Articles
- Page Path
- HOME > J Pathol Transl Med > Volume 49(6); 2015 > Article
-
Original Article
Therapeutic Effects of Umbilical Cord Blood Derived Mesenchymal Stem Cell-Conditioned Medium on Pulmonary Arterial Hypertension in Rats - Jae Chul Lee1,2,3, Choong Ik Cha3, Dong-Sik Kim2, Soo Young Choe1
-
Journal of Pathology and Translational Medicine 2015;49(6):472-480.
DOI: https://doi.org/10.4132/jptm.2015.09.11
Published online: October 16, 2015
1Department of Biology, School of Life Sciences, Chungbuk National University, Cheongju, Korea
2Department of Surgery, Brain Korea 21 PLUS Project for Medical Sciences and HBP Surgery and Liver Transplantation, Korea University College of Medicine, Seoul, Korea
3Department of Anatomy, Seoul National University College of Medicine, Seoul, Korea
-
Corresponding Author: Soo Young Choe, PhD Department of Biology, School of Life Sciences, Chungbuk National University, 1 Chungdae-ro, Seowon-gu, Cheongju 28644, Korea Tel: +82-43-261-2297 Fax: +82-43-275-2291 E-mail:leejc@chungbuk.ac.kr
Dong-Sik Kim, MD, PhD Department of Surgery, Brain Korea 21 PLUS Project for Medical Sciences and HBP Surgery and Liver Transplantation, Korea University College of Medicine, 145 Anam-ro, Seongbuk-gu, Seoul 02841, Korea Tel: +82-2-2286-1431 Fax: +82-2-2286-1428 E-mail:beas100@korea.ac.kr
© 2015 The Korean Society of Pathologists/The Korean Society for Cytopathology
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/3.0/) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
Abstract
-
Background:
- Human umbilical cord blood-derived mesenchymal stem cells (hUCB-MSCs) may have multiple therapeutic applications for cell based therapy including the treatment of pulmonary artery hypertension (PAH). As low survival rates and potential tumorigenicity of implanted cells could undermine the mesenchymal stem cell (MSC) cell-based therapy, we chose to investigate the use of conditioned medium (CM) from a culture of MSC cells as a feasible alternative.
-
Methods:
- CM was prepared by culturing hUCB-MSCs in three-dimensional spheroids. In a rat model of PAH induced by monocrotaline, we infused CM or the control unconditioned culture media via the tail-vein of 6-week-old Sprague-Dawley rats.
-
Results:
- Compared with the control unconditioned media, CM infusion reduced the ventricular pressure, the right ventricle/(left ventricle+interventricular septum) ratio, and maintained respiratory function in the treated animals. Also, the number of interleukin 1α (IL-1α), chemokine (C-C motif) ligand 5 (CCL5), and tissue inhibitor of metalloproteinase 1 (TIMP-1)–positive cells increased in lung samples and the number of terminal deoxynucleotidyl transferase–mediated deoxyuridine triphosphate nick-end labeling technique (TUNEL)–positive cells decreased significantly in the CM treated animals.
-
Conclusions:
- From our in vivo data in the rat model, the observed decreases in the TUNEL staining suggest a potential therapeutic benefit of the CM in ameliorating PAH-mediated lung tissue damage. Increased IL-1α, CCL5, and TIMP-1 levels may play important roles in this regard.
- Animals
- Six-week-old male Sprague-Dawley rats were used. All rats were housed in climate-controlled conditions with a 12-hour light/12-hour dark cycle, and had free access to food and water. All animal experiments were approved by the appropriate Institutional Review Boards of the Seoul National University College of Medicine (Seoul, Korea; SNU-101122-2) and conducted in accordance with National Institutes of Health Guide for the Care Use of Laboratory Animals (NIH publication No. 86-23, revised in 1996).
- Pulmonary arterial hypertension rat model
- PAH was induced by subcutaneous injection of 50 mg/kg monocrotaline (MCT; Sigma-Aldrich, St. Louis, MO, USA) dissolved in 0.5 N HCl. The rats were grouped into a control group (C group) (n=20), injection of α–minimal essential medium (αMEM) followed by MCT group (M group) (n=20), and injection of MCT followed by hUCB-MSC-CM transfusion group (CM group) (n=20). αMEM and hUCB-MSC-CM (0.5 μL/hr) were transfused by tail-vein 7 days after MCT injection. The animals were sacrificed at 7, 14, 21, and 28 days after hUCB-MSC-CM transfusion. Tissues were removed and immediately frozen at –70°C for enzyme analysis.
- Cell preparation and culture of hUCB-MSCs
- hUCB-MSCs were obtained from the Biomedical Research Institute (Seoul, Korea). Isolated human MSCs were expanded in culture as previously described [6]. hUCB-MSCs were maintained in αMEM (Gibco, Grand Island, NY, USA) supplemented with 10% fetal bovine serum (Gibco), 100 U/mL penicillin (Gibco), and 100 g/mL streptomycin (Gibco). Passages up to 5 were used for experiments.
- Preparation of hUCB-MSC-CM
- To generate hUCB-MSC-CM spheroids [16,17], 30 μL of cell suspension (1×106 cells/mL) were applied to the lid of a Petri dish containing phosphate buffered saline (PBS). After 24 hours of incubation, spheroids formed in the drops were retrieved. For the three-dimensional bioreactor culture, hUCB-MSC spheroids (4.2×107 cells) were cultured in a siliconized spinner flask (Bellco, Vineland, NJ, USA) containing 70 mL αMEM with stirring at 70 rpm. To obtain CM, the medium was changed to αMEM without serum, and the cells were cultured for 2 days. CM was collected by centrifugation [14].
- Determination of the organ weights and right hypertrophy index
- The rats were weighed and observed for general appearance during the study period. The animals were sacrificed at the scheduled time. The wet weights of the excised right ventricle (RV), left ventricle (LV), and interventricular septum (IVS) were measured. The weights of the LV and IVS were added (LV+IVS) to determine the RV to LV+IVS ratio [RV/(LV+IVS)], which was used to determine the right hypertrophy index.
- Pulmonary hemodynamics
- Rats were anaesthetized by intraperitoneal injection of urethane and secured on a surgical stage. An 8-mm-long right internal jugular vein was isolated and ligated at the distal end. The vessel was cut at the proximal end of ligation. A catheter filled with heparinized saline was rapidly inserted along the incision and slowly advanced for about 5 cm to enter the pulmonary artery. The standard of pulmonary hypertension was defined as a systolic pulmonary pressure (SPAP) larger than 50 mm Hg [18]. Hemodynamic parameters were recorded at baseline and at 7, 14, 21, and 28 days.
- Immunohistochemistry
- Excised lung tissues were incubated overnight in 10% buffered formalin. Four-micrometer-thick sections were cut from paraffin embedded tissue blocks, deparaffinized in xylene, and rehydrated in graded alcohol solutions (70%–100%). Heat antigen retrieval was achieved by boiling the tissue sections in antigen retrieval solution in pH 6.0 or pH 9.0 (Dako, Carpinteria, CA, USA) for 10 minutes in a microwave prior to incubation at 4°C overnight with primary antibodies against interleukin 1α (IL-1α), chemokine (C-C motif) ligand 5 (CCL5), and tissue inhibitor of metalloproteinase 1 (TIMP-1; Abcam, Cambridge, MA, USA). After incubation with the appropriate biotinylated secondary antibodies for 30 minutes at 4°C and subsequently with streptavidin (Dako, Kyoto, Japan), color development was done using diaminobenzidine (DAB) as a chromogen and counterstained with hematoxylin.
- Western blot analysis
- The tissue was homogenized in 10 mM Tris HCl buffer, pH 7.4 containing 0.5 mM ethylenediaminetetraacetic acid, pH 8.0, 0.25 M sucrose, 1 mM phenylmethylsulfonyl fluoride, 1 mM Na4VO3, and a protease inhibitor cocktail (Roche-Boehringer-Mannheim, Mannheim, Germany). After centrifugation, the supernatant was subjected to sodium dodecyl sulfate polyacrylamide gel electrophoresis (SDS-PAGE). Samples equivalent to 25 μg of protein content were loaded and size-separated by 8%–12% SDS-PAGE. The proteins on the acrylamide gel were transferred to a polyvinylidene difluoride membrane (Millipore, Bedford, MA, USA) at 400 mA in a transfer buffer containing 25 mM Tris and 192 mM glycine, pH 8.4. The nitrocellulose membrane was blocked in tris-buffered saline with 5% non-fat dry milk at room temperature for 1 hour in 0.1% Tween 20 and incubated with the appropriated primary antibodies, including anti–IL-1α (Santa Cruz Biotechnology, Santa Cruz, CA, USA), anti-CCL5 (Fitzgerald Industries International, Concord, MA, USA), anti–TIMP-1 (Abcam), and anti-caspase-3, anti-Bcl-2, anti-actin (Santa Cruz Biotechnology) at 4°C for overnight. The membranes were then incubated with horseradish peroxidase-conjugated secondary antibody (Cell Signaling Technology, Danvers, MA, USA) for 1 hour at room temperature. After washing, the membranes were visualized by a chemiluminescent ECL-detection kit from GE-Healthcare (Piscataway, NJ, USA).
- Cytokine array and gene expression in lung tissues
- The lung samples were collected at termination (4 days after hUCB-MSC-CM injection) and quickly frozen in liquid nitrogen. A rat cytokine array (ARY008, R&D Systems, Minneapolis, MN, USA) was used to screen the lung homogenates according to the manufacturer’s instructions. The samples were pooled per treatment group and equal amounts of protein were loaded on the blots.
- In situ terminal deoxynucleotidyl transferase–mediated deoxyuridine triphosphate nick-end labeling technique assay for lung cell apoptosis
- Apoptotic cells in the tissue sections were detected by the terminal deoxynucleotidyl transferase-mediated deoxyuridine triphosphate nick-end labeling technique (TUNEL) using a commercial apoptosis kit (TACS TM TdT Kit, R&D Systems), according to the supplier’s instructions. In brief, the lung tissue sections were de-paraffinized with xylene and ethanol and rinsed with PBS. The sections were then treated with proteinase K in PBS followed by quenching of endogenous peroxidase. A biotinylated dNTP mix was added to the 3’-OH ends of DNA by terminal deoxynucleotidyl transferase (TdT). After incubating with streptavidin-horseradish peroxidase, the sections were stained with DAB and counterstained with methyl green. Finally, the sections were dehydrated in ethanol, cleared with xylene, and mounted with coverslips in a permanent medium. According to the supplier’s instructions, experimental controls included for the assay were TACS-nuclease–treated thyroid tissue sections as the positive control and the omission of the TdT reaction step as the negative control.
- Statistical analyses
- Results were expressed as the mean±standard deviation. An unpaired two-tailed t test and Mann-Whitney test were used, and a p-value less than .05 was considered statistically significant. SPSS ver. 14.0 for Windows (SPSS Inc., Chicago, IL, USA) was used for all statistical analyses.
MATERIALS AND METHODS
- Changes in body and organ weights and systolic pulmonary artery pressure after injection with hUCB-MSC-CM in PAH rats
- hUCB-MSC-CM has the potential to increase cell differentiation and induce immune modulation in various disease models [19,20]. However, the role of hUCB-MSC-CM in PAH has not been well elucidated. To address this, in our rat model of PAH, following MCT treatment, we treated rats with hUCB-MSC-CM and sham treated for the control group. There was a significant decrease in body weight at 14, 21, and 28 days in the MCT group (M group) compared to the control group (C group). However, body weight increased at 21 and 28 days in the conditioned media treated group (CM group) compared to the M group. The M group also showed increased weights of the RV at 21 and 28 days. The sum weight of LV+IVS was not significantly different between the C, M, and CM groups at the time point tested. The ratio of RV to LV+IVS, namely RV/LV+IVS, was significantly higher at 14, 21, and 28 days in the M group compared with the C group. However, the RV/LV+IVS ratio was significantly decreased at 28 days in the CM group compared with the M group. Also, LV+IVS was significantly lower in both M and CM groups compared to the C group at 14, 21, and 28 days. The lung weight was significantly increased in the M group compared with the C group at 21 and 28 days. However, the lung weight was significantly decreased in the CM group compared to the M group at 28 days (Table 1). The mean SPAP was also significantly increased in the M group compared to the C and CM groups at 14, 21, and 28 days (Table 2).
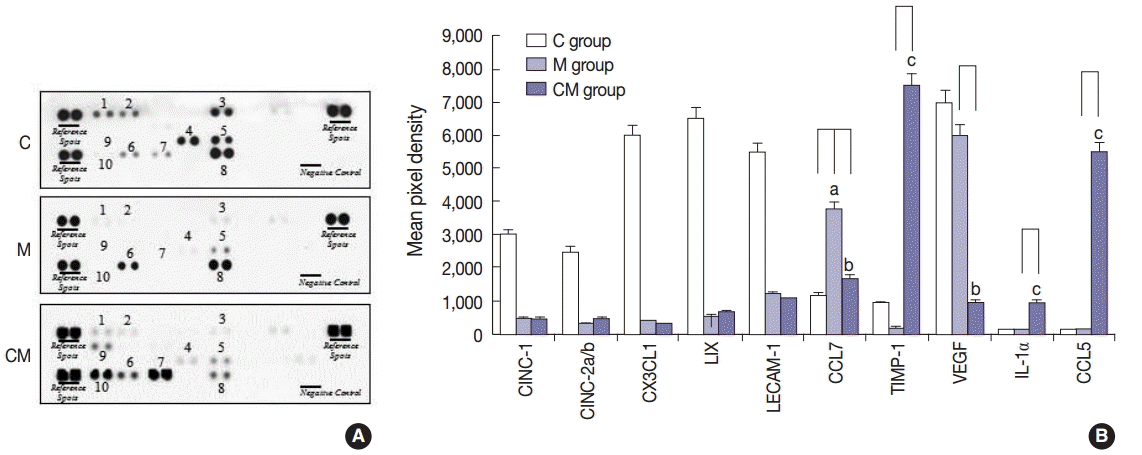
- Cytokine profile in the lung tissues after hUCB-MSC-CM treatment
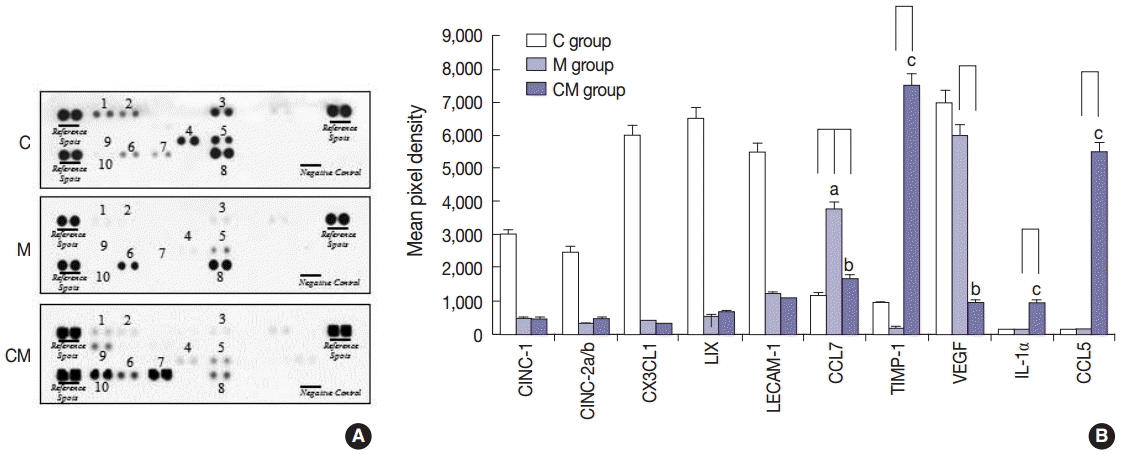
- A profile of the cytokines in the lung homogenates was made to investigate potential changes from hUCB-MSC-CM treatment (Fig. 1). Ten pro-inflammatory cytokines that included cytokine-induced neutrophil chemoattractant-1 (CINC-1), cytokine-induced neutrophil chemoattractant-2a/b (CINC-2a/b), chemokine (C-X-C motif) ligand 1 (CX3CL1), lipopolysaccharide-induced CXC chemokine (LIX), leukocyte endothelial cell adhesion molecule 1 (LECAM-1), chemokine (C-X-C motif) ligand 7, TIMP-1, vascular endothelial growth factor (VEGF), IL-1α, and CCL5 were examined in the C, M, and CM groups. CINC-1, CINC-2a/b, CX3CL1, LIX, LECAM-1, TIMP-1, and VEGF were lower in the M and CM groups, whereas TIMP-1, IL-1α, and CCL5 were higher in the CM group compared to the C and M groups. CCL7 was higher in the M group, whereas CCL7 was lower in the CM group compared to the M group (Fig. 1).
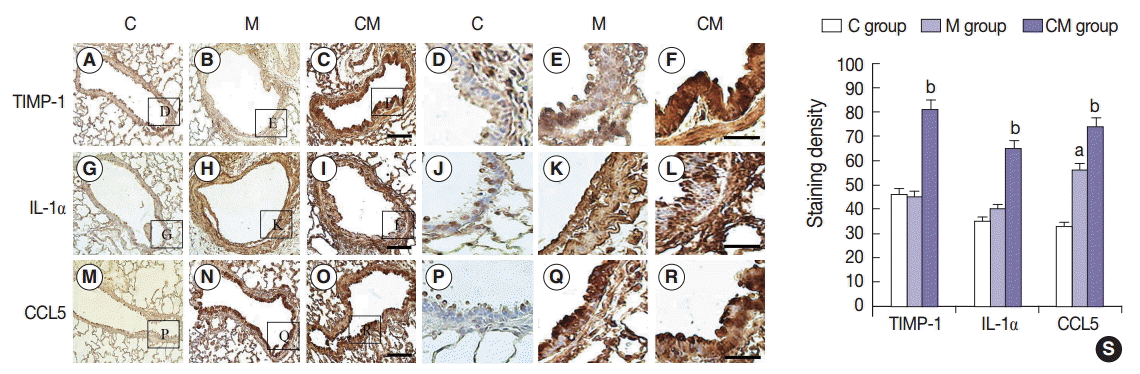
- Immunohistochemistry analysis of lung samples
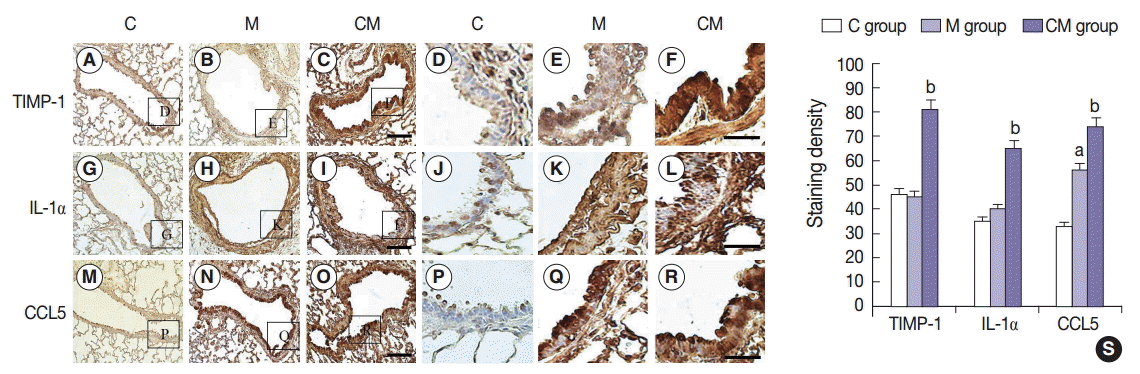
- Immunohistochemistry (IHC) staining of the lung tissue revealed that TIMP-1-, IL-1α-, and CCL5-positive cells were more prevalent in the CM group, and then followed by the M group in comparison with the C group at 28 days (Fig. 2A–R). These results confirmed that hUCB-MSC-CM increased the expression of certain immunomodulating cytokines (at the protein level) in the lungs of treated animals. Three weeks after hUCB-MSC-CM transfusion, TIMP-1-, IL-1α-, and CCL5-positive cells were still observed at the transplanted lung area in the CM group. The increased levels of TIMP-1, IL-1α, and CCL5 immunoreactivity observed in the M group were statistically significant (p<.05). The increased levels of CCL5 immunoreactivity were also significant in the CM group compared with the M group (Fig. 2S).
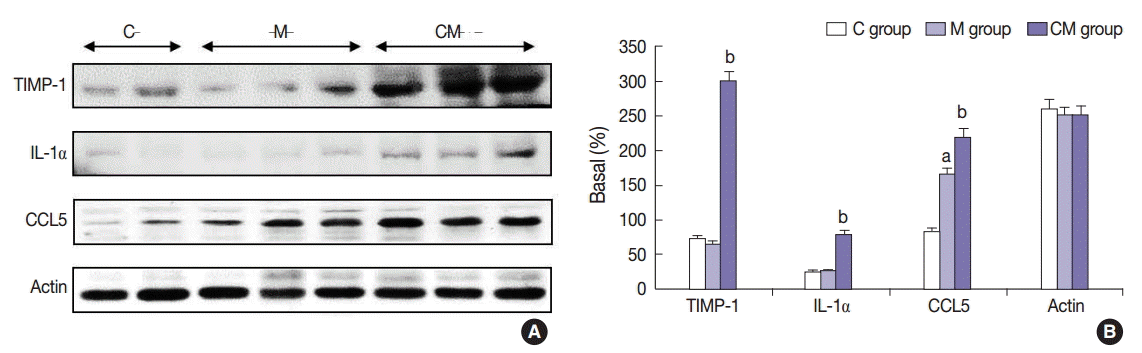
- Western blot analysis
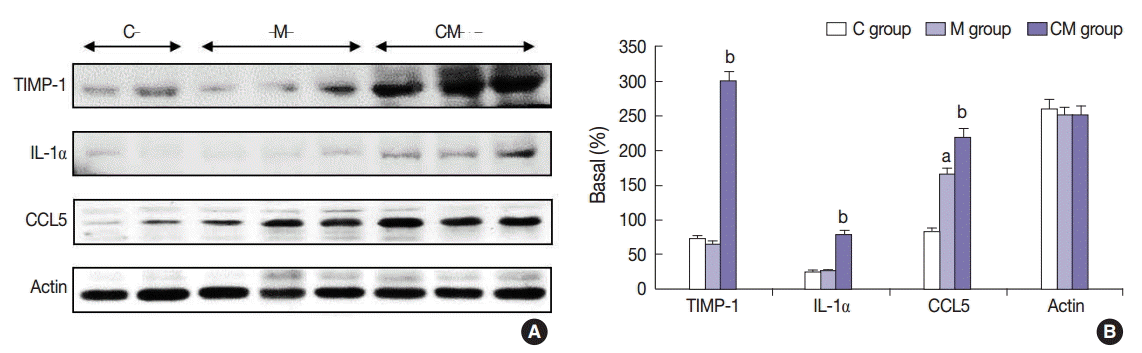
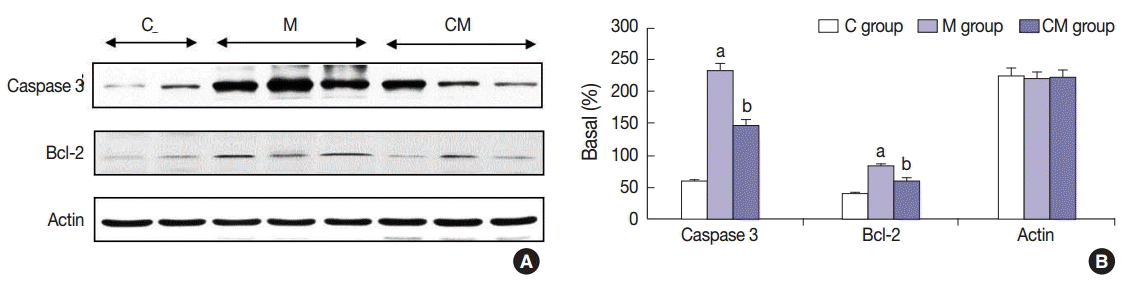
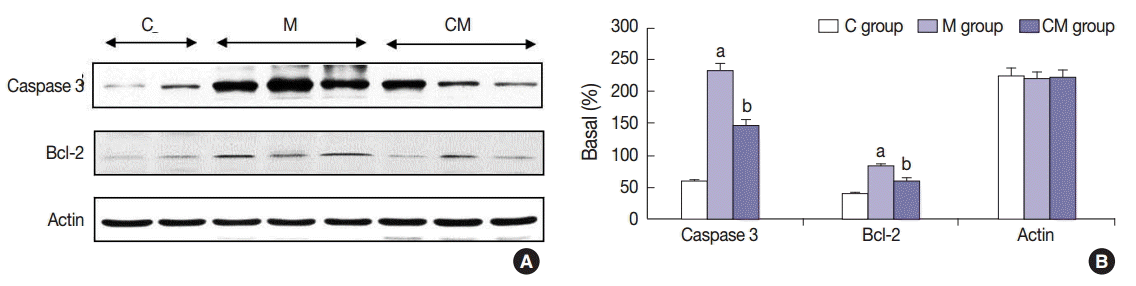
- The protein expressions of CCL5 at 28 days were significantly increased in the M group compared to the C group. The protein expressions of TIMP-1, IL-1α, and CCL5 at 28 days were significantly increased in the CM group compared to the M group (Fig. 3). The protein expressions of caspase-3 and Bcl-2 were significantly increased in the M group compared to the C group at 28 days. The protein expressions of caspase-3 and Bcl-2 were significantly decreased in the CM group compared to the M group at 28 days (Fig. 4).
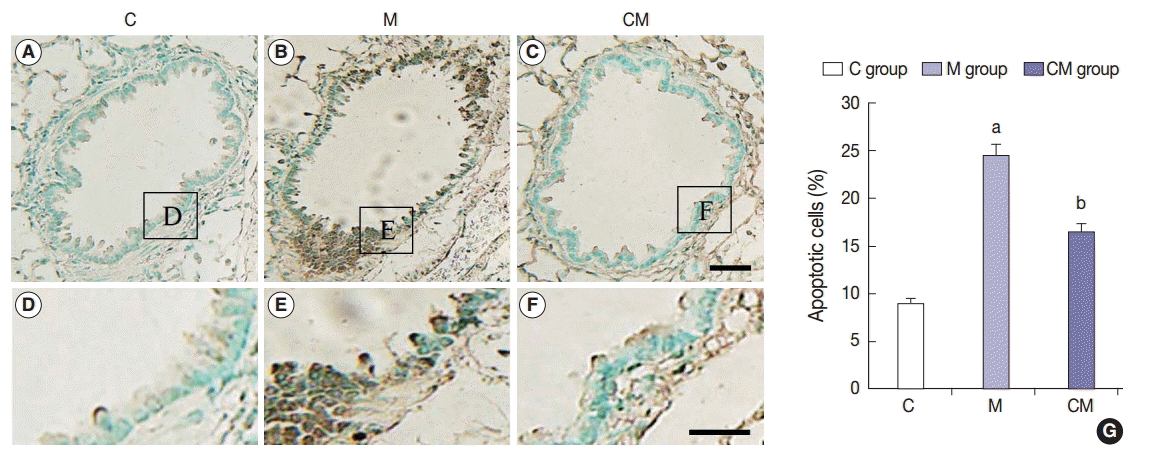
- TUNEL apoptosis assay
- The TUNEL staining was performed to detect apoptotic DNA in the lung tissue. The assayed C group did not have any positive staining (Fig. 5A, D). However, the M group had lung tissues with a positive TUNEL staining as seen by the presence of dark brown nuclei (Fig. 5B, E). CM group also contained cells with brown nuclei, indicating apoptotic DNA (Fig. 5C, F). Apoptotic cells were significantly more prevalent in the M group than in the C group, but they were less prevalent in the CM group than in the M group (Fig. 5G). The results indicated that hUCB-MSC-CM could attenuate apoptosis in the lung tissues of treated PAH rats.
RESULTS
- In this study, we tested the effects of CM infusion on PAH affected lung tissue in a rat model. It was previously demonstrated that CM of hUCB-MSCs contain active levels of a number of disease modifying growth factors and cytokines [21,22]. CM of hUCB-MSCs contain sizable levels of angiopoietin, hepatocyte growth factor, interleukin-4, insulin-like growth factor, placental growth factor, vascular endothelial cell growth factor, angiogenin, stem cell factor, and tyrosine hydroxylase [5,23-25]. Our previous studies demonstrated the neuroprotective effects of conditioned media from hADSC and hNSC in rat models of stroke and Huntington’s disease [14,15]. Therefore, we chose to test the CM prepared from hUCB-MSCs in a PAH rat model for therapeutic signals.
- MSCs are multipotent stromal cells that have self-renewal capacity, and can differentiate into a variety of cell types such as osteoblasts, chondrocytes, myocytes, and adipocytes [26,27]. MSCs have been isolated from several different sources such as embryonic tissue, bone marrow, adipose tissue, and the placenta [28]. MSCs are the source of many immune-dampening cytokines and in this regard, have demonstrated potency in a number of disease models [29,30]. In addition the secreted factors from MSCs display antiapoptotic, proliferative activity and the cells may be involved in the removal of harmful factors from their vicinity [23,31].
- From our study, we detected relatively high concentrations of CCL5, TIMP-1, and IL-1α in hUCB-MSC-CM treated lung tissues compared with MCT alone (M) and control (C) groups as confirmed by a rat cytokine array panel (Fig. 1). Cytokines play important roles in a number of biological processes including innate immunity, apoptosis, angiogenesis, cell growth, and differentiation [32]. These processes play important roles in disease protection and recovery.
- Lipopolysaccharide-induced CXC chemokine (also termed CXCL5) is a member of the CXC chemokine family, and is a potent neutrophil chemoattractant [33]. A TIMP-1 is a naturally occurring inhibitor of metalloproteinases [34-36], and TIMPs inhibit tumorigenesis, cellular invasion, metastasis, and angiogenesis. TIMPs may also promote tumor growth and inhibit apoptosis. These opposite roles of TIMPs in tumor regression and progression have been attributed to modulation by the tissue microenvironment [37]. Many cytokines induce endothelial cells to express adhesion molecules and lead to secretion of chemokines that attract white blood cells to a site of injury [38,39]. In our study, for the hUCB-MSC-CM treated PAH induced animals, the lung tissues showed significant increases in the number of IL-1α-positive pulmonary arterioles compared with the control group.
- IL-1α (and also tumor necrosis factor α) are known to stimulate proliferation of endothelial cells and fibroblasts that increase the blood supply at the site of injury and repair damage [40]. The IL-1 family includes the structurally related proteins IL-1α, IL-1β, and IL-1 receptor antagonist that bind to the same receptor. The IL-1 family plays an important role in interstitial lung diseases. Previous research has demonstrated that IL-1α expression levels in the lung correlated with the development of pulmonary fibrosis in rodents exposed to bleomycin or radiation [41,42]. Furthermore, studies have demonstrated up-regulation of IL-1α expression in fibro-proliferative areas within the lungs of idiopathic pulmonary fibrosis patients [43]. IHC for IL-1α, CCL5, and TIMP-1 confirmed the lung cell increases for these three cytokines previously seen in the lung homogenates for the three cytokines (Fig. 2). How the characteristics of the above cytokines may either ameliorate or exacerbate the effects of PAH remain to be explored.
- The numbers of TUNEL-positive cells in the lung areas were also significantly reduced by the infusion of hUCB-MSC-CM (Fig. 5). The hUCB-MSC-CM treatment was initiated 28 days after induction of PAH. Therefore, the reduction of apoptosis could be due to protective mechanisms of the hUCB-MSC-CM. These therapeutic effects could provide a clinically relevant benefit to patients. For our study, although no cells were implanted, our data demonstrated that an infusion of hUCB-MSC-CM can significantly reduce lung cell apoptosis due to PAH in our rat model. This novel therapeutic modality could be a viable treatment for PAH and bypass several technical limitations of a direct MSC cell transplantation. The present study also revealed certain changes in chemokine, cytokine, and growth factor levels after hUCB-MSC-CM transfusion in a PAH rat model. Through a complex interaction of these mediators involved in immunomodulation and inflammation, we may expect a positive effect on reducing the impact of PAH on lung cells. Exactly how these cytokines and factors interact to impact the survival of the lung tissue cells remains to be explored. As there are several treatment options available for PAH in people, an effective therapy in prolonging survival remains elusive. Our data with factors present in hUCB-MSC-CM may present an exciting opportunity for more effective therapies.
- The limitations of our study included the small sample size and a short follow-up of the treated animals. Future studies with larger sample sizes and a longer duration of treatment will be required, along with standardizing the quality and amount of hUCB-MSC-CM, frequency, and the duration required for the treatment.
DISCUSSION
Acknowledgments

| Day | Group | Body weight (g) | RV (g) | LV + IVS (g) | RV / (LV + IVS) (%) |
|---|---|---|---|---|---|
| 7 | Control | 318.63 ± 14.78 | 0.132 ± 0.02 | 0.611 ± 0.02 | 0.21 ± 0.01 |
| M | 278.50 ± 32.71 | 0.155 ± 0.03 | 0.543 ± 0.03 | 0.28 ± 0.02 | |
| CM | 280.46 ± 29.82 | 0.164 ± 0.02 | 0.561 ± 0.03 | 0.29 ± 0.02 | |
| 14 | Control | 343.65 ± 24.52 | 0.156 ± 0.02 | 0.731 ± 0.03 | 0.21 ± 0.02 |
| M | 256.71 ± 45.57a | 0.234 ± 0.03 | 0.671 ± 0.02 | 0.34 ± 0.03a | |
| CM | 271.21 ± 38.82 | 0.224 ± 0.04 | 0.699 ± 0.03 | 0.32 ± 0.02 | |
| 21 | Control | 393.81 ± 24.62 | 0.166 ± 0.03 | 0.782 ± 0.03 | 0.21 ± 0.02 |
| M | 249.67 ± 47.29a | 0.314 ± 0.06a | 0.677 ± 0.05 | 0.46 ± 0.05a | |
| CM | 271.00 ± 51.55b | 0.284 ± 0.05 | 0.631 ± 0.03 | 0.45 ± 0.03 | |
| 28 | Control | 394.00 ± 41.61 | 0.171 ± 0.02 | 0.801 ± 0.03 | 0.21 ± 0.02 |
| M | 229.71 ± 44.82a | 0.394 ± 0.08a | 0.751 ± 0.06 | 0.52 ± 0.07a | |
| CM | 319.29 ± 36.62b | 0.261 ± 0.06b | 0.732 ± 0.04 | 0.35 ± 0.04b |
Values are presented as mean ± standard deviation.
hUCB-MSCs-CM, conditioned medium from human umbilical-cord blood derived mesenchymal cells; PAH, pulmonary artery hypertension; RV, right ventricle; LV, left ventricle; IVS, interventricular septum; M, monocrotaline; CM, hUCB-MSCs-CM.
ap < .05 compared with the C group;
bp < .05 compared with the M group.
| Day | C group | M group | CM group |
|---|---|---|---|
| 7 | 22.7 ± 0.6 | 24.5 ± 2.1 | 23.2 ± 3.4 |
| 14 | 22.0 ± 1.1 | 37.8 ± 3.2a | 30.9 ± 4.6 |
| 21 | 23.3 ± 0.9 | 50.2 ± 4.7a | 39.2 ± 5.2b |
| 28 | 22.9 ± 2.1 | 58.0 ± 6.4a | 37.8 ± 4.1b |
- 1. Farber HW, Loscalzo J. Pulmonary arterial hypertension. N Engl J Med 2004; 351: 1655-65. ArticlePubMed
- 2. Liang OD, Mitsialis SA, Chang MS, et al. Mesenchymal stromal cells expressing heme oxygenase-1 reverse pulmonary hypertension. Stem Cells 2011; 29: 99-107. ArticlePubMedPMCPDF
- 3. Can MM, Tanboga IH, Demircan HC, et al. Enhanced hemostatic indices in patients with pulmonary arterial hypertension: an observational study. Thromb Res 2010; 126: 280-2. ArticlePubMed
- 4. Fukumoto Y, Shimokawa H. Recent progress in the management of pulmonary hypertension. Circ J 2011; 75: 1801-10. ArticlePubMed
- 5. Liu Q, Luo Z, He S, et al. Conditioned serum-free medium from umbilical cord mesenchymal stem cells has anti-photoaging properties. Biotechnol Lett 2013; 35: 1707-14. ArticlePubMedPDF
- 6. Lee JC, Kim KC, Yang YS, et al. Microarray analysis after umbilical cord blood derived mesenchymal stem cells injection in monocrotaline-induced pulmonary artery hypertension rats. Anat Cell Biol 2014; 47: 217-26. ArticlePubMedPMC
- 7. Zhao YD, Courtman DW, Ng DS, et al. Microvascular regeneration in established pulmonary hypertension by angiogenic gene transfer. Am J Respir Cell Mol Biol 2006; 35: 182-9. ArticlePubMed
- 8. Saigawa T, Kato K, Ozawa T, et al. Clinical application of bone marrow implantation in patients with arteriosclerosis obliterans, and the association between efficacy and the number of implanted bone marrow cells. Circ J 2004; 68: 1189-93. ArticlePubMed
- 9. Kang H, Kim KH, Lim J, et al. The therapeutic effects of human mesenchymal stem cells primed with sphingosine-1 phosphate on pulmonary artery hypertension. Stem Cells Dev 2015; 24: 1658-71. ArticlePubMedPMC
- 10. Umar S, de Visser YP, Steendijk P, et al. Allogenic stem cell therapy improves right ventricular function by improving lung pathology in rats with pulmonary hypertension. Am J Physiol Heart Circ Physiol 2009; 297: H1606-16. ArticlePubMed
- 11. Kim AK, Kim MH, Kim S, et al. Stem-cell therapy for peripheral arterial occlusive disease. Eur J Vasc Endovasc Surg 2011; 42: 667-75. ArticlePubMed
- 12. Lee KB, Kang ES, Kim AK, et al. Stem cell therapy in patients with thromboangiitis obliterans: assessment of the long-term clinical outcome and analysis of the prognostic factors. Int J Stem Cells 2011; 4: 88-98. ArticlePubMedPMCPDF
- 13. Yang SS, Kim NR, Park KB, et al. A phase I study of human cord blood-derived mesenchymal stem cell therapy in patients with peripheral arterial occlusive disease. Int J Stem Cells 2013; 6: 37-44. ArticlePubMedPMCPDF
- 14. Cho YJ, Song HS, Bhang S, et al. Therapeutic effects of human adipose stem cell-conditioned medium on stroke. J Neurosci Res 2012; 90: 1794-802. ArticlePubMed
- 15. Lim HC, Lee ST, Chu K, et al. Neuroprotective effect of neural stem cell-conditioned media in in vitro model of Huntington’s disease. Neurosci Lett 2008; 435: 175-80. ArticlePubMed
- 16. Lee EJ, Park SJ, Kang SK, et al. Spherical bullet formation via E-cadherin promotes therapeutic potency of mesenchymal stem cells derived from human umbilical cord blood for myocardial infarction. Mol Ther 2012; 20: 1424-33. ArticlePubMedPMC
- 17. Pereira T, Ivanova G, Caseiro AR, et al. MSCs conditioned media and umbilical cord blood plasma metabolomics and composition. PLoS One 2014; 9: e113769. ArticlePubMedPMC
- 18. Lipke DW, Arcot SS, Gillespie MN, Olson JW. Temporal alterations in specific basement membrane components in lungs from monocrotaline-treated rats. Am J Respir Cell Mol Biol 1993; 9: 418-28. ArticlePubMed
- 19. Bieback K, Kern S, Kluter H, Eichler H. Critical parameters for the isolation of mesenchymal stem cells from umbilical cord blood. Stem Cells 2004; 22: 625-34. ArticlePubMedPDF
- 20. Wang M, Yang Y, Yang D, et al. The immunomodulatory activity of human umbilical cord blood-derived mesenchymal stem cells in vitro. Immunology 2009; 126: 220-32. ArticlePubMedPMC
- 21. Jeong SY, Kim DH, Ha J, et al. Thrombospondin-2 secreted by human umbilical cord blood-derived mesenchymal stem cells promotes chondrogenic differentiation. Stem Cells 2013; 31: 2136-48. ArticlePubMedPDF
- 22. Kim JY, Kim DH, Kim JH, et al. Soluble intracellular adhesion molecule-1 secreted by human umbilical cord blood-derived mesenchymal stem cell reduces amyloid-beta plaques. Cell Death Differ 2012; 19: 680-91. ArticlePubMedPMCPDF
- 23. Jin H, Sanberg PR, Henning RJ. Human umbilical cord blood mononuclear cell-conditioned media inhibits hypoxic-induced apoptosis in human coronary artery endothelial cells and cardiac myocytes by activation of the survival protein Akt. Cell Transplant 2013; 22: 1637-50. ArticlePubMedPDF
- 24. Yang S, Sun HM, Yan JH, et al. Conditioned medium from human amniotic epithelial cells may induce the differentiation of human umbilical cord blood mesenchymal stem cells into dopaminergic neuron-like cells. J Neurosci Res 2013; 91: 978-86. ArticlePubMed
- 25. Yang C, Lei D, Ouyang W, et al. Conditioned media from human adipose tissue-derived mesenchymal stem cells and umbilical cord-derived mesenchymal stem cells efficiently induced the apoptosis and differentiation in human glioma cell lines in vitro. Biomed Res Int 2014; 2014: 109389.ArticlePubMedPMCPDF
- 26. Jiang Y, Jahagirdar BN, Reinhardt RL, et al. Pluripotency of mesenchymal stem cells derived from adult marrow. Nature 2002; 418: 41-9. ArticlePubMedPDF
- 27. Mareschi K, Ferrero I, Rustichelli D, et al. Expansion of mesenchymal stem cells isolated from pediatric and adult donor bone marrow. J Cell Biochem 2006; 97: 744-54. ArticlePubMed
- 28. Secco M, Zucconi E, Vieira NM, et al. Multipotent stem cells from umbilical cord: cord is richer than blood! Stem Cells 2008; 26: 146-50. ArticlePubMedPDF
- 29. Kim ES, Jeon HB, Lim H, et al. Conditioned media from human umbilical cord blood-derived mesenchymal stem cells inhibits melanogenesis by promoting proteasomal degradation of MITF. PLoS One 2015; 10: e0128078. ArticlePubMedPMC
- 30. Kim JY, Kim DH, Kim DS, et al. Galectin-3 secreted by human umbilical cord blood-derived mesenchymal stem cells reduces amyloid-beta42 neurotoxicity in vitro. FEBS Lett 2010; 584: 3601-8. ArticlePubMed
- 31. Flynn A, Barry F, O’Brien T. UC blood-derived mesenchymal stromal cells: an overview. Cytotherapy 2007; 9: 717-26. ArticlePubMed
- 32. Dranoff G. Cytokines in cancer pathogenesis and cancer therapy. Nat Rev Cancer 2004; 4: 11-22. ArticlePubMedPDF
- 33. Chandrasekar B, Melby PC, Sarau HM, et al. Chemokine-cytokine cross-talk. The ELR+ CXC chemokine LIX (CXCL5) amplifies a proinflammatory cytokine response via a phosphatidylinositol 3-kinase-NF-kappa B pathway. J Biol Chem 2003; 278: 4675-86. PubMed
- 34. Brew K, Dinakarpandian D, Nagase H. Tissue inhibitors of metalloproteinases: evolution, structure and function. Biochim Biophys Acta 2000; 1477: 267-83. ArticlePubMed
- 35. Bourboulia D, Stetler-Stevenson WG. Matrix metalloproteinases (MMPs) and tissue inhibitors of metalloproteinases (TIMPs): Positive and negative regulators in tumor cell adhesion. Semin Cancer Biol 2010; 20: 161-8. ArticlePubMedPMC
- 36. Jiang Y, Goldberg ID, Shi YE. Complex roles of tissue inhibitors of metalloproteinases in cancer. Oncogene 2002; 21: 2245-52. ArticlePubMedPDF
- 37. Stetler-Stevenson WG. Tissue inhibitors of metalloproteinases in cell signaling: metalloproteinase-independent biological activities. Sci Signal 2008; 1: re6.ArticlePubMedPMC
- 38. Furie MB, Randolph GJ. Chemokines and tissue injury. Am J Pathol 1995; 146: 1287-301. PubMedPMC
- 39. Kaneider NC, Leger AJ, Kuliopulos A. Therapeutic targeting of molecules involved in leukocyte-endothelial cell interactions. FEBS J 2006; 273: 4416-24. ArticlePubMed
- 40. Kolb M, Margetts PJ, Anthony DC, Pitossi F, Gauldie J. Transient expression of IL-1beta induces acute lung injury and chronic repair leading to pulmonary fibrosis. J Clin Invest 2001; 107: 1529-36. ArticlePubMedPMC
- 41. Johnston CJ, Piedboeuf B, Rubin P, Williams JP, Baggs R, Finkelstein JN. Early and persistent alterations in the expression of interleukin-1 alpha, interleukin-1 beta and tumor necrosis factor alpha mRNA levels in fibrosis-resistant and sensitive mice after thoracic irradiation. Radiat Res 1996; 145: 762-7. PubMed
- 42. Phan SH, Kunkel SL. Lung cytokine production in bleomycin-induced pulmonary fibrosis. Exp Lung Res 1992; 18: 29-43. ArticlePubMed
- 43. Pan LH, Ohtani H, Yamauchi K, Nagura H. Co-expression of TNF alpha and IL-1 beta in human acute pulmonary fibrotic diseases: an immunohistochemical analysis. Pathol Int 1996; 46: 91-9. PubMed
REFERENCES
Figure & Data
References
Citations

- Comparative analysis on the anti-inflammatory/immune effect of mesenchymal stem cell therapy for the treatment of pulmonary arterial hypertension
Seyeon Oh, Albert Y. Jang, Sehyun Chae, Seungbum Choi, Jeongsik Moon, Minsu Kim, Edda Spiekerkoetter, Roham T. Zamanian, Phillip C. Yang, Daehee Hwang, Kyunghee Byun, Wook-Jin Chung
Scientific Reports.2021;[Epub] CrossRef - MSCs Therapy Reverse the Gut Microbiota in Hypoxia-Induced Pulmonary Hypertension Mice
Lingjie Luo, Qinhua Chen, Lei Yang, Zhenxia Zhang, Jihong Xu, Deming Gou
Frontiers in Physiology.2021;[Epub] CrossRef - The promise of mesenchymal stem cell therapy for acute respiratory distress syndrome
Ben Antebi, Arezoo Mohammadipoor, Andriy I. Batchinsky, Leopoldo C. Cancio
Journal of Trauma and Acute Care Surgery.2018; 84(1): 183. CrossRef - Let-7a-transfected mesenchymal stem cells ameliorate monocrotaline-induced pulmonary hypertension by suppressing pulmonary artery smooth muscle cell growth through STAT3-BMPR2 signaling
Gesheng Cheng, Xingye Wang, Yongxin Li, Lu He
Stem Cell Research & Therapy.2017;[Epub] CrossRef - Reduced immunoreactivities of B-type natriuretic peptide in pulmonary arterial hypertension rats after ranolazine treatment
Jae Chul Lee, Kwan Chang Kim, Soo Young Choe, Young Mi Hong
Anatomy & Cell Biology.2016; 49(1): 7. CrossRef - Right sided heart failure and pulmonary hypertension: New insights into disease mechanisms and treatment modalities
Diana Drogalis-Kim, John Jefferies, Ivan Wilmot, Juan Alejos
Progress in Pediatric Cardiology.2016; 43: 71. CrossRef - Notice of Retraction: Therapeutic Effects of Umbilical Cord Blood Derived Mesenchymal Stem Cell-Conditioned Medium on Pulmonary Arterial Hypertension in Rats
Jae Chul Lee, Choong Ik Cha, Dong-Sik Kim, Soo Young Choe
Journal of Pathology and Translational Medicine.2016; 50(4): 325. CrossRef
 PubReader
PubReader ePub Link
ePub Link-
 Cite this Article
Cite this Article
- Cite this Article
-
- Close
- Download Citation
- Close
- Figure





Fig. 1.
Fig. 2.
Fig. 3.
Fig. 4.
Fig. 5.
| Day | Group | Body weight (g) | RV (g) | LV + IVS (g) | RV / (LV + IVS) (%) |
|---|---|---|---|---|---|
| 7 | Control | 318.63 ± 14.78 | 0.132 ± 0.02 | 0.611 ± 0.02 | 0.21 ± 0.01 |
| M | 278.50 ± 32.71 | 0.155 ± 0.03 | 0.543 ± 0.03 | 0.28 ± 0.02 | |
| CM | 280.46 ± 29.82 | 0.164 ± 0.02 | 0.561 ± 0.03 | 0.29 ± 0.02 | |
| 14 | Control | 343.65 ± 24.52 | 0.156 ± 0.02 | 0.731 ± 0.03 | 0.21 ± 0.02 |
| M | 256.71 ± 45.57 |
0.234 ± 0.03 | 0.671 ± 0.02 | 0.34 ± 0.03 |
|
| CM | 271.21 ± 38.82 | 0.224 ± 0.04 | 0.699 ± 0.03 | 0.32 ± 0.02 | |
| 21 | Control | 393.81 ± 24.62 | 0.166 ± 0.03 | 0.782 ± 0.03 | 0.21 ± 0.02 |
| M | 249.67 ± 47.29 |
0.314 ± 0.06 |
0.677 ± 0.05 | 0.46 ± 0.05 |
|
| CM | 271.00 ± 51.55 |
0.284 ± 0.05 | 0.631 ± 0.03 | 0.45 ± 0.03 | |
| 28 | Control | 394.00 ± 41.61 | 0.171 ± 0.02 | 0.801 ± 0.03 | 0.21 ± 0.02 |
| M | 229.71 ± 44.82 |
0.394 ± 0.08 |
0.751 ± 0.06 | 0.52 ± 0.07 |
|
| CM | 319.29 ± 36.62 |
0.261 ± 0.06 |
0.732 ± 0.04 | 0.35 ± 0.04 |
| Day | C group | M group | CM group |
|---|---|---|---|
| 7 | 22.7 ± 0.6 | 24.5 ± 2.1 | 23.2 ± 3.4 |
| 14 | 22.0 ± 1.1 | 37.8 ± 3.2 |
30.9 ± 4.6 |
| 21 | 23.3 ± 0.9 | 50.2 ± 4.7 |
39.2 ± 5.2 |
| 28 | 22.9 ± 2.1 | 58.0 ± 6.4 |
37.8 ± 4.1 |
Values are presented as mean ± standard deviation. hUCB-MSCs-CM, conditioned medium from human umbilical-cord blood derived mesenchymal cells; PAH, pulmonary artery hypertension; RV, right ventricle; LV, left ventricle; IVS, interventricular septum; M, monocrotaline; CM, hUCB-MSCs-CM. p < .05 compared with the C group; p < .05 compared with the M group.
Values are presented as mean ± standard deviation. hUCB-MSCs-CM, conditioned medium from human umbilical-cord blood derived mesenchymal cells; PAH, pulmonary artery hypertension; C, control; M, monocrotaline; CM, hUCB-MSCs-CM. p < .05 compared with the C group; p < .05 compared with the M group.

 E-submission
E-submission