Current Concepts and Occurrence of Epithelial Odontogenic Tumors: I. Ameloblastoma and Adenomatoid Odontogenic Tumor
Article information
Abstract
Ameloblastomas and adenomatoid odontogenic tumors (AOTs) are common epithelial tumors of odontogenic origin. Ameloblastomas are clinico-pathologically classified into solid/multicystic, unicystic, desmoplastic, and peripheral types, and also divided into follicular, plexiform, acanthomatous, granular types, etc., based on their histological features. Craniopharyngiomas, derived from the remnants of Rathke's pouch or a misplaced enamel organ, are also comparable to the odontogenic tumors. The malignant transformation of ameloblastomas results in the formation of ameloblastic carcinomas and malignant ameloblastomas depending on cytological dysplasia and metastasis, respectively. AOTs are classified into follicular, extrafollicular, and peripheral types. Ameloblastomas are common, have an aggressive behavior and recurrent course, and are rarely metastatic, while AOTs are hamartomatous benign lesions derived from the complex system of the dental lamina or its remnants. With advances in the elucidation of molecular signaling mechanisms in cells, the cytodifferentiation of epithelial tumor cells in ameloblastomas and AOTs can be identified using different biomarkers. Therefore, it is suggested that comprehensive pathological observation including molecular genetic information can provide a more reliable differential diagnosis for the propagation and prognosis of ameloblastomas and AOTs. This study aimed to review the current concepts of ameloblastomas and AOTs and to discuss their clinico-pathological features relevant to tumorigenesis and prognosis.
EPITHELIAL ODONTOGENIC TUMORS
The pathogenetic mechanism of odontogenic tumors is closely related to the developmental processes of teeth. As a result, the molecular signaling mechanisms for normal enamel organs and odontogenic tumors have been closely compared. For the organogenesis of tooth germs, the reciprocal induction between odontogenic mesenchyme and enamel epithelium sequentially progresses to differentiate odontoblasts into odontogenic mesenchyme and ameloblasts in the enamel epithelium.1,2 During these processes of tooth formation, the tumorigenesis of odontogenic epithelium occurs in the state of immature odontogenic tissues, resulting in different histological features and variable potentials of tumor propagation. However, different epithelial odontogenic tumors can be classified into ameloblastomas, adenomatoid odontogenic tumors (AOTs), calcifying epithelial odontogenic tumors, odontogenic ghost cell tumors, squamous odontogenic tumors, ameloblastic fibromas, ameloblastic fibroodontomas, or odontomas, etc. This study was conducted to review the current concepts and occurrences of ameloblastomas and AOTs.
AMELOBLASTOMA
Once designated as an adamantinoma in 1885 (Malassez) but renamed to ameloblastoma in 1930 (Ivey and Churchil), ameloblastoma is a representative benign tumor of odontogenic epithelium. Some authors still misuse the term adamantinoma to describe ameloblastomas, even though an adamantinoma, meaning "very hard," is a rare bone tumor that differs in histology and frequency of malignancy from ameloblastomas.3 Ameloblastomas are common, have an aggressive behavior and recurrent course, and are rarely metastatic. However, adamantinomatous craniopharyngioma and adamantinoid basal cell carcinoma are terms that are still used to describe their histological characteristics.4
Characteristics of ameloblastoma
Ameloblastoma is a slow growing odontogenic epithelial tumor of the jaw and accounts for about 1% of all oral tumors and about 18% of odontogenic tumors. It is primarily seen in adults in the third to fifth decades of life, with almost equal sex predilection.5 Radiographically, it appears as an expansile radiolucency with thinned and perforated cortices, frequently causing root resorption. These features are common in giant cell tumors, aneurysmal bone cysts, and renal cell carcinoma metastasis, thus a definitive diagnosis can be made through histopathology.
A total of 5,213 cases of ameloblastomas are found in literature, including 3,677 cases of ameloblastomas available in literature from 1960 to 1993,5 340 cases of ameloblastomas in the Malaysian population (1993 through 2008),6 and 1,196 cases of ameloblastomas diagnosed during the years of 1993 to 2009 in Chulalongkorn University, Bangkok.7 These studies revealed the mean age of patients as 36.1 years, with 2,709 (51.9%) patients being male, and mandibular tumors (81.7%) outnumbering maxillary tumors. In the variants of ameloblastomas, approximately 10.7% were unicystic ameloblastomas and 1.8% were peripheral ameloblastomas.
In Korea, about 500 cases of ameloblastomas were sporadically reported, and among them, 452 cases were available to be statistically analyzed.8-10 The ratio of males to females was about 57:43, and the average age was approximately 33.8 years. The most frequent site of involvement was around the mandibular molar area (57.7%). The tumors were composed of conventional ameloblastomas (48.9%), unicystic ameloblastomas (25.3%), and peripheral ameloblastomas (3.1%). The recurrence rate for conventional ameloblastomas (17.1%) was significantly higher than for the unicystic type (9.1%). According to the histology, the acanthomatous, plexiform, and follicular patterns of the conventional ameloblastomas had similar recurrence rates of 16.2%, 15.9%, and 12.7%, respectively.
Molecular mechanisms of ameloblastoma
Every cellular changes, including proliferation, differentiation, senescence, tumorigenesis, etc., occur through the activation or inactivation of related molecular signaling pathways. The overexpression or underexpression of important signaling molecules may play an important role in the tumorigenesis of ameloblastomas. First of all, several proteins expressed in the enamel epithelium during the early stage of tooth formation, including amelogenin,11 tuftelin,12 and ameloblastin,13,14 are specifically expressed in tumor cells of ameloblastomas, but enamelin and amelotin expressed in the mineralizing stage of tooth formation are rarely positive in tumor cells of ameloblastomas.15,16
Different proteins functioning in the signaling of the proliferation and differentiation of enamel epithelium and odontogenic tumors have been identified by many authors. The cellular and stromal proteins of syndecan-1,17 perlecan, α-dystroglycan, integrin β1,18 CD10, and osteopontin19 are overexpressed in ameloblastomas that exhibit locally invasive behavior and a high risk of recurrence. Solid ameloblastomas show an intense expression of fibronectin at the epithelial-mesenchymal interface, whereas desmoplastic ameloblastomas reveal no immunoexpression of fibronectin at this site. Ameloblastomas present a stronger immunoreaction of tenascin than AOTs, especially at the epithelial-mesenchymal interface, while AOTs and desmoplastic ameloblastomas exhibit an intense labeling for type I collagen. These expression patterns of matrix proteins agree with the more locally invasive behavior of ameloblastomas in comparison to AOTs.20
The local invasiveness of ameloblastoma is also related to the expressions of metalloproteinase (MMP)-2 and MMP-9. The overexpression of MMP-9 in ameloblastomas is possibly modulated by the unmethylation of the gene and may influence the aggressive behavior and high recurrence rates of ameloblastoma.21
The facts that receptor activator of nuclear factor κB ligand (RANKL) and MMP-9 are expressed in ameloblastomas may suggest that ameloblastoma cells have the potential to induce osteoclastogenesis, resulting in the rapid destruction of bone marrow.22,23 The higher expression rates of interleukin (IL)-1α and IL-6 are associated with tumor size in ameloblastomas. Therefore, it is suggested that the IL-1α and IL-6 cytokines play a role in the aggressive behavior of ameloblastomas by increasing bone resorption.24
The overexpression of cyclin D1, a member of G1 cyclins controlling the cell-cycle transit from the G1 to S phase, is found in some plexiform ameloblastomas but is relatively mild in AOTs. The peripheral columnar and central stellate reticulum-like cells of ameloblastomas exhibit a dominant immunoreaction of cyclin D1, which is almost non-existent in the squamous and granular cells of ameloblastomas.25
Midkine is a heparin-binding growth factor overexpressed in various human cancers and is expressed in ameloblastomas and ameloblastic carcinomas. This midkine expression in the majority of ameloblastomas may suggest a role of the protein in tumor development, progression, and behavior.26
Epithelial growth factor receptor expression had no statistical significance for local recurrence, patient age, or tumor size, whereas a significant relation existed between CD10 expression and the Ki-67 labeling index, which is significantly related to the recurrence of ameloblastomas.27
The overexpression of p53 and murine double minute 2 (MDM2) are associated with the pathogenesis and oncogenesis of ameloblastomas.28 The overexpression of p-mTOR, p-4E-BP1, and p-p70S6K in the nucleus also are related to the invasiveness of ameloblastomas.29
The transforming growth factor-β (TGF-β)/SMAD signaling pathway is commonly activated in ameloblastomas, AOTs, and calcifying cystic odontogenic tumors. Meanwhile, the TGF-β/SMAD immunoreaction is significantly reduced in ameloblastomas in comparison to AOTs and calcifying cystic odontogenic tumors. These changes may lead to the more aggressive biological behavior of ameloblastomas through increased cell proliferation and reduced apoptosis and differentiation.30
WNT proteins (except WNT-10b) are heterogeneously expressed in different types of ameloblastomas, indicating that WNT expression is closely related to tumor cell differentiation and invasion.31 However, the WNT-related bone-forming genes of WDR5 and runt-related transcription factor 2 (RUNX2) are down-regulated in ameloblastomas.32
Alterations in the Sonic Hedgehog signaling pathway, including protein patched homolog (PTCH) gene mutations, have been associated with the pathogenesis of some odontogenic tumors including ameloblastomas and are closely related to nevoid basal cell carcinoma syndrome through the genetic alteration of PTCH. Therefore, ameloblastoma diagnosis possibly warrants a search for associated cutaneous basal cell carcinomas and other benign and malignant tumors related to nevoid basal cell carcinoma syndrome.33-35
There appears to be no difference in the expression of E-cadherin or β-catenin between tooth germs and solid and unicystic ameloblastomas. The expression of these molecules seems to be mainly related to the process of cell differentiation.36
Rho GTPases that regulate the cell cycle, shape, polarization, invasion, migration, and apoptosis are overexpressed in ameloblastomas. Therefore, it is suggested that these GTPases play a role in the neoplastic epithelial cell phenotype determination of ameloblastomas (polarized or non-polarized), as well as in variant (solid or unicystic) and subtype (follicular or plexiform) determination.37
On the other hand, tumor necrosis factor-related apoptosis-inducing ligand (TRAIL) and TRAIL receptor are diffusely expressed in ameloblastomas and are possibly involved in the neoplastic transformation of odontogenic epithelium. FAS (CD95), FAS ligand, and caspase-3 are also expressed in ameloblastomas.38 These expressions may suggest some intrinsic regulation of neoplastic cell proliferation and death in ameloblastomas, thus explaining their slow growth and inability to metastasize.39
Histological types of ameloblastoma
Histologically, ameloblastomas are classified into follicular, plexiform, acanthomatous, and granular cell types. Less common histologic variants are clear cell, desmoplastic, basal cell, papilliferous, and keratoameloblastoma types. Generally, one-third of ameloblastomas are plexiform, one-third are follicular, and other variants such as acanthomatous types usually occur in older patients.
Follicular type
The follicular type of ameloblastoma frequently forms various sizes of dental follicle structures and possesses an outer arrangement of columnar or palisaded ameloblast-like cells and inner zone of triangular-shaped cells resembling the stellate reticulum in the bell stage of the tooth germ (Fig. 1A, E). Histopathology shows cells that have a tendency for nuclei to move away from the basement membrane. This process is referred to as "reverse polarization." The central cells sometimes degenerate to form central microcysts.
Photomicrographs of the different types of ameloblastoma. (A, E) Follicular type. (B, F) Plexiform type. (C, G) Acanthomatous type. (D, H) Desmoplastic type. Panels (E), (F), (G), and (H) are the magnifications of panels (A), (B), (C), and (D), respectively.
The follicular type of ameloblastomas grow and usually form multicystic nodules, resulting in multilocular tumor masses with frequent recurrence compared to the other types of ameloblastomas.40
Plexiform type
The plexiform type contains epithelium that proliferates in a "fish net pattern," or in a "cord-like fashion anastomosing each other," hence the name "plexiform." There are layers of cells between the proliferating epithelium with well-formed desmosomal junctions, simulating spindle cell layers (Fig. 1B, F). Plexiform ameloblastomas with multiple cyst formation are rare and benign odontogenic tumors which may reach grotesque proportions, affecting a large region of the jaw.41
Acanthomatous type
Acanthomatous ameloblastoma is the extremely rare variant exhibiting solid epithelial cell nests with peripheral palisading ameloblastic cells and central squamous cell differentiation (Fig. 1C, G).42 The acanthomatous ameloblastoma masquerades as a squamous cell carcinoma and also appears as a "hybrid" ameloblastoma admixed with a pronounced desmoplastic pattern.43
Granular cell type
Granular cell ameloblastoma is a variant of ameloblastoma, where the tumor cells located in the central portion of the follicles have granular eosinophilic cytoplasm and the peripheral tumor cells resemble ameloblasts. The granular cells are a transitional or matured phase in the life cycle of ameloblastomas, starting with normal stellate reticulum-like cells, leading to a production of granules and finally resulting in degeneration and the formation of cystic areas, differing from the aggressive tumor nature reported previously.44 However, the granular cell type is rare and accounts for only 4% of ameloblastomas.45
Others
Keratoameloblastoma is a very rare ameloblastoma variant defined by extensive squamous metaplasia and keratinization. Hemangiomatous ameloblastoma is also described as a rare ameloblastic variant.46
Basal cell ameloblastoma is also a very rare variant of ameloblastoma resembling basaloid squamous cell carcinoma but possessing the pathological features of conventional ameloblastoma.47
Adenoid ameloblastoma is a rare variant in this category and can cause problems in diagnosis due to the presence of areas resembling AOT and the occurrence of varying degrees of dentinoid formation.48
Clinico-pathological classification of ameloblastoma
According to the World Health Organization (WHO), ameloblastomas are classified into the following types depending on the origin of tumorigenesis: solid/multicystic, extraosseous/peripheral, desmoplastic, and unicystic. The common solid/multicystic ameloblastomas arise from enamel epithelial rests in jaw bone, while unicystic ameloblastomas arise from the epithelium of odontogenic cysts. Desmoplastic ameloblastomas exhibit active stromal proliferation, while peripheral ameloblastomas arise from dental lamina rests and oral mucosa epithelium. These tumor types differ in biological behavior and rate of recurrence. Therefore, each type of ameloblastoma requires different forms of treatment.49 Additionally craniopharyngiomas arising from the rests of Rathke's pouch epithelium are comparable to ameloblastomas.
Solid/multicystic ameloblastoma (intraosseous ameloblastoma, central ameloblastoma, conventional ameloblastoma)
Conventional ameloblastomas are the most common and prevalent among odontogenic tumors. Clinico-radiographically, they present as intraosseous lesions which exhibit slow, painless swelling or expansion of the jaw, with multilocular expansile radiolucency most frequently found in the mandibular molar/ramus area. Depending on the histological features, the conventional ameloblastoma is also called a solid/multicystic ameloblastoma, in contrast to the unicystic ameloblastoma.50
Unicystic ameloblastoma (mural ameloblastoma)
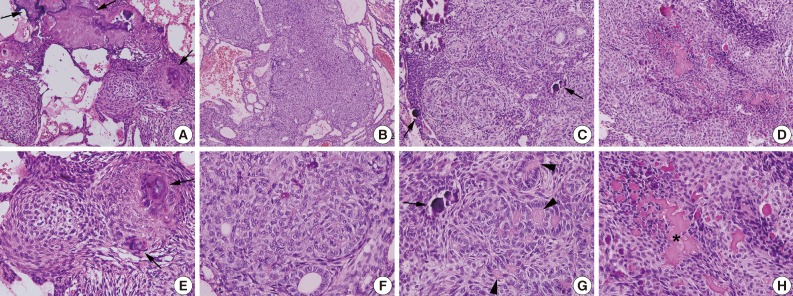
Unicystic ameloblastomas refer to the cystic lesions that exhibit clinical, radiographic, or gross features of a mandibular cyst but upon histologic examination show typical ameloblastoma epithelium lining part of the cyst cavity, with or without luminal and/or mural tumor growth (Fig. 2A, B).51 The unicystic ameloblastoma is believed to be less aggressive than the conventional ameloblastoma. Since this tumor shows considerable clinical and radiographical similarities with a dentigerous cyst, a differential diagnosis is necessary. Moreover, the recurrence of unicystic ameloblastomas may be long delayed, and long-term postoperative follow-up is essential for the proper management of these patients.52

Photomicrographs of unicystic ameloblastoma (A, B) and ameloblastic carcinoma (C). (A, B) Ameloblastoma occurs from the epithelium of odontogenic keratocyst. (C) Ameloblastic carcinoma shows severe atypia of tumor cells.
Unicystic ameloblastomas also demonstrate the variable propensity for recurrence. As a result, there is a difference in the biological behavior between mural unicystic ameloblastomas and those which are simply cystic or exhibit intraluminal proliferation.53 However, unicystic ameloblastomas have a distinct trait of less aggressive behavior compared to conventional ameloblastomas.54
Desmoplastic ameloblastoma
Desmoplastic ameloblastoma is a benign but locally invasive variant of the solid/multicystic ameloblastoma (Fig. 1D, H). The desmoplastic ameloblastoma accounts for only 4% to 13% of all ameloblastomas and can induce abundant stromal osteoplasia.55 Simple desmoplastic ameloblastomas (88.0%) and desmoplastic ameloblastomas with osteoplasia (12.0%) were the histologic variants reported, however, desmoplastic ameloblastomas showed slower growth rate than conventional ameloblastomas.56 In the immunoreactions of proliferating cell nuclear antigen (PCNA) and Ki-67, the desmoplastic ameloblastoma demonstrated a significantly lower proliferation rate compared to solid/multicystic ameloblastomas, unicystic ameloblastomas, and ameloblastic carcinomas.57
Peripheral ameloblastoma
Peripheral ameloblastoma is a rare extraosseous odontogenic tumor with histological characteristics similar to those found in conventional intraosseous ameloblastomas. Peripheral ameloblastomas clearly originated from odontogenic epithelial remnants located at the outside of tooth germs rather than from common oral epithelium.58,59 This tumor accounts for approximately 2-10% of all ameloblastomas, similar to classical ameloblastomas.60
Craniopharyngioma compared with odontogenic tumors
Craniopharyngiomas are low-grade epithelial neoplasms occurring almost exclusively in the sellar/suprasellar region. Craniopharyngiomas are generally considered to arise from the remnants of Rathke's pouch or a misplaced enamel organ. The overall incidence rate of craniopharyngiomas is approximately 1.3 per million. During adulthood, there is a peak incidence between 40 and 44 years. Based on the striking histological similarity of craniopharyngiomas and odontogenic tumors, craniopharyngiomas can be divided into adamantinomatous and papillary variants according to the WHO classification of odontogenic tumors. Approximately 50% of craniopharyngiomas corresponded histologically to calcifying odontogenic cysts, 24% to ameloblastomas, and 15% exhibited features of both calcifying odontogenic cysts and ameloblastomas either within the same specimen or in specimens derived from different resections. Only a rare 5% of craniopharyngiomas resembled calcifying epithelial odontogenic tumors, with approximately 2% of craniopharyngiomas resembling AOTs. However, no odontogenic counterparts could be established for papillary craniopharyngiomas.61
The adamantinomatous craniopharyngioma is a locally aggressive neoplasm with a significant rate of recurrence.62 All adamantinomatous craniopharyngiomas show a variable degree of enamel protein expression, mainly in ghost cells that includes amelogenin, enamelin, and enamelysin. Lymphoid enhancer binding factor 1 (LEF1) is also heterogeneously expressed in adamantinomatous craniopharyngiomas. Remarkably, the expression pattern of LEF1 is identical to that of nuclear β-catenin accumulation. In contrast, none of the papillary craniopharyngiomas expresses enamel proteins or LEF1. These findings suggest that adamantinomatous craniopharyngiomas consistently exhibit odontogenic epithelial differentiation. Since adamantinomatous craniopharyngiomas are possibly caused by a β-catenin mutation, the inappropriate activation of β-catenin/LEF1 complex-dependent transcription may play a critical role in their tumorigenesis.63
Only shadow cells present in adamantinomatous craniopharyngiomas are positive for human hair keratin, which may indicate their follicular differentiation. Rathke's cleft cyst ciliated cuboidal cells are cytokeratin (CK)-7+/CK-8+/CK-14-, and metaplastic squamous cells are CK-7+/CK-8-/CK-14+. These findings suggest that adamantinomatous craniopharyngiomas may be related to heterotopic ectodermal tissue, which can differentiate into hair follicles, while papillary craniopharyngiomas may arise from Rathke's cleft cyst.49
In the immunological expressions of KL1 (high molecular weight cytokeratins), 5D3 (low molecular weight cytokeratins) and involucrin (characteristic of terminally differentiated keratinocytes), adamantinomatous craniopharyngiomas are similar to those reported in ameloblastomas for squamous differentiation, implying that adamantinomatous craniopharyngiomas and ameloblastomas are homologous lesions.64
The papillary type of craniopharyngiomas occurs almost exclusively in adult patients, shows frequent radiologic solidity and absence of calcification, demonstrates a macroscopic papillary nature, and, microscopically, exhibits a well-differentiated papillary squamous epithelium without calcification, palisaded cells, or keratoid nodules.65
Malignant transformation in craniopharyngiomas, although rare, does occur. It assumes varied histologic appearances, usually after multiple recurrences and radiation therapy, and has a nearly uniform fatal outcome. De novo malignancy in odontogenic tumors of the sella is even more unusual but also has an ominous prognosis.66
Malignant transformation of ameloblastomas
The malignant transformation of ameloblastomas can exhibit an aggressive clinical course, including multiple recurrences, a short disease-free interval, pulmonary metastasis, and extensive skull-base infiltration. They constitute less than 1% of all ameloblastomas. Malignancies in ameloblastomas may involve local dysplastic change or metastasis in the tissue. The former are classified as ameloblastic carcinomas, the latter as malignant ameloblastomas.
Ameloblastic carcinoma
Ameloblastic carcinoma is a rare, odontogenic, malignant tumor that has the features of ameloblastoma in addition to cytologic atypia with or without metastasis. It is classified as primary type; secondary type, intraosseous; secondary type, and peripheral type according to the WHO classification of 2005.67
The majority of cases reported are secondary type ameloblastic carcinoma. The mandible is the most common site of occurrence for both ameloblastic carcinoma types. The tumor cells resemble the cells seen in ameloblastomas but exhibit cytologic atypia (Fig. 2C), including plexiform invasion, hyperchromatism, mitosis, and necrosis that are associated with history of recurrence and death by disease, as well as tumors with clear cells, especially in the secondary type of ameloblastic carcinoma. Secondary type ameloblastic carcinoma appears to correlate with recurrence and mortality.68 Direct extension of the tumor with lymph node involvement and metastasis to various sites (frequently the lungs) have been reported. Wide local excision is the treatment of choice. Regional lymph node dissection should be considered and performed selectively.69
In the analysis of six ameloblastic carcinomas from the literature up to 2009, the mean age was 49.2 years with a wide age range (7-91 years). The rate of occurrence was higher in males, and the most common site of occurrence was the mandible. Most cases (70%) involved the posterior portion of the jaw. Metastatic lesions were detected in 22% of patients during follow-up, and the lung was the most common area of distant metastasis.70
Ameloblastic carcinomas may present de novo, ex ameloblastomas, or ex odontogenic cysts. Most ameloblastic carcinomas are presumed to present de novo. The clinical course of ameloblastic carcinomas is typically aggressive, with extensive local destruction.71
Peripheral ameloblastic carcinoma is an extremely rare odontogenic tumor derived from the remnants of dental lamina and/or mucosal epithelium of the oral mucosa. The histology of the biopsy tissue and surgically-removed specimens reveal characteristic features resembling squamous cell carcinoma, basal cell carcinoma, and benign follicles of ameloblastoma. These neoplastic structures, as well as the proliferation and elongation of the mucosal epithelium, comprise an extensive network. The varied cytopathologic findings may be related to the proliferation and transformation of basal cells of the mucosal epithelium toward ameloblastic carcinoma and variable squamous differentiation.72
A rare variant of spindle-cell ameloblastic carcinoma resulting in extensive metastasis and unfavorable outcomes have been reported in about seven cases in the literature.73 Ultrastructural and immunohistochemical examinations also show the spindle-cell component of the tumor to be epithelial in character.73
In genome analysis, the CpG methylation of p16 (cyclin-dependent kinase inhibitor 2A) is observed in all ameloblastic carcinoma samples, but only one ameloblastoma specimen exhibits the mutation. Therefore, it is presumed that p16 alteration may play a role in the malignant progression of ameloblastic carcinoma.74
Malignant ameloblastoma
The WHO defines malignant ameloblastoma as a lesion exhibiting features of an ameloblastoma in primary and metastatic growths. The WHO classification emphasizes metastasis as a diagnostic criterion but is rather vague in defining its histopathologic aspects. It is advocated that the term malignant ameloblastoma be reserved for those lesions that, in spite of a seemingly innocuous histology, have produced metastatic growth. The WHO classification should be modified to include ameloblastic carcinoma as a diagnostic term for lesions that combine features of an ameloblastoma with a less-differentiated histomorphology.75
It is not possible to distinguish conventional intraosseous ameloblastomas from malignant ameloblastomas according to histopathologic features. It is necessary to pay special attention, especially in elderly patients, and to carry out further clinical, radiological, and pathohistological diagnostic procedures, such as immunohistochemical analysis.76 When metastases occur, although uncommon, lungs (71%) constitute the most frequent site involved, followed by cervical lymph nodes (28%). The female-to-male ratio is about 1:1.1. Primary tumor is diagnosed in about 28% of cases at ages ≤20 years, with a maxilla-to-mandible ratio of approximately 1:5.2. The mean disease-free interval and survival for pulmonary metastasis is about 14.4 years and 3 years, respectively, and about 13 years and 6 years for cervical metastasis, respectively.76 So far, less than 50 cases of ameloblastoma with metastases have been reported.77
ADENOMATOID ODONTOGENIC TUMOR
Characteristics of adenomatoid odontogenic tumor
AOT is an uncommon, hamartomatous, benign, epithelial lesion of odontogenic origin that was first described by Driebaldt in 1907 as a pseudo-adenoameloblastoma. The current WHO classification of odontogenic tumors defines AOT as being composed of odontogenic epithelium in a variety of histoarchitectural patterns, embedded in mature connective tissue stroma, and characterized by slow but progressive growth.78
The introduction of the name "AOT" has resulted in simpler and fruitful surgical management like enucleation and curettage with no reports of recurrence. In the past, a similar lesion with the diagnosis of adeno-ameloblastoma resulted in unnecessary mutilating surgery.79
Compared to ameloblastomas, AOT is a benign, nonaggressive tumor with limited growth and no tendency of recurrence. This is often misdiagnosed as an odontogenic cyst and accounts for about 1% to 9% of all odontogenic tumors. It is predominantly found in young and female patients, is more often located in the maxilla and is typically associated with an unerupted permanent tooth. There are three variants of AOT-follicular, extrafollicular, and peripheral.80
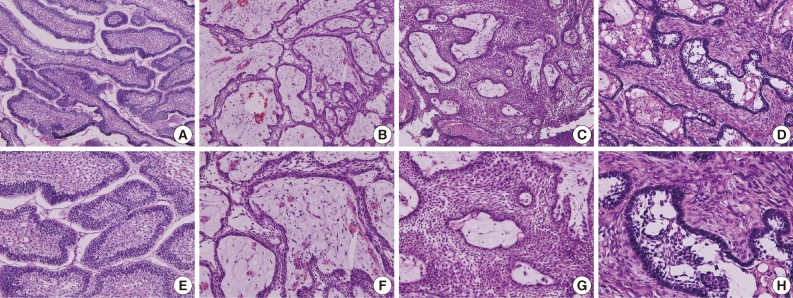
Among 15 cases of AOT, the anterior maxilla was the most common site (66.6%), and radiographically, most cases showed a unilocular radiolucency with well-defined borders (57.1%). Histologically, most cases exhibited a predominantly solid growth pattern (46.7%) or a similar proportion of solid and cribriform patterns (46.7%) (Fig. 3A, B, E, F). Eosinophilic amorphous material ("tumor droplets") was found in all cases (100%) (Fig. 3C, D, G, H). Most tumors contained duct-like spaces (93.3%) (Fig. 3B, C, F, G) and convoluted structures (60.0%), whereas a minor proportion of the cases presented with calcifying epithelial odontogenic tumor-like areas (26.7%). Variable amounts of calcified materials were found in most AOTs (80.0%) (Fig. 3A, D, E, H), whereas osteodentin and perivascular hyalinization were seen only rarely (6.7% for each). About five (33.3%) cases had areas mimicking a dentigerous cyst and were mostly diagnosed in females (80.0%).81
Variable histological features of adenomatoid odontogenic tumor. (A, E) Multiple epithelial follicles with calcifications (arrows). (B, F) Whirling epithelial cells with adenomatoid structures. (C, G) Multifocal calcification (arrows) with eosinophilic coagulum (arrowheads). (D, H) Abortive tooth materials (asterisk) produced by epithelial tumor cells.
AOT has numerous dispersed or clustered radiopaque foci compared to the presence of calcification in calcifying cystic odontogenic tumors showing a thin radiopaque line and discrete radiopaque foci. Therefore, radiolucency with numerous radiopaque foci (particularly when the radiolucency surrounds a portion of the root or entire tooth) is suggestive of an AOT rather than a calcifying cystic odontogenic tumor.82
In a review of 272 AOTs with special emphasis on radiological features, the patient age at time of diagnosis ranged from 3 years to 82 years (mean, 18.4 years). The maxilla-to-mandible ratio was 1.7:1. Small opacities were present in 77% of the lesions and were associated with expansion of the cortex. The significant radiological features in patients aged 30 years and above were root resorption and lesions crossing the midline.83
According to all reports regarding AOTs and cited in PubMed since 1990, AOTs account for approximately 1% to 9% of all odontogenic tumors. These tumors are predominantly found in young female patients, are more often located in the maxilla, and are typically associated with an unerupted permanent tooth. AOTs frequently resemble other odontogenic lesions such as dentigerous cysts or ameloblastomas.84
Molecular mechanisms of adenomatoid odontogenic tumor
AOT is a noninvasive tumor that never infiltrates surrounding normal tissues. However, the mean values of the labeling index for Ki-67 in solid ameloblastomas and AOTs are 4% and 1%, respectively. These values for B-cell lymphoma 2 (BCL-2) in solid ameloblastomas and AOTs are 63% and 26%, respectively. These findings suggest the harmatomatous behavior of AOT compared to ameloblastomas.85 AOT demonstrates a similar level of PCNA immunoreaction to ameloblastomas but exhibits weaker expressions of p53 and MDM2. This implies that AOT has less aggressive behavior than ameloblastomas.86,87 In AOTs, some epithelial tumor cells show a strong cytoplasmic reaction to β-catenin,88 possess whirling epithelial cells expressing cyclin D1, and have spindle-shaped tumor cells that are positive for podoplanin.89 These findings are suggestive for the active proliferation of AOTs.25
Since the consistent and strong immunolocalization of hepatocyte growth factor (HGF) and c-met in squamous cells are present in AOTs, the HGF/c-met interaction may have an influence on squamous differentiation in the odontogenic epithelium of AOT.90 Enamel proteins including amelogenin, ameloblastin, and amelotin, as well as TGF-β/SMADs, are more intensely expressed in AOTs than in ameloblastomas. Therefore, AOTs exhibit less aggressive biological behavior and increased cytodifferentiation and apoptosis than ameloblastomas.16,30
Types of adenomatoid odontogenic tumor
AOT is an uncommon, progressively growing, and asymptomatic benign non-invasive lesion that occurs twice as often in females and usually in the second decade of life. The three variants of AOT are characteristic-a follicular variant (73%) associated with an impacted and displaced tooth, an extrafollicular variant (24%) mimicking a radicular cyst around the apex of a tooth, and a peripheral (epulis-like) variant (3%) exhibiting a periodontal bone defect or ectopic growth. On reappraisal of the origin and pathogenesis of AOT, it would seem that this tumor or hamartomatous lesion is derived from the odontogenic epithelium of the dental lamina complex or its remnants.91
Follicular type
The follicular variant of the AOT is thought to originate from the reduced enamel epithelium of the dental follicle. All the variants of AOT exhibit identical histologic features. The follicular variant (male:female ratio, 1:1.9) accounts for 73.0% to 97.2% of all AOTs and is three times as frequent as the extrafollicular variant. The follicular variant is diagnosed earlier in life (mean age, 17 years) compared to the extrafollicular variant (mean age, 24 years), whereas 53.1% of all the variants occur during the teenage years (13-19 years). Follicular AOT is associated with one embedded tooth in 93.2% of cases. Maxillary permanent canines account for 41.7% of the cases, and all four canines are involved in 60.1% of AOT-associated cases.92
Extrafollicular type
Compared to the follicular variant of AOT, the origin of the extrafollicular variant remains unclear. However, the available reviews in the literature suggest that some extrafollicular AOTs may arise as a secondary phenomenon within pre-existing odontogenic cysts or cystic tumors. For example, the tumor may originate from the epithelial lining of an odontogenic cyst or unicystic ameloblastoma.93,94 A rare subvariant of the extrafollicular type of AOT may radiographically mimic periapical diseases, which is initially suspicious of small periapical pathology. This subvariant is very rare, with only 12 cases reported in the literature.95
Peripheral type
Peripheral AOT occurring far distant from tooth germ structures is rarely encountered. So far, there have been only 14 reported cases of peripheral AOTs. A marked female predominance was apparent in peripheral AOTs, and approximately 90% of the peripheral AOTs occurred in the maxilla,96 showing a striking tendency to occur in the anterior maxilla. The tumor primarily manifested in the incisor and can involve the maxillary antrum.97
SUMMARY
Ameloblastomas and AOTs are benign epithelial tumors of odontogenic origin. The former demonstrates an aggressive behavior and frequent recurrence, while the latter exhibits limited growth and no tendency of recurrence. The differential diagnosis between ameloblastomas and AOTs is essential. Due to the local invasive growth and the possible potential for the malignant transformation of ameloblastomas, it is recommended by WHO that ameloblastomas be classified into solid/multicystic, unicystic, desmoplastic, and peripheral ameloblastomas according to their clinicopathological features rather than the previous histological types including follicular, plexiform, acanthomatous, granular types, etc. AOTs are also classified into follicular, extrafollicular, and peripheral types, even though all types exhibit similar histological features. With advance in the elucidation of molecular signaling mechanisms in cells, the cytodifferentiation of epithelial tumor cells in ameloblastomas and AOTs can be identified using different biomarkers. Therefore, it is suggested that comprehensive pathological observation including molecular genetic information can provide a more reliable differential diagnosis for the propagation and prognosis of ameloblastomas and AOTs.
Notes
No potential conflict of interest relevant to this article was reported.