Articles
- Page Path
- HOME > J Pathol Transl Med > Forthcoming articles > Article
-
Original Article
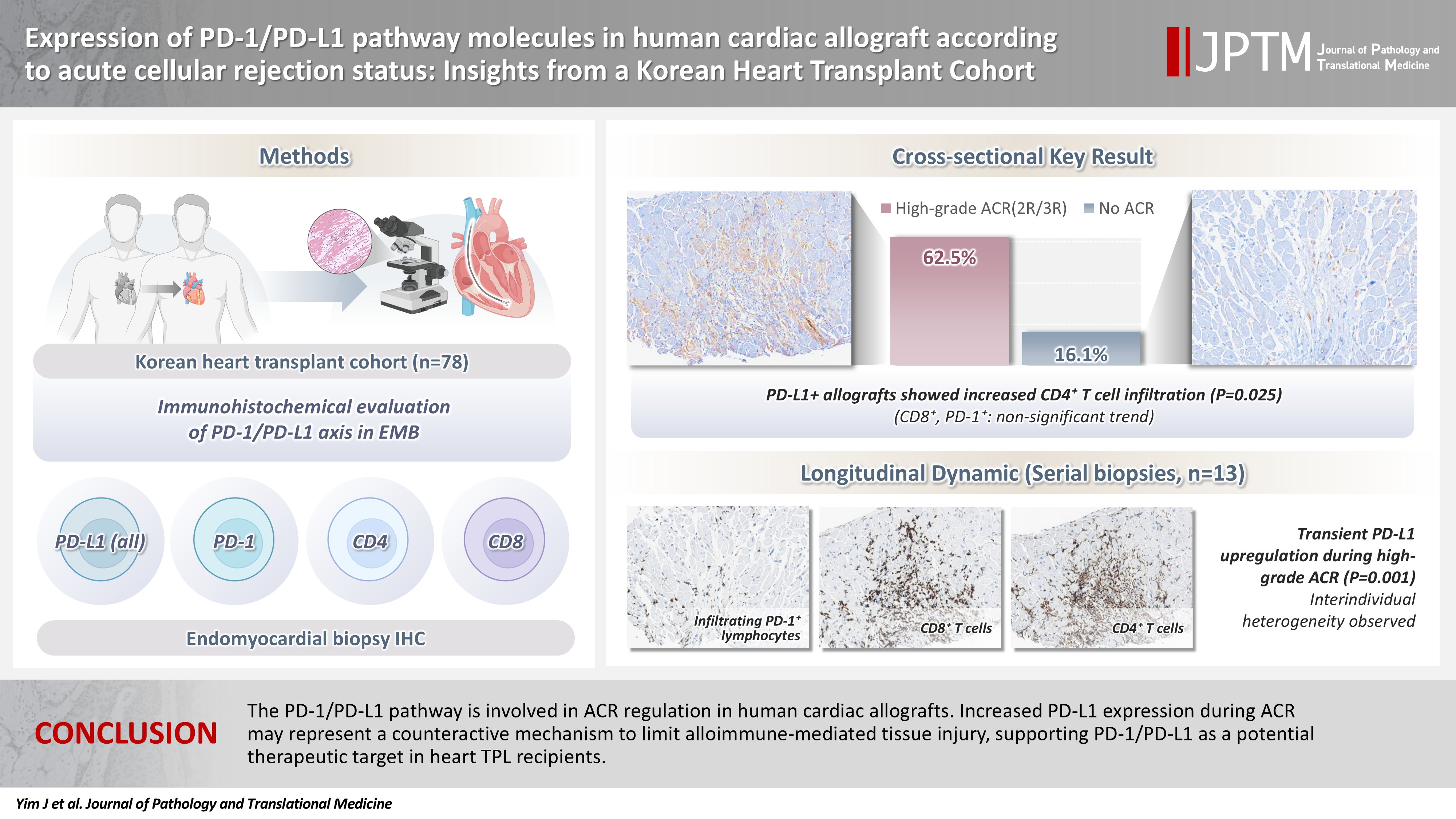
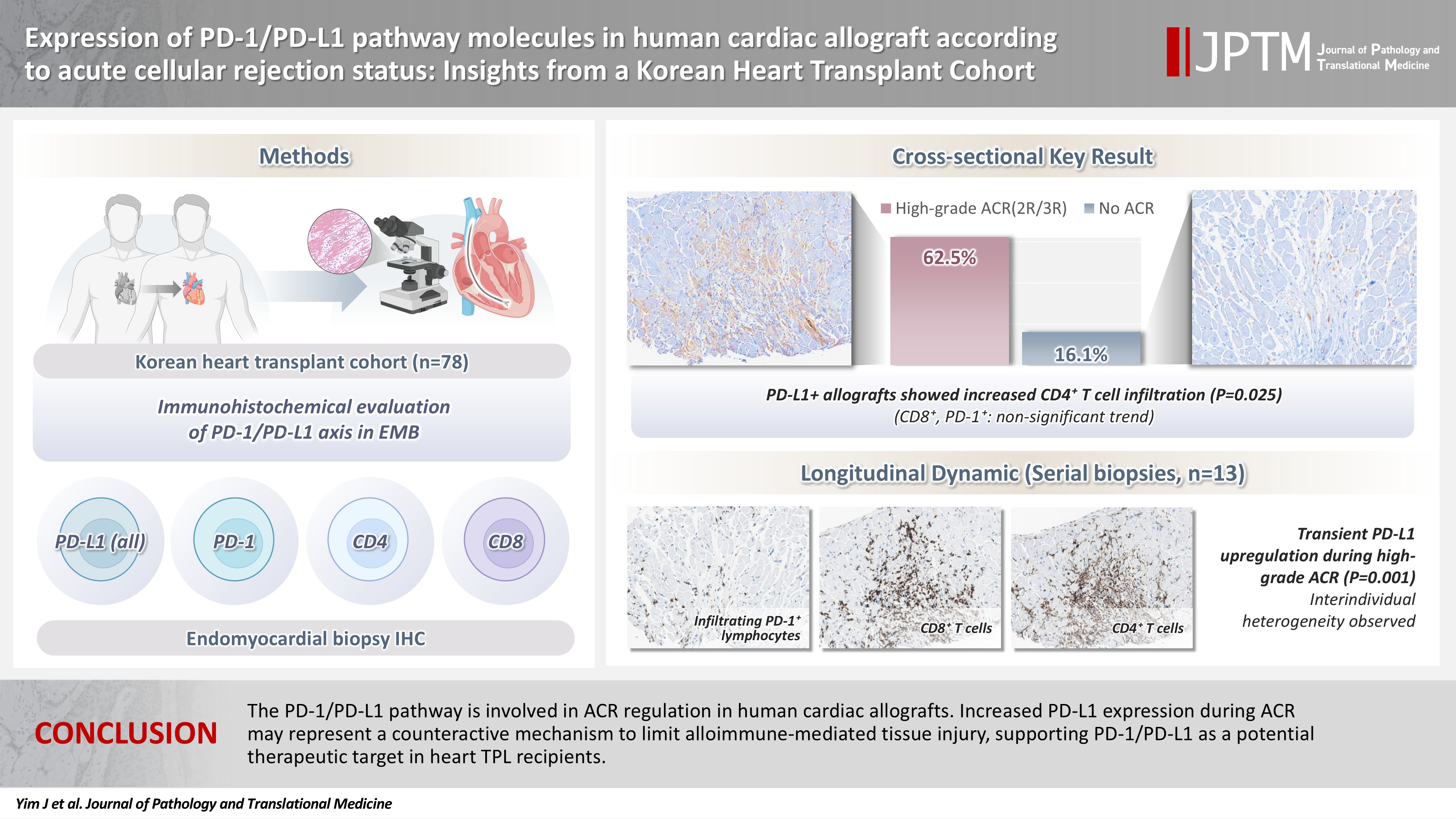
Expression of PD-1/PD-L1 pathway molecules in human cardiac allograft according to acute cellular rejection status: insights from a Korean Heart Transplant Cohort -
Jeemin Yim1,2
 , Yoon Kyung Jeon1,3,4
, Yoon Kyung Jeon1,3,4 , Doo Hyun Chung1,5,6
, Doo Hyun Chung1,5,6 , Jaemoon Koh1,3
, Jaemoon Koh1,3
-
DOI: https://doi.org/10.4132/jptm.2026.01.02
Published online: March 27, 2026
1Department of Pathology, Seoul National University College of Medicine, Seoul, Korea
2Department of Pathology, Seoul Metropolitan Government-Seoul National University Boramae Medical Center, Seoul, Korea
3Department of Pathology, Seoul National University Hospital, Seoul, Korea
4Cancer Research Institute, Seoul National University College of Medicine, Seoul, Korea
5Department of Biomedical Sciences, Seoul National University College of Medicine, Seoul, Korea
6Ischemic/Hypoxic Disease Institute, Seoul National University College of Medicine, Seoul, Korea
- Corresponding author: Jaemoon Koh, MD, PhD Department of Pathology, Seoul National University College of Medicine, 103 Daehak-ro, Jongno-gu, Seoul 03080, Korea Tel: +82-2-740-0029, Fax: +82-2-743-5530 E-mail: tarda@naver.com
© The Korean Society of Pathologists/The Korean Society for Cytopathology
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (https://creativecommons.org/licenses/by-nc/4.0) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
- 780 Views
- 55 Download
Abstract
-
Background
- Acute cellular rejection (ACR) following heart transplantation (TPL) compromises graft function and survival. The programmed cell death-1 (PD-1)/PD-1 ligand-1 (PD-L1) pathway represents an immune checkpoint that maintains peripheral immune tolerance, but its expression and significance in human cardiac allografts with ACR remain unclear. Thus, we investigated PD-1/PD-L1 expression in endomyocardial biopsies from heart TPL recipients to clarify the role of this pathway in the ACR of human cardiac allografts and explore the potential of therapeutic modulation of PD-1/PD-L1 in this setting.
-
Methods
- Endomyocardial biopsies of 78 patients with heart TPL were subjected to immunohistochemistry for PD-L1, PD-1, CD4, and CD8. PD-L1 expression and quantities of PD-1+, CD4+, and CD8+ infiltrating lymphocytes were evaluated according to clinicopathological features, ACR presence, and clinical outcomes.
-
Results
- Allografts with high-grade ACR (International Society for Heart and Lung Transplantation grades 2R and 3R) demonstrated markedly higher PD-L1 expression than did those without ACR (62.5% vs. 16.1%, p < .001). PD-L1 expression was positively associated with CD4+ lymphocyte infiltration (p = .025), whereas CD8 and PD-1+ lymphocyte counts were higher in PD-L1-positive allografts without reaching statistical significance (p = .059 and p = .390, respectively). Serial biopsies revealed that PD-L1 expression was upregulated in patients with high-grade ACR compared with that in previous non-ACR tissues, and follow-up biopsies were performed after ACR resolution.
-
Conclusions
- The PD-1/PD-L1 pathway is involved in ACR regulation in human cardiac allografts. Increased PD-L1 expression during ACR may represent a counteractive mechanism to limit alloimmune-mediated tissue injury, supporting PD-1/PD-L1 as a potential therapeutic target in heart TPL recipients.
- Heart transplantation (TPL) is the only treatment of choice for prolonging the survival and quality of life of patients with severe heart failure [1]. Despite advances in immunosuppressive therapy, acute cellular rejection (ACR) continues to impair long-term graft survival. According to the International Society for Heart and Lung Transplantation (ISHLT) registry, ACR accounts for approximately 11% of heart transplant deaths in the first 3 years post-TPL [2], usually occurring within the first year (often in the first 6 months post-TPL) and affecting 20%–40% of recipients, with each episode predisposing to graft dysfunction and failure [3,4]. The gold standard for ACR diagnosis is histopathological grading of endomyocardial biopsy (EMB) for lymphocytic infiltration and myocyte injury. Routine EMB surveillance, as per ISHLT guidelines, enables early detection and management of rejection [5-8].
- ACR arises from the recipient T-cell recognition of donor alloantigens. T-lymphocytes orchestrate allograft injury via antigen-specific activation and costimulatory signaling. Critically, co-inhibitory immune checkpoints temper T-cell activation to maintain peripheral tolerance and prevent excessive tissue damage [9,10]. Among these, the programmed cell death-1 (PD-1) receptor and its ligand programmed death-ligand 1 (PD-L1; also known as B7-H1) constitute a key inhibitory pathway activated during chronic antigen exposure. Engagement of PD-1 on T cells by PD-L1, expressed on antigen-presenting and parenchymal cells, attenuates effector T cell responses and helps preserve transplant tolerance [11,12]. Notably, this pathway is utilized by cancer and chronic viruses to evade immunity. Conversely, PD-1/PD-L1 blockade therapies restore T-cell activity in oncology [11,12]. In TPL models, PD-1/PD-L1 interactions are essential for alloimmune response downregulation and graft survival promotion [13,14]. For instance, PD-L1 expression critically modulates immune responses, contributing to cardiac allograft tolerance in mice and regulating CD8+ T cell–mediated injury in the heart [15,16]. In humans, PD-L1 expression correlates with biopsy-proven ACR severity and rejection risk [17,18]. Moreover, blocking PD-1 and augmenting its signaling (e.g., with PD-L1.Ig fusion proteins) accelerate rejection and prolong allograft survival, respectively [19,20]. PD-1–deficient mice developed severe myocarditis and rapidly rejected cardiac allografts, underscoring the protective role of PD-1 [21]. Notably, patients with solid organ transplants who receive anti–PD-1 cancer therapy manifest acute T cell-mediated graft rejection, highlighting the PD-1/PD-L1 axis as a pivotal mediator of allograft tolerance in humans [22].
- Nonetheless, until recently, PD-1/PD-L1 involvement in human cardiac allograft rejection was not well characterized [23]. Emerging clinical evidence indicates that PD-L1 expression is induced in cardiac allografts during ACR [18]. In a recent study on heart transplant recipients, PD-L1 was upregulated in cardiomyocytes in proportion to ACR severity [18], suggesting an intrinsic counter-regulatory mechanism; nevertheless, PD-L1 upregulation alone may not fully prevent allograft damage [24]. While characterization of PD-1/PD-L1 expression in human cardiac allografts has begun in other populations, a comprehensive understanding of PD-1/PD-L1 status and its precise correlation with rejection severity—particularly within specific cohorts such as East Asians—remains largely unknown. Thus, we investigated PD-1 and PD-L1 expression in EMB samples from heart TPL patients, aiming to clarify the role of this checkpoint pathway in the ACR of human cardiac allografts and to explore the potential of therapeutic modulation of PD-1/PD-L1 in this setting.
INTRODUCTION
- Patients
- Overall, 78 patients who underwent heart TPL and were followed up at Seoul National University Hospital (SNUH; Seoul, Republic of Korea) between March 2010 and May 2015 were included in this study to evaluate their treatment response and long-term survival. Acute rejection routine surveillance was performed in all patients for at least 1 year by protocol EMBs. Briefly, patients were monitored at the cardiac center for the first 2 weeks following TPL and followed up with routine clinical visits every 3 months for 1-year post-TPL. Protocol EMBs were performed 10 days after TPL with subsequent biopsies, depending on whether the steroids were to be weaned off or reduced to a maintenance dose. Clinical data were obtained from the medical records, and all EMB pathological materials were reviewed.
- Histological EMB evaluation
- Histology-based rejection grading for cardiac allografts was performed according to the 2004 ISHLT guidelines [8] as follows: (1) grade 0R, no rejection; (2) grade 1R, mild rejection—interstitial and/or perivascular infiltrate with up to 1 focus of myocyte injury; (3) grade 2R, moderate rejection—two or more foci of infiltrate with associated myocyte injury; and (4) grade 3R, severe rejection—a diffuse process of myocyte injury in which distinct foci are difficult to delineate. The most severe grade observed across multiple sections was recorded as the ACR grade of the cardiac allograft. In clinical practice, ISHLT grades 2R and 3R are considered high-grade ACR and require treatment [8]. Thus, in this study, the correlation between ACR and clinicopathological features was compared between patients with grades 0R/1R and those with grades 2R/3R.
- Immunohistochemistry
- Immunohistochemistry (IHC) was performed using a rabbit anti–PD-L1 (E1L3N) XP monoclonal antibody (mAb) (Cell Signaling Technology, Danvers, MA, USA), a mouse anti–PD-1 mAb (clone MRQ-22, Cell Marque, Rocklin, CA, USA), a mouse anti-CD4 mAb (clone 4B12, Thermo Fisher Scientific, Rockford, IL, USA), and the Benchmark XT autostainer (Ventana Medical Systems, Tucson, AZ, USA). IHC for CD8 (rabbit IgG, clone SP16, Thermo Fisher Scientific) was performed using a Bond-Max automated immunostainer (Leica Microsystems, Melbourne, Australia). All EMB tissues were subjected to PD-L1 expression immunohistochemical analysis. IHC for PD-1, CD4, and CD8 was performed on EMB samples, demonstrating a grade 1R-3R ACR because ACR grade 0 EMB tissues were infiltrated by few or no lymphocytes.
- PD-L1 IHC was evaluated based on membranous and/or cytoplasmic staining intensity and proportion in cardiac tissue, including myocardiocytes, endothelial cells, and immune cells, and scored as follows: 0, no staining; 1, weak intensity in any proportion or moderate intensity in 5% of total area in EMB tissue; 2, moderate intensity in ≥5% of total area in EMB tissue; and 3, strong intensity in ≥5% of total area in EMB tissue. Patients with PD-L1 IHC scores of 2 or 3 were considered positive for PD-L1 expression. In addition, a PD-L1 H-score was calculated for each biopsy to capture the overall burden of PD-L1 expression. For this purpose, we estimated the percentage of PD-L1–positive cells in the entire biopsy section (0%–100%) and recorded the predominant staining intensity (0–3). The H-score was then derived as intensity × percentage of positive cells, yielding a value between 0 and 300. This continuous H-score was used particularly to evaluate longitudinal changes in PD-L1 expression in serial biopsies.
- CD4+, CD8+, and PD-1+ lymphocyte enumeration
- For each specimen, two representative high-power fields (HPFs, 400× magnification) were selected from the areas infiltrated by the highest number of lymphocytes after examination of hematoxylin and eosin–stained slides under a microscope. Identical areas were taken from CD4-, CD8, and PD-1 immunostained slides, and CD4+, CD8+, and PD-1+ lymphocytes were manually counted. The data are presented as the mean number of cells per HPF.
- Statistical analysis
- All statistical analyses were performed using the SPSS software ver. 23 (IBM Corp., New York, NY, USA). Comparisons between variables were performed using the χ2 test, Fisher’s exact test, or Student’s t-test. Post-transplant survival (PS) was measured from the date of heart TPL to the date of death from any cause. Survival analysis was performed using the Kaplan-Meier method with the log-rank test. Two-sided p-values < .05 were considered statistically significant.
MATERIALS AND METHODS
- Patient characteristics
- The characteristics of the patients with heart TPL are summarized in Table 1. The median patient age was 54 years, ranging from 6 to 76 years. Among others, the underlying diseases leading to cardiac TPL included dilated cardiomyopathy (CMP; n = 43), ischemic CMP (n = 16), congestive heart failure (n = 7), and amyloidosis (n = 4). The body mass index was 20.9 ± 4.1 (mean ± standard deviation [SD]), and 23 patients (29.5%) exhibited diabetes mellitus. A ventilator was applied to two patients before heart TPL. The ejection fraction measured 25.3 ± 12.1 and 63.9 ± 6.6 before and after heart TPL, respectively. Total ischemic time and the pulmonary artery pressure before heart TPL were 182.9 ± 56.5 minutes and 47.1 ± 14.1 mmHg, respectively. The median follow-up duration for all patients was 124.5 months.
- Correlation between ACR and clinical features
- EMB histopathological evaluation revealed ACR grade 0R, 1R, 2R, and 3R in 25 (32.0%), 37 (47.4%), 12 (15.5%), and four (5.1%) of the 78 patients with heart TPL, respectively. ACR of ≥grade 2R was observed in 20.6% (16/78) of the patients during the follow-up period. The correlations between the clinical features of the patients and a high-grade ACR are summarized in Table 2. No clinical factors were significantly associated with high-grade ACR in patients with cardiac TPL.
- Analysis of PD-L1 expression and PD-1+, CD4+, and CD8+ lymphocytes according to ACR status
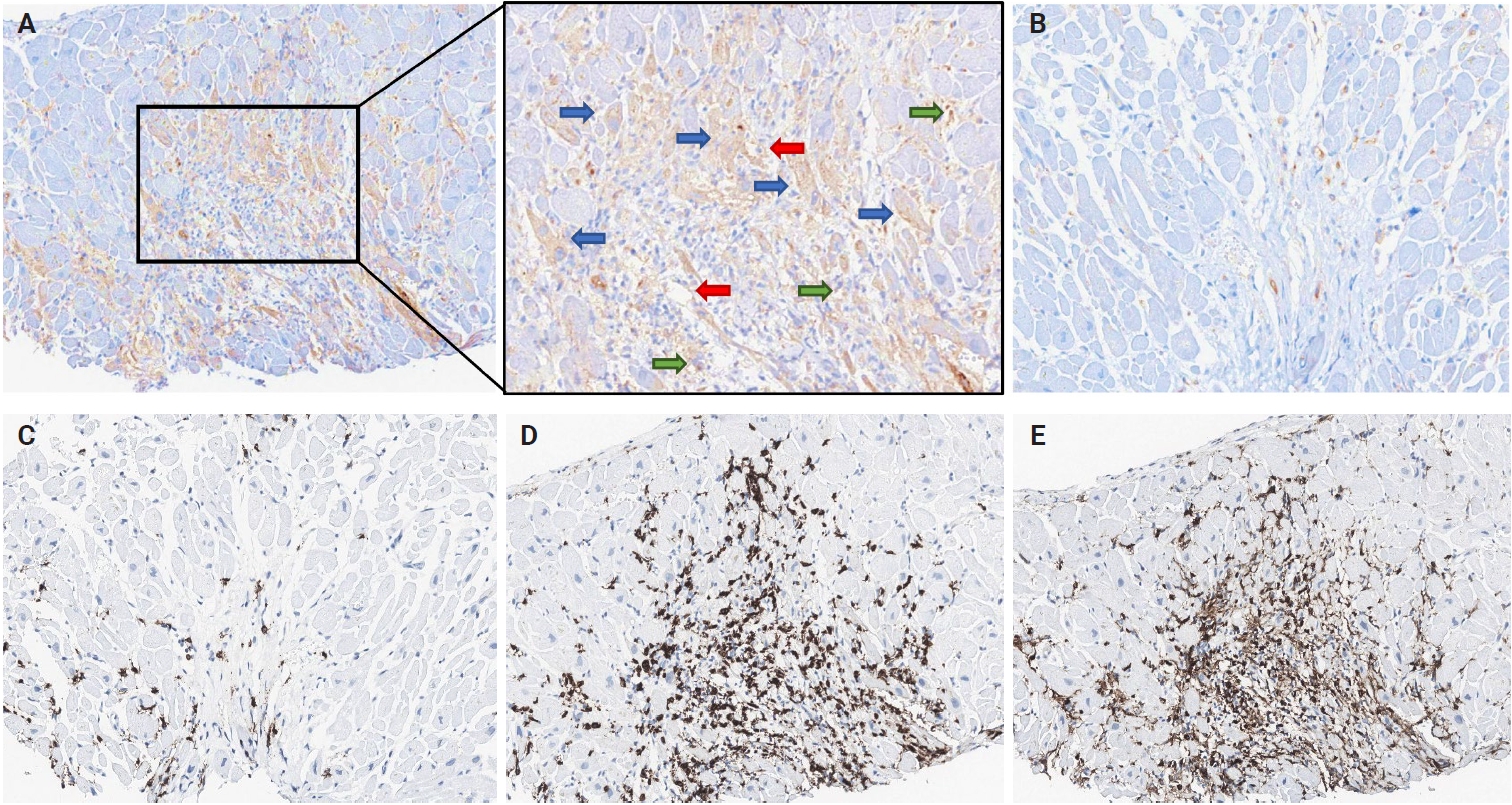
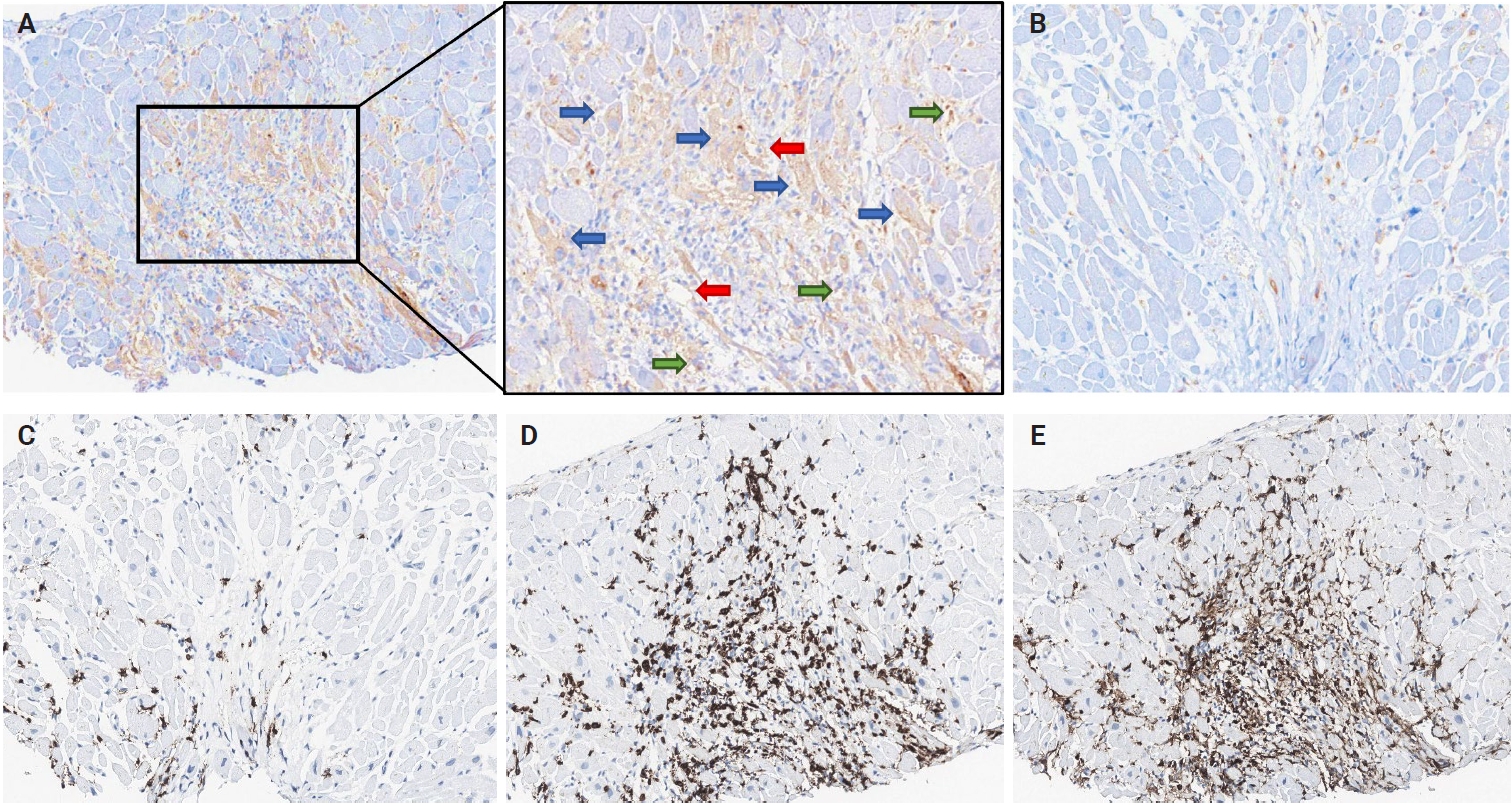
- Representative IHC images of PD-L1, PD-1, CD4, and CD8 in EMBs are displayed in Fig. 1. PD-L1–expressing cells in cardiac allografts were mostly macrophages or endothelial cells at the immune cell infiltration site in the myocardium. Additionally, PD-L1 expression has been occasionally observed in cardiomyocytes. Overall, PD-L1 expression in cardiac allografts was positive in 25.6% (20/78) of patients with heart TPL. The number of PD-1+, CD4+, and CD8+ cells per HPF, evaluated in 39 of the 78 patients, excluding those with ACR grade 0R, was 18.5 ± 33.3, 116.4 ± 117.8, and 105.2 ± 89.2 (mean ± SD), respectively.
- The correlations between high-grade ACR and pathological features, including PD-L1 expression and lymphocyte infiltration, are summarized in Table 2. EMB tissues with high-grade ACR tended to be infiltrated by higher numbers of CD4+ and CD8+ lymphocytes. Notably, patients with high-grade ACR exhibited significantly higher PD-L1 expression than did those without high-grade ACR (62.5% vs. 16.1%, p < .001).
- The correlations between PD-L1 expression and the clinicopathological features of patients with heart TPL are summarized in Table 3. There was no significant association between PD-L1 expression and clinical features. Conversely, EMB tissues with PD-L1 positivity were infiltrated with a higher number of CD4+ and CD8+ lymphocytes (p = .025 and p = .059, respectively) (Table 3). Moreover, the number of PD-1+ lymphocytes was higher in the PD-L1–positive group (35.6 ± 58.7) than in the PD-L1–negative group (14.8 ± 24.7), but did not reach statistical significance (p = .390) (Table 3). These findings suggest that the PD-1/PD-L1 pathway may be involved in ACR in human heart transplants.
- PD-L1 expression evaluation in serial EMBs with ACR in heart TPL
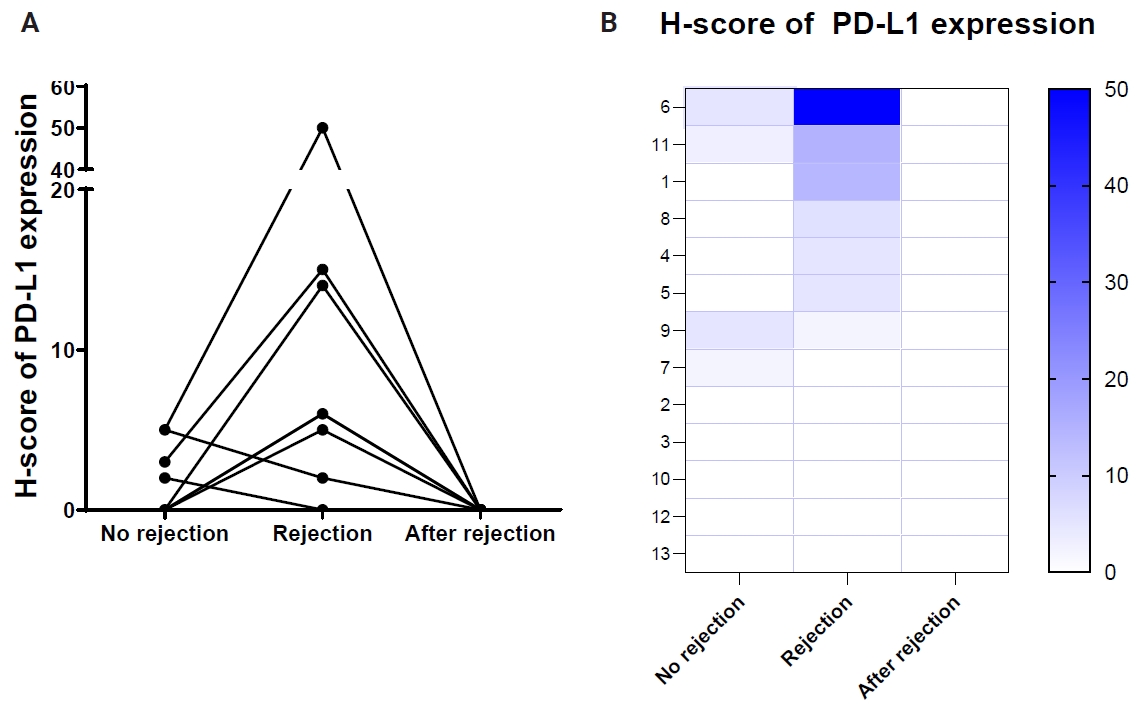
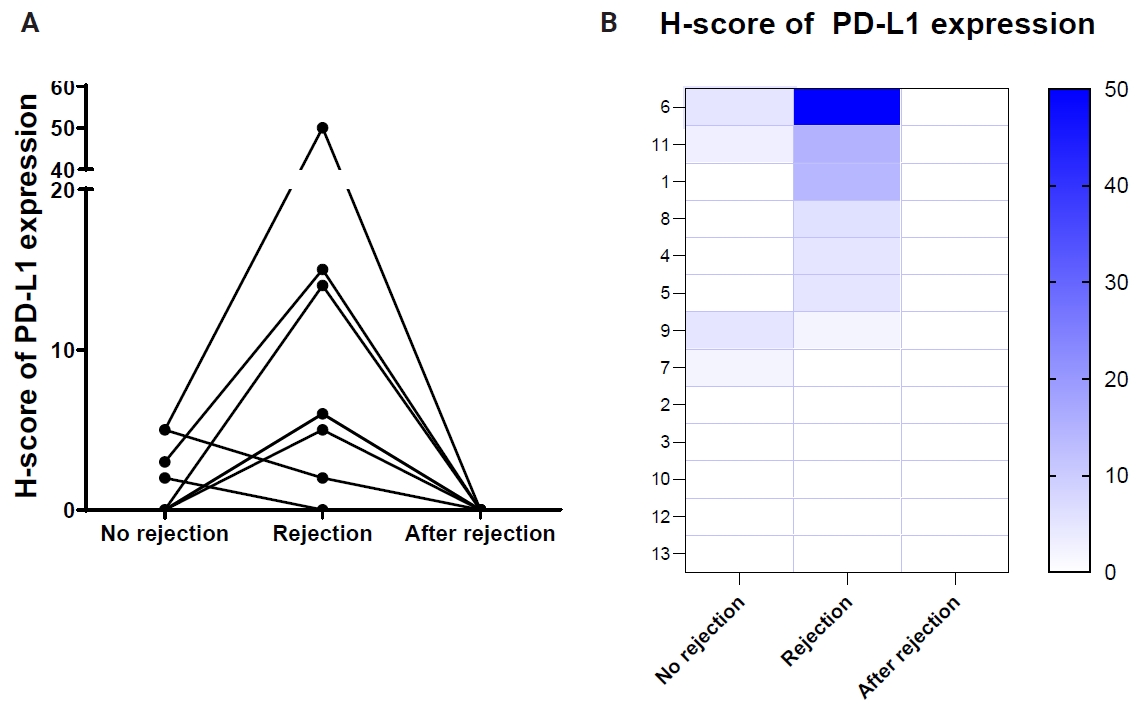
- Serial EMB samples from patients who developed ACR of more than grade 2R on follow-up were available in 13 patients (Table 4). In six patients, the H-score of PD-L1 expression in EMBs increased in cardiac allografts after developing high-grade ACR compared to that in previous EMB tissues without high-grade ACR (Fig. 2). Conversely, five cases demonstrated no PD-L1 H-score differences in EMBs taken at the time without high-grade ACR and at the time of high-grade ACR, and two cases revealed a slight decrease in the PD-L1 H-scores in EMBs with high-grade ACR compared to previous EMBs without ACR. Overall, PD-L1 expression levels significantly increased in EMBs with high-grade ACR compared with previous EMBs without ACR (p =.001) (Fig. 2) and subsequently decreased in follow-up EMBs after ACR subsided (Fig. 2).
- Prognostic significance of PD-1/PD-L1 pathway status in heart TPL
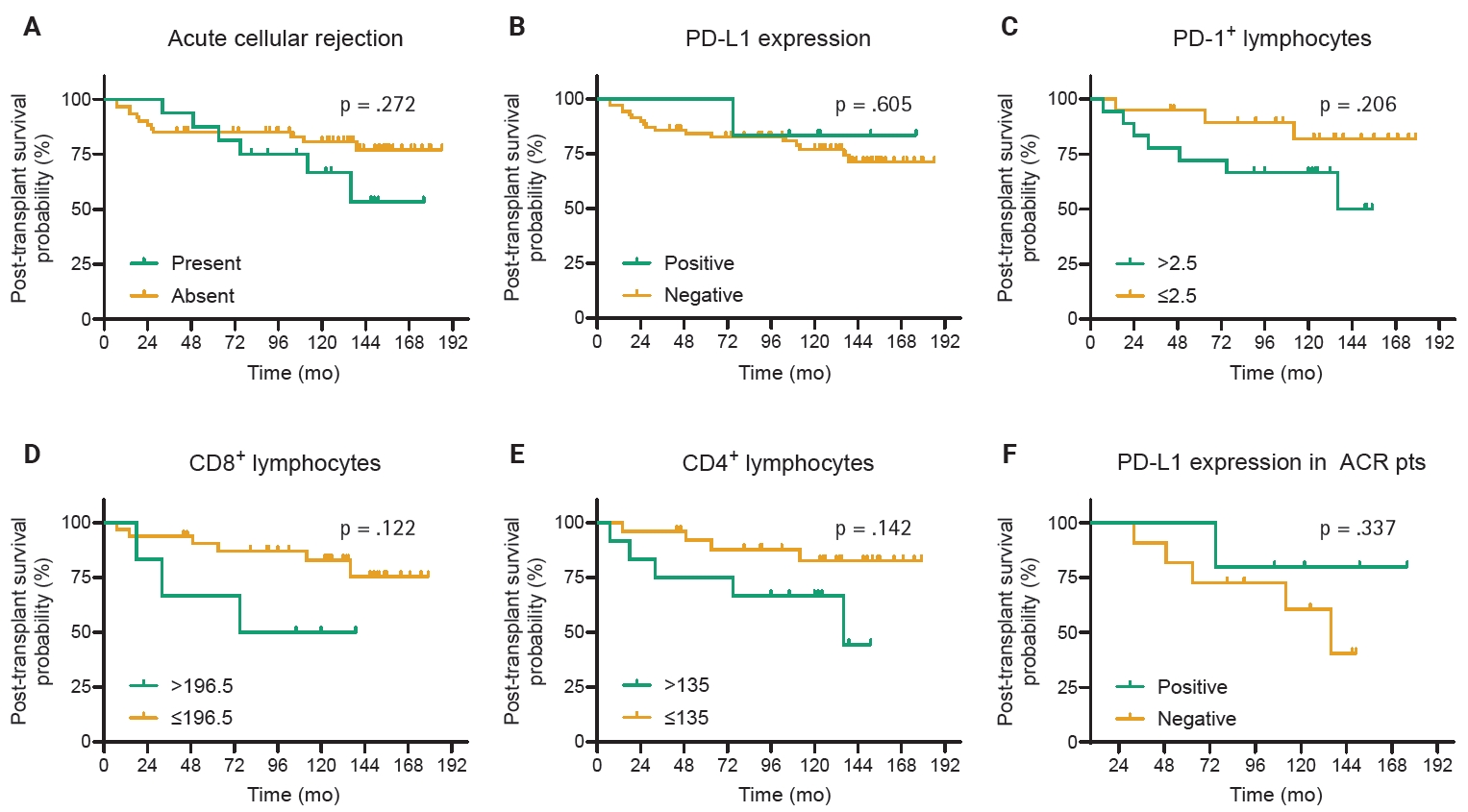
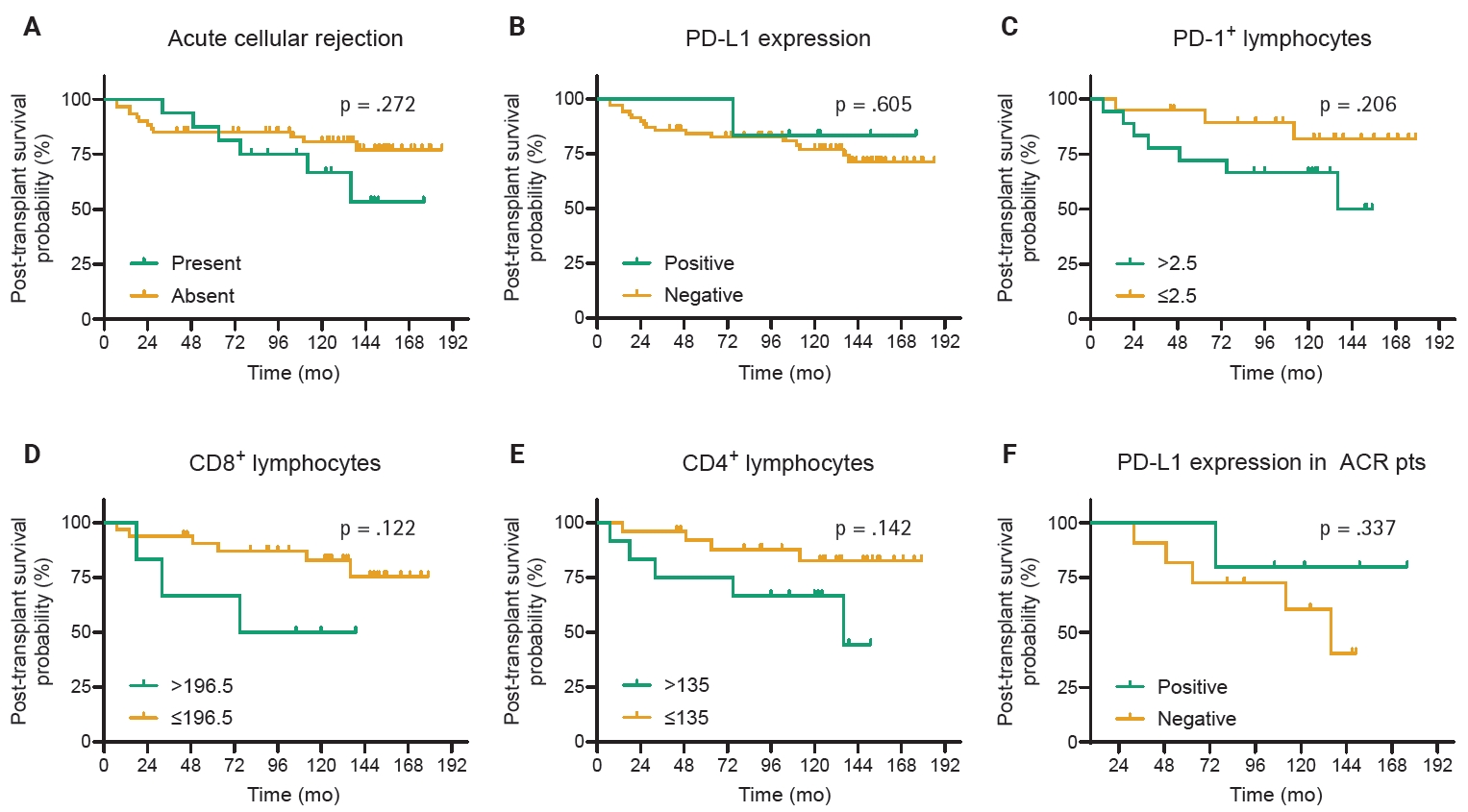
- Univariate survival analysis revealed that no clinical factors were associated with survival in patients with heart TPL. The episodes of high-grade ACR, PD-L1 expression, and the number of PD-1+, CD4+, or CD8+ lymphocytes in the EMB tissues were not associated with PS (Fig. 3).
RESULTS
- To the best of our knowledge, this is the first study in East Asia to examine PD-1 and PD-L1 expression in EMBs from heart transplant recipients, including one of the largest cohorts (78 patients) reported to date on this topic [17,18,25]. PD-L1 was significantly upregulated in human cardiac allografts during high-grade ACR, with a concomitant increase in PD-1-positive T lymphocyte infiltration, thereby suggesting that the PD-1/PD-L1 immune checkpoint pathway is actively involved in T-cell-mediated rejection regulation during heart TPL. In addition to prior work conducted primarily in non–East Asian populations [17,18,25], our study provides additional data on PD-1/PD-L1 expression in an East Asian heart transplant cohort, including recipients with serially evaluated endomyocardial biopsies.
- This observation is consistent with evidence from various experimental models demonstrating that the PD-1/PD-L1 axis is crucial for dampening alloimmune injury [13–16]. In murine transplant models, the absence of PD-1 or PD-L1 accelerates cardiac allograft rejection and severe myocarditis, demonstrating the important role of this pathway in maintaining peripheral tolerance [15,16,21]. Specifically, donor tissue-derived PD-L1 has been identified as a key mediator of graft acceptance, and cardiac allografts in mice lacking PD-L1 on the parenchymal cells, or specifically on the endothelium, experience more aggressive T-cell infiltration and damage [16,21]. Moreover, pro-inflammatory cytokines, such as interferon-γ, can strongly induce PD-L1 on graft cells, suggesting that the alloimmune response itself induces this protective checkpoint upregulation [16]. Accordingly, PD-1 signaling blockade exacerbates rejection in animal models, while augmenting the pathway (e.g., with PD-L1.Ig fusion proteins) leads to graft protection and prolonged graft survival [19,20]. Collectively, these preclinical findings support our human data and indicate that PD-L1 induction in cardiac allografts functions as a counter-regulatory mechanism to mitigate acute cellular rejection.
- Unlike most previous studies that analyzed cross-sectional biopsy samples, our study evaluated serial EMBs from individual patients before, during, and after rejection episodes. This longitudinal approach revealed that PD-L1 expression was dynamically upregulated in allograft tissues early in high-grade ACR development and declined after the rejection resolution, paralleling PD-1 T-cell infiltration changes. Therefore, PD-L1 overexpression may be a transient adaptive mechanism that suppresses T-cell-mediated tissue injury. Consistently, Choudhary et al. [18] reported that PD-L1 expression in transplanted hearts correlated with rejection severity and decreased after successful anti-rejection therapy. Furthermore, Peyster et al. [17] found that patients who never experienced rejection had higher baseline proportions of PD-L1+ and FoxP3+ cells in their allografts than did those who developed ACR. Collectively, these studies support the concept that PD-L1 contributes to an immune-tolerant graft microenvironment, whereas diminished PD-L1 expression may predispose patients to rejection.
- Another unique aspect of our study is the compartmental analysis of PD-L1 expression within the allograft and the quantitative assessment of PD-L1 on graft cardiomyocytes, endothelial cells, and infiltrating immune cells. Through this comprehensive approach, we found that all three compartments upregulated PD-L1 during ACR, reflecting a concerted multicellular checkpoint response within the cardiac graft, which is consistent with the findings of Bracamonte-Baran et al. [25], who demonstrated that PD-L1 expression in non-hematopoietic cardiac cells (particularly in the endothelium) is crucial for modulating T cell infiltration in heart transplants. In their study, higher PD-L1 levels in graft endothelial cells were associated with significantly reduced CD8+ T-cell infiltrates, whereas endothelial PD-L1 loss was associated with dense CD8+ T-cell infiltration and more severe rejection. Furthermore, in a mouse model, they demonstrated that the lack of PD-L1 in cardiac endothelial cells led to accelerated rejection, implicating graft-expressed PD-L1 in the protection against alloimmune injury. These results reinforce our human data and earlier murine evidence, underscoring the fact that PD-L1 upregulation by grafted parenchymal cells actively suppresses alloreactive T cells. Variability in the dominant site of PD-L1 expression (endothelial vs. parenchymal) may explain differences in graft resilience to immune attacks.
- Despite the overall trend of PD-L1 induction in rejection, we observed heterogeneity. Among the 13 patients with serial EMBs available, seven (five with no change and two with a slight decrease) did not exhibit appreciable PD-L1 upregulation at the time of high-grade ACR compared with their prior EMBs. Notably, all these patients still responded to anti-rejection therapy and survived the episode, indicating that the absence of PD-L1 upregulation did not preclude successful rejection control. Therefore, when the PD-1/PD-L1 axis is not engaged, alternative immune-regulatory pathways may compensate; conversely, in some fulminant rejections, the immune response may outpace PD-L1 induction. Indeed, prior transplant biopsy analyses have reported that although PD-L1 is frequently induced during rejection, its presence alone is not always sufficient to prevent ongoing tissue damage [16,18]. Thus, PD-L1 likely functions as a multiple redundant checkpoint that collectively modulates alloimmunity [23], implying that other co-inhibitory receptors (e.g., cytotoxic T-lymphocyte–associated protein 4, T-cell Immunoglobulin and Mucin-domain containing protein 3, and lymphocyte-activation gene 3) may be concurrently involved in restraining rejection, especially in cases where PD-L1 expression remains low [23]. Our results underscore this immune heterogeneity and the need to explore additional inhibitory pathways in transplant rejection, as the therapeutic targeting of PD-1/PD-L1 alone may not uniformly prevent rejection in all settings.
- In addition to this heterogeneity within cellular rejection, another important consideration is antibody-mediated rejection (AMR), which was beyond the scope of the present analysis. In the present study, we focused on T cell–mediated acute cellular rejection and did not specifically evaluate pathological or serological features of AMR. To our knowledge, human data directly linking PD-L1 expression to AMR in heart TPL are extremely limited. A small single-center pilot study that analyzed PD-L1 expression in endomyocardial biopsies from patients with AMR, ACR, and no rejection did not find a clear difference in PD-L1 levels between rejection types, although cases with overlapping AMR and ACR showed higher PD-L1 expression and tended to exhibit faster histological resolution of rejection [26]. These preliminary observations, together with prior work mainly focused on cellular rejection and chronic allograft injury, underline the need for future studies that integrate PD-L1 assessment with detailed AMR phenotyping to clarify whether PD-L1 plays a distinct role in humoral rejection.
- Additional limitations of our study should be acknowledged. In this study, immunostaining for PD-1, CD4, and CD8 was restricted to EMBs with ACR grade ≥1R. Consequently, we could not systematically quantify low-level baseline immune cell infiltration in grade 0R allografts, and future work incorporating these markers across all rejection grades, including 0R, will be needed to more fully define the spectrum of immune activation within the cardiac allograft.
- These findings carry potential clinical implications for graft monitoring and risk stratification. As PD-L1 expression in allografts correlates closely with rejection activity, it could be investigated as a biomarker to aid in ACR diagnosis or prediction. For instance, Novysedlak et al. [27] found that PD-L1 (combined with platelet endothelial cell adhesion molecule-1) was significantly elevated in lung transplant biopsies with ACR and proposed PD-L1 as a useful diagnostic marker for rejection. Similarly, in heart TPL, tracking PD-L1 levels in protocol biopsies or even non-invasively via novel imaging modalities might help identify patients at a higher rejection risk [28,29]. A graft that fails to upregulate PD-L1 in the face of immune stress or exhibits a PD-L1 expression abrupt decline may be more susceptible to uncontrolled T-cell attack. This notion is corroborated by the observation that patients whose allografts eventually experienced rejection tended to have lower baseline PD-L1+ cell fractions and more pronounced CD8+ T cell infiltrates, whereas grafts maintaining high PD-L1 and plentiful regulatory immune cells were more likely to remain quiescent [17]. Incorporating PD-L1 assessment, potentially alongside other immunoregulatory markers such as FoxP3, into post-transplant surveillance may enhance subclinical rejection detection and predict which patients are at the greatest risk for severe ACR. The PD-L1 expression increase observed during ACR may represent a compensatory immunoregulatory response designed to attenuate tissue-damaging lymphocyte activity, highlighting its biological importance.
- In our cohort with long-term follow-up (median 124.5 months), neither high-grade ACR, PD-L1 expression status, nor the densities of PD-1⁺, CD8⁺, or CD4⁺ lymphocytes were significantly associated with all-cause post-TPL survival, indicating that even after more than a decade of observation, these immunological markers did not predict long-term outcomes; specifically, their expression patterns likely reflect active immune regulation during acute rejection rather than survival determinants. The absence of an association may reflect the effective rescue of rejection episodes with immunosuppression, mitigating long-term effects, and the influence of other factors, such as cardiac allograft vasculopathy and infection, may outweigh the contribution of PD-L1–related immune regulation. Taken together, these findings suggest that PD-L1 expression should be interpreted primarily as a marker of ongoing immunological counterresponses rather than as a prognostic biomarker for graft survival.
- On the therapeutic front, our data and recent studies provide a rationale for targeting the PD-1/PD-L1 pathway to promote transplant tolerance [23,30]. Augmenting this checkpoint—for example, using PD-1 agonists or strategies to boost PD-L1 expression in the graft—could reinforce the inherent protection of the allograft against T-cell attacks. Experimental approaches, such as PD-L1.Ig fusion proteins (soluble PD-1 agonists), have already prolonged cardiac allograft survival in preclinical models [19], and methods to increase PD-L1 expression in donor tissues via gene therapy or pharmacologic induction are being explored [30]. Any such intervention must be balanced against risks of oversuppression of the immune system (e.g., infection or malignancy). Our findings highlight a cautionary note: therapeutic blockade of the PD-1/PD-L1 axis, as employed in cancer immunotherapy, may precipitate rejection in transplant recipients. Indeed, documented cases demonstrate abrupt acute rejection of transplanted organs in patients receiving PD-1 inhibitors for malignancy [22,31]; moreover, a recent pharmacovigilance analysis further underscored this risk, reporting a high incidence of allograft rejection following checkpoint inhibitor therapy [32]. Collectively, these observations indicate that intact PD-1/PD-L1 signaling is critical for maintaining immune tolerance to transplanted organs in humans, because disrupting this pathway can unleash alloreactive T-cell responses and precipitate rejection [22]. Thus, while modulating the PD-1/PD-L1 pathway offers promising opportunities to improve graft outcomes, caution is warranted in transplant patients.
- Ultimately, elucidating the patterns and effects of PD-1/PD-L1 signaling in heart transplantation provides a foundation for improved rejection monitoring and the development of targeted immunotherapies that enhance graft tolerance without undue risk.
DISCUSSION
Ethics Statement
This study was performed according to the recommendations of the World Medical Association Declaration of Helsinki and was approved by the Institutional Review Board of SNUH (IRB No. H-1412-106-634). Formal written informed consent was not required with a waiver by the appropriate IRB and/or national research ethics committee.
Availability of Data and Material
The datasets generated and analyzed during the current study are not publicly available due to ethical and privacy restrictions (they contain information that could compromise participant confidentiality), but are available from the corresponding author upon reasonable request and with appropriate institutional approval.
Code Availability
Not applicable.
Author Contributions
Conceptualization: JK, DHC, YKJ. Data curation: JK. Formal analysis: JK, JY. Investigation: JY, JK. Methodology: JK. Project administration: JK. Resources: JK, DHC, YKJ. Supervision: JK, DHC, YKJ. Validation: JY, JK. Visualization: JY, JK. Writing—original draft: JY, JK. Writing—review & editing: JK, JY, YKJ, DHC. Approval of final manuscript: all authors
Conflicts of Interest
The authors declare that they have no potential conflicts of interest.
Funding Statement
This research was supported by grants of the Korea Health Technology R&D Project through the Korea Health Industry Development Institute (KHIDI), funded by the Ministry of Health and Welfare, Republic of Korea (RS-2024-00409089).
| High-grade acute cellular rejectiona | |||
|---|---|---|---|
| Absent (n = 62) | Present (n = 16) | p-value | |
| Age (yr) | |||
| <60 | 38 (77.6) | 11 (22.4) | .773 |
| ≥60 | 24 (82.8) | 5 (17.2) | |
| Sex | |||
| Male | 48 (81.4) | 11 (18.6) | .520 |
| Female | 14 (73.7) | 5 (26.3) | |
| CAD | |||
| Absent | 50 (79.4) | 13 (20.6) | >.99 |
| Present | 12 (80.0) | 3 (20.0) | |
| Diabetes | |||
| Absent | 45 (81.8) | 10 (18.2) | .540 |
| Present | 17 (73.9) | 6 (26.1) | |
| Hypertension | |||
| Absent | 45 (76.3) | 14 (23.7) | .330 |
| Present | 17 (89.5) | 2 (10.5) | |
| Smoking | |||
| Never | 43 (81.1) | 10 (18.9) | .873 |
| Ever | 19 (76.0) | 6 (24.0) | |
| Ventilator | |||
| Not applied | 60 (78.9) | 16 (21.1) | .334 |
| Applied | 2 (100) | 0 | |
| Dialysis | |||
| Never | 52 (77.6) | 15 (22.4) | .273 |
| Ever | 10 (90.9) | 1 (9.1) | |
| Body mass index (kg/m2) | 21.0 ± 4.1 | 20.7 ± 4.2 | .828 |
| Preoperative EF (%) | 25.5 ± 12.6 | 24.5 ± 10.3 | .764 |
| Postoperative EF (%) | 64.1 ± 6.4 | 62.9 ± 7.5 | .548 |
| Ischemic time (min) | 184.2 ± 58.7 | 177.6 ± 48.6 | .719 |
| Pulmonary artery pressure (mmHg) | 46.4 ± 14.4 | 50.6 ± 12.8 | .379 |
| Post-transplant survival (mo) | 32.6 ± 20.0 | 34.0 ± 19.4 | .806 |
| PD-L1 expression | |||
| Negative | 52 (89.7) | 6 (10.3) | <.001 |
| Positive | 10 (50.0) | 10 (50.0) | |
| PD-1+ lymphocytes (number/HPF) | 11.3 ± 20.5 | 31.3 ± 46.7 | .148 |
| CD8+ lymphocytes (number/HPF) | 89.8 ± 78.3 | 129.8 ± 102.3 | .175 |
| CD4+ lymphocytes (number/HPF) | 82.7 ± 72.4 | 170.5 ± 154.9 | .054 |
Values are presented as number (%) or mean ± SD.
PD-1, programmed cell death-1; PD-L1, programmed death-ligand 1; CAD, coronary artery disease; EF, ejection fraction; HPF, high power field; SD, standard deviation.
aCases with International Society for Heart and Lung Transplantation (ISHLT) grade 2R and 3R were considered to have high-grade acute cellular rejection.
- 1. de Jonge N, Kirkels JH, Klopping C, et al. Guidelines for heart transplantation. Neth Heart J 2008; 16: 79-87. ArticlePubMedPMCPDF
- 2. Lund LH, Edwards LB, Kucheryavaya AY, et al. The registry of the International Society for Heart and Lung Transplantation: thirty-first official adult heart transplant report--2014; focus theme: retransplantation. J Heart Lung Transplant 2014; 33: 996-1008. ArticlePubMed
- 3. Soderlund C, Ohman J, Nilsson J, et al. Acute cellular rejection the first year after heart transplantation and its impact on survival: a single-centre retrospective study at Skane University Hospital in Lund 1988-2010. Transpl Int 2014; 27: 482-92. ArticlePubMed
- 4. Sinphurmsukskul S, Ariyachaipanich A, Siwamogsatham S, et al. Endomyocardial biopsy and prevalence of acute cellular rejection in heart transplantation. Transplant Proc 2021; 53: 318-23. ArticlePubMed
- 5. Velleca A, Shullo MA, Dhital K, et al. The International Society for Heart and Lung Transplantation (ISHLT) guidelines for the care of heart transplant recipients. J Heart Lung Transplant 2023; 42: e1-141.
- 6. Patel JK, Kobashigawa JA. Should we be doing routine biopsy after heart transplantation in a new era of anti-rejection? Curr Opin Cardiol 2006; 21: 127-31. ArticlePubMed
- 7. Billingham ME. Endomyocardial biopsy diagnosis of acute rejection in cardiac allografts. Prog Cardiovasc Dis 1990; 33: 11-8. ArticlePubMed
- 8. Stewart S, Winters GL, Fishbein MC, et al. Revision of the 1990 working formulation for the standardization of nomenclature in the diagnosis of heart rejection. J Heart Lung Transplant 2005; 24: 1710-20. ArticlePubMed
- 9. Francisco LM, Sage PT, Sharpe AH. The PD-1 pathway in tolerance and autoimmunity. Immunol Rev 2010; 236: 219-42. ArticlePubMedPMC
- 10. Li XC, Rothstein DM, Sayegh MH. Costimulatory pathways in transplantation: challenges and new developments. Immunol Rev 2009; 229: 271-93. ArticlePubMed
- 11. Ito T, Ueno T, Clarkson MR, et al. Analysis of the role of negative T cell costimulatory pathways in CD4 and CD8 T cell-mediated alloimmune responses in vivo. J Immunol 2005; 174: 6648-56. ArticlePubMedPDF
- 12. Zou W, Wolchok JD, Chen L. PD-L1 (B7-H1) and PD-1 pathway blockade for cancer therapy: mechanisms, response biomarkers, and combinations. Sci Transl Med 2016; 8: 328rv4.ArticlePubMedPMC
- 13. Sandner SE, Clarkson MR, Salama AD, et al. Role of the programmed death-1 pathway in regulation of alloimmune responses in vivo. J Immunol 2005; 174: 3408-15. ArticlePubMedPDF
- 14. Ma D, Duan W, Li Y, et al. PD-L1 deficiency within islets reduces allograft survival in mice. PLoS One 2016; 11: e0152087. ArticlePubMedPMC
- 15. Riella LV, Watanabe T, Sage PT, et al. Essential role of PDL1 expression on nonhematopoietic donor cells in acquired tolerance to vascularized cardiac allografts. Am J Transplant 2011; 11: 832-40. ArticlePubMed
- 16. Grabie N, Gotsman I, DaCosta R, et al. Endothelial programmed death-1 ligand 1 (PD-L1) regulates CD8+ T-cell mediated injury in the heart. Circulation 2007; 116: 2062-71. ArticlePubMed
- 17. Peyster EG, Wang C, Ishola F, et al. In situ immune profiling of heart transplant biopsies improves diagnostic accuracy and rejection risk stratification. JACC Basic Transl Sci 2020; 5: 328-40. ArticlePubMedPMC
- 18. Choudhary A, Brinkley DM, Besharati S, et al. PD-L1 (programmed death ligand 1) as a marker of acute cellular rejection after heart transplantation. Circ Heart Fail 2021; 14: e008563. ArticlePubMedPMC
- 19. Dudler J, Li J, Pagnotta M, Pascual M, von Segesser LK, Vassalli G. Gene transfer of programmed death ligand-1.Ig prolongs cardiac allograft survival. Transplantation 2006; 82: 1733-7. ArticlePubMed
- 20. Ozkaynak E, Wang L, Goodearl A, et al. Programmed death-1 targeting can promote allograft survival. J Immunol 2002; 169: 6546-53. ArticlePubMedPDF
- 21. Wang J, Okazaki I, Yoshida T, et al. PD-1 deficiency results in the development of fatal myocarditis in MRL mice. Int Immunol 2010; 22: 443-52. ArticlePubMed
- 22. Lipson EJ, Bagnasco SM, Moore JJ, et al. Tumor regression and allograft rejection after administration of anti-PD-1. N Engl J Med 2016; 374: 896-8. ArticlePubMedPMC
- 23. Del Bello A, Treiner E. Immune checkpoints in solid organ transplantation. Biology (Basel) 2023; 12: 1358.ArticlePubMedPMC
- 24. Riella LV, Paterson AM, Sharpe AH, Chandraker A. Role of the PD-1 pathway in the immune response. Am J Transplant 2012; 12: 2575-87. ArticlePubMedPMC
- 25. Bracamonte-Baran W, Gilotra NA, Won T, et al. Endothelial stromal PD-L1 (programmed death ligand 1) modulates CD8(+) T-cell infiltration after heart transplantation. Circ Heart Fail 2021; 14: e007982. ArticlePubMedPMC
- 26. Szymanska S, Sobieszczanska-Malek M, Wieckowska-Karkucinska A, Grajkowska W. Tissue expression of ligand to programmed death receptor 1 (PD-L1) in endomyocardial biopsies of patients after heart transplantation: association with allograft rejection, pilot study. World J Surg Surgical Res 2021; 4: 1350.
- 27. Novysedlak R, Balko J, Tavandzis J, et al. Elevated PD-L1 and PECAM-1 as diagnostic biomarkers of acute rejection in lung transplantation. Transpl Int 2024; 37: 13796.ArticlePubMedPMC
- 28. Malih S, Lin W, Tang Z, et al. Noninvasive PET imaging of tumor PD-L1 expression with (64)Cu-labeled Durvalumab. Am J Nucl Med Mol Imaging 2024; 14: 31-40. ArticlePubMedPMC
- 29. Niemeijer AN, Leung D, Huisman MC, et al. Whole body PD-1 and PD-L1 positron emission tomography in patients with non-small-cell lung cancer. Nat Commun 2018; 9: 4664.ArticlePubMedPMCPDF
- 30. Handelsman S, Overbey J, Chen K, Lee J, Haj D, Li Y. PD-L1's role in preventing alloreactive T cell responses following hematopoietic and organ transplant. Cells 2023; 12: 1609.ArticlePubMedPMC
- 31. Owonikoko TK, Kumar M, Yang S, et al. Cardiac allograft rejection as a complication of PD-1 checkpoint blockade for cancer immunotherapy: a case report. Cancer Immunol Immunother 2017; 66: 45-50. ArticlePubMedPMCPDF
- 32. Cui X, Yan C, Xu Y, et al. Allograft rejection following immune checkpoint inhibitors in solid organ transplant recipients: a safety analysis from a literature review and a pharmacovigilance system. Cancer Med 2023; 12: 5181-94. ArticlePubMedPMCPDF
REFERENCES
Figure & Data
References
Citations

 PubReader
PubReader ePub Link
ePub Link-
 Cite this Article
Cite this Article
- Cite this Article
-
- Close
- Download Citation
- Close
- Figure
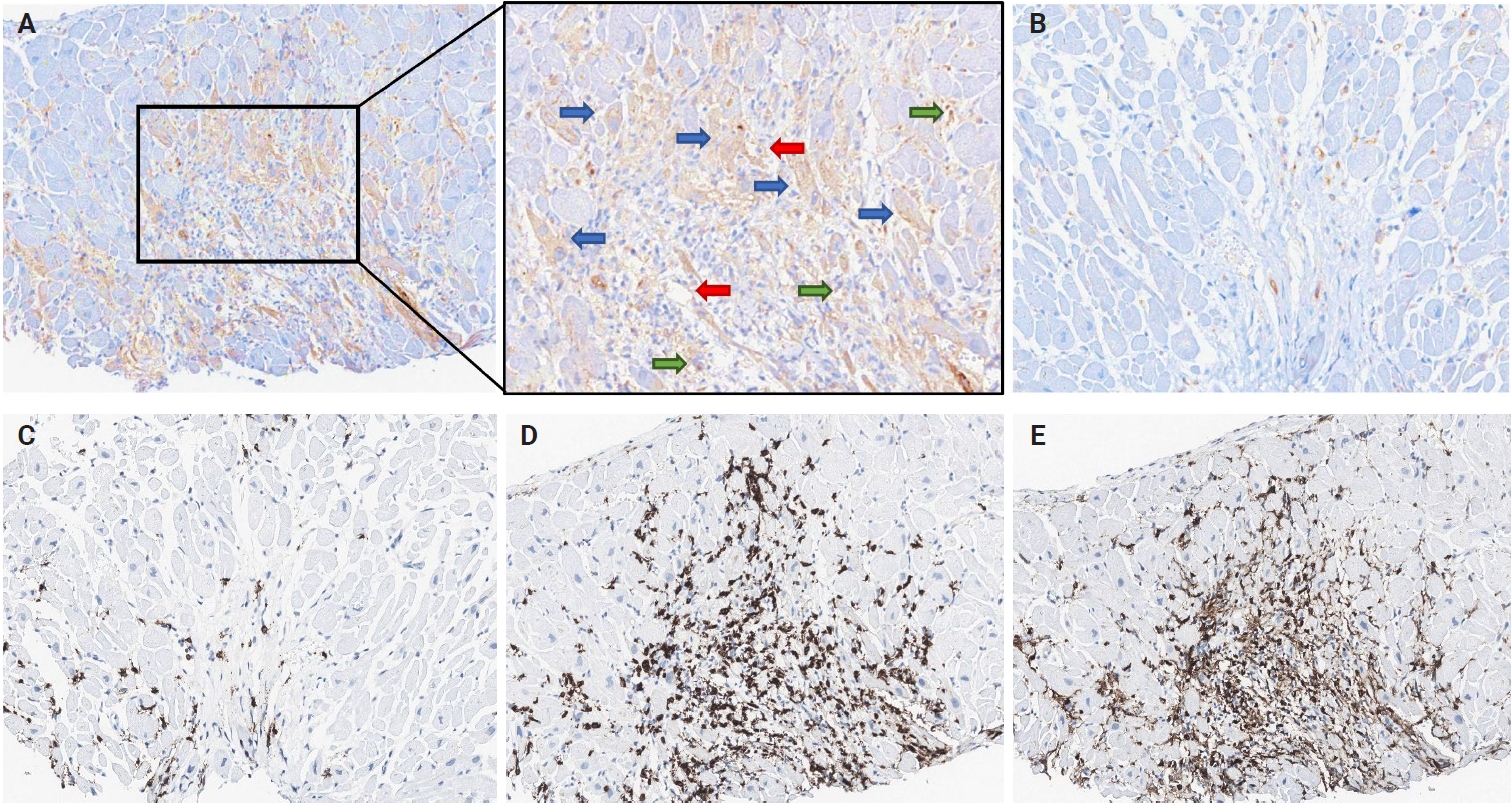
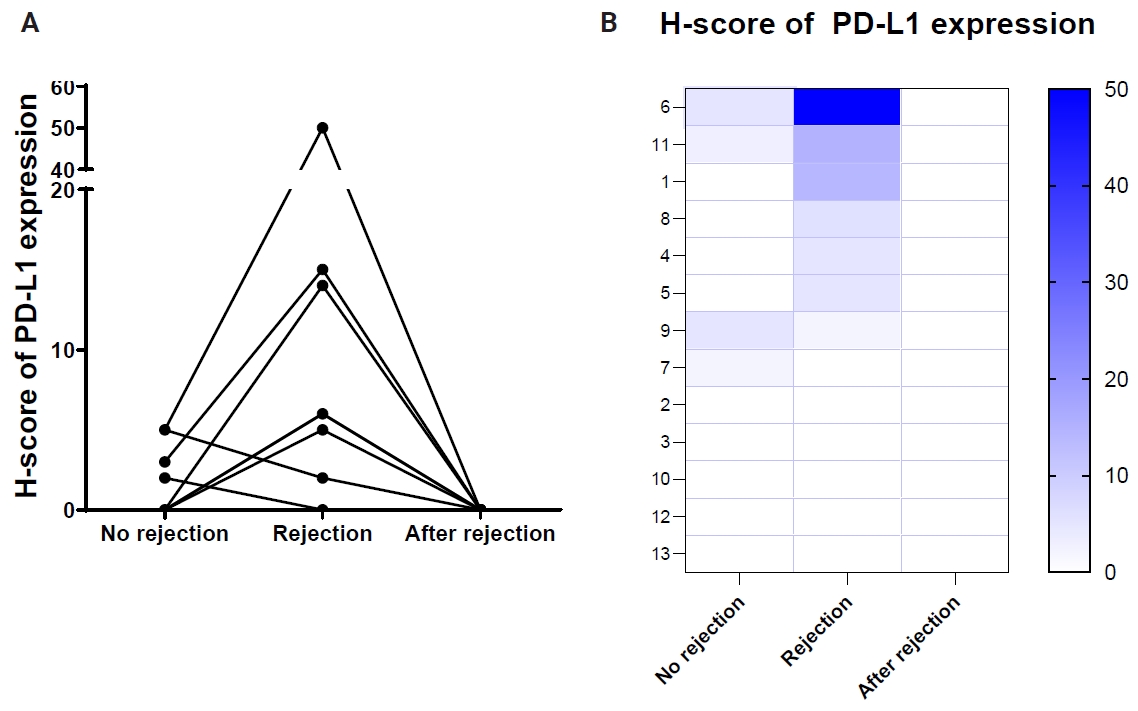
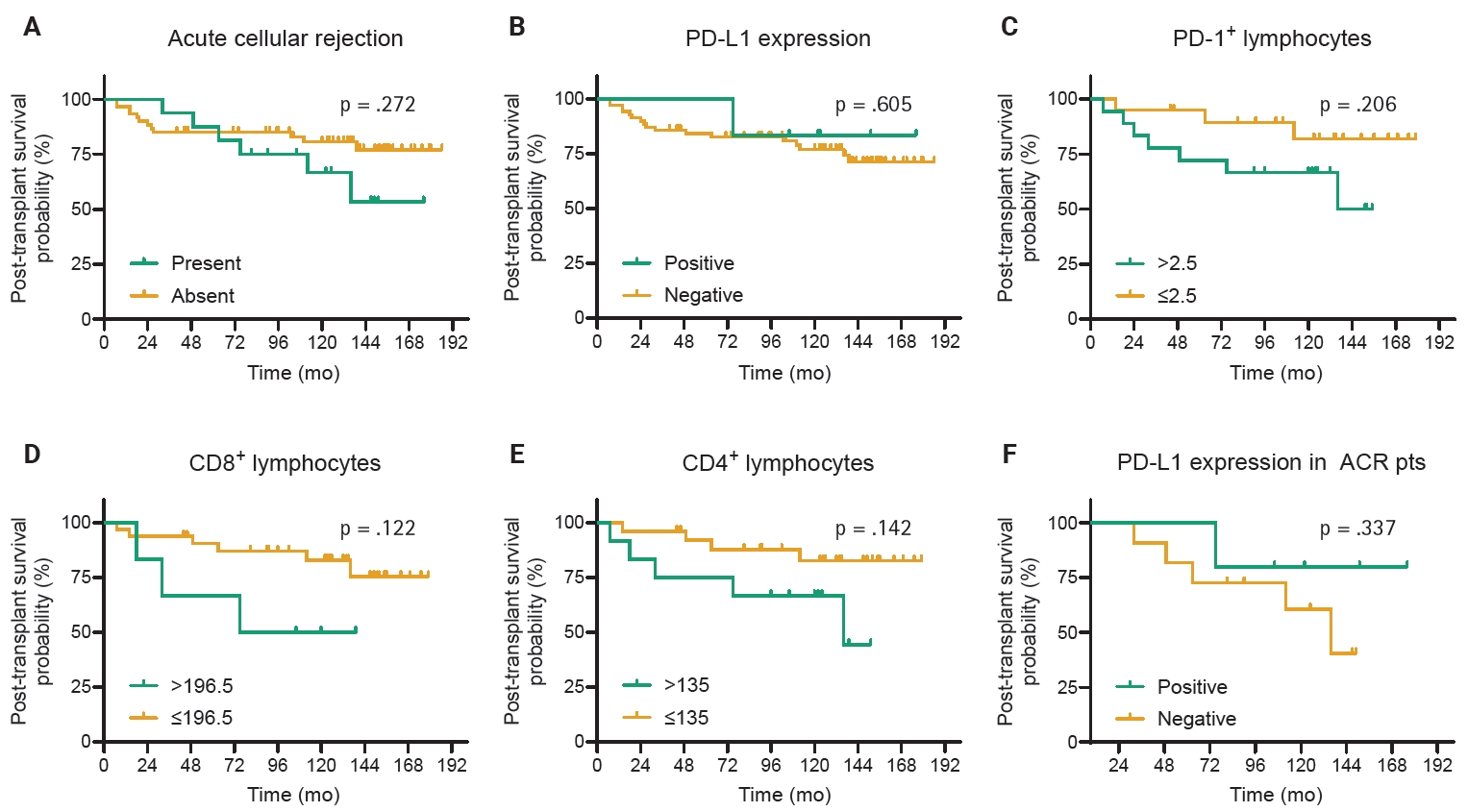
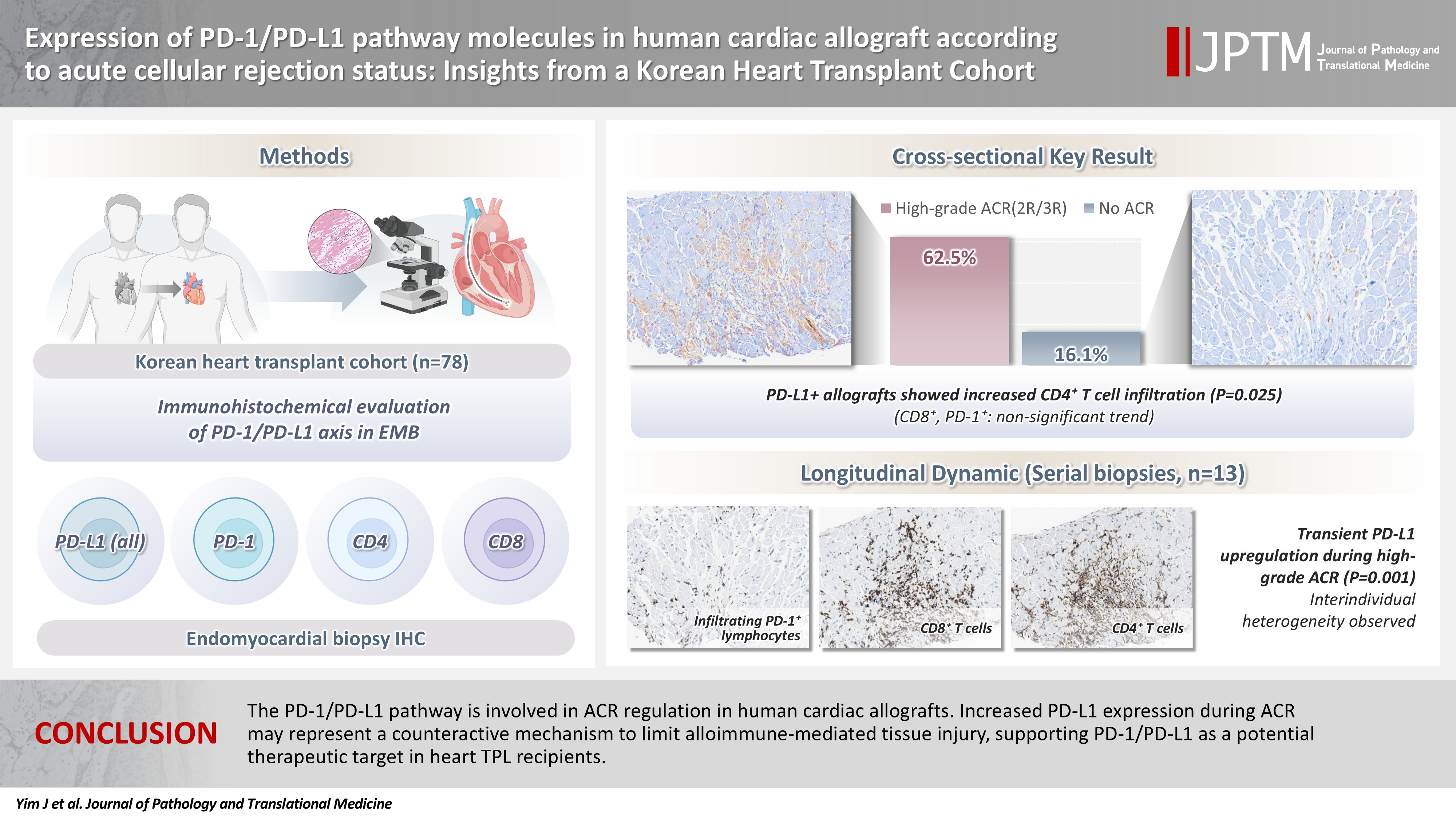
Fig. 1.
Fig. 2.
Fig. 3.
Graphical abstract
| Variable | Value |
|---|---|
| Age (yr) | 47.7 (6–76) |
| Pediatrics | 14 (17.9) |
| Adult | 64 (82.1) |
| Sex | |
| Male | 59 (75.6) |
| Female | 19 (24.4) |
| Diagnosis | |
| CHF | 7 (9.0) |
| Dilated CMP | 43 (55.1) |
| Hypertrophic CMP | 1 (1.3) |
| Ischemic CMP | 16 (20.5) |
| Restrictive CMP | 2 (2.5) |
| Valvulopathy | 2 (2.5) |
| Amyloidosis | 4 (5.2) |
| Myocarditis | 1 (1.3) |
| Endocarditis | 1 (1.3) |
| Heart anomaly | 1 (1.3) |
| Smoking | |
| Never | 53 (67.9) |
| Ever | 25 (32.1) |
| Diabetes mellitus | |
| Absent | 55 (70.5) |
| Present | 23 (29.5) |
| Ventilator | |
| Not applied | 76 (97.5) |
| Applied | 2 (2.5) |
| BMI (kg/m2) | 20.9 (11.3–37.8) |
| Pre-operative EF (%) | 25.3 (12–77) |
| Post-operative EF (%) | 63.9 (49–77) |
| Ischemic time (min) | 182.9 (59–283) |
| Pulmonary artery pressure (mmHg) | 47.1 (25–84) |
| High-grade acute cellular rejection |
|||
|---|---|---|---|
| Absent (n = 62) | Present (n = 16) | p-value | |
| Age (yr) | |||
| <60 | 38 (77.6) | 11 (22.4) | .773 |
| ≥60 | 24 (82.8) | 5 (17.2) | |
| Sex | |||
| Male | 48 (81.4) | 11 (18.6) | .520 |
| Female | 14 (73.7) | 5 (26.3) | |
| CAD | |||
| Absent | 50 (79.4) | 13 (20.6) | >.99 |
| Present | 12 (80.0) | 3 (20.0) | |
| Diabetes | |||
| Absent | 45 (81.8) | 10 (18.2) | .540 |
| Present | 17 (73.9) | 6 (26.1) | |
| Hypertension | |||
| Absent | 45 (76.3) | 14 (23.7) | .330 |
| Present | 17 (89.5) | 2 (10.5) | |
| Smoking | |||
| Never | 43 (81.1) | 10 (18.9) | .873 |
| Ever | 19 (76.0) | 6 (24.0) | |
| Ventilator | |||
| Not applied | 60 (78.9) | 16 (21.1) | .334 |
| Applied | 2 (100) | 0 | |
| Dialysis | |||
| Never | 52 (77.6) | 15 (22.4) | .273 |
| Ever | 10 (90.9) | 1 (9.1) | |
| Body mass index (kg/m2) | 21.0 ± 4.1 | 20.7 ± 4.2 | .828 |
| Preoperative EF (%) | 25.5 ± 12.6 | 24.5 ± 10.3 | .764 |
| Postoperative EF (%) | 64.1 ± 6.4 | 62.9 ± 7.5 | .548 |
| Ischemic time (min) | 184.2 ± 58.7 | 177.6 ± 48.6 | .719 |
| Pulmonary artery pressure (mmHg) | 46.4 ± 14.4 | 50.6 ± 12.8 | .379 |
| Post-transplant survival (mo) | 32.6 ± 20.0 | 34.0 ± 19.4 | .806 |
| PD-L1 expression | |||
| Negative | 52 (89.7) | 6 (10.3) | <.001 |
| Positive | 10 (50.0) | 10 (50.0) | |
| PD-1+ lymphocytes (number/HPF) | 11.3 ± 20.5 | 31.3 ± 46.7 | .148 |
| CD8+ lymphocytes (number/HPF) | 89.8 ± 78.3 | 129.8 ± 102.3 | .175 |
| CD4+ lymphocytes (number/HPF) | 82.7 ± 72.4 | 170.5 ± 154.9 | .054 |
| PD-L1 expression | |||
|---|---|---|---|
| Absent (n = 58) | Present (n = 20) | p-value | |
| Age (yr) | |||
| <60 | 35 (71.4) | 14 (28.6) | .593 |
| ≥60 | 23 (79.3) | 6 (20.7) | |
| Sex | |||
| Male | 47 (79.7) | 12 (20.3) | .074 |
| Female | 11 (57.9) | 8 (42.1) | |
| CAD | |||
| Absent | 49 (77.8) | 14 (22.2) | .192 |
| Present | 9 (60.0) | 6 (40.0) | |
| Diabetes | |||
| Absent | 43 (78.2) | 12 (21.8) | .263 |
| Present | 15 (65.2) | 8 (34.8) | |
| Hypertension | |||
| Absent | 42 (71.2) | 17 (28.8) | .369 |
| Present | 16 (84.2) | 3 (15.8) | |
| Smoking | |||
| Never | 41 (77.4) | 12 (22.6) | .488 |
| Ever | 17 (68.0) | 8 (32.0) | |
| Ventilator | |||
| Not applied | 56 (73.7) | 20 (26.3) | .273 |
| Applied | 2 (100) | 0 | |
| Dialysis | |||
| Never | 50 (74.6) | 17 (25.4) | .894 |
| Ever | 8 (72.7) | 3 (27.3) | |
| Body mass index (kg/m2) | 21.1 ± 4.0 | 20.1 ± 4.9 | .480 |
| Preoperative EF (%) | 25.9 ± 12.5 | 21.4 ± 8.8 | .282 |
| Postoperative EF (%) | 63.7 ± 6.8 | 65.3 ± 4.9 | .521 |
| Ischemic time (min) | 183.9 ± 55.8 | 176.9 ± 63.7 | .733 |
| Pulmonary artery pressure (mmHg) | 46.0 ± 13.7 | 57.2 ± 14.5 | .066 |
| Post-transplant survival (mo) | 31.5 ± 19.2 | 42.4 ± 22.1 | .104 |
| PD-1+ lymphocytes (number/HPF) | 14.8 ± 24.7 | 35.6 ± 58.7 | .390 |
| CD8+ lymphocytes (number/HPF) | 91.6 ± 76.4 | 158.0 ± 119.2 | .059 |
| CD4+ lymphocytes (number/HPF) | 95.3 ± 105.2 | 198.4 ± 135.4 | .025 |
| Patient No. | Prior rejection | Rejection | Post-rejection |
|---|---|---|---|
| 1 | |||
| ACR grade | 0R | 2R | 0R |
| PD-L1 H-score | 0 | 14 | 0 |
| 2 | |||
| ACR grade | 1R | 2R | 1R |
| PD-L1 H-score | 0 | 0 | 0 |
| 3 | |||
| ACR grade | 0R | 2R | 0R |
| PD-L1 H-score | 0 | 0 | 0 |
| 4 | |||
| ACR grade | 0R | 2R | 0R |
| PD-L1 H-score | 0 | 5 | 0 |
| 5 | |||
| ACR grade | 0R | 2R | 0R |
| PD-L1 H-score | 0 | 5 | 0 |
| 6 | |||
| ACR grade | 0R | 3R | 0R |
| PD-L1 H-score | 5 | 50 | 0 |
| 7 | |||
| ACR grade | 0R | 2R | 0R |
| PD-L1 H-score | 2 | 0 | 0 |
| 8 | |||
| ACR grade | 0R | 2R | 0R |
| PD-L1 H-score | 0 | 6 | 0 |
| 9 | |||
| ACR grade | 1R | 2R | 1R |
| PD-L1 H-score | 5 | 2 | 0 |
| 10 | |||
| ACR grade | 1R | 2R | 1R |
| PD-L1 H-score | 0 | 0 | 0 |
| 11 | |||
| ACR grade | 2R | 3R | 0R |
| PD-L1 H-score | 3 | 15 | 0 |
| 12 | |||
| ACR grade | 0R | 2R | 2R |
| PD-L1 H-score | 0 | 0 | 0 |
| 13 | |||
| ACR grade | 1R | 3R | 2R |
| PD-L1 H-score | 0 | 0 | 0 |
Values are presented as mean (range) or number (%). CHF, chronic heart failure; CMP, cardiomyopathy; BMI, body mass index; EF, ejection fraction.
Values are presented as number (%) or mean ± SD. PD-1, programmed cell death-1; PD-L1, programmed death-ligand 1; CAD, coronary artery disease; EF, ejection fraction; HPF, high power field; SD, standard deviation. Cases with International Society for Heart and Lung Transplantation (ISHLT) grade 2R and 3R were considered to have high-grade acute cellular rejection.
Values are presented as number (%) or mean ± SD. PD-L1, programmed death-ligand 1; CAD, coronary artery disease; EF, ejection fraction; HPF, high power field; SD, standard deviation.
PD-L1, programmed death-ligand 1; ACR, acute cellular rejection; H-score, histochemical score.

 E-submission
E-submission