Articles
- Page Path
- HOME > J Pathol Transl Med > Volume 50(4); 2016 > Article
-
Brief Case Report
Gastric Mixed Adenoneuroendocrine Carcinoma with Squamous Differentiation: A Case Report - Han-Ik Bae, Chaeyoon Lee1, Young-Min Jo, Ohkyung Kwon1, Wansik Yu1, Mee-Seon Kim2, An Na Seo
-
Journal of Pathology and Translational Medicine 2016;50(4):318-321.
DOI: https://doi.org/10.4132/jptm.2015.10.17
Published online: January 11, 2016
Department of Pathology, Kyungpook National University Hospital, Kyungpook National University Medical Center, Kyungpook National University School of Medicine, Daegu, Korea
1Department of Surgery, Gastric Cancer Center, Kyungpook National University Medical Center, Kyungpook National University School of Medicine, Daegu, Korea
2Department of Pathology, Samsung Changwon Hospital, Sungkyunkwan University School of Medicine, Changwon, Korea
- Corresponding Author An Na Seo, MD, PhD Department of Pathology, Kyungpook National University Hospital, Kyungpook National University Medical Center, Kyungpook National University School of Medicine, 807 Hoguk-ro, Buk-gu, Daegu 41404, Korea Tel: +82-53-200-3403, Fax: +82-53-200-3399, E-mail: san_0729@naver.com
© 2016 The Korean Society of Pathologists/The Korean Society for Cytopathology
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/3.0/) which permits unrestricted noncommercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
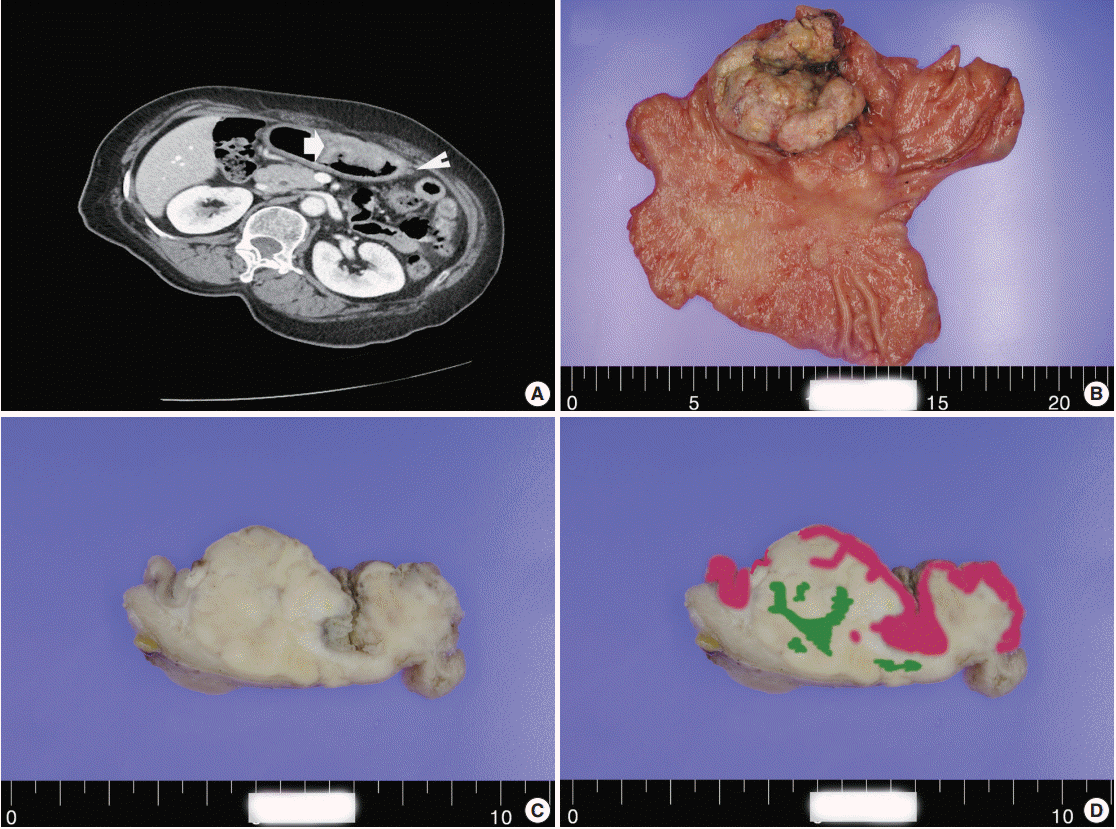
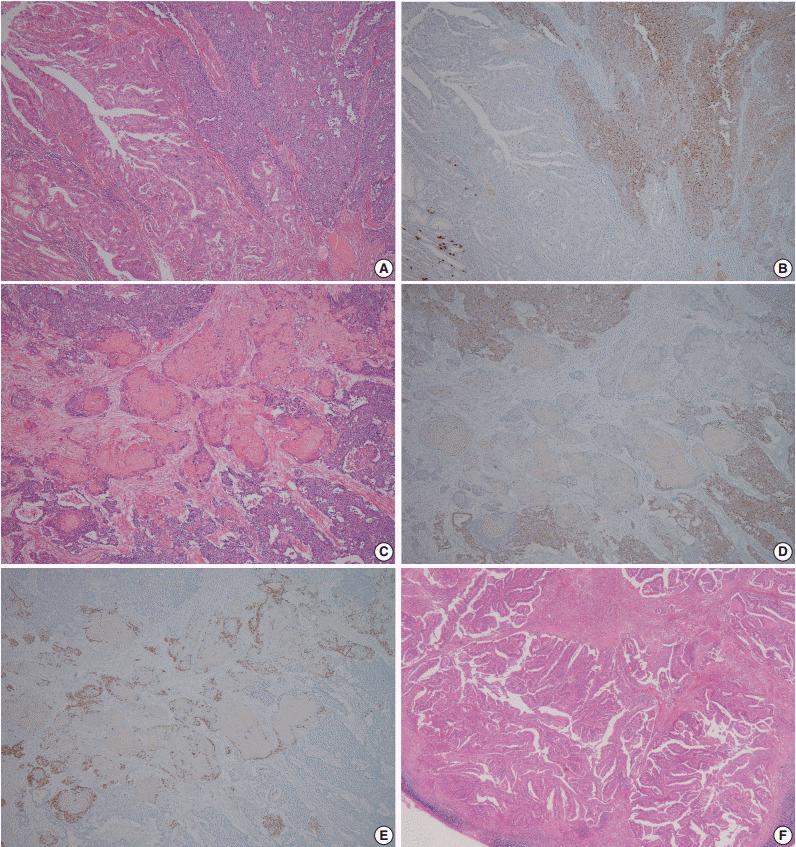
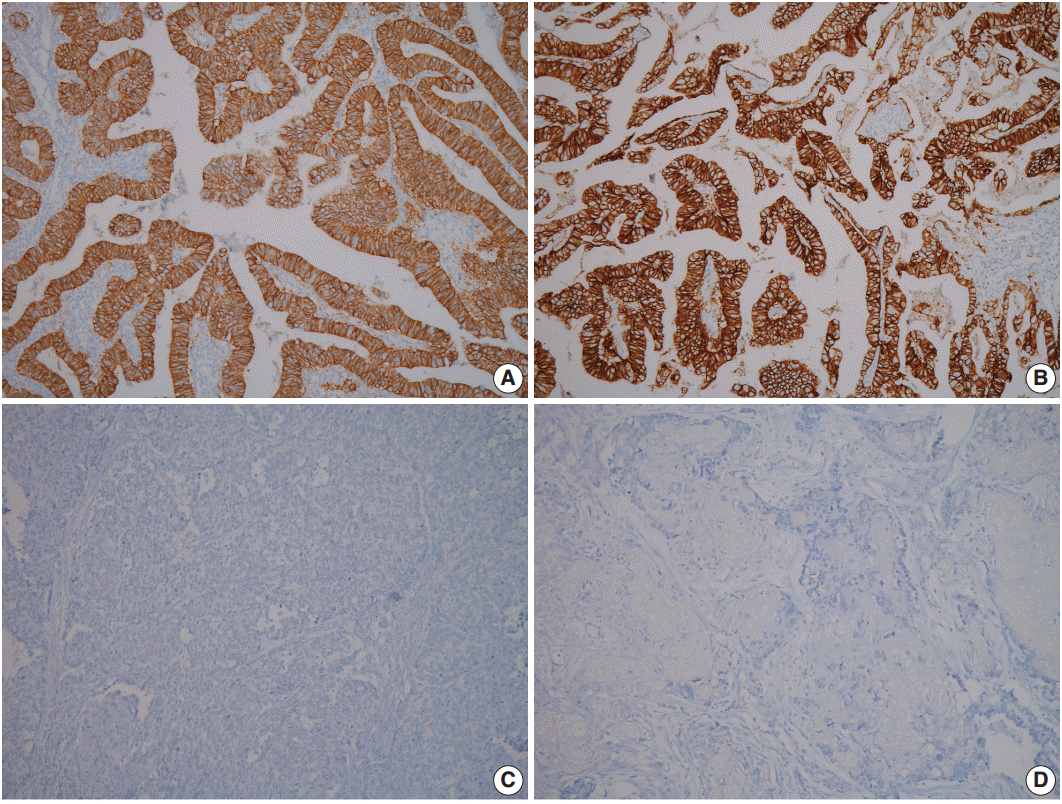
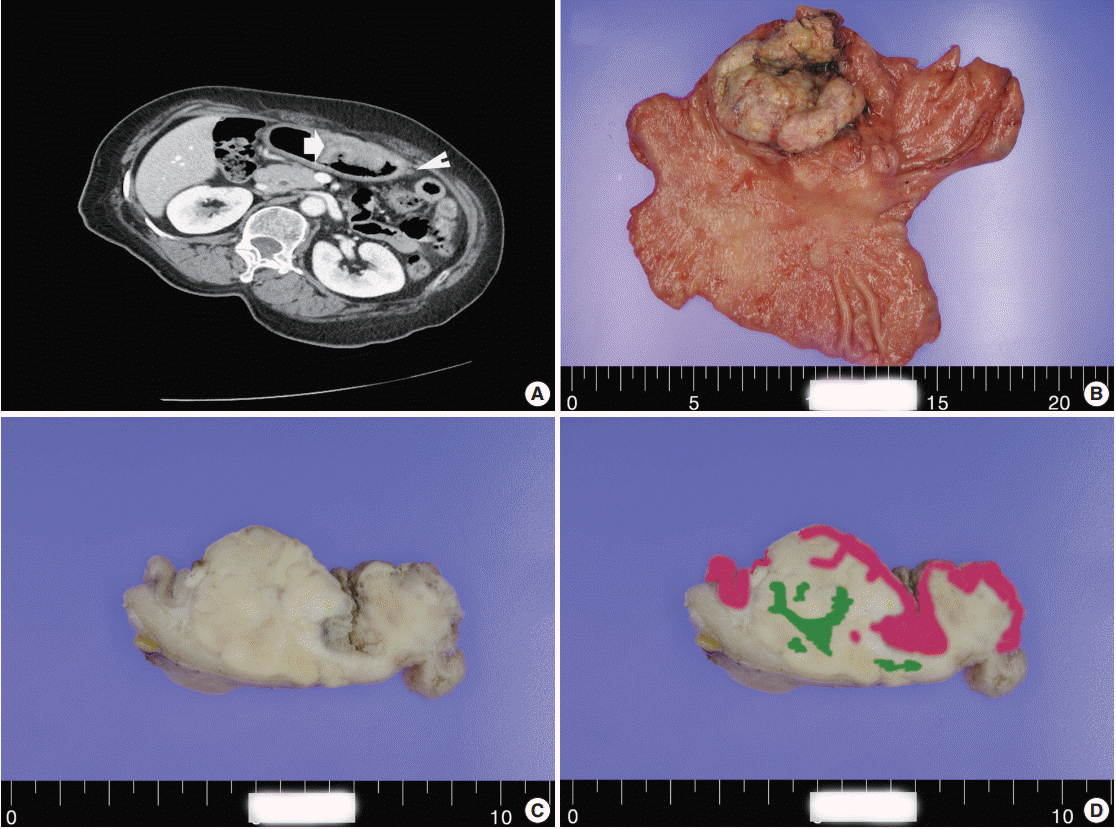
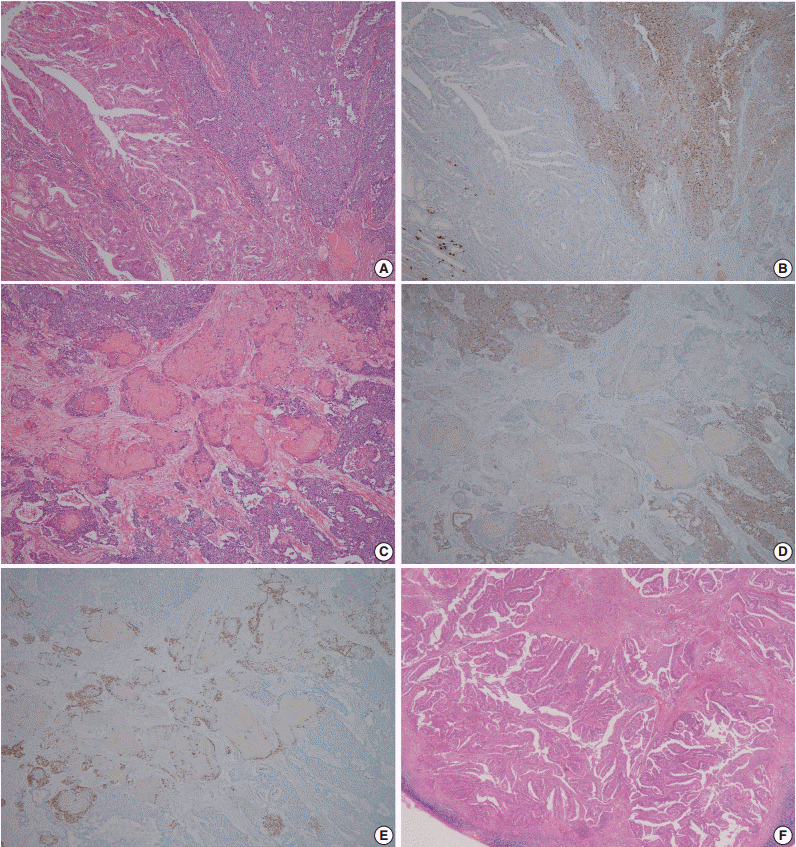
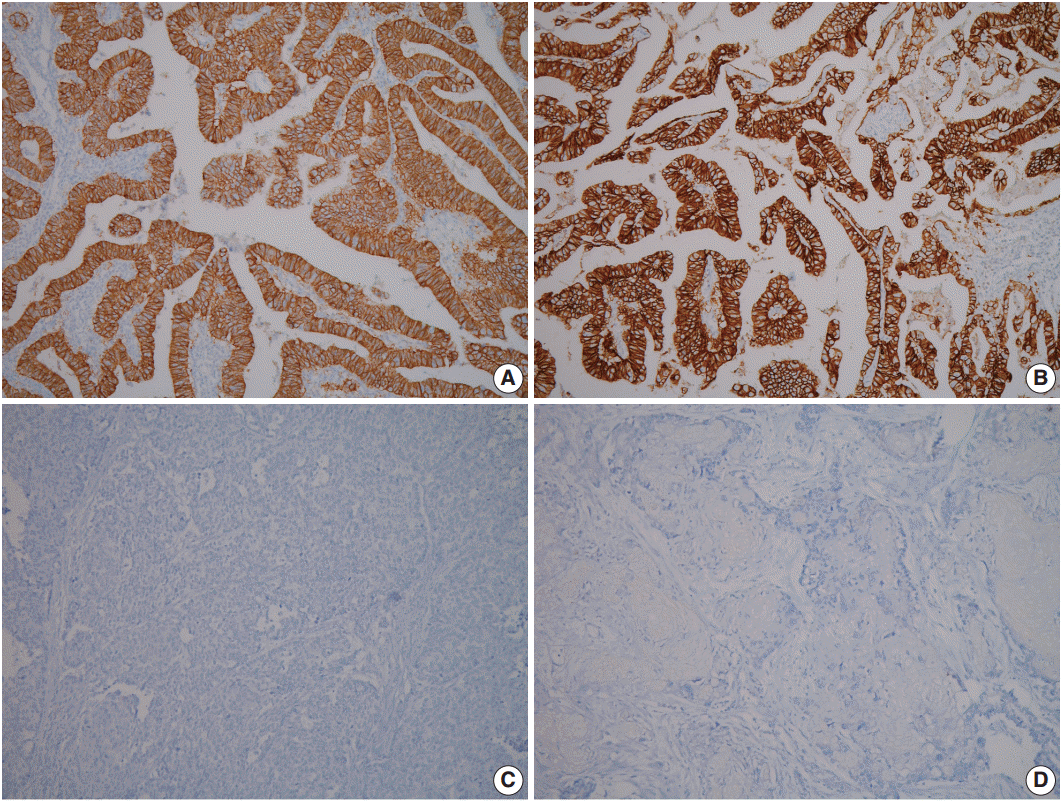
- A 60-year-old woman presented with generalized weakness, anorexia, weight loss, and fasting epigastric pain over a 5-month period. The patient had suffered from allergic urticaria for several years, but this had been treated and subsequently controlled with standard medication. She denied any previous alcohol or herbal medication intake and had no significant family history. Esophagogastroduodenoscopy revealed an 8.5 × 7.0-cm-sized ulcerofungating mass in the anterior wall of the lower body. Biopsy of the mass was diagnosed as moderately differentiated adenocarcinoma. Preliminary laboratory test results were within normal limits, whereas a tumor marker level test revealed elevated carbohydrate antigen 19-9 (98.8 U/mL). Computed tomography demonstrated a large hypodense ulcerofungating mass in the gastric anterior wall along with multiple perigastric lymphadenopathies (Fig. 1A). The patient underwent D2 subtotal gastrectomy and early postoperative intraperitoneal chemotherapy. Gross examination of the resected stomach showed a smooth serosal surface and a large ulcerofungating mass measuring 8.5 × 7.0 cm, consistent with Borrmann type 2, in the lower anterior wall (Fig. 1B). On sectioning, the tumor had a heterogeneous appearance with a mixture of white and yellow lesions with infiltrative boundaries, invading to the serosal layer (Fig. 1C). Microscopically, the tumor comprised 55% large cell NEC, 35% moderately differentiated adenocarcinoma, and 10% well differentiated SqCC. The adenocarcinoma component was in the upper portion of the tumor, whereas the NEC component presented in the deep portion, with a small proportion in the upper portion, up to the mucosal layer (Fig. 1D). In particular, the collision between the adenocarcinoma and NEC components was found in the ulcerated tumor area (Fig. 2A). Additionally, transition from NEC to SqCC was found in some areas (Fig. 2C). As expected, immunohistochemical staining for synaptophysin and chromogranin A were positive in the NEC component but negative in the adenocarcinoma and SqCC components (Fig. 2B, D). The SqCC component but not the NEC or adenocarcinoma component revealed strong nuclear expression for p63 (Fig. 2E). The tumor mitotic count was 95 mitoses per 10 high-power fields, and the Ki-67 labeling index was 96%. The tumor penetrated the serosa, and 14 of the 48 regional lymph nodes showed metastasis, which, interestingly, was composed of only the adenocarcinoma component (Fig. 2F), but distant metastasis was not detected. Interestingly, for c-erbB2, the NEC component revealed negativity, whereas the adenocarcinoma component showed complete membranous staining of strong intensity in primary gastric cancer and in its corresponding metastatic regional lymph nodes (Fig. 3). During seven cycles of adjuvant chemotherapy, the patient experienced sudden abdominal pain due to mechanical obstruction by an adhesion at the operation site. Since then, she has stopped receiving chemotherapy and is currently receiving supportive care. The patient has been monitored for 18 months post-surgery and remains alive without evidence of disease recurrence.
- The study was performed in accordance with the guidelines of the Institutional Review Board of Kyungpook National University Medical Center and was exempt from written informed consent. The information of this case was retrospectively obtained from medical records in the database of Kyungpook National University Medical Center.
CASE REPORT
- Gastric MANECs are mixed exocrine and endocrine carcinomas, with one component exceeding at least 30% of the tumor lesion [1,2]. The exocrine component is characterized by adenocarcinoma with various differentiated grades, and the neuroendocrine component is usually represented by NEC [1]. To our knowledge, there have been only four reported cases of gastric MANEC containing a SqCC component [3-5]. Pericleous et al. [5] reported a case of MANEC with a SqCC component where only the NEC component metastasized to the liver. In addition, Shibuya et al. [4] reported a small cell undifferentiated carcinoma with adenocarcinoma and SqCC components, and hepatic nodules showed an atypical carcinoid component. In contrast, in the current case, only the adenocarcinoma component metastasized to the regional lymph nodes, with no distant metastasis detected.
- The pathogenesis of gastric NEC is unclear. Based on the presence of NEC with other glandular or squamous cell components, the proposed origin of gastric MANEC is pluripotent stem cells that have the potential to differentiate into other cell types producing mucin or keratin [6,7]. Owing to the heterogeneous composition of MANEC, treatment approaches should focus on the most aggressive tumor components. However, the prognosis and behavior of gastric MANEC with trilineage histologic differentiation are unknown because of its rarity. Therefore, collaborative multicenter studies including such patients are required to determine optimal treatment regimens, prevent relapse, and improve prognosis.
- Herein, we report a gastric MANEC with trilineage histologic differentiation including large cell NEC, adenocarcinoma, and SqCC, of which only the adenocarcinoma component metastasized to the regional lymph nodes, without distant metastasis.
DISCUSSION
- 1. Bosman FT, Carneiro F, Hruban RH, Theise ND. WHO classification of tumours of the digestive system. 4th ed. Lyon: IARC Press, 2010.
- 2. Volante M, Rindi G, Papotti M. The grey zone between pure (neuro)endocrine and non-(neuro)endocrine tumours: a comment on concepts and classification of mixed exocrine-endocrine neoplasms. Virchows Arch 2006; 449: 499-506. ArticlePubMedPDF
- 3. Haratake J, Horie A, Inoshita S. Gastric small cell carcinoma with squamous and neuroendocrine differentiation. Pathology 1992; 24: 116-20. ArticlePubMed
- 4. Shibuya H, Azumi N, Abe F. Gastric small-cell undifferentiated carcinoma with adeno and squamous cell carcinoma components. Acta Pathol Jpn 1985; 35: 473-80. ArticlePubMed
- 5. Pericleous M, Toumpanakis C, Lumgair H, et al. Gastric mixed adenoneuroendocrine carcinoma with a trilineage cell differentiation: case report and review of the literature. Case Rep Oncol 2012; 5: 313-9. ArticlePubMedPMC
- 6. Bartley AN, Rashid A, Fournier KF, Abraham SC. Neuroendocrine and mucinous differentiation in signet ring cell carcinoma of the stomach: evidence for a common cell of origin in composite tumors. Hum Pathol 2011; 42: 1420-9. ArticlePubMed
- 7. Kim KM, Kim MJ, Cho BK, Choi SW, Rhyu MG. Genetic evidence for the multi-step progression of mixed glandular-neuroendocrine gastric carcinomas. Virchows Arch 2002; 440: 85-93. ArticlePubMedPDF
REFERENCES
Figure & Data
References
Citations

- Comparison of Metastatic Patterns Among Neuroendocrine Tumors, Neuroendocrine Carcinomas, and Nonneuroendocrine Carcinomas of Various Primary Organs
Hyung Kyu Park, Ghee Young Kwon
Journal of Korean Medical Science.2023;[Epub] CrossRef - Mixed large cell neuroendocrine carcinoma and squamous cell carcinoma of the colon: detailed molecular characterisation of two cases indicates a distinct colorectal cancer entity
Christine Woischke, Peter Jung, Andreas Jung, Jörg Kumbrink, Sibylle Eisenlohr, Christoph Josef Auernhammer, Michael Vieth, Thomas Kirchner, Jens Neumann
The Journal of Pathology: Clinical Research.2021; 7(1): 75. CrossRef - Multiregion Comprehensive Genomic Profiling of a Gastric Mixed Neuroendocrine-Nonneuroendocrine Neoplasm with Trilineage Differentiation
Faheem Farooq, Kevin Zarrabi, Keith Sweeney, Joseph Kim, Jela Bandovic, Chiraag Patel, Minsig Choi
Journal of Gastric Cancer.2018; 18(2): 200. CrossRef
 PubReader
PubReader ePub Link
ePub Link-
 Cite this Article
Cite this Article
- Cite this Article
-
- Close
- Download Citation
- Close
- Figure




 E-submission
E-submission