 22. Ordonez NG. Desmoplastic small round cell tumor: I: a histopathologic study of 39 cases with emphasis on unusual histological patterns. Am J Surg Pathol 1998; 22: 1303–13. ArticlePubMed
- 23. Sawyer JR, Tryka AF, Lewis JM. A novel reciprocal chromosome translocation t(11;22)(p13;q12) in an intraabdominal desmoplastic small round-cell tumor. Am J Surg Pathol 1992; 16: 411–6. ArticlePubMed
- 24. Demicco EG. Sarcoma diagnosis in the age of molecular pathology. Adv Anat Pathol 2013; 20: 264–74. ArticlePubMed
- 25. Neuville A, Chibon F, Coindre JM. Grading of soft tissue sarcomas: from histological to molecular assessment. Pathology 2014; 46: 113–20. ArticlePubMed
- 26. Chibon F, Lagarde P, Salas S, et al. Validated prediction of clinical outcome in sarcomas and multiple types of cancer on the basis of a gene expression signature related to genome complexity. Nat Med 2010; 16: 781–7. ArticlePubMed
- 27. Min KW. Usefulness of electron microscopy in the diagnosis of “small” round cell tumors of the sinonasal region. Ultrastruct Pathol 1995; 19: 347–63. ArticlePubMed
- 28. Ghadially FN. Ultrastructural pathology of the cell and matrix. 4th ed. Newton: Butterworth-Heinemann, 1997.
- 29. Shindo D, Ikematsu Y, Lim SH, Yonenaga I. Digital electron microscopy on advanced materials. Mater Charact 2000; 44: 375–84. Article
- 30. Tucker JA. The continuing value of electron microscopy in surgical pathology. Ultrastruct Pathol 2000; 24: 383–9. ArticlePubMed
- 31. Turbat-Herrera EA, Herrera GA. Electron microscopy renders the diagnostic capabilities of cytopathology more precise: an approach to everyday practice. Ultrastruct Pathol 2005; 29: 475–82. ArticlePubMed
- 32. Laforga JB. Malignant epithelioid gastrointestinal stromal tumors: report of a case with cytologic and immunohistochemical studies. Acta Cytol 2005; 49: 435–40. ArticlePubMed
- 33. Sorensen PH, Lessnick SL, Lopez-Terrada D, Liu XF, Triche TJ, Denny CT. A second Ewing’s sarcoma translocation, t(21;22), fuses the EWS gene to another ETS-family transcription factor, ERG. Nat Genet 1994; 6: 146–51. ArticlePubMed
- 34. Chen Y, Jung SM, Chao TC. Malignant rhabdoid tumor of the small intestine in an adult: a case report with immunohistochemical and ultrastructural findings. Dig Dis Sci 1998; 43: 975–9. ArticlePubMed
- 35. Leuschner I, Radig K, Harms D. Desmoplastic small round cell tumor. Semin Diagn Pathol 1996; 13: 204–12. PubMed
- 36. Castleberry RP, Pritchard J, Ambros P, et al. The International Neuroblastoma Risk Groups (INRG): a preliminary report. Eur J Cancer 1997; 33: 2113–6. ArticlePubMed
- 37. Devall JM, Frush KM, Steiner L. Small blue round cell tumor: an unusual case presentation in the foot. J Am Podiatr MedAssoc 2011; 101: 363–9. Article
- 38. Santacruz MR, Proctor L, Thomas DB, Gehrig PA. Extraskeletal myxoid chondrosarcoma: a report of a gynecologic case. Gynecol Oncol 2005; 98: 498–501. ArticlePubMed
- 39. Shakked RJ, Geller DS, Gorlick R, Dorfman HD. Mesenchymal chondrosarcoma: clinicopathologic study of 20 cases. Arch Pathol Lab Med 2012; 136: 61–75. ArticlePubMed
- 40. Chong SM, Nilsson BS, Quah TC, Wee A. Malignant melanoma: an uncommon cause of smallround cell malignancy in childhood. Acta Cytol 1997; 41: 609–10. ArticlePubMed
- 41. Bianchi L, Orlandi A, Iraci S, Spagnoli LG, Nini G. Solid alveolar rhabdomyosarcoma of the hand in adolescence: a clinical, histologic, immunologic, and ultrastructural study. Pediatr Dermatol 1995; 12: 343–7. ArticlePubMed
- 42. Kilpatrick SE, Doyon J, Choong PF, Sim FH, Nascimento AG. The clinicopathologic spectrum of myxoid and round cell liposarcoma. A study of 95 cases. Cancer 1996; 77: 1450–8. ArticlePubMed
- 43. Tsokos M, Alaggio RD, Dehner LP, Dickman PS. Ewing sarcoma/peripheral primitive neuroectodermal tumor and related tumors. Pediatr Dev Pathol 2012; 15(1 Suppl):108–26. ArticlePubMedPMC
- 44. Abe S. Ultrastructural and immunohistochemical study of Ewing’s sarcoma and related tumors: morphological neural markers suggesting neural histogenesis in 32 small round-cell sarcomas. J Orthop Sci 1997; 2: 75–83. Article
- 45. Fisher C. The comparative roles of electron microscopy and immunohistochemistry in the diagnosis of soft tissue tumours. Histopathology 2006; 48: 32–41. ArticlePubMed
- 46. Joo M, Kang YK, Kim HS, Lee HK, Park YK. Mesenchymal chondrosarcoma of the hyoid bone: a case report. J Korean Med Sci 1998; 13: 696–700. ArticlePubMedPMC
- 47. Llombart-Bosch A, Contesso G, Peydro-Olaya A. Histology, immunohistochemistry, and electron microscopy of small round cell tumors of bone. Semin Diagn Pathol 1996; 13: 153–70. PubMed
- 48. Martinez-Tello FJ, Navas-Palacios JJ. Ultrastructural study of conventional chondrosarcomas and myxoid- and mesenchymal-chondrosarcomas. Virchows Arch A Pathol Anat Histol 1982; 396: 197–211. ArticlePubMed
- 49. Roy S, Meachim G. Chondrocyte ultrastructure in adult human articular cartilage. Ann Rheum Dis 1968; 27: 544–58. ArticlePubMedPMC
- 50. Kuroda N, Sogoh T, Ohara M, et al. Clear cell chondrosarcoma: an ultrastructural study. Med Mol Morphol 2009; 42: 185–8. ArticlePubMed
- 51. Corradi D, Bacchini P, Campanini N, Bertoni F. Aggressive clear cell chondrosarcomas: do distinctive characteristics exist?: a report of 4 cases. Arch Pathol Lab Med 2006; 130: 1673–9. ArticlePubMed
- 52. Mawad JK, Mackay B, Raymond AK, Ayala AG. Electron microscopy in the diagnosis of smallround cell tumors of bone. Ultrastruct Pathol 1994; 18: 263–8. ArticlePubMed
- 53. Erlandson RA. The ultrastructural distinction between rhabdomyosarcoma and other undifferentiated “sarcomas”. Ultrastruct Pathol 1987; 11: 83–101. ArticlePubMed
- 54. Abe S, Imamura T, Park P, et al. Small round-cell type of malignant peripheral nerve sheath tumor. Mod Pathol 1998; 11: 747–53. PubMed
- 55. Kurtkaya-Yapicier O, Scheithauer B, Woodruff JM. The pathobiologic spectrum of Schwannomas. Histol Histopathol 2003; 18: 925–34. PubMed
- 56. Kim NR, Chung DH, Park CY, Ha SY. Malignant peripheral nerve sheath tumor of the uterine cervix expressing both S-100 protein and HMB-45. J Obstet Gynaecol Res 2009; 35: 1136–41. ArticlePubMed
- 57. Eyden B. The myofibroblast: a study of normal, reactive and neoplastic tissues, with an emphasis on ultrastructure. part 2 - tumours and tumour-like lesions. J Submicrosc Cytol Pathol 2005; 37: 231–96. PubMed
- 58. Silverman JF, Landreneau RJ, Sturgis CD, et al. Small-cell variant of synovial sarcoma: fine-needle aspiration with ancillary features and potential diagnostic pitfalls. Diagn Cytopathol 2000; 23: 118–23. ArticlePubMed
- 59. Rong R, Doxtader EE, Tull J, de la Roza G, Zhang S. Metastatic poorly differentiated monophasic synovial sarcoma to lung with unknown primary: a molecular genetic analysis. Int J Clin Exp Pathol 2009; 3: 217–21. PubMedPMC
- 60. Dickersin GR. Synovial sarcoma: a review and update, with emphasis on the ultrastructural characterization of the nonglandular component. Ultrastruct Pathol 1991; 15: 379–402. ArticlePubMed
- 61. Sandberg AA, Stone JF, Czarnecki L, Cohen JD. Hematologic masquerade of rhabdomyosarcoma. Am J Hematol 2001; 68: 51–7. ArticlePubMed
- 62. Ganesan P, Thulkar S, Rajan A, Bakhshi S. Solid variant of alveolar rhabdomyosarcoma mimicking non-Hodgkin lymphoma: case report and review of literature. J Pediatr Hematol Oncol 2008; 30: 772–4. ArticlePubMed
- 63. Kim NR, Lee WK, Lee JI, Cho HY. Multiple jejunal myeloid sarcomas presenting with intestinal obstruction in a non-leukemic patient: a case report with ultrastructural observations. Korean J Pathol 2012; 46: 590–4. ArticlePubMedPMC
- 64. Dohner H, Estey EH, Amadori S, et al. Diagnosis and management of acute myeloid leukemia in adults: recommendations from an international expert panel, on behalf of the European LeukemiaNet. Blood 2010; 115: 453–74. ArticlePubMed
- 65. Dickersin GR. Electron microscopy of leukemias and lymphomas. Clin Lab Med 1987; 7: 199–247. ArticlePubMed
- 66. Goedhals J, Beukes CA, Cooper S. The ultrastructural features of plasmablastic lymphoma. Ultrastruct Pathol 2006; 30: 427–33. ArticlePubMed
- 67. Anderson DR. Ultrastructure of normal and leukemic leukocytes in human peripheral blood. J Ultrastruct Res 1966; 9: 1–42. ArticlePubMed
- 68. Takemori N, Hirai K, Onodera R, Saito N. Ultrastructural study of periodic lamellar granules in human neutrophils. Cell Tissue Res 1995; 281: 69–76. ArticlePubMed
- 69. Petitt M, Doeden K, Harris A, Bocklage T. Cutaneous extrarenal rhabdoid tumor with myogenic differentiation. J Cutan Pathol 2005; 32: 690–5. ArticlePubMed
- 70. Haberler C, Laggner U, Slavc I, et al. Immunohistochemical analysis of INI1 protein in malignant pediatric CNS tumors: Lack of INI1 in atypical teratoid/rhabdoid tumors and in a fraction of primitive neuroectodermal tumors without rhabdoid phenotype. Am J Surg Pathol 2006; 30: 1462–8. ArticlePubMed
- 71. Peng HQ, Stanek AE, Teichberg S, Shepard B, Kahn E. Malignant rhabdoid tumor of the kidney in an adult: a case report and review of the literature. Arch Pathol Lab Med 2003; 127: e371–3. ArticlePubMed
- 72. Parham DM, Weeks DA, Beckwith JB. The clinicopathologic spectrum of putative extrarenal rhabdoid tumors: an analysis of 42 cases studied with immunohistochemistry or electron microscopy. Am J Surg Pathol 1994; 18: 1010–29. ArticlePubMed
- 73. Magro CM, Crowson AN, Mihm MC. Unusual variants of malignant melanoma. Mod Pathol 2006; 19 Suppl 2: S41–70. Article
References
Figure & Data
References
Citations
Citations to this article as recorded by 

- Electron Microscopy in the Context of a Children's Research Hospital
Cam Robinson
Microscopy and Microanalysis.2020; 26(S2): 1610. CrossRef - Primary bilateral corneal nerve sheath neoplasm in a dog
Marina L. Leis, M. Elyse Salpeter, Bianca S. Bauer, Dale L. Godson, Bruce H. Grahn
Veterinary Ophthalmology.2017; 20(4): 365. CrossRef - Hirnbasissyndrom infolge eines Tumors bei einer 17 Monate alten Deutsch-Holstein-Färse
Wolf Wippermann, Sandra Schöniger, Kerstin Gerlach, Gerald Fritz Schusser, Gabor Köller, Alexander Starke
Tierärztliche Praxis Ausgabe G: Großtiere / Nutztiere.2016; 44(03): 180. CrossRef - The Continuing Value of Ultrastructural Observation in Central Nervous System Neoplasms in Children
Na Rae Kim, Sung-Hye Park
Journal of Pathology and Translational Medicine.2015; 49(6): 427. CrossRef
 PubReader
PubReader ePub Link
ePub Link-
 Cite this Article
Cite this Article
- Cite this Article
-
- Close
- Download Citation
- Close
- Figure

 E-submission
E-submission