Articles
- Page Path
- HOME > J Pathol Transl Med > Volume 48(3); 2014 > Article
-
Original Article
Extrapulmonary Lymphangioleiomyoma: Clinicopathological Analysis of 4 Cases - Dae Hyun Song, In Ho Choi, Sang Yun Ha, Kang Min Han, Jae Jun Lee, Min Eui Hong, Yoon-La Choi, Kee-Taek Jang, Sang Yong Song, Chin A Yi1, Joungho Han
-
Korean Journal of Pathology 2014;48(3):188-192.
DOI: https://doi.org/10.4132/KoreanJPathol.2014.48.3.188
Published online: June 26, 2014
Department of Pathology, Samsung Medical Center, Sungkyunkwan University School of Medicine, Seoul, Korea.
1Department of Radiology, Samsung Medical Center, Sungkyunkwan University School of Medicine, Seoul, Korea.
- Corresponding Author: Joungho Han, M.D. Department of Pathology, Samsung Medical Center, Sungkyunkwan University School of Medicine, 81 Irwon-ro, Gangnam-gu, Seoul 135-710, Korea. Tel: +82-2-3410-2800, Fax: +82-2-3410-0025, hanjho@skku.edu
© 2014 The Korean Society of Pathologists/The Korean Society for Cytopathology
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/3.0/) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
Abstract
-
Background
- Lymphangioleiomyomatosis (LAM) is a slowly progressive neoplastic disease that predominantly affects females. Usually, LAM affects the lung; it can also affect extrapulmonary sites, such as the mediastinum, the retroperitoneum, or the lymph nodes, although these locations are rare. A localized form of LAM can manifest as extrapulmonary lesions; this form is referred to as extrapulmonary lymphangioleiomyoma (E-LAM). Due to the rare occurrence of E-LAM and its variable, atypical location, E-LAM is often difficult to diagnose. Herein, we report the clinicopathological information from four E-LAM cases, and also review previous articles investigating this disease.
-
Methods
- Four patients with E-LAM were identified at the Samsung Medical Center (Seoul, Korea) from 1995 to 2012. All E-LAM lesions underwent surgical excision.
-
Results
- All patients were females within the age range of 43 to 47 years. Two patients had para-aortic retroperitoneal masses, while the other two patients had pelvic lesions; two out of the four patients also had accompanying pulmonary LAM. In addition, no patient displayed any evidence of tuberous sclerosis. Histologically, two patients exhibited nuclear atypism with cytologic degeneration.
-
Conclusions
- E-LAM should be considered in the differential diagnosis of patients presenting with pelvic or para-aortic masses. We also conclude that further clinical and pathological evaluation is needed in patients with E-LAM and nuclear atypism.
- Four patients with E-LAM were identified at the Samsung Medical Center (Seoul, Korea) from 1995 to 2012. All cases were surgically resected. Complete clinicopathological data, including age, sex, E-LAM location, other clinical conditions, radiologic findings, size, follow-up period, and presence of TS were collected for each patient. Hematoxylin and eosin slides were reviewed to evaluate the histologic features of the E-LAM lesions in each patient. Immunohistochemical staining was also performed, including staining for smooth muscle actin (1:1,000, 1A4, Dako, Glostrup, Denmark), human malanoma black-45 (HMB-45; 1:80, Dako), the progesterone receptor (1:800, 16, Novocastra, Newcastle upon Tyne, UK), and p53 (1:10,000, BP53.12, Invitrogen, Carlsbad, CA, USA).
MATERIALS AND METHODS
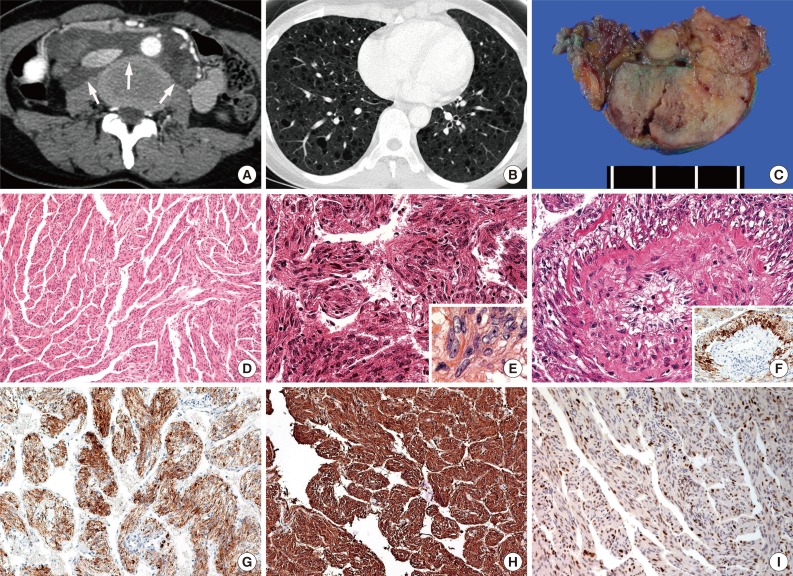
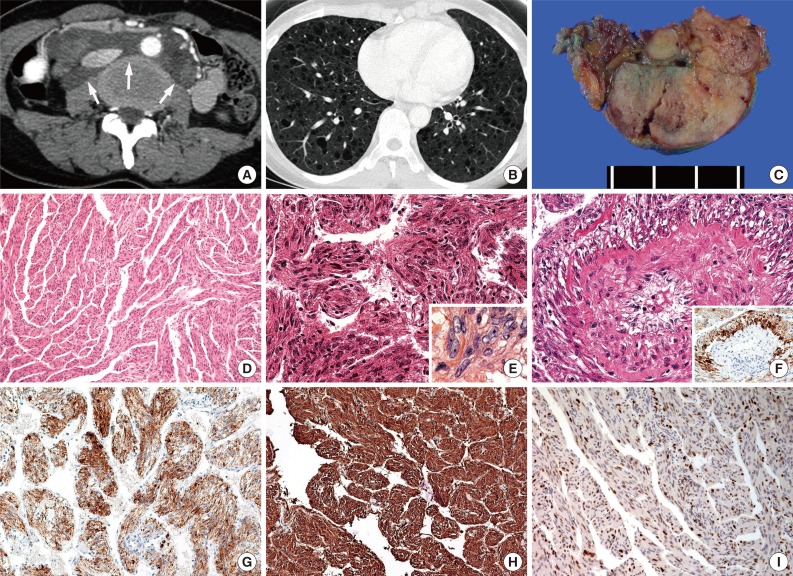
- The clinicoradiological information for each patient is summarized in Table 1. All patients were female, and all were in the age range of 43 to 47 years old. Two patients (cases nos. 2 and 4) exhibited low-attenuated lesions (9 cm and 10 cm in diameter, respectively) in the para-aortic retroperitoneal area as assessed by computed tomography (CT) (Fig. 1A). The radiological conclusions from these scans were lymphangioma and lymphoma, respectively. One patient (case no. 1) exhibited a parametrial mass 6.5 cm in diameter, with a large number of additional lesions displaying an extensive mix of high and low attenuation by CT. In the remaining patient (case no. 3), E-LAM was incidentally detected in several lymph nodes of the right pelvis. This patient exhibited squamous cell carcinoma of the uterine cervix; thus, radical hysterectomy with bilateral pelvic lymph node dissection was performed. Two of the four patients also had accompanying P-LAM. The P-LAM exhibited characteristic radiologic features of well-defined, uniformly thin-walled cysts that were diffusely distributed throughout both lungs (Fig. 1B). Lung biopsies were not performed for these two patients. Furthermore, none of the patients exhibited any evidence of TS. The follow-up periods ranged from a third of a month to 95 months. All patients were alive at the end of follow-up.
- Gross examination of multiple dissections revealed soft, well-circumscribed, ivory and gray solid masses (Fig. 1C). The histopathologic findings were summarized in Table 2. Microscopically, these masses were composed of spindle cells, aggregated lymphocytes, and small vessels. The spindle cells formed anastomosing trabecular bundles with slit-like spaces, similar to a labyrinth (Fig. 1D). Several spindle cell areas showed vaguely palisading patterns. In cases nos. 1 and 4, relatively low cellularity was observed in these areas. The nuclei of the spindle cells exhibited bland-looking nuclear features, such as fine chromatin structures distributed openly and evenly, and inconspicuous nucleoli. In cases nos. 2 and 3, diffuse nuclear crowding was observed at low magnification. The tumor cells exhibited spindle-shaped or round nuclei, with a coarse chromatin pattern. Nuclear membrane wrinkling and conspicuous nucleoli were also identified in the tumor cells (Fig. 1E). These characteristics were also accompanied by cytoplasmic degeneration, with obscure cytoplasmic membranes, and amorphous pink material intermingled between the tumor cells. Perivascular epithelioid E-LAM (PEE) cell proliferation was detected particularly at the intratumoral area of case no. 2 (Fig. 1F). The proliferated PEE cells exhibited round or polygonal-shaped nuclei. The PEE cells were located around the vessels and extended up to the vessel adventitia. A coarse chromatin pattern and conspicuous nucleoli were identified in PEE cells. Case no. 3 did not exhibit any blood vessels near the tumoral lymph node lesions, and PEE proliferation was not observed, although several foci of lymphoid aggregation were observed. Mitosis or necrosis was not seen in any case.
- Immunohistochemical analysis revealed that the spindle-shaped and round E-LAM cells exhibited diffuse staining for HMB-45 (Fig. 1F, G). The PEE cells showed higher reactivity for HMB-45 compared with the surrounding the spindle shaped tumor cells. Furthermore, the spindle cells and the PEE cells showed diffuse staining for both smooth muscle actin and the progesterone receptor (Fig. 1H, I). All samples were negative for p53.
RESULTS
- Lung tumors such as LAM, angiomyolipoma, and clear cell "sugar" tumors belong to the PEComa family.5 The World Health Organization has defined PEComa as "mesenchymal tumors composed of distinctive cells that show a focal association with blood vessel walls and usually express melanocytic and smooth-muscle markers." Perivascular epithelioid cells (PEC) are difficult to detect in normal tissue; thus, the role of PEC in these lung tumors is not yet well-established. The LAM cells in this study showed identical immunohistochemical staining for HMB-45 and smooth muscle actin as for PEC. These LAM cells have been shown to be related to lymphatic smooth muscle cells.6 The majority of LAM cases originate at the lung and manifest as diffuse cystic lesions. Microscopically, P-LAM cells are spindle-shaped or round and exhibit bland-looking nuclei. These cells form either a well-defined cystic wall or randomly distributed, proliferative foci. In contrast to P-LAM, which is manifested by diffuse lesions, E-LAM is manifested by localized lesions. Furthermore, E-LAM tumors are extremely rare, and the precise nature of these tumors is still being investigated.
- Several cases of E-LAM have been reported. Matsui et al.6 described 22 cases of E-LAM, including the relevant clinicopathological information from each case. All 22 patients with E-LAM were female, and their ages ranged from 22 to 67 years old. Moreover, 19 of the 22 patients also exhibited P-LAM. The follow-up periods ranged from 1 to 13 years, and one patient died from P-LAM. The three primary locations of the E-LAM lesions were the posterior mediastinum, the upper retroperitoneum adjacent to the abdominal aorta, and the pelvic cavity. To the best of our knowledge, only two previous reports have described E-LAM cases in Korea.7,8 The first case of E-LAM was reported by Kim et al.7 in 2005, and a second case was reported by Han et al.8 in 2008. The six cases of E-LAM that have been identified in Korea, including the four present cases, are summarized in Table 1. All patients were female, and their ages ranged from 21 to 41 years old. The tumors were present in a sporadic form and were located in the pelvic cavity in four patients and in the para-aortic retroperitoneum in two patients. Three patients also had accompanying P-LAM. The youngest patient died from P-LAM 6 months after the initial operation. Currently, E-LAM is very difficult to diagnose by radiology in the absence of accompanying P-LAM.
- Differential diagnoses performed with radiological techniques should include lymphoma, schwannoma, paraganglioma at the para-aortic area, and metastatic tumors in the lymph nodes. The two present cases with para-aortic masses appeared to be lymphoma and paraganglioma by radiological examinations. Pathologically, determining the extent of immunoreactivity for HMB-45 and smooth muscle actin is helpful in differential diagnosis between schwannoma, leiomyoma, leimyosarcoma, and paraganglioma. The distinctive structural pattern of E-LAM lesions, which consist of an anastomosing trabecular bundle with slit-like spaces similar to a labyrinth, is also a valuable clue in differential diagnosis from PEComa. Using intraoperative frozen biopsies, the discrimination of paraganglioma and metastatic leiomyosarcoma from E-LAM was extremely difficult, and may not actually be possible.
- The majority of E-LAM cases are also accompanied by P-LAM.6 Out of the six cases of E-LAM in Korea, including the four cases in our study, three patients exhibited concurrent P-LAM and E-LAM. It is currently vigorously contested whether E-LAM and/or P-LAM lesions are metastatic or not. Hayashi et al.9 described metastatic potential in the LAM lesions of 10 patients. These authors suggested that the primary site of LAM may be the uterus or the pelvic cavity near the uterus, thus raising the possibility that the P-LAM lesions were metastatic lesions originally derived from the uterus. They introduced PEE cells as involvement of vascular wall of LAM cells. Out of the nine cases of uterine LAM, seven exhibited involvement of the vascular wall; however, intravascular involvement was not detected and endothelial cells were preserved in all cases.9 In our study, case 2 exhibited PEE cells with preserved endothelial cells and no intravascular involvement. And this case of E-LAM was not accompanied by P-LAM. Our study is the first to report the finding that PEE cells exhibit stronger staining for HMB-45 compared with spindle LAM cells. To accurately evaluate the degree of metastasis of LAM lesions, it is vital to determine whether the PEE cells represent an invasion or not. To date, no definitive evidence has been obtained regarding whether LAM lesions exhibit invasion or metastasis.
- Cases 2 and 3 revealed cytologic atypism, compared with cases 1 and 4, which did not. The cases of E-LAM that exhibited cellular atypism (cases 2 and 3) were not accompanied by P-LAM. Considering case 2 showed both PEE and cellular atypism, the cellular atypism of E-LAM may be a valuable feature for determining whether or not the E-LAM is accompanied by PEE cells. Case 3 showed cellular atypism; however, PEE cell was not detected in this case. However, the opportunity to evaluate the presence of PEE in case 3 was somewhat limited, since no medium-sized vascular structures were observed in the intratumoral area. In PEComa, variable degrees of tumor size, infiltration, nuclear grade, cellularity, mitotic activity, necrosis, and vascular invasion are considered to indicate malignancy.10 PEComa with nuclear pleomorphism was considered to have uncertain malignant potential by Folpe et al.10 However, the criteria for nuclear atypism in E-LAM have not yet been defined. Further clinicopathological correlating study including more cases with nuclear atypism, are needed.
- In addition, based on the conclusions of several previous reports,11,12 we immunostained for p53 in the key slides of all our cases in an attempt to identify possible differences between atypical nuclei and bland nuclei. However, none of the slides in our study stained positive for p53.
- In conclusion, here we report four rare cases of E-LAM in our hospital. For females in whom para-aortic or pelvic masses are observed, E-LAM should be considered in the differential diagnosis. Further clinical and pathological evaluation of E-LAM with nuclear atypism is necessary.
DISCUSSION
- 1. McCormack FX, Travis WD, Colby TV, Henske EP, Moss J. Lymphangioleiomyomatosis: calling it what it is: a low-grade, destructive, metastasizing neoplasm. Am J Respir Crit Care Med 2012; 186: 1210-1212. ArticlePubMedPMC
- 2. Meraj R, Wikenheiser-Brokamp KA, Young LR, McCormack FX. Lymphangioleiomyomatosis: new concepts in pathogenesis, diagnosis, and treatment. Semin Respir Crit Care Med 2012; 33: 486-497. ArticlePubMed
- 3. Mavroudi M, Zarogoulidis P, Katsikogiannis N, et al. Lymphangioleiomyomatosis: current and future. J Thorac Dis 2013; 5: 74-79. PubMedPMC
- 4. Kim KW, Seo JB, Lee HJ, Im JG, Kim SH. Renal and pulmonary lymphangioleiomyomatosis: a case report. Eur J Radiol 2000; 36: 126-129. ArticlePubMed
- 5. Chang H, Jung W, Kang Y, Jung WY. Pigmented perivascular epithelioid cell tumor (PEComa) of the kidney: a case report and review of the literature. Korean J Pathol 2012; 46: 499-502. ArticlePubMedPMC
- 6. Matsui K, Tatsuguchi A, Valencia J, et al. Extrapulmonary lymphangioleiomyomatosis (LAM): clinicopathologic features in 22 cases. Hum Pathol 2000; 31: 1242-1248. ArticlePubMed
- 7. Kim HS, Park MI, Suh KS. Lymphangiomyomatosis arising in the pelvic cavity: a case report. J Korean Med Sci 2005; 20: 904-907. ArticlePubMedPMC
- 8. Han JM, Lee KH, Kim SJ, et al. A case of lymphangioleiomyomatosis originated in the pelvic cavity. J Gynecol Oncol 2008; 19: 195-198. ArticlePubMedPMC
- 9. Hayashi T, Kumasaka T, Mitani K, et al. Prevalence of uterine and adnexal involvement in pulmonary lymphangioleiomyomatosis: a clinicopathologic study of 10 patients. Am J Surg Pathol 2011; 35: 1776-1785. PubMed
- 10. Folpe AL, Mentzel T, Lehr HA, Fisher C, Balzer BL, Weiss SW. Perivascular epithelioid cell neoplasms of soft tissue and gynecologic origin: a clinicopathologic study of 26 cases and review of the literature. Am J Surg Pathol 2005; 29: 1558-1575. PubMed
- 11. Hayashi T, Koike K, Kumasaka T, et al. Uterine angiosarcoma associated with lymphangioleiomyomatosis in a patient with tuberous sclerosis complex: an autopsy case report with immunohistochemical and genetic analysis. Hum Pathol 2012; 43: 1777-1784. ArticlePubMed
- 12. Hewedi IH, Radwan NA, Shash LS. Diagnostic value of progesterone receptor and p53 expression in uterine smooth muscle tumors. Diagn Pathol 2012; 7: 1.ArticlePubMedPMCPDF
REFERENCES
| Case No. | Age (yr) | Sex | Location | Size (number) | Radiologic impression | Other condition | TS | FUP (mo) |
|---|---|---|---|---|---|---|---|---|
| 1 | 47 | F | Parametrium | 6.5 (× 1) | E-LAM | P-LAM | No | 95, alive, NER |
| 2 | 46 | F | Retroperitoneum (para-aortic) | 9 (× 1) | Paraganglioma | No | No | 17, alive, NER |
| 3 | 46 | F | Right pelvic lymph node | Up to 0.4 (× 3) | No detection | Sqcc in uterine cervix | No | 19, alive, NER |
| 4 | 43 | F | Retroperitoneum (para-aortic) | 10 (× 1) | Lymphoma | P-LAM | No | 1/3, alive, NER |
| Kim et al. [7] | 21 | F | Pelvic cavity | 9.7 | N/A | P-LAM | No | 6, died for P-LAM |
| Han et al. [8] | 46 | F | Pelvic cavity | 5 | N/A | No | N/A | 6, NER |
Figure & Data
References
Citations

- Surgical Management of Solitary Extrapulmonary Lymphangioleiomyomatosis in the Mesentery: A Case Report
Jack Menzie, Chih C Kuan, Travis Ackermann, Yeng Kwang Tay
Cureus.2024;[Epub] CrossRef - Lymphangioleiomyomatosis with Tuberous Sclerosis Complex—A Case Study
Aleksandra Marciniak, Jolanta Nawrocka-Rutkowska, Agnieszka Brodowska, Andrzej Starczewski, Iwona Szydłowska
Journal of Personalized Medicine.2023; 13(11): 1598. CrossRef - A case of lymphangioleiomyomatosis with endometrial cancer diagnosed by retroperitoneoscopic para-aortic lymph node dissection
Aiko Ogasawara, Shogo Yamaguchi, Hiroaki Inui, Mieko Hanaoka, Daisuke Shintani, Sho Sato, Masanori Yasuda, Akira Yabuno
JAPANESE JOURNAL OF GYNECOLOGIC AND OBSTETRIC ENDOSCOPY.2022; 38(1): 158. CrossRef - Primary retroperitoneal PEComa: an incidental finding
Bárbara Monteiro Marinho, António Gâmboa Canha, Donzília Sousa Silva, José Davide Pinto Silva
BMJ Case Reports.2022; 15(11): e250466. CrossRef - Imaging Findings of Thoracic Lymphatic Abnormalities
Jingshuo (Derek) Sun, Thomas Shum, Fardad Behzadi, Mark M. Hammer
RadioGraphics.2022; 42(5): 1265. CrossRef - Extrapulmonary uterine lymphangioleiomyomatosis (LAM) and dysfunctional uterine bleeding: the first presentation of LAM in a tuberous sclerosis complex patient
Lucy Grant, Saliya Chipwete, San Soo Hoo, Anjali Bhatnagar
BMJ Case Reports.2019; 12(2): e226358. CrossRef - Summary of the Japanese Respiratory Society statement for the treatment of lung cancer with comorbid interstitial pneumonia
Takashi Ogura, Nagio Takigawa, Keisuke Tomii, Kazuma Kishi, Yoshikazu Inoue, Eiki Ichihara, Sakae Homma, Kazuhisa Takahashi, Hiroaki Akamatsu, Satoshi Ikeda, Naohiko Inase, Tae Iwasawa, Yuichiro Ohe, Hiromitsu Ohta, Hiroshi Onishi, Isamu Okamoto, Kazumasa
Respiratory Investigation.2019; 57(6): 512. CrossRef - Incidental lymphangioleiomyomatosis in the lymph nodes of gynecologic surgical specimens
Ikumi Kuno, Hiroshi Yoshida, Hanako Shimizu, Takashi Uehara, Masaya Uno, Mitsuya Ishikawa, Tomoyasu Kato
European Journal of Obstetrics & Gynecology and Reproductive Biology.2018; 231: 93. CrossRef - Solitary extrapulmonary lymphangioleiomyomatosis of the liver: A case report and literature review
Weiwei Fu, Yujun Li, Hong Li, Ping Yang, Xiaoming Xing
Experimental and Therapeutic Medicine.2016; 12(3): 1499. CrossRef - Incidental Pelvic and Para-aortic Lymph Node Lymphangioleiomyomatosis Detected During Surgical Staging of Pelvic Cancer in Women Without Symptomatic Pulmonary Lymphangioleiomyomatosis or Tuberous Sclerosis Complex
Joseph T. Rabban, Brandie Firetag, Ankur R. Sangoi, Miriam D. Post, Charles J. Zaloudek
American Journal of Surgical Pathology.2015; 39(8): 1015. CrossRef

Fig. 1
| Case No. | Age (yr) | Sex | Location | Size (number) | Radiologic impression | Other condition | TS | FUP (mo) |
|---|---|---|---|---|---|---|---|---|
| 1 | 47 | F | Parametrium | 6.5 (× 1) | E-LAM | P-LAM | No | 95, alive, NER |
| 2 | 46 | F | Retroperitoneum (para-aortic) | 9 (× 1) | Paraganglioma | No | No | 17, alive, NER |
| 3 | 46 | F | Right pelvic lymph node | Up to 0.4 (× 3) | No detection | Sqcc in uterine cervix | No | 19, alive, NER |
| 4 | 43 | F | Retroperitoneum (para-aortic) | 10 (× 1) | Lymphoma | P-LAM | No | 1/3, alive, NER |
| Kim et al. [7] | 21 | F | Pelvic cavity | 9.7 | N/A | P-LAM | No | 6, died for P-LAM |
| Han et al. [8] | 46 | F | Pelvic cavity | 5 | N/A | No | N/A | 6, NER |
| Case No. | Nuclear crowding | Nucleoli | Coarse chromatin | Irregularity of nuclear membrane | Cytoplasmic | Perivascular epithelioid E-LAM cells | Slit-like space | Mitosis/Necrosis | HMB-45/SMA |
|---|---|---|---|---|---|---|---|---|---|
| 1 | – | Inconspicuous | – | – | – | – | + | –/– | +/+ |
| 2 | + | Conspicuous | + | + | + | + | + | –/– | +/+ |
| 3 | + | Conspicuous | + | + | + | – | + | –/– | +/+ |
| 4 | – | Inconspicuous | – | – | – | – | + | –/– | +/+ |
TS, tuberous sclerosis, FUP, follow-up period; F, female; N/A, not available; P-LAM, pulmonary lymphangioleioyomatosis; NER, no evidence of recurrence; E-LAM, extrapulmonary lymphangioleimyoma; Sqcc, squamous cell carcinoma.
E-LAM, extrapulmonary lymphangioleiomyoma; HMB-45, human melanoma black-45; SMA, smooth muscle actin.

 E-submission
E-submission
 PubReader
PubReader Cite this Article
Cite this Article