Articles
- Page Path
- HOME > J Pathol Transl Med > Volume 47(4); 2013 > Article
-
Original Article
Bronchial Schwannomas: Clinicopathologic Analysis of 7 Cases - Yoon Yang Jung,, Min Eui Hong, Joungho Han, Tae Sung Kim1, Jhingook Kim2, Young-Mog Shim2, Hojoong Kim3
-
Korean Journal of Pathology 2013;47(4):326-331.
DOI: https://doi.org/10.4132/KoreanJPathol.2013.47.4.326
Published online: August 26, 2013
Department of Pathology, Samsung Medical Center, Sungkyunkwan University School of Medicine, Seoul, Korea.
1Department of Radiology, Samsung Medical Center, Sungkyunkwan University School of Medicine, Seoul, Korea.
2Department of Thoracic Surgery, Samsung Medical Center, Sungkyunkwan University School of Medicine, Seoul, Korea.
3Department of Internal Medicine, Samsung Medical Center, Sungkyunkwan University School of Medicine, Seoul, Korea.
- Corresponding Author: Joungho Han, M.D. Department of Pathology, Samsung Medical Center, Sungkyunkwan University School of Medicine, 81 Irwon-ro, Gangnam-gu, Seoul 135-710, Korea. Tel: +82-2-3410-2800, Fax: +82-2-3410-0025, 'hanjho@skku.edu'
- *Present address of Yoon Yang Jung. Department of Pathology, Chung-Ang University Hospital, Seoul, Korea
© 2013 The Korean Society of Pathologists/The Korean Society for Cytopathology
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/3.0/) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
Abstract
-
Background
- It has long been recognized that bronchial schwannomas are extremely rare. As such, diagnosing tumors in this extraordinary location can sometimes be problematic.
-
Methods
- We reviewed seven cases of bronchoscopically or surgically resected endobronchial schwannomas and evaluated their clinical and pathologic features.
-
Results
- The present study included five female and two male patients, with ages ranging from 16 to 81 years (mean age, 44.9 years). The clinical presentation varied according to tumor size and location. Patients with more centrally (trachea or main bronchus) located tumors experienced respiratory symptoms (80%) more often than patients with more peripherally (lobar or segmental bronchus) located tumors (0%). Histologically, the tumors were composed of spindle cells that stained with S100 protein. Some of the tumors showed typical Antoni A areas with Verocay body formation. Five of six patients (83.3%) underwent complete tumor removal by rigid bronchoscopy.
-
Conclusions
- Pathologists should consider endobronchial schwannoma in the differential diagnosis of a spindle cell tumor involving the bronchus. Additionally, our results showed that rigid bronchoscopy is an effective tool for tumor removal in endobronchial schwannoma patients.
- Patients
- During a 19-year period (1995-2013), a total of seven patients with endobronchial schwannomas were bronchoscopically or surgically treated at Samsung Medical Center in Seoul, Korea. Each patient's clinical information, including age at the time of diagnosis, sex, clinical presentation, notable past history, and radiologic and bronchoscopic findings, were obtained from our electronic medical record database. This study was approved by the Institutional Review Board of Samsung Medical Center (SMC 2013-04-095).
- Tumor classification according to its location
- We classified tumors into central or peripheral type according to tumor location. Our definition differed from the classification of Kasahara et al.,7 which classified tumors located in the trachea or in the proximal bronchus and that are visible by bronchofiberscopy as the central type. In the current study, we simply considered the tumor location regardless of bronchoscopic accessibility. The tumor was classified as being centrally located when the tumor was located in the trachea or main bronchus. When the tumor was located in the lobar bronchus or segmental bronchus, we considered the tumor to be peripherally located.
MATERIALS AND METHODS
- A summary of the clinical, radiologic, bronchoscopic features, and treatment details is presented in Table 1.
- Clinical features
- There were five female and two male patients, showing female predominance (57.1%), and the patients' ages ranged from 16 to 81 with a mean age of 44.9 years. Each patient's history and clinical presentation is described below. Three cases (42.8%) involved the main bronchi, one case (14.3%) involved both the carina and main bronchus, one case (14.3%) involved the carina, one case (14.3%) involved the lobar bronchus, and one case (14.3%) involved the segmental bronchus. Clinical presentations included cough, dyspnea, hemoptysis, pleuritic chest pain, and postobstructive pneumonia. One patient was asymptomatic, and the tumor was detected during a routine chest radiographic examination. Among five cases with centrally located schwannomas (carina and main bronchus), four patients (80%) experienced respiratory symptoms such as dyspnea. The patient with carinal involvement also had hemoptysis. However, none of the patients with peripherally located tumors (lobar and segmental bronchus) experienced this symptom. The patient with lobar bronchus involvement had pleuritic chest pain. The patient with segmental bronchus involvement of the tumor was asymptomatic. The duration of symptoms between onset and presentation ranged from one month to 19 months (average, 7 months), with the exception of the asymptomatic patient.
- Radiologic features
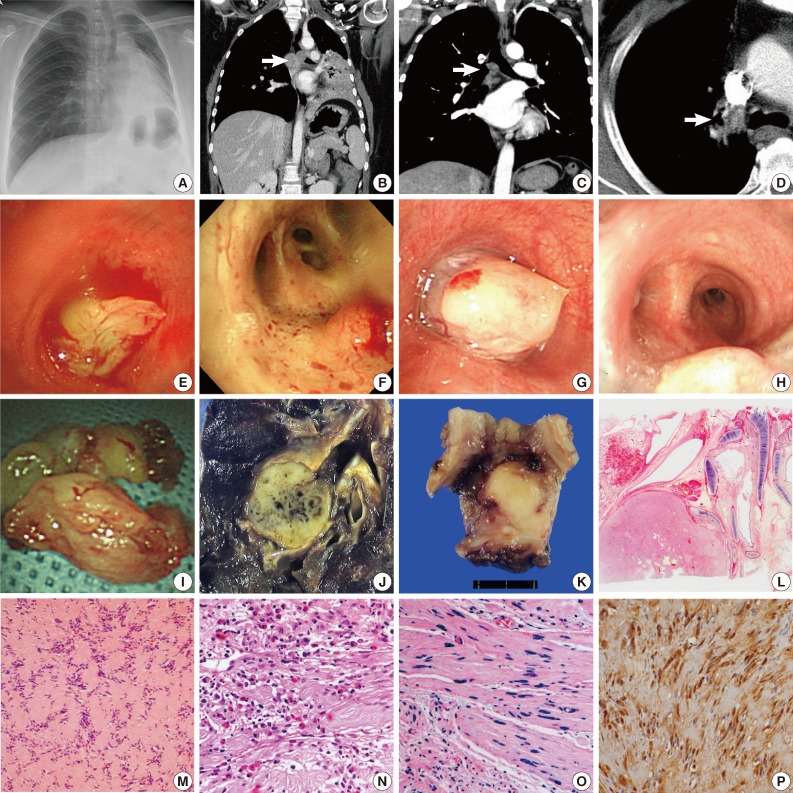
- The tumor size varied from 1.5 to 4 cm (mean size, 2.7 cm). A variety of radiologic impressions were encountered in these cases. These included malignant tumors, such as squamous cell carcinoma, carcinoid tumor, adenoid cystic carcinoma, mucoepidermoid carcinoma, and metastatic breast cancer, as well as benign tumors such as leiomyoma, cylindroma, inflammatory pseudotumor, and schwannoma. Two patients (cases nos. 3 and 5) had pneumonia (Fig. 1A, B). In case no. 2, the tumor showed lobulation with extraluminal extension (Fig. 1C), and the radiologist had a suspicion of malignant tumor. In case no. 4, the patient had a past medical history of breast cancer, and an endobronchial nodule was revealed on computed tomographic (CT) scan (Fig. 1D). Both the clinician and the radiologist suspected metastatic breast cancer. An impression of schwannoma was noted in only one case. In case no. 3, the radiologist placed the impression of schwannoma on the differential diagnosis list.
- Intervention
- Bronchoscopy was performed in every case. Even in the patient with the tumor located in the segmental bronchus (case no. 6), tumors were observed by bronchoscopy. The tumors were described as lobulated (Fig. 1E), well-circumscribed (Fig. 1F), round (Fig. 1G), or raised/smooth surfaced (Fig. 1H) lesions. Visible vessels were also noted in one case. With the exception of one case, all patients had the initial treatment of rigid bronchoscopic tumor removal (Fig. 1I). Only the patient with segmental bronchus tumor involvement (case no. 6) had surgical resection as the initial treatment (Fig. 1J). Among the six patients with initial bronchoscopic resection, five patients had complete tumor removal without recurrence. One patient (case no. 2) experienced tumor recurrence. The patient underwent surgical resection for the recurred tumor (Fig. 1K). None of the patients died of bronchial schwannoma.
- Histological features
- Histologically, the tumors were visualized as relatively well-circumscribed masses with or without adjacent bronchial cartilage (Fig. 1L). All cases showed similar microscopic characteristics of schwannoma: cellular areas (Antoni type A), more loosely structured areas (Antoni type B), and formation of Verocay bodies (Fig. 1M). Cytologically, tumor cells were bland-looking spindle cells with indistinct cell borders and club-shaped nuclei. Hemorrhage, collagen deposition, and eosinophilic infiltration (Fig. 1N) were also noted in some cases. Two cases (cases nos. 4 and 5) exhibited ancient change (Fig. 1O). On light microscopy, differential diagnosis included leiomyoma and inflammatory myoblastic tumor. Every case demonstrated positivity for S100 protein immunohistochemical staining (Fig. 1P), which helped confirm the diagnosis of endobronchial schwannoma.
RESULTS
- Since Verocay and Antoni described the histological aspects of schwannoma in the early 1900s, pathologists have been well aware of the histopathologic features of this tumor. A schwannoma is a benign peripheral nerve sheath neoplasm that is composed almost entirely of Schwann cells. Microscopically, the tumor consists of distinctive architectural patterns: cellular "Antoni type A" areas and loosely structured "Antoni type B" areas. Additionally, these tumors have another characteristic feature: "Verocay bodies." A typical Verocay body consists of a stacked arrangement of horizontal rows of palisaded nuclei that are separated by areas of acellular pink zones composed of cytoplasmic processes of the Schwann cells.8
- While schwannoma involving various parts of the body is a relatively frequently encountered histologic diagnosis for pathologists, it rarely presents as an endobronchial lesion. Therefore, a clinician may not suspect a schwannoma in a patient with an endobronchial lesion. Benign endobronchial tumors themselves are uncommon, and of these, endobronchial schwannoma is a rare entity, constituting approximately 2% of benign tracheobronchial tumors.9 Although rare, schwannomas can occur in any area of the tracheobronchial tree, with intraluminal or extraluminal extensions.7,10,11
- The clinical presentation of endobronchial schwannoma varies and depends on the tumor location, size, and degree of bronchial obstruction. Symptoms include dry or productive cough, fever, hemoptysis, dyspnea, and postobstructive pneumonia, and any one of these can be the first sign of bronchial schwannoma.12
- As described above, the clinical symptoms of our patients included cough, fever, hemoptysis, dyspnea, and pleuritic chest pain. Four out of five patients (80%) with more central tumor locations (carina and main bronchus) experienced dyspnea, while neither of the two patients with peripheral locations (lobar and segmental bronchus) had this symptom. Notably, the patient with the most peripherally located tumor (anterior segmental bronchus of the left lower lobe anterobasal segment) was asymptomatic, and the tumor was only discovered on a routine chest radiograph. The duration of symptoms ranged from one to 19 months, with an average of seven months, excluding the asymptomatic patient.
- Since symptoms are nonspecific, a diagnosis of schwannoma cannot be made on the basis of clinical presentation. Pre-operatively, several other differential diagnoses for the endobronchial lesion may be considered, including malignant lesions. In our cases, the clinical and radiologic impressions included malignant lesions such as squamous cell carcinoma, adenoid cystic carcinoma, and small cell carcinoma, as well as benign lesions including inflammatory pseudotumor, leiomyoma, and neurogenic tumor. In addition, in three of our cases, radiologists included the possibility of malignant tumor on CT scan. As clinical and radiologic impressions vary, and CT imaging cannot be used to differentiate the nature of the tumor, biopsy is required in most cases.
- After a biopsy specimen is obtained, the differential diagnoses can be narrowed into fewer entities by pathology. However, assigning a definite diagnosis on the basis of light microscopy alone is difficult. Initially, tumors that consist of spindled cells, such as leiomyoma, inflammatory myofibroblastic tumor, and meningioma, might be in the differential diagnoses list. One of our patients had a previous biopsy diagnosis of inflammatory pseudotumor at an outside hospital. Although rare, a variety of spindle cell tumors can occur in the tracheobronchial tree, and all of those can be in the differential diagnosis list.
- Leiomyoma constitutes around 2% of benign lung tumors, and 45% of pulmonary leiomyomas are endobronchial.13 Patients with endobronchial leiomyoma usually experience respiratory symptoms due to partial or complete airway obstruction.14 On microscopy, leiomyoma are composed of bundles and whorls spindle cells with abundant elongated eosinophilic cytoplasm. The tumors show positivity for desmin and smooth muscle actin, but not for S100 protein.
- Inflammatory myofibroblastic tumor is a rare tumor that is usually located in the lung, but can sometimes involve the upper respiratory tract.15 Histologically, the tumor consists of uniform, bland, fibroblast-like spindle cells arranged in fascicles or in a storiform pattern. Characteristically, the tumor is accompanied by an inflammatory infiltrate of plasma cells.16
- Meningioma rarely occurs in the pulmonary region, with approximately thirty reports of primary pulmonary meningiomas in the literature.17 These tumors can contain fascicularly arranged tumor cells resembling fibroblasts.17 Positivity for epithelial membrane antigen is not pathognomic, but it is expressed in meningiomas and can be helpful in differential diagnosis.18
- In the diagnostic process, the identification of the characteristics of schwannoma, including typical Antoni A formation and Verocay bodies in hematoxylin and eosin stains and S100 positivity aids in confirming the correct diagnosis of schwannoma.19 Although schwannomas are histologically benign, the clinical course depends on the tumor location, size, and degree of bronchial obstruction.12 Kasahara et al.7 reported a case of schwannoma leading to patient death with a complication of pneumonia. Considering that schwannoma can lead to other complications, proper management based on each patient's condition is required.
- Until recently, the standard treatment for endobronchial schwannoma has been surgical resection. However, bronchoscopic treatment has been recently utilized for benign tracheobronchial tumors, and has been shown to be a safe and effective tool.4,5 However, to date, there is only limited information in the literature on bronchoscopic removal of endobronchial schwannoma.6
- In our cases, aside from one patient with a segmental bronchial tumor, six patients had bronchoscopic tumor removal as the initial treatment. Five of six patients (83.3%) had successful removal of the tumor without recurrence, revealing a high efficacy of bronchoscopic removal of endobronchial schwannoma. Unfortunately, this procedure cannot always achieve complete resection. In case no. 2, the patient underwent rigid bronchoscopic tumor removal at an outside hospital, but residual tumor was observed a month after the intervention, and she required surgery for complete resection of the tumor. Our study revealed that rigid bronchoscopy is a very effective tool for the management of schwannoma, but also showed that it cannot promise complete resection. Therefore, before performing a bronchoscopic intervention, clinicians and surgeons need a thorough evaluation of resectability considering tumor size and location. In asymptomatic patients, watchful waiting can also be an option.
- In conclusion, although endobronchial schwannoma is rare, awareness of the possibility of schwannoma involving the bronchus might be helpful in making a correct diagnosis. After diagnosing the tumor, proper management based on each patient's clinical setting is required. In addition, our study revealed successful results following bronchoscopic removal of endobronchial schwannomas.
DISCUSSION
- 1. Zachariades N, Skoura C, Papageorgiou G, Chrissomali E. Giant ancient neurilemmoma of the cervical region: report of case. J Oral Maxillofac Surg 2001; 59: 668–672. PMID: 11381392. ArticlePubMed
- 2. Murphy J, Giunta JL. Atypical central neurilemmoma of the mandible. Oral Surg Oral Med Oral Pathol 1985; 59: 275–278. PMID: 3856819. ArticlePubMed
- 3. Villanueva J, Gigoux C, Solé F. Central neurilemmoma of maxilla: a case report. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 1995; 79: 41–43. PMID: 7614159. ArticlePubMed
- 4. Suzuki H, Sekine Y, Motohashi S, et al. Endobronchial neurogenic tumors treated by transbronchial electrical snaring and Nd-YAG laser abrasion: report of three cases. Surg Today 2005; 35: 243–246. PMID: 15772797. ArticlePubMed
- 5. Kwon YS, Kim H, Koh WJ, et al. Clinical characteristics and efficacy of bronchoscopic intervention for tracheobronchial leiomyoma. Respirology 2008; 13: 908–912. PMID: 18811890. ArticlePubMed
- 6. Lee BR, Choi YD, Kim YI, Lim SC, Kwon YS. Endobronchial schwannoma treated by rigid bronchoscopy with argon plasma coagulation. Tuberc Respir Dis 2012; 73: 174–177. Article
- 7. Kasahara K, Fukuoka K, Konishi M, et al. Two cases of endobronchial neurilemmoma and review of the literature in Japan. Intern Med 2003; 42: 1215–1218. PMID: 14714962. ArticlePubMed
- 8. Joshi R. Learning from eponyms: Jose Verocay and Verocay bodies, Antoni A and B areas, Nils Antoni and schwannomas. Indian Dermatol Online J 2012; 3: 215–219. PMID: 23189261. ArticlePubMedPMC
- 9. Shah H, Garbe L, Nussbaum E, Dumon JF, Chiodera PL, Cavaliere S. Benign tumors of the tracheobronchial tree: endoscopic characteristics and role of laser resection. Chest 1995; 107: 1744–1751. PMID: 7781378. ArticlePubMed
- 10. Stack PS, Steckler RM. Tracheal neurilemmoma: case report and review of the literature. Head Neck 1990; 12: 436–439. PMID: 2211106. ArticlePubMed
- 11. Shirakusa T, Takada S, Yamazaki S, et al. Intrabronchial neurilemmoma: review of cases in Japan. Thorac Cardiovasc Surg 1989; 37: 388–390. PMID: 2694443. ArticlePubMed
- 12. Oxner CR, Shinners MJ, Godin DA, Lyn IT, Brent RB, McFadden PM. Obstructing tracheobronchial schwannoma. J La State Med Soc 2005; 157: 159–161. PMID: 16173316. PubMed
- 13. Wilson RW, Kirejczyk W. Pathological and radiological correlation of endobronchial neoplasms: Part I, Benign tumors. Ann Diagn Pathol 1997; 1: 31–46. PMID: 9869824. ArticlePubMed
- 14. White SH, Ibrahim NB, Forrester-Wood CP, Jeyasingham K. Leiomyomas of the lower respiratory tract. Thorax 1985; 40: 306–311. PMID: 4023981. ArticlePubMedPMC
- 15. Rodrigues M, Taylor RJ, Sun CC, Wolf JS. Inflammatory myofibroblastic tumor of the larynx in a 2-year-old male. ORL J Otorhinolaryngol Relat Spec 2005; 67: 101–105. PMID: 15821353. ArticlePubMed
- 16. Coffin CM, Hornick JL, Fletcher CD. Inflammatory myofibroblastic tumor: comparison of clinicopathologic, histologic, and immunohistochemical features including ALK expression in atypical and aggressive cases. Am J Surg Pathol 2007; 31: 509–520. PMID: 17414097. ArticlePubMed
- 17. van der Meij JJ, Boomars KA, van den Bosch JM, van Boven WJ, de Bruin PC, Seldenrijk CA. Primary pulmonary malignant meningioma. Ann Thorac Surg 2005; 80: 1523–1525. PMID: 16181912. ArticlePubMed
- 18. Moran CA, Hochholzer L, Rush W, Koss MN. Primary intrapulmonary meningiomas: a clinicopathologic and immunohistochemical study of ten cases. Cancer 1996; 78: 2328–2333. PMID: 8941002. ArticlePubMed
- 19. Bosch X, Ramirez J, Font J, et al. Primary intrapulmonary benign schwannoma: a case with ultrastructural and immunohistochemical confirmation. Eur Respir J 1990; 3: 234–237. PMID: 2178968. ArticlePubMed
References
Figure & Data
References
Citations

- Video-assisted thoracic surgery for an endobronchial ancient schwannoma obstructing the left main bronchus
Jiyeon Kang, Yeon Soo Kim, Ji-Ye Kim
Journal of Surgical Case Reports.2024;[Epub] CrossRef - Two cases of large tracheobronchial schwannomas completely resected by rigid bronchoscopy with multiple instruments
Changhwan Kim, Hae‐Seong Nam, Yousang Ko
Respirology Case Reports.2023;[Epub] CrossRef - Tracheobronchial schwannoma: a case report and literature review
Guo Lina, Hou Pengguo, Xiao Zhihua, Wang Jianxin, Bai Baoqin, Zhang Mingyue, Sun Junping
Journal of International Medical Research.2023; 51(1): 030006052211498. CrossRef - Malignant and Benign Tracheobronchial Neoplasms: Comprehensive Review with Radiologic, Bronchoscopic, and Pathologic Correlation
Francis Girvin, Alexander Phan, Sharon Steinberger, Eugene Shostak, Jamie Bessich, Fang Zhou, Alain Borczuk, Geraldine Brusca-Augello, Margaret Goldberg, Joanna Escalon
RadioGraphics.2023;[Epub] CrossRef - Clinicopathological Characteristics and Pathogenesis of Granular Cell Tumours of the Airways
Jesús Machuca-Aguado, Fernando Cózar-Bernal, Enrique Rodríguez-Zarco, Juan José Ríos-Martin, Miguel Ángel Idoate Gastearena
Journal of Bronchology & Interventional Pulmonology.2023; 30(4): 390. CrossRef - Treatment of primary tracheal schwannoma with endoscopic resection: A case report
Yong-Shuai Shen, Xiang-Dong Tian, Yi Pan, Hua Li
World Journal of Clinical Cases.2022; 10(28): 10279. CrossRef - Primary bronchial schwannoma: A case report
Yosuke Aoyama, Atsushi Miyamoto, Takeshi Fujii, Sakashi Fujimori, Meiyo Tamaoka, Daiya Takai
Medicine.2022; 101(40): e31062. CrossRef - Endobronchial schwannoma in adult: A case report
Touil Imen, Boudaya Mohamed Sadok, Aloui Raoudha, Souhir Ksissa, Brahem Yosra, Ben Attig Yosr, Ksontini Meriem, Bouchareb Soumaya, Keskes Boudawara Nadia, Boussoffara Leila, Knani Jalel
Respiratory Medicine Case Reports.2021; 33: 101396. CrossRef - Primary intratracheal schwannoma misdiagnosed as severe asthma in an adolescent: A case report
Hui-Rong Huang, Pei-Qiang Li, Yi-Xin Wan
World Journal of Clinical Cases.2021; 9(17): 4388. CrossRef - PD‐1/PD‐L1 negative schwannoma mimicking obstructive bronchial malignancy: A case report
Daibing Zhou, Xiaoyan Xing, Jie Fan, Youzhi Zhang, Jie Liu, Yi Gong
Thoracic Cancer.2020; 11(8): 2335. CrossRef - Case report: A tracheobronchial schwannoma in a child
Li Zhang, Wen Tang, Qing-Shan Hong, Pei-feng Lv, Kui-Ming Jiang, Rui Du
Respiratory Medicine Case Reports.2020; 30: 101047. CrossRef - Recurrent transmural tracheal schwannoma resected by video-assisted thoracoscopic window resection
Huiguo Chen, Kai Zhang, Mingjun Bai, Haifeng Li, Jian Zhang, Lijia Gu, Weibin Wu
Medicine.2019; 98(51): e18180. CrossRef - Primary intratracheal schwannoma resected during bronchoscopy using argon plasma coagulation
Purva V Sharma, Yash B Jobanputra, Tatiana Perdomo Miquel, J Ryan Schroeder, Adam Wellikoff
BMJ Case Reports.2018; : bcr-2018-225140. CrossRef - Dumbbell posterior mediastinal schwannoma invading trachea: Multidisciplinary management – weight off the chest
Abhijeet Singh, VallandramamR Pattabhiraman, Arjun Srinivasan, Sivaramakrishnan Mahadevan
Lung India.2018; 35(3): 269. CrossRef - Primary tracheal schwannoma a review of a rare entity: current understanding of management and followup
Shadi Hamouri, Nathan M. Novotny
Journal of Cardiothoracic Surgery.2017;[Epub] CrossRef - A Case of Primary Tracheal Schwannoma
Sung Min Choi, Ji Hong You, Sang Bae Lee, Seong Han Kim, Yon Soo Kim
Kosin Medical Journal.2017; 32(2): 258. CrossRef - Endobronchial Neurilemmoma Mimicking a Bronchial Polyp
Ryoung Eun Ko, Seung Yong Park, Yeong Hun Choe, So Ri Kim, Heung Bum Lee, Yong Chul Lee, Seoung Ju Park
Soonchunhyang Medical Science.2015; 21(2): 176. CrossRef - Optimal treatment for primary benign intratracheal schwannoma: A case report and review of the literature
XIAHUI GE, FENGFENG HAN, WENBIN GUAN, JINYUAN SUN, XUEJUN GUO
Oncology Letters.2015; 10(4): 2273. CrossRef
 PubReader
PubReader ePub Link
ePub Link-
 Cite this Article
Cite this Article
- Cite this Article
-
- Close
- Download Citation
- Close
- Figure

 E-submission
E-submission