Post-transplant liver biopsies: a concise and practical approach for beginners
Article information
Abstract
Exposure to post-transplant liver biopsies varies among pathology residencies and largely depends on the institution's training program, particularly if the hospital has a liver transplant program. The interpretation of biopsies from transplanted livers presents its own set of challenges, even for those with a solid understanding of non-transplant medical liver biopsies. In this review, we aim to provide a succinct, step-by-step approach to help you interpret liver transplant biopsies. This article may be beneficial for residents interested in liver pathology, gastrointestinal and liver pathology fellows in the early stages of training, clinical gastroenterology and hepatology fellows, hepatologists and general pathologists who are curious about this niche.
INTRODUCTION
Liver transplantation has emerged as a vital therapeutic option for patients with liver failure and/or end-stage liver disease. While it offers significant benefits, it also presents risks and necessitates thorough monitoring to prevent rejection or failure of the transplanted liver. This monitoring is done radiologically and through serial blood liver function tests. The liver function test panel includes alkaline phosphatase, aspartate aminotransferase, alanine aminotransferase, albumin, gamma-glutamyl transferase, bilirubin, prothrombin time, and international normalized ratio. If abnormalities in the liver function tests cannot be explained clinically or radiologically, a biopsy is needed to determine any treatable etiology.
Assessing post-transplant liver biopsies can be challenging because of the wide range of potential pathologies. Breaking down the assessment of these biopsies into six steps may provide a simplified approach, particularly for beginners. In each step, assess the biopsy for one broad category of etiologies and begin building a differential diagnosis list. After the final step, the comprehensive list of possible diagnoses can be further refined based on the clinical scenario.
STEP 1: DE NOVO DISEASES
De novo liver disease refers to the development of new liver pathology that was not present in the liver before transplantation. To assess this category of entities, begin by disregarding the fact that this is a post-transplant biopsy and assess whether the microscopic findings are pathognomonic of any known liver pathology. Employ the same approach you would use for non-transplant medical liver biopsies. Table 1 summarizes the pathognomonic histologic findings of the most commonly encountered liver pathologies. These findings are also illustrated in Figs. 1–3.
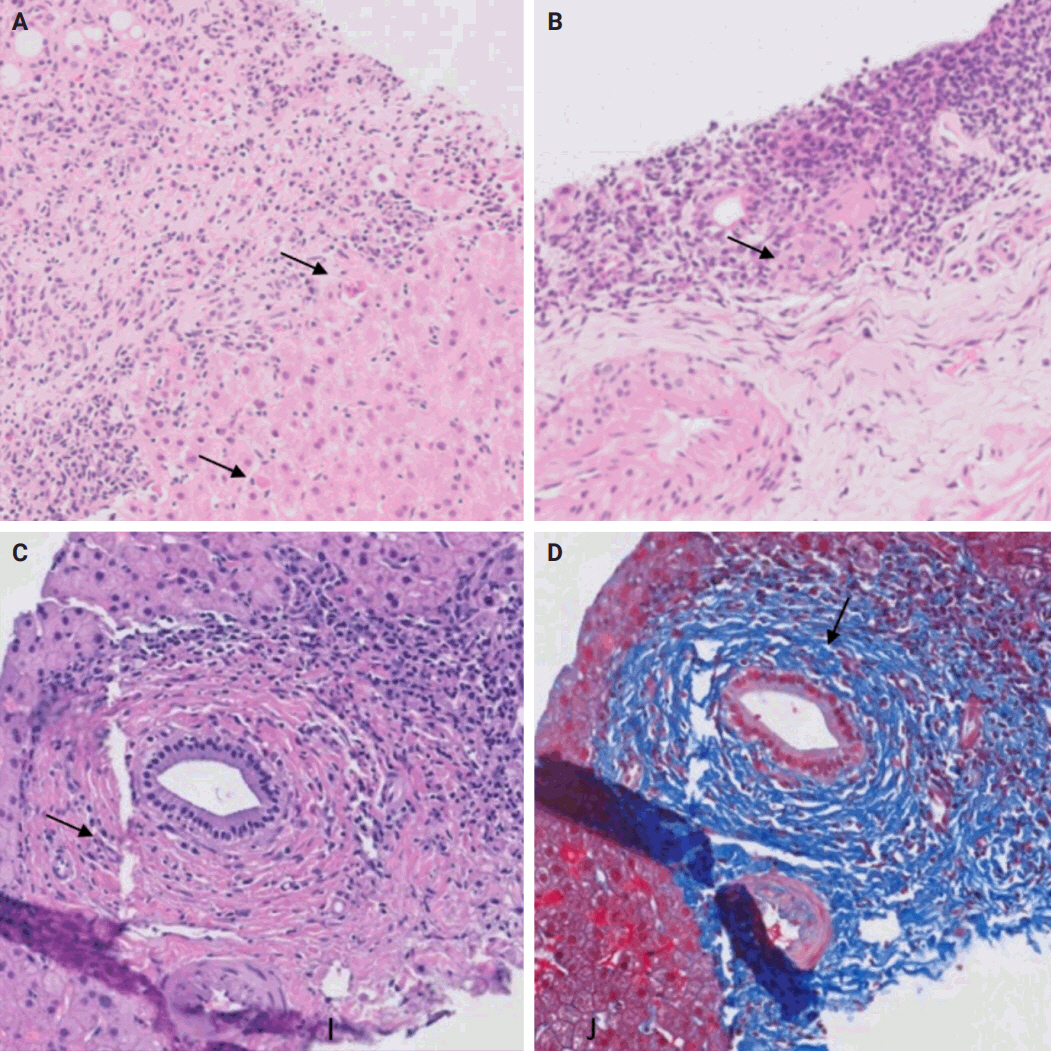
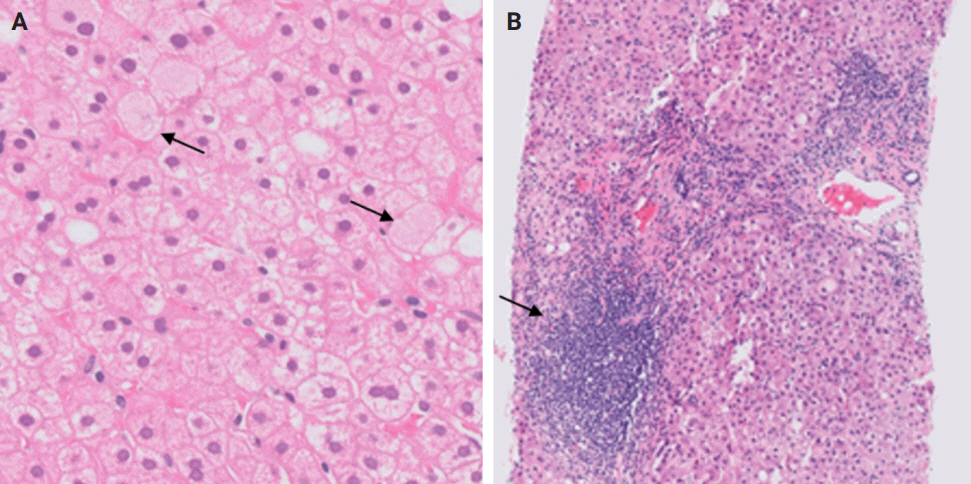
(A) Autoimmune hepatitis: plasma cell-rich infiltrate with periportal apoptotic hepatocytes (arrows). (B) Primary biliary cholangitis: lymphocyte-rich infiltrate with poorly formed granuloma (arrow). (C, D) Primary sclerosing cholangitis: concentric periductal fibrosis (arrow) (D, Trichrome).
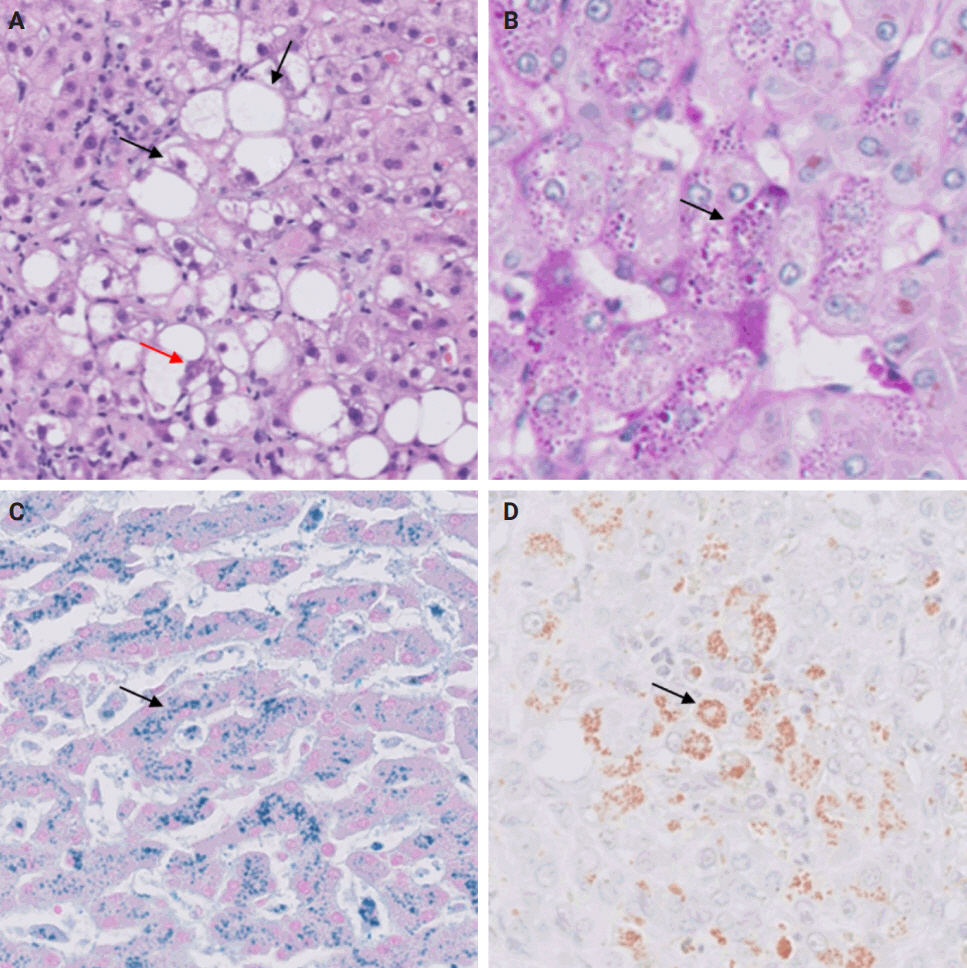
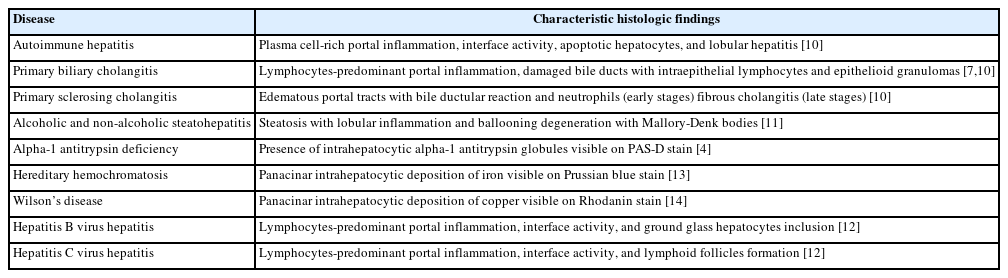
(A) Steatohepatitis: macrovesicular steatosis with ballooned hepatocytes (black arrows) and Mallory-Denk bodies (red arrow). (B) Alpha-1 antitrypsin deficiency: intrahepatocytic alpha-1 antitrypsin globules (arrow) (periodic acid–Schiff–diastase). (C) Hemochromatosis: intrahepatocytic iron accumulation (arrow) (Perl’s stain). (D) Wilson’s disease: intrahepatocytic copper accumulation (arrow) (Rhodanine stain).
STEP 2: RECURRENCE OF PRIMARY LIVER DISEASE
In this step, evaluate the primary liver disease that necessitated the transplant. If the disease is prone to recur, examine the biopsy for its distinct histologic features. Incorporate the possibility of disease recurrence into your growing list of differential diagnoses.
Wilson’s disease and alpha-1 antitrypsin deficiency are two primary liver diseases that typically do not recur following liver transplantation. Wilson’s disease arises from a mutation in the ATP7B gene, responsible for encoding a copper-transporting ATPase pivotal in excreting excess copper from the liver into bile [1]. Liver transplantation is considered curative as the transplanted liver possesses a functional ATP7B gene, facilitating effective regulation of copper metabolism [2]. Alpha-1 antitrypsin deficiency stems from mutations in the SERPINA1 gene, leading to the production of abnormal alpha-1 antitrypsin protein, which accumulates in hepatocytes, causing inflammation and fibrosis [3]. Liver transplantation is curative for liver dysfunction caused by alpha-1 antitrypsin deficiency as the transplanted liver can produce normal alpha-1 antitrypsin protein [4].
Hereditary hemochromatosis, characterized by increased intestinal iron absorption due to a mutation in the HFE gene, poses a different challenge [5]. It is controversial whether hemochromatosis can recur post-liver transplantation, although it is usually curative [5,6]. Additionally, due to their pathophysiology, primary biliary cholangitis, primary sclerosing cholangitis, autoimmune hepatitis, steatohepatitis, and hepatotropic viral hepatitis are also known to recur after liver transplantation [7-12].
STEP 3: REJECTION
In this step, you should assess the possibility of rejection and include it in the list of potential diagnoses. There are two types of rejection that can occur after liver transplantation: T cell–mediated rejection (TCMR) and antibody-mediated rejection (AMR).
TCMR is more commonly encountered and encompasses acute cellular rejection, chronic ductopenic rejection, and chronic rejection with foam cell arteriopathy. Given its prevalence in clinical practice, it is important to highlight the Banff scoring system. This system is commonly employed for both diagnosing and grading acute cellular rejection. It categorizes the typical histopathological findings of acute cellular rejection into three main areas: portal inflammation, bile duct inflammation, and venous endothelial inflammation. These categories are then graded on a scale ranging from none (0) to severe (3). A cumulative score of 0–1 indicates a negative result for acute cellular rejection, while a score of 2–3 is considered indeterminate or borderline. Mild rejection is denoted by a score of 3–4, moderate rejection by a score of 5–7, and severe rejection by a score of 8–9 [15,16].
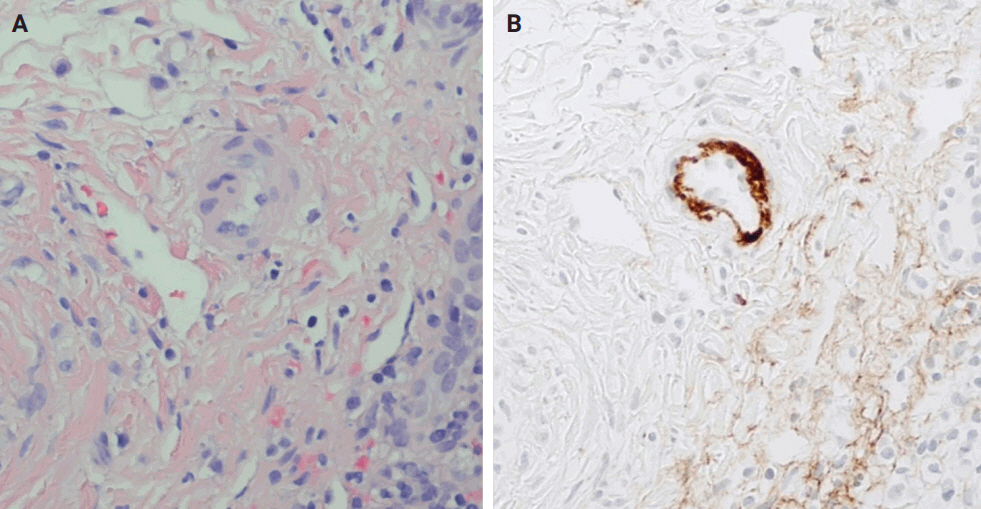
AMR includes hyperacute AMR (almost eliminated due to its rarity), acute AMR (aAMR), and chronic active AMR. Both of the latter two entities are associated with an elevated serologic titer of donor-specific antibodies, which are antibodies specific to the donor's human leukocyte antigens and C4d deposition in portal microvascular endothelium (portal vein and capillaries) (best seen on C4d immunostain) [16,17].
Plasma cell-rich rejection is a mixed TCMR and aAMR which occurs in patients with original disease other than autoimmune hepatitis [16].
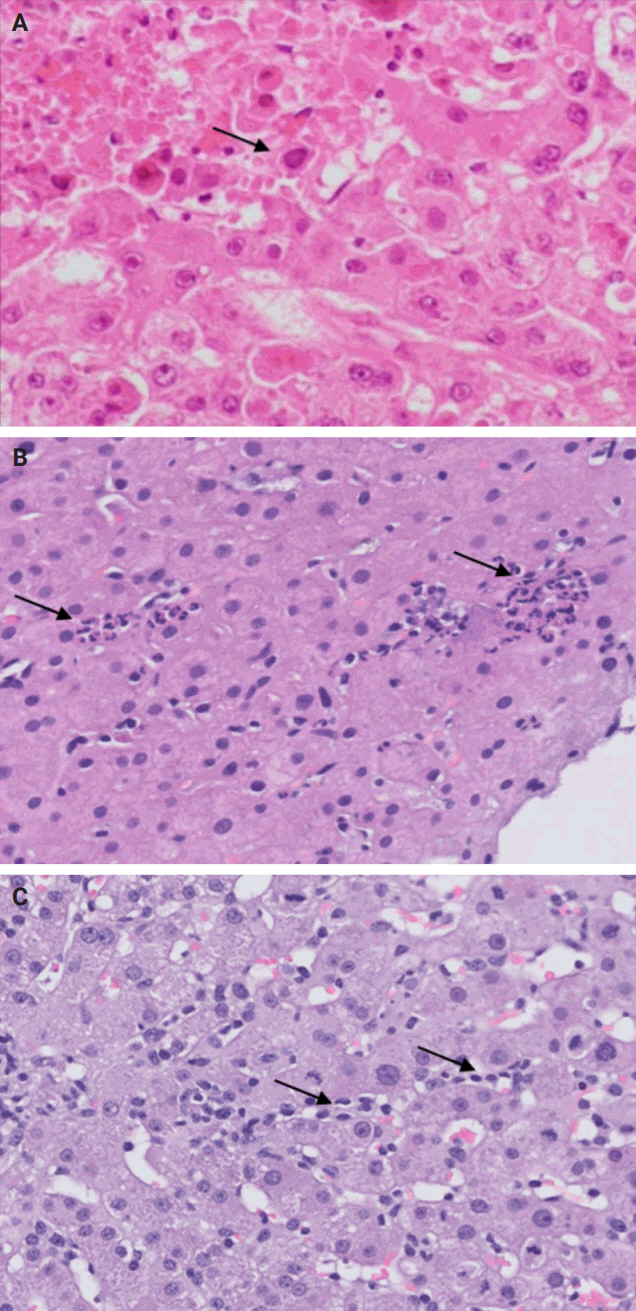
The distinctive histologic features of these rejections are outlined in Table 2. Figs. 4 and 5 highlight the pathognomonic features of each subtype of rejection.
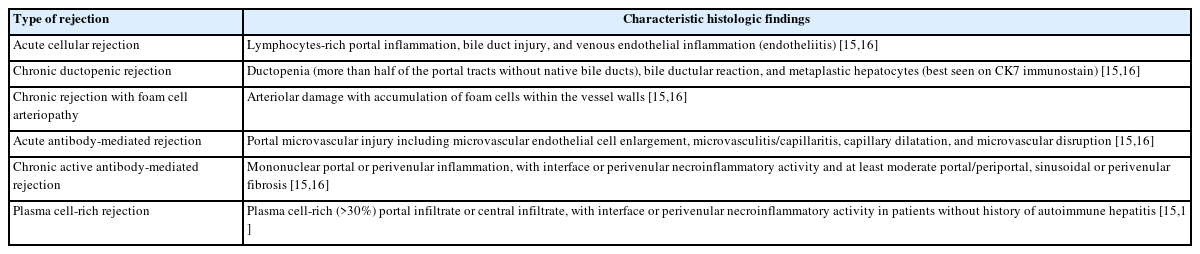
Types of rejection in patients following orthotopic liver transplantation with their characteristic histological findings
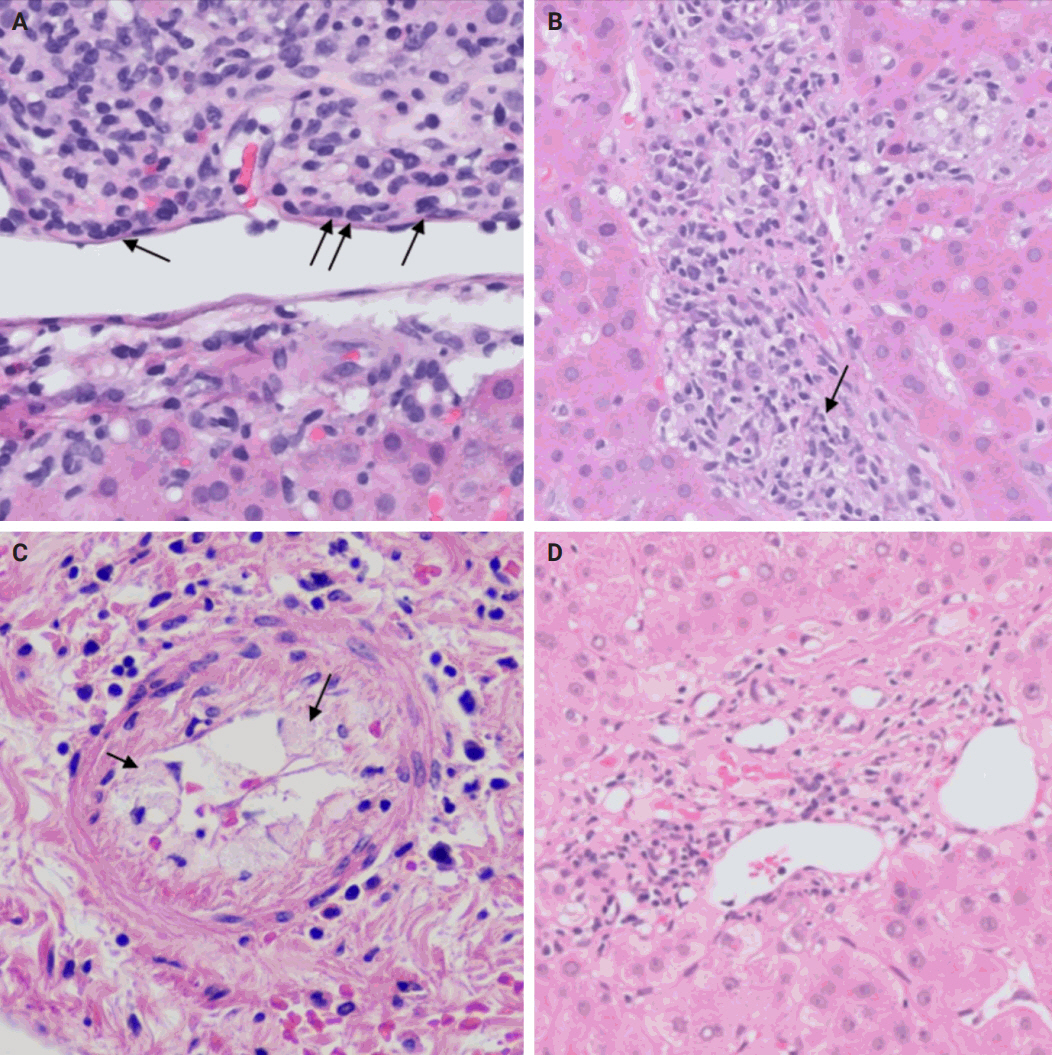
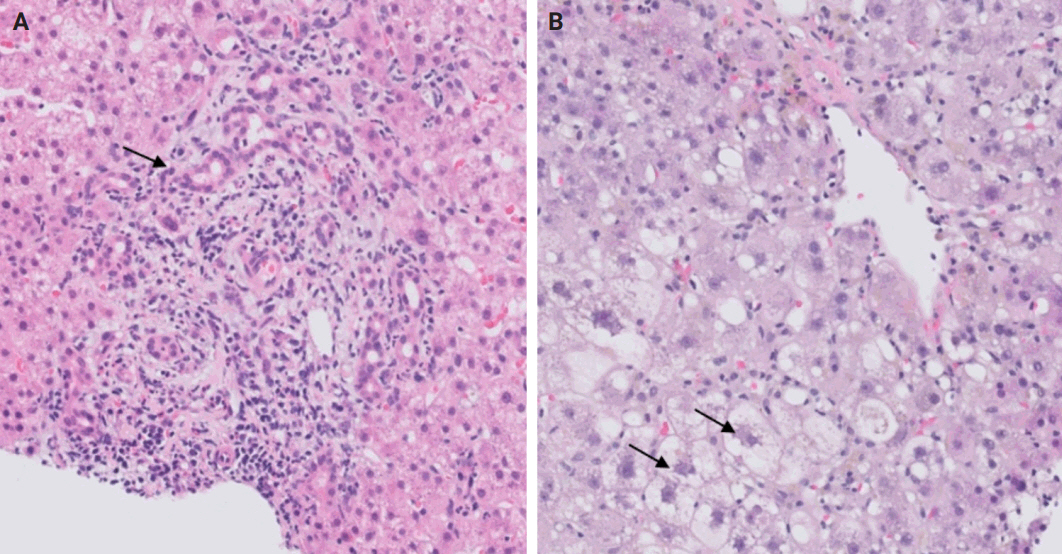
(A) Acute cellular rejection: endothelitis (arrows). (B) Plasma cell-rich rejection: centrilobular plasma cell-rich inflammatory infiltrate (arrow) (Courtesy of Maria Isabel Fiel MD). (C) Chronic rejection with foam cell arteriopathy: foam cell accumulation in the arterial wall (arrows) (Courtesy of Maria Isabel Fiel MD). (D) Chronic ductopenic rejection: absence of native bile duct in a portal tract.
STEP 4: BLOOD AND BILE FLOW OBSTRUCTION
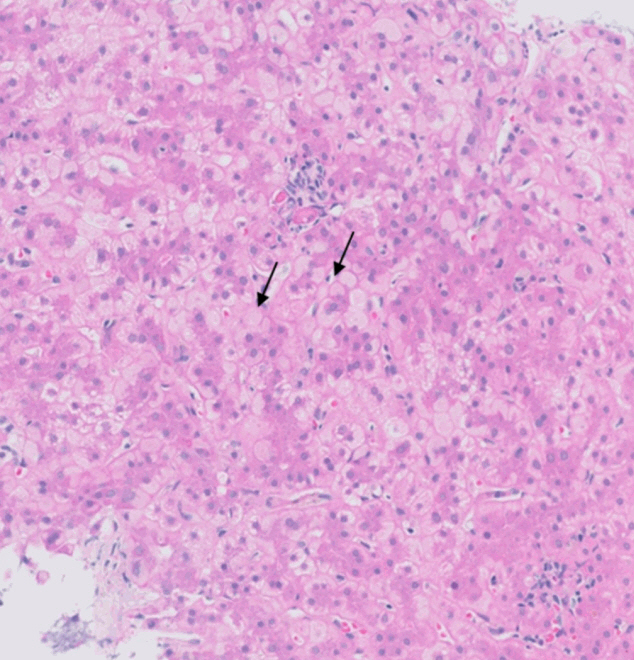
During liver transplantation, precise vascular and biliary anastomoses are crucial for ensuring the proper function and integration of the transplanted liver into the recipient’s body. However, these anastomoses carry the risk of obstruction and failure. Therefore, it is necessary to assess for obstruction or thrombosis of the hepatic artery, hepatic vein, portal vein, and bile ducts when evaluating a post-transplant liver. Table 3 summarizes the histologic features associated with these clinical scenarios. Additionally, the restoration of blood flow to the transplanted liver after ischemia can lead to liver damage known as reperfusion injury [18]. Table 3 also outlines the histologic characteristics of reperfusion injury [19-23]. All the histologic characteristics are also illustrated in Figs. 6 and 7.

Potential complications of orthotopic liver transplantation with their characteristic histological findings
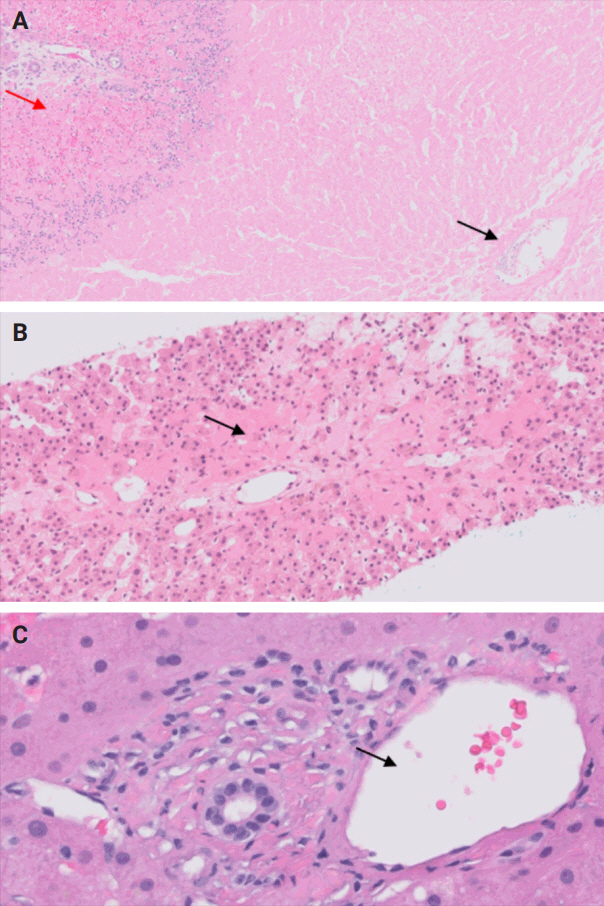
(A) Hepatic artery thrombosis: centrilobular (black arrow) and periportal coagulative necrosis (red arrow). (B) Hepatic vein thrombosis: centrilobular congestion and necrosis (arrow). (C) Portal vein thrombosis: dystrophic (dilated) portal vein (arrow).
STEP 5: OPPORTUNISTIC NON-HEPATOTROPIC VIRAL INFECTIONS
Following liver transplantation, patients receive immunosuppressive therapy, increasing their susceptibility to various opportunistic infections and/or reactivation of dormant infections, such as cytomegalovirus (CMV), Epstein-Barr virus, and other opportunistic pathogens. Bacterial and fungal infections commonly affect organs other than the liver and often do not require biopsy. However, viral infections can directly involve the liver and may necessitate biopsy to distinguish them from rejection. During this step, the focus is on identifying histologic features specific to non-hepatotropic viral infections, which could also be confirmed by immunostaining. Table 4 provides a summary of the histologic characteristics observed in viral infections following liver transplant [24,25]. Some of these features are shown in Fig. 8.
STEP 6: DRUG-INDUCED LIVER INJURY
Following liver transplantation, patients receive immunosuppressive therapy, medical prophylaxis as well as ongoing treatment for existing comorbidities. Each of these medications have varying degrees of likelihood to cause drug-induced liver injury (DILI) and should be considered as potential offending agents when developing your differential diagnosis. However, diagnosing DILI is challenging due to the diverse histopathological patterns observed, which may overlap with patterns seen in other post-transplant diseases. During this step, it's important to review the patient's medication list to identify any drugs that match any pathognomonic histologic findings observed. The inclusion of DILI in the differential diagnosis may also be reasonable if the histologic findings cannot be explained by other steps in this evaluation process.
Keep in mind that patients typically receive prophylactic antimicrobial medications and immunosuppressants in the postoperative period, many of which can induce DILI. In terms of antiviral medications, valganciclovir (Valcyte) is indicated for CMV prophylaxis for the first 3–6 months post-transplantation in all patients [26]. For antibiotic coverage, prophylaxis against Pneumocystis jirovecii pneumonia is typically administered for 12 months after transplantation [26]. Trimethoprim/sulfamethoxazole is the most common medication used; however, atovaquone or dapsone can be used as an alternative in patients with a sulfa allergy [26]. In patients with latent tuberculosis, prophylactic isoniazid is warranted for 9 months duration [26]. Finally, fungal prophylaxis is indicated in low-risk patients for 1 month after transplant but may be used for longer duration in higher risk patients [26]. This is typically accomplished with clotrimazole, fluconazole, or itraconazole [26]. Commonly used medications for immunosuppression and antimicrobial prophylaxis, along with their likelihood of causing DILI, are listed in Table 5.
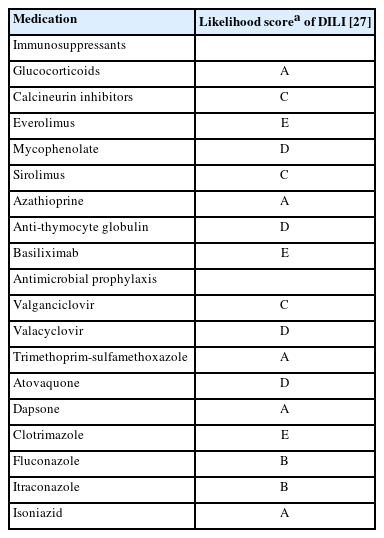
Common immunosuppressants and antimicrobial prophylactic medications with their corresponding likelihood of causing DILI, according to the NIH
A rare manifestation of DILI is the development of pseudo-ground-glass inclusions, which can occur with immunosuppressive therapy. These inclusions can mimic the appearance of hepatitis B inclusions but will be negative for hepatitis B virus surface antigen [28]. These inclusions are highlighted in Fig. 9.
CONCLUSION
Evaluation of post-transplant liver biopsies presents a unique challenge in pathology training, influenced significantly by institutional exposure and the presence of specialized liver transplant programs. By emphasizing key diagnostic considerations such as de novo diseases, disease recurrence, rejection, vascular complications, viral infections, and DILI, our structured, step-by-step approach outlined in this review aims to facilitate a systematic interpretation of these biopsies, bridging the knowledge gap for pathologists at various stages of their careers.
Notes
Ethics Statement
Institutional Review Board approval was waived due to the use of retrospective, de-identified data. This study adhered to the guidelines enacted by the Office of Human Research Protection that is supported by the U.S. Department of Health & Human Services. All information included in this report is de-identified and no personal details that may be used to identify the patient are included in this report.
Availability of Data and Material
Data sharing not applicable to this article as no datasets were generated or analyzed during the study.
Code Availability
Not applicable.
Author Contributions
Conceptualization: TEJ. Investigation: DH, MBO, TEJ, MS. Project administration: TEJ, MBO, DH, MS. Resources: TEJ. Supervision: TEJ. Visualization: TEJ, MBO, DH. Writing—original draft: MBO, DH, MS. Writing—review & editing: TEJ, DH, MBO, MS. Approval of final manuscript: all authors.
Conflicts of Interest
The authors declare that they have no potential conflicts of interest.
Funding Statement
No funding to disclose.
Acknowledgments
Hwa Jeong Lee, MD for contributing a case of herpes simplex virus hepatitis. Maria Isabel Fiel, MD for contributing a case of chronic rejection with foam cell arteriopathy.