Search
- Page Path
- HOME > Search
- The evolving role of TRPS1 in dermatopathology: insights from the past 4 years
- Mokhtar H. Abdelhammed, Woo Cheal Cho
- J Pathol Transl Med. 2026;60(2):129-143. Published online January 29, 2026
- DOI: https://doi.org/10.4132/jptm.2025.11.25
- 3,015 View
- 216 Download
-
 Abstract
Abstract
 PDF
PDF - Over the past 4 years, trichorhinophalangeal syndrome type 1 (TRPS1) has rapidly gained attention among practicing pathologists, with numerous studies emerging that both support and question its diagnostic utility. Initially regarded as a highly specific marker for tumors of mammary origin, TRPS1 is now recognized to have broader expression patterns, including in a variety of cutaneous neoplasms. This is likely due to embryologic parallels between breast tissue and skin adnexal structures, an overlap that was underappreciated in early investigations. Although TRPS1 lacks absolute specificity—even among cutaneous neoplasms—it can still offer meaningful diagnostic value when interpreted alongside conventional immunohistochemical markers and within the appropriate morphologic context. Noteworthy diagnostic applications include mammary Paget disease, primary extramammary Paget disease, rare adnexal neoplasms such as endocrine mucin-producing sweat gland carcinoma and primary cutaneous NUT adnexal carcinoma, and cutaneous metastases from breast carcinoma. In this review, we present the most comprehensive and up-to-date evaluation of the utility and limitations of TRPS1 immunohistochemistry in dermatopathology. Our aim is to deepen understanding of this emerging marker and provide practical guidance on its optimal integration with established immunohistochemical panels to enhance diagnostic accuracy in routine practice.
- Characterization of undifferentiated carcinoma of the salivary gland: clinicopathological and immunohistochemical analyses in comparison with lymphoepithelial carcinoma
- Sangjoon Choi, Gyuheon Choi, Hee Jin Lee, Joon Seon Song, Yoon Se Lee, Seung-Ho Choi, Kyung-Ja Cho
- J Pathol Transl Med. 2025;59(6):361-370. Published online September 8, 2025
- DOI: https://doi.org/10.4132/jptm.2025.07.07
- 3,827 View
- 304 Download
-
 Abstract
Abstract
 PDF
PDF - Background
This study aimed to reclassify a subset of poorly differentiated salivary gland carcinoma that do not conform to any entities of the current World Health Organization (WHO) classification into the category of undifferentiated carcinoma (UDC) because they lack specific histologic differentiation or immunophenotype. Methods: Cases of salivary gland carcinomas from Asan Medical Center (2002–2020) that did not fit any existing WHO classification criteria and were diagnosed as poorly differentiated carcinoma, high-grade carcinoma, or UDC, were retrospectively reviewed. Immunohistochemical (IHC) staining for p40, neuroendocrine markers, androgen receptor (AR), and gross cystic disease fluid protein 15 (GCDFP-15) and Epstein-Barr virus (EBV) in situ hybridization (ISH) were performed. Clinical data were collected from the electronic medical records. Results: Six salivary gland carcinomas did not align with any specific entities and lacked distinct differentiation. Two of six cases displayed lymphoepithelial carcinoma (LEC)-like morphology but were negative or showed negligible immunoreactivity for p40 and EBV ISH, distinguishing them from LEC of the salivary gland. Two cases showed strong AR positivity, suggesting a potential overlap with salivary duct carcinoma (SDC) but lacked classic SDC morphologies and GCDFP-15 expression. No cases expressed neuroendocrine markers. Conclusions: This study proposes reclassifying these poorly differentiated or high-grade salivary gland carcinomas as UDC based on their indeterminate differentiation and IHC profiles. This may lead to a clearer diagnostic category and enhance our understanding of these high-grade tumors.
- Primary Merkel cell carcinoma of the salivary gland: a clinicopathologic study of four cases with a review of literature
- Gyuheon Choi, Joon Seon Song, Hee Jin Lee, Gi Hwan Kim, Young Ho Jung, Yoon Se Lee, Kyung-Ja Cho
- J Pathol Transl Med. 2025;59(3):171-179. Published online April 30, 2025
- DOI: https://doi.org/10.4132/jptm.2025.03.25
- 4,650 View
- 159 Download
- 1 Crossref
-
 Abstract
Abstract
 PDF
PDF - Background
Primary Merkel cell carcinoma of the salivary gland is currently not listed in the World Health Organization classification. However, cases of Merkel cell type neuroendocrine carcinomas of the salivary gland with perinuclear cytokeratin 20 positivity have been intermittently reported. We here investigated the clinicopathologic features of additional cases.
Methods
Data of four cases of Merkel cell type small cell neuroendocrine carcinoma of the salivary gland were retrieved. To confirm the tumors’ primary nature, clinical records and pathologic materials were reviewed. Optimal immunohistochemical staining was performed to support the diagnosis.
Results
All tumors were located in the parotid gland. Possibilities of metastasis were excluded in all cases through a meticulous clinicopathological review. Tumor histology was consistent with the diagnosis of small cell neuroendocrine carcinoma. Tumors’ immunohistochemical phenotypes were consistent with Merkel cell carcinoma, including Merkel cell polyomavirus large T antigen positivity in two of the four cases.
Conclusions
Merkel cell carcinomas can originate in salivary glands and are partly associated with Merkel cell polyomavirus infection as in cutaneous Merkel cell carcinomas. -
Citations
Citations to this article as recorded by- Parotid intranodal metastasis of Merkel cell carcinoma: a rare case report
Tong Gao, Dengshun Wang, Hongwei Yu, Yu’e Wang, Haibin Lu
BMC Oral Health.2025;[Epub] CrossRef
- Parotid intranodal metastasis of Merkel cell carcinoma: a rare case report
- Welcoming the new, revisiting the old: a brief glance at cytopathology reporting systems for lung, pancreas, and thyroid
- Rita Luis, Balamurugan Thirunavukkarasu, Deepali Jain, Sule Canberk
- J Pathol Transl Med. 2024;58(4):165-173. Published online July 15, 2024
- DOI: https://doi.org/10.4132/jptm.2024.06.11
- 5,711 View
- 284 Download
- 1 Web of Science
- 2 Crossref
-
 Abstract
Abstract
 PDF
PDF - This review addresses new reporting systems for lung and pancreatobiliary cytopathology as well as the most recent edition of The Bethesda Reporting System for Thyroid Cytopathology. The review spans past, present, and future aspects within the context of the intricate interplay between traditional morphological assessments and cutting-edge molecular diagnostics. For lung and pancreas, the authors discuss the evolution of reporting systems, emphasizing the bridge between past directives and more recent collaborative efforts of the International Academy of Cytology and the World Health Organization in shaping universal reporting systems. The review offers a brief overview of the structure of these novel systems, highlighting their strengths and pinpointing areas that require further refinement. For thyroid, the authors primarily focus on the third edition of The Bethesda System for Reporting Thyroid Cytopathology, also considering the two preceding editions. This review serves as an invaluable resource for cytopathologists, offering a panoramic view of the evolving landscape of cytopathology reporting and pointing out the integrative role of the cytopathologist in an era of rapid diagnostic and therapeutic advancements.
-
Citations
Citations to this article as recorded by- WHO Reporting System for Lung Cytopathology: Insights Into the Insufficient/Inadequate/Non‐Diagnostic, Atypical and Suspicious for Malignancy Categories and How to Use Them
Zahra Maleki, Sule Canberk, Andrew Field
Cytopathology.2025; 36(5): 434. CrossRef - Reproducibility of the Bethesda system for reporting thyroid cytopathology (TBSRTC): An observational study of 100 patients
Kishori Moni Panda, Reena Naik, Mohd Ghouse Mohiddin
Indian Journal of Pathology and Oncology.2024; 11(4): 385. CrossRef
- WHO Reporting System for Lung Cytopathology: Insights Into the Insufficient/Inadequate/Non‐Diagnostic, Atypical and Suspicious for Malignancy Categories and How to Use Them
- Revisiting the utility of identifying nuclear grooves as unique nuclear changes by an object detector model
- Pedro R. F. Rende, Joel Machado Pires, Kátia Sakimi Nakadaira, Sara Lopes, João Vale, Fabio Hecht, Fabyan E. L. Beltrão, Gabriel J. R. Machado, Edna T. Kimura, Catarina Eloy, Helton E. Ramos
- J Pathol Transl Med. 2024;58(3):117-126. Published online April 30, 2024
- DOI: https://doi.org/10.4132/jptm.2024.03.07
- 5,917 View
- 275 Download
-
 Abstract
Abstract
 PDF
PDF - Background
Among other structures, nuclear grooves are vastly found in papillary thyroid carcinoma (PTC). Considering that the application of artificial intelligence in thyroid cytology has potential for diagnostic routine, our goal was to develop a new supervised convolutional neural network capable of identifying nuclear grooves in Diff-Quik stained whole-slide images (WSI) obtained from thyroid fineneedle aspiration.
Methods
We selected 22 Diff-Quik stained cytological slides with cytological diagnosis of PTC and concordant histological diagnosis. Each of the slides was scanned, forming a WSI. Images that contained the region of interest were obtained, followed by pre-formatting, annotation of the nuclear grooves and data augmentation techniques. The final dataset was divided into training and validation groups in a 7:3 ratio.
Results
This is the first artificial intelligence model based on object detection applied to nuclear structures in thyroid cytopathology. A total of 7,255 images were obtained from 22 WSI, totaling 7,242 annotated nuclear grooves. The best model was obtained after it was submitted 15 times with the train dataset (14th epoch), with 67% true positives, 49.8% for sensitivity and 43.1% for predictive positive value.
Conclusions
The model was able to develop a structure predictor rule, indicating that the application of an artificial intelligence model based on object detection in the identification of nuclear grooves is feasible. Associated with a reduction in interobserver variability and in time per slide, this demonstrates that nuclear evaluation constitutes one of the possibilities for refining the diagnosis through computational models.
- TRPS1 expression in non-melanocytic cutaneous neoplasms: an immunohistochemical analysis of 200 cases
- Yi A. Liu, Phyu P. Aung, Yunyi Wang, Jing Ning, Priyadharsini Nagarajan, Jonathan L. Curry, Carlos A. Torres-Cabala, Doina Ivan, Victor G. Prieto, Qingqing Ding, Woo Cheal Cho
- J Pathol Transl Med. 2024;58(2):72-80. Published online February 26, 2024
- DOI: https://doi.org/10.4132/jptm.2024.01.23
- 8,374 View
- 409 Download
- 15 Web of Science
- 15 Crossref
-
 Abstract
Abstract
 PDF
PDF Supplementary Material
Supplementary Material - Background
Although trichorhinophalangeal syndrome type 1 (TRPS1) was initially thought to be highly sensitive and specific for carcinomas and mesenchymal tumors of mammary origin, more recent data suggest its expression is not limited to breast neoplasms but also can be seen in other cutaneous neoplasms, such as extramammary Paget disease and squamous cell carcinoma (SCC) in situ.
Methods
Two-hundred cases of non-melanocytic cutaneous neoplasm, including basal cell carcinomas (BCCs) (n = 41), SCCs (n = 35), Merkel cell carcinomas (MCCs) (n = 25), and adnexal neoplasms (n = 99), were tested for TRPS1 expression using a monoclonal anti- TRPS1 rabbit anti-human antibody.
Results
TRPS1 expression was present in almost all cases of SCC (94%), with a median H-score of 200, while it was either absent or only focally present in most BCCs (90%), with a median H-score of 5. The difference between BCCs and SCCs in H-score was significant (p < .001). All MCCs (100%) lacked TRPS1 expression. TRPS1 expression was frequently seen in most adnexal neoplasms, benign and malignant, in variable intensity and proportion but was consistently absent in apocrine carcinomas. All endocrine mucin-producing sweat gland carcinomas (EMPSGCs) (100%, 6/6) showed diffuse and strong TRPS1 immunoreactivity, with a median H-score of 300, which was significantly different (p < .001) than that of BCCs.
Conclusions
Our study shows that TRPS1 may be an effective discriminatory marker for BCCs and SCCs. It also has a role in distinguishing BCCs from EMPSGCs. -
Citations
Citations to this article as recorded by- Metastatic Vulvar Paget's Disease Presenting in a Supraclavicular Lymph Node: A Diagnostic Challenge on Fine Needle Aspiration Cytology
Thiri Htoo Aung, Neha Seth, Anam Khan, Kasturi Das
Diagnostic Cytopathology.2026;[Epub] CrossRef - The evolving role of TRPS1 in dermatopathology: insights from the past 4 years
Mokhtar H. Abdelhammed, Woo Cheal Cho
Journal of Pathology and Translational Medicine.2026; 60(2): 129. CrossRef - Correspondence: Primary Cutaneous NUT Adnexal Carcinoma: A Case Report With Novel Clinical and Pathological Observations
Woo Cheal Cho
Journal of Cutaneous Pathology.2026;[Epub] CrossRef - Trichorhinophalangeal syndrome type 1 (TRPS1) in breast pathology: diagnostic utility and pitfalls
Atif Ali Hashmi, Edi Brogi, Hannah Y. Wen
Diagnostic Pathology.2025;[Epub] CrossRef - Refining NTRK Fusion Detection in Papillary Thyroid Carcinoma Through Pan-TRK Immunohistochemistry and Histopathologic Features
Hyun Lee, Sue Youn Kim, Ji Min Park, Seung-Hyun Jung, Ozgur Mete, Chan Kwon Jung
Endocrine Pathology.2025;[Epub] CrossRef - Endocrine mucin-producing sweat gland carcinoma: Case report and literature review
Nan Guo, Zhenlin Fan, Yitong Chen, Qian Li, Limin Guo
European Journal of Ophthalmology.2025;[Epub] CrossRef - Updates on utility of immunohistochemistry in diagnosis of metastatic breast cancer
Hongxia Sun, Aysegul A. Sahin, Qingqing Ding
Human Pathology.2025; 162: 105821. CrossRef - Primary Cutaneous NUT Adnexal Carcinoma With BRD4::NUTM1 Fusion: A 19-Year Follow-Up
Elsayed Ibrahim, Richard K. Yang, Maria A. Gubbiotti, Victor G. Prieto, Woo Cheal Cho
The American Journal of Dermatopathology.2025; 47(9): 731. CrossRef - Primary mucinous carcinoma of the skin with co-expression of TRPS1 and GATA3: a case report
Liling Song, Ning Zhu, Lei Jiang, Dong Gao, Guohua Yu
Frontiers in Oncology.2025;[Epub] CrossRef - Diagnostic Algorithm for Secondary Extramammary Paget Disease from Institutional Cases and Literature Review
Salin Kiratikanon, Ayaka Fukui, Masahiro Hirata, Jakob M. T. Moran, Masakazu Fujimoto, Mai P. Hoang
Cancers.2025; 17(24): 4014. CrossRef - TRPS1 Expression Is Frequently Seen in a Subset of Cutaneous Mesenchymal Neoplasms and Tumors of Uncertain Differentiation: A Potential Diagnostic Pitfall
Moon Joo Kim, Yi A. Liu, Yunyi Wang, Jing Ning, Woo Cheal Cho
Dermatopathology.2024; 11(3): 200. CrossRef - TRPS1 expression in MPNST is correlated with PRC2 inactivation and loss of H3K27me3
Rossana Lazcano, Davis R. Ingram, Gauri Panse, Alexander J. Lazar, Wei-Lien Wang, Jeffrey M. Cloutier
Human Pathology.2024; 151: 105632. CrossRef - Syringocystadenoma Papilliferum-Like Features in Poroma: An Unusual Morphologic Pattern of Poroma or True Synchronous Occurrence of 2 Distinct Neoplasms?
Mouaz Alsawas, Fiorinda F. Muhaj, Phyu P. Aung, Priyadharsini Nagarajan, Woo Cheal Cho
The American Journal of Dermatopathology.2024; 46(12): 871. CrossRef - A Comprehensive Review of TRPS1 as a Diagnostic Immunohistochemical Marker for Primary Breast Carcinoma: Latest Insights and Diagnostic Pitfalls
Antonia-Carmen Georgescu, Tiberiu-Augustin Georgescu, Simona-Alina Duca-Barbu, Lucian Gheorghe Pop, Daniela Oana Toader, Nicolae Suciu, Dragos Cretoiu
Cancers.2024; 16(21): 3568. CrossRef - Expression of TRPS1 in Metastatic Tumors of the Skin: An Immunohistochemical Study of 72 Cases
Kassiani Boulogeorgou, Christos Topalidis, Triantafyllia Koletsa, Georgia Karayannopoulou, Jean Kanitakis
Dermatopathology.2024; 11(4): 293. CrossRef
- Metastatic Vulvar Paget's Disease Presenting in a Supraclavicular Lymph Node: A Diagnostic Challenge on Fine Needle Aspiration Cytology
- Trouble-makers in cytologic interpretation of the uterine cervix
- Eunah Shin, Jaeeun Yu, Soon Won Hong
- J Pathol Transl Med. 2023;57(3):139-146. Published online May 15, 2023
- DOI: https://doi.org/10.4132/jptm.2023.04.25
- 12,598 View
- 483 Download
- 4 Web of Science
- 5 Crossref
-
 Abstract
Abstract
 PDF
PDF - The development and standardization of cytologic screening of the uterine cervix has dramatically decreased the prevalence of squamous cell carcinoma of the uterine cervix. Advances in the understanding of biology of human papillomavirus have contributed to upgrading the histologic diagnosis of the uterine cervix; however, cytologic screening that should triage those that need further management still poses several difficulties in interpretation. Cytologic features of high grade intraepithelial squamous lesion (HSIL) mimics including atrophy, immature metaplasia, and transitional metaplasia, and glandular lesion masquerades including tubal metaplasia and HSIL with glandular involvement are described with accentuation mainly on the differential points. When the cytologic features lie in a gray zone between the differentials, the most important key to the more accurate interpretation is sticking to the very basics of cytology; screening the background and cellular architecture, and then scrutinizing the nuclear and cytoplasmic details.
-
Citations
Citations to this article as recorded by- Cytology–Biopsy Concordance in High-Risk Human Papillomavirus–Positive Women with Abnormal Cytology Findings: Menopause-Stratified Analysis
Isik Sozen, Gozde Sahin, Yuksel Ulu, Dilara Yitiz, Basak Ozge Kayan, Ilkbal Temel Yuksel
Medicina.2026; 62(4): 631. CrossRef - Pathologists Recommend Repeat Pap Testing: In Clinical Practice What Do Gylecologist Do?
Gizem Ay Haldız
Muğla Sıtkı Koçman Üniversitesi Tıp Dergisi.2026; 13(1): 1. CrossRef - Risk of cervical stenosis after cervical excision in postmenopausal patients
Eva Hauge, Line Winther Gustafson, Mette Tranberg, Pinar Bor
European Journal of Obstetrics & Gynecology and Reproductive Biology.2025; 308: 208. CrossRef - Pitfalls in Gynecological Cytology: Review of the Common and Less Frequent Entities in Pap Test
Danijela Vrdoljak-Mozetič, Snježana Štemberger-Papić, Damjana Verša Ostojić, Roberta Rubeša, Marko Klarić, Senija Eminović
Acta Cytologica.2024; 68(3): 281. CrossRef - Cytological features of human papillomavirus‐infected immature squamous metaplastic cells from cervical intraepithelial neoplasia grade 2
Mitsuaki Okodo, Kaori Okayama, Koji Teruya, Ruku Shinohara, Shuichi Mizuno, Rei Settsu, Yasuyoshi Ishii, Masahiko Fujii, Hirokazu Kimura, Mizue Oda
Journal of Medical Virology.2023;[Epub] CrossRef
- Cytology–Biopsy Concordance in High-Risk Human Papillomavirus–Positive Women with Abnormal Cytology Findings: Menopause-Stratified Analysis
- Diagnostic distribution and pitfalls of glandular abnormalities in cervical cytology: a 25-year single-center study
- Jung-A Sung, Ilias P. Nikas, Haeryoung Kim, Han Suk Ryu, Cheol Lee
- J Pathol Transl Med. 2022;56(6):354-360. Published online November 9, 2022
- DOI: https://doi.org/10.4132/jptm.2022.09.05
- 9,639 View
- 162 Download
- 6 Web of Science
- 4 Crossref
-
 Abstract
Abstract
 PDF
PDF - Background
Detection of glandular abnormalities in Papanicolaou (Pap) tests is challenging. This study aimed to review our institute’s experience interpreting such abnormalities, assess cytohistologic concordance, and identify cytomorphologic features associated with malignancy in follow-up histology.
Methods
Patients with cytologically-detected glandular lesions identified in our pathology records from 1995 to 2020 were included in this study.
Results
Of the 683,197 Pap tests performed, 985 (0.144%) exhibited glandular abnormalities, 657 of which had tissue follow-up available. One hundred eighty-eight cases were cytologically interpreted as adenocarcinoma and histologically diagnosed as malignant tumors of various origins. There were 213 cases reported as atypical glandular cells (AGC) and nine cases as adenocarcinoma in cytology, yet they were found to be benign in follow-up histology. In addition, 48 cases diagnosed with AGC and six with adenocarcinoma cytology were found to have cervical squamous lesions in follow-up histology, including four squamous cell carcinomas. Among the cytomorphological features examined, nuclear membrane irregularity, three-dimensional clusters, single-cell pattern, and presence of mitoses were associated with malignant histology in follow-up.
Conclusions
This study showed our institute’s experience detecting glandular abnormalities in cervical cytology over a 25-year period, revealing the difficulty of this task. Nonetheless, the present study indicates that several cytological findings such as membrane irregularity, three-dimensional clusters, single-cell pattern, and evidence of proliferation could help distinguishing malignancy from a benign lesion. -
Citations
Citations to this article as recorded by- “Atypical Glandular Cells” on Cervical Cytology: Correlation Between Glandular Cell Component Volume and Histological Follow‐Up
Havva Gokce Terzioglu, Alessa Aragao, Julieta E. Barroeta
Diagnostic Cytopathology.2026; 54(2): 71. CrossRef - Expertise in Gynecological Pathology Impacts Diagnosis of Atypical Glandular Cell Category in Cervical Cytology
Havva Gökce Terzioglu, Alessa Aragao, Julieta E. Barroeta
Journal of Lower Genital Tract Disease.2025; 29(4): 297. CrossRef - Comparison of Cytological and/or Histopathological Results of Patients with Single and Multiple HPV Positivity
Fatih Mehmet Kaya, Şafak Ersöz, Cihan Comba, Ömer Demir
Acta Cytologica.2025; : 1. CrossRef - Analysis of atypical glandular cells in ThinPrep Pap smear and follow-up histopathology
Tengfei Wang, Yinan Hua, Lina Liu, Bing Leng
Baylor University Medical Center Proceedings.2024; 37(3): 403. CrossRef
- “Atypical Glandular Cells” on Cervical Cytology: Correlation Between Glandular Cell Component Volume and Histological Follow‐Up
- Adrenal hemangioblastoma
- Joo-Yeon Koo, Kyung-Hwa Lee, Joon Hyuk Choi, Ho Seok Chung, Chan Choi
- J Pathol Transl Med. 2022;56(3):161-166. Published online February 28, 2022
- DOI: https://doi.org/10.4132/jptm.2021.12.28
- 6,186 View
- 159 Download
- 1 Web of Science
- 1 Crossref
-
 Abstract
Abstract
 PDF
PDF - Hemangioblastoma (HB) is a rare benign tumor that most commonly occurs in the cerebellum. HB is composed of neoplastic stromal cells and abundant small vessels. However, the exact origin of stromal cells is controversial. Extraneural HBs have been reported in a small series, and peripheral HBs arising in the adrenal gland are extremely rare. Herein, we report a case of sporadic adrenal HB in a 54-year-old woman. The tumor was a well-circumscribed, yellow mass measuring 4.2 cm in diameter. Histologically, the tumor was composed of small blood vessels and vacuolated stromal cells with clear cytoplasm. On immunohistochemical stain, the stromal cells were positive for S-100 protein, neuron-specific enolase, and synaptophysin. The tumor did not reveal mutation of VHL alleles. We herein present a case of HB of the adrenal gland and review of the literature.
-
Citations
Citations to this article as recorded by- Familial Von Hippel–Lindau Disease: A Case Series of Cerebral Hemangioblastomas with MRI, Histopathological, and Genetic Correlations
Claudiu Matei, Ioana Boeras, Dan Orga Dumitriu, Cosmin Mutu, Adriana Popescu, Mihai Gabriel Cucu, Alexandru Calotă-Dobrescu, Bogdan Fetica, Diter Atasie
Life.2025; 15(11): 1649. CrossRef
- Familial Von Hippel–Lindau Disease: A Case Series of Cerebral Hemangioblastomas with MRI, Histopathological, and Genetic Correlations
- Clinicopathologic implication of PD-L1 gene alteration in primary adrenal diffuse large B cell lymphoma
- Ki Rim Lee, Jiwon Koh, Yoon Kyung Jeon, Hyun Jung Kwon, Jeong-Ok Lee, Jin Ho Paik
- J Pathol Transl Med. 2022;56(1):32-39. Published online November 16, 2021
- DOI: https://doi.org/10.4132/jptm.2021.10.05
- 6,082 View
- 170 Download
- 2 Web of Science
- 1 Crossref
-
 Abstract
Abstract
 PDF
PDF - Background
Primary adrenal (PA) diffuse large B cell lymphoma (DLBCL) was previously reported as an aggressive subset of DLBCL, but its genetic features were not sufficiently characterized. From our previous study of DLBCL with programmed death-ligand 1 (PD-L1) gene alterations, we focused on PD-L1 gene alterations in PA-DLBCL with clinicopathologic implications.
Methods
We performed fluorescence in situ hybridization for PD-L1 gene translocation and amplification in PA-DLBCL (n = 18) and comparatively analyzed clinicopathologic characteristics with systemic non-adrenal (NA)-DLBCL (n = 90).
Results
PA-DLBCL harbored distinctive features (vs. NADLBCL), including high international prognostic index score (3–5) (72% [13/18] vs. 38% [34/90], p = .007), poor Eastern Cooperative Oncology Group performance score (≥ 2) (47% [7/15] vs. 11% [10/90], p = .003), elevated serum lactate dehydrogenase (LDH) (78% [14/18] vs. 51% [44/87], p = .035) and MUM1 expression (87% [13/15] vs. 60% [54/90], p = .047). Moreover, PA-DLBCL showed frequent PD-L1 gene alterations (vs. NA-DLBCL) (39% [7/18] vs. 6% [5/86], p = .001), including translocation (22% [4/18] vs. 3% [3/87], p = .016) and amplification (17% [3/18] vs. 2% [2/87], p = .034). Within the PA-DLBCL group, PD-L1 gene–altered cases (vs. non-altered cases) tended to have B symptoms (p = .145) and elevated LDH (p = .119) but less frequent bulky disease (≥ 10 cm) (p = .119). In the survival analysis, PA-DLBCL had a poor prognosis for overall survival (OS) and progression-free survival (PFS) (vs. NA-DLBCL; p = .014 and p = .004). Within the PA-DLBCL group, PD-L1 translocation was associated with shorter OS and PFS (p < .001 and p = .012).
Conclusions
PA-DLBCL is a clinically aggressive and distinct subset of DLBCL with frequent PD-L1 gene alterations. PD-L1 gene translocation was associated with poor prognosis in PA-DLBCL. -
Citations
Citations to this article as recorded by- Case Report: Diagnostic value of spectral CT in primary adrenal lymphoma
Xiang Zhuang, Xi xi Jin, Li wen Feng, Hui Zhang
Frontiers in Oncology.2026;[Epub] CrossRef
- Case Report: Diagnostic value of spectral CT in primary adrenal lymphoma
- Adenocarcinoma of the minor salivary gland with concurrent MAML2 and EWSR1 alterations
- Sangjoon Choi, Junhun Cho, Seung Eun Lee, Chung-Hwan Baek, Yi-Kyung Kim, Hyung-Jin Kim, Young Hyeh Ko
- J Pathol Transl Med. 2021;55(2):132-138. Published online January 22, 2021
- DOI: https://doi.org/10.4132/jptm.2020.12.11
- 8,142 View
- 130 Download
- 13 Web of Science
- 9 Crossref
-
 Abstract
Abstract
 PDF
PDF - Salivary gland tumors are histologically diverse, and each entity has distinctive histopathological and molecular features. We report two cases of salivary gland tumors with unique histological and molecular findings, which have not been documented previously. The tumors were located in the base of the tongue in both patients. Most tumor cells were arranged in cords and nests, giving a trabecularlike appearance. Focally, glandular structures with intraluminal mucin and perivascular pseudorosette-like configurations were identified. Tumor cells had eosinophilic to clear cytoplasm, and showed mild nuclear atypia. They were positive for pancytokeratin and negative for S-100, p63, c-KIT, androgen receptor, and neuroendocrine markers. Multiple foci of capsular or lymphovascular invasion were identified, but the Ki-67 labeling index was low (< 5%). Fluorescence in situ hybridization revealed concurrent alterations of MAML2 and EWSR1 gene. Further investigations with a larger number of cases with similar histological and molecular features will accurately classify this tumor.
-
Citations
Citations to this article as recorded by- Diagnostic and Prognostic Utility of MAML2 Gene Rearrangement in Mucoepidermoid Carcinoma of Salivary Glands and Its Correlation with Histologic Grading Systems
Akshara Ravichandran, Daphne Fonseca, Sahithi Shilpa Arya, G. Sandhya Devi, Suseela Kodandapani, Chandrasekhara Rao, T. Subramanyeshwar Rao
Indian Journal of Otolaryngology and Head & Neck Surgery.2026; 78(3): 1506. CrossRef - A novel fusion of EWSR1::PRKD1 in cribriform adenocarcinoma of salivary glands: A rare case report
Zecra Yahia
Human Pathology Reports.2025; 42: 300801. CrossRef - Salivary Gland Neoplasms With a Unique Trabecular Histology and MAML2 Translocation
Bokyung Ahn, Seung-Ho Choi, Doeun Kim, Deokhoon Kim, Kyung-Ja Cho
American Journal of Surgical Pathology.2023; 47(10): 1085. CrossRef - Mesonephric-like Adenocarcinoma of the Ovary: Clinicopathological and Molecular Characteristics
Hyun Hee Koh, Eunhyang Park, Hyun-Soo Kim
Diagnostics.2022; 12(2): 326. CrossRef - The evolving role of molecular pathology in the diagnosis of salivary gland tumours with potential pitfalls
Kanwalpreet Kaur, Shailee Mehta, Sangita Vanik, Priti Trivedi, Nirmalya Banerjee, Harsh Dhar, Sourav Datta, Subhadeep Karanjai
European Archives of Oto-Rhino-Laryngology.2022; 279(8): 3769. CrossRef - Alveolar Soft Part Sarcoma of the Uterus: Clinicopathological and Molecular Characteristics
Yurimi Lee, Kiyong Na, Ha Young Woo, Hyun-Soo Kim
Diagnostics.2022; 12(5): 1102. CrossRef - Endometrioid Carcinomas of the Ovaries and Endometrium Involving Endocervical Polyps: Comprehensive Clinicopathological Analyses
Jihee Sohn, Yurimi Lee, Hyun-Soo Kim
Diagnostics.2022; 12(10): 2339. CrossRef - Mesonephric-like Differentiation of Endometrial Endometrioid Carcinoma: Clinicopathological and Molecular Characteristics Distinct from Those of Uterine Mesonephric-like Adenocarcinoma
Sujin Park, Go Eun Bae, Jiyoung Kim, Hyun-Soo Kim
Diagnostics.2021; 11(8): 1450. CrossRef - Mesonephric-like Adenocarcinoma of the Uterine Corpus: Comprehensive Immunohistochemical Analyses Using Markers for Mesonephric, Endometrioid and Serous Tumors
Hyunjin Kim, Kiyong Na, Go Eun Bae, Hyun-Soo Kim
Diagnostics.2021; 11(11): 2042. CrossRef
- Diagnostic and Prognostic Utility of MAML2 Gene Rearrangement in Mucoepidermoid Carcinoma of Salivary Glands and Its Correlation with Histologic Grading Systems
- Primary squamous cell carcinoma of the salivary gland: immunohistochemical analysis and comparison with metastatic squamous cell carcinoma
- Uiree Jo, Joon Seon Song, Seung-Ho Choi, Soon Yuhl Nam, Sang Yoon Kim, Kyung-Ja Cho
- J Pathol Transl Med. 2020;54(6):489-496. Published online August 31, 2020
- DOI: https://doi.org/10.4132/jptm.2020.07.19
- 11,255 View
- 211 Download
- 18 Web of Science
- 19 Crossref
-
 Abstract
Abstract
 PDF
PDF Supplementary Material
Supplementary Material - Background
Primary squamous cell carcinoma (SCC) of the salivary gland is a rare disease, and distinguishing primary SCC from metastatic SCC is difficult. This study investigated the histological and immunohistochemical differences between primary and metastatic salivary gland SCC to improve the accuracy of diagnosis and to explore the pathogenesis of this disease.
Methods
Data of 16 patients who underwent surgery for SCC of salivary glands between 2000 and 2018 at Asan Medical Center were retrieved. Eight patients had a history of SCC at other sites, and eight patients had only salivary gland SCC. Immunostaining for p16, p53, androgen receptor (AR), gross cystic disease fluid protein 15 (GCDFP-15), and c-erbB2, as well as mucicarmine staining, were compared between the two groups.
Results
Most tumors were located in the center of the salivary glands with extraparenchymal extension. The histology of primary SCC of the salivary gland was consistent with moderately differentiated SCC with extensive desmoplastic reaction and peritumoral inflammation. Involvement of the salivary gland ducts and transition into the ductal epithelium were observed in two cases. Metastatic SCC resembled the primary tumor histologically and was associated with central necrosis. Both groups exhibited negative mucin staining. Two, one, and one primary SCC case exhibited AR, GCDFP-15, and c-erbB2 positivity, respectively.
Conclusions
A subset of primary SCCs originated in salivary ducts or was related to salivary duct carcinoma. Distinguishing primary from metastatic SCC of the salivary gland is difficult using histologic features and immunoprofiles. A comprehensive review of the medical history is essential. -
Citations
Citations to this article as recorded by- Solitary Parotid Metastasis as an Atypical Site of Recurrence in Lung Squamous Cell Carcinoma: A Diagnostic Difficulty
Mamadou Alpha Prateaux, Mohamed Amine Haouane, Issam Rharrassi, Mohamed Amine Azami
Cureus.2026;[Epub] CrossRef - Clinical diagnosis, treatment, and survival analysis of 61 cases of salivary duct carcinoma: a retrospective study
Shubin Dong, Mengru Li, Zhiwei Zhang, Bowei Feng, Wei Ding, Jiang Chang, Feng Liu
PeerJ.2025; 13: e19626. CrossRef - Characterization of undifferentiated carcinoma of the salivary gland: clinicopathological and immunohistochemical analyses in comparison with lymphoepithelial carcinoma
Sangjoon Choi, Gyuheon Choi, Hee Jin Lee, Joon Seon Song, Yoon Se Lee, Seung-Ho Choi, Kyung-Ja Cho
Journal of Pathology and Translational Medicine.2025; 59(6): 361. CrossRef - Primary salivary gland squamous cell carcinoma with sialolithiasis in the submandibular gland: A case report and literature review
Sawako Ono, Katsutoshi Hirose, Yuji Hirata, Marie Yamada, Satoko Nakamura, Hidetaka Yamamoto
Journal of Oral and Maxillofacial Surgery, Medicine, and Pathology.2024; 36(5): 768. CrossRef - A case of primary squamous cell carcinoma of the parotid gland and review of the literature
Jingli Zhao, Xinrong Nan, Chuhuan Zhou, Nan Jiang, Liangliang Tian
Journal of Case Reports and Images in Oncology.2024; 10(1): 7. CrossRef - Metastatic cutaneous squamous cell carcinoma accounts for nearly all squamous cell carcinomas of the parotid gland
Patrick J. Bradley, Göran Stenman, Lester D. R. Thompson, Alena Skálová, Roderick H. W. Simpson, Pieter J. Slootweg, Alessandro Franchi, Nina Zidar, Alfons Nadal, Henrik Hellquist, Michelle D. Williams, Ilmo Leivo, Abbas Agaimy, Alfio Ferlito
Virchows Archiv.2024; 485(1): 3. CrossRef - Common skin cancers and their association with other non-cutaneous primary malignancies: a review of the literature
Lindsay Holic
Medical Oncology.2024;[Epub] CrossRef - Salivary duct carcinoma with squamous differentiation: histomorphological and immunophenotypical analysis of six cases
Melad N Dababneh, Christopher C Griffith, Kelly R Magliocca, Ivan J Stojanov
Histopathology.2024; 85(4): 590. CrossRef - Comprehensive Next Generation Sequencing Reveals that Purported Primary Squamous Cell Carcinomas of the Parotid Gland are Genetically Heterogeneous
Justin A. Bishop, Masato Nakaguro, Ilan Weinreb, Doreen Palsgrove, Lisa M. Rooper, Travis W. Vandergriff, Brian Carlile, Jeffrey A. Sorelle, Jeffrey Gagan, Toshitaka Nagao
Head and Neck Pathology.2024;[Epub] CrossRef - Salivary gland fine needle aspiration: a focus on diagnostic challenges and tips for achieving an accurate diagnosis
Carla Saoud, Hansen Lam, Sandra I. Sanchez, Zahra Maleki
Diagnostic Histopathology.2023; 29(8): 357. CrossRef - Salivary gland pathologies: evolution in classification and association with unique genetic alterations
Michał Żurek, Łukasz Fus, Kazimierz Niemczyk, Anna Rzepakowska
European Archives of Oto-Rhino-Laryngology.2023; 280(11): 4739. CrossRef - A retrospective study of nonneoplastic and neoplastic disorders of the salivary glands
Sorin Vamesu, Oana Andreea Ursica, Ana Maria Gurita, Raluca Ioana Voda, Mariana Deacu, Mariana Aschie, Madalina Bosoteanu, Georgeta Camelia Cozaru, Anca Florentina Mitroi, Cristian Ionut Orasanu
Medicine.2023; 102(42): e35751. CrossRef - Pembrolizumab as a first line therapy in a patient with extensive mucoepidermoid salivary gland carcinoma. A complete clinical, radiological and pathological response. A very specific case
Raed Farhat, Noam Asna, Yaniv Avraham, Ashraf Khater, Majd Asakla, Alaa Safia, Sergio Szvalb, Nidal Elkhatib, Shlomo Merchavy
Discover Oncology.2022;[Epub] CrossRef - Morphologic CT and MRI features of primary parotid squamous cell carcinoma and its predictive factors for differential diagnosis with mucoepidermoid carcinoma
Xiaohua Ban, Huijun Hu, Yue Li, Lingjie Yang, Yu Wang, Rong Zhang, Chuanmiao Xie, Cuiping Zhou, Xiaohui Duan
Insights into Imaging.2022;[Epub] CrossRef - A Rare Case of Primary Squamous Cell Carcinoma of the Submandibular Salivary Gland: Brief Overview of Diagnostic Ambiguity and Treatment Challenges
Pawan Hingnikar, Anendd Jadhav, Nitin D Bhola
Cureus.2022;[Epub] CrossRef - Necrotizing Sialometaplasia of the Hard Palate: Diagnosis and
Treatment
Sangeun Lee, Yun Sung Lim, Kyuho Lee, Bo Hae Kim
Journal of Clinical Otolaryngology Head and Neck Surgery.2022; 33(4): 236. CrossRef - Parotid Salivary Duct Carcinoma With a Prominent Squamous Component: Immunohistochemical Profile, Diagnostic Pitfalls, and Therapeutic Implications
Naomi Hardy, Joshua Thompson, Ranee Mehra, Cinthia B. Drachenberg, Kyle Hatten, John C. Papadimitriou
International Journal of Surgical Pathology.2021; 29(7): 726. CrossRef - Intrasalivary Thymic Carcinoma: A Case Report and Literature Review
Michał Kunc, Alexandra Kamieniecki, Grzegorz Walczak, Tomasz Nowicki, Bartosz Wasąg, Bogusław Mikaszewski, Dominik Stodulski, Wojciech Biernat
Head and Neck Pathology.2021; 16(3): 857. CrossRef - Cancer Stem Cell Markers in Squamous Cell Carcinomas of the Salivary Glands
Mattis Bertlich, Julia Kitz, Marie Kruizenga, Jennifer Lee Spiegel, Martin Canis, Friedrich Ihler, Frank Haubner, Bernhard G. Weiss, Mark Jakob
Oncology.2021; 99(6): 402. CrossRef
- Solitary Parotid Metastasis as an Atypical Site of Recurrence in Lung Squamous Cell Carcinoma: A Diagnostic Difficulty
- A retrospective cytohistological correlation of fine-needle aspiration cytology with classification by the Milan System for Reporting Salivary Gland Cytopathology
- Ji Hyun Park, Yoon Jin Cha, Ja Yeong Seo, Jae Yol Lim, Soon Won Hong
- J Pathol Transl Med. 2020;54(5):419-425. Published online July 8, 2020
- DOI: https://doi.org/10.4132/jptm.2020.06.09
- 7,749 View
- 206 Download
- 12 Web of Science
- 15 Crossref
-
 Abstract
Abstract
 PDF
PDF - Background
Before publication of the new classification system named the Milan System for Reporting Salivary Gland Cytopathology (MSRSGC) in 2018, there was no standard classification for salivary gland lesions obtained by fine-needle aspiration (FNA). We therefore aimed to evaluate the diagnostic utility of this system by retrospectively reviewing FNA samples using the MSRSGC and to determine their risk of developing into neoplasms and becoming malignant.
Methods
Retrospective slide review and classification of salivary gland FNAs obtained over a 6-year period (2013–2018) at a single center were performed by two pathologists. The risks of neoplasm and malignancy for each category also were calculated.
Results
This study surveyed 374 FNAs (371 patients) performed over a six-year period and selected 148 cases that included documented surgical follow-up (39.6%). Among the surgically treated cases, the distributions of FNA categories were as follows: non-diagnostic (ND; 16.9%), non-neoplastic (NN; 2.7%), atypia of undetermined significance (AUS; 3.4%), benign (BN; 54.7%), salivary gland neoplasm of uncertain malignant potential (SUMP; 10.1%), suspicious for malignancy (SM; 6.8%), and malignant (M; 5.4%). The risk of malignancy (ROM) was 24.0% for ND, 0% for NN, 40.0% for AUS, 2.5% for BN, 46.7% for SUMP, 100% for SM, and 87.5% for M. The overall diagnostic accuracy was 95.9% (142/148 cases).
Conclusions
The newly proposed MSRSGC appears to be a reliable system for classification of salivary gland lesions according to the associated ROM. -
Citations
Citations to this article as recorded by- Optimizing preoperative assessment of salivary gland lesions: updates and practical implications in the Milan System 2nd edition
Patrizia Straccia, Vincenzo Fiorentino, Alessia Piermattei, Qianqian Zhang, Belen Padial-Urtueta, Federica Cianfrini, Antonino Mule’, Esther Diana Rossi
Virchows Archiv.2026;[Epub] CrossRef - The Impact of Lesion-Specific and Sampling-Related Factors on Success of Salivary Gland Fine-Needle Aspiration Cytology
Marcel Mayer, Mohammad Marwan Alfarra, Kathrin Möllenhoff, Marianne Engels, Christoph Arolt, Alexander Quaas, Philipp Wolber, Louis Jansen, Lisa Nachtsheim, Maria Grosheva, Jens Peter Klussmann, Sami Shabli
Head and Neck Pathology.2025;[Epub] CrossRef - The Myriad Spectrum of Salivary Gland Lesions: Cytohistological Correlation on Fine Needle Aspiration Cytology, Core Needle Biopsy, and Resections in a 5‐Year Single Institutional Experience of North India
Zachariah Chowdhury, Pallavi Majumdar, Sumeet Narain, Komal Lamba
Diagnostic Cytopathology.2025; 53(8): 391. CrossRef - Diagnostic Performance of the Milan System for Reporting Salivary Gland Cytopathology and a Proposed Algorithm for Fine-Needle Aspiration Cytology of Salivary Gland Lesions
Norihide Mochizuki, Hirotaka Fujita, Takuma Tajiri, Masataka Ueda, Makiko Kurata, Chie Inomoto, Tomoko Sugiyama, Daisuke Maki, Shuichi Shiraishi, Tomohisa Machida, Hitoshi Ito, Yohei Masugi, Naoya Nakamura
Acta Cytologica.2025; 69(4): 324. CrossRef - A study of fine needle aspiration cytology and histopathology correlation of salivary gland neoplasms in a tertiary care hospital: an observational study
Asima Malik, Ahlam Mushtaq, Salma Bhat, Suhail Naik
International Journal of Contemporary Pediatrics.2025; 13(1): 23. CrossRef - The Milan system for reporting salivary gland cytopathology – Assessment of utility and the risk of malignancy
Annu E. Prakash, Renu Sukumaran, Nileena Nayak, K. Lakshmi, Anitha Mathews, Jayasree Kattoor
Indian Journal of Cancer.2024; 61(3): 575. CrossRef - Salivary gland fine-needle aspiration biopsy: quality assurance results from a tertiary cancer center
Fanni Ratzon, Dominique L. Feliciano, Nora Katabi, Bin Xu, Oscar Lin, Xiao-Jun Wei
Journal of the American Society of Cytopathology.2023; 12(3): 206. CrossRef - Cytohistological correlation and risk stratification of salivary gland lesions using the Milan System for Reporting Salivary Gland Cytopathology: A tertiary care centre experience
Tarun Kumar, Prerna Tewari, Jitendra Singh Nigam, Shreekant Bharti, Surabhi, Ruchi Sinha, Punam Prasad Bhadani
Cytopathology.2023; 34(3): 225. CrossRef - Assessment of Risk of Malignancy of Fine-needle Aspiration Cytology in Salivary Gland Lesions Using the Milan System for Reporting Salivary Gland Cytopathology Categorization: A Systematic Review and Meta-analysis
Amit Kumar, Subhash Chandra, Bishnupati Singh, Swati Sharma, Ankita Tandon, Ajoy Kumar Shahi
The Journal of Contemporary Dental Practice.2023; 23(10): 1039. CrossRef - Milan Sınıflandırma Sistemi’ne Göre Değerlendirilen Tükürük Bezi İnce İğne Aspirasyon Sitolojilerinin Histopatolojik Tanı Uyumu
Özlem SARAYDAROĞLU, Selin YİRMİBEŞ
Uludağ Üniversitesi Tıp Fakültesi Dergisi.2023; 49(3): 285. CrossRef - Milan system for reporting salivary gland cytopathology: Adoption and outcomes in a community setting
Samih J. Nassif, Ali R. Sasani, Garrey T. Faller, Jennifer L. Harb, Jagdish K. Dhingra
Head & Neck.2022; 44(6): 1462. CrossRef - Nondiagnostic salivary gland FNAs are associated with decreased risk of malignancy compared with “all‐comer” patients: Analysis of the Milan System for Reporting Salivary Gland Cytopathology with a focus on Milan I: Nondiagnostic
Shu K. Lui, Troy Tenney, Patrick C. Mullane, Kartik Viswanathan, Daniel J. Lubin
Cancer Cytopathology.2022; 130(10): 800. CrossRef - Application of the Milan System for Reporting Salivary Gland Cytopathology: A systematic review and meta‐analysis
Zhaoyang Wang, Huan Zhao, Huiqin Guo, Changming An
Cancer Cytopathology.2022; 130(11): 849. CrossRef - Multiplexed single‐cell analysis of FNA allows accurate diagnosis of salivary gland tumors
Juhyun Oh, Tae Yeon Yoo, Talia M. Saal, Lisa Tsay, William C. Faquin, Jonathan C.T. Carlson, Daniel G. Deschler, Sara I. Pai, Ralph Weissleder
Cancer Cytopathology.2022; 130(8): 581. CrossRef - Cytologic analysis of vitreous fluids: A retrospective review of our 24 years of experience
Gabriel L. Collins, Elizabeth W. Hubbard, Christopher T. Clark, Lisa D. Duncan, Laurentia Nodit
Diagnostic Cytopathology.2021; 49(10): 1122. CrossRef
- Optimizing preoperative assessment of salivary gland lesions: updates and practical implications in the Milan System 2nd edition
- 2019 Practice guidelines for thyroid core needle biopsy: a report of the Clinical Practice Guidelines Development Committee of the Korean Thyroid Association
- Chan Kwon Jung, Jung Hwan Baek, Dong Gyu Na, Young Lyun Oh, Ka Hee Yi, Ho-Cheol Kang
- J Pathol Transl Med. 2020;54(1):64-86. Published online January 15, 2020
- DOI: https://doi.org/10.4132/jptm.2019.12.04
- 28,694 View
- 1,082 Download
- 54 Web of Science
- 62 Crossref
-
 Abstract
Abstract
 PDF
PDF - Ultrasound-guided core needle biopsy (CNB) has been increasingly used for the pre-operative diagnosis of thyroid nodules. Since the Korean Society of the Thyroid Radiology published the ‘Consensus Statement and Recommendations for Thyroid CNB’ in 2017 and the Korean Endocrine Pathology Thyroid CNB Study Group published ‘Pathology Reporting of Thyroid Core Needle Biopsy’ in 2015, advances have occurred rapidly not only in the management guidelines for thyroid nodules but also in the diagnostic terminology and classification schemes. The Clinical Practice Guidelines Development Committee of the Korean Thyroid Association (KTA) reviewed publications on thyroid CNB from 1995 to September 2019 and updated the recommendations and statements for the diagnosis and management of thyroid nodules using CNB. Recommendations for the resolution of clinical controversies regarding the use of CNB were based on expert opinion. These practical guidelines include recommendations and statements regarding indications for CNB, patient preparation, CNB technique, biopsy-related complications, biopsy specimen preparation and processing, and pathology interpretation and reporting of thyroid CNB.
-
Citations
Citations to this article as recorded by- Reevaluation of malignancy risk in nondiagnostic thyroid nodules with long‐term follow‐up via surgical resection or core needle biopsy: A retrospective study
Ji‐Seon Jeong, Young Jun Choi, Jeong Hyun Lee, Jung Hwan Baek, Yu‐Mi Lee, Tae‐Yon Sung, Dong Eun Song
Cancer Cytopathology.2026;[Epub] CrossRef - Clinical observation of thyroid metastatic lesion in a patient withrectal cancer
K. M. Blikyan, S. V. Lukyanov, A. B. Alnikin, N. S. Lukyanov, P. V. Konovalenko
Endocrine Surgery.2026; 19(3): 40. CrossRef - Current Evidence, Selective Indications, and the Role of Lymph-Node Assessment in Intraoperative Frozen Section in Thyroid Cancer Surgery: A Literature Review
Gregorio Scerrino, Marco Marciano', Bianca Vicari, Maria Aurora Bullaro, Renato Di Vuolo, Pierina Richiusa, Giuseppina Orlando, Vito Rodolico, Giuseppina Melfa
Journal of Clinical Medicine.2026; 15(4): 1611. CrossRef - Core needle biopsy as a first-line diagnostic tool for selected thyroid nodules: a real-world evaluation of diagnostic performance and safety
Xing Li, Yi Pan, Yanmei Ou, Xin Gao, Yue Gao, Luwei Liu, Yinze Li, Yong Xu, Wengui Xu
Frontiers in Oncology.2026;[Epub] CrossRef - Evaluation and Management of Thyroid Nodules: A Joint Consensus Statement From the British Thyroid Association (BTA), British Association of Endocrine and Thyroid Surgeons (BAETS) and Collaborating Bodies
Ram Moorthy, Saba P. Balasubramanian, Kate Farnell, Mairead Kelly, Gitta Madani, Mufaddal Moonim, Carla Moran, Julia Priestley, Michael Stechman, Emma Watts, Kristien Boelaert
Clinical Endocrinology.2026; 104(6): 682. CrossRef - Usefulness of core needle biopsy of thyroid for the diagnosis of IgG4 Hashimoto's thyroiditis
Chenxu Zhao, Yang Yu, Jumei Liu, Yang Zhang, Lei Chen, Guizhi Lu, Ying Gao
Journal of Translational Internal Medicine.2026; 14(2): 306. CrossRef - Comparison of ultrasound-guided biopsy techniques for level IV lymph nodes: semiautomatic vs. Menghini modified needles in a retrospective dual-center study
Gang Liu, Yixin Zhu, Guoru Wu, Guangyin Yu, Hao Luo, Lu Pang, Qiongxian Long, Lin Zhu, Yu Shi
BMC Medical Imaging.2026;[Epub] CrossRef - Clinical Value of Core Needle Biopsy as a Second-Line Approach After Non-Conclusive Fine-Needle Aspiration in Thyroid Nodules: A Paired Analysis
Vladan Markovic, Slobodanka Mitrovic, Tijana Maksic, Irfan Corovic, Marija Sekulic, Mladen Maksic, Vesna Grbovic
Diagnostics.2026; 16(7): 1104. CrossRef - Advantages of thyroid core needle biopsy: an emerging selective first-line biopsy modality
Jae Ho Shin, Yeseul Kim, Min Kyoung Lee, Jung Hwan Baek, So Lyung Jung
Ultrasonography.2026; 45(3): 205. CrossRef - Preoperative hydrodissection for predicting extrathyroidal extension in thyroid tumors
Yeseul Kim, Jae Ho Shin, Sung-Hye You, Bo Kyu Kim, Byungjun Kim, Kyeong Jin Kim
Minimally Invasive Therapy & Allied Technologies.2026; : 1. CrossRef - Comparison of core-needle biopsy and repeat fine-needle aspiration biopsy for thyroid nodules with initially inconclusive findings: a systematic review, diagnostic accuracy meta-analysis, and meta-regression
Hendra Zufry, Timotius Ivan Hariyanto
Journal of the American Society of Cytopathology.2025; 14(3): 159. CrossRef - Ultrasound-guided core-needle biopsy for diagnosis of thyroid cancer
D.D. Dolidze, S.D. Kovantsev, Z.A. Bagatelia, A.V. Bumbu, Yu.V. Barinov, G.M. Chechenin, N.V. Pichugina, D.G. Gogolashvili
Pirogov Russian Journal of Surgery.2025; (3): 87. CrossRef - Superior Diagnostic Yield of Core Needle Biopsy Over Fine Needle Aspiration in Diagnosing Follicular-Patterned Neoplasms: A Multicenter Study Focusing on Bethesda IV Results
Leehi Joo, Jung Hwan Baek, Jungbok Lee, Dong Eun Song, Sae Rom Chung, Young Jun Choi, Jeong Hyun Lee
Korean Journal of Radiology.2025; 26(6): 604. CrossRef - Diagnostic yield of fine needle aspiration with simultaneous core needle biopsy for thyroid nodules
Mohammad Ali Hasannia, Ramin Pourghorban, Hoda Asefi, Amir Aria, Elham Nazar, Hojat Ebrahiminik, Alireza Mohamadian
Journal of Pathology and Translational Medicine.2025; 59(3): 180. CrossRef - Lessons learned from the first 2 years of experience with thyroid core needle biopsy at an Indonesian national referral hospital
Agnes Stephanie Harahap, Maria Francisca Ham, Retno Asti Werdhani, Erwin Danil Julian, Rafi Ilmansyah, Chloe Indira Arfelita Mangunkusumso, Tri Juli Edi Tarigan
Journal of Pathology and Translational Medicine.2025; 59(3): 149. CrossRef - Preoperative Fine-Needle Aspiration in Goiter With Compressive Symptoms: A Systematic Review and Meta-analysis
Moeen Sbeit, Rania Faris, Ohad Ronen
Endocrine Practice.2025; 31(8): 1038. CrossRef - Risk Stratification of Thyroid Nodules Diagnosed as Follicular Neoplasm on Core Needle Biopsy
Byeong-Joo Noh, Won Jun Kim, Jin Yub Kim, Ha Young Kim, Jong Cheol Lee, Myoung Sook Shim, Yong Jin Song, Kwang Hyun Yoon, In-Hye Jung, Hyo Sang Lee, Wooyul Paik, Dong Gyu Na
Endocrinology and Metabolism.2025; 40(4): 610. CrossRef - Diagnostic performances of five US risk stratification systems for malignancy in focal [18F]FDG-PET/CT thyroid incidentalomas
Chae Young Shin, Hye Shin Ahn, Dong Gyu Na, Hyo Sang Lee, Eon-Woo Shin
European Radiology.2025; 35(12): 7701. CrossRef - Malignancy risk in AUS thyroid lesions: comparison between FNA and CNB with implications for NIFTP diagnosis
Yeseul Kim, Jae Ho Shin, You-Na Sung, Dawon Park, Harim Oh, Hyo Seon Ryu, Kyeong Jin Kim, Hyun Joo Kim, Sin Gon Kim, Hoon Yub Kim, Kwang Yoon Jung, Seung-Kuk Baek, Sangjeong Ahn
Frontiers in Endocrinology.2025;[Epub] CrossRef - Comparison of Diagnostic Yield Between Fine Needle Aspiration Cytology and Core Needle Biopsy in the Diagnosis of Thyroid Nodule
Yeongrok Lee, Myung Jin Ban, Do Hyeon Kim, Jin-Young Kim, Hyung Kwon Byeon, Jae Hong Park
Diagnostics.2025; 15(20): 2566. CrossRef - Repeatedly non-diagnostic thyroid nodules: the experience of two thyroid clinics
Filippo EGALINI, Mattia ROSSI, Chiara MELE, Yanina LIZET CASTILLO, Francesca MALETTA, Barbara PULIGHEDDU, Ezio GHIGO, Ruth ROSSETTO GIACCHERINO, Loredana PAGANO, Mauro PAPOTTI
Minerva Endocrinology.2025;[Epub] CrossRef - A comparative analysis of core needle biopsy and repeat fine needle aspiration in patients with inconclusive initial cytology of thyroid nodules
Xuejiao Su, Can Yue, Wanting Yang, Buyun Ma
Frontiers in Endocrinology.2024;[Epub] CrossRef - A Narrative Review of the 2023 Korean Thyroid Association Management Guideline for Patients with Thyroid Nodules
Eun Kyung Lee, Young Joo Park, Chan Kwon Jung, Dong Gyu Na
Endocrinology and Metabolism.2024; 39(1): 61. CrossRef - Doing more with less: integrating small biopsies in cytology practice
Anjali Saqi, Michiya Nishino, Mauro Saieg, Amy Ly, Abberly Lott Limbach
Journal of the American Society of Cytopathology.2024; 13(4): 233. CrossRef - 2023 Update of the Korean Thyroid Association Guidelines for the Management of Thyroid Nodules
Eun Kyung Lee, Young Joo Park
Clinical Thyroidology®.2024; 36(4): 153. CrossRef - Korean Thyroid Association Guidelines on the Management of Differentiated Thyroid Cancers; Part I. Initial Management of Differentiated Thyroid Cancers - Chapter 2. Surgical Management of Thyroid Cancer 2024
Yoon Young Cho, Cho Rok Lee, Ho-Cheol Kang, Bon Seok Koo, Hyungju Kwon, Sun Wook Kim, Won Woong Kim, Jung-Han Kim, Dong Gyu Na, Young Joo Park, Kyorim Back, Young Shin Song, Seung Hoon Woo, Ho-Ryun Won, Chang Hwan Ryu, Jee Hee Yoon, Min Kyoung Lee, Eun Ky
International Journal of Thyroidology.2024; 17(1): 30. CrossRef - Korean Thyroid Association Management Guidelines for Patients with Thyroid Nodules 2024
Young Joo Park, Eun Kyung Lee, Young Shin Song, Su Hwan Kang, Bon Seok Koo, Sun Wook Kim, Dong Gyu Na, Seung-Kuk Baek, So Won Oh, Min Kyoung Lee, Sang-Woo Lee, Young Ah Lee, Yong Sang Lee, Ji Ye Lee, Dong-Jun Lim, Leehi Joo, Yuh-Seog Jung, Chan Kwon Jung,
International Journal of Thyroidology.2024; 17(1): 208. CrossRef - Korean Thyroid Association Guidelines on the Management of Differentiated Thyroid Cancers; Overview and Summary 2024
Young Joo Park, Eun Kyung Lee, Young Shin Song, Bon Seok Koo, Hyungju Kwon, Keunyoung Kim, Mijin Kim, Bo Hyun Kim, Won Gu Kim, Won Bae Kim, Won Woong Kim, Jung-Han Kim, Hee Kyung Kim, Hee Young Na, Shin Je Moon, Jung-Eun Moon, Sohyun Park, Jun-Ook Park, J
International Journal of Thyroidology.2024; 17(1): 1. CrossRef - Educational exchange in thyroid core needle biopsy diagnosis: enhancing pathological interpretation through guideline integration and peer learning
Agnes Stephanie Harahap, Chan Kwon Jung
Journal of Pathology and Translational Medicine.2024; 58(5): 205. CrossRef - Current role of interventional radiology in thyroid nodules
Onur Taydas, Erbil Arik, Omer Faruk Sevinc, Ahmet Burak Kara, Mustafa Ozdemir, Hasret Cengiz, Zulfu Bayhan, Mehmet Halil Ozturk
Frontiers in Endocrinology.2024;[Epub] CrossRef - Neck Schwannoma Masking as Thyroid Tumour: Into the Deep of Diagnostics and Anatomy
Serghei Covantsev, Anna Bumbu, Anna Sukhotko, Evghenii Zakurdaev, Ivan Kuts, Andrey Evsikov
Diagnostics.2024; 14(20): 2332. CrossRef - Thermal ablation for Bethesda III and IV thyroid nodules: current diagnosis and management
Wen-Hui Chan, Pi-Ling Chiang, An-Ni Lin, Yen-Hsiang Chang, Wei-Che Lin
Ultrasonography.2024; 43(6): 395. CrossRef - A new LNC89/LNC60-Col11a2 axis revealed by whole-transcriptome analysis may be associated with goiters related to excess iodine nutrition
Guanying Nie, Shuang Li, Wei Zhang, Fangang Meng, Zixuan Ru, Jiahui Li, Dianjun Sun, Ming Li
Frontiers in Endocrinology.2024;[Epub] CrossRef - A simplified four-tier classification for thyroid core needle biopsy
M. Paja, J. L. Del Cura, R. Zabala, I. Korta, Mª T. Gutiérrez, A. Expósito, A. Ugalde
Journal of Endocrinological Investigation.2024; 48(4): 895. CrossRef - Risk of thyroid cancer in a lung cancer screening population of the National Lung Screening Trial according to the presence of incidental thyroid nodules detected on low-dose chest CT
Hyobin Seo, Kwang Nam Jin, Ji Sang Park, Koung Mi Kang, Eun Kyung Lee, Ji Ye Lee, Roh-Eul Yoo, Young Joo Park, Ji-hoon Kim
Ultrasonography.2023; 42(2): 275. CrossRef - Preoperative Risk Stratification of Follicular-patterned Thyroid Lesions on Core Needle Biopsy by Histologic Subtyping and RAS Variant-specific Immunohistochemistry
Meejeong Kim, Sora Jeon, Chan Kwon Jung
Endocrine Pathology.2023; 34(2): 247. CrossRef - Differential regional importance mapping for thyroid nodule malignancy prediction with potential to improve needle aspiration biopsy sampling reliability
Liping Wang, Yuan Wang, Wenliang Lu, Dong Xu, Jincao Yao, Lijing Wang, Lei Xu
Frontiers in Oncology.2023;[Epub] CrossRef - Preoperative evaluation of thyroid nodules – Diagnosis and management strategies
Tapoi Dana Antonia, Lambrescu Ioana Maria, Gheorghisan-Galateanu Ancuta-Augustina
Pathology - Research and Practice.2023; 246: 154516. CrossRef - 2023 Korean Thyroid Association Management Guidelines for Patients with Thyroid Nodules
Young Joo Park, Eun Kyung Lee, Young Shin Song, Soo Hwan Kang, Bon Seok Koo, Sun Wook Kim, Dong Gyu Na, Seung-Kuk Baek, So Won Oh, Min Kyoung Lee, Sang-Woo Lee, Young Ah Lee, Yong Sang Lee, Ji Ye Lee, Dong-Jun Lim, Leehi Joo, Yuh-Seog Jung, Chan Kwon Jung
International Journal of Thyroidology.2023; 16(1): 1. CrossRef - Fast Track Management of Primary Thyroid Lymphoma in the Very Elderly Patient
Pierre Yves Marcy, Frederic Bauduer, Juliette Thariat, Olivier Gisserot, Edouard Ghanassia, Bruno Chetaille, Laurys Boudin, Jean Baptiste Morvan
Current Oncology.2023; 30(6): 5816. CrossRef - Reevaluating diagnostic categories and associated malignancy risks in thyroid core needle biopsy
Chan Kwon Jung
Journal of Pathology and Translational Medicine.2023; 57(4): 208. CrossRef - Diagnostic performance of shear wave elastography in thyroid nodules with indeterminate cytology: A systematic review and meta-analysis
Yuxuan Qiu, Zhichao Xing, Qianru Yang, Yan Luo, Buyun Ma
Heliyon.2023; 9(10): e20654. CrossRef - Comparison of the diagnostic value of fine needle aspiration and ultrasound in thyroid pathology
P. S. Glushkov, R. Kh. Azimov, N. L. Aleshenko, E. A. Maruchak, Y. P. Sych, G. N. Minkova, K. A. Shemyatovsky, V. A. Gorsky
Endocrine Surgery.2023; 17(3): 43. CrossRef - Comparison of Core Needle Biopsy and Repeat Fine-Needle Aspiration in Avoiding Diagnostic Surgery for Thyroid Nodules Initially Diagnosed as Atypia/Follicular Lesion of Undetermined Significance
Leehi Joo, Dong Gyu Na, Ji-hoon Kim, Hyobin Seo
Korean Journal of Radiology.2022; 23(2): 280. CrossRef - Diagnostic efficacy, performance and safety of side-cut core needle biopsy for thyroid nodules: comparison of automated and semi-automated biopsy needles
Ji Yeon Park, Seong Yoon Yi, Soo Heui Baek, Yu Hyun Lee, Heon-Ju Kwon, Hee Jin Park
Endocrine.2022; 76(2): 341. CrossRef - Thyroid Cancer Diagnostics Related to Occupational and Environmental Risk Factors: An Integrated Risk Assessment Approach
Gabriela Maria Berinde, Andreea Iulia Socaciu, Mihai Adrian Socaciu, Andreea Cozma, Armand Gabriel Rajnoveanu, Gabriel Emil Petre, Doina Piciu
Diagnostics.2022; 12(2): 318. CrossRef - Approach to Bethesda system category III thyroid nodules according to US-risk stratification
Jieun Kim, Jung Hee Shin, Young Lyun Oh, Soo Yeon Hahn, Ko Woon Park
Endocrine Journal.2022; 69(1): 67. CrossRef - Clinicopathological and Molecular Features of Secondary Cancer (Metastasis) to the Thyroid and Advances in Management
Marie Nguyen, George He, Alfred King-Yin Lam
International Journal of Molecular Sciences.2022; 23(6): 3242. CrossRef - Diagnostic Performance of Thyroid Core Needle Biopsy Using the Revised Reporting System: Comparison with Fine Needle Aspiration Cytology
Kwangsoon Kim, Ja Seong Bae, Jeong Soo Kim, So Lyung Jung, Chan Kwon Jung
Endocrinology and Metabolism.2022; 37(1): 159. CrossRef - Core Needle Biopsy Can Early and Precisely Identify Large Thyroid Masses
Antonio Matrone, Luigi De Napoli, Liborio Torregrossa, Aleksandr Aghababyan, Piermarco Papini, Carlo Enrico Ambrosini, Rosa Cervelli, Clara Ugolini, Fulvio Basolo, Eleonora Molinaro, Rossella Elisei, Gabriele Materazzi
Frontiers in Oncology.2022;[Epub] CrossRef - Primary thyroid leiomyosarcoma with transvenous extension to the right atrium: a case report
Juraj Dubrava, Peter Martanovic, Marina Pavlovicova, Pavel Babal, Akhil Narang, Maria Mattioli, Nidhish Tiwari, Zhiyu Liu, Mariame Chakir
European Heart Journal - Case Reports.2022;[Epub] CrossRef - Radiofrequency ablation for management of thyroid nodules in quarantine zone of COVID-19 pandemic setting in Indonesia
Kristanto Yuli Yarso, Sumadi Lukman Anwar
Annals of Medicine and Surgery.2022; 81: 104132. CrossRef - A Matched-Pair Analysis of Nuclear Morphologic Features Between Core Needle Biopsy and Surgical Specimen in Thyroid Tumors Using a Deep Learning Model
Faridul Haq, Andrey Bychkov, Chan Kwon Jung
Endocrine Pathology.2022; 33(4): 472. CrossRef - Diagnostic performance of core needle biopsy as a first‐line diagnostic tool for thyroid nodules according to ultrasound patterns: Comparison with fine needle aspiration using propensity score matching analysis
Hye Shin Ahn, Inyoung Youn, Dong Gyu Na, Soo Jin Kim, Mi Yeon Lee
Clinical Endocrinology.2021; 94(3): 494. CrossRef - Hydrodissection: A Novel Approach for Safe Core Needle Biopsy of Small High-Risk Subcapsular Thyroid Nodules
Hojat Ebrahiminik, Hossein Chegeni, Javad Jalili, Rambod Salouti, Hadi Rokni, Afshin Mohammadi, Ali Mosaddegh Khah, Seyed Mohammad Tavangar, Zahra Ebrahiminik
CardioVascular and Interventional Radiology.2021; 44(10): 1651. CrossRef - Application of biomarkers in the diagnosis of uncertain samples of core needle biopsy of thyroid nodules
Yan Xiong, Xin Li, Li Liang, Dong Li, Limin Yan, Xueying Li, Jiting Di, Ting Li
Virchows Archiv.2021; 479(5): 961. CrossRef - VE1 immunohistochemistry is an adjunct tool for detection of BRAFV600E mutation: Validation in thyroid cancer patients
Faiza A. Rashid, Sobia Tabassum, Mosin S. Khan, Hifzur R. Ansari, Muhammad Asif, Ahmareen K. Sheikh, Syed Sameer Aga
Journal of Clinical Laboratory Analysis.2021;[Epub] CrossRef - The Diagnostic Value of the American College of Radiology Thyroid Imaging Reporting and Data System Classification and Shear-Wave Elastography for the Differentiation of Thyroid Nodules
Gül Bora Makal, Aydın Aslan
Ultrasound in Medicine & Biology.2021; 47(5): 1227. CrossRef - Comparison of the diagnostic performance of the modified Korean Thyroid Imaging Reporting and Data System for thyroid malignancy with three international guidelines
Eun Ju Ha, Jung Hee Shin, Dong Gyu Na, So Lyung Jung, Young Hen Lee, Wooyul Paik, Min Ji Hong, Yeo Koon Kim, Chang Yoon Lee
Ultrasonography.2021; 40(4): 594. CrossRef - VE1 Immunohistochemistry Improves the Limit of Genotyping for Detecting BRAFV600E Mutation in Papillary Thyroid Cancer
Sonam Choden, Somboon Keelawat, Chan Kwon Jung, Andrey Bychkov
Cancers.2020; 12(3): 596. CrossRef - The 2019 core-needle biopsy practice guidelines
So Yeong Jeong, Jung Hwan Baek
Ultrasonography.2020; 39(3): 311. CrossRef - Re: The 2019 core-needle biopsy practice guidelines
Ji-hoon Kim
Ultrasonography.2020; 39(3): 313. CrossRef
- Reevaluation of malignancy risk in nondiagnostic thyroid nodules with long‐term follow‐up via surgical resection or core needle biopsy: A retrospective study
- Primary Necrobiotic Xanthogranulomatous Sialadenitis with Submandibular Gland Localization without Skin Involvement
- Myunghee Kang, Na Rae Kim, Dong Hae Chung, Jae Yeon Seok, Dong Young Kim
- J Pathol Transl Med. 2019;53(4):261-265. Published online January 16, 2019
- DOI: https://doi.org/10.4132/jptm.2019.01.08
- 9,656 View
- 171 Download
- 2 Web of Science
- 6 Crossref
-
 Abstract
Abstract
 PDF
PDF - Necrobiotic xanthogranulomatous reaction is a multiorgan, non-Langerhans cell histiocytosis with an unknown etiology. Occurrence in the salivary gland is extremely rare. We recently identified a case of necrobiotic xanthogranulomatous sialadenitis in a 73-year-old Korean woman who presented with a painless palpable lesion in the chin. There was no accompanying cutaneous lesion. Partial resection and subsequent wide excision with neck dissection were performed. Pathological examination showed a severe inflammatory lesion that included foamy macrophages centrally admixed with neutrophils, eosinophils, lymphocytes, plasma cells, and scattered giant cells, as well as necrobiosis. During the 12-month postoperative period, no grossly remarkable change in size was noted. Necrobiotic xanthogranulomatous inflammation may be preceded by or combined with hematologic malignancy. Although rare, clinicians and radiologists should be aware that an adhesive necrobiotic xanthogranuloma in the salivary gland may present with a mass-like lesion. Further evaluation for hematologic disease and close follow-up are needed when a pathologic diagnosis is made.
-
Citations
Citations to this article as recorded by- Salivary gland macrophages in health and disease: heterogeneity, niche crosstalk, and therapeutic avenues
Xinglei Li, Yan Feng, Huixin Xue, Xinxin Ni
Frontiers in Immunology.2025;[Epub] CrossRef - Five Cases of Xanthogranulomatous Sialadenitis
Satoshi Kiyama, Hiroyuki Iuchi, Kotoko Ito, Kengo Nishimoto, Tsutomu Matsuzaki, Masaru Yamashita
Practica Oto-Rhino-Laryngologica.2022; 115(4): 315. CrossRef - Xanthogranulomatous change in a pleomorphic adenoma: An extremely rare variant/degenerative change. Is it fine needle aspiration induced?
Mukta Pujani, Dipti Sidam, Kanika Singh, Aparna Khandelwal, Khushbu Katarya
Diagnostic Cytopathology.2021;[Epub] CrossRef - A Case of Xanthogranulomatous Sialadenitis with Facial Palsy Mimicking Malignancy
Sang Hyun Kim, Sun Woo Kim, Sang Hyuk Lee
Korean Journal of Otorhinolaryngology-Head and Neck Surgery.2021; 64(6): 422. CrossRef - Xanthogranulomatous Sialadenitis, an Uncommon Reactive Change is Often Associated with Warthin’s Tumor
Lihong Bu, Hui Zhu, Emilian Racila, Sobia Khaja, David Hamlar, Faqian Li
Head and Neck Pathology.2020; 14(2): 525. CrossRef - A Case of Xanthogranulomatous Sialadenitis of the Sublingual Gland:A Review of Literature
Naoya KITAMURA, Seiji OHNO, Tetsuya YAMAMOTO
Journal of Japanese Society of Oral Medicine.2019; 25(1): 20. CrossRef
- Salivary gland macrophages in health and disease: heterogeneity, niche crosstalk, and therapeutic avenues
- Cytopathologic Features of Secretory Carcinoma of Salivary Gland: Report of Two Cases
- Young Ah Kim, Jae Won Joung, Sun-Jae Lee, Hoon-Kyu Oh, Chang Ho Cho, Woo Jung Sung
- J Pathol Transl Med. 2019;53(1):70-74. Published online December 28, 2018
- DOI: https://doi.org/10.4132/jptm.2018.11.09
- 9,185 View
- 150 Download
- 7 Web of Science
- 6 Crossref
-
 Abstract
Abstract
 PDF
PDF - Secretory carcinoma of the salivary gland (SC) is a newly introduced rare salivary gland tumor that shares histological, immunohistochemical, and genetic characteristics with secretory carcinoma of the breast. Here, we report the cytologic features of two cases of SC confirmed by surgical resection. In these two cases, SC was incidentally detected in a 64-year-old female and a 56-yearold male. Fine needle aspiration cytology revealed nests of tumor cells with a papillary or glandular structure floating in mucinous secretions. The tumor cells demonstrated uniform, round, smooth nuclear contours and distinct nucleoli. Multiple characteristic cytoplasmic vacuoles were revealed. Singly scattered tumor cells frequently showed variable sized cytoplasmic vacuoles. The cytopathologic diagnosis of SC should be considered when characteristic cytological findings are revealed. Further immunohistochemistry and gene analyses are helpful to diagnose SC.
-
Citations
Citations to this article as recorded by- Salivary Gland Secretory Carcinoma; Review of 13 Years World‐Wide Experience and Meta‐Analysis
Eyal Yosefof, Tomer Boldes, Daniel Dan, Eyal Robenshtok, Yulia Strenov, Gideon Bachar, Thomas Shpitzer, Aviram Mizrachi
The Laryngoscope.2024; 134(4): 1716. CrossRef - Efficacy of Fine-Needle Aspiration Cytology in Diagnosing Secretory Carcinoma of Salivary Gland: A Systematic Review and Meta-Analysis
Pooja Sharma Kala, Mamta Gupta, Naveen Thapliyal
Acta Cytologica.2024; 68(2): 83. CrossRef - An Underappreciated Cytomorphological Feature of Secretory Carcinoma of Salivary Gland on Fine Needle Aspiration Biopsy: Case Report with Literature Review
Yinan Hua, Bing Leng, Kenneth E. Youens, Lina Liu
Head and Neck Pathology.2022; 16(2): 567. CrossRef - Prognostic factors in mammary analogue secretory carcinomas of the parotid gland: Systematic review and meta‐analysis
Stefan Janik, Muhammad Faisal, Blazen Marijić, Stefan Grasl, Matthaeus Ch. Grasl, Gregor Heiduschka, Boban M. Erovic
Head & Neck.2022; 44(3): 792. CrossRef - A systematic review of secretory carcinoma of the salivary gland: where are we?
Lísia Daltro Borges Alves, Andreia Cristina de Melo, Thayana Alves Farinha, Luiz Henrique de Lima Araujo, Leandro de Souza Thiago, Fernando Luiz Dias, Héliton Spíndola Antunes, Ana Lucia Amaral Eisenberg, Luiz Claudio Santos Thuler, Daniel Cohen Goldember
Oral Surgery, Oral Medicine, Oral Pathology and Oral Radiology.2021; 132(4): e143. CrossRef - Clinical characteristics of acinic cell carcinoma and secretory carcinoma of the parotid gland
Tetsuya Terada, Ryo Kawata, Keiki Noro, Masaaki Higashino, Shuji Nishikawa, Shin-ichi Haginomori, Yoshitaka Kurisu, Hiroko Kuwabara, Yoshinobu Hirose
European Archives of Oto-Rhino-Laryngology.2019; 276(12): 3461. CrossRef
- Salivary Gland Secretory Carcinoma; Review of 13 Years World‐Wide Experience and Meta‐Analysis
- Adrenal Cortical Neoplasm with Uncertain Malignant Potential Arising in the Heterotopic Adrenal Cortex in the Liver of a Patient with Beckwith-Wiedemann Syndrome
- Eun Na Kim, Dong Eun Song, Hee Mang Yoon, Beom Hee Lee, Chong Jai Kim
- J Pathol Transl Med. 2019;53(2):129-135. Published online November 26, 2018
- DOI: https://doi.org/10.4132/jptm.2018.11.13
- 8,287 View
- 110 Download
- 5 Web of Science
- 5 Crossref
-
 Abstract
Abstract
 PDF
PDF - Patients with Beckwith-Wiedemann syndrome (BWS) are predisposed to developing embryonal tumors, with hepatoblastoma being the most common type. Our patient showed hemihypertrophy, macroglossia, and paternal uniparental disomy in chromosome 11 and was diagnosed with BWS. When the patient was 9 months old, a 2.5×1.5 cm oval hypoechoic exophytic mass was detected in the inferior tip of his right liver. Preoperative imaging identified it as hepatoblastoma; however, histologic, immunohistochemistry, and electron microscopic findings were compatible with adrenal cortical neoplasm with uncertain malignant potential. The origin of the adrenal tissue seemed to be heterotopic. Here, we describe for the first time an adrenal cortical neoplasm with uncertain malignant potential arising in the heterotopic adrenal cortex located in the liver of a patient with BWS.
-
Citations
Citations to this article as recorded by- Adrenocortical tumors and hereditary syndromes
Kanakamani Jeyaraman, Paola Concolino, Henrik Falhammar
Expert Review of Endocrinology & Metabolism.2025; 20(1): 1. CrossRef - Functional adrenocortical carcinoma with adrenohepatic fusion: A case report
Pastor Escárcega-Fujigaki, Guillermo Hernández-Peredo Rezk, José de Jesús Loeza- Oliva, Anallely Luna-Hernández, Bethsaida Natali Arreguín-Cortés, Rafael López-Cruz
Journal of Pediatric Surgery Case Reports.2024; 107: 102841. CrossRef - Molecular and Clinical Features of Adrenocortical Tumors in Beckwith–Wiedemann Spectrum
Diana Carli, Federico Rondot, Maria Luca, Anna Campello, Stefano Gabriele Vallero, Elisa Tirtei, Andrea Gazzin, Simona Cardaropoli, Francesca Montanari, Claudio Graziano, Paola Quarello, Abu Saadat, Angela Sparago, Giovanni Battista Ferrero, Franca Fagiol
Cancers.2024; 16(23): 3967. CrossRef - Beckwith–Wiedemann syndrome: Clinical, histopathological and molecular study of two Tunisian patients and review of literature
Hela Sassi, Yasmina Elaribi, Houweyda Jilani, Imen Rejeb, Syrine Hizem, Molka Sebai, Nadia Kasdallah, Habib Bouthour, Samia Hannachi, Jasmin Beygo, Ali Saad, Karin Buiting, Dorra H’mida Ben‐Brahim, Lamia BenJemaa
Molecular Genetics & Genomic Medicine.2021;[Epub] CrossRef - Adrenocortical Tumors in Children With Constitutive Chromosome 11p15 Paternal Uniparental Disomy: Implications for Diagnosis and Treatment
Emilia Modolo Pinto, Carlos Rodriguez-Galindo, Catherine G. Lam, Robert E. Ruiz, Gerard P. Zambetti, Raul C. Ribeiro
Frontiers in Endocrinology.2021;[Epub] CrossRef
- Adrenocortical tumors and hereditary syndromes
- PLAG1, SOX10, and Myb Expression in Benign and Malignant Salivary Gland Neoplasms
- Ji Hyun Lee, Hye Ju Kang, Chong Woo Yoo, Weon Seo Park, Jun Sun Ryu, Yuh-Seog Jung, Sung Weon Choi, Joo Yong Park, Nayoung Han
- J Pathol Transl Med. 2019;53(1):23-30. Published online November 14, 2018
- DOI: https://doi.org/10.4132/jptm.2018.10.12
- 13,734 View
- 384 Download
- 28 Web of Science
- 34 Crossref
-
 Abstract
Abstract
 PDF
PDF - Background
Recent findings in molecular pathology suggest that genetic translocation and/oroverexpression of oncoproteins is important in salivary gland tumorigenesis and diagnosis. Weinvestigated PLAG1, SOX10, and Myb protein expression in various salivary gland neoplasm tissues.
Methods
A total of 113 cases of surgically resected salivary gland neoplasms at the NationalCancer Center from January 2007 to March 2017 were identified. Immunohistochemical stainingof PLAG1, SOX10, and Myb in tissue samples was performed using tissue microarrays.
Results
Among the 113 cases, 82 (72.6%) were benign and 31 (27.4%) were malignant. PLAG1 showednuclear staining and normal parotid gland was not stained. Among 48 cases of pleomorphicadenoma, 29 (60.4%) were positive for PLAG1. All other benign and malignant salivary glandneoplasms were PLAG1-negative. SOX10 showed nuclear staining. In normal salivary gland tissuesSOX10 was expressed in cells of acinus and intercalated ducts. In benign tumors, SOX10 expressionwas observed in all pleomorphic adenoma (48/48), and basal cell adenoma (3/3), but not inother benign tumors. SOX10 positivity was observed in nine of 31 (29.0%) malignant tumors.Myb showed nuclear staining but was not detected in normal parotid glands. Four of 31 (12.9%)malignant tumors showed Myb positivity: three adenoid cystic carcinomas (AdCC) and onemyoepithelial carcinoma with focal AdCC-like histology.
Conclusions
PLAG1 expression is specificto pleomorphic adenoma. SOX10 expression is helpful to rule out excretory duct origin tumor,but its diagnostic value is relatively low. Myb is useful for diagnosing AdCC when histology isunclear in the surgical specimen. -
Citations
Citations to this article as recorded by- Pleomorphic adenoma: A comprehensive review
Wilson Duplessis, Jason K. Wasserman
Seminars in Diagnostic Pathology.2026; 43(2): 150991. CrossRef - Pleomorphic adenoma gene 1 (PLAG1) protects p53-/- myoepithelial cells from mitochondria-related apoptosis caused by hypoxia
Nodoka Kindaichi, Yoshiki Mukudai, Yuzo Abe, Masataka Watanabe, Maki Nara, Konomi Yamada, Asami Houri, Toshikazu Shimane, Tatsuo Shirota
Journal of Oral and Maxillofacial Surgery, Medicine, and Pathology.2025; 37(4): 654. CrossRef - Retrospective Clinicopathological Study of 33 Cases of Pleomorphic Salivary Adenoma Diagnosed in Benghazi
Siraj S. Najem, Elhoni Ashour, Rehab Elmaddani, Ali M. Elmurtadi
Libyan Journal of Dentistry .2025; 8(2): 29. CrossRef - Pleomorphic adenoma of palatal minor salivary glands
Afrah Aldelaimi, Tahrir Aldelaimi, Suzan Abdulkareem
Revista Española de Cirugía Oral y Maxilofacial.2025;[Epub] CrossRef - Immunohistochemical Characterization of a Large Cohort of Triple Negative Breast Cancer
Rachel Han, Sharon Nofech-Mozes, Dina Boles, Hannah Wu, Nikolina Curcin, Elzbieta Slodkowska
International Journal of Surgical Pathology.2024; 32(2): 239. CrossRef - Proceedings of the 2024 North American Society of Head and Neck Pathology Companion Meeting, Baltimore, MD, March 24, 2024: Navigating Ancillary Studies in Basaloid/Blue Salivary Tumors
Kristine S. Wong
Head and Neck Pathology.2024;[Epub] CrossRef - Insights into the molecular alterations of PLAG1 and HMGA2 associated with malignant phenotype acquisition in pleomorphic adenoma
Reydson Alcides de Lima-Souza, Gustavo de Souza Vieira, Talita de Carvalho Kimura, João Figueira Scarini, Luccas Lavareze, Tayná Figueiredo Maciel, Moisés Willian Aparecido Gonçalves, Erika Said Abu Egal, Albina Altemani, Fernanda Viviane Mariano
Critical Reviews in Oncology/Hematology.2024; 204: 104494. CrossRef - Expanding the Molecular Spectrum of Carcinoma Ex Pleomorphic Adenoma
Reydson Alcides de Lima-Souza, Albina Altemani, Michal Michal, Fernanda Viviane Mariano, Ilmo Leivo, Alena Skálová
American Journal of Surgical Pathology.2024; 48(12): 1491. CrossRef - Utility of SOX10 and estrogen receptor immunohistochemical expression in endometrial carcinoma of Egyptian patients
Mona A. Kora, Alyaa A. Moselhy, Rania A. Abdallah
Egyptian Journal of Pathology.2024; 44(2): 190. CrossRef - Exploring Advanced Diagnostic Techniques for Salivary Gland Disorders: A Narrative Overview
Chuan-Xiang Li-, Liu Zhang, Ya-Ru Yan, Yong-Jie Ding, Ying-Ni Lin, Jian-Ping Zhou, Ning Li, Hong-Peng Li, Shi-Qi Li, Xian-Wen Sun, Qing-Yun Li
Asian journal of Current Research in Clinical Cancer.2024; 4(1): 1. CrossRef - The Challenge of “Monomorphic” Mucoepidermoid Carcinoma—Report of a Rare Case with Pure Spindle-Clear Cell Morphology
Xinyi Qu, Edwin Jun Chen Chew, Sathiyamoorthy Selvarajan, Bingcheng Wu, Abbas Agaimy, Fredrik Petersson
Head and Neck Pathology.2023; 17(3): 864. CrossRef - SOX10
Albert L Sy, Mai P Hoang
Journal of Clinical Pathology.2023; 76(10): 649. CrossRef - Activating Transcription Factor 1 (ATF1) Immunohistochemical Marker Distinguishes HCCC from MEC
Wafaey Badawy, Asmaa S. Abdelfattah, Haneen A. Sallam
Journal of Molecular Pathology.2023; 4(3): 178. CrossRef - Rare case of pleomorphic adenoma presenting as peritonsilar tumor
Anđelina Jovanović, Svetlana Valjarević, Milan Jovanović
Medicinska istrazivanja.2023; 56(3): 95. CrossRef - Pleomorphic Adenoma of a Minor Salivary Gland of the Hard Palate: A Case Report
Ishank Panchal, Anil Wanjari
Cureus.2023;[Epub] CrossRef - Advanced Diagnostic Methods for Salivary Glands Diseases: A Narrative Review Study
Malak Mohammed AlOsaimi, Abdulaziz Mohammed AlSubaheen, Taif Saleh Jameel, Rand Abdulrahman AlSalamah, Dalal Naseh AlAnzi, Norah Ameen AlOushan, Fahad Fadhel AlShammari, Cristalle Soman
Clinical Cancer Investigation Journal.2023; 12(4): 19. CrossRef - Clinical Significance of SOX10 Expression in Human Pathology
Hisham F. Bahmad, Aran Thiravialingam, Karthik Sriganeshan, Jeffrey Gonzalez, Veronica Alvarez, Stephanie Ocejo, Alvaro R. Abreu, Rima Avellan, Alejandro H. Arzola, Sana Hachem, Robert Poppiti
Current Issues in Molecular Biology.2023; 45(12): 10131. CrossRef - NR4A3 Immunostain Is a Highly Sensitive and Specific Marker for Acinic Cell Carcinoma in Cytologic and Surgical Specimens
Kartik Viswanathan, Shaham Beg, Bing He, Taotao Zhang, Richard Cantley, Daniel J Lubin, Qiuying Shi, Zahra Maleki, Saeed Asiry, Rema Rao, Nora Katabi, Masato Nakaguro, William C Faquin, Peter M Sadow, Momin T Siddiqui, Theresa Scognamiglio
American Journal of Clinical Pathology.2022; 157(1): 98. CrossRef - Recent Advances on Immunohistochemistry and Molecular Biology for the Diagnosis of Adnexal Sweat Gland Tumors
Nicolas Macagno, Pierre Sohier, Thibault Kervarrec, Daniel Pissaloux, Marie-Laure Jullie, Bernard Cribier, Maxime Battistella
Cancers.2022; 14(3): 476. CrossRef - Diagnostic accuracy of human transcriptional activator (Myb) expression by ELISA technique versus immunohistochemistry in detecting salivary gland carcinomas
Yousra Refaey, OlfatGamil Shaker, Ayman Abdelwahab, ImanAdel Mohamed Abdelmoneim, Fat’heyaMohamed Zahran
Journal of International Oral Health.2022; 14(1): 61. CrossRef - SLUG is a key regulator of epithelial-mesenchymal transition in pleomorphic adenoma
Hyesung Kim, Seung Bum Lee, Jae Kyung Myung, Jeong Hwan Park, Eunsun Park, Dong Il Kim, Cheol Lee, Younghoon Kim, Chul-Min Park, Min Bum Kim, Gil Chai Lim, Bogun Jang
Laboratory Investigation.2022; 102(6): 631. CrossRef - Assessment of MEF2C as a novel myoepithelial marker using normal salivary gland and pleomorphic adenoma: An immunohistochemical study
Ikuko Takakura, Satoko Kujiraoka, Rika Yasuhara, Junichi Tanaka, Fumio Ide, Kenji Mishima
Journal of Oral and Maxillofacial Surgery, Medicine, and Pathology.2022; 34(4): 523. CrossRef - Update on selective special types of breast neoplasms: Focusing on controversies, differential diagnosis, and molecular genetic advances
Shi Wei
Seminars in Diagnostic Pathology.2022; 39(5): 367. CrossRef - Cutaneous Melanomas: A Single Center Experience on the Usage of Immunohistochemistry Applied for the Diagnosis
Costantino Ricci, Emi Dika, Francesca Ambrosi, Martina Lambertini, Giulia Veronesi, Corti Barbara
International Journal of Molecular Sciences.2022; 23(11): 5911. CrossRef - Distinct clinicopathological and genomic features in solid and basaloid adenoid cystic carcinoma of the breast
Juan Ji, Fang Zhang, Fanglei Duan, Hong Yang, Jun Hou, Yang Liu, Jie Dai, Qiong Liao, Xian Chen, Qingsong Liu
Scientific Reports.2022;[Epub] CrossRef - NR4A3 fluorescence in situ hybridization analysis in cytologic and surgical specimens of acinic cell carcinoma
Qiuying Shi, Bin Zhang, Caroline Bsirini, Liqiong Li, Ellen J. Giampoli, Kelly R. Magliocca, Michelle Reid, Zhongren Zhou
Human Pathology.2022; 127: 86. CrossRef - Evaluation of NR4A3 immunohistochemistry (IHC) and fluorescence in situ hybridization and comparison with DOG1 IHC for FNA diagnosis of acinic cell carcinoma
John M. Skaugen, Raja R. Seethala, Simion I. Chiosea, Michael S. Landau
Cancer Cytopathology.2021; 129(2): 104. CrossRef -
MYB-NFIB Translocation by FISH in Adenoid Cystic Carcinoma of the Head and Neck in Nigerian Patients: A Preliminary Report
Adepitan A. Owosho, Olufunlola M. Adesina, Oluwole Odujoko, Olujide O. Soyele, Akinwumi Komolafe, Robert Bauer, Kallie Holte, Kurt F. Summersgill
Head and Neck Pathology.2021; 15(2): 433. CrossRef - Liquid-based cytology of oral brushings in a case of adenoid cystic carcinoma arising from the palate
Ryo MAKINO, Akihiko KAWAHARA, Hideyuki ABE, Yorihiko TAKASE, Chihiro FUKUMITSU, Kazuya MURATA, Tomoko YOSHIDA, Yukako SHINODA, Yoshiki NAITO, Jun AKIBA
The Journal of the Japanese Society of Clinical Cytology.2021; 60(1): 33. CrossRef - MYB Translocations in Both Myoepithelial and Ductoglandular Epithelial Cells in Adenoid Cystic Carcinoma: A Histopathologic and Genetic Reappraisal in Six Primary Cutaneous Cases
Keisuke Goto, Kazuyoshi Kajimoto, Takashi Sugino, Shin-ichi Nakatsuka, Makoto Yoshida, Mai Noto, Michihiro Kono, Toshihiro Takai
The American Journal of Dermatopathology.2021; 43(4): 278. CrossRef - Co-expression of Myoepithelial and Melanocytic Features in Carcinoma Ex Pleomorphic Adenoma
Costantino Ricci, Federico Chiarucci, Francesca Ambrosi, Tiziana Balbi, Barbara Corti, Ottavio Piccin, Ernesto Pasquini, Maria Pia Foschini
Head and Neck Pathology.2021; 15(4): 1385. CrossRef - Juvenile onset pleomorphic adenoma presenting as giant tumor of parotid gland in a young female
Surender Verma, Shivika Aggarwal, Pradeep Garg, Anjali Verma, Mridul Gera, Swaran S. Yadav
Journal of Dr. NTR University of Health Sciences.2021; 10(4): 286. CrossRef - Cytopathology and diagnostics of Warthin's tumour
Mirna Sučić, Nives Ljubić, Leila Perković, Dunja Ivanović, Leo Pažanin, Tena Sučić Radovanović, Dubravka Župnić‐Krmek, Fabijan Knežević
Cytopathology.2020; 31(3): 193. CrossRef - Clear cell papillary neoplasm of the breast with MAML2 gene rearrangement: Clear cell hidradenoma or low-grade mucoepidermoid carcinoma?
Raima A. Memon, Carlos N Prieto Granada, Shi Wei
Pathology - Research and Practice.2020; 216(10): 153140. CrossRef
- Pleomorphic adenoma: A comprehensive review
- Hyalinizing Trabecular Tumor of the Thyroid Gland, a Diagnostic Challenge in Fine-Needle Aspiration Cytology: Case Report
- Ye-Young Rhee, Hong Kyu Jung, Se Hoon Kim, Soo Hee Kim
- J Pathol Transl Med. 2018;52(4):252-256. Published online June 11, 2018
- DOI: https://doi.org/10.4132/jptm.2018.04.28
- 11,844 View
- 187 Download
- 8 Web of Science
- 11 Crossref
-
 Abstract
Abstract
 PDF
PDF - Hyalinizing trabecular tumor (HTT) is a rare thyroid tumor with low to minimal malignant potential. HTT is often misinterpreted as other thyroid tumors, including papillary thyroid carcinoma (PTC) and medullary thyroid carcinoma (MTC), on fine-needle aspiration (FNA) cytology, because of its overlapping cytologic features, such as nuclear grooves and intranulcear pseudoinclusions. Although cytopathologists cannot definitely conclude HTT by FNA cytology, suspicion of HTT is necessary to avoid misdiagnosing HTT as PTC or MTC and to avoid unnecessary aggressive treatment. Here, we report a case of HTT with novel cytologic features in CellPrep liquid based cytology that was diagnosed as suspicious for papillary carcinoma by FNA and finally diagnosed as HTT in the surgical specimen.
-
Citations
Citations to this article as recorded by- Hyalinizing trabecular tumor of the thyroid: A comprehensive review of clinicopathological features, diagnostic dilemmas, and emerging molecular insights
Yinghe Huang, Shanshan Liu, Yilei Wen
Annals of Diagnostic Pathology.2026; 80: 152539. CrossRef - Plasma cells and plasmacytoid features in thyroid lesions
Qianqian Zhang, Angela Feraco, Belen Padial Urtueta, Elisabetta Merenda, Luisa Cioni, Alessia Piermattei, Patrizia Straccia, Federica Cianfrini, Antonino Mule, Liron Pantanowitz, Esther Diana Rossi
Virchows Archiv.2026;[Epub] CrossRef - Hyalinizing Trabecular Tumor: A Case Series with Literature Review
Abdulwahid M. Salih, Rebaz O. Mohammed, Hiwa O. Baba, Shko H. Hassan, Muhammed Bag A. Ali, Imad J. Habibullah, Imad S. Sedeeq, Dyari Q. Hamad, Karokh K. Mohammed, Marwan N. Hassan, Abdullah A. Qadir, Harun A. Ahmed, Meer M. Abdulkareem, Fahm
Judi Clinical Journal.2025; 1(1): 1. CrossRef - Cytomorphological traits of fine-needle aspirates of hyalinizing trabecular tumor of the thyroid gland: A brief report
Fei Wang, Yufei Liu
Indian Journal of Pathology and Microbiology.2024; 67(1): 128. CrossRef - Total thyroidectomy can still remain the method of choice in some Bethesda III cases
Jindrich Lukas, Barbora Hintnausova, Vlasta Sykorova, Martin Syrucek, Marek Maly, Jaroslava Duskova
Biomedical Papers.2023; 167(1): 61. CrossRef - Diagnostic clues for hyalinizing trabecular tumor on fine needle aspiration cytology
Lone Nielsen, Ana María Colino Gallardo, Pablo Pérez Alonso, Luis Ortega Medina, Esthefanía Latorre García, Cristina Díaz del Arco, Reyes Bergillos Jiménez, Lorenzo Alarcón García, Marta Cruz Blanco, Jesús Vega González, Montserrat De la Torre Serrano, Ma
Cytojournal.2023; 20: 19. CrossRef - Clinical Characteristics of the Hyalinizing Trabecular Tumor
Byung-Chang Kim, Shin Jeong Pak, Jae Won Cho, Won Woong Kim, Yu-mi Lee, Tae-Yon Sung, Jung Hwan Baek, Ki-Wook Chung
Journal of Endocrine Surgery.2022; 22(4): 116. CrossRef - A Case of Multifocal Hyalinizing Trabecular Tumors of the Thyroid
Gland
Suhwan Jeong, Hanaro Park
Journal of Clinical Otolaryngology Head and Neck Surgery.2021; 32(3): 308. CrossRef - The Diagnosis of Hyalinizing Trabecular Tumor: A Difficult and Controversial Thyroid Entity
Esther Diana Rossi, Mauro Papotti, William Faquin, Luigi Maria Larocca, Liron Pantanowitz
Head and Neck Pathology.2020; 14(3): 778. CrossRef - A large series of hyalinizing trabecular tumors: Cytomorphology and ancillary techniques on fine needle aspiration
Marco Dell’Aquila, Carmen Gravina, Alessandra Cocomazzi, Sara Capodimonti, Teresa Musarra, Stefania Sfregola, Vincenzo Fiorentino, Luca Revelli, Maurizio Martini, Guido Fadda, Liron Pantanowitz, Luigi Maria Larocca, Esther Diana Rossi
Cancer Cytopathology.2019; 127(6): 390. CrossRef - GLIS rearrangements in thyroid nodules: A key to preoperative diagnosis of hyalinizing trabecular tumor
Marina N. Nikiforova, Yuri E. Nikiforov, N. Paul Ohori
Cancer Cytopathology.2019; 127(9): 560. CrossRef
- Hyalinizing trabecular tumor of the thyroid: A comprehensive review of clinicopathological features, diagnostic dilemmas, and emerging molecular insights
- Fine-Needle Aspiration Cytology of Carcinosarcoma in the Salivary Gland: An Extremely Rare Case Report
- Hyo Jung An, Hye Jin Baek, Jin Pyeong Kim, Min Hye Kim, Dae Hyun Song
- J Pathol Transl Med. 2018;52(2):136-139. Published online December 28, 2017
- DOI: https://doi.org/10.4132/jptm.2017.07.27
- 8,736 View
- 136 Download
- 5 Web of Science
- 5 Crossref
-
 Abstract
Abstract
 PDF
PDF - Carcinosarcoma of the salivary gland is an extremely rare tumor that is composed of both malignant epithelial and mesenchymal components. Diagnosing carcinosarcoma with fine-needle aspiration cytology is challenging because of its overlapping cytomorphologic characteristics with other high-grade malignant salivary gland tumors. Among the many features, including pleomorphic oncocytoid epithelial components, necrotic background, and mitoses, recognizing the singly scattered atypical spindle cells is most essential in carcinosarcoma. We present a case of a 66-year-old male patient with characteristic features of carcinosarcoma, who was successfully treated by wide local excision and subsequent radiation therapy.
-
Citations
Citations to this article as recorded by- A Rare Osteoid Forming Carcinosarcoma Ex‐Pleomorphic Adenoma of the Parotid Gland
Nyein Nyein Htun, Daniel Nguyen, Beverly Y. Wang, Anoosh Montaser, Behdokht Nowroozizadeh, Suraiya Saleem
Case Reports in Pathology.2025;[Epub] CrossRef - Carcinosarcoma of the parotid gland: a case report and review of the literature
Swachi Jain, Mohammed Abdelwahed, Daniel Hector Chavarria, Lucio Pereira, Gary Stone, Alan Johnson, Jian Yi Li
Journal of Medical Case Reports.2024;[Epub] CrossRef - Is Primary Poorly Differentiated Sarcomatoid Malignancy of the Parotid Gland Sarcomatoid Undifferentiated/Dedifferentiated Melanoma? Report of Three Unusual Cases Diagnosed by Fine-Needle Aspiration Combined with Histological, Immunohistochemical, and Mol
Jerzy Klijanienko, Julien Masliah-Planchon, Olivier Choussy, Guillaume Rougier, Antoine Dubray Vautrin, Maria Lesnik, Nathalie Badois, Wahib Ghanem, Jan Klos, Christophe Le Tourneau, Gregoire Marret, Raymond Barnhill, Adel K. El-Naggar
Acta Cytologica.2024; 68(2): 107. CrossRef - Carcinosarcoma of the deep lobe of the parotid gland in the parapharyngeal region: A case report
Yue-Yang Tang, Gui-Quan Zhu, Zhi-Jian Zheng, Li-Hong Yao, Zi-Xin Wan, Xin-Hua Liang, Ya-Ling Tang
World Journal of Clinical Cases.2023; 11(31): 7663. CrossRef - Carcinosarcoma of Submandibular Salivary Gland with a Rare Sarcomatous Variant
Shalini Bhalla, Naseem Akhtar, Puneet Prakash, Malti Kumari, Madhu Mati Goel
Indian Journal of Surgical Oncology.2019; 10(1): 61. CrossRef
- A Rare Osteoid Forming Carcinosarcoma Ex‐Pleomorphic Adenoma of the Parotid Gland
- Duodenal Adenocarcinoma of Brunner Gland Origin: A Case Report
- Ji Hye Moon, Kyoungbun Lee, Han-Kwang Yang, Woo Ho Kim
- J Pathol Transl Med. 2018;52(3):179-182. Published online December 27, 2017
- DOI: https://doi.org/10.4132/jptm.2017.10.09
- 10,300 View
- 175 Download
- 7 Web of Science
- 7 Crossref
-
 Abstract
Abstract
 PDF
PDF - We report a case of adenocarcinoma originating from the duodenal Brunner glands in a 47-year-old female patient. The lesion was 0.8 cm in extent and located at the posterior wall of the first part of the duodenum. Histologically, the tumor showed transition from non-neoplastic Brunner glands through dysplastic epithelium into adenocarcinoma. The carcinoma cells were strongly positive for MUC6 protein, which is an epithelial marker for the Brunner glands. Tumor protein p53 was overexpressed in the carcinoma cells, but not in the non-neoplastic or dysplastic epithelium. Dystrophic calcification was predominant. This is the first case report of duodenal adenocarcinoma of Brunner gland origin in Korea.
-
Citations
Citations to this article as recorded by- Duodenal Adenocarcinoma with a Gastric Phenotype Demonstrating a Rapidly Progressive Course
Sho Matsuyama, Akihisa Fukuda, Nobukazu Agatsuma, Masahito Hoki, Takahiro Utsumi, Hiroshi Seno
Internal Medicine.2025; 64(11): 1684. CrossRef - Relationship Between Immunophenotypes, Genetic Profiles, and Clinicopathologic Characteristics in Small Bowel Adenocarcinoma
Aitoshi Hoshimoto, Atsushi Tatsuguchi, Takeshi Yamada, Sho Kuriyama, Ryohei Hamakubo, Takayoshi Nishimoto, Jun Omori, Naohiko Akimoto, Katya Gudis, Keigo Mitsui, Shu Tanaka, Shunji Fujimori, Tsutomu Hatori, Akira Shimizu, Katsuhiko Iwakiri
American Journal of Surgical Pathology.2024; 48(2): 127. CrossRef - Recurrence of Brunner Gland Adenocarcinoma After Duodenectomy and Gastrojejunostomy
Paul Hong, Marcel Ghanim, Abdul Haseeb, Xianzhong Ding, Ayokunle T. Abegunde
ACG Case Reports Journal.2023; 10(6): e01060. CrossRef - HER2-positive adenocarcinoma arising from heterotopic pancreas tissue in the duodenum: A case report
Yoshifumi S Hirokawa, Takashi Iwata, Yoshinaga Okugawa, Koji Tanaka, Hiroyuki Sakurai, Masatoshi Watanabe
World Journal of Gastroenterology.2021; 27(28): 4738. CrossRef - Brunner’s gland adenoma is a rare cause of upper gastrointestinal bleeding: A case report and literature review
Nader Bakheet, Ahmed Cordie, Mohamed Nabil alkady, Ibrahim Naguib
Arab Journal of Gastroenterology.2020; 21(2): 122. CrossRef - Brunner's Gland Adenocarcinoma in an Aged Western Gorilla (Gorilla gorilla)
Jun Sasaki, Ippei Muneuchi, Kanako Ushio, Chihiro Sochi, Youichi Irie, Kazunori Yoshizumi, Waturu Hashimoto
Journal of Comparative Pathology.2020; 181: 47. CrossRef - Adenocarcinoma of the duodenum arising from Brunner’s gland resected by partial duodenectomy: a case report
Tetsuya Mochizuki, Nobuaki Fujikuni, Koichi Nakadoi, Masahiro Nakahara, Kazuaki Tanabe, Shuji Yonehara, Toshio Noriyuki
Surgical Case Reports.2019;[Epub] CrossRef
- Duodenal Adenocarcinoma with a Gastric Phenotype Demonstrating a Rapidly Progressive Course
- Cytologic Characteristics of Thymic Adenocarcinoma with Enteric Differentiation: A Study of Four Fine-Needle Aspiration Specimens
- Ah-Young Kwon, Joungho Han, Hae-yon Cho, Seokhwi Kim, Heejin Bang, Jiyeon Hyeon
- J Pathol Transl Med. 2017;51(5):509-512. Published online August 4, 2017
- DOI: https://doi.org/10.4132/jptm.2017.03.22
- 9,362 View
- 126 Download
- 6 Web of Science
- 8 Crossref
-
 Abstract
Abstract
 PDF
PDF - Thymic adenocarcinoma is extremely rare. Although its histologic features have been occasionally reported, a lack of description of the cytologic features has hampered the prompt and accurate diagnosis of this condition. Herein, we describe the cytologic findings and histology of four aspiration cytology specimens of thymic adenocarcinoma. The specimens were obtained from primary tumors, metastatic lymph nodes, and pericardial effusions. All four specimens showed three-dimensional glandular clusters with a loss of polarity and nuclear overlapping. One specimen had extensive extracellular mucinous material. Three specimens contained tumor cells with intracytoplasmic vacuoles. While the specimen with extracellular mucin showed relatively mild cytologic atypia, other specimens exhibited more atypical cytologic changes: irregular nuclear membranes, a coarse chromatin pattern, and prominent nucleoli. The cytologic features were correlated with the histologic features in each case of enteric type thymic adenocarcinoma. The differential diagnosis included other thymic carcinomas, yolk sac tumors, and metastatic adenocarcinoma from the lung or colorectum.
-
Citations
Citations to this article as recorded by- Primary thymic adenocarcinoma with signet ring cell-like features: A case report and literature review
Ya Jiang, Ziran Gao, Wenmang Xu, Yuanyuan Wang
Asian Journal of Surgery.2026; 49(1): 589. CrossRef - Case report: Primary adenocarcinoma NOS of the thymus and cytological features
Jonathan Willner, Osvaldo Hernandez, Lea Azour, Andre L. Moreira
Diagnostic Cytopathology.2023;[Epub] CrossRef - Systemic chemotherapy for unresectable or recurrent primary thymic adenocarcinoma of enteric type
Xiaofang Gao
International Cancer Conference Journal.2022; 12(1): 46. CrossRef - Thymic adenocarcinoma accompanied by type A thymoma and pulmonary minimally invasive adenocarcinoma and harboring distinct gene alterations
Yi-Wen Zheng, Lin-Lin Bai, Gui-Yang Jiang, Xu-Yong Lin, Yang Liu, Hong-Tao Xu
Medicine.2021; 100(15): e25254. CrossRef - A case report: primary thymic adenocarcinoma with enteric differentiation
Yuuki Kou, Hirokazu Tanaka, Nobuhisa Yamazaki, Hiroyoshi Watanabe, Makoto Sonobe
The Journal of the Japanese Association for Chest Surgery.2020; 34(2): 107. CrossRef - Primary thymic adenocarcinoma with an aggressive clinical course: An autopsy case showing signet ring cell‐like features
Ayako Shiono, Takashi Fujino, Kyoichi Kaira, Tomomi Kato, Masanori Yasuda, Kunihiko Kobayashi, Hiroshi Kagamu
Thoracic Cancer.2020; 11(12): 3609. CrossRef - Primary Thymic Signet Ring Cell Adenocarcinoma: A Currently Unrecognized Variant
Richard Benedict Supan Roxas, Marie Christine Fajatin Bernardo, Araceli Pacis Jacoba, Janet Lim-Dy, Anarose Cariaga Alvarado, Jasna Metovic, Laura Annaratone, Mauro Papotti
International Journal of Surgical Pathology.2019; 27(3): 315. CrossRef - Disseminated and massive tumor burden in a case of primary thymic mucinous adenocarcinoma
Hui-Wen Liu, Chih-Yi Liu, Yi-Chen Yeh
Journal of Cancer Research and Practice.2019; 6(3): 151. CrossRef
- Primary thymic adenocarcinoma with signet ring cell-like features: A case report and literature review
- Mammary Carcinoma Arising in Microglandular Adenosis: A Report of Five Cases
- Mimi Kim, Milim Kim, Yul Ri Chung, So Yeon Park
- J Pathol Transl Med. 2017;51(4):422-427. Published online April 4, 2017
- DOI: https://doi.org/10.4132/jptm.2016.11.11
- 16,233 View
- 204 Download
- 14 Web of Science
- 14 Crossref
-
 Abstract
Abstract
 PDF
PDF - Mammary carcinoma arising in microglandular adenosis (MGA) is extremely rare, and MGA is regarded as a non-obligate precursor of triple-negative breast cancer. We report five cases of carcinoma arising in MGA of the breast. All cases showed a spectrum of proliferative lesions ranging from MGA to atypical MGA, ductal carcinoma in situ or invasive carcinoma. Immunohistochemically, all cases were triple-negative and expression of S-100 protein gradually decreased as the lesions progressed from MGA to atypical MGA and carcinoma. Three cases showed acinic cell differentiation with reactivity to α1-antitrypsin, and one case was metaplastic carcinoma. During clinical follow-up, one patient developed local recurrence. Carcinoma arising in MGA is a rare but distinct subset of triple-negative breast cancer with characteristic histologic and immunohistochemical findings.
-
Citations
Citations to this article as recorded by- Microglandular adenosis associated with invasive breast carcinoma: Tertiary care oncology centre experience of an under-recognized entity
Ayushi Sahay, Asawari Patil, Trupti Pai, Poonam Panjwani, Shalaka Joshi, Palak Popat Thakkar, Sangeeta B. Desai, Tanuja M. Shet
Annals of Diagnostic Pathology.2026; 80: 152576. CrossRef - Two similar but distinct types of breast acinar cell carcinoma: evidence from histological, immunohistochemical and molecular features
Mingfang Sun, Lin Fu, Hongjiu Ren, Jian Wang, Xuyong Lin, Qingfu Zhang
Histopathology.2025; 87(6): 904. CrossRef - Acinic Cell Carcinoma of the Breast: A Population‐Based Clinicopathologic Study
Faruk Skenderi, Giridhara Rathnaiah Babu, Una Glamoclija, Emir Veledar, Zoran Gatalica, Janez Lamovec, Semir Vranic
Cancer Reports.2025;[Epub] CrossRef - Acinic Cell Carcinoma of the Breast: A Case Report and Review of Literature
Ihab S Atta
Cureus.2024;[Epub] CrossRef - Histological and electron microscopic features of the extracellular matrix of invasive breast ductal carcinoma of no special type. Report of 5 cases and literature review
M.V. Mnikhovich, A.V. Romanov, T.M. Nguyen, T.V. Bezuglova, D.A. Pastukhova
Ultrastructural Pathology.2023; 47(4): 261. CrossRef - Ductal carcinoma in situ arises from microglandular adenosis and atypical microglandular adenosis in a young woman
Nguyen Thu Huong, Tran-Thi Hue, Nguyen Duy Hung, Nguyen Minh Duc
Journal of Clinical Imaging Science.2023; 13: 15. CrossRef - Primary acinic cell carcinoma of the breast: A case report and literature review
Zhi-Min Deng, Yi-Ping Gong, Feng Yao, Ma-Li Wu, Zi-Tao Wang, Jing-Ping Yuan, Yan-Xiang Cheng
Heliyon.2023; 9(9): e20160. CrossRef - Two cases of mammary acinic cell carcinomas with microglandular structures mimicking microglandular adenosis
Fang Yu, Li Niu, Bicheng Wang, Wei Fan, Jian Xu, Qiongrong Chen
Pathology International.2022; 72(6): 343. CrossRef - Metaplastic Matrix-Producing Carcinoma and Apocrine Lobular Carcinoma In Situ Associated with Microglandular Adenosis: A Unique Case Report
Nektarios Koufopoulos, Dionysios Dimas, Foteini Antoniadou, Kyparissia Sitara, Dimitrios Balalis, Ioannis Boutas, Alina Roxana Gouloumis, Adamantia Kontogeorgi, Lubna Khaldi
Diagnostics.2022; 12(6): 1458. CrossRef - Salivary gland-like breast tumors: A review of diagnostic features and prognosis
Vicente Marco
Revista de Senología y Patología Mamaria.2022; 35: S43. CrossRef - Acinic cell carcinoma of the breast: A comprehensive review
Azra Ajkunic, Faruk Skenderi, Nada Shaker, Saghir Akhtar, Janez Lamovec, Zoran Gatalica, Semir Vranic
The Breast.2022; 66: 208. CrossRef - Unusual cerebrospinal fluid finding of intracytoplasmic granules in metaplastic carcinoma of the breast with acinar differentiation
Ruhani Sardana, Anil V Parwani, Xiaoyan Cui, Jayalakshmi Balakrishna
Diagnostic Cytopathology.2021;[Epub] CrossRef - Salivary gland-type mammary carcinoma arising in microglandular adenosis: A case report and clinicopathological review of the literature
Victoria Rico, Yukiko Shibahara, Marjorie Monteiro, Elzbieta Slodkowska, Samantha Tam, Pearl Zaki, Carlo De Angelis, Edward Chow, Katarzyna Joanna Jerzak
Cancer Treatment and Research Communications.2020; 24: 100178. CrossRef - Triple negative metaplastic breast carcinoma presenting in the background of atypical microglandular adenosis with candidacy for atezolizumab immunotherapy
Hector Chavarria, Sean Hacking, Cao Jin, Nidhi Kataria, Florin Glodan, Tawfiqul Bhuiya, Mansoor Nasim
Human Pathology: Case Reports.2020; 21: 200398. CrossRef
- Microglandular adenosis associated with invasive breast carcinoma: Tertiary care oncology centre experience of an under-recognized entity
- Morphological and Functional Changes in the Thyroid Follicles of the Aged Murine and Humans
- Junguee Lee, Shinae Yi, Yea Eun Kang, Hyeon-Woo Kim, Kyong Hye Joung, Hae Joung Sul, Koon Soon Kim, Minho Shong
- J Pathol Transl Med. 2016;50(6):426-435. Published online October 14, 2016
- DOI: https://doi.org/10.4132/jptm.2016.07.19
- 19,967 View
- 484 Download
- 46 Web of Science
- 48 Crossref
-
 Abstract
Abstract
 PDF
PDF - Background
Although both thyroid histology and serum concentrations of hormones are known to change with age, only a few reports exist on the relationship between the age-related structural and functional changes of the thyroid follicles in both mice and humans. Our objectives were to investigate age-related histological changes of the thyroid follicles and to determine whether these morphological changes were associated with the functional activity of the follicles.
Methods
The thyroid glands of mice at 18 weeks and at 6, 15, and 30 months of age were histologically examined, and the serum levels of thyroid hormones were measured in 11-week-old and 20-month-old mice. Samples of human thyroid tissue from 10 women over 70 years old and 10 women between 30 and 50 years of age were analyzed in conjunction with serum thyroid hormone level.
Results
The histological and functional changes observed in the thyroid follicles of aged mice and women were as follows: variable sizing and enlargement of the follicles; increased irregularity of follicles; Sanderson’s polsters in the wall of large follicles; a large thyroglobulin (Tg) globule or numerous small fragmented Tg globules in follicular lumens; oncocytic change in follicular cells; and markedly dilated follicles empty of colloid. Serum T3 levels in 20-month-old mice and humans were unremarkable.
Conclusions
Thyroid follicles of aged mice and women show characteristic morphological changes, such as cystic atrophy, empty colloid, and Tg globules. -
Citations
Citations to this article as recorded by- OCT-Based Differentiation of papillary thyroid carcinoma through quantitative optical attenuation and microstructural characterization
Zengming Li, Chao Zhao, Weizheng Mao, Xiaofeng Shi, Hang Zhao, Jun Ma
Journal of Modern Optics.2026; 73(3): 219. CrossRef - Influence of donor–recipient age differences in allogenic rat thyroid transplantation
Ayumi Arauchi, Katsuhisa Matsuura, Tatsuya Shimizu
Regenerative Therapy.2026; 31: 101047. CrossRef - Histological Evidence of Thyroid Disruption in Wild Mice from Conventional and Organic Farming Environments
Nádia M. P. Coelho, Ricardo Camarinho, Patrícia Garcia, Filipe Bernardo, Armindo S. Rodrigues
Environments.2026; 13(2): 66. CrossRef - Risk factors for thyroid nodules in a health examination population: a cross-sectional study and development of a simplified predictive model
Hangtian Yu, Jingle Cao, Jing Han, Yang Li, Wenyu Li, Zihan Li, Jinjia Zhang, YaLi Zhang
Frontiers in Endocrinology.2026;[Epub] CrossRef - Iodine and Thyroid Dysfunction in Ageing: Nutritional, Pharmacologic, and Microbial Modifiers in Older Adults
Corina-Aurelia Zugravu, Marta Petre, Ciprian Constantin
Geriatrics.2026; 11(1): 12. CrossRef - Advances in Thyroid Gland Regeneration: The Integrated Approach of Cell Biology and Bioengineering
Tomasz Górnicki, Maria Grazia Soriano, Giuseppe Raffaele, Gabriele Maria Fortunato, Walis Jones, Antonella Cecchettini, Patrycja Szymczyk-Ziółkowska, Krzysztof Kaliszewski, Piotr Dzięgiel, Federico Vozzi, Carmelo Di Maria, Bartosz Kempisty, Giovanni Vozzi
Tissue Engineering Part B: Reviews.2026;[Epub] CrossRef - Characterization of primary cilia in different epithelial cells of thyroid gland
B. Pérez-Fernández, V. Vázquez-Román, J. M. Fernández-Santos, I. Martín-Lacave
Histochemistry and Cell Biology.2025;[Epub] CrossRef - Characterizing thyroid follicles histogenesis in the human fetuses: A morphological approach
Chacchu Bhattarai, Phanindra Prasad Poudel, Mahendra Raj Pandey, Chandni Gupta, Sneha Guruprasad Kalthur
Tissue and Cell.2025; 93: 102710. CrossRef - Effect of a Low-Molecular-Weight Allosteric Agonist of the Thyroid-Stimulating Hormone Receptor on Basal and Thyroliberin-Stimulated Activity of Thyroid System in Diabetic Rats
Kira V. Derkach, Alena S. Pechalnova, Viktor N. Sorokoumov, Inna I. Zorina, Irina Y. Morina, Elizaveta E. Chernenko, Egor A. Didenko, Irina V. Romanova, Alexander O. Shpakov
International Journal of Molecular Sciences.2025; 26(2): 703. CrossRef - Human Relevance of Pharmaceutical Drug‐Induced Thyroid Tumors in Rats, Labeling Implications, and Carcinogenicity Study Requirements
B. D. Hollingshead, Z. A. Radi
Journal of Applied Toxicology.2025; 45(8): 1426. CrossRef - Early developmental exposure to sodium perchlorate disrupts thyroid, liver, and testicular development in mice
Kyle Bouten, Roman Parpart, Marielle Calanza, Michael Minicozzi
Integrative And Comparative Biology.2025; 65(1): 11. CrossRef - Experimental induction of hypothyroidism and goitre in goat kids: Clinical, pathological and histomorphometric findings
Luis Adrián Colque-Caro, Claudio Gustavo Barbeito, Dadín Prando Moore, Juan Francisco Micheloud
Research in Veterinary Science.2025; 197: 105926. CrossRef - The role of the primary cilium in thyroid function and dysfunction with implications for thyroid disease
Inés Martín-Lacave, Victoria Vázquez-Román, Beatriz Pérez-Fernández, José María Fernández-Santos
Histochemistry and Cell Biology.2025;[Epub] CrossRef - Genetic Rescue of a Subset of Thyroid Follicular Cells Restores Thyroid Function in Dyshormonogenic Duoxa−/− Mice
Sami Djerbib, Helmut Grasberger, Steven Goossens, Jody Haigh, Lieven Haenebalcke, Tim Pieters, Jukka Kero, Françoise Miot, Xavier De Deken
Thyroid®.2025; 35(11): 1230. CrossRef - Arsenic-Induced Thyroid Hormonal Alterations and Their Putative Influence on Ovarian Follicles in Balb/c Mice
Nandheeswari K, Jayapradha P, Sree Vaishnavi Nalla, Itishree Dubey, Sapana Kushwaha
Biological Trace Element Research.2024; 202(9): 4087. CrossRef - Histological evidence of hypothyroidism in mice chronically exposed to conventional farming
Nádia Coelho, Ricardo Camarinho, Patrícia Garcia, Armindo S. Rodrigues
Environmental Toxicology and Pharmacology.2024; 106: 104387. CrossRef - Optimizing decellularization protocols for human thyroid tissues: a step towards tissue engineering and transplantation
Özge Karabıyık Acar, Gülnihal Bozdağ, Ezgi Hacıhasanoğlu, A Alperen Tuncer, Erhan Aysan, Gamze Torun Köse
Biomedical Materials.2024; 19(4): 045034. CrossRef - A comparative cross-sectional study on the quality of life in Grave’s disease patients: urban vs. rural perspectives
Shivang Mishra, Anurag Kumar Singh, Sumit Rajotiya, Sourav Debnath, Sachin Kumar, Pratima Singh, Snehpreet, Preeti Raj, Mahaveer Singh, Hemant Bareth, Deepak Nathiya, Balvir Singh Tomar
Frontiers in Public Health.2024;[Epub] CrossRef - Effect of thyroid disruption on ovarian development following maternal exposure to Bisphenol S
Lina Chouchene, Sana Boughammoura, Mariem Ben Rhouma, Rania Mlouka, Mohamed Banni, Imed Messaoudi, Kaouthar Kessabi
Environmental Science and Pollution Research.2024; 31(39): 52596. CrossRef - Fish collagen peptides supplementation ameliorates skeletal muscle injury in high-fat diet mice associated with thyroid function recovery
Yuhui Yang, Hao Yang, Xu Tian, Wenhan Jian, Nazish Muzaffar, Guowei Le, Yanli Xie, Peng Li, Yuncong Xu
Journal of Functional Foods.2024; 123: 106612. CrossRef - Thyroid‐on‐a‐Chip: An Organoid Platform for In Vitro Assessment of Endocrine Disruption
Daniel J. Carvalho, Anna M. Kip, Mírian Romitti, Marta Nazzari, Andreas Tegel, Matthias Stich, Christian Krause, Florian Caiment, Sabine Costagliola, Lorenzo Moroni, Stefan Giselbrecht
Advanced Healthcare Materials.2023;[Epub] CrossRef - Serious lesions in Green turtles (Chelonia mydas) afflicted by fatal Spirorchiidiasis found stranded in south and southeastern Brazil
Hassan Jerdy, Bruna Barreto, Max Werneck, Rachel Ann Hauser-Davis, Paula Baldassin, Patrick Gabriel, Aline Luize de Moraes Souza, Maria Aparecida da Silva, Aline Felix, Rachel Ribeiro Rodrigues, Mariah Bianchi, Carla Barbosa, Gessica Gomes Vieira, Lara Ri
International Journal for Parasitology: Parasites and Wildlife.2023; 20: 73. CrossRef - 基于光学相干层析技术的甲状腺乳头状癌图像识别
李增鸣 Li Zengming, 赵潮 Zhao Chao, 张旭 Zhang Xu, 毛伟征 Mao Weizheng, 赵航 Zhao Hang, 史晓凤 Shi Xiaofeng, 马君 Ma Jun
Laser & Optoelectronics Progress.2023; 60(4): 0417002. CrossRef - Differentiating Thyroid Follicular Adenoma from Follicular Carcinoma via G-Protein Coupled Receptor-Associated Sorting Protein 1 (GASP-1)
Yuan Rong, Cesar Torres-Luna, George Tuszynski, Richard Siderits, Frank N. Chang
Cancers.2023; 15(13): 3404. CrossRef - The effects of glyphosate-based herbicide on the hypothalamic-pituitary thyroid axis are tissue-specific and dependent on age exposure
Jeane Maria Oliveira, Jamilli Zenzeluk, Paula Bargi-Souza, Raphael Escorsim Szawka, Marco Aurelio Romano, Renata Marino Romano
Environmental Pollution.2023; 334: 122216. CrossRef - Borealin/CDCA8 deficiency alters thyroid development and results in papillary tumor-like structures
Hortense Didier-Mathon, Athanasia Stoupa, Dulanjalee Kariyawasam, Sonny Yde, Beatrix Cochant-Priollet, Lionel Groussin, Frédéric Sébag, Nicolas Cagnard, Patrick Nitschke, Dominique Luton, Michel Polak, Aurore Carré
Frontiers in Endocrinology.2023;[Epub] CrossRef - Notch signaling in thyrocytes is essential for adult thyroid function and mammalian homeostasis
Lluc Mosteiro, Thi Thu Thao Nguyen, Simona Hankeova, Daniel Alvarez-Sierra, Mike Reichelt, Shannon M. Vandriel, Zijuan Lai, Feroza K. Choudhury, Dewakar Sangaraju, Binita M. Kamath, Alexis Scherl, Ricardo Pujol-Borrell, Robert Piskol, Christian W. Siebel
Nature Metabolism.2023; 5(12): 2094. CrossRef - Toward Systems-Level Metabolic Analysis in Endocrine Disorders and Cancer
Aliya Lakhani, Da Hyun Kang, Yea Eun Kang, Junyoung O. Park
Endocrinology and Metabolism.2023; 38(6): 619. CrossRef - Сравнение морфологических изменений щитовидной железы крыс послевлияния разных видов алиментарной депривации
Р. В. Янко
Biological Journal of Armenia.2022; : 24. CrossRef - Zinc Depletion Inhibits the Synthesis and Secretion of Thyroglobulin by Inducing Endoplasmic Reticulum Stress in PCCL3 Thyroid Cells
Kisang Kwon, Eun-Ryeong Lee, Kyung-Hee Kang, Tae-Sik Hwang, Seung-Whan Kim, Hyewon Park, O-Yu Kwon
International Journal of Biology and Biomedical Engineering.2022; 16: 290. CrossRef - Metimazol ile Oluşturulan Sıçan Hipotiroidi Modelinde Kognitif Fonksiyonlar, Anksiyete ve Depresyon Benzeri Davranışların Değerlendirilmesi: Pilot Çalışma
Oğuzhan Ekin EFE, Selda EMRE AYDINGÖZ, Karl Michael LUX, Eda ÖZER, Ayşegül SÜZER, Meral TUNCER
Uludağ Üniversitesi Tıp Fakültesi Dergisi.2022; 48(2): 147. CrossRef - Loss of thyroid gland circadian PER2 rhythmicity in aged mice and its potential association with thyroid cancer development
Junguee Lee, Hae Joung Sul, Hyunsu Choi, Dong Hyun Oh, Minho Shong
Cell Death & Disease.2022;[Epub] CrossRef - HISTOTOPOGRAPHIC AND STRUCTURAL FEATURES OF CANINE THYROID GLAND FOLLICLES UNDER SINGLE DYNAMIC MOTOR LOADS
Andrey V. Bezdenezhnykh, Sofia S. Kolodezhnaya, Daria E. Nazarova, Anna P. Bavrina
Morphological newsletter.2022; 30(4): 22. CrossRef - Thyroid hormone receptor β sumoylation is required for thyrotropin regulation and thyroid hormone production
Sujie Ke, Yan-Yun Liu, Rajendiran Karthikraj, Kurunthachalam Kannan, Jingjing Jiang, Kiyomi Abe, Anna Milanesi, Gregory A. Brent
JCI Insight.2021;[Epub] CrossRef - Thyroid Gland Alterations in Old-Aged Wistar Rats: A Comprehensive Stereological, Ultrastructural, Hormonal, and Gene Expression Study
Marko Miler, Vladimir Ajdžanović, Jasmina Živanović, Jelena Marković Filipović, Branka Šošić-Jurjević, Verica Milošević
Microscopy and Microanalysis.2021; 27(2): 437. CrossRef - Age-dependent effects on radiation-induced carcinogenesis in the rat thyroid
Mutsumi Matsuu-Matsuyama, Kazuko Shichijo, Katsuya Matsuda, Nariaki Fujimoto, Hisayoshi Kondo, Shiro Miura, Tomomi Kurashige, Yuji Nagayama, Masahiro Nakashima
Scientific Reports.2021;[Epub] CrossRef - Relevant dose of the environmental contaminant, tributyltin, promotes histomorphological changes in the thyroid gland of male rats
Paula Rodrigues-Pereira, Sofia Macedo, Tiago Bordeira Gaspar, Sule Canberk, Samia Selmi-Ruby, Valdemar Máximo, Paula Soares, Leandro Miranda-Alves
Molecular and Cellular Endocrinology.2020; 502: 110677. CrossRef - Morphological and functional changes in neonatally X-irradiated thyroid gland in rats
Nariaki Fujimoto, Mutsumi Matsuu-Matsuyama, Masahiro Nakashima
Endocrine Journal.2020; 67(2): 231. CrossRef - Optical coherence tomography for thyroid pathology: 3D analysis of tissue microstructure
Iulian Emil Tampu, Michaela Maintz, Daniela Koller, Kenth Johansson, Oliver Gimm, Arrigo Capitanio, Anders Eklund, Neda Haj-Hosseini
Biomedical Optics Express.2020; 11(8): 4130. CrossRef - Thyroid surgery in the elderly
Nicole Ruszkay, David Goldenberg, Guy Slonimsky
Operative Techniques in Otolaryngology-Head and Neck Surgery.2020; 31(3): 206. CrossRef - Liver X Receptor β Related to Tumor Progression and Ribosome Gene Expression in Papillary Thyroid Cancer
Seonhyang Jeong, In-Kyu Kim, Hyunji Kim, Moon Jung Choi, Jandee Lee, Young Suk Jo
Endocrinology and Metabolism.2020; 35(3): 656. CrossRef - A historical excursus of diagnostic methods for Hashimoto thyroiditis and Graves' disease
Maria Trovato
Gazzetta Medica Italiana Archivio per le Scienze Mediche.2020;[Epub] CrossRef - Cross‐sectional comparison of health‐span phenotypes in young versus geriatric marmosets
Corinna N. Ross, Jessica Adams, Olga Gonzalez, Edward Dick, Luis Giavedoni, Vida L. Hodara, Kimberley Phillips, Anna D Rigodanzo, Balakuntalam Kasinath, Suzette D. Tardif
American Journal of Primatology.2019;[Epub] CrossRef - Effects of nonylphenols on embryonic development and metamorphosis of Xenopus laevis: FETAX and amphibian metamorphosis toxicity test (OECD TG231)
Yang Xu, Sun Jung Park, Myung Chan Gye
Environmental Research.2019; 174: 14. CrossRef - Basal Autophagy Deficiency Causes Thyroid Follicular Epithelial Cell Death in Mice
Tomomi Kurashige, Yasuyo Nakajima, Mika Shimamura, Mutsumi Matsuyama, Masanobu Yamada, Masahiro Nakashima, Yuji Nagayama
Endocrinology.2019; 160(9): 2085. CrossRef - Aging Is Associated with Low Thyroid State and Organ-Specific Sensitivity to Thyroxine
Helena Rakov, Meri De Angelis, Kostja Renko, Georg Sebastian Hönes, Denise Zwanziger, Lars Christian Moeller, Karl-Werner Schramm, Dagmar Führer
Thyroid®.2019; 29(12): 1723. CrossRef - High-fat, simple-carbohydrate diet intake induces hypothalamic–pituitary–thyroid axis dysregulation in C57BL/6J male mice
Nagaraj Banavara Swarnalatha, Neena Roy, Mahesh Manjunath Gouda, Rajeish Moger, Asha Abraham
Applied Physiology, Nutrition, and Metabolism.2018; 43(4): 371. CrossRef - Biomarker Sensitivity to Vehicle Exhaust in Experimentally Exposed European Starlings
Michelle A. North, Jaime Rodriguez-Estival, Judit E. G. Smits
Environmental Science & Technology.2017; 51(22): 13427. CrossRef
- OCT-Based Differentiation of papillary thyroid carcinoma through quantitative optical attenuation and microstructural characterization
- A Pyloric Gland-Phenotype Ovarian Mucinous Tumor Resembling Lobular Endocervical Glandular Hyperplasia in a Patient with Peutz-Jeghers Syndrome
- Eun Na Kim, Gu-Hwan Kim, Jiyoon Kim, In Ah Park, Jin Ho Shin, Yun Chai, Kyu-Rae Kim
- J Pathol Transl Med. 2017;51(2):159-164. Published online August 22, 2016
- DOI: https://doi.org/10.4132/jptm.2016.07.01
- 10,704 View
- 210 Download
- 10 Web of Science
- 9 Crossref
-
 Abstract
Abstract
 PDF
PDF - We describe an ovarian mucinous neoplasm that histologically resembles lobular endocervical glandular hyperplasia (LEGH) containing pyloric gland type mucin in a patient with Peutz-Jeghers syndrome (PJS). Although ovarian mucinous tumors rarely occur in PJS patients, their pyloric gland phenotype has not been clearly determined. The histopathologic features of the ovarian mucinous tumor were reminiscent of LEGH. The cytoplasmic mucin was stained with periodic acid-Schiff reaction after diastase treatment but was negative for Alcian blue pH 2.5, suggesting the presence of neutral mucin. Immunohistochemically, the epithelium expressed various gastric markers, including MUC6, HIK1083, and carbonic anhydrase-IX. Multiple ligation-dependent probe amplification detected a germline heterozygous deletion mutation at exons 1–7 of the STK11 gene (c.1-?_920+?del) in peripheral blood leukocytes and mosaic loss of heterozygosity in ovarian tumor tissue. Considering that LEGH and/or gastric-type cervical adenocarcinoma can be found in patients with PJS carrying germline and/or somatic STK11 mutations, our case indicates that STK11 mutations have an important role in the proliferation of pyloric-phenotype mucinous epithelium at various anatomical locations.
-
Citations
Citations to this article as recorded by- Molecular evidence of a clonal relationship of synchronous/multifocal gastric‐type lesions of the female genital tract
Min Shi, Hong Yang, Fang Zhang, Ting Hou, Huageng Huang, Yi Lu, Yehan Zhou, Ting Lan, Juan Ji, Jun Hou, Chengmin Zhou, Zhou Zhang, Sheng Qin, Zongyao Huang, Yang Liu
The Journal of Pathology.2026; 268(1): 27. CrossRef - Serine/threonine kinase 11 (STK11) associated adnexal tumors: from biology to therapeutic impact
Guanxiang Huang, Wenyu Lin, Tingting Jiang, Yuanjun Cai, Chengbin Lin, Pengming Sun
Human Genomics.2025;[Epub] CrossRef - Novel ultrasound features and diagnostic clues of gastric-type endocervical adenocarcinoma: a case series
Liwen Yang, Yangyang Wang, Jian Cai, Ying Xiong, Juan Li, Qi Zhou, Nan Ye, Hua Lai, Tianjiao Liu, Liuying Zhou
Frontiers in Oncology.2025;[Epub] CrossRef - Ovarian Mucinous Tumor Presenting Atypical Lobular Endocervical Glandular Hyperplasia-Like Appearance in a Patient With Germline STK11 p.F354L Variant: A Case Report
Hiroshi Yoshida, Kengo Hiranuma, Mariko Nakahara, Mayumi Kobayashi-Kato, Yasuhito Tanase, Masaya Uno, Kouya Shiraishi, Mitsuya Ishikawa, Tomoyasu Kato
International Journal of Surgical Pathology.2024; 32(2): 394. CrossRef - Preoperative multimodal ultrasonic imaging in a case of Peutz-Jeghers syndrome complicated by atypical lobular endocervical glandular hyperplasia: a case report and literature review
Liwen Yang, Duan Duan, Ying Xiong, Tianjiao Liu, Lijun Zhao, Fan Lai, Dingxian Gu, Liuying Zhou
Hereditary Cancer in Clinical Practice.2024;[Epub] CrossRef - Gastric‐type glandular lesions of the female genital tract excluding the cervix: emerging pathological entities
Richard W‐C Wong, Karen L Talia, W Glenn McCluggage
Histopathology.2024; 85(1): 20. CrossRef - Gastric-phenotype Mucinous Carcinoma of the Fallopian Tube with Secondary Ovarian Involvement in a Woman with Peutz-Jeghers Syndrome: A Case Report
Mónica Bronte Anaut, Javier Arredondo Montero, Maria Pilar Fernández Seara, Rosa Guarch Troyas
International Journal of Surgical Pathology.2023; 31(1): 92. CrossRef - Molecular characterization of gastric-type endocervical adenocarcinoma using next-generation sequencing
Swati Garg, Teddy S. Nagaria, Blaise Clarke, Orit Freedman, Zanobia Khan, Joerg Schwock, Marcus Q. Bernardini, Amit M. Oza, Kathy Han, Adam C. Smith, Tracy L. Stockley, Marjan Rouzbahman
Modern Pathology.2019; 32(12): 1823. CrossRef - The developing spectrum of gastric-type cervical glandular lesions
Karen L. Talia, W. Glenn McCluggage
Pathology.2018; 50(2): 122. CrossRef
- Molecular evidence of a clonal relationship of synchronous/multifocal gastric‐type lesions of the female genital tract
- Evaluation of the VE1 Antibody in Thyroid Cytology Using Ex Vivo Papillary Thyroid Carcinoma Specimens
- Yon Hee Kim, Hyunee Yim, Yong-Hee Lee, Jae Ho Han, Kyi Beom Lee, Jeonghun Lee, Euy Young Soh, Seon-Yong Jeong, Jang-Hee Kim
- J Pathol Transl Med. 2016;50(1):58-66. Published online December 14, 2015
- DOI: https://doi.org/10.4132/jptm.2015.10.10
- 12,216 View
- 81 Download
- 9 Web of Science
- 11 Crossref
-
 Abstract
Abstract
 PDF
PDF - Background
Recently, VE1, a monoclonal antibody against the BRAFV600E mutant protein, has been investigated in terms of its detection of the BRAFV600E mutation. Although VE1 immunostaining and molecular methods used to assess papillary thyroid carcinoma in surgical specimens are in good agreement, evaluation of VE1 in thyroid cytology samples is rarely performed, and its diagnostic value in cytology has not been well established. In present study, we explored VE1 immunoexpression in cytology samples from ex vivo papillary thyroid carcinoma specimens in order to minimize limitations of low cellularity and sampling/targeting errors originated from thyroid fineneedle aspiration and compared our results with those obtained using the corresponding papillary thyroid carcinoma tissues. Methods: The VE1 antibody was evaluated in 21 cases of thyroid cytology obtained directly from ex vivo thyroid specimens. VE1 immunostaining was performed using liquid-based cytology, and the results were compared with those obtained using the corresponding tissues. Results: Of 21 cases, 19 classic papillary thyroid carcinomas had BRAFV600E mutations, whereas two follicular variants expressed wild-type BRAF. VE1 immunoexpression varied according to specimen type. In detection of the BRAFV600E mutation, VE1 immunostaining of the surgical specimen exhibited 100% sensitivity and 100% specificity, whereas VE1 immunostaining of the cytology specimen exhibited only 94.7% sensitivity and 0% specificity. Conclusions: Our data suggest that VE1 immunostaining of a cytology specimen is less specific than that of a surgical specimen for detection of the BRAFV600E mutation, and that VE1 immunostaining of a cytology specimen should be further evaluated and optimized for clinical use. -
Citations
Citations to this article as recorded by- Principles of Analytic Validation of Immunohistochemical Assays: Guideline Update
Jeffrey D. Goldsmith, Megan L. Troxell, Sinchita Roy-Chowdhuri, Carol F. Colasacco, Mary Elizabeth Edgerton, Patrick L. Fitzgibbons, Regan Fulton, Thomas Haas, Patricia L. Kandalaft, Tanja Kalicanin, Christina Lacchetti, Patti Loykasek, Nicole E. Thomas,
Archives of Pathology & Laboratory Medicine.2024; 148(6): e111. CrossRef - Inter- and Intra-observer Reproducibility of Thyroid Fine Needle Aspiration Cytology: An investigation of Bethesda 2023 Using Immunohistochemical BRAFV600E Antibody
Erdogan Bahattin, Dündar Emine, Çetin Kısmet Çivi, Yılmaz Fatih
Journal of Cytology.2024; 41(4): 221. CrossRef - VE1 immunohistochemistry is an adjunct tool for detection of BRAFV600E mutation: Validation in thyroid cancer patients
Faiza A. Rashid, Sobia Tabassum, Mosin S. Khan, Hifzur R. Ansari, Muhammad Asif, Ahmareen K. Sheikh, Syed Sameer Aga
Journal of Clinical Laboratory Analysis.2021;[Epub] CrossRef - Effective utilization of liquid-based cytology for thyroid lesions
Yukie YAMAYA
The Journal of the Japanese Society of Clinical Cytology.2021; 60(3): 164. CrossRef - Diagnostic Efficacy of BRAFV600E Immunocytochemistry in Thyroid Aspirates in Bethesda Category IV and Papillary Thyroid Carcinoma
Nidhi Anand, Tushar Agrawal, Anurag Gupta, Saumya Shukla, Roma Pradhan, Nuzhat Husain
Journal of Cytology.2021; 38(3): 113. CrossRef - RETRACTED: The Relationship between BRAF V600E Mutation and the Clinicopathological Features of the Papillary Thyroid Carcinoma
Yi Shi, Yiming Jiang, Maiweilidan Yimingjiang, Xuelian Pang, Zhiping Ma, Wenli Cui, Wei Zhang
International Journal of Clinical Medicine.2020; 11(05): 216. CrossRef - The immunocytochemical expression of VE‐1 (BRAF V600E‐related) antibody identifies the aggressive variants of papillary thyroid carcinoma on liquid‐based cytology
Patrizia Straccia, Chiara Brunelli, Esther D. Rossi, Paola Lanza, Maurizio Martini, Teresa Musarra, Celestino Pio Lombardi, Alfredo Pontecorvi, Guido Fadda
Cytopathology.2019; 30(5): 460. CrossRef - Utility of the BRAF p.V600E immunoperoxidase stain in FNA direct smears and cell block preparations from patients with thyroid carcinoma
Amber L. Smith, Michelle D. Williams, John Stewart, Wei‐Lien Wang, Savitri Krishnamurthy, Maria E. Cabanillas, Sinchita Roy‐Chowdhuri
Cancer Cytopathology.2018; 126(6): 406. CrossRef - Refinement of the criteria for ultrastructural peritubular capillary basement membrane multilayering in the diagnosis of chronic active/acute antibody-mediated rejection
Heounjeong Go, Sung Shin, Young Hoon Kim, Duck Jong Han, Yong Mee Cho
Transplant International.2017; 30(4): 398. CrossRef - Thyroid Fine-Needle Aspiration Cytology Practice in Korea
Yoon Jin Cha, Ju Yeon Pyo, SoonWon Hong, Jae Yeon Seok, Kyung-Ju Kim, Jee-Young Han, Jeong Mo Bae, Hyeong Ju Kwon, Yeejeong Kim, Kyueng-Whan Min, Soonae Oak, Sunhee Chang
Journal of Pathology and Translational Medicine.2017; 51(6): 521. CrossRef - Use of monoclonal antibodies to detect specific mutations in formalin-fixed, paraffin-embedded tissue sections
Zhenying Guo, Ricardo V. Lloyd
Human Pathology.2016; 53: 168. CrossRef
- Principles of Analytic Validation of Immunohistochemical Assays: Guideline Update
- A Rare Case of Primary Tubular Adenocarcinoma of the Thymus, Enteric Immunophenotype: A Case Study and Review of the Literature
- Hae Yoen Jung, Hyundeuk Cho, Jin-Haeng Chung, Sang Byoung Bae, Ji-Hye Lee, Hyun Ju Lee, Si-Hyong Jang, Mee-Hye Oh
- J Pathol Transl Med. 2015;49(4):331-334. Published online June 1, 2015
- DOI: https://doi.org/10.4132/jptm.2015.04.16
- 11,697 View
- 84 Download
- 14 Web of Science
- 15 Crossref
-
 Abstract
Abstract
 PDF
PDF - Thymic carcinomas are uncommon malignant tumors, and thymic adenocarcinomas are extremely rare. Here, we describe a case of primary thymic adenocarcinoma in a 59-year-old woman. Histological examination of the tumor revealed tubular morphology with expression of cytokeratin 20 and caudal-type homeobox 2 according to immunohistochemistry, suggesting enteric features. Extensive clinical and radiological studies excluded the possibility of an extrathymic primary tumor. A review of the literature revealed only two global cases of primary tubular adenocarcinomas of the thymus with enteric immunophenotype.
-
Citations
Citations to this article as recorded by- Non-mucinous, enteric-type thymic adenocarcinoma: genetic analysis of a case
Eiji Narusawa, Yoichi Ohtaki, Genichiro Ishii, Seshiru Nakazawa, Natsuko Kawatani, Tomohiro Yazawa, Kazuki Numajiri, Yuka Yoshida, Keisuke Nimura, Ken Shirabe
General Thoracic and Cardiovascular Surgery Cases.2026;[Epub] CrossRef - Unresectable Primary Enteric‐Type Thymic Adenocarcinoma Treated With FOLFOX Chemotherapy: A Case Report
Carl He, Georgia Bentick, Patrick Hosking, Andrew Mant
Cancer Reports.2025;[Epub] CrossRef - Enteric thymic adenocarcinoma: Understanding a unique pathology in the mediastinum
Raja Chhabra, Kartik Mittal, Anmol Tufchi, Sajjan Rajpurohit, Deepak Kumar Mittal, Aditya Vidushi, Swati Saxena, Md Ali Osama
Indian Journal of Pathology and Microbiology.2025; 68(4): 820. CrossRef - Clinical characteristics and prognosis of primary thymic adenocarcinoma: A single‐center retrospective analysis
Qian Hong, Rui Han, Chen Chen, Fuquan Wang, Sining Zhang, Chenguang Zhao, Fang Li, Juwei Mu, Jiagen Li
Thoracic Cancer.2024; 15(24): 1815. CrossRef - Case report: Primary adenocarcinoma NOS of the thymus and cytological features
Jonathan Willner, Osvaldo Hernandez, Lea Azour, Andre L. Moreira
Diagnostic Cytopathology.2023;[Epub] CrossRef - Enteric-type thymic adenocarcinoma: a case report and literature review focusing on prognosis based on histological subtypes
Rurika Hamanaka, Kei Nakano, Takaaki Tsuboi, Kazuhito Hatanaka, Mitsutomo Kohno, Ryota Masuda, Masayuki Iwazaki
General Thoracic and Cardiovascular Surgery.2022; 70(5): 501. CrossRef - Metastatic thymic-enteric adenocarcinoma responding to chemoradiation plus anti-angiogenic therapy: A case report
Man Li, Xiao-Yu Pu, Li-Hua Dong, Peng-Yu Chang
World Journal of Clinical Cases.2021; 9(7): 1676. CrossRef - A case report: primary thymic adenocarcinoma with enteric differentiation
Yuuki Kou, Hirokazu Tanaka, Nobuhisa Yamazaki, Hiroyoshi Watanabe, Makoto Sonobe
The Journal of the Japanese Association for Chest Surgery.2020; 34(2): 107. CrossRef - Thymic enteric type adenocarcinoma: A case report with cytological features
Marie Tamai, Mitsuaki Ishida, Yusuke Ebisu, Hisashi Okamoto, Chika Miyasaka, Chisato Ohe, Yoshiko Uemura, Tomohito Saito, Tomohiro Murakawa, Koji Tsuta
Diagnostic Cytopathology.2018; 46(1): 92. CrossRef - Histologic characteristics of thymic adenocarcinomas: Clinicopathologic study of a nine-case series and a review of the literature
Ah-Young Kwon, Joungho Han, Jinah Chu, Yong Soo Choi, Byeong-Ho Jeong, Myung-Ju Ahn, Yong Chan Ahn
Pathology - Research and Practice.2017; 213(2): 106. CrossRef - Characterization of genetic aberrations in a single case of metastatic thymic adenocarcinoma
Yeonghun Lee, Sehhoon Park, Se-Hoon Lee, Hyunju Lee
BMC Cancer.2017;[Epub] CrossRef - Cytologic Characteristics of Thymic Adenocarcinoma with Enteric Differentiation: A Study of Four Fine-Needle Aspiration Specimens
Ah-Young Kwon, Joungho Han, Hae-yon Cho, Seokhwi Kim, Heejin Bang, Jiyeon Hyeon
Journal of Pathology and Translational Medicine.2017; 51(5): 509. CrossRef - Mucinous cystic tumor with CK20 and CDX2 expression of the thymus: Is this a benign counterpart of adenocarcinoma of the thymus, enteric type?
Jun Akiba, Hiroshi Harada, Shintaro Yokoyama, Toshihiro Hashiguchi, Akihiko Kawahara, Masahiro Mitsuoka, Shinzo Takamori, Hirohisa Yano
Pathology International.2016; 66(1): 29. CrossRef - Colon cancer chemotherapy for a patient with CDX2-expressing metastatic thymic adenocarcinoma: a case report and literature review
Akihiko Sawaki, Mikiya Ishihara, Yuji Kozuka, Hiroyasu Oda, Satoshi Tamaru, Yumiko Sugawara, Yoshiki Yamashita, Toshiro Mizuno, Taizo Shiraishi, Naoyuki Katayama
International Cancer Conference Journal.2016; 5(2): 113. CrossRef - Metastatic Thymic Adenocarcinoma from Colorectal Cancer
Mina Lee, Suk Jin Choi, Yong Han Yoon, Joung-Taek Kim, Wan Ki Baek, Young Sam Kim
The Korean Journal of Thoracic and Cardiovascular Surgery.2015; 48(6): 447. CrossRef
- Non-mucinous, enteric-type thymic adenocarcinoma: genetic analysis of a case
- Accuracy of Core Needle Biopsy Versus Fine Needle Aspiration Cytology for Diagnosing Salivary Gland Tumors
- In Hye Song, Joon Seon Song, Chang Ohk Sung, Jong-Lyel Roh, Seung-Ho Choi, Soon Yuhl Nam, Sang Yoon Kim, Jeong Hyun Lee, Jung Hwan Baek, Kyung-Ja Cho
- J Pathol Transl Med. 2015;49(2):136-143. Published online March 12, 2015
- DOI: https://doi.org/10.4132/jptm.2015.01.03
- 18,794 View
- 244 Download
- 81 Web of Science
- 85 Crossref
-
 Abstract
Abstract
 PDF
PDF - Background
Core needle biopsy is a relatively new technique used to diagnose salivary gland lesions, and its role in comparison with fine needle aspiration cytology needs to be refined. Methods: We compared the results of 228 ultrasound-guided core needle biopsy and 371 fine needle aspiration procedures performed on major salivary gland tumors with their postoperative histological diagnoses. Results: Core needle biopsy resulted in significantly higher sensitivity and more accurate tumor subtyping, especially for malignant tumors, than fine needle aspiration. No patient developed major complications after core needle biopsy. Conclusions: We recommend ultrasoundguided core needle biopsy as the primary diagnostic tool for the preoperative evaluation of patients with salivary gland lesions, especially when malignancy is suspected. -
Citations
Citations to this article as recorded by- Parotid Secretory Carcinoma: A Distinct Entity from Acinic Cell Carcinoma
Muhammad Adzha Musa, Muhamad Arif Ahmad Radzi, Moharzudi Mohamed, Ikram Hakim
Indian Journal of Otolaryngology and Head & Neck Surgery.2026; 78(2): 1209. CrossRef - Fine-needle aspiration or core needle biopsy? A meta-analysis of diagnostic accuracy and procedural outcomes in salivary gland tumors
Kenan Kassem, Alaa Safia, Uday Abd Elhadi, Shlomo Merchavy
European Journal of Radiology.2026; 194: 112532. CrossRef - Controversial issues in core needle biopsy for diagnosing parotid neoplasms
N. V. Vishneva, P. A. Demenchuk, O. Yu. Petropavlovskaya, E. I. Selifanova, A. N. Lanina, N. V. Kalakutsky, A. I. Yaremenko
Parodontologiya.2026; 30(4): 407. CrossRef - Deep learning-based computer-aided diagnosis for parotid gland tumors on MRI
Yuki Irifune, Sohei Mitani, Koji Kinoshita, Naoto Kawaguchi, Takashi Kitani, Eriko Sato, Yuki Hosokawa, Masaharu Isshiki, Kazuki Yoshida, Takashi Mukaigawa, Naohito Hato
Auris Nasus Larynx.2026; 53(3): 349. CrossRef - A Case of Parotid Nodular Oncocytic Hyperplasia Diagnosed Preoperatively by Core Needle Biopsy
Yuki Kimata, Shinichi Sato, Hisanobu Tamaki
Practica Oto-Rhino-Laryngologica.2026; 119(4): 231. CrossRef - Optimizing preoperative assessment of salivary gland lesions: updates and practical implications in the Milan System 2nd edition
Patrizia Straccia, Vincenzo Fiorentino, Alessia Piermattei, Qianqian Zhang, Belen Padial-Urtueta, Federica Cianfrini, Antonino Mule’, Esther Diana Rossi
Virchows Archiv.2026;[Epub] CrossRef - Frozen Section Analysis in Submandibular Gland Tumors: Optimizing Intraoperative Decision-Making
Amir Bolooki, Felix Johnson, Anna Stenzl, Zhaojun Zhu, Benedikt Gabriel Hofauer
Cancers.2025; 17(5): 895. CrossRef - The Myriad Spectrum of Salivary Gland Lesions: Cytohistological Correlation on Fine Needle Aspiration Cytology, Core Needle Biopsy, and Resections in a 5‐Year Single Institutional Experience of North India
Zachariah Chowdhury, Pallavi Majumdar, Sumeet Narain, Komal Lamba
Diagnostic Cytopathology.2025; 53(8): 391. CrossRef - Salivary Duct Carcinoma: A 12-Year Single Center Experience
Hyowon Ahn, Dongbin Ahn, Ji Hye Kwak
Korean Journal of Otorhinolaryngology-Head and Neck Surgery.2025; 68(6): 232. CrossRef - Cytological Spectrum of Salivary Gland Lesions Using the Milan System and their Histopathological Correlation: Retrospective Study in a Peripheral Medical College of Eastern India
Ujjwal Bandyopadhyay, Sarmila Guha Banerjee, Sirshak Dutta
Indian Journal of Otolaryngology and Head & Neck Surgery.2025; 77(11): 4887. CrossRef - Diagnostic performance of ultrasound guided salivary gland core needle biopsy and fine needle aspiration in children
Vicente Oliveira, Anthea Lafrenière, Nikolaus Wolter, Joao Amaral, Alessandro Gasparetto, Dimitri Parra Rojas
Pediatric Radiology.2025; 56(2): 393. CrossRef - Giant Pleomorphic Adenoma of Submandibular Gland
Harendra Kumar, Qazi Saquib Rizwan, Mayank Gupta, Tarun Kumar
Indian Journal of Otolaryngology and Head & Neck Surgery.2024; 76(1): 1361. CrossRef - CT-guided core needle biopsies of head and neck tumors: a comprehensive monocenter analysis of safety and outcomes
Thomas Joseph Vogl, Heinrich Johannes Ketelsen, Scherwin Mahmoudi, Jan-Erik Scholtz, Vitali Koch, Leon David Grünewald, Peter Wild, Timo Stoever, Simon Bernatz
European Radiology.2024; 34(8): 5370. CrossRef - Indications for Submandibulectomy Within a 20-Year Period
Amir Bolooki, Anna Stenzl, Christopher Weusthof, Benedikt Hofauer
Ear, Nose & Throat Journal.2024;[Epub] CrossRef - Treatment of Warthin’s Tumors of the Parotid Gland With Radiofrequency Ablation: A Systematic Review of the Current Literature
Kenny Do, Eric Kawana, Sisi Tian, Jo-Lawrence Bigcas
Ear, Nose & Throat Journal.2024;[Epub] CrossRef - Evaluation of the anterior processes of the parotid gland: an ultrasonographic study
Tarık Ali Uğur, Hümeyra Tercanlı
Surgical and Radiologic Anatomy.2024; 46(6): 915. CrossRef - Salivary Gland Fine-Needle Aspiration
Federica Policardo, Antonino Mule’, Esther Diana Rossi
Surgical Pathology Clinics.2024; 17(3): 347. CrossRef - Implementation of the Milan System for Reporting Salivary Gland Cytology: A Two-Year Outcome Cytopathology Data of a Tertiary Care Center
Soudamini Mahapatra, Chinmaya Sundar Ray, Aparajita Mishra, Dileswari Pradhan
Cureus.2024;[Epub] CrossRef - Preoperative cytopathological investigatory aids in the diagnosis of salivary gland lesions
S Vidyalakshmi, K Shanmugasamy
Journal of Oral and Maxillofacial Pathology.2024; 28(2): 172. CrossRef - Machine Learning on Ultrasound Texture Analysis Data for Characterizing of Salivary Glandular Tumors: A Feasibility Study
Li-Jen Liao, Ping-Chia Cheng, Feng-Tsan Chan
Diagnostics.2024; 14(16): 1761. CrossRef - Association of Membranous Basal Cell Adenoma and Basal Cell Adenocarcinoma With Brooke-Spiegler Syndrome
Lexi Goehring, Debby Rampisela, Jordan L Pleitz
Cureus.2024;[Epub] CrossRef - A Retrospective 8‐Year Single Institutional Study in Germany Regarding Diagnosis, Treatment, and Outcome of Malignant Parotid Tumors
S. Andrianopoulou, L. S. Fiedler, B. M. Lippert, O. C. Bulut, Mohamed Rahouma
International Journal of Surgical Oncology.2024;[Epub] CrossRef - High Field MRI in Parotid Gland Tumors: A Diagnostic Algorithm
Chiara Gaudino, Andrea Cassoni, Martina Lucia Pisciotti, Resi Pucci, Chiara Veneroso, Cira Rosaria Tiziana Di Gioia, Francesca De Felice, Patrizia Pantano, Valentino Valentini
Cancers.2024; 17(1): 71. CrossRef - Nationwide Incidence Trends of Pediatric Parotid Malignancy in Korea and a Retrospective Analysis of Single-Institution Surgical Experience of Parotidectomy
Hyun Seong Kim, Seo Young Kim, Eun-Jae Chung, Seong Keun Kwon, Soon-Hyun Ahn, Yuh-Seog Jung, Jungirl Seok
Korean Society of Head and Neck Oncology.2024; 40(2): 7. CrossRef - The Usefulness of Ultrasound-Guided Core Needle Biopsy Compared to Fine Needle Aspiration in Pre-Operative Diagnosis of Cystic-Predominant Parotid Tumors
Youn Jin Cho, Young Rok Jo, Hyun Jun Hong, Hye Ran Lee
Korean Journal of Otorhinolaryngology-Head and Neck Surgery.2023; 66(8): 532. CrossRef - The Value of Ultrasound-guided Core Needle Biopsy in Differentiating Benign from Malignant Salivary Gland Lesions
Mohammad Ali Kazemi, Farzaneh Amini, Bita Kargar, Maryam Lotfi, Keyvan Aghazadeh, Hashem Sharifian, Behnaz Moradi, Javid Azadbakht
Indian Journal of Otolaryngology and Head & Neck Surgery.2023; 75(2): 266. CrossRef - Schnellschnittdiagnostik bei Tumoren des Trigonum submandibulare
S. Riemann, A. Knopf
HNO.2023; 71(3): 164. CrossRef - Myoepithelial Carcinoma Ex Pleomorphic Adenoma of the Submandibular Gland: A Case Report
Georgia Syrnioti, Antonia Syrnioti, Alharith Abdullah, Xuehui Lui, Ernesto Mendoza
Cureus.2023;[Epub] CrossRef - Intraductal Carcinoma: The Carcinoma In Situ of the Salivary Gland
Rhema Thomas, Tijjani Umar, Farzad Borumandi
Journal of Craniofacial Surgery.2023; 34(5): e432. CrossRef - Fine-Needle Aspiration Cytology for Parotid Tumors
Masataka Taniuchi, Tetsuya Terada, Ryo Kawata
Life.2022; 12(11): 1897. CrossRef - Utility of the Milan System for Reporting Salivary Gland Cytology, with focus on the incidence and histologic correlates of atypia of undetermined significance (AUS) and salivary gland neoplasm of uncertain malignant potential (SUMP): A 3‐year institution
Christopher M. Cormier, Shweta Agarwal
Cancer Cytopathology.2022; 130(4): 303. CrossRef - Percutaneous CT-Guided Core Needle Biopsies of Head and Neck Masses: Review of 184 Cases at a Single Academic Institution, Common and Special Techniques, Diagnostic Yield, and Safety
R.W. Jordan, D.P. Shlapak, J.C. Benson, F.E. Diehn, D.K. Kim, V.T. Lehman, G.B. Liebo, A.A. Madhavan, J.M. Morris, P.P. Morris, J.T. Verdoorn, C.M. Carr
American Journal of Neuroradiology.2022; 43(1): 117. CrossRef - Nodular fasciitis of the submandibular gland
Ting Suen Wong, Richard Wei Chern Gan, Laszlo Karsai, Bun Yin Winson Wong
BMJ Case Reports.2022; 15(4): e245584. CrossRef - Validation of the Milan system for reporting salivary gland cytopathology: a single institution’s 10-year experience
Christopher Felicelli, Joseph Reznicek, Yevgen Chornenkyy, Lucy Jager, Daniel Johnson
Journal of the American Society of Cytopathology.2022; 11(5): 264. CrossRef - Application of the Milan system for reporting salivary gland cytopathology using cell blocks
Grégoire B. Morand, Raihanah Alsayegh, Alex M. Mlynarek, Marianne Plourde, Tiffany Mach, Marco A. Mascarella, Michael P. Hier, Livia Florianova, Marc P. Pusztaszeri
Virchows Archiv.2022; 481(4): 575. CrossRef - Comparisons among the Ultrasonography Prediction Model, Real-Time and Shear Wave Elastography in the Evaluation of Major Salivary Gland Tumors
Ping-Chia Cheng, Wu-Chia Lo, Chih-Ming Chang, Ming-Hsun Wen, Po-Wen Cheng, Li-Jen Liao
Diagnostics.2022; 12(10): 2488. CrossRef - A Novel Sonographic Scoring Model in the Prediction of Major Salivary Gland Tumors
Wu‐Chia Lo, Chih‐Ming Chang, Chi‐Te Wang, Po‐Wen Cheng, Li‐Jen Liao
The Laryngoscope.2021;[Epub] CrossRef - Assessing the diagnostic accuracy for pleomorphic adenoma and Warthin tumor by employing the Milan System for Reporting Salivary Gland Cytopathology: An international, multi‐institutional study
Derek B. Allison, Alexander P. Smith, Daniel An, James Adam Miller, Khurram Shafique, Sharon Song, Kartik Viswanathan, Elizabeth Eykman, Rema A. Rao, Austin Wiles, Güliz A. Barkan, Ritu Nayar, Guido Fadda, Celeste N. Powers, Esther Diana Rossi, Momin T. S
Cancer Cytopathology.2021; 129(1): 43. CrossRef - Magnetic resonance imaging of salivary gland tumours: Key findings for imaging characterisation
Davide Maraghelli, Michele Pietragalla, Cesare Cordopatri, Cosimo Nardi, Anna Julie Peired, Giandomenico Maggiore, Stefano Colagrande
European Journal of Radiology.2021; 139: 109716. CrossRef - The Milan System, from Its Introduction to Its Current Adoption in the Diagnosis of Salivary Gland Cytology
Esther Diana Rossi
Journal of Molecular Pathology.2021; 2(2): 114. CrossRef - Utility of the Milan system for reporting salivary gland cytopathology during rapid on‐site evaluation (ROSE) of salivary gland aspirates
Aanchal Kakkar, Mukin Kumar, Priyadarsani Subramanian, Arshad Zubair, Rajeev Kumar, Alok Thakar, Deepali Jain, Sandeep R. Mathur, Venkateswaran K. Iyer
Cytopathology.2021; 32(6): 779. CrossRef - Contribution of small tissue biopsy and flow cytometry to preoperative cytological categorization of salivary gland fine needle aspirates according to the Milan System: Single center experience on 287 cases
Tolga Bağlan, Serpil Dizbay Sak, Cevriye Cansız Ersöz, Koray Ceyhan
Diagnostic Cytopathology.2021; 49(4): 509. CrossRef - Is Milan for kids?: The Milan System for Reporting Salivary Gland Cytology in pediatric patients at an academic children's hospital with cytologic‐histologic correlation
Swati P. Satturwar, Maren Y. Fuller, Sara E. Monaco
Cancer Cytopathology.2021; 129(11): 884. CrossRef - Radiographic Interpretation in Oral Medicine and Hospital Dental Practice
Katherine France, Anwar A.A.Y. AlMuzaini, Mel Mupparapu
Dental Clinics of North America.2021; 65(3): 509. CrossRef - Carcinoma ex pleomorphic adenoma of major salivary glands: CT and MR imaging findings
Can Wang, Qiang Yu, Siyi Li, Jingjing Sun, Ling Zhu, Pingzhong Wang
Dentomaxillofacial Radiology.2021; 50(7): 20200485. CrossRef - Salivary gland carcinoma in children and adolescents: The EXPeRT/PARTNER diagnosis and treatment recommendations
Aurore Surun, Dominik T. Schneider, Andrea Ferrari, Teresa Stachowicz‐Stencel, Jelena Rascon, Anna Synakiewicz, Abbas Agaimy, Kata Martinova, Denis Kachanov, Jelena Roganovic, Ewa Bien, Gianni Bisogno, Ines B. Brecht, Frédéric Kolb, Juliette Thariat, Anto
Pediatric Blood & Cancer.2021;[Epub] CrossRef - A Call for Universal Acceptance of the Milan System for Reporting Salivary Gland Cytopathology
Eric Barbarite, Sidharth V. Puram, Adeeb Derakhshan, Esther D. Rossi, William C. Faquin, Mark A. Varvares
The Laryngoscope.2020; 130(1): 80. CrossRef - Preoperative biopsy in parotid malignancies: Variation in use and impact on surgical margins
Liliya Benchetrit, Sina J. Torabi, Elliot Morse, Saral Mehra, Rahmatullah Rahmati, Heather A. Osborn, Benjamin L. Judson
The Laryngoscope.2020; 130(6): 1450. CrossRef - α‐Synuclein Real‐Time Quaking‐Induced Conversion in the Submandibular Glands of Parkinson's Disease Patients
Sireesha Manne, Naveen Kondru, Huajun Jin, Vellareddy Anantharam, Xuemei Huang, Arthi Kanthasamy, Anumantha G. Kanthasamy
Movement Disorders.2020; 35(2): 268. CrossRef - The Accessory Parotid Gland and its Clinical Significance
Mateusz A. Rosa, Dominik P. Łazarz, Jakub R. Pękala, Bendik Skinningsrud, Sigurd S. Lauritzen, Bernard Solewski, Przemysław A. Pękala, Jerzy A. Walocha, Krzysztof A. Tomaszewski
Journal of Craniofacial Surgery.2020; 31(3): 856. CrossRef - Comparison of core needle biopsy and fine‐needle aspiration in diagnosis of ma lignant salivary gland neoplasm: Systematic review and meta‐analysis
Jungheum Cho, Junghoon Kim, Ji Sung Lee, Choong Guen Chee, Youngjune Kim, Sang Il Choi
Head & Neck.2020; 42(10): 3041. CrossRef - The Milan system for reporting salivary gland cytopathology: The clinical impact so far. Considerations from theory to practice
Esther Diana Rossi, William C. Faquin
Cytopathology.2020; 31(3): 181. CrossRef - The role of core needle biopsy in parotid glands lesions with inconclusive fine needle aspiration
Farrokh Heidari, Firouzeh Heidari, Benyamin Rahmaty, Neda Jafari, Kayvan Aghazadeh, Saeed Sohrabpour, Ebrahim Karimi
American Journal of Otolaryngology.2020; 41(6): 102718. CrossRef - Role of Fine Needle Aspiration Cytology in the Diagnosis of Parotid Gland Tumors: Analysis of 193 Cases
Rahim Dhanani, Haissan Iftikhar, Muhammad Sohail Awan, Nida Zahid, Sehrish Nizar Ali Momin
International Archives of Otorhinolaryngology.2020; 24(04): e508. CrossRef - Cytohistological correlation of salivary gland tumours with emphasis on Milan system for reporting: A novel step towards internal quality assurance
Anandraj Vaithy.K, ATM Venkat Raghava, E S Keerthika Sri, K R Umadevi
IP Archives of Cytology and Histopathology Research.2020; 5(4): 283. CrossRef - Diagnosing Recently Defined and Uncommon Salivary Gland Lesions in Limited Cellularity Specimens: Cytomorphology and Ancillary Studies
Esther Diana Rossi, Zubair Baloch, William Faquin, Liron Pantanowitz
AJSP: Reviews and Reports.2020; 25(5): 210. CrossRef - Peripheral T Cell Lymphoma of Parotid Gland: A Diagnostic Challenge
J. G. Aishwarya, Satish Nair, C. N. Patil, Swarna Shivakumar, N. Shrivalli, Ashish Shah
Indian Journal of Otolaryngology and Head & Neck Surgery.2019; 71(S1): 533. CrossRef - Potential utility of core needle biopsy in the diagnosis of IgG4-related dacryoadenitis and sialadenitis
Kenichi Takano, Tsuyoshi Okuni, Keisuke Yamamoto, Ryuta Kamekura, Ryoto Yajima, Motohisa Yamamoto, Hiroki Takahashi, Tetsuo Himi
Modern Rheumatology.2019; 29(2): 393. CrossRef - Retrospective assessment of the effectiveness of the Milan system for reporting salivary gland cytology: A systematic review and meta‐analysis of published literature
Sahar J Farahani, Zubair Baloch
Diagnostic Cytopathology.2019; 47(2): 67. CrossRef - Values of fine‐needle aspiration cytology of parotid gland tumors: A review of 996 cases at a single institution
Manabu Suzuki, Ryo Kawata, Masaaki Higashino, Shuji Nishikawa, Tetsuya Terada, Shin‐Ichi Haginomori, Yoshitaka Kurisu, Yoshinobu Hirose
Head & Neck.2019; 41(2): 358. CrossRef - Positive Surgical Margins in Submandibular Malignancies: Facility and Practice Variation
Liliya Benchetrit, Elliot Morse, Benjamin L. Judson, Saral Mehra
Otolaryngology–Head and Neck Surgery.2019; 161(4): 620. CrossRef - The growth rate and the positive prediction of needle biopsy of clinically diagnosed Warthin’s tumor
Jungirl Seok, Woo-Jin Jeong, Soon-Hyun Ahn, Young Ho Jung
European Archives of Oto-Rhino-Laryngology.2019; 276(7): 2091. CrossRef - The Difference in the Clinical Features Between Carcinoma ex Pleomorphic Adenoma and Pleomorphic Adenoma
Jungirl Seok, Se Jin Hyun, Woo-Jin Jeong, Soon-Hyun Ahn, Hyojin Kim, Young Ho Jung
Ear, Nose & Throat Journal.2019; 98(8): 504. CrossRef - Fine‐needle aspiration cytology and radiological imaging in parotid gland tumours: Our experience in 103 patients
Clare Perkins, Edward Toll, Philip Reece
Clinical Otolaryngology.2019; 44(6): 1124. CrossRef - Application of the Milan system of reporting salivary cytopathology – A retrospective cytohistological correlation study
Ramya Katta, Devi Padmavathi Chaganti
Journal of Dr. NTR University of Health Sciences.2019; 8(1): 11. CrossRef - Ultrasound‐guided core needle biopsy in salivary glands: A meta‐analysis
Hee Joon Kim, Jong Seung Kim
The Laryngoscope.2018; 128(1): 118. CrossRef - Accuracy and effectiveness of ultrasound-guided core-needle biopsy in the diagnosis of focal lesions in the salivary glands
Jose Luis del Cura, Gloria Coronado, Rosa Zabala, Igone Korta, Ignacio López
European Radiology.2018; 28(7): 2934. CrossRef - The Milan System for Reporting Salivary Gland Cytopathology (MSRSGC): an ASC-IAC–sponsored system for reporting salivary gland fine-needle aspiration
Esther Diana Rossi, Zubair Baloch, Marc Pusztaszeri, William C. Faquin
Journal of the American Society of Cytopathology.2018; 7(3): 111. CrossRef - Routine and Advanced Ultrasound of Major Salivary Glands
Kunwar Suryaveer Singh Bhatia, Yuk-Ling Dai
Neuroimaging Clinics of North America.2018; 28(2): 273. CrossRef - The Milan System for Reporting Salivary Gland Cytopathology (MSRSGC): An ASC-IAC-Sponsored System for Reporting Salivary Gland Fine-Needle Aspiration
Esther Diana Rossi, Zubair W. Baloch, Marc Pusztaszeri, William C. Faquin
Acta Cytologica.2018; 62(3): 157. CrossRef - Evaluation and diagnosis of salivary gland neoplasms
Erica Jackson Mayland, Anna M. Pou
Operative Techniques in Otolaryngology-Head and Neck Surgery.2018; 29(3): 129. CrossRef - Feasibility and Safety of Multicenter Tissue and Biofluid Sampling for α-Synuclein in Parkinson’s Disease: The Systemic Synuclein Sampling Study (S4)
Lana M. Chahine, Thomas G. Beach, Nicholas Seedorff, Chelsea Caspell-Garcia, Christopher S. Coffey, Michael Brumm, Charles H. Adler, Geidy E. Serrano, Carly Linder, Sherri Mosovsky, Tatiana Foroud, Holly Riss, Dixie Ecklund, John Seibyl, Danna Jennings, V
Journal of Parkinson’s Disease.2018; 8(4): 517. CrossRef - Preoperative diagnostic of parotid gland neoplasms: fine-needle aspiration cytology or core needle biopsy?
Peter Zbären, Asterios Triantafyllou, Kenneth O. Devaney, Vincent Vander Poorten, Henrik Hellquist, Alessandra Rinaldo, Alfio Ferlito
European Archives of Oto-Rhino-Laryngology.2018; 275(11): 2609. CrossRef - A comparison study of the reporting systems for salivary gland fine needle aspirations: Are they really different?
Diana Montezuma, Sule Canberk, Ozlem Aydın, Mehmet Polat Dermirhas, André F. Vieira, Süha Goksel, Ümit İnce, Fernando Schmitt
Diagnostic Cytopathology.2018; 46(10): 859. CrossRef - Pediatric salivary gland carcinomas: Diagnostic and therapeutic management
Céleste Rebours, Vincent Couloigner, Louise Galmiche, Odile Casiraghi, Cécile Badoual, Sabah Boudjemaa, Anthony Chauvin, Monique Elmaleh, Brice Fresneau, Sylvie Fasola, Erea‐Noël Garabédian, Thierry Van Den Abeele, Daniel Orbach
The Laryngoscope.2017; 127(1): 140. CrossRef - Agreement between rapid on‐site evaluation and the final cytological diagnosis of salivary gland specimens
S. Wangsiricharoen, S. Lekawanvijit, S. Rangdaeng
Cytopathology.2017; 28(4): 321. CrossRef - Mesenchymal neoplasms of the head and neck: a cytopathologic analysis on fine needle aspiration
James Lee, Samia Kazmi, Christopher J. VandenBussche, Syed Z. Ali
Journal of the American Society of Cytopathology.2017; 6(3): 105. CrossRef - Clinical Results of Surgical Treatment in Parotid Tumors
Ahmet Kara
Journal of Otolaryngology-ENT Research.2017;[Epub] CrossRef - Parotid gland metastases of distant primary tumours: A diagnostic challenge
Achim M. Franzen, Thomas Günzel, Anja Lieder
Auris Nasus Larynx.2016; 43(2): 187. CrossRef - Modern Radiology in the Management of Head and Neck Cancer
G.J.C. Burkill, R.M. Evans, V.V. Raman, S.E.J. Connor
Clinical Oncology.2016; 28(7): 440. CrossRef - Fine‐needle aspiration and core needle biopsy: An update on 2 common minimally invasive tissue sampling modalities
Paul A. VanderLaan
Cancer Cytopathology.2016; 124(12): 862. CrossRef - Staging and follow-up of high-grade malignant salivary gland tumours: The role of traditional versus functional imaging approaches – A review
Nicole Freling, Flavio Crippa, Roberto Maroldi
Oral Oncology.2016; 60: 157. CrossRef - Biopsy of parotid masses: Review of current techniques
Sananda Haldar, Joseph D Sinnott, Kemal M Tekeli, Samuel S Turner, David C Howlett
World Journal of Radiology.2016; 8(5): 501. CrossRef - Review on the applications of ultrasonography in dentomaxillofacial region
Şehrazat Evirgen
World Journal of Radiology.2016; 8(1): 50. CrossRef - Comprehensive Cytomorphologic Analysis of Pulmonary Adenoid Cystic Carcinoma: Comparison to Small Cell Carcinoma and Non-pulmonary Adenoid Cystic Carcinoma
Seokhwi Kim, Jinah Chu, Hojoong Kim, Joungho Han
Journal of Pathology and Translational Medicine.2015; 49(6): 511. CrossRef
- Parotid Secretory Carcinoma: A Distinct Entity from Acinic Cell Carcinoma
- Oncocytic Lipoadenoma: A Rare Case of Parotid Gland Tumor and Review of the Literature
- Chen-lin Chi, Tseng-tong Kuo, Li-yu Lee
- J Pathol Transl Med. 2015;49(2):144-147. Published online March 12, 2015
- DOI: https://doi.org/10.4132/jptm.2014.02.10
- 11,745 View
- 69 Download
- 8 Web of Science
- 9 Crossref
-
 Abstract
Abstract
 PDF
PDF - Oncocytic lipoadenoma is a rare tumor, with only 18 cases having been reported since the first in 1998. We encountered a case of oncocytic lipoadenoma presenting as a slowly growing parotid mass in a 71-year-old man. This tumor is characteristically comprised of a mixture of oncocytes and adipocytes. The present case is one of five reported cases of oncocytic lipoadenoma showing sebaceous differentiation. The results of immunohistochemical study with DOG1 antibody supported the origination of this tumor in the striated duct.
-
Citations
Citations to this article as recorded by- Multimodal Imaging of Oncocytic Lipoadenoma Arising from the Parotid Deep Lobe with Medial Extension into the Parapharyngeal Space: A Case Report with Histopathologic Findings and Literature Review
Jong-Uk Lee, Hye Jin Baek, Kwang Ho Choi, Eun Cho, Hyo Jung An
Diagnostics.2026; 16(9): 1366. CrossRef - Oncocytic lipoadenoma of the parotid gland: a case report and a review of the literature
Jood K Alotaibi, Turki Mohammed Almuhaimid, Ghada Abdallah Moumneh
Journal of Surgical Case Reports.2024;[Epub] CrossRef - Oncocytic sialolipoma of parotid gland: Case report and literature review
VenuPatel Sureja, KoyyeRavindranath Tagore
Indian Journal of Pathology and Microbiology.2023; 66(3): 591. CrossRef - Complex Component of Oncocytic and Non-Oncocytic Lipoadenomas in the Parotid Gland: A Case Report
Fuyuki Sato, Takashi Nakajima, Takashi Sugino
Diagnostics.2021; 11(8): 1478. CrossRef - Oncocyitic lipoadenoma of the parotid gland
Renato PIANTANIDA, Alberto CARANTI, Adele CHIESA, Jessica BARIZZI, Ulrike PERRIARD, Filippo BARUCCA, Antonio PELLANDA
Otorinolaringologia.2021;[Epub] CrossRef - A case of oncocytic lipoadenoma of the submandibular gland and its diagnostic cytology challenges
Khaled A. Murshed, Ammar Khalafalla, Belal Alani, Hanan Farghaly, Moustafa Alkhalil
Diagnostic Cytopathology.2020; 48(4): 364. CrossRef - An extremely rare case of giant oncocytic adenolipoma of the parotid gland
Dipesh Shakya, Ajit Nepal
Clinical Case Reports.2020; 8(12): 2390. CrossRef - A rare cause of primary hyperparathyroidism: Parathyroid lipoadenoma
Sabri Özden, Servet Güreşci, Barış Saylam, Gül Dağlar
Auris Nasus Larynx.2018; 45(6): 1245. CrossRef - Oncocytic osteolipoadenoma of the submandibular gland
Domenico Corradi, Rodolfo Monaco, Giulia D'Angelo, Paola Bini, Teore Ferri, Enrico M Silini
Histopathology.2016; 69(1): 148. CrossRef
- Multimodal Imaging of Oncocytic Lipoadenoma Arising from the Parotid Deep Lobe with Medial Extension into the Parapharyngeal Space: A Case Report with Histopathologic Findings and Literature Review
- Cytokeratin-Positive Gastrointestinal Stromal Tumor of Biphasic Morphology: A Case Report
- Sung Sun Kim, Yoo Duk Choi, Jae Hyuk Lee, Chan Choi
- Korean J Pathol. 2014;48(5):375-378. Published online October 27, 2014
- DOI: https://doi.org/10.4132/KoreanJPathol.2014.48.5.375
- 9,305 View
- 39 Download
- 2 Crossref
-
 PDF
PDF -
Citations
Citations to this article as recorded by- CYTOKERATINS: NOT AN EPITHELIAL ENTITY ANYMORE?
Geetpriya Kaur, Devicharan Shetty, Seema Sikka, Aparna Pathak
INTERNATIONAL JOURNAL OF SCIENTIFIC RESEARCH.2022; : 15. CrossRef - Gastrointestinal stromal tumors of the stomach in a 10-year-old child
Saeed Nasher, Fayed Al-Yousofy, Faisal Ahmed
Journal of Pediatric Surgery Case Reports.2021; 74: 102044. CrossRef
- CYTOKERATINS: NOT AN EPITHELIAL ENTITY ANYMORE?
- Cytomorphological Findings and Histological Correlation of Low-Grade Cribriform Cystadenocarcinoma of Salivary Gland in Fine-Needle Aspiration: A Case Study
- Young Sin Ko, Ja Seung Koo
- Korean J Pathol. 2013;47(6):592-595. Published online December 24, 2013
- DOI: https://doi.org/10.4132/KoreanJPathol.2013.47.6.592
- 9,587 View
- 70 Download
- 13 Crossref
-
 Abstract
Abstract
 PDF
PDF Low-grade cribriform cystadenocarcinoma (LGCCC) of the salivary gland is a rare tumor. We report the cytologic features and histologic correlation of a patient with LGCCC. A 57-year-old man had a hardly palpable, nontender mass in the right cheek area followed over nine months. Radiologic analysis revealed a 1.2 cm multiseptated, cystic, solid nodule in an anterior superficial lobe of the right parotid gland. Fine-needle aspiration cytology revealed many irregular overlapping sheets or clusters of ductal epithelial cells forming solid, pseudopapillary, and cribriform architectures. Nuclei of the tumor cells revealed inconspicuous atypia with minimal size variation. On the basis of these findings, we confirmed a diagnosis of ductal epithelial proliferative lesion, favoring neoplasm, with uncertain malignant potential. Tumor excision was performed, revealing a tiny multicystic nodule (0.7 cm). Histopathologically, this tumor showed the characteristic morphology of LGCCC. This is the first report of cytomorphological findings of LGCCC in Korea.
-
Citations
Citations to this article as recorded by- Duct tales of a parotid gland swelling
Swati Raj, Monika Singh, Mamta Gupta, Naveen Thapliyal
Cytojournal.2023; 20: 22. CrossRef - Salivary Gland Intraductal Carcinoma: How Do 183 Reported Cases Fit Into a Developing Classification
Lester D.R. Thompson, Justin A. Bishop
Advances in Anatomic Pathology.2023; 30(2): 112. CrossRef - Intraductal carcinoma of the parotid gland
Yukiya HIRATA, Kayoko HIGUCHI, Toshitaka NAGAO, Yoko ZUKERAN, Takao KINJO, Naoki WADA
The Journal of the Japanese Society of Clinical Cytology.2022; 61(6): 431. CrossRef - Intraductal carcinomas of the salivary glands: systematic review and classification of 93 published cases
Andrea Palicelli
APMIS.2020; 128(3): 191. CrossRef - What do we know about the cytological features of pure intraductal carcinomas of the salivary glands?
Andrea Palicelli
Cytopathology.2020; 31(3): 185. CrossRef - Diagnosing Recently Defined and Uncommon Salivary Gland Lesions in Limited Cellularity Specimens: Cytomorphology and Ancillary Studies
Esther Diana Rossi, Zubair Baloch, William Faquin, Liron Pantanowitz
AJSP: Reviews and Reports.2020; 25(5): 210. CrossRef - Low-grade intraductal carcinoma of salivary glands: A systematic review of this rare entity
Francesco Giovacchini, Caterina Bensi, Stefano Belli, Maria Elena Laurenti, Martina Mandarano, Daniele Paradiso, Michele Giansanti, Antonio Tullio
Journal of Oral Biology and Craniofacial Research.2019; 9(1): 96. CrossRef - The rare entity of cystadenocarcinoma (CAC) in parotid gland: A single-center experience
Bing Guo, Yu-an Cao, Xingjun Qin, Chunyue Ma
Journal of Cranio-Maxillofacial Surgery.2019; 47(5): 826. CrossRef - Cytopathology approach to rare salivary gland lesions with oncocytic features
Siba El Hussein, Samer N. Khader
Diagnostic Cytopathology.2019; 47(10): 1090. CrossRef - Unicystic high‐grade intraductal carcinoma of the parotid gland: cytological and histological description with clinic–pathologic review of the literature
Andrea Palicelli, Paola Barbieri, Narciso Mariani, Paola Re, Stefania Galla, Raffaele Sorrentino, Francesca Locatelli, Nunzio Salfi, Guido Valente
APMIS.2018; 126(9): 771. CrossRef - Low-grade cribriform cystadenocarcinoma arising from a minor salivary gland: a case report
Masashi Kimura, Shinji Mii, Shinichi Sugimoto, Kosuke Saida, Shojiroh Morinaga, Masahiro Umemura
Journal of Oral Science.2016; 58(1): 145. CrossRef - A Case of Cystadenocarcinoma Arising from Parotid Gland
Jong Chul Hong, Tae Kyoung Koh, Min Gyoung Pak, Heon Soo Park
Korean Journal of Otorhinolaryngology-Head and Neck Surgery.2016; 59(4): 300. CrossRef - Mammary analogue secretory carcinoma of parotid gland
Atsuko NASU, Sakae HATA, Masaru FUJITA, Toyoko YAMAUCHI, Satoko NAKAMURA, Takehiro TANAKA, Kouichi ICHIMURA, Hiroyuki YANAI
The Journal of the Japanese Society of Clinical Cytology.2016; 55(2): 112. CrossRef
- Duct tales of a parotid gland swelling
- Diffuse Large B-Cell Lymphoma Arising in Warthin's Tumor: Case Study and Review of the Literature
- Güliz Özkök, Funda Taşlı, Nazan Özsan, Rafet Öztürk, Hakan Postacı
- Korean J Pathol. 2013;47(6):579-582. Published online December 24, 2013
- DOI: https://doi.org/10.4132/KoreanJPathol.2013.47.6.579
- 9,404 View
- 55 Download
- 16 Crossref
-
 Abstract
Abstract
 PDF
PDF Warthin's tumor is the second most common type of salivary gland tumor. Microscopically, Warthin's tumor displays a proliferative epithelial component and lymphoid stroma. Carcinomas arising from the epithelial component are well known, but malignant transformations of the lymphoid stroma are rare. When they do occur, they are most commonly B-cell type non-Hodgkin lymphomas. A 60-year-old male patient underwent surgical resection of a parotid mass. After superficial parotidectomy, microscopic examination indicated that the tumor was of epithelial components with basaloid and oncocytic columns of cells neighboring lymphoid components. In addition to the lymphoid follicles with distinct germinal centers, there were large, bizarre and extremely atypical neoplastic cells seen in the lymphoid component. Large neoplastic cells were diffusely CD20 and CD30 positive. The patient was diagnosed with "Warthin's tumor and diffuse large B-cell lymphoma with expression of CD30." The histopathologic and clinical features are discussed along with a review of the literature.
-
Citations
Citations to this article as recorded by- From the archives of MD Anderson Cancer Center: Small lymphocytic lymphoma/chronic lymphocytic leukemia diagnosed in Warthin tumor
Jing Zhou, L. Jeffrey Medeiros
Annals of Diagnostic Pathology.2025; 79: 152533. CrossRef - Warthin’s Tumor Involved by Monoclonal B-Cell Lymphocytosis: A Case Report
Alexie Kindy, Ziang Wang, Robin Gautam, Robert Bennett, Zijian Wang
Cureus.2025;[Epub] CrossRef - Warthin tumor concomitant with mantle cell lymphoma: a case report and review of literature
Hai-Chao Tong, Shuang Ma, Lan Chen, Xiangyun Meng, Ying-Chun Li, Le-Yao Li, Lingyun Dong, Wan-Lin Zhang, Tyler Wildes, Lian-He Yang, Endi Wang
Diagnostic Pathology.2024;[Epub] CrossRef - A Rare Diagnosis of Parotid Gland Follicular Lymphoma Arising in Warthin Tumor: Case Report and Literature Review
Ido Vaknin, Irit Allon, Shirley Zafrir-Haver, Alex Abramson
Medicina.2024; 60(12): 2086. CrossRef - Warthin’s Tumor of the Parotid Gland With Degeneration to Diffuse Large B-cell Lymphoma: A Case Report and Review of Literature
Mauricio Gutierrez-Alvarez, Cynthia Martinez, Ana Priscila Campollo Lopez, Kevin Fuentes, Jorge Alberto Robles Aviña
Cureus.2023;[Epub] CrossRef - Pathogenesis Analysis of Salivary Gland Tumors Through the Expression of Programmed Death-Ligand 1 (PD-L1)
Aisyah Izzatul Muna, Maria Evata Krismawati Surya, Meiske Margaretha, Jane Kosasih, Mei Syafriadi
Indian Journal of Otolaryngology and Head & Neck Surgery.2023; 75(4): 3098. CrossRef - Role of cytotoxic T-lymphocyte antigen 4 (CTLA-4) expression in the pathogenesis of Warthin’s tumor growth
Alvionika Nadyah Qotrunnada, Tecky Indriana, Jane Kosasih, Meiske Margaretha, Mei Syafriadi
Dental Journal (Majalah Kedokteran Gigi).2022; 55(4): 194. CrossRef - Lymphoepithelial Carcinoma of Salivary Glands
Lester D.R. Thompson, Rumeal D. Whaley
Surgical Pathology Clinics.2021; 14(1): 75. CrossRef - Follicular Lymphoma Diagnosed in Warthin Tumor: A Case Report and Review of the Literature
Fnu Alnoor, Jatin S. Gandhi, Matthew K. Stein, Joel F. Gradowski
Head and Neck Pathology.2020; 14(2): 386. CrossRef - Prevalence of Lymphoid Neoplasia in a Retrospective Analysis of Warthin Tumor: A Single Institution Experience
F. N. U. Alnoor, Jatin S. Gandhi, Matthew K. Stein, Jorge Solares, Joel F. Gradowski
Head and Neck Pathology.2020; 14(4): 944. CrossRef - A Case of Diffuse Large B Cell Lymphoma in Submandibular Gland
Sang Hoo Park, Min Gyoung Pak, Dong Kun Lee
Journal of Clinical Otolaryngology Head and Neck Surgery.2020; 31(1): 111. CrossRef - Analysis of the Clinical Relevance of Histological Classification of Benign Epithelial Salivary Gland Tumours
Henrik Hellquist, António Paiva-Correia, Vincent Vander Poorten, Miquel Quer, Juan C. Hernandez-Prera, Simon Andreasen, Peter Zbären, Alena Skalova, Alessandra Rinaldo, Alfio Ferlito
Advances in Therapy.2019; 36(8): 1950. CrossRef - Diffuse large B-cell lymphoma arising from follicular lymphoma with warthin’s tumor of the parotid gland - immunophenotypic and genetic features: A case report
Chang-Song Wang, Xia Chu, Di Yang, Lei Ren, Nian-Long Meng, Xue-Xia Lv, Tian Yun, Yan-Sha Cao
World Journal of Clinical Cases.2019; 7(22): 3895. CrossRef - Presentation of Chronic Lymphocytic Leukemia/Small Lymphocytic Lymphoma in a Warthin Tumor: Case Report and Literature Review
Hadeel Jawad, Peter McCarthy, Gerard O’Leary, Cynthia C. Heffron
International Journal of Surgical Pathology.2018; 26(3): 256. CrossRef - Bilateral Warthin tumor in psoriatic patients in therapy with multiple immunosuppressive therapy
M Burlando, E Cozzani, C Chinazzo, M larosa, M Boggio, A Parodi
International Journal of Immunopathology and Pharmacology.2015; 28(1): 138. CrossRef - Nodular Lymphocyte-Predominant Hodgkin Lymphoma in a Warthin Tumor of the Parotid Gland
Arianna Di Napoli, Giuseppe Mallel, Armando Bartolazzi, Elena Cavalieri, Roberto Becelli, Claudia Cippitelli, Luigi Ruco
International Journal of Surgical Pathology.2015; 23(5): 419. CrossRef
- From the archives of MD Anderson Cancer Center: Small lymphocytic lymphoma/chronic lymphocytic leukemia diagnosed in Warthin tumor
- Fine-Needle Aspiration Cytology of Low-Grade Cribriform Cystadenocarcinoma with Many Psammoma Bodies of the Salivary Gland
- Ji Yun Jeong, Dongbin Ahn, Ji Young Park
- Korean J Pathol. 2013;47(5):481-485. Published online October 25, 2013
- DOI: https://doi.org/10.4132/KoreanJPathol.2013.47.5.481
- 9,011 View
- 49 Download
- 12 Crossref
-
 Abstract
Abstract
 PDF
PDF Low-grade cribriform cystadenocarcinoma (LGCCC) is a rare salivary gland tumor that was recently defined as a variant of cystadenocarcinoma by the 2005 World Health Orgazniation (WHO) classification system. We report cytologic findings of an unusual case of LGCCC with many psammoma bodies. A 90-year-old man presented a palpable mass on his left parotid gland. Fine-needle aspiration (FNA) cytology showed tumor cells that were arranged in clusters and dispersed individually. The tumor cells showed mild atypia and had clear or dense cytoplasm with some vacuoles. Numerous psammoma bodies were noted. After surgical resection, the histologic examination revealed a mixed solid and cystic mass showing intraductal growth with focal stromal invasion. The S-100 protein expressed in the tumor cells, but smooth muscle actin and p63 were positive only in myoepithelial cells. Although LGCCCs resemble other salivary gland tumors, differentiating LGCCC during preoperative FNA is important to avoid unnecessary overtreatment.
-
Citations
Citations to this article as recorded by- Salivary Gland Intraductal Carcinoma: How Do 183 Reported Cases Fit Into a Developing Classification
Lester D.R. Thompson, Justin A. Bishop
Advances in Anatomic Pathology.2023; 30(2): 112. CrossRef - Duct tales of a parotid gland swelling
Swati Raj, Monika Singh, Mamta Gupta, Naveen Thapliyal
Cytojournal.2023; 20: 22. CrossRef - Intraductal carcinoma of the parotid gland
Yukiya HIRATA, Kayoko HIGUCHI, Toshitaka NAGAO, Yoko ZUKERAN, Takao KINJO, Naoki WADA
The Journal of the Japanese Society of Clinical Cytology.2022; 61(6): 431. CrossRef - Intraductal carcinoma of the retromolar trigone found with elevated serum CEA and CA19-9 levels: a case report
Mao KAWAKAMI, Nobuhiro UEDA, Yuka TAKAHASHI, Sho ARIKAWA, Nobuhiro YAMAKAWA, Tadaaki KIRITA
Japanese Journal of Oral and Maxillofacial Surgery.2021; 67(5): 292. CrossRef - Endoscopic trans‐pterygoid resection of a low‐grade cribriform cystadenocarcinoma of the infratemporal fossa
Vikram G. Ramjee, Landon J. Massoth, John P. Richards, Kibwei A. McKinney
World Journal of Otorhinolaryngology - Head and Neck Surgery.2020; 6(2): 115. CrossRef - Psammoma Bodies in a Large Myoepithelioma
Marcela Pessoa de Melo, Diego Filipe Bezerra Silva, Rodrigo Alves Ribeiro, Tony Santos Peixoto, Daliana Queiroga de Castro Gomes, Pollianna Muniz Alves, Cassiano Francisco Weege Nonaka, Bárbara Vanessa de Brito Monteiro
Journal of Craniofacial Surgery.2020; 31(4): e326. CrossRef - Low-grade intraductal carcinoma of salivary glands: A systematic review of this rare entity
Francesco Giovacchini, Caterina Bensi, Stefano Belli, Maria Elena Laurenti, Martina Mandarano, Daniele Paradiso, Michele Giansanti, Antonio Tullio
Journal of Oral Biology and Craniofacial Research.2019; 9(1): 96. CrossRef - What is your diagnosis? Submandibular mass in a dog
Julie Allen, Ashley M. Talley, Carol B. Grindem, Jennifer A. Neel
Veterinary Clinical Pathology.2018; 47(4): 676. CrossRef - Primary acinic cell carcinoma of the lung with psammoma bodies: A case report and review of literature
Xiu-Peng Zhang, Gui-Yang Jiang, Qing-Fu Zhang, Hong-Tao Xu, Qing-Chang Li, En-Hua Wang
Pathology - Research and Practice.2017; 213(4): 405. CrossRef - Cytology of low‐grade cribriform cystadenocarcinoma in salivary glands: Cytological and immunohistochemical distinctions from other salivary gland neoplasms
Yoshiki Ohta, Yuko Hirota, Yohko Kohno, Koji Kishimoto, Tomoko Norose, Nobuyuki Ohike, Masafumi Takimoto, Akira Shiokawa, Hidekazu Ota
Diagnostic Cytopathology.2016; 44(3): 241. CrossRef - Low-grade cribriform cystadenocarcinoma arising from a minor salivary gland: a case report
Masashi Kimura, Shinji Mii, Shinichi Sugimoto, Kosuke Saida, Shojiroh Morinaga, Masahiro Umemura
Journal of Oral Science.2016; 58(1): 145. CrossRef - A Case of Cystadenocarcinoma Arising from Parotid Gland
Jong Chul Hong, Tae Kyoung Koh, Min Gyoung Pak, Heon Soo Park
Korean Journal of Otorhinolaryngology-Head and Neck Surgery.2016; 59(4): 300. CrossRef
- Salivary Gland Intraductal Carcinoma: How Do 183 Reported Cases Fit Into a Developing Classification
- A Case of Multifocal Papillary Thyroid Carcinoma Consisting of One Encapsulated Follicular Variant with
BRAF K601E Mutation and Three Conventional Types withBRAF V600E Mutation - Wook Youn Kim, Young Sin Ko, Tae Sook Hwang, Hye Seung Han, So Dug Lim, Wan Seop Kim, Seo Young Oh
- Korean J Pathol. 2013;47(3):293-298. Published online June 25, 2013
- DOI: https://doi.org/10.4132/KoreanJPathol.2013.47.3.293
- 10,920 View
- 53 Download
- 7 Crossref
-
 Abstract
Abstract
 PDF
PDF Multifocal papillary thyroid carcinoma (mPTC) comprises about 20-30% of PTC. In mPTC, individual tumor foci can be identical or frequently composed of different histological types including follicular, solid, tall-cell or conventional patterns. We report a case of mPTC consisting of one encapsulated follicular variant of papillary thyroid carcinoma (FVPTC) and three conventional PTCs in a 44-year-old woman. This case genetically demonstrates unique features including the simultaneous presence of the
BRAF V600E (T1799A) mutation and theBRAF K601E (A1801G) mutation in conventional PTC and FVPTC, respectively.-
Citations
Citations to this article as recorded by- BRAF K601E Mutation in Oncocytic Carcinoma of the Thyroid: A Case Report and Literature Review
Antonio Matrone, Fabrizia Citro, Carla Gambale, Alessandro Prete, Elisa Minaldi, Raffaele Ciampi, Teresa Ramone, Gabriele Materazzi, Liborio Torregrossa, Rossella Elisei
Journal of Clinical Medicine.2023; 12(22): 6970. CrossRef - Case of aggressive metastatic follicular variant papillary thyroid carcinoma with BRAF K601E and BCORL1 mutations
Doaa Attia, Alexander Lurie, Qihui Zhai, Thomas Mesko, Robert Smallridge
BMJ Case Reports.2020; 13(6): e234208. CrossRef - BRAF gene: From human cancers to developmental syndromes
Muhammad Ramzan Manwar Hussain, Mukhtiar Baig, Hussein Sheik Ali Mohamoud, Zaheer Ulhaq, Daniel C. Hoessli, Ghaidaa Siraj Khogeer, Ranem Radwan Al-Sayed, Jumana Yousuf Al-Aama
Saudi Journal of Biological Sciences.2015; 22(4): 359. CrossRef - Clinical significance of BRAF V600E mutation in 154 patients with thyroid nodules
LINGYING YU, LIZHEN MA, QIAOFENG TU, YI ZHANG, YUEMING CHEN, DAOJUN YU, SHAOYU YANG
Oncology Letters.2015; 9(6): 2633. CrossRef - Clinicopathological Features of Rare BRAF Mutations in Korean Thyroid Cancer Patients
Uiju Cho, Woo Jin Oh, Ja Seong Bae, Sohee Lee, Young Sub Lee, Gyeong Sin Park, Youn Soo Lee, Chan Kwon Jung
Journal of Korean Medical Science.2014; 29(8): 1054. CrossRef - Recurrent Thyroid Papillary Carcinoma in Children Under Ten Years Old: Report of Two Cases and Literature Review
Byeong-Joo Noh, Ji-Youn Sung, Youn-Wha Kim, Yong-Koo Park
Korean Journal of Pathology.2014; 48(4): 297. CrossRef - Anaplastic Transformation of Papillary Thyroid Carcinoma in a Young Man: A Case Study with Immunohistochemical andBRAFAnalysis
Ji Hye Park, Hyeong Ju Kwon, Cheong Soo Park, SoonWon Hong
Korean Journal of Pathology.2014; 48(3): 234. CrossRef
- BRAF K601E Mutation in Oncocytic Carcinoma of the Thyroid: A Case Report and Literature Review
- Heterotopic Intestinal Cyst of the Submandibular Gland: A Case Study
- Mi Jung Kwon, Dong Hoon Kim, Hye-Rim Park, Soo Kee Min, Jinwon Seo, Eun Soo Kim, Si Whan Kim, Bumjung Park
- Korean J Pathol. 2013;47(3):279-283. Published online June 25, 2013
- DOI: https://doi.org/10.4132/KoreanJPathol.2013.47.3.279
- 9,687 View
- 42 Download
- 8 Crossref
-
 Abstract
Abstract
 PDF
PDF Heterotopic gastrointestinal cysts are rarely found in the oral cavity. Most of these cysts are lined with gastric mucosa and involve the tongue. There have been no reported heterotopic intestinal cysts of the submandibular gland that are completely lined with colonic mucosa. An 8-year-old girl presented with an enlarging swelling in the left submandibular area, and a 4-cm unilocular cyst was fully excised. The cyst was completely lined with colonic mucosa that was surrounded by smooth muscle layer, and the lining cells were positive for CDX-2, an intestinal marker, indicating a high degree of differentiation. The pathogenesis remains unclear, but it may be related to the misplacement of embryonic rests within the oral cavity during early fetal development. Although heterotopic intestinal cysts rarely occur in the submandibular gland, they should be considered in the differential diagnosis of facial swellings in the pediatric population.
-
Citations
Citations to this article as recorded by- Large oral heterotopic gastrointestinal cyst in a child: A case report and update
Débora Frota Colares, Julliany Taverny Sousa, André Luis Alves Borges, Bárbara de Assis Araújo, José Sandro Pereira da Silva, Lélia Batista de Souza
Journal of Oral and Maxillofacial Surgery, Medicine, and Pathology.2025; 37(5): 1137. CrossRef - Heterotopic gastrointestinal cyst of the oral cavity: A rare clinical report and literature review
Andrea Maldonado, Rubén Muñoz, Jesús Cabrera, José Alcides Almeida de Arruda, Bruno Augusto Benevenuto de Andrade, Mariana Villarroel-Dorrego, Isabella Bittencourt do Valle
Journal of Stomatology Oral and Maxillofacial Surgery.2025; 126(5): 102406. CrossRef - Atypical Extraoral Presentation of a Heterotopic Gastrointestinal Cyst on the Face: A Case Report
Anita Dhupar, Anupama Mukherjee, Anita E Spadigam, Praveen S Kumar
Cureus.2024;[Epub] CrossRef - Heterotopic gastrointestinal cyst in the floor of mouth: a case report
Naoaki SAITO, Satoshi MARUYAMA, Yusuke KATO, Ryoko TAKEUCHI, Jun-ichi TANUMA, Tadaharu KOBAYASHI
Japanese Journal of Oral and Maxillofacial Surgery.2023; 69(1): 27. CrossRef - A case report of oral heterotopic gastrointestinal cysts (HGIC) and review of the literature
Gursimran Kaur Bains, Richard Pilkington, Joanna Stafford, Sunil Bhatia
Oral Surgery.2022; 15(1): 71. CrossRef - A Rare Case of Ectopic Colonic Mucosa Presenting With Airway Compromise in a Neonate
Justin Hall, Fatima Z Aly, Julia Comer, Michael P Gebhard, Thomas Schrepfer
Cureus.2022;[Epub] CrossRef - Ultrasonic Features of Uncommon Congenital Heterotopic Colon and Pancreas in the Neck: An Extremely Rare Case Report
Yingli Wei, Zhihao Pan, Xiaoling Kang, Cuiqing Huang, Dan Chen
Frontiers in Pediatrics.2021;[Epub] CrossRef - Quiste gastrointestinal heterotópico en la cavidad oral
Beatriz Arango de Samper, Eliana Elisa Muñoz López, Estefanía Morales González
Latin American Journal of Oral and Maxillofacial Surgery.2021; 1(1): 40. CrossRef
- Large oral heterotopic gastrointestinal cyst in a child: A case report and update
- ThinPrep Cytological Findings of Desmoplastic Small Round Cell Tumor with Extensive Glandular Differentiation: A Case Study
- Hyun-Jung Kim, Byeong Seok Sohn, Ji-Eun Kwon, Jeong Yeon Kim, Kyeongmee Park
- Korean J Pathol. 2013;47(2):182-187. Published online April 24, 2013
- DOI: https://doi.org/10.4132/KoreanJPathol.2013.47.2.182
- 8,965 View
- 71 Download
- 11 Crossref
-
 Abstract
Abstract
 PDF
PDF Desmoplastic small round cell tumor (DSRCT) is a rare and highly aggressive neoplasm. The cytological diagnosis of this tumor has only been reported in a few cases. In most of these cases, the diagnosis was made using fine-needle aspiration cytology. Most DSRCTs resemble disseminated carcinomatoses in their clinical manifestation as well as cytomorphologically, even in young-adult patients. These authors report a case of using peritoneal-washing and pleural-effusion ThinPrep cytology to diagnose DSRCT, with extensive glandular differentiation and mucin vacuoles. We found that fibrillary stromal fragment, clinical setting, and adjunctive immunocytochemical staining were most helpful for avoiding misdiagnosis.
-
Citations
Citations to this article as recorded by- Desmoplastic Small Round Cell Tumor: Study of Cytomorphologic and Immunophenotypical Features in Seven Cases, One With Unusual Rhabdoid Morphology
Sameer Chhetri Aryal, Khalid Shittu, Mohamed Mustafa, Fatimah I. Alruwaii, Kyle D. Perry, Lisi Yuan
Diagnostic Cytopathology.2026;[Epub] CrossRef - A Rare and Aggressive Abdominopelvic Tumor: A Case of Desmoplastic Small Round Cell Tumor in a Young Male
Yağmur Sena Tosun, Aytan Babazade, Emine Sena Sözen, Betül Erişmiş, Enes Seyda Şahiner
Caucasian Medical Journal.2025; 3(1): 4. CrossRef - Desmoplastic Small Round Cell Tumor Involving Serous Fluid: Cytologic Features and Diagnostic Pitfalls: A Series of 8 Cases
Nibras L Fakhri, Qiong Gan
American Journal of Clinical Pathology.2023; 160(4): 417. CrossRef - A Review of Effusion Cytomorphology of Small Round Cell Tumors
Lucy M. Han, Christopher J. VandenBussche, Mads Abildtrup, Ashish Chandra, Poonam Vohra
Acta Cytologica.2022; 66(4): 336. CrossRef - Intra-abdominal desmoplastic small blue round cell tumor: A case report
Tareq Hamed Al Taei, Hasan Al Fardan, Sarah Ali Al Mail
Radiology Case Reports.2022; 17(12): 4502. CrossRef - Desmoplastic Small Round Cell Tumor of the Kidney: Report of a Case, Literature Review, and Comprehensive Discussion of the Distinctive Morphologic, Immunohistochemical, and Molecular Features in the Differential Diagnosis of Small Round Cell Tumors Affec
Carlos A. Galliani, Michele Bisceglia, Antonio Del Giudice, Giuseppe Cretì
Advances in Anatomic Pathology.2020; 27(6): 408. CrossRef - Intra-abdominal desmoplastic small round cell tumors: CT and FDG-PET/CT findings with histopathological association
JINGJING CHEN, ZENGJIE WU, BINBIN SUN, DACHENG LI, ZHENGUANG WANG, FANGJUN LIU, HUI HUA
Oncology Letters.2016; 11(5): 3298. CrossRef - Desmoplastic small round cell tumor with sphere‐like clusters mimicking adenocarcinoma
Yukinori Hattori, Akihiko Yoshida, Naoshi Sasaki, Yasuo Shibuki, Kenji Tamura, Koji Tsuta
Diagnostic Cytopathology.2015; 43(3): 214. CrossRef - Tumor intraabdominal desmoplásico de células pequeñas y redondas
Andrés Alejandro Briseño-Hernández, Deissy Roxana Quezada-López, Lilia Edith Corona-Cobián, Agar Castañeda-Chávez, Alfonso Tonatiuh Duarte-Ojeda, Michel Dassaejv Macías-Amezcua
Cirugía y Cirujanos.2015; 83(3): 243. CrossRef - Intra-abdominal desmoplastic small round cell tumour
Andrés Alejandro Briseño-Hernández, Deissy Roxana Quezada-López, Lilia Edith Corona-Cobián, Agar Castañeda-Chávez, Alfonso Tonatiuh Duarte-Ojeda, Michel Dassaejv Macías-Amezcua
Cirugía y Cirujanos (English Edition).2015; 83(3): 243. CrossRef - Diagnostic Pitfalls of Differentiating Desmoplastic Small Round Cell Tumor (DSRCT) From Wilms Tumor (WT)
Michael A. Arnold, Lynn Schoenfield, Berkeley N. Limketkai, Christina A. Arnold
American Journal of Surgical Pathology.2014; 38(9): 1220. CrossRef
- Desmoplastic Small Round Cell Tumor: Study of Cytomorphologic and Immunophenotypical Features in Seven Cases, One With Unusual Rhabdoid Morphology
- Fine Needle Aspiration Cytology of Thyroid Follicular Neoplasm: Cytohistologic Correlation and Accuracy
- Changyoung Yoo, Hyun Joo Choi, Soyoung Im, Ji Han Jung, Kiouk Min, Chang Suk Kang, Young-Jin Suh
- Korean J Pathol. 2013;47(1):61-66. Published online February 25, 2013
- DOI: https://doi.org/10.4132/KoreanJPathol.2013.47.1.61
- 13,682 View
- 87 Download
- 21 Crossref
-
 Abstract
Abstract
 PDF
PDF Background This study evaluated the accuracy of fine needle aspiration cytology (FNAC) in cases of follicular neoplasm (FN) on the basis of histologic diagnosis, and reviewed the cytologic findings of FN according to the FNAC.
Methods Among the 66 cases diagnosed with thyroid FN by FNAC during the 7-year period from 2003 to 2009, 36 cases that had undergone thyroid surgery were available for review. Cytologic diagnosis was compared with the histologic diagnosis of each case.
Results Among the 36 cases with a cytologic diagnosis of thyroid FN, histologic diagnosis was as follows: 20 follicular adenomas (55.6%), 3 Hurthle cell adenomas (8.3%), 2 follicular carcinomas (5.6%), 8 nodular goiters (22.2%), 2 papillary carcinomas (5.6%), and 1 Hashimoto's thyroiditis (2.8%), resulting in a diagnostic accuracy of FNAC for thyroid FN of 69.5%.
Conclusions This study shows that FNAC for thyroid FN is a useful primary screening method because when FN is diagnosed by FNAC, the rate of FN histologic diagnosis is relatively high, however, adequate sampling and experience is a prerequisite for this procedure.
-
Citations
Citations to this article as recorded by- Practical and challenging issue in thyroid cytopathology
Qianqian Zhang, Belen Padial Urtueta, Elisabetta Merenda, Gabriele Rotondaro, Noemi Morelli, Alessia Piermattei, Patrizia Straccia, Federica Cianfrini, Angela Feraco, Alessia Granitto, Antonino Mule, Esther Diana Rossi
Human Pathology.2026; 169: 106019. CrossRef - Evaluation and Management of Thyroid Nodules: A Joint Consensus Statement From the British Thyroid Association (BTA), British Association of Endocrine and Thyroid Surgeons (BAETS) and Collaborating Bodies
Ram Moorthy, Saba P. Balasubramanian, Kate Farnell, Mairead Kelly, Gitta Madani, Mufaddal Moonim, Carla Moran, Julia Priestley, Michael Stechman, Emma Watts, Kristien Boelaert
Clinical Endocrinology.2026; 104(6): 682. CrossRef - Indications for Core Needle Biopsy in Low-Suspicion Thyroid Nodules on US
Myoung Kyoung Kim, Jung Hee Shin, Haejung Kim, Soo Yeon Hahn
Journal of the Korean Society of Radiology.2026;[Epub] CrossRef - Diagnostic approach to FNA biopsy of cystic lesions of the head and neck
Stefen Andrianus, Olivia Leung, Zubair Baloch
Cancer Cytopathology.2025;[Epub] CrossRef - Malignancy risk in AUS thyroid lesions: comparison between FNA and CNB with implications for NIFTP diagnosis
Yeseul Kim, Jae Ho Shin, You-Na Sung, Dawon Park, Harim Oh, Hyo Seon Ryu, Kyeong Jin Kim, Hyun Joo Kim, Sin Gon Kim, Hoon Yub Kim, Kwang Yoon Jung, Seung-Kuk Baek, Sangjeong Ahn
Frontiers in Endocrinology.2025;[Epub] CrossRef - Correlation of Bethesda Categories in Thyroid Lesions With Histopathology: A Single Tertiary Care Center Experience
Sreenidhi Sreeram, Leena Dennis Joseph, Subalakshmi Balasubramanian
Cureus.2025;[Epub] CrossRef - Prevalence and Predictors of Malignancy in Contralateral Thyroid Lobe in Patients Undergoing Completion Thyroidectomy
Pradipta Kumar Parida, Siddhartha Pradhan, Chapity Preetam, Pradeep Pradhan, Dillip Kumar Samal, Saurav Sarkar
Indian Journal of Otolaryngology and Head & Neck Surgery.2022; 74(S2): 2053. CrossRef - Ultrasonographic and cytologic assessments of follicular neoplasms of the thyroid: Predictive features differentiating follicular carcinoma from follicular adenoma
Hye Shin Ahn, Hee Sung Kim, Min Ji Hong, Paula Soares
PLOS ONE.2022; 17(7): e0271437. CrossRef - 2019 Practice guidelines for thyroid core needle biopsy: a report of the Clinical Practice Guidelines Development Committee of the Korean Thyroid Association
Chan Kwon Jung, Jung Hwan Baek, Dong Gyu Na, Young Lyun Oh, Ka Hee Yi, Ho-Cheol Kang
Journal of Pathology and Translational Medicine.2020; 54(1): 64. CrossRef - Preoperative diagnostic categories of fine needle aspiration cytology for histologically proven thyroid follicular adenoma and carcinoma, and Hurthle cell adenoma and carcinoma: Analysis of cause of under- or misdiagnoses
Hee Young Na, Jae Hoon Moon, June Young Choi, Hyeong Won Yu, Woo-Jin Jeong, Yeo Koon Kim, Ji-Young Choe, So Yeon Park, Paula Soares
PLOS ONE.2020; 15(11): e0241597. CrossRef - Core needle biopsy of thyroid nodules: outcomes and safety from a large single-center single-operator study
Jooae Choe, Jung Hwan Baek, Hye Sun Park, Young Jun Choi, Jeong Hyun Lee
Acta Radiologica.2018; 59(8): 924. CrossRef - Cytological Features That Differentiate Follicular Neoplasm from Mimicking Lesions
Kanghee Han, Hwa-Jeong Ha, Joon Seog Kong, Jung-Soon Kim, Jae Kyung Myung, Jae Soo Koh, Sunhoo Park, Myung-Soon Shin, Woo-Tack Song, Hye Sil Seol, Seung-Sook Lee
Journal of Pathology and Translational Medicine.2018; 52(2): 110. CrossRef - Comparison of the Diagnostic Efficacy of Ultrasound‐Guided Core Needle Biopsy With 18‐ Versus 20‐Gauge Needles for Thyroid Nodules
Hye Shin Ahn, Mirinae Seo, Su Min Ha, Hee Sung Kim
Journal of Ultrasound in Medicine.2018; 37(11): 2565. CrossRef - Subclassification of Bethesda Atypical and Follicular Neoplasm Categories According to Nuclear and Architectural Atypia Improves Discrimination of Thyroid Malignancy Risk
Joel Xue Yi Lim, Min En Nga, Dedrick Kok Hong Chan, Wee Boon Tan, Rajeev Parameswaran, Kee Yuan Ngiam
Thyroid.2018; 28(4): 511. CrossRef - The expression profile of integrin receptors and osteopontin in thyroid malignancies varies depending on the tumor progression rate and presence of BRAF V600E mutation
Galina Chernaya, Nina Mikhno, Tatiana Khabalova, Svetlana Svyatchenko, Lyudmila Mostovich, Sergey Shevchenko, Lyudmila Gulyaeva
Surgical Oncology.2018; 27(4): 702. CrossRef - The Usefulness of Immunocytochemistry of CD56 in Determining Malignancy from Indeterminate Thyroid Fine-Needle Aspiration Cytology
Hyunseo Cha, Ju Yeon Pyo, Soon Won Hong
Journal of Pathology and Translational Medicine.2018; 52(6): 404. CrossRef - Core Needle Biopsy of the Thyroid: 2016 Consensus Statement and Recommendations from Korean Society of Thyroid Radiology
Dong Gyu Na, Jung Hwan Baek, So Lyung Jung, Ji-hoon Kim, Jin Yong Sung, Kyu Sun Kim, Jeong Hyun Lee, Jung Hee Shin, Yoon Jung Choi, Eun Ju Ha, Hyun Kyung Lim, Soo Jin Kim, Soo Yeon Hahn, Kwang Hwi Lee, Young Jun Choi, Inyoung Youn, Young Joong Kim, Hye Sh
Korean Journal of Radiology.2017; 18(1): 217. CrossRef - Radiofrequency ablation of small follicular neoplasms: initial clinical outcomes
Su Min Ha, Jin Yong Sung, Jung Hwan Baek, Dong Gyu Na, Ji-hoon Kim, Hyunju Yoo, Ducky Lee, Dong Whan Choi
International Journal of Hyperthermia.2017; : 1. CrossRef - A meta‐analytic review of the Bethesda System for Reporting Thyroid Cytopathology: Has the rate of malignancy in indeterminate lesions been underestimated?
Patrizia Straccia, Esther Diana Rossi, Tommaso Bizzarro, Chiara Brunelli, Federica Cianfrini, Domenico Damiani, Guido Fadda
Cancer Cytopathology.2015; 123(12): 713. CrossRef - Impact of NRAS Mutations on the Diagnosis of Follicular Neoplasm of the Thyroid
Ja-Seong Bae, Seung Kyu Choi, Sora Jeon, Yourha Kim, Sohee Lee, Youn Soo Lee, Chan Kwon Jung
International Journal of Endocrinology.2014; 2014: 1. CrossRef - Diagnosis of Thyroid Follicular Neoplasm: Fine-Needle Aspiration Versus Core-Needle Biopsy
Ra Gyoung Yoon, Jung Hwan Baek, Jeong Hyun Lee, Young Jun Choi, Min Ji Hong, Dong Eun Song, Jae Kyun Kim, Jong Ho Yoon, Won Bae Kim
Thyroid.2014; 24(11): 1612. CrossRef
- Practical and challenging issue in thyroid cytopathology
- Finding and Characterizing Mammary Analogue Secretory Carcinoma of the Salivary Gland
- Min Jung Jung, Joon Seon Song, Sang Yoon Kim, Soon Yuhl Nam, Jong-Lyel Roh, Seung-Ho Choi, Sung-Bae Kim, Kyung-Ja Cho
- Korean J Pathol. 2013;47(1):36-43. Published online February 25, 2013
- DOI: https://doi.org/10.4132/KoreanJPathol.2013.47.1.36
- 15,531 View
- 118 Download
- 74 Crossref
-
 Abstract
Abstract
 PDF
PDF Background A new tumor entity of the salivary glands, mammary analogue secretory carcinoma (MASC) with
ETV6-NTRK3 translocation, has recently been proposed. MASC was originally diagnosed as adenocarcinoma, not otherwise specified (ANOS), or acinic cell carcinoma (AciCC) by the current World Health Organization classification. We aimed to identify MASC cases by molecular tests, and to characterize their clinical, histological, and immunohistochemical features.Methods Thirty cases of MASC candidates were selected after review of 196 salivary gland tumors, and subjected to break-apart
ETV6 fluorescencein situ hybridization (FISH), and immunohistochemical study for S100 protein, gross cystic disease fluid protein 15, DOG1, estrogen receptor, and progesterone receptor.Results Valid FISH results were obtained in 23 cases, and 13 positive cases were retrieved. MASCs were histologically varied, and the most frequent features observed in 10 cases were low-grade papillary/cystic/glandular patterns, intraluminal secretory materials, ovoid/wrinkled nuclei, and relatively abundant granular eosinophilic cytoplasms, corresponding to papillary-cystic or follicular types of AciCC. All cases showed diffuse immunopositivity for S100 protein. Three cases developed recurrences, but all patients remained alive.
Conclusions MASC could be a molecularly well-defined salivary gland neoplasm, encompassing some portions of AciCC and ANOS, but its histological spectrum and clinical implication require further investigation.
-
Citations
Citations to this article as recorded by- Rare Tumors of Oral Cavity: A Case Report and Literature Review on Secretory Carcinoma of Minor Salivary Glands
Emilio Salerno, Silvio Abati, Gianluigi Arrigoni, Daniela Finocchiaro, Giorgio Gastaldi, Alessandra Lissoni, Andrea Galli, Matteo Trimarchi
Clinical and Experimental Dental Research.2025;[Epub] CrossRef - Salivary Gland Secretory Carcinoma; Review of 13 Years World‐Wide Experience and Meta‐Analysis
Eyal Yosefof, Tomer Boldes, Daniel Dan, Eyal Robenshtok, Yulia Strenov, Gideon Bachar, Thomas Shpitzer, Aviram Mizrachi
The Laryngoscope.2024; 134(4): 1716. CrossRef - Salivary gland secretory carcinoma presenting as a cervical soft tissue mass: a case report
Parisa Mokhles, Alireza Sadeghipour, Pegah Babaheidarian, Saleh Mohebbi, Zahra Keshtpour Amlashi, Mohammad Hadi Gharib, Mohammad Saeid Ahmadi, Zeinab Khastkhodaei
Journal of Medical Case Reports.2024;[Epub] CrossRef - Mammary analogue secretory carcinoma of the head and neck — Clinicopathological, imaging features and prognosis analysis
Runjia Liu, Chuanzheng Sun, Likang Zhao, Shiyu Zhou, Tao Xie, Ji Zhang, Dengpeng Tang, Lei Li, Yan Xi
Journal of Radiation Research and Applied Sciences.2024; 17(2): 100914. CrossRef - Mammary analogue secretory carcinoma involving submandibular gland: Diagnostic pitfall with review of literature
Nimisha Dhankar, Nidhi Verma, Abhinav Agarwal, Ravi Mehar, Sunil Pasricha
Journal of Cancer Research and Therapeutics.2024; 20(5): 1658. CrossRef - Metastatic salivary gland mammary analogue secretory carcinoma (MASC) of parotid gland – A rare case report in the literature review
Aynur Aliyeva, Ziya Karimov, Togay Muderris
Acta Oto-Laryngologica Case Reports.2023; 8(1): 38. CrossRef - Secretory carcinoma of minor salivary glands of buccal mucosa
Noshad Ali Langah, Abdul Ahad, Shayan Khalid Ghaloo, Muhammad Faisal, Raza Tasawar Hussain, Fareed Akbar Shah
International Journal of Surgery Case Reports.2023;[Epub] CrossRef - An Underappreciated Cytomorphological Feature of Secretory Carcinoma of Salivary Gland on Fine Needle Aspiration Biopsy: Case Report with Literature Review
Yinan Hua, Bing Leng, Kenneth E. Youens, Lina Liu
Head and Neck Pathology.2022; 16(2): 567. CrossRef - Clinicopathological investigation of secretory carcinoma cases including a successful treatment outcome using entrectinib for high-grade transformation: a case report
Kensuke Suzuki, Hiroshi Harada, Masayuki Takeda, Chisato Ohe, Yoshiko Uemura, Akihiko Kawahara, Shunsuke Sawada, Akira Kanda, Bhaswati Sengupta, Hiroshi Iwai
BMC Medical Genomics.2022;[Epub] CrossRef - DOG1 as an Immunohistochemical Marker of Acinic Cell Carcinoma: A Systematic Review and Meta-Analysis
Vincenzo Fiorentino, Patrizia Straccia, Pietro Tralongo, Teresa Musarra, Francesco Pierconti, Maurizio Martini, Guido Fadda, Esther Diana Rossi, Luigi Maria Larocca
International Journal of Molecular Sciences.2022; 23(17): 9711. CrossRef - Secretory carcinoma of the sinonasal cavity and pharynx: A retrospective analysis of four cases and literature review
Changli Yue, Xiaoli Zhao, Donglin Ma, Yingshi Piao
Annals of Diagnostic Pathology.2022; 61: 152052. CrossRef - Secretory carcinoma of the salivary gland: a multi‐institutional clinicopathologic study of 90 cases with emphasis on grading and prognostic factors
Bin Xu, Kartik Viswanathan, Kavita Umrau, Thair A. D. Al‐Ameri, Snjezana Dogan, Kelly Magliocca, Ronald A. Ghossein, Nicole A. Cipriani, Nora Katabi
Histopathology.2022; 81(5): 670. CrossRef - A systematic review of secretory carcinoma of the salivary gland: where are we?
Lísia Daltro Borges Alves, Andreia Cristina de Melo, Thayana Alves Farinha, Luiz Henrique de Lima Araujo, Leandro de Souza Thiago, Fernando Luiz Dias, Héliton Spíndola Antunes, Ana Lucia Amaral Eisenberg, Luiz Claudio Santos Thuler, Daniel Cohen Goldember
Oral Surgery, Oral Medicine, Oral Pathology and Oral Radiology.2021; 132(4): e143. CrossRef - A case report of surgical resection of secretory carcinoma in the maxillary and ethmoid sinus
Kurt Willis, Martin Bullock, Matthew H. Rigby
International Journal of Surgery Case Reports.2021; 81(C): 105750. CrossRef - High‐grade salivary gland carcinoma with the ETV6‐NTRK3 gene fusion: A case report and literature review of secretory carcinoma with high‐grade transformation
Satsuki Asai, Shinji Sumiyoshi, Yosuke Yamada, Ichiro Tateya, Toshitaka Nagao, Sachiko Minamiguchi, Hironori Haga
Pathology International.2021; 71(6): 427. CrossRef - High-grade Transformation/Dedifferentiation in Salivary Gland Carcinomas: Occurrence Across Subtypes and Clinical Significance
Alena Skalova, Ilmo Leivo, Henrik Hellquist, Abbas Agaimy, Roderick H.W. Simpson, Göran Stenman, Vincent Vander Poorten, Justin A. Bishop, Alessandro Franchi, Juan C. Hernandez-Prera, David Slouka, Stefan M. Willems, Kerry D. Olsen, Alfio Ferlito
Advances in Anatomic Pathology.2021; 28(3): 107. CrossRef - Radiological features of head and neck mammary analogue secretory carcinoma: 11 new cases with a systematic review of 29 cases reported in 28 publications
Ryo Kurokawa, Mariko Kurokawa, Akira Baba, Yoshiaki Ota, Toshio Moritani, Ashok Srinivasan
Neuroradiology.2021; 63(11): 1901. CrossRef - A case of secretory carcinoma of the salivary glands in the lower lip
Reiko OHARA, Haruki SATO, Kensuke NAGANAWA, Taihei HAYAKAWA, Tatsuya KATAOKA, Ichiro OH-IWA
Japanese Journal of Oral and Maxillofacial Surgery.2021; 67(2): 83. CrossRef - Undifferentiated and dedifferentiated head and neck carcinomas
Alessandro Franchi, Alena Skalova
Seminars in Diagnostic Pathology.2021; 38(6): 127. CrossRef - Cytopathological Findings of Secretory Carcinoma of the Salivary Gland and the Diagnostic Utility of Giemsa Staining
Yuria Egusa, Midori Filiz Nishimura, Satoko Baba, Kengo Takeuchi, Takuma Makino, Tomoyasu Tachibana, Asami Nishikori, Azusa Fujita, Hiroyuki Yanai, Yasuharu Sato
Diagnostics.2021; 11(12): 2284. CrossRef - Diagnosis and treatment of secretory carcinoma arising from the oral minor salivary gland
Masaru Ogawa, Satoshi Yokoo, Takahiro Yamaguchi, Keisuke Suzuki, Mai Seki-Soda, Takahiro Shimizu, Jun Kurihara, Takaya Makiguchi
Medicine.2021; 100(51): e28390. CrossRef - A biphasic sessile mass of the buccal mucosa
Tiffany M. Peters, James A. Phero, Brent A. Golden, Alice E. Curran
Oral Surgery, Oral Medicine, Oral Pathology and Oral Radiology.2020; 130(6): 612. CrossRef - Don't stop the champions of research now: a brief history of head and neck pathology developments
Lester D.R. Thompson, James S. Lewis, Alena Skálová, Justin A. Bishop
Human Pathology.2020; 95: 1. CrossRef - High Grade Transformation in Mucoepidermoid Carcinoma of the Minor Salivary Gland with Polyploidy of the Rearranged MAML2 Gene
Hyun Lee, Jong-Lyel Roh, Young-Jun Choi, Jene Choi, Kyung-Ja Cho
Head and Neck Pathology.2020; 14(3): 822. CrossRef - Pan‐Trk immunohistochemistry is a sensitive and specific ancillary tool for diagnosing secretory carcinoma of the salivary gland and detecting ETV6–NTRK3 fusion
Bin Xu, Mohamed R Haroon Al Rasheed, Cristina R Antonescu, Deepu Alex, Denise Frosina, Ronald Ghossein, Achim A Jungbluth, Nora Katabi
Histopathology.2020; 76(3): 375. CrossRef - Characterization of novel genetic alterations in salivary gland secretory carcinoma
Kiyong Na, Juan C. Hernandez-Prera, Jae-Yol Lim, Ha Young Woo, Sun Och Yoon
Modern Pathology.2020; 33(4): 541. CrossRef - Mammary analogue secretory carcinoma: An Indian experience of a novel entity
Zeba Nisar, JaydeepN Pol, RakhiV Jagdale, MadhuraD Phadke, GirishA Kadkol
Indian Journal of Pathology and Microbiology.2020; 63(5): 134. CrossRef - Secretory Carcinoma of Salivary Gland with High-Grade Histology Arising in Hard Palate: A Case Report
Kiyofumi Takabatake, Keisuke Nakano, Hotaka Kawai, Saori Yoshida, Haruka Omori, May Wathone Oo, Shan Qiusheng, Kenichiro Uchida, Katsuaki Mishima, Hitoshi Nagatsuka
Reports.2020; 3(2): 6. CrossRef - Secretory carcinoma of the salivary gland (mammary analogue secretory carcinoma) in children
I. V. Sidorov, I. S. Kletskaya, D. M. Konovalov
Arkhiv patologii.2020; 82(2): 43. CrossRef - Secretory carcinoma of the major salivary gland: Provincial population‐based analysis of clinical behavior and outcomes
Gareth Ayre, Martin Hyrcza, Jonn Wu, Eric Berthelet, Alena Skálová, Tom Thomson
Head & Neck.2019; 41(5): 1227. CrossRef - Higher Ki67 Index, Nodal Involvement, and Invasive Growth Were High Risk Factors for Worse Prognosis in Conventional Mammary Analogue Secretory Carcinoma
Jingjing Sun, Lizhen Wang, Zhen Tian, Yuhua Hu, Ronghui Xia, Jiang Li
Journal of Oral and Maxillofacial Surgery.2019; 77(6): 1187. CrossRef - Mammary analogue secretory carcinoma of salivary gland diagnosed on submandibular gland cytology: A case report and review of the literature
Ethar Al‐Husseinawi, Soheila Hamidpour, Evanthia Omoscharka
Cytopathology.2019; 30(3): 318. CrossRef - Secretory Carcinoma of Minor Salivary Gland in Buccal Mucosa: A Case Report and Review of the Literature
Durga Paudel, Michiko Nishimura, Bhoj Raj Adhikari, Daichi Hiraki, Aya Onishi, Tetsuro Morikawa, Puja Neopane, Sarita Giri, Koki Yoshida, Jun Sato, Masayuki Ono, Yoshitaka Kamino, Hiroki Nagayasu, Yoshihiro Abiko
Case Reports in Pathology.2019; 2019: 1. CrossRef - Estrogen Receptor, Progesterone Receptor, and HER-2 Expression in Recurrent Pleomorphic Adenoma
Ana Amélia de Souza, Albina Altemani, Ney Soares de Araujo, Lucas Novaes Texeira, Vera Cavalcanti de Araújo, Andresa Borges Soares
Clinical Pathology.2019;[Epub] CrossRef - A Case of Secretory Carcinoma That Occurred in the Buccal Submucosa
Saito Yoshiro, Kurasawa Yuya, Moriya Takashi, Ikeda Kenitiro, Kushihashi Yukiomi, Egawa Shunya, Katsuta Hideyuki, Shimane Toshikazu
International Journal of Otolaryngology and Head & Neck Surgery.2019; 08(06): 191. CrossRef - Sinonasal Secretory Carcinoma of Salivary Gland with High Grade Transformation: A Case Report of this Under-Recognized Diagnostic Entity with Prognostic and Therapeutic Implications
Bin Xu, Ruth Aryeequaye, Lu Wang, Nora Katabi
Head and Neck Pathology.2018; 12(2): 274. CrossRef - Primary mammary analogue secretory carcinoma of the lung: a case report
Tao Huang, Jonathan B. McHugh, Gerald J. Berry, Jeffrey L. Myers
Human Pathology.2018; 74: 109. CrossRef - Salivary Secretory Carcinoma With a Novel ETV6-MET Fusion
Lisa M. Rooper, Theodoros Karantanos, Yi Ning, Justin A. Bishop, Sarah W. Gordon, Hyunseok Kang
American Journal of Surgical Pathology.2018; 42(8): 1121. CrossRef - Secretory carcinoma: The eastern Canadian experience and literature review
David Forner, Martin Bullock, Daniel Manders, Timothy Wallace, Christopher J. Chin, Liane B. Johnson, Matthew H. Rigby, Jonathan R. Trites, Mark S. Taylor, Robert D. Hart
Journal of Otolaryngology - Head & Neck Surgery.2018;[Epub] CrossRef - Newly described salivary gland tumors
Alena Skalova, Michal Michal, Roderick HW Simpson
Modern Pathology.2017; 30: S27. CrossRef - Diagnóstico, tratamiento y seguimiento de un tumor de reciente descripción: el carcinoma análogo secretor de mama (MASC) de glándula salival. A propósito de 2 nuevos casos
Marina Alexandra Gavín-Clavero, M. Victoria Simón-Sanz, Ana M. López-López, Alberto Valero-Torres, Esther Saura-Fillat
Revista Española de Cirugía Oral y Maxilofacial.2017; 39(4): 221. CrossRef - Systematic review of mammary analog secretory carcinoma of salivary glands at 7 years after description
Bacem A. Khalele
Head & Neck.2017; 39(6): 1243. CrossRef - Salivary Gland Secretory Carcinoma With High-Grade Transformation, CDKN2A/B Loss, Distant Metastasis, and Lack of Sustained Response to Crizotinib
Nicole A. Cipriani, Elizabeth A. Blair, Joshua Finkle, Jennifer L. Kraninger, Christopher M. Straus, Victoria M. Villaflor, Daniel Thomas Ginat
International Journal of Surgical Pathology.2017; 25(7): 613. CrossRef - A systematic review including an additional pediatric case report: Pediatric cases of mammary analogue secretory carcinoma
Amanda L. Ngouajio, Sarah M. Drejet, D. Ryan Phillips, Don-John Summerlin, John P. Dahl
International Journal of Pediatric Otorhinolaryngology.2017; 100: 187. CrossRef - Newly Described Entities in Salivary Gland Pathology
Alena Skálová, Douglas R. Gnepp, James S. Lewis, Jennifer L. Hunt, Justin A. Bishop, Henrik Hellquist, Alessandra Rinaldo, Vincent Vander Poorten, Alfio Ferlito
American Journal of Surgical Pathology.2017; 41(8): e33. CrossRef - Mammary Analogue Secretory Carcinoma of Salivary Glands: Diagnostic Pitfall with Distinct Immunohistochemical Profile and Molecular Features
Oliver Bissinger, Carolin Götz, Andreas Kolk, Henning A. Bier, Abbas Agaimy, Henning Frenzel, Sven Perner, Julika Ribbat-Idel, Klaus Dietrich Wolff, Wilko Weichert, Caroline Mogler
Rare Tumors.2017; 9(3): 89. CrossRef - Mammary Analogue Secretory Carcinoma of Salivary Gland. A Case Report Emphasizing its Diagnostic Histological, Immunohistochemistry and Molecular Findings
Monalisa Hui, Shantveer G Uppin, Vamshi Krishna Thamtam, Abhiram Kalle
International Journal of Head and Neck Surgery.2017; 8(4): 160. CrossRef - A case of mammary analog secretory carcinoma of the lower lip
Takako Aizawa, Taro Okui, Ken Kitagawa, Yoshikazu Kobayashi, Koji Satoh, Hideki Mizutani
Journal of Oral and Maxillofacial Surgery, Medicine, and Pathology.2016; 28(3): 277. CrossRef - Mammary analogue secretory carcinoma of parotid: Is preoperative cytological diagnosis possible?
Nikita Oza, Kintan Sanghvi, Tanuja Shet, Asawari Patil, Santosh Menon, Mukta Ramadwar, Shubhada Kane
Diagnostic Cytopathology.2016; 44(6): 519. CrossRef - Lysozyme Expression Can be Useful to Distinguish Mammary Analog Secretory Carcinoma from Acinic Cell Carcinoma of Salivary Glands
Fernanda Viviane Mariano, Camila Andrea Concha Gómez, Juliana de Souza do Nascimento, Harim Tavares dos Santos, Erika Said Egal, Victor Angelo Martins Montalli, Pablo Agustin Vargas, Oslei Paes de Almeida, Albina Altemani
Head and Neck Pathology.2016; 10(4): 429. CrossRef - Cytogenetic and immunohistochemical characterization of mammary analogue secretory carcinoma of salivary glands
Syed A. Khurram, Jemel Sultan-Khan, Neil Atkey, Paul M. Speight
Oral Surgery, Oral Medicine, Oral Pathology and Oral Radiology.2016; 122(6): 731. CrossRef - The role of DOG1 immunohistochemistry in dermatopathology
Keisuke Goto
Journal of Cutaneous Pathology.2016; 43(11): 974. CrossRef - Extended immunologic and genetic lineage of mammary analogue secretory carcinoma of salivary glands
Hao Ni, Xue-ping Zhang, Xiao-tong Wang, Qiu-yuan Xia, Jing-huan Lv, Xuan Wang, Shan-shan Shi, Rui Li, Xiao-jun Zhou, Qiu Rao
Human Pathology.2016; 58: 97. CrossRef - A New Hitherto Unreported Histopathologic Manifestation of Mammary Analogue Secretory Carcinoma: “Masked MASC” Associated With Low-grade Mucinous Adenocarcinoma and Low-grade In Situ Carcinoma Components
Fredrik Petersson, Michael Michal, Dmitry V. Kazakov, Petr Grossmann, Michal Michal
Applied Immunohistochemistry & Molecular Morphology.2016; 24(9): e80. CrossRef - Papillary-cystic pattern is characteristic in mammary analogue secretory carcinomas but is rarely observed in acinic cell carcinomas of the salivary gland
Min-Shu Hsieh, Yueh-Hung Chou, Shin-Joe Yeh, Yih-Leong Chang
Virchows Archiv.2015; 467(2): 145. CrossRef - Mammary analog secretory carcinoma, low-grade salivary duct carcinoma, and mimickers: a comparative study
Todd M Stevens, Andra O Kovalovsky, Claudia Velosa, Qiuying Shi, Qian Dai, Randall P Owen, Walter C Bell, Shi Wei, Pamela A Althof, Jennifer N Sanmann, Larissa Sweeny, William R Carroll, Gene P Siegal, Martin J Bullock, Margaret Brandwein-Gensler
Modern Pathology.2015; 28(8): 1084. CrossRef - Aspiration cytology of mammary analogue secretory carcinoma of the salivary gland
Min Jung Jung, Sang Yoon Kim, Soon Yuhl Nam, Jong‐Lyel Roh, Seung‐Ho Choi, Jeong Hyun Lee, Jung Hwan Baek, Kyung‐Ja Cho
Diagnostic Cytopathology.2015; 43(4): 287. CrossRef - A Unique Case of a Cutaneous Lesion Resembling Mammary Analog Secretory Carcinoma
Jennifer Albus, Jacqueline Batanian, Bruce M. Wenig, Claudia I. Vidal
The American Journal of Dermatopathology.2015; 37(4): e41. CrossRef - Mammary analogue secretory carcinoma of salivary glands: a new entity associated with ETV6 gene rearrangement
Hanna Majewska, Alena Skálová, Dominik Stodulski, Adéla Klimková, Petr Steiner, Czesław Stankiewicz, Wojciech Biernat
Virchows Archiv.2015; 466(3): 245. CrossRef - A comparative immunohistochemistry study of diagnostic tools in salivary gland tumors: usefulness of mammaglobin, gross cystic disease fluid protein 15, and p63 cytoplasmic staining for the diagnosis of mammary analog secretory carcinoma?
Fabrice Projetti, Magali Lacroix‐Triki, Elie Serrano, Sebastien Vergez, Béatrice Herbault Barres, Julie Meilleroux, Marie‐Bernadette Delisle, Emmanuelle Uro‐Coste
Journal of Oral Pathology & Medicine.2015; 44(4): 244. CrossRef - Mammary Analogue Secretory Carcinoma of Salivary Glands
Yohei Ito, Kenichiro Ishibashi, Ayako Masaki, Kana Fujii, Yukio Fujiyoshi, Hideo Hattori, Daisuke Kawakita, Manabu Matsumoto, Satoru Miyabe, Kazuo Shimozato, Toshitaka Nagao, Hiroshi Inagaki
American Journal of Surgical Pathology.2015; 39(5): 602. CrossRef - Cytopathological features of mammary analogue secretory carcinoma—Review of literature
Maiko Takeda, Takahiko Kasai, Kohei Morita, Mao Takeuchi, Takeshi Nishikawa, Akinori Yamashita, Shinji Mikami, Hiroshi Hosoi, Chiho Ohbayashi
Diagnostic Cytopathology.2015; 43(2): 131. CrossRef - Mammary Analog Secretory Carcinoma of Salivary Glands
Justin A. Bishop
Pathology Case Reviews.2015; 20(1): 7. CrossRef - Diagnostic utility of phosphorylated signal transducer and activator of transcription 5 immunostaining in the diagnosis of mammary analogue secretory carcinoma of the salivary gland: A comparative study of salivary gland cancers
Akihiko Kawahara, Tomoki Taira, Hideyuki Abe, Yorihiko Takase, Takashi Kurita, Eiji Sadashima, Satoshi Hattori, Ichio Imamura, Shinji Matsumoto, Hitomi Fujisaki, Kazunobu Sueyoshi, Jun Akiba, Masayoshi Kage
Cancer Cytopathology.2015; 123(10): 603. CrossRef - A Case of Mammary Analogue Secretory Carcinoma Arising from Parotid Gland
Hee Tae Kim, Cha Hee Lee, Han Su Kim, Hae Sang Park
Korean Journal of Otorhinolaryngology-Head and Neck Surgery.2015; 58(8): 563. CrossRef - WITHDRAWN: A biphasic sessile mass of the buccal mucosa
Tiffany M. Peters, Jose P. Zevallos, Brent A. Golden, Alice E. Curran
Oral Surgery, Oral Medicine, Oral Pathology and Oral Radiology.2015;[Epub] CrossRef - Mammary analogue secretory carcinoma: an evaluation of its clinicopathological and genetic characteristics
Peter P. Luk, Christina I. Selinger, Timothy J. Eviston, Trina Lum, Bing Yu, Sandra A. O’Toole, Jonathan R. Clark, Ruta Gupta
Pathology.2015; 47(7): 659. CrossRef - A case of mammary analogue secretory carcinoma arising in the submandibular region
Kotaro ISHII, Koji NAKAMATSU, Koji SATO, Chikashi MINEMURA, Wataru KUMAMARU, Hiroyo YOSHIKAWA
Japanese Journal of Oral and Maxillofacial Surgery.2015; 61(11): 564. CrossRef - DOG1, p63, and S100 protein: a novel immunohistochemical panel in the differential diagnosis of oncocytic salivary gland neoplasms in fine-needle aspiration cell blocks
Alessandra C. Schmitt, Ryan McCormick, Cynthia Cohen, Momin T. Siddiqui
Journal of the American Society of Cytopathology.2014; 3(6): 303. CrossRef - Salivary Gland Tumor “Wishes” to Add to the Next WHO Tumor Classification: Sclerosing Polycystic Adenosis, Mammary Analogue Secretory Carcinoma, Cribriform Adenocarcinoma of the Tongue and Other Sites, and Mucinous Variant of Myoepithelioma
Douglas R. Gnepp
Head and Neck Pathology.2014; 8(1): 42. CrossRef - Hepatoid differentiation in renal cell carcinoma: a rare histologic pattern with clinical significance
Jungweon Shim, Heounjeong Go, Young-Suk Lim, Kyung Chul Moon, Jae Y. Ro, Yong Mee Cho
Annals of Diagnostic Pathology.2014; 18(6): 363. CrossRef - Mammary analogue secretory carcinoma: Update on a new diagnosis of salivary gland malignancy
Roshan Sethi, Elliott Kozin, Aaron Remenschneider, Josh Meier, Paul VanderLaan, William Faquin, Daniel Deschler, Robert Frankenthaler
The Laryngoscope.2014; 124(1): 188. CrossRef - Mammary analog secretory carcinoma of salivary gland in a 5 year old: Case report
Matthew Keisling, Michael Bianchi, Judy Mae Pascasio
International Journal of Pediatric Otorhinolaryngology Extra.2014; 9(4): 163. CrossRef - Fine-Needle Aspiration Cytology of Mammary Analog Secretory Carcinoma Masquerading as Low-Grade Mucoepidermoid Carcinoma: Case Report with a Review of the Literature
Jaya Bajaj, Cecilia Gimenez, Farah Slim, Mohamed Aziz, Kasturi Das
Acta Cytologica.2014; 58(5): 501. CrossRef
- Rare Tumors of Oral Cavity: A Case Report and Literature Review on Secretory Carcinoma of Minor Salivary Glands
- Diagnostic Difficulties in Fine Needle Aspiration of Benign Salivary Glandular Lesions
- Hye Jung Jo, Hyo Jung Ahn, Soojin Jung, Hye-Kyoung Yoon
- Korean J Pathol. 2012;46(6):569-575. Published online December 26, 2012
- DOI: https://doi.org/10.4132/KoreanJPathol.2012.46.6.569
- 10,198 View
- 62 Download
- 12 Crossref
-
 Abstract
Abstract
 PDF
PDF Background The diagnostic accuracy of fine needle aspiration cytology (FNAC) of salivary lesions is relatively high, but cytologic interpretation might be confusing if the sample is lacking typical cytologic features.
Methods There were 77 cases of benign salivary lesions, consisting of pleomorphic adenoma (PA) in 61 cases, Warthin's tumor (WT) in 12 cases, and other benign lesions in 4 cases. The causes of the discrepancies between the FNAC and the histologic diagnoses were evaluated.
Results Major discrepancies were noted in 4 of the 61 PA cases, and in 1 of 12 WT cases. The causes of the major discrepancies were a mislabeled site in 1 PA and 1 WT case, and an interpretation error in 3 PA cases. Minor discrepancies were more common in the WT cases (7 of 12 cases) than in the PA cases (11 of 61 cases). The causes of the minor discrepancies were a mislabeled site in 1 PA and 1 WT case, an inadequate sample in 7 PA and 2 WT cases, a lack of typical cytomorphology in 2 PA and 2 WT cases, and an interpretation error in 1 PA and 2 WT cases.
Conclusions To increase the diagnostic accuracy in the benign salivary lesions, recognition of both characteristic and less typical cytomorphology is needed.
-
Citations
Citations to this article as recorded by- Deep Learning Based on Ultrasound Images Differentiates Parotid Gland Pleomorphic Adenomas and Warthin Tumors
Yajuan Li, Mingchi Zou, Xiaogang Zhou, Xia Long, Xue Liu, Yanfeng Yao
Ultrasonic Imaging.2025; 47(3-4): 107. CrossRef - Clinical decision making when cytology indicates a Warthin tumor
Minna Sirviö, Katri Aro, Mira Naukkarinen, Antti Mäkitie, Jussi Tarkkanen, Jetta Kelppe, Timo Atula
Scientific Reports.2024;[Epub] CrossRef - Different MRI-based radiomics models for differentiating misdiagnosed or ambiguous pleomorphic adenoma and Warthin tumor of the parotid gland: a multicenter study
Jing Yang, Qiu Bi, Yiren Jin, Yong Yang, Ji Du, Hongjiang Zhang, Kunhua Wu
Frontiers in Oncology.2024;[Epub] CrossRef - Diagnostic Accuracy of Fine Needle Aspiration Cytology (FNAC) in Salivary Gland Lesions with Histopathological Examination (HPE) Correlation in a Tertiary Care Centre in Southern India
Banumathi Kasinathan, Babu Manohar, H. Ganapathy
Indian Journal of Otolaryngology and Head & Neck Surgery.2023; 75(2): 871. CrossRef - Diagnostic performance of qualitative and radiomics approach to parotid gland tumors: which is the added benefit of texture analysis?
Federica Vernuccio, Federica Arnone, Roberto Cannella, Barbara Verro, Albert Comelli, Francesco Agnello, Alessandro Stefano, Rosalia Gargano, Vito Rodolico, Giuseppe Salvaggio, Roberto Lagalla, Massimo Midiri, Antonio Lo Casto
The British Journal of Radiology.2021;[Epub] CrossRef - Improving the diagnosis of common parotid tumors via the combination of CT image biomarkers and clinical parameters
Dan Zhang, Xiaojiao Li, Liang Lv, Jiayi Yu, Chao Yang, Hua Xiong, Ruikun Liao, Bi Zhou, Xianlong Huang, Xiaoshuang Liu, Zhuoyue Tang
BMC Medical Imaging.2020;[Epub] CrossRef Evaluation of Salivary Gland Lesions by Fine Needle Aspiration Cytology at a Tertiary Care Hospital, Western Nepal
Anuj Poudel, Bigya Shrestha, Sudeep Regmi
Pathology and Laboratory Medicine International.2020; Volume 12: 9. CrossRef- Ultrasound-guided fine-needle capillary cytology of parotid gland masses coupled with a rapid-on-site evaluation improves results
R. Barats, S. Evrard, L. Collin, S. Vergez, S. Gellée, M. Courtade-Saïdi
Morphologie.2018; 102(336): 25. CrossRef - The Value of Ultrasound-Guided Fine-Needle Aspiration Cytology by Cytopathologists in the Diagnosis of Major Salivary Gland Tumors
Shahrzad Negahban, Sadegh Shirian, Bijan Khademi, Ahmad Oryan, Roshanak Sadoughifar, Mohammadian-Panah Mohammad, Azita Aledavood, Khosrow Daneshbod, Yayha Daneshbod
Journal of Diagnostic Medical Sonography.2016; 32(2): 92. CrossRef - Salivary Gland Tumors: A Diagnostic Dilemma!
Ranjit Kumar Peravali, H. Hari Kishore Bhat, Varsha H. Upadya, Anmol Agarwal, Sushma Naag
Journal of Maxillofacial and Oral Surgery.2015; 14(S1): 438. CrossRef - Diagnostic problems of salivary gland tumors
Ruchita Tyagi, Pranab Dey
Diagnostic Cytopathology.2015; 43(6): 495. CrossRef - Pleomorphic Adenoma of the Accessory Parotid Gland: Case Report and Reappraisal of Intraoral Extracapsular Dissection for Management
Tibebu M. Tsegga, Jennifer D. Britt, Aragon R. Ellwanger
Journal of Oral and Maxillofacial Surgery.2015; 73(3): 564. CrossRef
- Deep Learning Based on Ultrasound Images Differentiates Parotid Gland Pleomorphic Adenomas and Warthin Tumors
- Primary Thymic Mucinous Adenocarcinoma: A Case Report
- Jamshid Abdul-Ghafar, Suk-Joong Yong, Woocheol Kwon, Il Hwan Park, Soon-Hee Jung
- Korean J Pathol. 2012;46(4):377-381. Published online August 23, 2012
- DOI: https://doi.org/10.4132/KoreanJPathol.2012.46.4.377
- 10,602 View
- 83 Download
- 21 Crossref
-
 Abstract
Abstract
 PDF
PDF Primary thymic mucinous adenocarcinoma is an extremely rare aggressive subtype of thymic carcinoma. With a review of literatures, only nine cases have been reported up to present. A 36-year-old woman was admitted for further evaluation and treatment of a mediastinal mass. The patient had no medical history of cancer. The clinicoradiological examination disclosed no tumor elsewhere. After the surgical excision of mediastinal mass, it was grossly a round semi-solid mass with mucin-filled cystic areas. Microscopically solid areas showed cords, small nests and dilated glands infiltrating the fibrotic parenchyma, while the cystic areas were lined by mucinous epithelium with tumor cells floating in extracellular-mucin pools. Some cystic walls underwent malignant transformation of the benign thymic epithelium. Immunohistochemically, the tumor cells were positive for cytokeratin (CK) 7, CK20, CD5, and CDX-2, and negative for thyroid transcription factor-1. In conclusion, the mucinous thymic adenocarcinoma should be recognized as a separate histopathological entity and considered in the differential diagnosis of mediastinal carcinomas.
-
Citations
Citations to this article as recorded by- Unresectable Primary Enteric‐Type Thymic Adenocarcinoma Treated With FOLFOX Chemotherapy: A Case Report
Carl He, Georgia Bentick, Patrick Hosking, Andrew Mant
Cancer Reports.2025;[Epub] CrossRef - Enteric thymic adenocarcinoma: Understanding a unique pathology in the mediastinum
Raja Chhabra, Kartik Mittal, Anmol Tufchi, Sajjan Rajpurohit, Deepak Kumar Mittal, Aditya Vidushi, Swati Saxena, Md Ali Osama
Indian Journal of Pathology and Microbiology.2025; 68(4): 820. CrossRef - Primary mucinous adenocarcinoma of the thymus
Kohei Soejima, Hidehito Matsuoka
The Journal of the Japanese Association for Chest Surgery.2024; 38(6): 545. CrossRef - Stage IV thymic mucinous adenocarcinoma under long-term disease control after primary tumor resection: A case report
Chihaya Maeda, Tomoyuki Hishida, Kyohei Masai, Keisuke Asakura, Katsura Emoto, Hisao Asamura
The Journal of the Japanese Association for Chest Surgery.2022; 36(2): 156. CrossRef - Lenvatinib-refractory thymic mucinous carcinoma with PIK3CA mutation
Akihiro Tsukaguchi, Shoichi Ihara, Hironao Yasuoka, Seigo Minami
International Cancer Conference Journal.2022; 12(1): 36. CrossRef - Thymic mucinous adenocarcinoma: A case report
Hideki Tsubouchi, Naoki Ozeki, Yuka Suzuki, Koji Kawaguchi, Takayuki Fukui, Toyofumi F. Chen-Yoshikawa
The Journal of the Japanese Association for Chest Surgery.2021; 35(5): 547. CrossRef - Metastatic thymic-enteric adenocarcinoma responding to chemoradiation plus anti-angiogenic therapy: A case report
Man Li, Xiao-Yu Pu, Li-Hua Dong, Peng-Yu Chang
World Journal of Clinical Cases.2021; 9(7): 1676. CrossRef - Primary Mucinous Adenocarcinoma of the Thymus: a Rare Type of Thymic Carcinoma—Case Report
Koichi Tomoshige, Tomoshi Tsuchiya, Keitaro Matsumoto, Takuro Miyazaki, Ryoichiro Doi, Ryusuke Machino, Satoshi Mizoguchi, Takamune Matumoto, Yutaka Maeda, Takeshi Nagayasu
SN Comprehensive Clinical Medicine.2021; 3(5): 1233. CrossRef - Primary mucinous adenocarcinoma of the thymus: A case report
Tomoka Hamahiro, Ryo Maeda, Takanori Ayabe, Yuichiro Sato, Masaki Tomita
Respiratory Medicine Case Reports.2021; 34: 101497. CrossRef - Primary Thymic Mucinous Adenocarcinoma: A Case Report Focusing on Radiological Findings and Review of the Literature
Young Hoon Koo, Jae Wook Lee, Jai Soung Park, Kyung Eun Shin, Heon Lee, Susie Chin
Iranian Journal of Radiology.2020;[Epub] CrossRef - Mucinous adenocarcinoma of the thymus: report of a case
Fumihiko Kinoshita, Fumihiro Shoji, Kazuki Takada, Gouji Toyokawa, Tatsuro Okamoto, Tokujiro Yano, Yoshinao Oda, Yoshihiko Maehara
General Thoracic and Cardiovascular Surgery.2018; 66(2): 111. CrossRef - Thymic enteric type adenocarcinoma: A case report with cytological features
Marie Tamai, Mitsuaki Ishida, Yusuke Ebisu, Hisashi Okamoto, Chika Miyasaka, Chisato Ohe, Yoshiko Uemura, Tomohito Saito, Tomohiro Murakawa, Koji Tsuta
Diagnostic Cytopathology.2018; 46(1): 92. CrossRef - Primary thymic adenocarcinomas: a clinicopathological and immunohistochemical study of 16 cases with emphasis on the morphological spectrum of differentiation
Neda Kalhor, Cesar A. Moran
Human Pathology.2018; 74: 73. CrossRef - Histologic characteristics of thymic adenocarcinomas: Clinicopathologic study of a nine-case series and a review of the literature
Ah-Young Kwon, Joungho Han, Jinah Chu, Yong Soo Choi, Byeong-Ho Jeong, Myung-Ju Ahn, Yong Chan Ahn
Pathology - Research and Practice.2017; 213(2): 106. CrossRef - Cytologic Characteristics of Thymic Adenocarcinoma with Enteric Differentiation: A Study of Four Fine-Needle Aspiration Specimens
Ah-Young Kwon, Joungho Han, Hae-yon Cho, Seokhwi Kim, Heejin Bang, Jiyeon Hyeon
Journal of Pathology and Translational Medicine.2017; 51(5): 509. CrossRef - Mucinous cystic tumor with CK20 and CDX2 expression of the thymus: Is this a benign counterpart of adenocarcinoma of the thymus, enteric type?
Jun Akiba, Hiroshi Harada, Shintaro Yokoyama, Toshihiro Hashiguchi, Akihiko Kawahara, Masahiro Mitsuoka, Shinzo Takamori, Hirohisa Yano
Pathology International.2016; 66(1): 29. CrossRef - Colon cancer chemotherapy for a patient with CDX2-expressing metastatic thymic adenocarcinoma: a case report and literature review
Akihiko Sawaki, Mikiya Ishihara, Yuji Kozuka, Hiroyasu Oda, Satoshi Tamaru, Yumiko Sugawara, Yoshiki Yamashita, Toshiro Mizuno, Taizo Shiraishi, Naoyuki Katayama
International Cancer Conference Journal.2016; 5(2): 113. CrossRef - Adenocarcinoma of the Thymus, Enteric Type
Bernhard Moser, Ana Iris Schiefer, Stefan Janik, Alexander Marx, Helmut Prosch, Wolfgang Pohl, Barbara Neudert, Anke Scharrer, Walter Klepetko, Leonhard Müllauer
American Journal of Surgical Pathology.2015; 39(4): 541. CrossRef - A Rare Case of Primary Thymic Adenocarcinoma Mimicking Small Cell Lung Cancer
Eun Na Cho, Hye Sung Park, Tae Hoon Kim, Min Kwang Byun, Hyung Jung Kim, Chul Min Ahn, Yoon Soo Chang
Tuberculosis and Respiratory Diseases.2015; 78(2): 112. CrossRef - A Rare Case of Primary Tubular Adenocarcinoma of the Thymus, Enteric Immunophenotype: A Case Study and Review of the Literature
Hae Yoen Jung, Hyundeuk Cho, Jin-Haeng Chung, Sang Byoung Bae, Ji-Hye Lee, Hyun Ju Lee, Si-Hyong Jang, Mee-Hye Oh
Journal of Pathology and Translational Medicine.2015; 49(4): 331. CrossRef - Primary mucinous adenocarcinoma of the thymus: a case report
Tadashi Sakane, Kotaro Mizuno, Risa Oda, Takuya Matsui, Makoto Ito, Takeshi Yamada
The Journal of the Japanese Association for Chest Surgery.2014; 28(7): 904. CrossRef
- Unresectable Primary Enteric‐Type Thymic Adenocarcinoma Treated With FOLFOX Chemotherapy: A Case Report
- Cytologic Findings of Thyroid Carcinoma Showing Thymus-like Differentiation: A Case Report
- Sunhee Chang, Mee Joo, Hanseong Kim
- Korean J Pathol. 2012;46(3):302-305. Published online June 22, 2012
- DOI: https://doi.org/10.4132/KoreanJPathol.2012.46.3.302
- 12,698 View
- 85 Download
- 16 Crossref
-
 Abstract
Abstract
 PDF
PDF Carcinoma showing thymus-like differentiation (CASTLE) is a rare carcinoma of the thyroid or adjacent soft tissue of the neck with a histologic resemblance to thymic epithelial tumors. Although the fine-needle aspiration (FNA) plays a central role in the initial evaluation of thyroid nodules, few reports about the cytologic findings of CASTLE have been found according to a review of literatures. We report cytologic findings of a case of CASTLE. A 34-year-old woman presented with a 2-month history of sore throat. The FNA showed that the smear was composed of three dimensional clusters and sheets. The tumor cells were round to ovoid with high nuclear : cytoplasmic ratios. The nuclei were vesicular with small nucleoli. There were some tumor cells showing keratinization. Some lymphocytes were found on the background and within clusters. The presence of poorly-differentiated tumor cells with a focal keratinization and a lymphocytic background on the FNA is suggestive of CASTLE.
-
Citations
Citations to this article as recorded by- Fine‐Needle Aspiration Cytologic Diagnosis of Intrathyroidal Thymic Carcinoma: A Review of the Literature
Qi Chen, Xiaoli Yu
Diagnostic Cytopathology.2025; 53(10): 507. CrossRef - Preoperative Cytological Diagnosis of Intrathyroidal Thymic Carcinoma: A Case Report With Review of the Literature
Makoto Yamada, Shota Sezaki, Tomonari Ikeya, Masatoshi Furuta, Shogo Mizuno, Yoshiro Otsuki
Diagnostic Cytopathology.2025;[Epub] CrossRef - Coexistence of intrathyroid thymic carcinoma and papillary thyroid carcinoma: a case report and literature review
Maryam Vajihinejad, Ali Ataei, Mohammad Pashmchi, Ali Aledavoud, Vahid Zand, Mohammad Ali Broomand, Mohammad Mohammadi, Niloofar Zare Reshkuiyeh
Frontiers in Oncology.2024;[Epub] CrossRef - Intrathyroidal Thymic Carcinoma: A Retrospective Case Series Study
Jinhui Liang, Mei Huang, Helang Huang, Li Li, Hailin Luo, Weidong Mao, Shan Gao, Haoxiang Xu
Ear, Nose & Throat Journal.2023; 102(9): 584. CrossRef - Carcinoma showing thymus‐like differentiation of the parotid gland: The brief report of cytomorphology and review of the literature
Tomoko Uchiyama, Chiyoko Terada, Yuma Tachibana, Hirokazu Nishiura, Maiko Takeda, Tomomi Fujii, Takahiro Kimura, Masahiro Tsutsumi, Chiho Ohbayashi
Diagnostic Cytopathology.2023;[Epub] CrossRef - A case of concurrent occurrence of carcinoma showing thymus-like differentiation and follicular variant of papillary thyroid cancer in the same thyroid
Takahito Kimura, Keisuke Enomoto, Masamitsu Kono, Masanobu Hiraoka, Saori Takeda, Naoko Kumashiro, Shun Hirayama, Eri Kimura, Shunji Tamagawa, Makiko Ohtani, Shin-Ichi Murata, Muneki Hotomi
Journal of Surgical Case Reports.2022;[Epub] CrossRef - Ultrasonographic Features of Intrathyroidal Thymic Carcinoma: Review and Analysis of 10 Cases
Wang, MD Yanhai, Yang, MD Hua, Liu, MD Hanqing, Luo, MD Xiaoli, Liu, BS Luying, Zhou, BS Pingting
ADVANCED ULTRASOUND IN DIAGNOSIS AND THERAPY.2022; 6(2): 58. CrossRef - Thyroid carcinoma with thymus-like differentiation (CASTLE) tumor: а сase report
A. A. Ilyin, V. V. Polkin, P. A. Isaev, F. E. Sevrukov, N. Yu. Dvinskych, M. I. Ryzhenkova, S. A. Ivanov, A. D. Kaprin
Head and neck tumors (HNT).2021; 11(2): 64. CrossRef - Metastatic Renal Cell Neoplasm Within a Papillary Thyroid
Carcinoma as Incidental Finding in an Asymptomatic Patient: a Case Report
Maria-Rosa Bella-Cueto, Mireia Pascua-Solé, Albert Cano-Palomares, M. Àngels Cabezuelo-Hernandez, Maria-Rosa Escoda-Giralt, Santiago Barcons-Vilaplana, Paula Serret-Miralles, Carmen Caral-Vanaclocha, Xavier Guirao-Garriga, Joan Prats-Lopez, Meritxell Meda
SN Comprehensive Clinical Medicine.2020; 2(7): 978. CrossRef - Intrathyroidal thymic carcinoma exhibiting neuroendocrine differentiation: Case report with cytomorphology, immunocytochemistry, and review of the literature focusing on cytology
Wen‐hao Ren, Kun Dong, Xiao‐zheng Huang, Yan‐li Zhu
Diagnostic Cytopathology.2019; 47(11): 1197. CrossRef - Management and Prognostic Factors for Thyroid Carcinoma Showing Thymus-Like Elements (CASTLE): A Case Series Study
Rui Gao, Xi Jia, Ting Ji, Jinteng Feng, Aimin Yang, Guangjian Zhang
Frontiers in Oncology.2018;[Epub] CrossRef - Multiple squamous cells in thyroid fine needle aspiration: Friends or foes?
Heather Gage, Elizabeth Hubbard, Laurentia Nodit
Diagnostic Cytopathology.2016; 44(8): 676. CrossRef - Clinical analysis of 82 cases of carcinoma showing thymus-like differentiation of the thyroid
WEI GE, YONG-ZHONG YAO, GANG CHEN, YI-TAO DING
Oncology Letters.2016; 11(2): 1321. CrossRef - Carcinoma Showing Thymus-Like Differentiation (CASTLE): Cytopathological Features and Differential Diagnosis
Jennifer A. Collins, Bo Ping, Justin A. Bishop, Syed Z. Ali
Acta Cytologica.2016; 60(5): 421. CrossRef - Carcinoma Showing Thymus-Like Elements of the Thyroid Gland: Report of Three Cases Including One Case with Breast Cancer History
Guanjun Zhang, Xi Liu, Wei Huang, Xiaofeng Li, Marianne Johnstone, Yuan Deng, Yongqiang Ke, Quentin M. Nunes, Hongyan Wang, Yili Wang, Xuebin Zhang
Pathology & Oncology Research.2015; 21(1): 45. CrossRef - Potential Role of Adjuvant Radiation Therapy in Cervical Thymic Neoplasm Involving Thyroid Gland or Neck
Jae Myoung Noh, Sang Yun Ha, Yong Chan Ahn, Dongryul Oh, Seung Won Seol, Young Lyun Oh, Joungho Han
Cancer Research and Treatment.2014; 47(3): 436. CrossRef
- Fine‐Needle Aspiration Cytologic Diagnosis of Intrathyroidal Thymic Carcinoma: A Review of the Literature
- Osteoclast-like Giant Cell Tumor of Parotid Gland with a Carcinomatous Component: A Case Report
- Jung Wook Yang, Hyeon Cheol Kim, Jeong Hee Lee, Jong Sil Lee, Dong Chul Kim, Dae Hyun Song, Jin Pyeong Kim, Gyung Hyuck Ko
- Korean J Pathol. 2012;46(3):297-301. Published online June 22, 2012
- DOI: https://doi.org/10.4132/KoreanJPathol.2012.46.3.297
- 9,925 View
- 92 Download
- 6 Crossref
-
 Abstract
Abstract
 PDF
PDF The giant cell tumor of the salivary gland is very rare, and 20 cases have been reported in the English-language literature. We report an additional case. A 57-year old man had noticed a mass in the right parotid area for several weeks. The diagnosis using aspiration cytology was a giant cell tumor possibly with a carcinomatous component. Superficial parotidectomy was carried out. The resected parotid gland contained a 1.8 cm-sized well-circumscribed brownish tumor. Histologically the tumor consisted of evenly distributed osteoclast-like giant cells, mononuclear cells and two small foci of a carcinomatous component. The osteoclast-like giant cells and mononuclear cells were positive for vimentin and CD68, and the carcinomatous component was positive for cytokeratin and epithelial membrane antigen. There was no metastatic lesion in the cervical lymph nodes. We believe this is the first case in Korea of an osteoclast-like giant cell tumor of the parotid gland.
-
Citations
Citations to this article as recorded by- Giant cell tumour of the parotid gland: a rare, unusual entity
Erna Ahsan, Kranthi Kumar Jandrasupalli, Prashant Durgapal, Divya Yadav
BMJ Case Reports.2025; 18(4): e261751. CrossRef - Genomic alteration in rare subtype of sarcomatoid salivary duct carcinoma
Ji-Seon Jeong, Kyung-Ja Cho, Deokhoon Kim, Yoon Se Lee, Joon Seon Song
Pathology - Research and Practice.2021; 228: 153678. CrossRef - Giant cell tumor of temporomandibular joint presenting as a parotid tumor: Challenges in the accurate subclassification of giant cell tumors in an unusual location
Rongqin Ren, Sandra Mueller, Adele O. Kraft, Celeste N. Powers
Diagnostic Cytopathology.2018; 46(4): 340. CrossRef - Osteoclast‐Like Giant Cell Tumor of the Parotid Gland: Report of a Case Diagnosed on Fine‐Needle Aspiration Cytology With Histological and Immunohistochemical Findings
Poonam Elhence, Meenakshi Rao, Amit Goyal, Amit Kumar, Pushpinder S. Khera, Shilajit Bhattacharya
Diagnostic Cytopathology.2016; 44(6): 548. CrossRef - Giant cell tumour of a temporomandibular joint presenting as a parotid mass: Case report and analysis of the 19 cases in the literature
Yun-Chen Huang, Jeng-Wen Chen, Yen-Lin Chen, Pei-Jen Lou
Journal of Cranio-Maxillofacial Surgery.2014; 42(8): 1778. CrossRef - Tumeur à cellules géantes de type ostéoclastique de la parotide
S. Rammeh, I. Hergli, M.K. M’farrej, N. Znaidi, S. Nechi, R. Zermani
Revue de Stomatologie, de Chirurgie Maxillo-faciale et de Chirurgie Orale.2014; 115(3): 185. CrossRef
- Giant cell tumour of the parotid gland: a rare, unusual entity
- Gastric Adenocarcinoma of Fundic Gland Type: Report of Three Cases
- Eun Su Park, Young Eun Kim, Cheol Keun Park, Takashi Yao, Ryoji Kushima, Kyoung-Mee Kim
- Korean J Pathol. 2012;46(3):287-291. Published online June 22, 2012
- DOI: https://doi.org/10.4132/KoreanJPathol.2012.46.3.287
- 13,229 View
- 117 Download
- 25 Crossref
-
 Abstract
Abstract
 PDF
PDF Recently, fundic gland type gastric adenocarcinoma (GA-FG) has been reported as a new entity. This report describes GA-FG among Koreans for the first time. From March 2008 to July 2010 we identified only three cases of GA-FG out of over 6,000 GAs resected by endoscopy or surgery. Cell differentiation by mucin proteins, pepsinogen-I, and H+/K+-ATPase was evaluated. All three cases were male patients and diagnosed as early stage GA. Histologically, GA-FGs were well-differentiated adenocarcinoma with pale gray-blue, basophilic columnar or cuboidal cells and mildly enlarged nuclei, resembling chief cells. All three cases were positive for pepsinogen-I and were classified as gastric mucin phenotype. Among three histologic subtypes of GA-FG, since tumors were mainly composed of chief cells, our three cases were classified as chief cell predominant type. In conclusion, GA-FG is very rare among Koreans and pepsinogen-I and MUC6 expression are typical immunohistochemical findings in GA-FG suggesting differentiation toward fundic glands.
-
Citations
Citations to this article as recorded by- Endoscopic Submucosal Dissection of Early Gastric Adenocarcinoma of Fundic Gland Type: A Case Report
Ming Zhong, Wei Wei, Huang Zhong, Hang Gong, Tingyu Wang
Revista Española de Enfermedades Digestivas.2025;[Epub] CrossRef - Oxyntic Gland Neoplasms - From Adenoma to Advanced Gastric Cancer: A Review of 29 Cases
Gi Hwan Kim, Jun Su Lee, Jeong Hoon Lee, Young Soo Park
Journal of Gastric Cancer.2024; 24(4): 378. CrossRef - Transcriptome analysis reveals the essential role of NK2 homeobox 1/thyroid transcription factor 1 (NKX2-1/TTF-1) in gastric adenocarcinoma of fundic-gland type
Kazushi Fukagawa, Yu Takahashi, Nobutake Yamamichi, Natsuko Kageyama-Yahara, Yoshiki Sakaguchi, Miho Obata, Rina Cho, Nobuyuki Sakuma, Sayaka Nagao, Yuko Miura, Naoki Tamura, Daisuke Ohki, Hiroya Mizutani, Seiichi Yakabi, Chihiro Minatsuki, Keiko Niimi, Y
Gastric Cancer.2023; 26(1): 44. CrossRef - Clinicopathological Features and the Prevalence of Oxyntic Gland Neoplasm: A Single-center Retrospective Study
Hikari Asahara, Toshitatsu Takao, Yumiko Asahara, Masakyo Asahara, Douglas Motomura, Hiroya Sakaguchi, Tetsuya Yoshizaki, Nobuaki Ikezawa, Madoka Takao, Yoshinori Morita, Takashi Toyonaga, Masato Komatsu, Ryoji Kushima, Yuzo Kodama
Internal Medicine.2023; 62(19): 2763. CrossRef - Clinicopathological features of gastric adenocarcinoma of fundic gland type
Bao-Zhen Guo, Zhen-Zhen Liu, Gao-Fei Shen, Fei Zhu, Hui-Fen Lian, Xin Li, Jun-Yi Zheng, Jin-Peng Li, Shui-Miao Deng, Rui Huang
World Chinese Journal of Digestology.2023; 31(6): 244. CrossRef - Endoscopic Resection for Gastric Adenocarcinoma of the Fundic Gland Type: A Case Series
Hwa Jin Lee, Gwang Ha Kim, Dong Chan Joo, Moon Won Lee, Bong Eun Lee, Kyungbin Kim
The Korean Journal of Gastroenterology.2023; 81(6): 259. CrossRef - Gastric adenocarcinoma of the fundic gland type: A review of the literature
Zhiyong Zhai, Wei Hu, Zhaoyu Huang, Zemin Chen, Sicun Lu, Wei Gong
JGH Open.2023; 7(12): 812. CrossRef - Clinicopathological features of early stage gastric adenocarcinoma of fundic gland type
Huan Zhang, Shuyan Wang, Yongping Zhang, Fusang Ye, Chunnian Wang
Medicine.2022; 101(2): e28469. CrossRef - Gastric Adenocarcinoma of Fundic Gland Type Treated by Endoscopic Submucosal Dissection
Yong Bo Park, Gwang Ha Kim, Kyungbin Kim, Tae Kyoung Ha, Guk Bin Park, Young Min Kwak
The Korean Journal of Helicobacter and Upper Gastrointestinal Research.2021; 21(1): 82. CrossRef - Gastric epithelial neoplasm of fundic-gland mucosa lineage: proposal for a new classification in association with gastric adenocarcinoma of fundic-gland type
Hiroya Ueyama, Takashi Yao, Yoichi Akazawa, Takuo Hayashi, Koichi Kurahara, Yumi Oshiro, Masayoshi Yamada, Ichiro Oda, Shin Fujioka, Chiaki Kusumoto, Masayoshi Fukuda, Kunihisa Uchita, Tomohiro Kadota, Yasuhiro Oono, Kazuhisa Okamoto, Kazunari Murakami, Y
Journal of Gastroenterology.2021; 56(9): 814. CrossRef - Endoscopic resection is a suitable initial treatment strategy for oxyntic gland adenoma or gastric adenocarcinoma of the fundic gland type
Masaya Iwamuro, Chiaki Kusumoto, Masahiro Nakagawa, Sayo Kobayashi, Masao Yoshioka, Tomoki Inaba, Tatsuya Toyokawa, Shinichiro Hori, Shouichi Tanaka, Kazuhiro Matsueda, Takehiro Tanaka, Hiroyuki Okada
Scientific Reports.2021;[Epub] CrossRef - A series of five patients with oxyntic gland adenoma: Deciphering the clinical and histological features of these rare gastric polyps
Jerry C. Nagaputra, Tracy Jie Zhen Loh, Sangeeta Mantoo, Rafay Azhar, Vikneswaran Namasivayam, Wei Qiang Leow
Human Pathology Reports.2021; 26: 300566. CrossRef - Gastric adenocarcinoma of the fundic gland: A review of clinicopathological characteristics, treatment and prognosis
Xiang-yu Meng, Guang Yang, Cheng-ji Dong, Ru-yi Zheng
Rare Tumors.2021;[Epub] CrossRef - Gastric adenocarcinoma of the fundic gland type: clinicopathological features of eight patients treated with endoscopic submucosal dissection
Chengfang Li, Xinglong Wu, Shuang Yang, Xiaorong Yang, Jin Yao, Hong Zheng
Diagnostic Pathology.2020;[Epub] CrossRef - Multiple gastric adenocarcinoma of fundic gland type: A case report
Ou Chen, Ze-Yong Shao, Xiong Qiu, Guang-Ping Zhang
World Journal of Clinical Cases.2019; 7(18): 2871. CrossRef - Gastric Adenocarcinoma of the Fundic Gland Type
Mark A Benedict, Gregory Y Lauwers, Dhanpat Jain
American Journal of Clinical Pathology.2018; 149(6): 461. CrossRef - Oxyntic Gland Adenoma Treated by Endoscopic Mucosal Resection
In Ji Song, Jin Woo Joo, Jun Chul Park, Sung Kwan Shin, Yong Chan Lee, Sang Kil Lee
The Korean Journal of Helicobacter and Upper Gastrointestinal Research.2017; 17(2): 94. CrossRef - Chief cell‐predominant gastric polyps: a series of 12 cases with literature review
Karen Chan, Ian S Brown, Trevor Kyle, Gregory Y Lauwers, Marian Priyanthi Kumarasinghe
Histopathology.2016; 68(6): 825. CrossRef - Twelve-year natural history of a gastric adenocarcinoma of fundic gland type
Yoshinori Sato, Takashi Fujino, Akira Kasagawa, Ryo Morita, Shun-ichiro Ozawa, Yasumasa Matsuo, Tadateru Maehata, Hiroshi Yasuda, Masayuki Takagi, Fumio Itoh
Clinical Journal of Gastroenterology.2016; 9(6): 345. CrossRef - Clinicopathological features of gastric adenocarcinoma of the fundic gland (chief cell predominant type) by retrospective and prospective analyses of endoscopic findings
Takashi Chiba, Katsuaki Kato, Takayuki Masuda, Shuichi Ohara, Noriyuki Iwama, Takenobu Shimada, Daisuke Shibuya
Digestive Endoscopy.2016; 28(7): 722. CrossRef - Gastric Adenocarcinoma of the Fundic Gland Type Treated by Endoscopic Mucosal Resection: A Case Report and Review of the Literature
Eleanor Lewin, Philip Daroca, Sanjay Sikka, Tong Wu, Yukihiro Nakanishi
Case Reports in Pathology.2016; 2016: 1. CrossRef - Gastric adenocarcinoma of the fundic gland (chief cell-predominant type): A review of endoscopic and clinicopathological features
Masaki Miyazawa, Mitsuru Matsuda, Masaaki Yano, Yasumasa Hara, Fumitaka Arihara, Yosuke Horita, Koichiro Matsuda, Akito Sakai, Yatsugi Noda
World Journal of Gastroenterology.2016; 22(48): 10523. CrossRef - Oxyntic gland adenoma endoscopically mimicking a gastric neuroendocrine tumor: A case report
Tae-In Lee
World Journal of Gastroenterology.2015; 21(16): 5099. CrossRef - Oxyntic gland polyp/adenoma
Rajkumar Vajpeyi, Jyoti Dekate
Diagnostic Histopathology.2014; 20(11): 446. CrossRef - Gastric adenocarcinoma of fundic gland type with unusual behavior
Tetsuya Ueo, Hirotoshi Yonemasu, Tetsuya Ishida
Digestive Endoscopy.2014; 26(2): 293. CrossRef
- Endoscopic Submucosal Dissection of Early Gastric Adenocarcinoma of Fundic Gland Type: A Case Report
- Squamoid Eccrine Ductal Carcinoma of the Scalp
- Yong-Han Jung, Hye-Jung Jo, Mi-Seon Kang
- Korean J Pathol. 2012;46(3):278-281. Published online June 22, 2012
- DOI: https://doi.org/10.4132/KoreanJPathol.2012.46.3.278
- 13,981 View
- 126 Download
- 13 Crossref
-
 Abstract
Abstract
 PDF
PDF Squamoid eccrine ductal carcinoma (SEDC) is an exceedingly rare tumor that shows both squamous and adnexal ductal differentiation. We report a case of this unusual tumor occurring on the occiput of a 53-year-old man. A histopathological examination revealed a nodular lesion infiltrating the dermis and subcutaneous tissue with numerous duct-like structure and squamoid differentiation foci. Five months later, the patient presented with a palpable mass at the site of the previous excision and the right side of the neck. Sono-guided fine needle aspiration of the right neck mass was performed and was diagnosed as a metastastasis of a lymph node. A right neck node dissection and re-excision of the occiput was performed. The histopathological findings were similar, but squamoid differentiation was more prominent than that in the previous lesion. Because of the rarity of SEDC, little is known about its biological behavior and optimal treatment.
-
Citations
Citations to this article as recorded by- Surgical Treatment of Squamoid Eccrine Ductal Carcinoma: A Systematic Review
Nehaa Sohail, Hannah R. Riva, Mojahed Mohammad K Shalabi, Benjamin Kelley, Stanislav N. Tolkachjov
Dermatologic Surgery.2026; 52(3): 290. CrossRef - Squamoid Eccrine Ductal Carcinoma: A Systematic Review
Baraa Saifi, Maria Maalouf, Areeba Hasan, Doha Fadel, Marita Kassab, Nancy Emmanuel
Current Dermatology Reports.2025;[Epub] CrossRef - Aggressive Squamoid Eccrine Ductal Carcinoma of the Face: A Rare and Challenging Diagnosis—Case Report and Literature Review
Bruno Špiljak, Damir Sauerborn, Matej Tomas, Brankica Gregorić Butina, Ivana Mahovne, Suzana Erić, Bruno Vidaković, Stjepanka Lešić
Medicina.2025; 61(4): 612. CrossRef - Squamoid Eccrine Ductal Carcinoma: Treatment and Outcomes
Megan M. Lim, Jillian A. Macdonald
The American Journal of Dermatopathology.2022; 44(4): 249. CrossRef - Recurrent Squamoid Eccrine Ductal Carcinoma After Mohs Micrographic Surgery
Cardwell Gavin, Brinker Alyson
Dermatologic Surgery.2021; 47(8): 1108. CrossRef - Carcinoma ecrino: dos casos de una neoplasia inusual y revisión de la literatura
Clara Matas-Nadal, Josep Manel Fernández-Armenteros, Felip Vilardell, Josep Manel Casanova, Rafael S. Aguayo Ortiz
Piel.2020; 35(3): 159. CrossRef - Squamoid eccrine ductal carcinoma: a case report
Zeynep Bayramoğlu, Betül Ünal
The European Research Journal.2020; 6(2): 173. CrossRef - Squamoid eccrine ductal carcinoma of the ear helix
Sunmin Yim, Yun Ho Lee, Seoung Wan Chae, Won‐Serk Kim
Clinical Case Reports.2019; 7(7): 1409. CrossRef - A Case of Eccrine Carcinoma Presenting with Neurological Manifestations
Lynne Michelle B Soldivillo, Rosalina Espiritu Picar, Allen Evaristo
Philippine Journal of Internal Medicine.2017; 55(1): 46. CrossRef - Squamoid eccrine ductal carcinoma of the scalp
Hsien Chan, Vicki Howard, Denis Moir, Delwyn Dyall‐Smith
Australasian Journal of Dermatology.2016;[Epub] CrossRef - Squamoid Eccrine Ductal Carcinoma
Michiel P.J. van der Horst, Adriana Garcia-Herrera, Dorota Markiewicz, Blanca Martin, Eduardo Calonje, Thomas Brenn
American Journal of Surgical Pathology.2016; 40(6): 755. CrossRef - Squamoid eccrine ductal carcinoma
Maria Isabel Ramos Saraiva, Marcella Amaral Horta Barbosa Vieira, Larissa Karine Leite Portocarrero, Rafael Cavanellas Fraga, Priscila Kakizaki, Neusa Yuriko Sakai Valente
Anais Brasileiros de Dermatologia.2016; 91(6): 799. CrossRef - Anatomoclinical study of 30 cases of sclerosing sweat duct carcinomas (microcystic adnexal carcinoma, syringomatous carcinoma and squamoid eccrine ductal carcinoma)
E. Frouin, M.D. Vignon‐Pennamen, B. Balme, B. Cavelier‐Balloy, U. Zimmermann, N. Ortonne, A. Carlotti, L. Pinquier, J. André, B. Cribier
Journal of the European Academy of Dermatology and Venereology.2015; 29(10): 1978. CrossRef
- Surgical Treatment of Squamoid Eccrine Ductal Carcinoma: A Systematic Review
- Clinicopathological Analysis of 21 Thymic Neuroendocrine Tumors
- Soomin Ahn, Jae Jun Lee, Sang Yun Ha, Chang Ohk Sung, Jhingook Kim, Joungho Han
- Korean J Pathol. 2012;46(3):221-225. Published online June 22, 2012
- DOI: https://doi.org/10.4132/KoreanJPathol.2012.46.3.221
- 11,546 View
- 95 Download
- 31 Crossref
-
 Abstract
Abstract
 PDF
PDF Background Thymic neuroendocrine carcinomas (NECs) are uncommon, for which there is no established information available because of a limited number of epidemiological study in Asia.
Methods We reviewed 21 cases of surgically resected thymic NECs, and evaluated their pathological and clinical features.
Results It showed male predominance (male/female ratio, 15/6) with wide age range from 20 to 72 years (mean age, 49 years). All 21 cases were divided into two types according to the World Health Organization criteria: atypical carcinoid (n=18) and large cell NEC (n=3). Three cases of atypical carcinoid (AC) were associated with ectopic Cushing's syndrome. All the patients (3/3) with large cell NEC (3/3) and 16.7% (3/18) of those with AC died of tumor progression. Common sites of metastasis included lung, lymph node, brain, lumbar spine, mediastinum, bone, and liver.
Conclusions In conclusion, thymic neuroendocrine tumors carry a poor prognosis. Regarding the tumor classification, our results showed that a vast majority of carcinoids in the thymus correspond to ACs. In addition, our results also indicate that typical carcinoid is a very rare entity. Some cases of AC exhibited a large size, solid pattern and they showed aggressive clinical behavior, which highlights the spectrum of histologic appearances of thymic NECs.
-
Citations
Citations to this article as recorded by- Pathology, molecular biology, medical oncology, and radiotherapy implications of lung neuroendocrine neoplasm classification: a multidisciplinary perspective
Giuseppe Pelosi, Alice Laffi, Gianpiero Catalano, Antonino Bruno, Mauro Papotti, Barbara Bassani, Fabrizio Bianchi, Eleonora Duregon, Tommaso Martino De Pas, Chiara Catania, Riccardo Ricotta, Riccardo Papa, Sergio Harari, Angelica Sonzogni, Ilaria Guerrie
Expert Review of Anticancer Therapy.2026; 26(2): 205. CrossRef - A Case of Stage IV Thymic Typical Carcinoid with Multiple Bone Metastases
Hisaya Chikaraishi, Tomohiro Maniwa, Hironobu Samejima, Masao Kobayashi, Julian Horiguchi, Ryu Kanzaki, Hidetoshi Satomi, Keiichiro Honma, Jiro Okami
Haigan.2025; 65(4): 286. CrossRef - Neuroendocrine neoplasms of the thymus
Paul D. Barone, Chen Zhang
Frontiers in Immunology.2024;[Epub] CrossRef - Thymic neuroendocrine tumours
Jan von der Thüsen
Diagnostic Histopathology.2023; 29(2): 114. CrossRef - The Utility of Fine Needle Aspiration (FNA) Biopsy in the Diagnosis of Mediastinal Lesions
Uma Kundu, Qiong Gan, Deepak Donthi, Nour Sneige
Diagnostics.2023; 13(14): 2400. CrossRef - Paediatric and adolescent ectopic Cushing's syndrome: systematic review
Chethan Yami Channaiah, Manjiri Karlekar, Vijaya Sarathi, Anurag Ranjan Lila, Shruthi Ravindra, Padma Vikram Badhe, Gaurav Malhotra, Saba Samad Memon, Virendra Ashokrao Patil, C S Pramesh, Tushar Bandgar
European Journal of Endocrinology.2023; 189(4): S75. CrossRef - Multiple endocrine neoplasia type 1 (MEN-1) and neuroendocrine neoplasms (NENs)
Grigoris Effraimidis, Ulrich Knigge, Maria Rossing, Peter Oturai, Åse Krogh Rasmussen, Ulla Feldt-Rasmussen
Seminars in Cancer Biology.2022; 79: 141. CrossRef - Multiple electrolyte disturbances as the presenting feature of multiple endocrine neoplasia type 1 (MEN-1)
Adrian Po Zhu Li, Sheela Sathyanarayan, Salvador Diaz-Cano, Sobia Arshad, Eftychia E Drakou, Royce P Vincent, Ashley B Grossman, Simon J B Aylwin, Georgios K Dimitriadis
Endocrinology, Diabetes & Metabolism Case Reports.2022;[Epub] CrossRef - Metastatic Thymic Carcinoid: Does Surgeon Have a Primary Role?
Kumud Gupta, Ravindra K. Dewan, Vinitha Viswambharan Nair, Rajat Saxena, Shaleen Prasad
The Indian Journal of Chest Diseases and Allied Sciences.2022; 56(4): 255. CrossRef - A resected case of large cell neuroendocrine carcinoma of the thymus
Masashi Umeda, Takahiko Misao, Tomoya Senoh, Yoshinobu Shikatani, Motoi Aoe
The Journal of the Japanese Association for Chest Surgery.2022; 36(7): 766. CrossRef - Treatment strategy and prognostic analysis of spinal metastases from thymomas: A retrospective study from a single center
Shuzhong Liu, Xi Zhou, An Song, Zhen Huo, Siyuan Yao, Yipeng Wang, Yong Liu
Clinical Neurology and Neurosurgery.2020; 196: 106056. CrossRef - Large Cell Neuroendocrine Carcinoma of the Mediastinum Successfully Treated with Systemic Chemotherapy after Palliative Radiotherapy
Takeaki Hidaka, Saki Okuzumi, Ako Matsuhashi, Hidenori Takahashi, Kazunori Hata, Seiichiro Shimizu, Yoshinobu Iwasaki
Internal Medicine.2019; 58(4): 563. CrossRef - Surgical management of spinal metastases of thymic carcinoma
Shuzhong Liu, Xi Zhou, An Song, Zhen Huo, William A. Li, Radhika Rastogi, Yipeng Wang, Yong Liu
Medicine.2019; 98(3): e14198. CrossRef - Resected thymic large cell neuroendocrine carcinoma
Shogo Ogata, Ryo Maeda, Masaki Tomita, Yuichiro Sato, Takanori Ayabe, Kunihide Nakamura
International Journal of Surgery Case Reports.2019; 60(C): 53. CrossRef - Metastatic or locally advanced mediastinal neuroendocrine tumours
Aadil Adnan, Shreyas Kudachi, Sudha Ramesh, Kumar Prabhash, Sandip Basu
Nuclear Medicine Communications.2019; 40(9): 947. CrossRef - Results of treatment for thymic neuroendocrine tumours: multicentre clinicopathological study†
Naoko Ose, Hajime Maeda, Masayoshi Inoue, Eiichi Morii, Yasushi Shintani, Hiroshi Matsui, Hirohito Tada, Tositeru Tokunaga, Kenji Kimura, Yasushi Sakamaki, Yukiyasu Takeuchi, Kenjiro Fukuhara, Hiroshi Katsura, Teruo Iwasaki, Meinoshin Okumura
Interactive CardioVascular and Thoracic Surgery.2018; 26(1): 18. CrossRef - Patterns of Failure Following Postoperative Radiation Therapy Based on “Tumor Bed With Margin” for Stage II to IV Type C Thymic Epithelial Tumor
Kyung Hwa Lee, Jae Myoung Noh, Yong Chan Ahn, Dongryul Oh, Jhingook Kim, Young Mog Shim, Jung-ho Han
International Journal of Radiation Oncology*Biology*Physics.2018; 102(5): 1505. CrossRef - Resected thymic large cell neuroendocrine carcinoma: report of a case
Hiromitsu Domen, Yasuhiro Hida, Masaaki Sato, Haruka Takahashi, Tatsuru Ishikawa, Yosuke Shionoya, Midori Hashimoto, Kaoru Nishiyama, Yuma Aoki, Kazuho Inoko, Syotaro Furukawa, Kazuomi Ichinokawa, Hidehisa Yamada
Surgical Case Reports.2018;[Epub] CrossRef - Successful treatment of malignant thymoma with sacrum metastases
Shuzhong Liu, Xi Zhou, An Song, Zhen Huo, William A. Li, Radhika Rastogi, Yipeng Wang, Yong Liu
Medicine.2018; 97(51): e13796. CrossRef - Incidental metastatic mediastinal atypical carcinoid in a patient with parathyroid adenoma: a case report
Zareen Kiran, Asma Ahmed, Owais Rashid, Saira Fatima, Faizan Malik, Saulat Fatimi, Mubassher Ikram
Journal of Medical Case Reports.2017;[Epub] CrossRef - Thymus neuroendocrine tumors with CTNNB1 gene mutations, disarrayed ß-catenin expression, and dual intra-tumor Ki-67 labeling index compartmentalization challenge the concept of secondary high-grade neuroendocrine tumor: a paradigm shift
Alessandra Fabbri, Mara Cossa, Angelica Sonzogni, Paolo Bidoli, Stefania Canova, Diego Cortinovis, Maria Ida Abbate, Fiorella Calabrese, Nazarena Nannini, Francesca Lunardi, Giulio Rossi, Stefano La Rosa, Carlo Capella, Elena Tamborini, Federica Perrone,
Virchows Archiv.2017; 471(1): 31. CrossRef - Thymic large cell neuroendocrine carcinoma – a rare and aggressive tumor: a case report
Efared Boubacar, Gabrielle Atsame-Ebang, Sani Rabiou, Ammor Fatimazahra, Asmae Mazti, Ibrahim S. Sidibé, Layla Tahiri, Nawal Hammas, Ouadnouni Yassine, Smahi Mohamed, Chbani Laila, El Fatemi Hinde
Journal of Medical Case Reports.2017;[Epub] CrossRef - Clinicopathological features of neoplasms with neuroendocrine differentiation occurring in the liver
Yoriko Nomura, Osamu Nakashima, Jun Akiba, Sachiko Ogasawara, Shogo Fukutomi, Rin Yamaguchi, Hironori Kusano, Masayoshi Kage, Koji Okuda, Hirohisa Yano
Journal of Clinical Pathology.2017; 70(7): 563. CrossRef - Retrosternal goiter and thymic carcinoid: A rare co-existence
Abdulsalam Yaseen Taha, Nezar A. Almahfooz, Hassanain H. Khudair
Journal of the Egyptian Society of Cardio-Thoracic Surgery.2017; 25(4): 369. CrossRef - A case of large-cell neuroendocrine carcinoma of the thymus involving a patient with long-term survival after surgery
Qiuming Kan, Kohei Tagawa, Teruaki Ishida, Mitsuyo Nishimura, Katsuhiko Aoyama
The Journal of the Japanese Association for Chest Surgery.2017; 31(7): 927. CrossRef - Neuroendokrine Neoplasien des Mediastinums
L. Brcic, M. Heidinger, H. Popper
Der Pathologe.2016; 37(5): 434. CrossRef - Outcome of primary neuroendocrine tumors of the thymus: A joint analysis of the International Thymic Malignancy Interest Group and the European Society of Thoracic Surgeons databases
Pier Luigi Filosso, Xiaopan Yao, Usman Ahmad, Yilei Zhan, James Huang, Enrico Ruffini, William Travis, Marco Lucchi, Andreas Rimner, Alberto Antonicelli, Francesco Guerrera, Frank Detterbeck
The Journal of Thoracic and Cardiovascular Surgery.2015; 149(1): 103. CrossRef - Clinical Significance of Persistent Tumor in Bone Marrow during Treatment of High-risk Neuroblastoma
Young Bae Choi, Go Eun Bae, Na Hee Lee, Jung-Sun Kim, Soo Hyun Lee, Keon Hee Yoo, Ki Woong Sung, Hong Hoe Koo
Journal of Korean Medical Science.2015; 30(8): 1062. CrossRef - Tumor genetics and survival of thymic neuroendocrine neoplasms: A multi‐institutional clinicopathologic study
Philipp Ströbel, Andreas Zettl, Konstantin Shilo, Wen‐Yu Chuang, Andrew G. Nicholson, Yoshihiro Matsuno, Anthony Gal, Rolf Hubert Laeng, Peter Engel, Carlo Capella, Mirella Marino, John Kwok-Cheung Chan, Andreas Rosenwald, William Travis, Teri J. Franks,
Genes, Chromosomes and Cancer.2014; 53(9): 738. CrossRef - Disseminated large cell neuroendocrine carcinoma associated with ectopic adrenocorticotropic hormone secretion
A Van der Walt, K Huddle, S Pather, A Korb
Journal of Endocrinology, Metabolism and Diabetes of South Africa.2014; 19(1): 40. CrossRef - Morphologic Alteration of Metastatic Neuroblastic Tumor in Bone Marrow after Chemotherapy
Go Eun Bae, Yeon-Lim Suh, Ki Woong Sung, Jung-Sun Kim
Korean Journal of Pathology.2013; 47(5): 433. CrossRef
- Pathology, molecular biology, medical oncology, and radiotherapy implications of lung neuroendocrine neoplasm classification: a multidisciplinary perspective
- Liquid-Based Cytology of Villoglandular Adenocarcinoma of the Cervix: A Report of 3 Cases
- Younghwa Choi, Haeryoung Kim, Haiyoung Choi, Daehyun Hwang, Gheeyoung Choe, Jin-Haeng Chung, So Yeon Park, Hye Seung Lee, Jin Ho Paik, Hyo Jin Park
- Korean J Pathol. 2012;46(2):215-220. Published online April 25, 2012
- DOI: https://doi.org/10.4132/KoreanJPathol.2012.46.2.215
- 9,665 View
- 62 Download
- 5 Crossref
-
 Abstract
Abstract
 PDF
PDF Villoglandular adenocarcinoma (VGA) is a rare subtype of cervical adenocarcinoma with a more favorable prognosis compared to conventional adenocarcinomas. Although the tumors are usually recognized on colposcopic examination due to the mainly exophytic growth pattern, they may be underdiagnosed as benign lesions by cytology because of their minimal cytologic atypia. We report the liquid-based cytology (LBC) findings of three histologically confirmed VGAs which we have recently identified. They were characterized by hypercellular smears on low-power examination with smooth-bordered three-dimensional papillary fragments. The nuclei were relatively uniform with irregular nuclear membranes. Nucleoli were small but distinct and macronucleoli were also seen. The abnormal architectural patterns such as papillary structures and nuclear overlapping and nuclear hyperchromasia are important clues to the diagnosis of VGA. In addition, nuclear membrane irregularity and prominent nucleoli can be recognized on LBC specimens, further facilitating its diagnosis.
-
Citations
Citations to this article as recorded by- Adenocarcinoma viloglandular da cérvice uterina (AVG): relato de um caso in situ e uma análise sobre os desafios para o diagnóstico citológico
Catharine de Araújo Crisóstomo Pontes, Carlos Eduardo Queiroz de Lima, Adrya Lúcia Peres Bezerra, Joelma Basilio Tompson, Rosângela Pontes de Andrade, Felipe Campos de Queiroz
Revista Brasileira de Patologia do Trato Genital Inferior.2026; 9(2): 41. CrossRef - Adenocarcinoma viloglandular da cérvice uterina: relato de um caso “in situ" e análise dos desafios para o diagnóstico citológico
Catharine de Araújo Crisóstomo Pontes, Carlos Eduardo Queiroz de Lima, Adrya Lúcia Peres Bezerra, Felipe Campos de Queiroz
Revista Brasileira de Patologia do Trato Genital Inferior.2026; 9(2): 50. CrossRef - Villoglandular Carcinoma of the Uterine Cervix: Clinic‐Cytopathological‐Histological Features of a Rare Case With Brief Review of the Literature
Neelam Sood, Ruchika Gupta, Sanjay Gupta
Cytopathology.2025; 36(6): 596. CrossRef - Villoglandular adenocarcinoma of the uterine cervix: a systematic review and meta-analysis
Anna K. Dietl, Matthias W. Beckmann, Konrad Aumann
Archives of Gynecology and Obstetrics.2021; 304(2): 317. CrossRef - Les lésions glandulaires du col utérin : cas no 4
Catherine Genestie, Chiraz Hadj Kacem, Pierre Duvillard
Annales de Pathologie.2016; 36(3): 192. CrossRef
- Adenocarcinoma viloglandular da cérvice uterina (AVG): relato de um caso in situ e uma análise sobre os desafios para o diagnóstico citológico
- Detection of Survivin and COX-2 in Thyroid Carcinoma: Anaplastic Carcinoma Shows Overexpression of Nuclear Survivin and Low COX-2 Expression
- Young A Kim, Meesoo Chang, Young Joo Park, Ji Eun Kim
- Korean J Pathol. 2012;46(1):55-60. Published online February 23, 2012
- DOI: https://doi.org/10.4132/KoreanJPathol.2012.46.1.55
- 10,835 View
- 68 Download
- 6 Crossref
-
 Abstract
Abstract
 PDF
PDF Background Overexpression of survivin, a member of the inhibitors of apoptosis protein, has been reported in various carcinomas, and its interaction with cyclooxygenase 2 (COX-2) results in accelerated tumor progression. The purpose of this study is to investigate the immunohistochemical expression of survivin and COX-2 in benign and malignant thyroid tissues and to define its association with pathologic and clinical features.
Methods We examined expression of survivin and COX-2 by immunohistochemistry in 334 benign and malignant thyroid tissues and evaluated their clinical significance.
Results Expression of survivin showed an increase along the spectrum of thyroid carcinoma progression; rarely positive in adenomatous goiter, moderately positive in papillary carcinoma, and strongly positive in anaplastic carcinoma (AC). Papillary microcarcinoma revealed the highest COX-2 positivity and AC demonstrated the lowest positivity among thyroid cancers. Node negative carcinomas showed higher COX-2 expression than node positive tumors. Survivin expression did not correlate with COX-2.
Conclusions Our findings suggest that survivin overexpression may be related to the pathogenesis of AC and can be a predictor of disease progression. COX-2 may be involved in the early phase of thyroid carcinoma.
-
Citations
Citations to this article as recorded by- Interplay between nuclear survivin and the PRC2 complex and its impact on H3K27me3-directed transcriptional repression
Adesh D. Vaidya, Alexander J. Fezovich, Sally P. Wheatley
Journal of Cell Science.2026;[Epub] CrossRef - Survivin as a diagnostic and therapeutic marker for thyroid cancer
Mohammad-Reza Mahmoudian-Sani, Arash Alghasi, Ali Saeedi-Boroujeni, Akram Jalali, Mohammad Jamshidi, Ali Khodadadi
Pathology - Research and Practice.2019; 215(4): 619. CrossRef - TFAP2B overexpression contributes to tumor growth and progression of thyroid cancer through the COX-2 signaling pathway
Xiaoyan Fu, Huayong Zhang, Zhipeng Chen, Zhongyuan Yang, Dingbo Shi, Tianrun Liu, Weichao Chen, Fan Yao, Xuan Su, Wuguo Deng, Miao Chen, Ankui Yang
Cell Death & Disease.2019;[Epub] CrossRef - Expression of nm23-H1 and COX-2 in thyroid papillary carcinoma and microcarcinoma
Marija Milkovic Perisa, Bozena Sarcevic, Koraljka Gall Troselj, Kresimir Grsic, Sanda Sitic, Sven Seiwerth
Oncology Letters.2017; 13(5): 3547. CrossRef - The Diagnostic Usefulness of HMGA2, Survivin, CEACAM6, and SFN/14-3-3 δ in Follicular Thyroid Carcinoma
Min Hye Jang, Kyeong Cheon Jung, Hye Sook Min
Journal of Pathology and Translational Medicine.2015; 49(2): 112. CrossRef - Evaluation of survivin expression and its prognostic value in papillary thyroid carcinoma
Sonja Selemetjev, Tijana Isic Dencic, Ilona Marecko, Jelena Jankovic, Ivan Paunovic, Svetlana Savin, Dubravka Cvejic
Pathology - Research and Practice.2014; 210(1): 30. CrossRef
- Interplay between nuclear survivin and the PRC2 complex and its impact on H3K27me3-directed transcriptional repression
- Diagnostic Features of Fine Needle Aspiration Cytology of Pleomorphic Adenoma, Adenoid Cystic Carcinoma, and Mucoepidermoid Carcinoma of Salivary Gland.
- Eun Sook Nam, Won Bo Jo, Jung Ho Han, Insun Kim
- J Pathol Transl Med. 1990;1(1):60-67.
- 6,970 View
- 302 Download
-
 Abstract
Abstract
 PDF
PDF - The evaluate the diagnostic findings of salivary gland tumors, we reexamined aspiration cytology smears of 7 cases of pleomorphic adenoma, 3 cases of adenoid cystic carcinoma, and 3 cases of mucoepidermoid carcinoma, performed during April 1986 to March 1990, which were comfurmed by surgical excision and histologic diagnosis. The results obtained are summarized as follows : 1. All cases of pleomorphic adenoma showed branching cellular clusters of epithelial and myoepithelial cells. Acellular elements including myxomatous and chondroid components were observed. There were no cellular pleomorphism and nucleoli. Keratinizing squamous epithelial cells and keratin pearls were noted. 2. The smears of adenoid cystic carcinoma showed cell balls or cell cords containing a central hyaline core. Nuclear atypism and the nucleoli were frequently observed. There were no keratinizing squamous epithelial cells. 3. The smears of mucoepidermoid carcinoma showed mainly sheets or clusters of intermediate cells and some mucin-producing cells. Some nuclear pleomorphism was observed. Mucinous material and many inflammatory cells were present in the background.
- Mature Teratoma in the Adrenal Gland.
- Eun Jung Cha
- Korean J Pathol. 2011;45:S98-S100.
- DOI: https://doi.org/10.4132/KoreanJPathol.2011.45.S1.S98
- 3,998 View
- 50 Download
-
 Abstract
Abstract
 PDF
PDF - A teratoma is a germ-cell tumor composed of tissue components representing derivatives of three germ layers. A teratoma in the region of adrenal gland is a rare retroperitoneal tumor. We now report a case of a primary adrenal teratoma. A 38-year-old woman presented with an incidentally detected adrenal mass. The computed tomography scan revealed a 9x8x7.5 cm fat density mass with calcification in the left adrenal gland. The surgically resected tumor was round and well circumscribed and the adrenal gland was present at the periphery of the tumor. The cut surface contained fat tissue and a hair containing cyst. Microscopically, the tumor consisted of adipose tissue, hair, skin appendage, nerve, muscle bundle and bone.
- Osteoclast-like Giant Cell Tumor of the Parotid Gland Accompanied with Carcinoma ex Pleomorphic Adenoma.
- Mi Jung Kwon, Eun Sook Nam, Seong Jin Cho, Hyung Sik Shin, Ji Hyun Kwon, Young Soo Rho
- Korean J Pathol. 2011;45:S84-S88.
- DOI: https://doi.org/10.4132/KoreanJPathol.2011.45.S1.S84
- 4,944 View
- 46 Download
- 2 Crossref
-
 Abstract
Abstract
 PDF
PDF - The origin of osteoclast-like giant cell tumor (OGCT) of the salivary gland has been debated because the prototypic cells of osteoclast-like cells and mononuclear stromal cells are largely unexplained in this gland. Bone marrow-derived CD14+ and CD45+ monocyte-derived multipotential cells (CD14+/CD45+ MOMC) may be one of the possible origins of OGCTs of salivary glands, which have never been explored in salivary OGCTs. We present a case of OGCT accompanied with carcinoma ex pleomorphic adenoma in the parotid gland of a 67-year-old Korean female. The tumor presented as a rapidly growing cervical mass comprising a central area of carcinoma ex pleomorphic adenoma and a peripheral circumferential area of OGCT. The immunohistochemical staining pattern was phenotypically consistent with bone marrow-derived CD14+/CD45+ MOMC. This case is the first report of a salivary OGCT in Korea.
-
Citations
Citations to this article as recorded by- Giant cell tumour of the parotid gland: a rare, unusual entity
Erna Ahsan, Kranthi Kumar Jandrasupalli, Prashant Durgapal, Divya Yadav
BMJ Case Reports.2025; 18(4): e261751. CrossRef - Tumeur à cellules géantes de type ostéoclastique de la parotide
S. Rammeh, I. Hergli, M.K. M’farrej, N. Znaidi, S. Nechi, R. Zermani
Revue de Stomatologie, de Chirurgie Maxillo-faciale et de Chirurgie Orale.2014; 115(3): 185. CrossRef
- Giant cell tumour of the parotid gland: a rare, unusual entity

 E-submission
E-submission
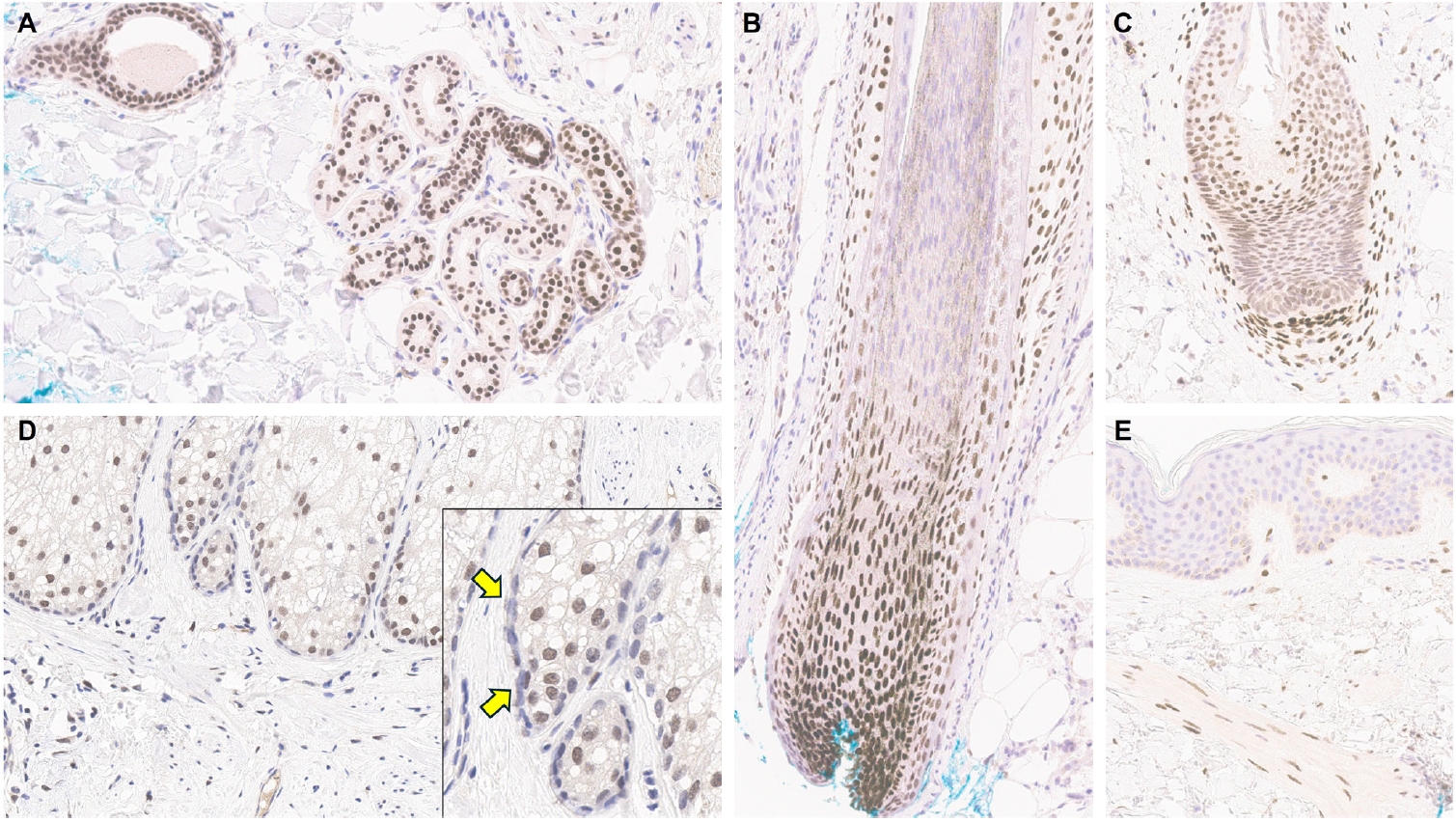
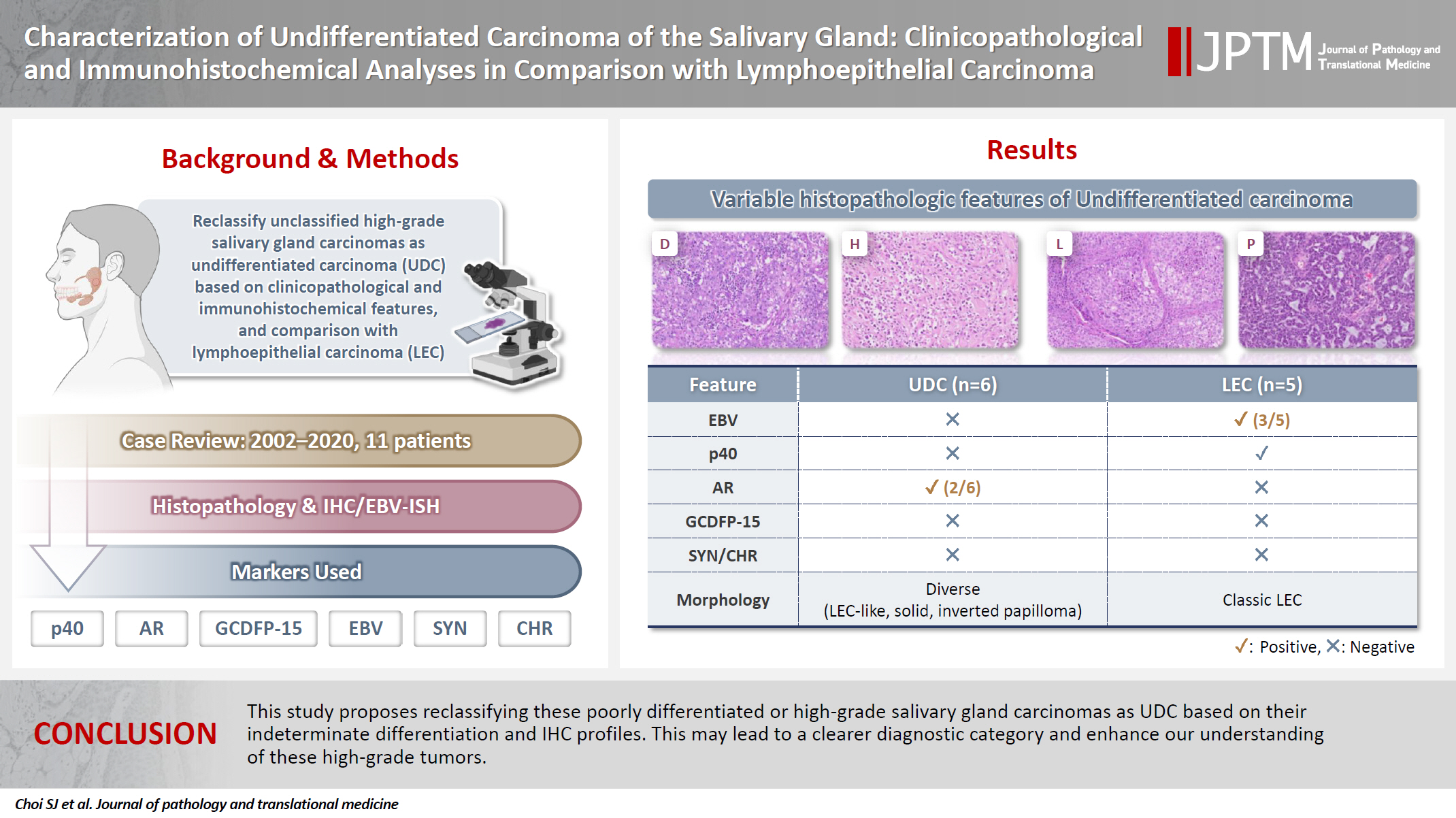












 First
First Prev
Prev



