HER2-low and ultralow breast cancer: interobserver challenges and lessons from a consensus study
Article information
Abstract
Background
The recent approval of trastuzumab deruxtecan for human epidermal growth factor receptor 2 (HER2)–low and HER2-ultralow breast cancer mandates an adequate assessment of these categories.
Methods
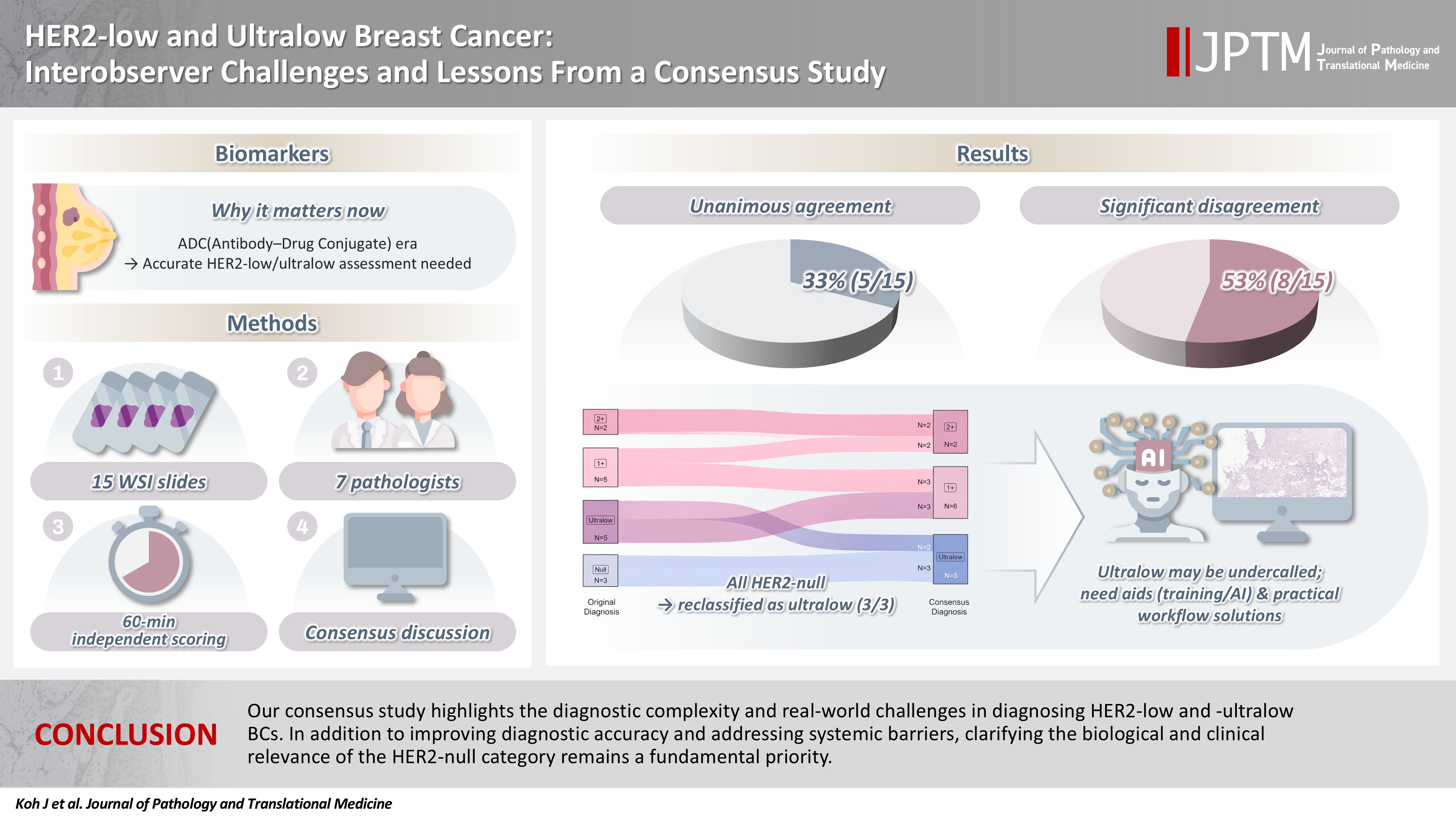
Seven breast pathologists from the Breast Pathology Study Group of the Korean Society of Pathologists held an on-site expert consensus meeting. Fifteen sets of virtual whole slide images (WSI) of hematoxylin and eosin stain and HER2 immunohistochemistry were provided. The pathologists were given 60 minutes to submit their diagnosis of HER2 expression into null, ultralow, 1+, 2+, or 3+. Afterwards, in-depth discussion and consensus diagnoses were made by real-time visualization of the WSI.
Results
After the consensus meeting, unanimous 100% agreements were seen only in five (33.3%) of the examined cases, which consisted of three 1+ cases and two 2+ cases. Two cases (13.3%) had mild disagreement, with only one pathologist’s disagreement. Of note, eight cases (53.3%) showed significant disagreement, defined by more than two pathologists’ disagreement. All HER2-null cases were reclassified as ultralow after consensus review, suggesting potential widespread underclassification of ultralow cases in clinical practice.
Conclusions
Experts had significant discrepancies in interpreting HER2-low/ultralow status. It is important to assess if the distinction between HER2-low and ultralow is strictly required and if HER2-null breast cancer exists in reality.
INTRODUCTION
Trastuzumab deruxtecan (T-DXd), an antibody drug conjugate (ADC) using human epidermal growth factor receptor 2 (HER2) as the target antigen and topoisomerase I inhibitor as the payload [1], has started a novel therapeutic era with unprecedented prolongation of progression-free survival and overall survival in patients with metastatic HER2-positive breast cancer (BC) [2]. Furthermore, the DESTINY-Breast 04 study proved its efficacy in patients with HER2-low BC [3], which is defined as HER2 immunohistochemistry (IHC) 1+ or HER2 IHC 2+ with a negative result on HER2 in situ hybridization (ISH). In response to this, the most recent American Society of Clinical Oncology/College of American Pathologists (ASCO/CAP) guideline, released in 2023, recommended that pathologists be aware of the HER2-low, which became an important trial entry marker [4].
Traditionally, BC has been classified as HER2-positive or HER2-negative based on IHC and ISH, with HER2-positive tumors defined as IHC 3+ or IHC 2+ with HER2 gene amplification [4]. Tumors lacking these features have historically been grouped as HER2-negative. More recently, HER2-low BC has been defined as tumors showing IHC 1+ or IHC 2+ with negative ISH results. An even more refined category, termed HER2-ultralow [5], refers to tumors with faint, incomplete membranous HER2 staining in less than 10% of tumor cells, whereas tumors lacking any convincing membranous staining are classified as HER2-null.
More recently, the DESTINY-Breast 06 study demonstrated similar trends in so-called HER2-ultralow BC, which includes BC with faint or barely perceptible membranous expression of HER2 in less than 10% of the tumor cells [5]. During the screening of DESTINY-Breast 06, it is reported that more than 50% of the locally assessed HER2-null BCs were upgraded into HER2-low or -ultralow after the interpretation by the central lab [6]. In addition, several studies reported significant interobserver variations in determining HER2-low or -ultralow BCs by IHC [7-9].
The Breast Pathology Study Group of the Korean Society of Pathologists previously conducted a nationwide study on the status of HER2-low diagnosis in the Republic of Korea to improve the diagnostic accuracy [10]. In line with this, the group held an expert consensus meeting with seven experienced breast pathologists to evaluate the diagnosis of HER2-low and -ultralow BCs. The meeting aimed to assess (1) the degree of interobserver variability among the breast pathologists, (2) the underlying causes of diagnostic discrepancies, and (3) real-world challenges that may arise in daily practice.
MATERIALS AND METHODS
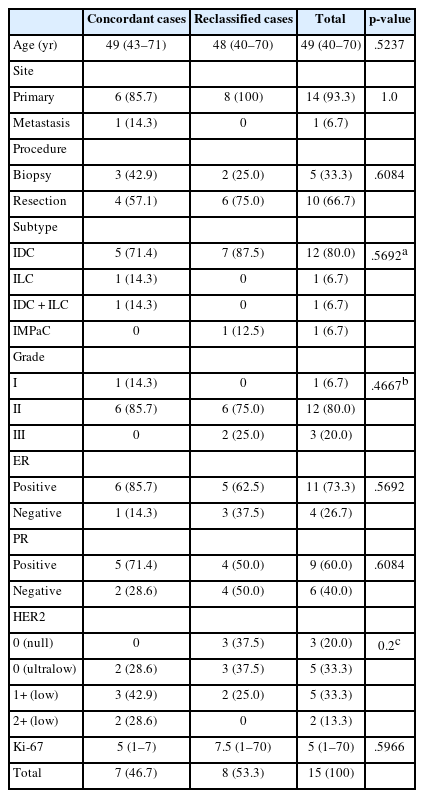
A breast pathologist (J.K.) reviewed the HER2 IHC slides of BC tissue samples from the Department of Pathology, Seoul National University Hospital (SNUH), and selected 15 cases four weeks prior to the consensus meeting. HER2 IHC was performed on formalin-fixed paraffin-embedded tissue sections of 4-µm thickness using the anti-HER2/neu (4B5) rabbit monoclonal antibody and the BenchMark XT automated IHC platform (Ventana Medical Systems, Tucson, AZ, USA). The whole slide images (WSI) files, including hematoxylin and eosin (H&E) stains and HER2 IHC, were digitally scanned at up to 40× magnification using the Leica Aperio GT450 device (Leica, Wetzlar, Germany). The WSIs were then anonymized and downloaded for use on-site at the consensus meeting. Clinicopathological characteristics were obtained retrospectively from medical records and pathology reports. Categorical variables were compared by Fisher’s exact test and continuous variables were compared using the Mann-Whitney test.
Seven breast pathologists from the Breast Pathology Study Group of the Korean Society of Pathologists held an on-site expert consensus meeting. Virtual WSI of H&E and HER2 IHC were reviewed individually by each pathologist during the consensus meeting. To minimize technical variability, all participants used the same model of laptop and the same image viewer software (Aperio ImageScope, Leica) under identical viewing conditions. The pathologists were blinded to the clinicopathological information and given 60 minutes to submit their individual diagnoses of HER2 expression, categorized as null, ultralow, 1+, 2+, and 3+. The original diagnoses and expert submissions were then shared and discussed. Mild disagreement was defined as only one pathologist differing from the consensus, and significant disagreement was defined as more than two pathologists disagreeing. An in-depth discussion was led by the moderator (G.G.), during which HER2 IHC WSIs were displayed in real-time on a shared high-resolution display to facilitate group review.
RESULTS
Study samples
The study set included 15 BC samples, of which 12 cases were invasive ductal carcinomas (IDCs). One sample was obtained from a liver metastasis, while the remaining cases were from the breast. There were five core needle biopsies and 10 surgical resection specimens. The distribution of original HER2 IHC diagnosis was as follows: HER2-null in 20.0% (3/15), ultralow in 33.3% (5/15), 1+ in 33.3% (5/15), and 2+ in 13.3% (2/15). The study set did not include any BCs with HER2 3+. Additional clinicopathological details of the study samples are summarized in Table 1.
Overall diagnostic concordance
After the consensus meeting, unanimous 100% agreement among the seven pathologists was observed in only 5 of the 15 examined cases (33.3%), which consisted of three HER2 1+ cases and two 2+ cases (Fig. 1). Two cases (13.3%) showed mild disagreement, and 8 cases (53.3%) showed significant disagreement. No notable differences in clinicopathological characteristics were observed between cases with concordant versus discordant diagnoses (Table 1), although cases with revised diagnoses tended to have numerically higher Ki-67 labeling indices.
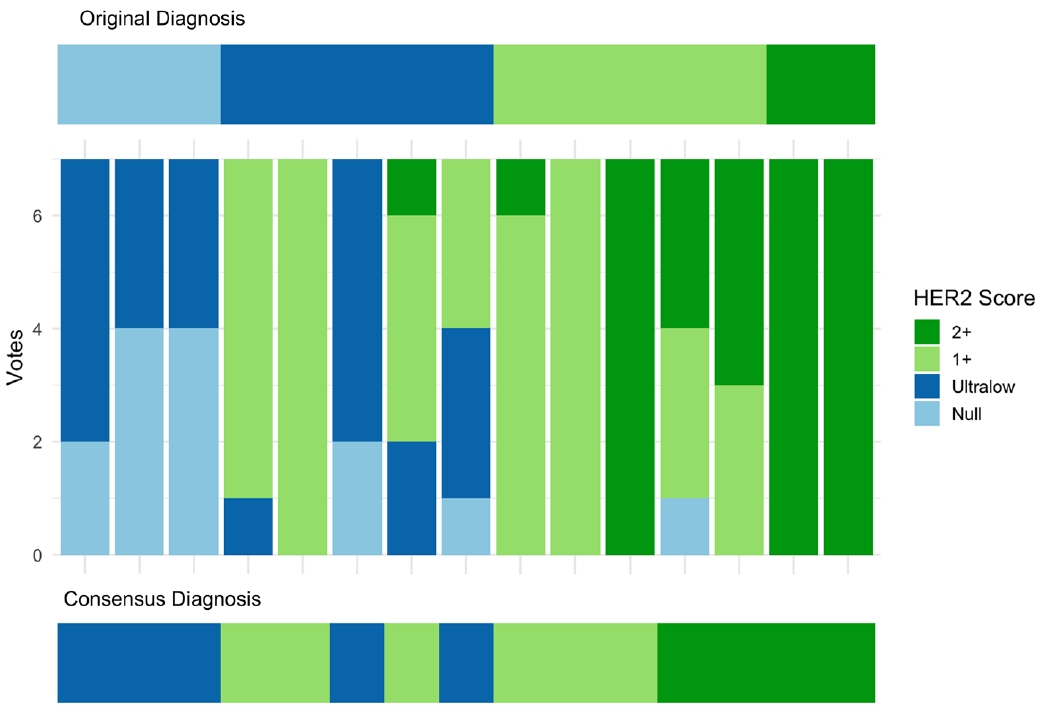
Individual voting patterns and consensus results for human epidermal growth factor receptor 2 (HER2) immunohistochemistry scoring. Stacked bar plots show the distribution of seven pathologists’ scores (null, ultralow, 1+, 2+) for each of the 15 cases. Consensus diagnoses for each case after discussion are shown at the bottom.
Among the eight significantly discordant cases, the original diagnoses varied: three were HER2-null, three were ultralow, and two were 1+. Notably, all three HER2-null cases were reclassified as ultralow after consensus review (Fig. 2), suggesting that HER2-ultralow may be underdiagnosed in real-world practice. Additionally, two cases initially called 1+ were upgraded to 2+, and one ultralow case was reclassified as 1+.

Sankey diagram showing changes between original human epidermal growth factor receptor 2 (HER2) immunohistochemistry diagnoses and consensus diagnoses. Each stream represents the reclassification of individual cases, with line width proportional to the number of cases. Most reclassification occurred between HER2-null and ultralow groups, while 1+ and 2+ categories were relatively stable.
Key illustrative cases with significant disagreement
Case 7 was a representative section from a surgically resected breast specimen with IDC, no special type (Fig. 3A). On low-power magnification, no discernible staining by HER2 was observed, and upon closer inspection, the vast majority of tumor cells remained negative; thus, the original diagnosis was HER2-null. During the consensus meeting, however, three pathologists identified tiny foci of cells with faint membranous staining. Their fields of view were shared on the screen in real time, and the panel unanimously agreed that the membrane staining was valid, resulting in a consensus diagnosis of HER2-ultralow. This case highlights the inherent difficulty in differentiating HER2-ultralow BC from HER2-null, as by definition, even a single tumor cell with membranous HER2 staining qualifies the case as HER2-ultralow.
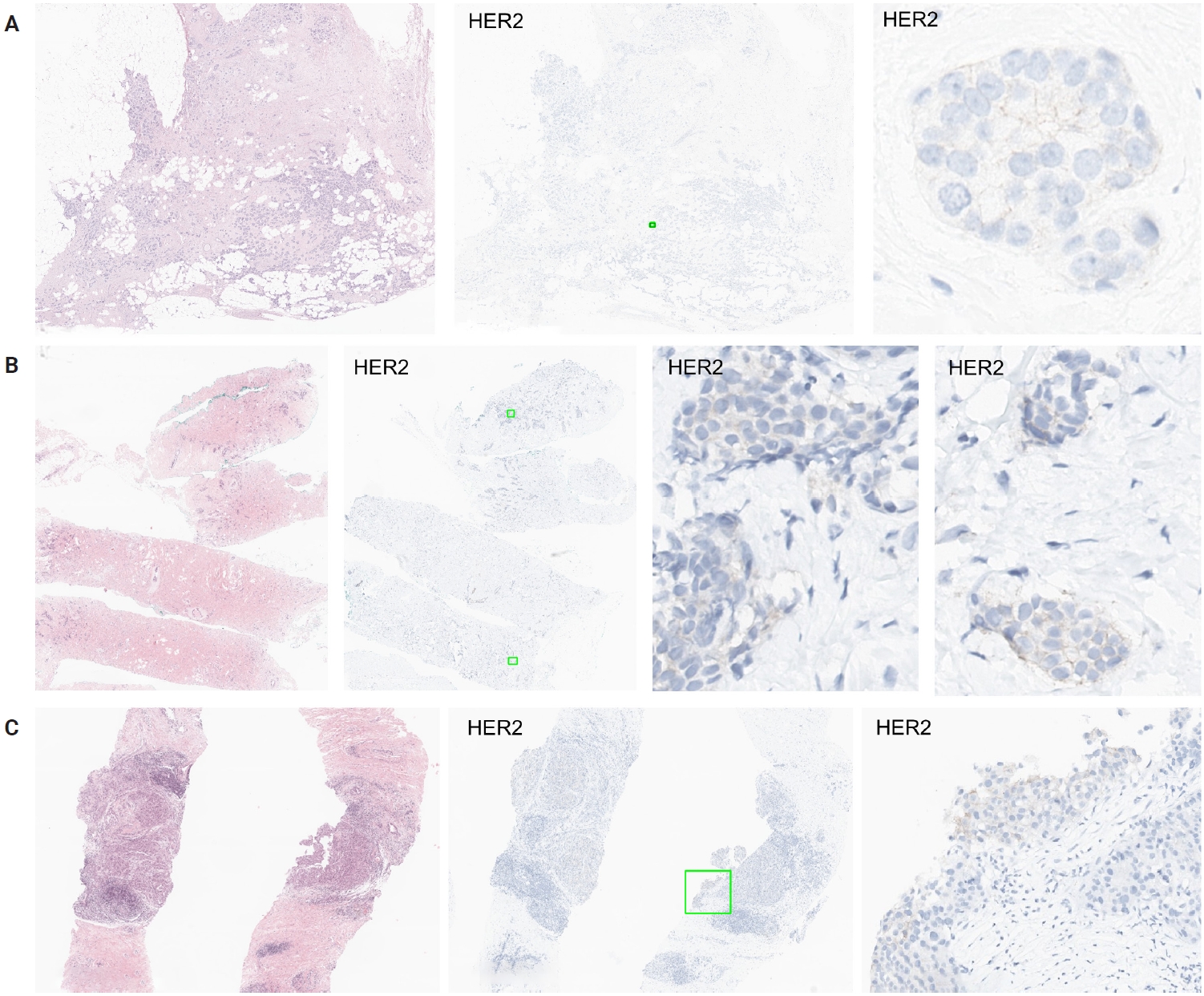
Representative challenging cases in human epidermal growth factor receptor 2 (HER2)–low/ultralow diagnosis. (A) Case 7: Initially diagnosed as HER2-null, but consensus review revealed small foci of faint membranous staining, leading to reclassification as HER2-ultralow. (B) Case 13: Disagreement due to difficulty distinguishing membranous from nonspecific cytoplasmic staining around the 10% cut-off. (C) Case 15: Debate centered on peripheral staining artifacts; careful distinction between true membranous staining and edge artifact is required for accurate classification.
Another case with significant disagreement was caused by the different perceptions of nonspecific cytoplasmic staining. It is well known that valid HER2 staining refers to membranous staining; therefore, cytoplasmic staining should not be interpreted as positive. In this case (case 13) (Fig. 3B), the diagnosis was debatable because some pathologists thought membranous staining was present in more than 10% of tumor cells, while others underestimated the percentage of positive cells as below 10% because they interpreted some stained cells as showing cytoplasmic rather than membranous staining. Coupled with the difficulty of distinguishing membranous from cytoplasmic staining, the semi-quantitative assessment near the 10% cut-off further complicates interpretation.
Next debate on case 15 (Fig. 3C) was related to edge artifacts. Before the emergence of the HER2-ultralow category, peripheral staining like that observed in this case was unequivocally considered HER2-negative, as it was focal and likely attributable to edge artifacts. Given that even a single cell with membranous staining qualifies as HER2-ultralow, it is essential to clarify the approach for identifying and excluding edge artifact to ensure diagnostic accuracy.
Real-world practical challenges
During the consensus discussion, several real-world challenges were identified that impact the consistent and accurate diagnosis of HER2-low and -ultralow BC in routine clinical practice. All participating pathologists emphasized the increasing workload associated with HER2 IHC interpretation, particularly in distinguishing between HER2-null, ultralow, and 1+ categories. The recent clinical emphasis on HER2-low classification has added further complexity, requiring meticulous examination of cases previously deemed HER2-negative.
In addition, some raised concerns about potential medicolegal consequences arising from diagnostic discrepancies, as variations in interpretation may lead to significant therapeutic implications. Participants even expressed concern about potential pressure to avoid classifying any metastatic BC cases as HER2-null, which may lead to diagnostic inflation. In cases where initial results are equivocal or debated, clinicians may request reevaluation of the immunostain or repeated staining. However, in many countries including the Republic of Korea, such reevaluation or repeated testing is not reimbursed, creating pragmatic barriers.
DISCUSSION
The emergence of HER2-low and -ultralow categories has redefined the landscape of HER2-targeted therapy and placed unprecedented emphasis on diagnostic precision in breast pathology. This distinction has become clinically relevant only recently, in parallel with the expanded eligibility for ADCs, particularly following clinical trials demonstrating therapeutic benefit even in tumors with extremely low HER2 expression. Nevertheless, our findings reveal substantial interobserver variability among experienced breast pathologists, with significant disagreement observed in more than half of the examined cases. Notably, all initially classified HER2-null cases were upgraded to ultralow upon consensus review, raising concern that a substantial proportion of HER2-ultralow BCs may be underrecognized in real-world settings. This diagnostic gap has critical therapeutic implications, as patients misclassified as HER2-null may be excluded from potentially beneficial T-DXd treatment.
Our study also highlights key diagnostic caveats, including non-specific background or edge staining, ambiguous membranous patterns, and the inherent difficulty of semi-quantitatively estimating HER2 expression with the cut-off value of 10%. These technical challenges, coupled with the rigid—albeit somewhat arbitrary—classification structure of current HER2 diagnosis workflows, limit reproducibility and raise questions about how HER2 diagnosis can be reliably implemented in real-world settings.
From a practical standpoint, these diagnostic challenges translate into a substantial increase in time required for the identification of HER2-ultralow cases, as it necessitates exhaustive slide review to detect rare tumor cells with extremely weak membranous staining. This level of scrutiny may not be feasible in high-volume routine practice.
The challenge is further compounded by the semi-quantitative nature of the 10% cut-off threshold. In this context, artificial intelligence–based image analysis tools may serve as supportive aids for detecting and quantifying weak or focal membranous staining, potentially improving reproducibility, although further validation is required.
Even with such potential supportive tools, the expert panel raised broader systemic concerns. Heavy diagnostic workloads, potential medicolegal conflicts surrounding borderline calls, and the lack of reimbursement for repeat testing were mentioned as significant barriers. Breast pathologists may also feel pressured to avoid classifying any metastatic BC case as HER2-null, due to concerns that the patient might lose access to a therapeutic option. This may contribute to diagnostic inflation and pose ethical concerns.
Several strategies were also suggested to improve diagnostic concordance. These include the development of training programs focused on HER2-low and -ultralow categories, and the integration of artificial intelligence tools to assist in the identification and quantification of weak or focal staining. Policy-level changes, including reimbursement for re-evaluation in borderline cases, are also urgently needed to support pathologists’ efforts.
More importantly, a fundamental question lies in whether HER2-null truly exists or not, and whether patients with HER2-null BC would not benefit from T-DXd therapy. The former concern is supported by our finding that all initially HER2-null cases were upgraded after the meeting. Similar phenomena have also been repeatedly reported in other groups [6-9,11]. Regarding the efficacy of T-DXd in HER2-null BCs, although limited in number, there are cases with objective response among HER2-null BC patients in the DAISY trial [12]. Furthermore, the observation that response rates in DB-06 did not linearly correlate with HER2 IHC scores [3] suggests that HER2 expression may serve merely as a trial entry marker, rather than a reliable biomarker for treatment response. Therefore, the true existence of HER2-null BCs should be clarified, and a scientific rationale must be established to justify whether maintaining this distinction between HER2-null and others has any meaningful diagnostic or therapeutic value. We hope the ongoing DESTINY-Breast15 trial will provide clearer answers to these questions [13].
In summary, our consensus study highlights the diagnostic complexity and real-world challenges in diagnosing HER2-low and -ultralow BCs. In addition to improving diagnostic accuracy and addressing systemic barriers, clarifying the biological and clinical relevance of the HER2-null category remains a fundamental priority.
Notes
Ethics Statement
This study was approved by the Institutional Review Board (IRB) of SNUH (IRB No. H-2504-142-1635), and formal written informed consent was not required with a waiver by the IRB.
Availability of Data and Material
The datasets generated or analyzed during the study are available from the corresponding author on reasonable request.
Code Availability
Not applicable.
Author Contributions
Conceptualization: GG. Data curation: JK, YJC, EYC, AL, JSK, SYP, MHK, JHJ, GG. Formal analysis: JK. Supervision: GG. Writing—original draft: JK. Writing— review &editing: JK, YJC, EYC, AL, JSK, SYP, MHK, JHJ, GG. Approval of final manuscript: all authors.
Conflicts of Interest
Jiwon Koh has advisory roles for AstraZeneca, Roche, Gilead Korea, and Daiichi Sankyo. S.Y.P., the editor-in-chief of the Journal of Pathology and Translational Medicine, was not involved in the editorial evaluation or decision to publish this article. All remaining authors have declared no conflicts of interest.
Funding Statement
No funding to declare.
Acknowledgments
The authors acknowledge the Breast Pathology Study Group of the Korean Society of Pathologists for its academic support and AstraZeneca Korea for their logistical support in organizing the consensus meeting.