Malignant Fat-Forming Solitary Fibrous Tumor of the Thigh
Article information
Fat-forming solitary fibrous tumor (SFT), previously known as lipomatous hemangiopericytoma, was described as an uncommon variant of extrapleural SFT in the 2013 World Health Organization (WHO) classification of soft tissue tumors.1 Fat-forming SFT is an uncommon, slow-growing neoplasm and is characterized by the typical histologic features of SFT with a mature adipocytic component. The fat-forming variant of SFT exhibits benign biological behavior; however, in rare case, it can have malignant histologic features. Fewer than 20 malignant cases have been reported in the English literature.2-5 Herein, we present a case of fat-forming SFT with malignant histologic features arising in the thigh, in addition to a review of the literature.
CASE REPORT
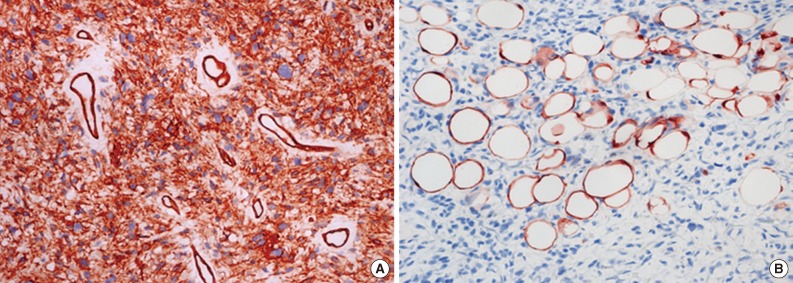
An 84-year-old man visited our hospital complaining of a palpable mass in his left thigh. Eighteen years prior, he underwent radical cystectomy for urothelial carcinoma. Magnetic resonance imaging (MRI) revealed an 11-cm-sized soft tissue mass at the deep aspect of the proximal thigh with infiltration to the surrounding muscle. The tumor exhibited low signal intensity and no obvious intra-tumoral fat component could be identified on T1-weighted MRI (Fig. 1A). The tumor demonstrated high signal intensity on T2-weighted MRI, and showed peripheral enhancement with extensive necrosis and hemorrhage on contrast-enhanced T1-weighted MRI with fat suppression. Wide excision was performed due to the suspicion of malignancy. Grossly, the tumor was relatively well circumscribed and had a yellowish-tan appearance with a glistering surface, extensive necrosis, and cystic degeneration (Fig. 1B). Microscopically, the tumor was encapsulated and had a partially infiltrative margin. The tumor demonstrated a SFT component characterized by a patternless architecture of oval to spindle cells surrounding the ectatic thin-walled branching vessels admixed with mature adipose tissue. The tumor had alternating hypercellular and hypocellular areas with extensive necrosis and hemorrhage. The tumor cells in the cellular area were pleomorphic and had numerous mitotic figures (up to 25 mitoses per 10 high power fields [HPF] and atypical mitosis) (Fig. 2A, B). The adipocytic components were located at the periphery of the tumor within the capsule, and comprised approximately 5% of the tumor. Fat lobules or scattered individual adipocytes were intermingled with pleomorphic stromal cells. Adipocytes had a clear cytoplasm with a flat nucleus located at the periphery of the cells (Fig. 2C, D). No lipoblasts were observed in the adipocytic component. Immunohistochemical staining demonstrated that the neoplastic cells were positive for CD34, CD99, and bcl-2, but were negative for S-100 protein, keratin, epithelial membrane antigen, CD31, desmin, and CD68. Mature adipose tissue between SFT components was positive for S-100 protein (Fig. 3). Based on these histologic and immunohistochemical findings, the tumor was diagnosed as a malignant fat-forming SFT of the thigh. The patient was followed postoperatively for 14 months, and there was no evidence of recurrence or metastasis.
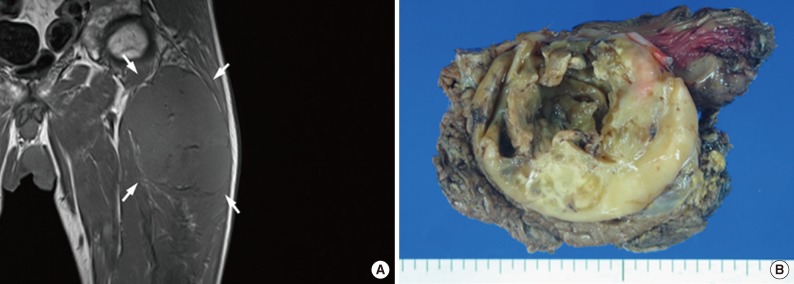
Radiologic and gross findings. (A) The tumor (arrows) exhibits diffuse low signal intensity on T1-weighted magnetic resonance imaging. (B) Grossly, the tumor is relatively well circumscribed, yellowish-tan in color, and shows necrosis.
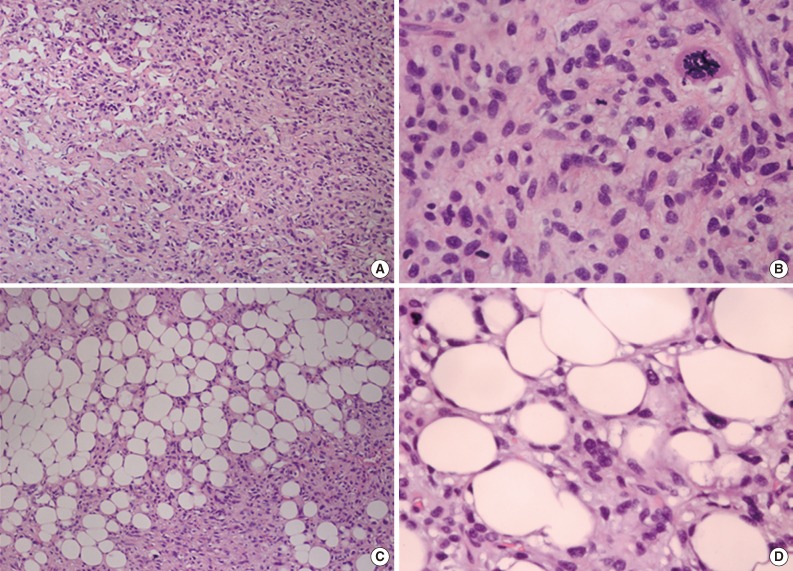
Microscopic findings. (A) Some areas of the tumor demonstrate an ordinary solitary fibrous tumor component, characterized by a patternless architecture of oval to spindle cells surrounding the ectatic thin-walled branching vessels. (B) The cellular area shows pleomorphic tumor cells and atypical mitotic figures. (C) Mature adipose tissue is identified within the tumor. (D) Adipocytes have a flat nucleus, which is located on the periphery of the cell, and are intermingled with atypical plump cells.
DISCUSSION
A SFT with a lipomatous component was initially termed lipomatous hemangiopericytoma as an unusual variant of a hemangiopericytoma. This variant was reclassified as a fat-forming SFT by the WHO in 2013.1 Fat-forming SFTs usually occur in older adults, predominantly in males, and have a broad anatomical distribution.1 Although fat-forming SFTs are typically nonrecurring and nonmetastasizing benign tumors, they can show malignant histologic features and malignant behavior.2-5 Reported cases of malignant fat-forming SFTs revealed that men and women are equally affected, that the age of patients ranged from 20 to 93 years, and that the maximal size of a malignant tumor was 20 cm (range, 3.4 to 20 cm). These tumors commonly involve the thigh and could also affect variable anatomical sites including the calf, neck, scalp, posterior thoracic cavity, ligamentum teres of the liver, arm, pelvis, lower back, vulva, and retroperitoneum.2-5 All of the reported malignant fat-forming SFTs were well emarcated and encapsulated. Tumor necrosis was relatively common; it was observed in eight reported cases. Immunohistochemically, most of the cases were positive for CD34 and/or CD99, and there was only one case of 10 that was negative for both CD34 and CD99.
Histologically, malignant fat-forming SFTs have an ordinary SFT component admixed with variable amounts of mature adipose tissue. The histological features suggestive of malignancy were increased cellularity, pleomorphism, mitotic count over 4 in 10 HPF, hemorrhage, necrosis, and infiltrative margin.1,4 The mitotic count ranged from 2 to 37 in 10 HPF.2-5 However, definitive criteria for malignancy in fat-forming SFTs have yet to be established. In our case, the tumor showed high cellularity, cellular pleomorphism with nuclear atypia, mitoses up to 25/10 HPF, hemorrhage, necrosis, and a partially infiltrative margin. Therefore, we determined this to be a case of malignant fat-forming SFT.
In most reported cases, mature adipocytes comprise the adipocytic component of the tumor. Multivacuolated lipoblasts with hyperchromatic scalloped nuclei or adipocytes with atypical stromal cells mimicking atypical lipomatous tumors could be observed.4 Our case had only mature adipocytic components and atypical lipoblasts were absent. The origin of the adipocyte of fat-forming SFT still remains unclear. Previous studies have suggested that SFT has an adipocytic component because immature pericytes within the tumor undergo the differentiation into adipocytes that normally occurs in embryologic development; thus, the variable adipocytic components, which range from immature, multivacuolated, lipoblast-like cells to mature adipocytes, can appear in SFTs during this differentiation process.6,7
For the diagnosis of a malignant fat-forming SFT, this disease must be differentiated from various soft-tissue sarcomas, particularly dedifferentiated liposarcoma, because it could have atypical multivacuolated lipoblasts.3,4 In such cases, the presence of a typical SFT pattern with dilated branching vessels is helpful for the diagnosis of malignant fat-forming SFT.4
Despite the histological features of malignant fat-forming SFT, its prognosis is not well-understood due to the low incidence and the absence of a large-scale case study. The prognostic index predicting local recurrence or latent metastasis is also yet to be established. In a study on ordinary SFT, positive resection margin, tumor size larger than 10 cm, mitoses of 4/10 HPF, nuclear pleomorphism, high cellularity, necrosis, and the presence of a malignant component have been suggested as prognostic factors for local recurrence and metastasis.8 Although most reported malignant fat-forming SFTs have a favorable prognosis, this disease still has the potential to be fatal. Two patients have died from multi-organ metastasis to the lung, spine, breast, bone, and soft tissue. These two cases showed significant atypical histologic features.4 In our case, the patient has survived without recurrence or metastasis for 14 months postoperatively. However, there remains a high potential for occurrence of late adverse events because the tumor exhibited many adverse pathologic factors, including a size of 11 cm, mitoses up to 25/10 HPF, pleomorphism, high cellularity and necrosis. Therefore, close follow-up is needed in our case.
Acknowledgments
This work was supported by the National Research Foundation of Korea Grant funded by the Korean Government (No. 2012-0009320).
Notes
No potential conflict of interest relevant to this article was reported.