Articles
- Page Path
- HOME > J Pathol Transl Med > Volume 47(2); 2013 > Article
-
Review & Perspective
Guideline Recommendations forEGFR Mutation Testing in Lung Cancer: Proposal of the Korean Cardiopulmonary Pathology Study Group - Hyo Sup Shim,*, Jin-Haeng Chung1,*, Lucia Kim2, Sunhee Chang3, Wan-Seop Kim4, Geon Kook Lee5, Soon-Hee Jung6, Se Jin Jang7
-
Korean Journal of Pathology 2013;47(2):100-106.
DOI: https://doi.org/10.4132/KoreanJPathol.2013.47.2.100
Published online: April 24, 2013
Department of Pathology, Yonsei University College of Medicine, Seoul, Korea.
1Department of Pathology, Seoul National University Bundang Hospital, Seongnam, Korea.
2Department of Pathology, Inha University School of Medicine, Incheon, Korea.
3Department of Pathology, Ilsan Paik Hospital, Inje University College of Medicine, Goyang, Korea.
4Department of Pathology, Konkuk University School of Medicine, Seoul, Korea.
5Department of Pathology, National Cancer Center, Goyang, Korea.
6Department of Pathology, Yonsei University Wonju College of Medicine, Wonju, Korea.
7Department of Pathology, Asan Medical Center, University of Ulsan College of Medicine, Seoul, Korea.
- Corresponding Author: Se Jin Jang, M.D. Department of Pathology, University of Ulsan College of Medicine, Asan Medical Center, 88 Olympic-ro 43-gil, Songpa-gu, Seoul 138-736, Korea. Tel: +82-2-3010-5966, Fax: +82-2-472-7898, jangsejin@amc.seoul.kr
- *Hyo Sup Shim and Jin-Haeng Chung contributed equally to this work.
© 2013 The Korean Society of Pathologists/The Korean Society for Cytopathology
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/3.0/) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
- Abstract
- PATIENT SELECTION
- SAMPLE SOURCES
- SAMPLE PROCESSING
- ESTABLISHING ADEQUATE TUMOR CONTENT FOR MUTATION TESTING
- METHODS FOR EGFR MUTATION TESTING
- TURNAROUND TIME
- REPEATED EXAMINATION
- REPORTING FORMAT
- PATHOLOGIST'S ROLES
- PERSPECTIVES AND ADDITIONAL RECOMMENDATIONS
- CONCLUSION
- Acknowledgments
- Acknowledgments
- NOTES
- REFERENCES
Abstract
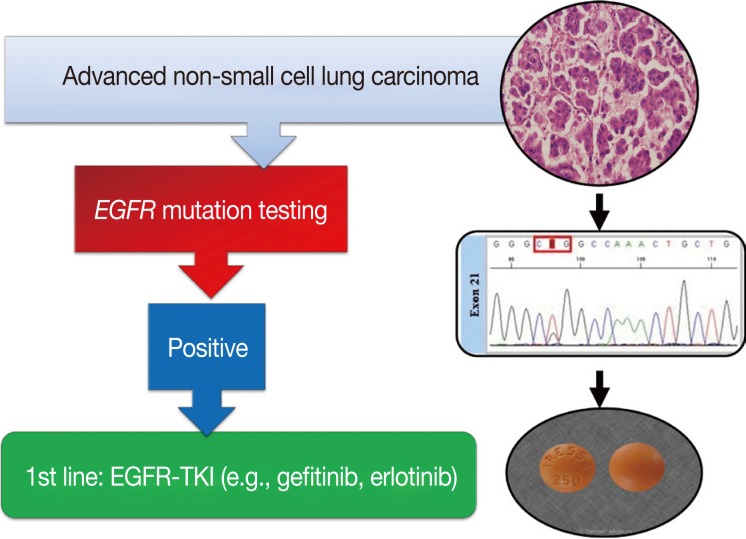
- Mutations of the epidermal growth factor receptor (EGFR) are the strongest predictive factor for response to EGFR tyrosine kinase inhibitors (TKIs), such as gefitinib and erlotinib. EGFR TKIs are approved in Korea as a first-line treatment for lung cancer patients with mutated EGFR. Rapid and accurate EGFR mutation testing is essential for patient selection and establishing targeted therapies with EGFR TKIs. Thus, a standard set of guideline recommendations for EGFR mutation testing suitable for the Korean medical community is necessary. In this article, we propose a set of guideline recommendations for EGFR mutation testing that was discussed and approved by the Cardiopulmonary Pathology Study Group of the Korean Society of Pathologists.
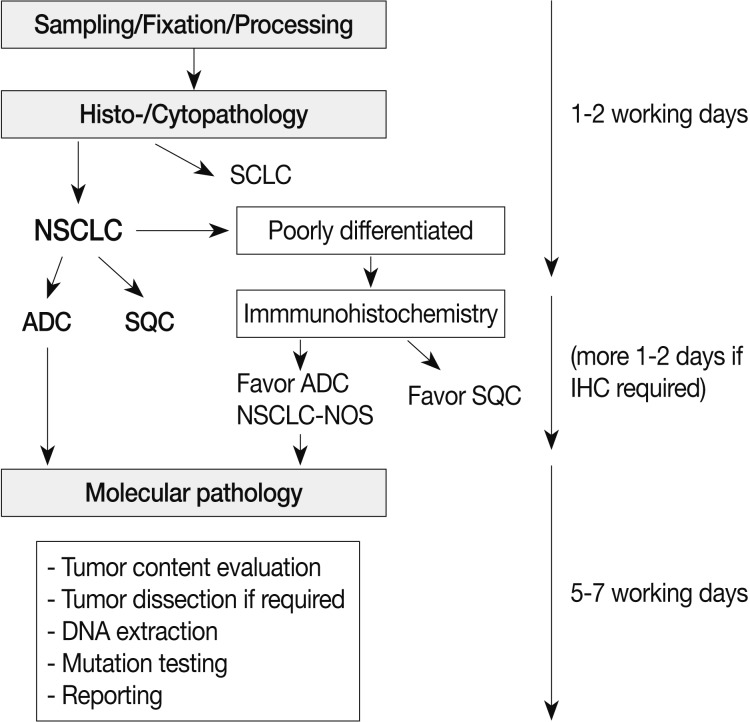
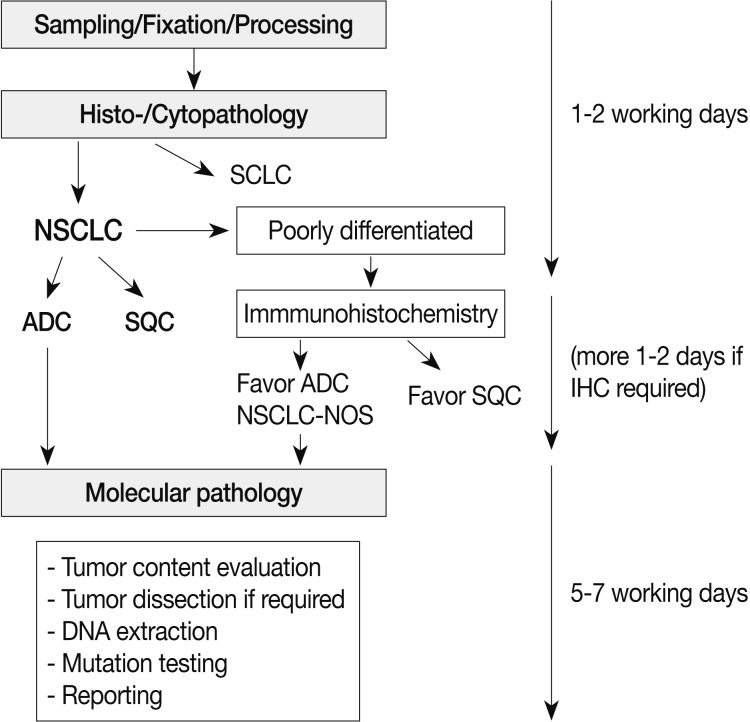
- The most important reason for EGFR mutation testing is to select patients who might benefit from EGFR TKI therapy. Patients that receive EGFR mutation testing are primarily those with advanced stage disease. EGFR mutations are more prevalent in female patients, never-smokers, and patients of Asian ethnicity. However, clinical features alone cannot entirely predict EGFR mutation status.6,7 Most of the guidelines published thus far recommend histologic type as the most important factor for determining whether EGFR mutational testing should be performed.8-10 Specifically, when patients are diagnosed with NSCLC including an adenocarcinoma component or NSCLC-not-otherwise-specified after immunohistochemistry, EGFR mutation testing is routinely recommended.9 Thus, pathologists should try to further classify poorly differentiated NSCLC into more specific types, such as adenocarcinoma or squamous cell carcinoma, whenever possible (Fig. 2). In addition, to preserve as much tissue as possible for molecular testing in small specimens, a minimum immunohistochemical panel such as thyroid transcription factor 1/napsin A/p63 or p40 is recommended.9,11,12
- EGFR mutations are detected in approximately 40% of Korean NSCLC patients with adenocarcinoma histology.13 Furthermore, it has been reported that EGFR mutations are more prevalent in specific subtypes of adenocarcinomas such as lepidic, papillary, or micropapillary, although it should be noted that these subtypes are not fully predictive of EGFR mutation status.14,15 Although previous studies have reported that a small fraction of squamous cell carcinomas or small cell carcinomas harbor EGFR mutations,16-19 routine examination is not recommended because the incidence in pure types is very low. However, in cases of female never-smokers, those with a combined tumor type, or when otherwise clinically indicated, mutation testing can be performed.
PATIENT SELECTION
- Various small biopsy and cytology specimens can be used as samples for mutation testing. More specifically, acceptable tissue specimens include transbronchial biopsy, gun biopsy, computed tomography-guided needle aspiration, endobronchial ultrasound-guided transbronchial needle aspiration, bronchial brushing/washing, and pleural fluid sampling.8,20,21 Many studies have shown that cytology specimens are suitable for assessing EGFR mutations, and that the results are highly concordant with those of corresponding histological specimens, especially when using more sensitive methods.20-24
- There have been several reports on the heterogeneous distribution of EGFR mutations and discordance of EGFR mutation status between primary tumors and corresponding metastatic tumors.25-27 In contrast, Yatabe et al.28 reported that a heterogeneous distribution of EGFR mutations is extremely rare in lung adenocarcinoma. Although there is an ongoing debate with respect to these reports, and further studies are needed,29,30 samples from a small portion of primary or metastatic tumor can be used equally.
SAMPLE SOURCES
- Routinely prepared samples are mostly formalin-fixed, paraffin-embedded (FFPE) tissues. Although there has been a report of fixation-related artifacts,31 routinely prepared FFPE tissues are the most practical and standard resource for EGFR mutation analysis. There is consensus that 10% neutral-buffered formalin is the optimum fixative for preparing FFPE samples,8,31 while the optimal fixation time ranges from 6 to 24 hours to avoid underfixation or overfixation, respectively.8,31
- Routinely prepared cytology specimens, such as alcohol-fixed smears or ThinPrep slides prepared by transferring cells in suspension20,23 and cell block specimens,32 are also suitable materials for EGFR mutation analysis.
SAMPLE PROCESSING
- Before mutation testing, the presence of tumor cells in the sample must be assessed by a pathologist. The ratio of tumor cells to normal cells is crucial for adequate mutation testing. For direct sequencing, the percentage of tumor cells in the sample should ideally be at least 50%, although reliable results can be influenced by a variety of factors. Thus, determination of the percentage of tumor cells in a given tissue or cell specimen is recommended. Macro- or microdissection may be used to increase the ratio of tumor to normal tissues if required. A study performed by Sun et al. showed that the following parameters correlate with the most reliable EGFR mutation results when using cytology samples: DNA concentration >25 µg/µL, content of >30 tumor cells, and tumor percentage >30%.23 The minimum number of tumor cells required for adequate testing and the minimum ratio of tumor to normal cells is influenced by the testing method (see below).
ESTABLISHING ADEQUATE TUMOR CONTENT FOR MUTATION TESTING
- Various methods can be used for detecting EGFR mutations.33-35 Pathologists should consider the available facilities and the pros and cons of each method, including the sensitivity and turnaround time. In addition, new techniques must be approved by the Korean government.
- Direct sequencing is considered to be the gold standard for EGFR mutation analysis. In Korea, most pathology laboratories perform direct sequencing for the detection of EGFR mutations using FFPE tissue samples. However, for directing DNA sequencing, a high ratio of tumor tissue to normal tissue content is required (more than 50% tumor content). In contrast, real time polymerase chain reaction (PCR)-based methods exhibit high sensitivity, requiring a mutant DNA content of only 1%.33 However, these methods can only detect previously known mutations or targeted sites. The peptide nucleic acid (PNA)-mediated PCR clamping method was recently developed and approved in Korea. The PNA clamping method exhibits high sensitivity compared with direct sequencing, and clinical outcomes are not significantly different between groups harboring EGFR mutations detected by direct sequencing or PNA-mediated PCR clamping.36-38 Highly sensitive methods can also be useful for detection of EGFR mutations associated with acquired resistance, such as T790M.39
METHODS FOR EGFR MUTATION TESTING
- Gefitinib is approved as a first-line treatment for advanced NSCLC-harboring EGFR mutations. Thus, the results of mutation analysis should be made available to physicians as soon as possible. Pathologic diagnosis and molecular testing are a combined and continuous process and should be supervised by a pathologist (Fig. 2). It is recommended that testing be completed within five to seven working days after ordering EGFR mutation testing.
- Several factors may influence turnaround time. In general, pathologic diagnoses and EGFR mutation testing performed within the same department rather than at separate laboratories would have a shorter turnaround time. When mutation testing is performed by an outside laboratory, communication and coordination between the pathology department and the external laboratory are encouraged.
TURNAROUND TIME
- Several criteria for repeated examination of EGFR mutation status have been recommended. The test should be repeated in cases of poor sequencing data, a cycle threshold too close to the defined cut-off limit (with pyrosequencing or PNA clamp kit), and/or mutation results that are not matched with previously well-defined clinical-pathologic characteristics. Specifically, pathologists should carefully interpret the results of EGFR mutations found in heavy smokers, solid or mucinous cancer types, or when EGFR mutations are concurrent with other exclusive driver mutations.
REPEATED EXAMINATION
- Molecular testing reports should contain the following information: pathologic number, age, sex, hospital unit number, biopsy site, sample source, requesting physician, requesting department, adequacy for testing (estimated tumor cell content), receipt day, report day, methodology used, exons tested and associated range of detectable mutations, mutation status, comments, testing technician, and corresponding pathologist.
REPORTING FORMAT
- The pathologist plays an essential role in EGFR mutation testing.40 The pathologist can either perform the test at the home institution or transfer the tissue to a reference laboratory for external examination. In both situations, the pathologist is responsible through the procedures. First, the pathologist should choose the most appropriate tissue to be tested.7,8,23 Second, the pathologist should verify that the selected tissue block for EGFR mutation testing contains sufficient tumor cells required for analysis. The proportion of the tumor cells in the tissue or cytology samples is very important to prevent contamination with non-tumor cells.7,23 Lastly, the pathologist is responsible for accurate and prompt reporting, which should include results from routing diagnostic information (such as histologic diagnosis), as well as from EGFR mutation testing. If the test is performed by an external reference laboratory, the pathologist integrates the test results into the pathology report of his/her institute.40 We recommend all patients with NSCLC having an adenocarcinoma component or NSCLC-not-otherwise-specified after immunohistochemistry should be tested for EGFR mutation. In the cases of squamous cell carcinomas or small cell carcinomas arising from never-smokers, mutation testing can be performed. As for the small biopsy or cytology specimens, macro- or microdissection may enhance mutation testing sensitivity.
PATHOLOGIST'S ROLES
- EGFR mutations and ALK rearrangements are currently used as predictive biomarkers for targeted lung cancer therapy. In addition, other driver mutations are now receiving attention, including ROS1 rearrangement,41 BRAF mutation,42,43 HER2 mutation,44 and RET rearrangement.45 In terms of molecular diagnostics, these other targetable mutations have developed the need for multiplex mutational testing. However, because most patients with lung cancer present with advanced-stage disease at the time of diagnosis, the diagnosis of lung cancer is often based on small specimens from a biopsy or cytology alone. Thus, each pathology department must develop a strategy to manage clinical samples and collaborate with clinicians.9 As mentioned above, these strategies include minimization of diagnostic stains in order to maximize the available tissue for molecular studies9,12 and reduction of the number of trimmings for slide sections.
PERSPECTIVES AND ADDITIONAL RECOMMENDATIONS
- As targetable mutations are discovered and corresponding targeted agents are developed, molecular diagnostics using clinical samples has become increasingly important. EGFR mutations are the most robust predictive factors for response to EGFR TKIs. Thus, each pathology department should maintain an optimal organization for the entire workflow of EGFR mutation testing, from sample collection to the final report. Lastly, pathologists should keep in mind that personalized medicine is driven by pathology and molecular diagnostics.
CONCLUSION
Acknowledgments
Acknowledgments
- 1. Cagle PT, Chirieac LR. Advances in treatment of lung cancer with targeted therapy. Arch Pathol Lab Med 2012; 136: 504-509. ArticlePubMedPDF
- 2. Janku F, Stewart DJ, Kurzrock R. Targeted therapy in non-small-cell lung cancer: is it becoming a reality? Nat Rev Clin Oncol 2010; 7: 401-414. ArticlePubMedPDF
- 3. Linardou H, Dahabreh IJ, Bafaloukos D, Kosmidis P, Murray S. Somatic EGFR mutations and efficacy of tyrosine kinase inhibitors in NSCLC. Nat Rev Clin Oncol 2009; 6: 352-366. ArticlePubMedPDF
- 4. Mok TS, Wu YL, Thongprasert S, et al. Gefitinib or carboplatin-paclitaxel in pulmonary adenocarcinoma. N Engl J Med 2009; 361: 947-957. ArticlePubMed
- 5. Fukuoka M, Wu YL, Thongprasert S, et al. Biomarker analyses and final overall survival results from a phase III, randomized, open-label, first-line study of gefitinib versus carboplatin/paclitaxel in clinically selected patients with advanced non-small-cell lung cancer in Asia (IPASS). J Clin Oncol 2011; 29: 2866-2874. ArticlePubMed
- 6. D'Angelo SP, Pietanza MC, Johnson ML, et al. Incidence of EGFR exon 19 deletions and L858R in tumor specimens from men and cigarette smokers with lung adenocarcinomas. J Clin Oncol 2011; 29: 2066-2070. ArticlePubMedPMC
- 7. Sun PL, Seol H, Lee HJ, et al. High incidence of EGFR mutations in Korean men smokers with no intratumoral heterogeneity of lung adenocarcinomas: correlation with histologic subtypes, EGFR/TTF-1 expressions, and clinical features. J Thorac Oncol 2012; 7: 323-330. ArticlePubMed
- 8. Pirker R, Herth FJ, Kerr KM, et al. Consensus for EGFR mutation testing in non-small cell lung cancer: results from a European workshop. J Thorac Oncol 2010; 5: 1706-1713. ArticlePubMed
- 9. Travis WD, Brambilla E, Noguchi M, et al. International Association for the Study of Lung Cancer/American Thoracic Society/European Respiratory Society international multidisciplinary classification of lung adenocarcinoma. J Thorac Oncol 2011; 6: 244-285. PubMedPMC
- 10. Salto-Tellez M, Tsao MS, Shih JY, et al. Clinical and testing protocols for the analysis of epidermal growth factor receptor mutations in East Asian patients with non-small cell lung cancer: a combined clinical-molecular pathological approach. J Thorac Oncol 2011; 6: 1663-1669. ArticlePubMed
- 11. Rekhtman N, Ang DC, Sima CS, Travis WD, Moreira AL. Immunohistochemical algorithm for differentiation of lung adenocarcinoma and squamous cell carcinoma based on large series of whole-tissue sections with validation in small specimens. Mod Pathol 2011; 24: 1348-1359. ArticlePubMedPDF
- 12. Noh S, Shim H. Optimal combination of immunohistochemical markers for subclassification of non-small cell lung carcinomas: a tissue microarray study of poorly differentiated areas. Lung Cancer 2012; 76: 51-55. ArticlePubMed
- 13. Yatabe Y. EGFR mutations and the terminal respiratory unit. Cancer Metastasis Rev 2010; 29: 23-36. ArticlePubMedPDF
- 14. Zakowski MF, Hussain S, Pao W, et al. Morphologic features of adenocarcinoma of the lung predictive of response to the epidermal growth factor receptor kinase inhibitors erlotinib and gefitinib. Arch Pathol Lab Med 2009; 133: 470-477. ArticlePubMedPMCPDF
- 15. Shim HS, Lee da H, Park EJ, Kim SH. Histopathologic characteristics of lung adenocarcinomas with epidermal growth factor receptor mutations in the International Association for the Study of Lung Cancer/American Thoracic Society/European Respiratory Society lung adenocarcinoma classification. Arch Pathol Lab Med 2011; 135: 1329-1334. ArticlePubMedPDF
- 16. Park SH, Ha SY, Lee JI, et al. Epidermal growth factor receptor mutations and the clinical outcome in male smokers with squamous cell carcinoma of lung. J Korean Med Sci 2009; 24: 448-452. ArticlePubMedPMC
- 17. Miyamae Y, Shimizu K, Hirato J, et al. Significance of epidermal growth factor receptor gene mutations in squamous cell lung carcinoma. Oncol Rep 2011; 25: 921-928. PubMed
- 18. Tatematsu A, Shimizu J, Murakami Y, et al. Epidermal growth factor receptor mutations in small cell lung cancer. Clin Cancer Res 2008; 14: 6092-6096. ArticlePubMedPDF
- 19. Shiao TH, Chang YL, Yu CJ, et al. Epidermal growth factor receptor mutations in small cell lung cancer: a brief report. J Thorac Oncol 2011; 6: 195-198. ArticlePubMed
- 20. Rekhtman N, Brandt SM, Sigel CS, et al. Suitability of thoracic cytology for new therapeutic paradigms in non-small cell lung carcinoma: high accuracy of tumor subtyping and feasibility of EGFR and KRAS molecular testing. J Thorac Oncol 2011; 6: 451-458. ArticlePubMed
- 21. Navani N, Brown JM, Nankivell M, et al. Suitability of endobronchial ultrasound-guided transbronchial needle aspiration specimens for subtyping and genotyping of non-small cell lung cancer: a multicenter study of 774 patients. Am J Respir Crit Care Med 2012; 185: 1316-1322. ArticlePubMedPMC
- 22. Allegrini S, Antona J, Mezzapelle R, et al. Epidermal growth factor receptor gene analysis with a highly sensitive molecular assay in routine cytologic specimens of lung adenocarcinoma. Am J Clin Pathol 2012; 138: 377-381. ArticlePubMed
- 23. Sun PL, Jin Y, Kim H, Lee CT, Jheon S, Chung JH. High concordance of EGFR mutation status between histologic and corresponding cytologic specimens of lung adenocarcinomas. Cancer Cytopathol 2012 12 05 [Epub]. http://dx.doi.org/10.1002/cncy.21260. Article
- 24. da Cunha Santos G, Saieg MA, Geddie W, Leighl N. EGFR gene status in cytological samples of nonsmall cell lung carcinoma: controversies and opportunities. Cancer Cytopathol 2011; 119: 80-91. PubMed
- 25. Taniguchi K, Okami J, Kodama K, Higashiyama M, Kato K. Intratumor heterogeneity of epidermal growth factor receptor mutations in lung cancer and its correlation to the response to gefitinib. Cancer Sci 2008; 99: 929-935. ArticlePubMedPMC
- 26. Schmid K, Oehl N, Wrba F, Pirker R, Pirker C, Filipits M. EGFR/KRAS/BRAF mutations in primary lung adenocarcinomas and corresponding locoregional lymph node metastases. Clin Cancer Res 2009; 15: 4554-4560. ArticlePubMedPDF
- 27. Gow CH, Chang YL, Hsu YC, et al. Comparison of epidermal growth factor receptor mutations between primary and corresponding metastatic tumors in tyrosine kinase inhibitor-naive non-small-cell lung cancer. Ann Oncol 2009; 20: 696-702. ArticlePubMed
- 28. Yatabe Y, Matsuo K, Mitsudomi T. Heterogeneous distribution of EGFR mutations is extremely rare in lung adenocarcinoma. J Clin Oncol 2011; 29: 2972-2977. ArticlePubMed
- 29. Chen ZY, Zhong WZ, Zhang XC, et al. EGFR mutation heterogeneity and the mixed response to EGFR tyrosine kinase inhibitors of lung adenocarcinomas. Oncologist 2012; 17: 978-985. ArticlePubMedPMCPDF
- 30. Bai H, Wang Z, Chen K, et al. Influence of chemotherapy on EGFR mutation status among patients with non-small-cell lung cancer. J Clin Oncol 2012; 30: 3077-3083. ArticlePubMedPMC
- 31. Eberhard DA, Giaccone G, Johnson BE. Non-Small-Cell Lung Cancer Working Group. Biomarkers of response to epidermal growth factor receptor inhibitors in Non-Small-Cell Lung Cancer Working Group: standardization for use in the clinical trial setting. J Clin Oncol 2008; 26: 983-994. ArticlePubMed
- 32. Nicholson AG, Gonzalez D, Shah P, et al. Refining the diagnosis and EGFR status of non-small cell lung carcinoma in biopsy and cytologic material, using a panel of mucin staining, TTF-1, cytokeratin 5/6, and p63, and EGFR mutation analysis. J Thorac Oncol 2010; 5: 436-441. ArticlePubMed
- 33. Pao W, Ladanyi M. Epidermal growth factor receptor mutation testing in lung cancer: searching for the ideal method. Clin Cancer Res 2007; 13: 4954-4955. ArticlePubMedPDF
- 34. Ellison G, Zhu G, Moulis A, Dearden S, Speake G, McCormack R. EGFR mutation testing in lung cancer: a review of available methods and their use for analysis of tumour tissue and cytology samples. J Clin Pathol 2013; 66: 79-89. ArticlePubMed
- 35. Lee HJ, Xu X, Kim H, et al. Comparison of direct sequencing, PNA clamping-real time polymerase chain reaction, and pyrosequencing methods for the detection of EGFR mutations in non-small cell lung carcinoma and the correlation with clinical responses to EGFR tyrosine kinase inhibitor treatment. Korean J Pathol 2013; 47: 52-60. ArticlePubMedPMC
- 36. Kim HJ, Kim WS, Shin KC, et al. Comparative analysis of peptide nucleic acid (PNA)-mediated real-time PCR clamping and DNA direct sequencing for EGFR mutation detection. Tuberc Respir Dis 2011; 70: 21-27. Article
- 37. Kim HJ, Lee KY, Kim YC, et al. Detection and comparison of peptide nucleic acid-mediated real-time polymerase chain reaction clamping and direct gene sequencing for epidermal growth factor receptor mutations in patients with non-small cell lung cancer. Lung Cancer 2012; 75: 321-325. ArticlePubMed
- 38. Han HS, Lim SN, An JY, et al. Detection of EGFR mutation status in lung adenocarcinoma specimens with different proportions of tumor cells using two methods of differential sensitivity. J Thorac Oncol 2012; 7: 355-364. ArticlePubMed
- 39. Arcila ME, Oxnard GR, Nafa K, et al. Rebiopsy of lung cancer patients with acquired resistance to EGFR inhibitors and enhanced detection of the T790M mutation using a locked nucleic acid-based assay. Clin Cancer Res 2011; 17: 1169-1180. ArticlePubMedPMCPDF
- 40. van Krieken JH, Jung A, Kirchner T, et al. KRAS mutation testing for predicting response to anti-EGFR therapy for colorectal carcinoma: proposal for an European quality assurance program. Virchows Arch 2008; 453: 417-431. ArticlePubMed
- 41. Bergethon K, Shaw AT, Ou SH, et al. ROS1 rearrangements define a unique molecular class of lung cancers. J Clin Oncol 2012; 30: 863-870. ArticlePubMedPMC
- 42. Marchetti A, Felicioni L, Malatesta S, et al. Clinical features and outcome of patients with non-small-cell lung cancer harboring BRAF mutations. J Clin Oncol 2011; 29: 3574-3579. ArticlePubMed
- 43. Paik PK, Arcila ME, Fara M, et al. Clinical characteristics of patients with lung adenocarcinomas harboring BRAF mutations. J Clin Oncol 2011; 29: 2046-2051. ArticlePubMedPMC
- 44. Arcila ME, Chaft JE, Nafa K, et al. Prevalence, clinicopathologic associations, and molecular spectrum of ERBB2 (HER2) tyrosine kinase mutations in lung adenocarcinomas. Clin Cancer Res 2012; 18: 4910-4918. ArticlePubMedPDF
- 45. Wang R, Hu H, Pan Y, et al. RET fusions define a unique molecular and clinicopathologic subtype of non-small-cell lung cancer. J Clin Oncol 2012; 30: 4352-4359. ArticlePubMed
REFERENCES
| Recommendation | |
|---|---|
| Patient selection | Pathologic diagnosis is the most important factor |
| Patients with non-small cell carcinoma, especially adenocarcinoma componenta | |
| Other types if clinically indicated | |
| Sample source | Primary and metastatic sites are equally suitable |
| Biopsy (formalin-fixed paraffin-embedded tissue) and cytology specimens are equally suitable | |
| Sample processing | Routine preparation for tissue or cytology is suitable |
| Tumor content | The presence of tumor cells must be verified by a pathologist |
| High percentage (ideally more than 50%) of tumor cells for direct sequencing | |
| Lower percentage acceptable for methods with higher sensitivity | |
| Method for mutation testing | Various methods can be used for mutation testing |
| New techniques must be approved by the Korean government | |
| The pathologist should consider available facilities and the pros and cons of each method | |
| Turnaround time | The entire workflow process should be supervised by the pathologist |
| Pathologic diagnosis: 1-2 working days | |
| Molecular diagnosis: 5-7 working days | |
| Repeat examination | The pathologist should consider repeating the examination under the following situations |
| Poor sequence data | |
| Cycle threshold too close to the defined cut-off limit | |
| Result are not matched with previously well-defined clinical-pathologic characteristics | |
| Reporting format | Sample information, type of method, mutation status, comments |
EGFR, epidermal growth factor receptor.
aIn this regard, poorly differentiated non-small cell carcinoma should be further classified into a more specific type whenever possible. A minimum immunohistochemical panel (such as thyroid transcription factor 1/napsin A/p63 or p40) is recommended in small specimens to preserve as much tissue as possible for molecular testing.
Figure & Data
References
Citations

- Establishment of a Flow Cytometry Protocol for Binarily Detecting Circulating Tumor Cells with EGFR Mutation
Cheng-Yu Chang, Chia-Chun Tu, Shian-Ren Lin, Chih-Hao Fang, Po-Wei Tseng, Wan-En Liao, Li-Yun Huang, Shiu-Lan Wang, Wan-Yu Lai, Yee Chao, Yen-Ling Chiu, Jan-Mou Lee
Diseases.2025; 13(12): 406. CrossRef - Favorable Conditions for the Detection of EGFR T790M Mutation Using Plasma Sample in Patients with Non-Small-Cell Lung Cancer
Insu Kim, Hee Yun Seol, Soo Han Kim, Mi-Hyun Kim, Min Ki Lee, Jung Seop Eom
Cancers.2023; 15(5): 1445. CrossRef - Novel Targets, Novel Treatments: The Changing Landscape of Non-Small Cell Lung Cancer
Dorine de Jong, Jeeban P. Das, Hong Ma, Jacienta Pailey Valiplackal, Conor Prendergast, Tina Roa, Brian Braumuller, Aileen Deng, Laurent Dercle, Randy Yeh, Mary M. Salvatore, Kathleen M. Capaccione
Cancers.2023; 15(10): 2855. CrossRef - Coordination games in cancer
Péter Bayer, Robert A. Gatenby, Patricia H. McDonald, Derek R. Duckett, Kateřina Staňková, Joel S. Brown, Jun Tanimoto
PLOS ONE.2022; 17(1): e0261578. CrossRef - Molecular biomarker testing for non–small cell lung cancer: consensus statement of the Korean Cardiopulmonary Pathology Study Group
Sunhee Chang, Hyo Sup Shim, Tae Jung Kim, Yoon-La Choi, Wan Seop Kim, Dong Hoon Shin, Lucia Kim, Heae Surng Park, Geon Kook Lee, Chang Hun Lee
Journal of Pathology and Translational Medicine.2021; 55(3): 181. CrossRef - Primary HHV-8 (-) Effusion-Based Non-Germinal Center B Cell Diffuse Large B Cell Lymphoma Successfully Treated with Standard Anthracycline-Based Chemoimmunotherapy
Justin J Kuhlman, Muhamad Alhaj Moustafa, Liuyan Jiang, Han W Tun
Journal of Blood Medicine.2021; Volume 12: 833. CrossRef - Molecular Testing of Lung Cancers
Hyo Sup Shim, Yoon-La Choi, Lucia Kim, Sunhee Chang, Wan-Seop Kim, Mee Sook Roh, Tae-Jung Kim, Seung Yeon Ha, Jin-Haeng Chung, Se Jin Jang, Geon Kook Lee
Journal of Pathology and Translational Medicine.2017; 51(3): 242. CrossRef - Strategic management of transthoracic needle aspirates for histological subtyping and EGFR testing in patients with peripheral lung cancer: An institutional experience
Choonhee Son, Eun‐Ju Kang, Mee Sook Roh
Diagnostic Cytopathology.2015; 43(7): 532. CrossRef - Ultrasonography-Guided Core Biopsy of Supraclavicular Lymph Nodes for Diagnosis of Metastasis and Identification of Epidermal Growth Factor Receptor (EGFR) Mutation in Advanced Lung Cancer
Jooae Choe, Mi Young Kim, Jung Hwan Baek, Chang-Min Choi, Hwa Jung Kim
Medicine.2015; 94(29): e1209. CrossRef - Reliable EGFR mutation testing in ultrasound-guided supraclavicular lymph node fine-needle aspirates: a cohort study with diagnostic performance analysis
Amir Awwad, Sandeep Tiwari, Vishakha Sovani, David R Baldwin, Maruti Kumaran
BMJ Open Respiratory Research.2015; 2(1): e000075. CrossRef - Simultaneous diagnostic platform of genotyping EGFR, KRAS, and ALK in 510 Korean patients with non‐small‐cell lung cancer highlights significantly higher ALK rearrangement rate in advanced stage
Tae‐Jung Kim, Chan Kwon Park, Chang Dong Yeo, Kihoon Park, Chin Kook Rhee, Jusang Kim, Seung Joon Kim, Sang Haak Lee, Kyo‐Young Lee, Hyoung‐Kyu Yoon
Journal of Surgical Oncology.2014; 110(3): 245. CrossRef - Novel EGFR mutation-specific antibodies for lung adenocarcinoma: Highly specific but not sensitive detection of an E746_A750 deletion in exon 19 and an L858R mutation in exon 21 by immunohistochemistry
An Na Seo, Tae-In Park, Yan Jin, Ping-Li Sun, Hyojin Kim, Hyun Chang, Jin-Haeng Chung
Lung Cancer.2014; 83(3): 316. CrossRef - Molecular Pathology of Lung Cancer: Current Status and Future Directions
Mee Sook Roh
Tuberculosis and Respiratory Diseases.2014; 77(2): 49. CrossRef
 PubReader
PubReader ePub Link
ePub Link-
 Cite this Article
Cite this Article
- Cite this Article
-
- Close
- Download Citation
- Close
- Figure


Fig. 1
Fig. 2
| Recommendation | |
|---|---|
| Patient selection | Pathologic diagnosis is the most important factor |
| Patients with non-small cell carcinoma, especially adenocarcinoma component |
|
| Other types if clinically indicated | |
| Sample source | Primary and metastatic sites are equally suitable |
| Biopsy (formalin-fixed paraffin-embedded tissue) and cytology specimens are equally suitable | |
| Sample processing | Routine preparation for tissue or cytology is suitable |
| Tumor content | The presence of tumor cells must be verified by a pathologist |
| High percentage (ideally more than 50%) of tumor cells for direct sequencing | |
| Lower percentage acceptable for methods with higher sensitivity | |
| Method for mutation testing | Various methods can be used for mutation testing |
| New techniques must be approved by the Korean government | |
| The pathologist should consider available facilities and the pros and cons of each method | |
| Turnaround time | The entire workflow process should be supervised by the pathologist |
| Pathologic diagnosis: 1-2 working days | |
| Molecular diagnosis: 5-7 working days | |
| Repeat examination | The pathologist should consider repeating the examination under the following situations |
| Poor sequence data | |
| Cycle threshold too close to the defined cut-off limit | |
| Result are not matched with previously well-defined clinical-pathologic characteristics | |
| Reporting format | Sample information, type of method, mutation status, comments |
In this regard, poorly differentiated non-small cell carcinoma should be further classified into a more specific type whenever possible. A minimum immunohistochemical panel (such as thyroid transcription factor 1/napsin A/p63 or p40) is recommended in small specimens to preserve as much tissue as possible for molecular testing.

 E-submission
E-submission