Articles
- Page Path
- HOME > J Pathol Transl Med > Volume 50(1); 2016 > Article
-
Brief Case Report
Indeterminate Dendritic Cell Tumor: A Case Report of a Rare Langerhans Cell Lineage Disease - Jin Roh, So-Woon Kim, Chan-Sik Park
-
Journal of Pathology and Translational Medicine 2016;50(1):78-81.
DOI: https://doi.org/10.4132/jptm.2015.07.03
Published online: August 4, 2015
Department of Pathology, Asan Medical Center, University of Ulsan College of Medicine, Seoul, Korea
- Corresponding Author: Chan-Sik Park, MD Department of Pathology, Asan Medical Center, University of Ulsan College of Medicine, 88 Olympic-ro 43-gil, Songpa-gu, Seoul 05505, Korea Tel: +82-2-3010-4560, Fax: +82-2-472-7898, E-mail: csikpark@amc.seoul.kr
• Received: May 20, 2015 • Revised: June 29, 2015 • Accepted: July 1, 2015
© 2016 The Korean Society of Pathologists/The Korean Society for Cytopathology
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/3.0/) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
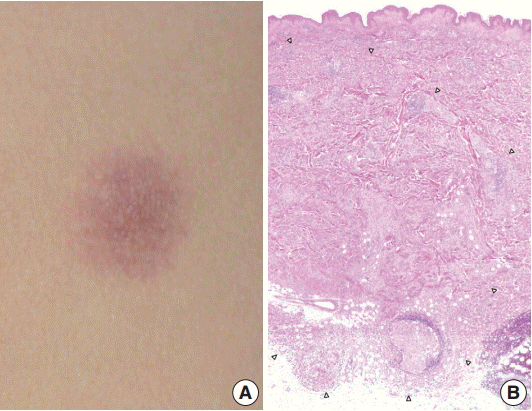
- A 29-year-old female patient presented with a 1.2-cm, ill-defined, erythematous subcutaneous nodule on the left flank (Fig. 1A), which appeared a month ago and increased in size over time. She had no cutaneous or extracutaneous symptoms. The patient was otherwise healthy with no remarkable medical history. Skeletal survey and bone scan revealed no bone lesions.
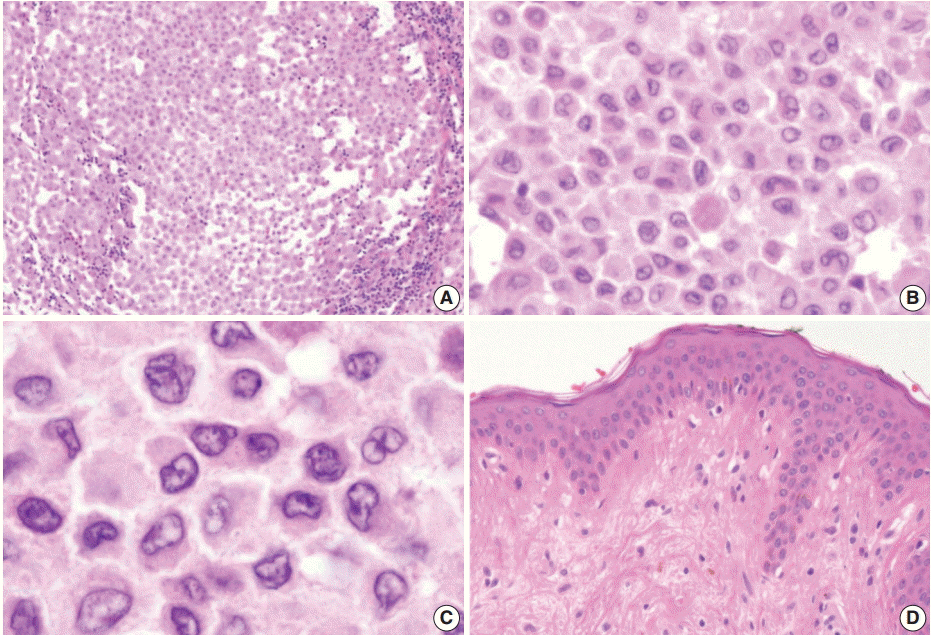
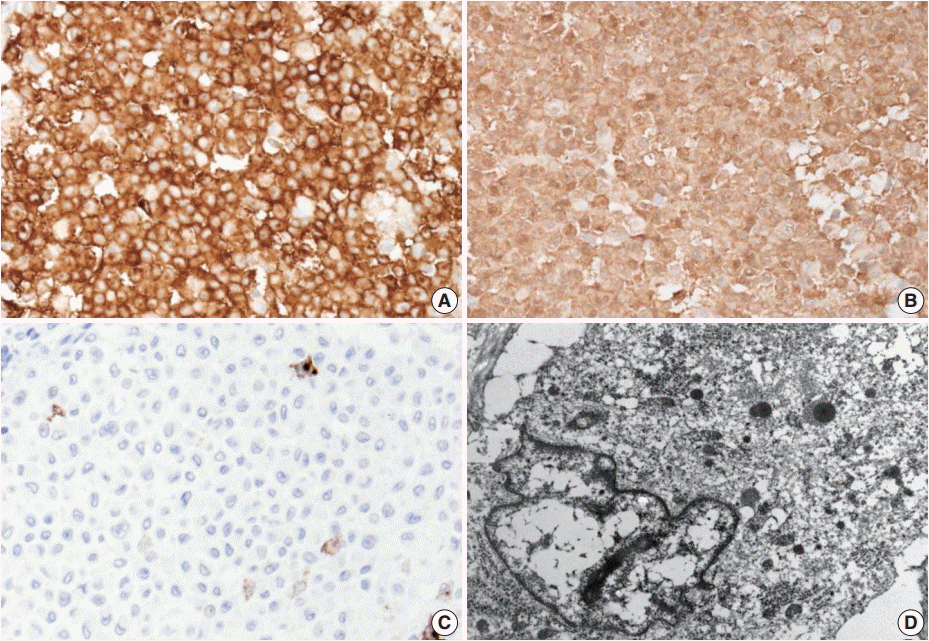
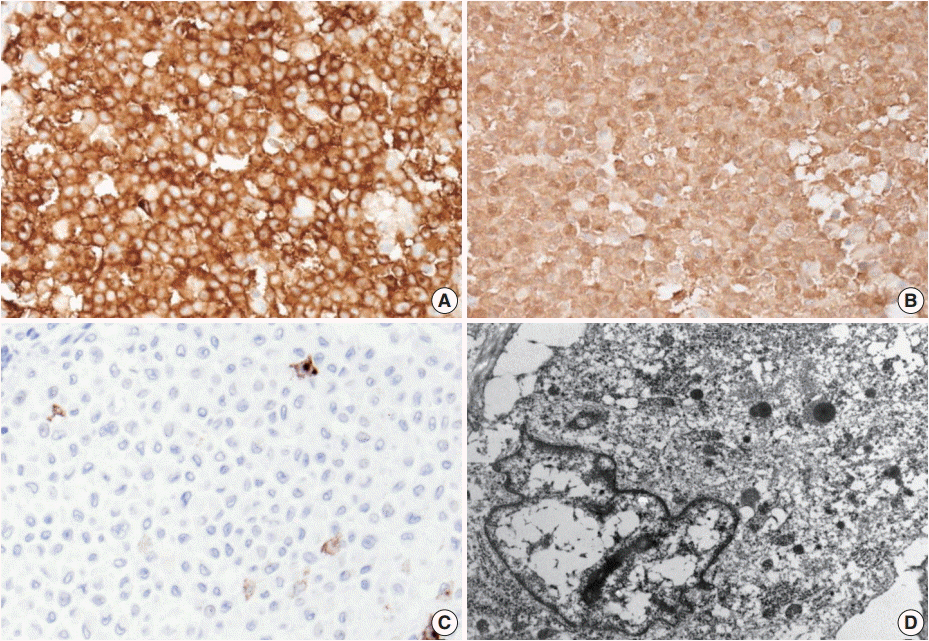
- Excision of the nodule was performed. Microscopically, the lesion showed poorly circumscribed infiltrates of tumor cells forming mostly solid sheets in the dermis and subcutis (Fig. 1B). No necrosis or adnexal invasion was noted. A small amount of peritumoral inflammatory cell infiltration, composed mostly of lymphocytes, was observed. Eosinophils were notably absent (Fig. 2A). Tumor cells showed monotonous morphology with indistinct cell borders. They had moderately abundant and lightly eosinophilic cytoplasm (Fig. 2B). The tumor cells had enfolded or reniform vesicular nuclei with inconspicuous nucleoli, and some of the tumor cells showed longitudinal nuclear grooves. Although occasional neoplastic cells had a large nucleus, mitotic figures were rarely seen (Fig. 2C). Characteristically, there was no epidermal involvement by tumor cells (Fig. 2D). The neoplastic cells in the tumor were diffusely immunopositive for S100 and CD1a (Fig. 3A, B). Langerin (CD207) was clearly negative in the tumor cells. Scattered langerin-positive bystander Langerhans cells serve as internal positive controls (Fig. 3C). Despite intensive scrutiny, no Birbeck granules were identified on electron microscopy (Fig. 3D).
CASE REPORT
- In this paper, we report a case of IDCT, an extraordinarily rare neoplasm. Most IDCTs occur in adults without predilection for either sex [3,5]. IDCT is almost always restricted to the skin without systemic symptoms [2,6]. Its rarity and resemblance to LCL often cause as diagnostic challenge for pathologists who are not familiar with this entity. Uniform expression of CD1a and S100 protein enables distinction of IDCT from other forms of non-Langerhans cell neoplasms, such as juvenile xanthogranuloma, xanthoma, histiocytic sarcoma, and reticulohistiocytosis [7]. The differential diagnosis also includes Langerhans cell lineage tumors, such as Langerhans cell histiocytosis (LCH) and Langerhans cell sarcoma (LCS). LCH is a rare condition and mostly occurs in childhood [7]. In cutaneous LCH, the lesions present as papules or plaques [3]. Tumor cells show typical morphology of Langerhans cells, which are indistinguishable from those of IDCT [5]. However, unlike IDCT, LCH shows evident epidermotropism with intraepidermal Langerhans cell microabscesses [3]. Presence of eosinophilic infiltration is another important difference from IDCT [3]. Immunopositivity for S100 protein, CD1a, and langerin (CD207), and the presence of Birbeck granules confirm the diagnosis of LCH [2]. Although LCH shows variable outcomes, most of the cases require long-term follow-up. LCS is another important tumor in the differential diagnosis. LCS is a very rare high-grade neoplasm [8]. Unlike in IDCT, epidermotropism and eosinophil infiltration are not usually evident in LCS [5]; however, most of the tumor cells show overtly malignant cytology including pleomorphic and hyperchromatic nuclei with frequent mitotic figures [8]. Langerhans cell differentiation of neoplastic cells of LCS must be confirmed by immunohistochemistry (IHC) or electron microscopy. LCS is an aggressive neoplasm and has very poor prognosis [8]. In the present case, langerin (CD207) immunonegativity of the tumor cells and the absence of Birbeck granules on electron microscopy were crucial for definitive diagnosis of IDCT [5].
- Regarding the utility of IHC in comparison with electron microscopy, langerin IHC is important in the differentiating between true Langerhans cells and Langerhans cell-like lesions [2]. Langerin is an antibody to a transmembrane C-type lectin that associates with Birbeck granule formation. Previous studies revealed high sensitivity and specificity of langerin for Langerhans cell differentiation [9,10].
- In summary, IDCT should be included in the differential diagnosis for a lesion composed of Langerhans cell-like cells without epidermotropism and eosinophilic infiltration. To our knowledge, the present study is the first report of IDCT in Korean pathologic literature. Making an accurate diagnosis of IDCT is important to avoid overly aggressive management.
DISCUSSION
Acknowledgments
Fig. 1.(A) An ill-defined, 1.2-cm, round, erythematous subcutaneous nodule on the left flank. (B) The ill-defined tumor is mainly located in the dermis and subcutis. The margin of the tumor is marked by arrowheads.
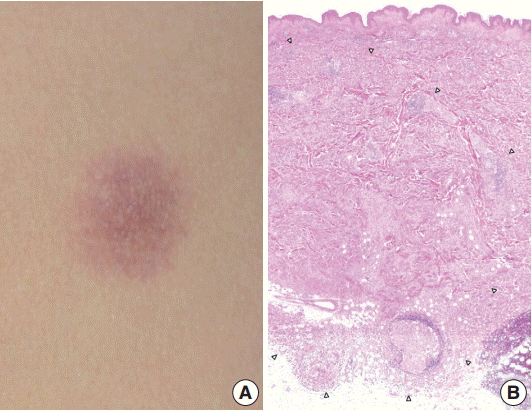

Fig. 2.(A) There is patchy infiltration by aggregates of the tumor cells. Tumor cells are usually monotonous and have indistinct cell borders. Clusters of lymphocytes are admixed with tumor cells in focal areas without eosinophils. (B) The constituent cells have ovoid cell morphology with abundant eosinophilic cytoplasm. (C) Their nuclei are oval and sometimes indented. Nuclear grooves are frequently seen. Although occasional enlarged nuclei are identified, mitoses are rarely seen. (D) There is no epidermal involvement.
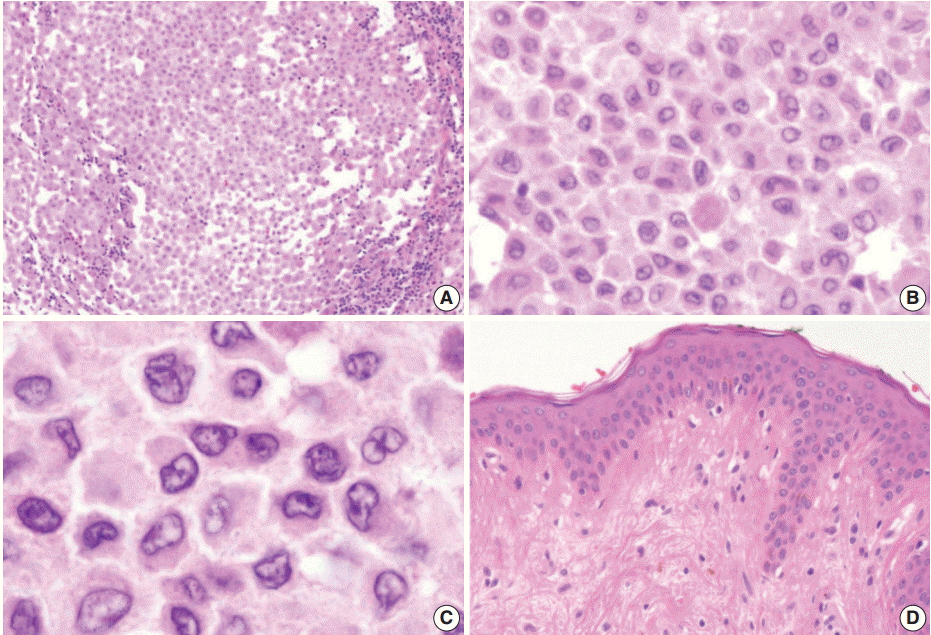

Fig. 3.The neoplastic cells diffusely express CD1a (A) and S100 protein (B). (C) Langerin (CD207) is typically negative. (D) No Birbeck granules are seen on transmission electron microscopy (×8,000).

- 1. Weiss LM, Chan JK, Fletcher CD. Other rare dendritic cell tumours. In: Swerdlow SH, Campo E, Harris NL, eds. WHO classification of tumours of haematopoietic and lymphoid tissues. 4th ed. Lyon: IARC Press, 2008; 365.
- 2. Ghanadan A, Kamyab K, Ramezani M, et al. Indeterminate cell histiocytosis: report of a case. Acta Med Iran 2014; 52: 788-90. PubMed
- 3. Rezk SA, Spagnolo DV, Brynes RK, Weiss LM. Indeterminate cell tumor: a rare dendritic neoplasm. Am J Surg Pathol 2008; 32: 1868-76. PubMed
- 4. Vener C, Soligo D, Berti E, et al. Indeterminate cell histiocytosis in association with later occurrence of acute myeloblastic leukaemia. Br J Dermatol 2007; 156: 1357-61. ArticlePubMed
- 5. Wang CH, Chen GS. Indeterminate cell histiocytosis: a case report. Kaohsiung J Med Sci 2004; 20: 24-30. ArticlePubMedPDF
- 6. Calonje E, Brenn T, Lazar A, McKee PH. McKee’s pathology of the skin with clinical correlations. 4th ed. Edinburgh: Saunders, 2011; 1398.
- 7. Bakry OA, Samaka RM, Kandil MA, Younes SF. Indeterminate cell histiocytosis with naive cells. Rare Tumors 2013; 5: e13.
- 8. Bohn OL, Ruiz-Argüelles G, Navarro L, Saldivar J, Sanchez-Sosa S. Cutaneous Langerhans cell sarcoma: a case report and review of the literature. Int J Hematol 2007; 85: 116-20. ArticlePubMed
- 9. Chikwava K, Jaffe R. Langerin (CD207) staining in normal pediatric tissues, reactive lymph nodes, and childhood histiocytic disorders. Pediatr Dev Pathol 2004; 7: 607-14. ArticlePubMedPDF
- 10. Lau SK, Chu PG, Weiss LM. Immunohistochemical expression of Langerin in Langerhans cell histiocytosis and non-Langerhans cell histiocytic disorders. Am J Surg Pathol 2008; 32: 615-9. ArticlePubMed
REFERENCES
Figure & Data
References
Citations
Citations to this article as recorded by 

- Widespread Cutaneous Indeterminate Dendritic Cell Tumor (IDCT) With ETV3::NOAC2 Rearrangement Successfully Treated With PUVA Therapy: A Case Report
Akshay Deshpande, Zbigniew Rudzki, Jaideep Bhat, Matthew Pugh, Susan Rose, Shankara Paneesha, Pier Paolo Piccaluga
Case Reports in Hematology.2024;[Epub] CrossRef - Indeterminate cell histiocytosis: A systematic review of the literature with a comprehensive revision of clinical, histopathological, and molecular features
Simone Zanella, Emilio Berti, Arturo Bonometti
Journal of the European Academy of Dermatology and Venereology.2023; 37(8): 1559. CrossRef - Indeterminate dendritic cell tumor (IDCT)
Evgenia V. Okladnikova, Vladimir M. Merk, Yuriy А. Krivopalov, Vladimir А. Khorzhevskii, Tatiana G. Ruksha
Vestnik dermatologii i venerologii.2023; 99(4): 143. CrossRef - Disseminated Indeterminate Dendritic Cell Tumor
Mary Z. Chhangte, Debopriya Paul, Reena Lamba, Biswajit Dey
Indian Dermatology Online Journal.2021; 12(3): 462. CrossRef - Neoplasias de células histiocitarias y dendríticas: revisión de la literatura
Cristina Díaz del Arco, Luis Ortega Medina, M. Jesús Fernández Aceñero
Revista Española de Patología.2018; 51(3): 160. CrossRef - Indeterminate cell histiocytosis in a Chinese patient with progressive and extensive nodular lesions and mixed indeterminate cell and macrophage‐monocyte lineage
Qiping Chen, Joyce S.‐S. Lee, Michelle W. Liang, Michelle M.F. Chan, Ramón M. Pujol, Luis Requena, Suat Hoon Tan
Journal of Cutaneous Pathology.2018; 45(6): 428. CrossRef - Recurrent Indeterminate Dendritic Cell Tumor of the Skin
Jin Woo Joo, Taek Chung, Yoon Ah Cho, Sang Kyum Kim
Journal of Pathology and Translational Medicine.2018; 52(4): 243. CrossRef - Indeterminate Dendritic Cell Tumor: A Report of Two New Cases Lacking the ETV3-NCOA2 Translocation and a Literature Review
Jonathan J. Davick, Jinah Kim, Mark R. Wick, Alejandro A. Gru
The American Journal of Dermatopathology.2018; 40(10): 736. CrossRef - A Case of Indeterminate Dendritic Cell Tumor: A Rare Neoplasm with Langerhans Cell Lineage
Jungyoon Moon, Ji Hoon Yang, Jaewon Lee, Jong Seo Park, Kwang Hyun Cho
Annals of Dermatology.2018; 30(6): 744. CrossRef - Indeterminate Dendritic Cell Tumor in Thoracic Spine: A Case Report
Sze Kiat Tan, Lee Onn Chieng, Karthik Madhavan, Andrew Rosenberg, Ian Cote
World Neurosurgery.2017; 108: 543. CrossRef
 PubReader
PubReader ePub Link
ePub Link-
 Cite this Article
Cite this Article
- Cite this Article
-
- Close
- Download Citation
- Close
- Figure
Indeterminate Dendritic Cell Tumor: A Case Report of a Rare Langerhans Cell Lineage Disease



Fig. 1. (A) An ill-defined, 1.2-cm, round, erythematous subcutaneous nodule on the left flank. (B) The ill-defined tumor is mainly located in the dermis and subcutis. The margin of the tumor is marked by arrowheads.
Fig. 2. (A) There is patchy infiltration by aggregates of the tumor cells. Tumor cells are usually monotonous and have indistinct cell borders. Clusters of lymphocytes are admixed with tumor cells in focal areas without eosinophils. (B) The constituent cells have ovoid cell morphology with abundant eosinophilic cytoplasm. (C) Their nuclei are oval and sometimes indented. Nuclear grooves are frequently seen. Although occasional enlarged nuclei are identified, mitoses are rarely seen. (D) There is no epidermal involvement.
Fig. 3. The neoplastic cells diffusely express CD1a (A) and S100 protein (B). (C) Langerin (CD207) is typically negative. (D) No Birbeck granules are seen on transmission electron microscopy (×8,000).
Fig. 1.
Fig. 2.
Fig. 3.
Indeterminate Dendritic Cell Tumor: A Case Report of a Rare Langerhans Cell Lineage Disease

 E-submission
E-submission