Articles
- Page Path
- HOME > J Pathol Transl Med > Volume 49(6); 2015 > Article
-
Review
Acute Atherosis of the Uterine Spiral Arteries: Clinicopathologic Implications - Joo-Yeon Kim1, Yeon Mee Kim1,2
-
Journal of Pathology and Translational Medicine 2015;49(6):462-471.
DOI: https://doi.org/10.4132/jptm.2015.10.23
Published online: November 4, 2015
1Department of Pathology, Haeundae Paik Hospital, Inje University College of Medicine, Busan, Korea
2Department of Molecular Medicine, Kungpook National University School of Medicine, Daegu, Korea
- Corresponding Author: Yeon Mee Kim, MD Department of Pathology, Haeundae Paik Hospital, Inje University College of Medicine, 875 Haeun-daero, Haeundae-gu, Busan 48108, Korea Tel: +82-51-797-3100 Fax: +82-51-797-3101 E-mail: ykim.haeundae@gmail.com
• Received: September 19, 2015 • Revised: October 20, 2015 • Accepted: October 23, 2015
© 2015 The Korean Society of Pathologists/The Korean Society for Cytopathology
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/3.0/) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
- Abstract
- POOR PLACENTATION AND ACUTE ATHEROSIS
- HISTOLOGIC CHARACTERISTICS AND TOPOGRAPHIC DISTRIBUTION OF ACUTE ATHEROSIS
- FREQUENCY OF ACUTE ATHEROSIS
- PLACENTAL LESIONS ASSOCIATED WITH ACUTE ATHEROSIS
- PATHOGENESIS OF ACUTE ATHEROSIS
- THE CLINICAL IMPLICATIONS OF ACUTE ATHEROSIS
- CONCLUSION
- NOTES
- Acknowledgments
- REFERENCES
Abstract
- Acute atherosis is unique vascular changes of the placenta associated with poor placentation. It is characterized by subendothelial lipid-filled foam cells, fibrinoid necrosis of the arterial wall, perivascular lymphocytic infiltration, and it is histologically similar to early-stage atherosclerosis. Acute atherosis is rare in normal pregnancies, but is frequently observed in non- transformed spiral arteries in abnormal pregnancies, such as preeclampsia, small for gestational age (SGA), fetal death, spontaneous preterm labor and preterm premature rupture of membranes. In preeclampsia, spiral arteries fail to develop physiologic transformation and retain thick walls and a narrow lumen. Failure of physiologic transformation of spiral arteries is believed to be the main cause of uteroplacental ischemia, which can lead to the production of anti-angiogenic factors and induce endothelial dysfunction and eventually predispose the pregnancy to preeclampsia. Acute atherosis is more frequently observed in the spiral arteries of the decidua of the placenta (parietalis or basalis) than in the decidual or myometrial segments of the placental bed. The presence and deeper location of acute atherosis is associated with poorer pregnancy outcomes, more severe disease, earlier onset of preeclampsia, and a greater frequency of SGA neonates in patients with preeclampsia. Moreover, the idea that the presence of acute atherosis in the placenta may increase the risk of future cardiovascular disease in women with a history of preeclampsia is of growing concern. Therefore, placental examination is crucial for retrospective investigation of pregnancy complications and outcomes, and accurate placental pathology based on universal diagnostic criteria in patients with abnormal pregnancies is essential for clinicopathologic correlation.
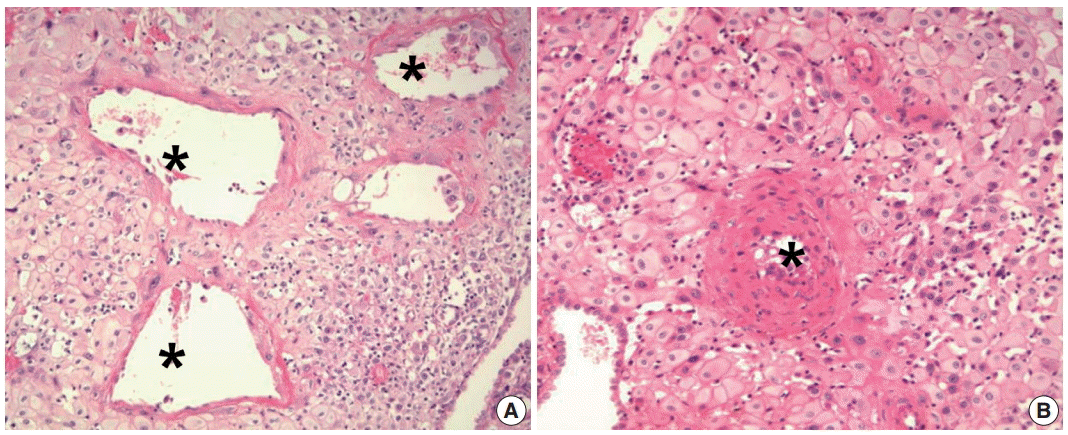
- Normal physiologic transformation of the uterine spiral arteries during early pregnancy is considered a foundation of successful pregnancy [58]. The spiral arteries are normally transformed into large dilated vessels, with dramatic structural changes to the vessel wall. The key findings of normal transformation of the spiral arteries are (1) dilation of the lumen, (2) trophoblast invasion into the vessel wall, and (3) replacement of the muscular and elastic tissue of the arterial wall by a thick fibrinoid material (Fig. 1A) [58,59]. These changes maximize the delivery of maternal blood to the intervillous space of the placenta, so that a sufficient blood supply through transformed spiral arteries enables the transfer of enough nutrition and oxygen from the mother to the fetus. Maternal blood from dilated spiral arteries meets fetal blood in the intervillous space, while the intervillous space drains blood back to the utero-placental veins [60]. For successful placentation, trophoblast invasion from the maternal-fetal interface to the myometrium through the decidua during the first 3 months of pregnancy is critical.
- Poor placentation is defined as the failure of physiologic transformation of spiral arteries and appears to arise from an inadequate or shallow trophoblast invasion [8,58,61]. Poor placentation is also a leading cause of preeclampsia and other abnormal pregnancies, such as spontaneous abortion, SGA, preterm labor and preterm PROM [7,9,62-65]. In preeclampsia, spiral arteries of the myometrial segment of the uterus fail to achieve physiologic transformation and retain thick walls and a narrow lumen; this is believed to be the main cause of uteroplacental ischemia (Fig. 1B) [7,9]. Uteroplacental ischemia can lead to the production of anti-angiogenic factors [66-68], such as soluble fms-like tyrosine kinase 1 [64,69], and endoglin [70,71], which can induce endothelial dysfunction and eventually predispose the pregnancy to preeclampsia. Spiral arteries that fail to achieve physiologic transformation are prone to develop acute atherosis [72-79].
POOR PLACENTATION AND ACUTE ATHEROSIS
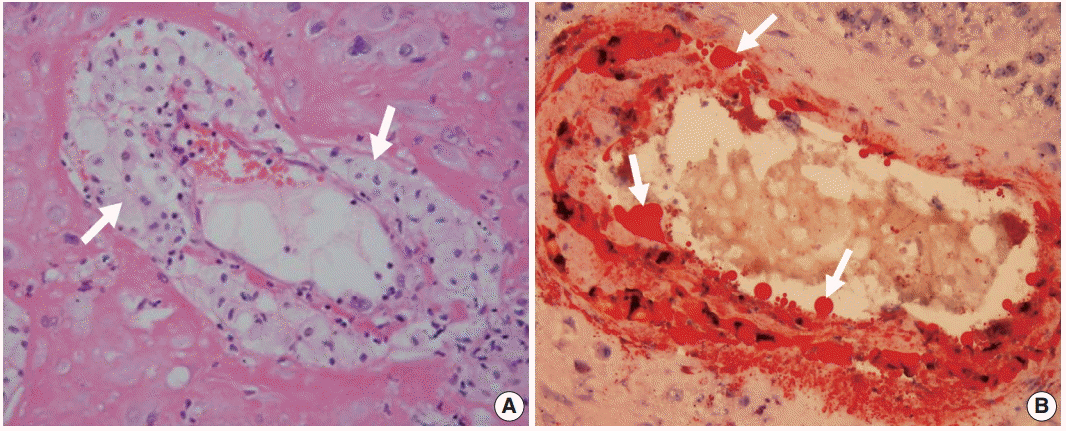
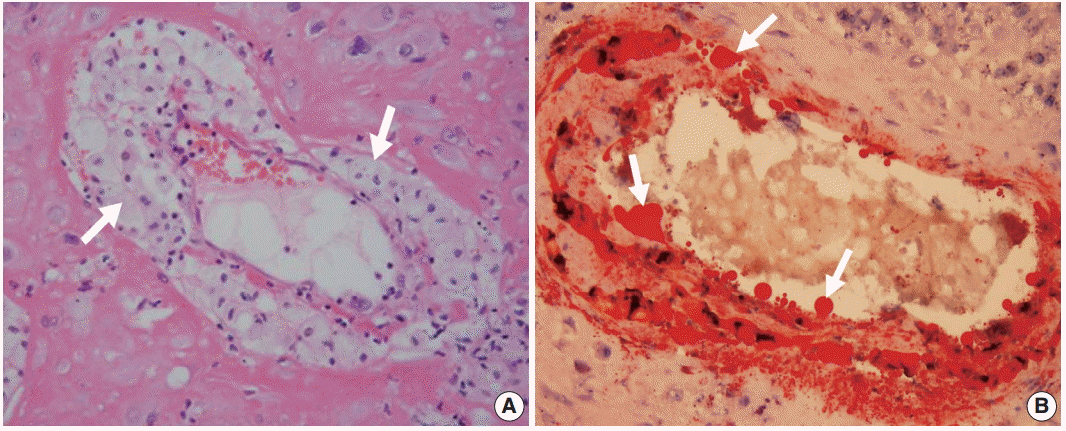
- Histologic findings of acute atherosis consist of the presence of fibrinoid necrosis of the artery wall, a subendothelial collection of lipid-laden macrophages, and vascular or perivascular lymphocytic infiltration in non-transformed uterine spiral arteries (Fig. 2A) [1,11]. Lipids in the spiral arteries with acute atherosis are stained red with oil-red O (Fig. 2B).
- Acute atherosis was more frequently observed in the spiral arteries of the decidua (parietalis or basalis) of the placenta than in the decidual or myometrial segments of the placental bed. The depth of acute atherosis is associated with the severity of preeclampsia. Briefly, the presence of atherosis in the myometrial segment is associated with a severe form and an earlier onset of preeclampsia than those without this lesion in the myometrial segment [51,80].
HISTOLOGIC CHARACTERISTICS AND TOPOGRAPHIC DISTRIBUTION OF ACUTE ATHEROSIS
- Acute atherosis used to be considered a characteristic finding of spiral arteries of patients with preeclampsia and has been reported to occur in 5% to 40% of patients with preeclampsia [5,7,8,10-13,16-19,23,24,26-31,40,42,45,46]. The variable frequency of acute atherosis may be explained by the following reasons: (1) variation in the number of tissue sections taken (size of sample), (2) location of tissue sections, (3) variation in tissue staining methods (hematoxylin and eosin [H&E] staining only or additional immunohistochemical staining) for the diagnosis of acute atherosis, and (4) differences in the pathologist’s diagnostic skill. In a previous study with over 14,000 placenta samples using only H&E staining for the diagnosis of acute atherosis and taking an average of two sections from each placenta based on routine histology laboratory tasks [51], the prevalence of acute atherosis in uncomplicated pregnancies was 0.4% and the frequency of acute atherosis varied based on the specific obstetrical syndrome: preeclampsia, 10%; fetal death, 9%; mid-trimester spontaneous abortion, 2.5%; SGA neonates without preeclampsia, 1.7%; and spontaneous preterm labor, 1.2%. Acute atherosis is associated with more severe disease, earlier onset of preeclampsia, and a greater frequency of SGA neonates in patients with preeclampsia [51,80].
FREQUENCY OF ACUTE ATHEROSIS
- A previous study found that acute atherosis is associated with an increased risk of placental lesions consistent with maternal underperfusion, fetal vascular thrombo-occlusive disease and chronic chorioamnionitis, but not with other chronic inflammatory lesions [81].
- The correlation between acute atherosis and chronic chorioamnionitis indicates the presence of circulating maternal T cells and adaptive immune response may also play a role in the genesis of acute atherosis [73,77,78,82-91].
- In chronic chorioamnionitis, maternal T cells infiltrating the chorion laeve cause trophoblast apoptosis, which resembles allograft rejection [92]. Chronic chorioamnionitis is also associated with anti-fetal HLA maternal sensitization [93] and complement deposition in the umbilical vein endothelium [94], which has been associated with a novel form of fetal systemic inflammatory response characterized by the over-expression of T-cell chemokines such as CXCL10 [95].
- Higher concentrations of CXCL9, CXCL10, and CXCL11 have been found in mothers with chronic placental inflammation compared to those without [92]. Similarly, T lymphocytes have been detected in the early stages of atheroma formation [96]. Moreover, differential expression of three interferon (IFN) gammainducible CXC chemokines, IFN-inducible protein 10 (CXCL10 or IP-10), monokine induced by IFN-y (CXCL9 or Mig) and IFN-inducible T-cell α chemoattractant (CXCL11 or I-TAC) were found in atherosclerosis [96].
PLACENTAL LESIONS ASSOCIATED WITH ACUTE ATHEROSIS
- Multiple factors including excessive decidual inflammation [42,47,48,97], immune dysregulation at the maternal-fetal interface [27] and immunological mismatch between the mother and fetus [27] have been proposed as causes or initiators of acute atherosis.
- Recently, Staff et al. [48] suggested four serial mechanisms for the development of acute atherosis, with excessive decidual inflammation as the final common pathway: (1) shear flow stress caused by abnormal blood flow in inadequately remodeled spiral arteries, (2) decidual inflammation, including maternal alloreactivity to feto-paternal HLA-C or minor histocompatibility antigens, (3) background (systemic) maternal inflammatory stress secondary to the changes induced by pregnancy and preeclampsia, and (4) maternal genetic predisposition (for example, polymorphism in regulator of G protein signaling 2).
- Elevation of signs of intravascular inflammation have been reported in both normal [98-101] and abnormal pregnancies, such as spontaneous preterm labor with intact membranes [102-108], preterm PROM [109-114], preeclampsia [115-144], SGA [119,134,136,138,145-154], and pyelonephritis [99,155-157]. Since chronic vascular inflammation is one of the main causes of atherosclerosis and acute atherosis, the possibility of activation of cholesterol crystal-induced inflammasome in macrophages [158] should be investigated as an important link between cholesterol metabolism and acute atherosis.
PATHOGENESIS OF ACUTE ATHEROSIS
- Maternal serum lipid level of patients with acute atherosis in the placenta
- In atherosclerosis, medium-sized and large arteries fueled by lipids, as well as the deposition of excess cholesterol in the blood stream, initiate atherosclerosis [159]. Similarly, acute atherosis in non-transformed uterine spiral arteries also show variable amounts of lipid deposition in the wall of spiral arteries, and it is stained red with oil-red O staining (Fig. 2B). Moreover, serum triglycerides are about 50% higher in preeclamptic women than in normal pregnant women. However, there are no differences in other lipid profiles, including total cholesterol and high-density lipoprotein [51,160-162]. Whether there are differences in low-density lipoprotein remains controversial [143-145,163].
- Acute atherosis and future cardiovascular risk
- Accumulating evidence suggests that women with a history of preeclampsia show a high prevalence of major cardiovascular risk factors [53-57]. Similarities between preeclampsia and atherosclerosis, as well as between acute atherosis of the spiral arteries and coronary atherosclerosis, have been observed. Intravascular inflammation [98,118,120,164,165], changes in lipid metabolism [162,166-168], and macrophage infiltration of the intima and media are seen in both acute atherosis and atherosclerosis [27,169,170]. Therefore, hyperlipidemia and abnormal lipid metabolism combined with intravascular inflammation can defect endothelial cell function and may lead to atherosclerosis in non-pregnant women who have a past history of preeclampsia [72-79]. We recommend that women who have a past history of preeclampsia be considered at high risk for cardiovascular disease and recommend implementation of regular screenings and prevention programs.
THE CLINICAL IMPLICATIONS OF ACUTE ATHEROSIS
- The presence and deeper location of acute atherosis is associated with worse pregnancy outcomes, more severe disease, earlier onset, and a greater frequency of SGA neonates in patients with preeclampsia. Moreover, the idea that acute atherosis in the placenta may increase the risk of future cardiovascular disease in women with a history of preeclampsia is of growing concern. Therefore, placental examination is crucial for investigation of pregnancy complications and outcomes, and accurate placental pathology based on universal diagnostic criteria in patients with abnormal pregnancies is essential for clinicopathologic correlation.
CONCLUSION
Acknowledgments
Fig. 1.Spiral artery changes during pregnancy. (A) Normal physiologic transformation of spiral arteries in a normal pregnancy. The lumen of the spiral artery (asterisks) is dilated. Trophoblastic cells are infiltrating the wall of the spiral artery. (B) Failure of physiologic transformation of spiral arteries in a patient with preeclampsia. The lumen of the arteries (asterisk) is not dilated. The medial layers of the spiral arteries are intact. Although many interstitial trophoblasts surround the spiral artery, trophoblasts have not invaded the vessel wall.
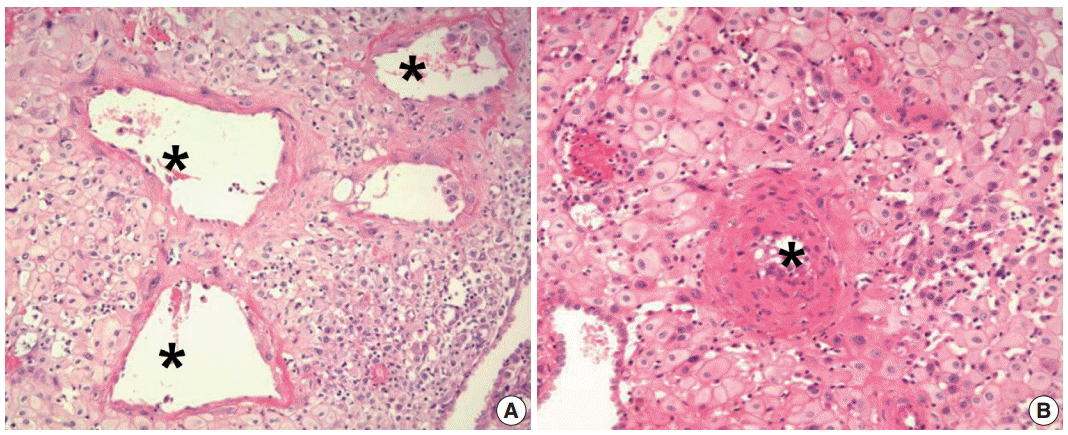

Fig. 2. Acute atherosis in decidual spiral arteries. (A) Many lipid-laden macrophages (arrows) are seen in the spiral arteries. (B) Acute atherosis on oil-red O staining. Fat droplets (arrows) in the non-transformed spiral artery are stained red.

- 1. Hertig AT. Vascular pathology in the hypertensive albuminuric toxemias of pregnancy. Clinics 1945; 4: 602-14.
- 2. Zeek PM, Assali NS. Vascular changes in the decidua associated with eclamptogenic toxemia of pregnancy. Am J Clin Pathol 1950; 20: 1099-109. ArticlePubMed
- 3. Hanssens M, Pijnenborg R, Keirse MJ, Vercruysse L, Verbist L, Van Assche FA. Renin-like immunoreactivity in uterus and placenta from normotensive and hypertensive pregnancies. Eur J Obstet Gynecol Reprod Biol 1998; 81: 177-84. ArticlePubMed
- 4. Sexton LI, Hertig AT, Reid DE, Kellogg FS, Patterson WA. Premature separation of the normally implanted placenta; a clinicopathological study of 476 cases. Am J Obstet Gynecol 1950; 59: 13-24. PubMed
- 5. Maqueo M, Chavezazuela J, Dosaldelavega M. Placental pathology in eclampsia and preeclampsia. Obstet Gynecol 1964; 24: 350-6. ArticlePubMed
- 6. Driscoll SG. The pathology of pregnancy complicated by diabetes mellitus. Med Clin N Am 1965; 49: 1053-67. Article
- 7. Robertson WB, Brosens I, Dixon HG. The pathological response of the vessels of the placental bed to hypertensive pregnancy. J Pathol Bacteriol 1967; 93: 581-92. ArticlePubMed
- 8. Brosens IA, Robertson WB, Dixon HG. The role of the spiral arteries in the pathogenesis of preeclampsia. Obstet Gynecol Annu 1972; 1: 177-91. PubMed
- 9. Brosens I, Renaer M. On the pathogenesis of placental infarcts in pre-eclampsia. J Obstet Gynaecol Br Commonw 1972; 79: 794-9. ArticlePubMed
- 10. Emmrich P, Birke R, Gödel E. Morphology of myometrial and decidual arteries in normal pregnancy, in toxemia of pregnancy, and in maternal diabetes (author’s transl). Pathol Microbiol (Basel) 1975; 43: 38-61. PubMed
- 11. De Wolf F, Robertson WB, Brosens I. The ultrastructure of acute atherosis in hypertensive pregnancy. Am J Obstet Gynecol 1975; 123: 164-74. ArticlePubMed
- 12. Robertson WB, Brosens I, Dixon G. Uteroplacental vascular pathology. Eur J Obstet Gynecol Reprod Biol 1975; 5: 47-65. ArticlePubMed
- 13. Robertson WB, Brosens I, Dixon G. Maternal uterine vascular lesions in the hypertensive complications of pregnancy. Perspect Nephrol Hypertens 1976; 5: 115-27. PubMed
- 14. De Wolf F, Brosens I, Renaer M. Fetal growth retardation and the maternal arterial supply of the human placenta in the absence of sustained hypertension. Br J Obstet Gynaecol 1980; 87: 678-85. ArticlePubMed
- 15. Abramowsky CR, Vegas ME, Swinehart G, Gyves MT. Decidual vasculopathy of the placenta in lupus erythematosus. N Engl J Med 1980; 303: 668-72. ArticlePubMed
- 16. Kitzmiller JL, Watt N, Driscoll SG. Decidual arteriopathy in hypertension and diabetes in pregnancy: immunofluorescent studies. Am J Obstet Gynecol 1981; 141: 773-9. ArticlePubMed
- 17. Sheppard BL, Bonnar J. An ultrastructural study of utero-placental spiral arteries in hypertensive and normotensive pregnancy and fetal growth retardation. Br J Obstet Gynaecol 1981; 88: 695-705. ArticlePubMed
- 18. De Wolf F, Carreras LO, Moerman P, Vermylen J, Van Assche A, Renaer M. Decidual vasculopathy and extensive placental infarction in a patient with repeated thromboembolic accidents, recurrent fetal loss, and a lupus anticoagulant. Am J Obstet Gynecol 1982; 142: 829-34. ArticlePubMed
- 19. Hustin J, Foidart JM, Lambotte R. Maternal vascular lesions in preeclampsia and intrauterine growth retardation: light microscopy and immunofluorescence. Placenta 1983; 4 Spec No: 489-98. PubMed
- 20. Althabe O, Labarrere C, Telenta M. Maternal vascular lesions in placentae of small-for-gestational-age infants. Placenta 1985; 6: 265-76. ArticlePubMed
- 21. Labarrere C, Alonso J, Manni J, Domenichini E, Althabe O. Immunohistochemical findings in acute atherosis associated with intrauterine growth retardation. Am J Reprod Immunol Microbiol 1985; 7: 149-55. ArticlePubMedPDF
- 22. Labarrere C, Althabe O. Chronic villitis of unknown etiology and maternal arterial lesions in preeclamptic pregnancies. Eur J Obstet Gynecol Reprod Biol 1985; 20: 1-11. ArticlePubMed
- 23. Khong TY, De Wolf F, Robertson WB, Brosens I. Inadequate maternal vascular response to placentation in pregnancies complicated by pre-eclampsia and by small-for-gestational age infants. Br J Obstet Gynaecol 1986; 93: 1049-59. ArticlePubMed
- 24. McFadyen IR, Price AB, Geirsson RT. The relation of birthweight to histological appearances in vessels of the placental bed. Br J Obstet Gynaecol 1986; 93: 476-81. ArticlePubMed
- 25. Labarrere CA, Catoggio LJ, Mullen EG, Althabe OH. Placental lesions in maternal autoimmune diseases. Am J Reprod Immunol Microbiol 1986; 12: 78-86. ArticlePubMedPDF
- 26. Khong TY, Pearce JM, Robertson WB. Acute atherosis in preeclampsia: maternal determinants and fetal outcome in the presence of the lesion. Am J Obstet Gynecol 1987; 157: 360-3. ArticlePubMed
- 27. Labarrere CA. Acute atherosis: a histopathological hallmark of immune aggression? Placenta 1988; 9: 95-108. ArticlePubMed
- 28. Frusca T, Morassi L, Pecorelli S, Grigolato P, Gastaldi A. Histological features of uteroplacental vessels in normal and hypertensive patients in relation to birthweight. Br J Obstet Gynaecol 1989; 96: 835-9. ArticlePubMed
- 29. Khong TY. Acute atherosis in pregnancies complicated by hypertension, small-for-gestational-age infants, and diabetes mellitus. Arch Pathol Lab Med 1991; 115: 722-5. PubMed
- 30. Meekins JW, Pijnenborg R, Hanssens M, McFadyen IR, van Asshe A. A study of placental bed spiral arteries and trophoblast invasion in normal and severe pre-eclamptic pregnancies. Br J Obstet Gynaecol 1994; 101: 669-74. ArticlePubMedPDF
- 31. Meekins JW, Pijnenborg R, Hanssens M, van Assche A, McFadyen IR. Immunohistochemical detection of lipoprotein(a) in the wall of placental bed spiral arteries in normal and severe preeclamptic pregnancies. Placenta 1994; 15: 511-24. ArticlePubMed
- 32. Salafia CM, Parke AL. Placental pathology in systemic lupus erythematosus and phospholipid antibody syndrome. Rheum Dis Clin North Am 1997; 23: 85-97. ArticlePubMed
- 33. Brosens I, Dixon HG, Robertson WB. Fetal growth retardation and the arteries of the placental bed. Br J Obstet Gynaecol 1977; 84: 656-63. ArticlePubMed
- 34. Gerretsen G, Huisjes HJ, Elema JD. Morphological changes of the spiral arteries in the placental bed in relation to pre-eclampsia and fetal growth retardation. Br J Obstet Gynaecol 1981; 88: 876-81. PubMed
- 35. Nayar R, Lage JM. Placental changes in a first trimester missed abortion in maternal systemic lupus erythematosus with antiphospholipid syndrome; a case report and review of the literature. Hum Pathol 1996; 27: 201-6. ArticlePubMed
- 36. Hayati AR, Azizah A, Wahidah A. Incidence of acute atherosis in complete molar pregnancy. Malays J Pathol 1998; 20: 113-4. PubMed
- 37. Khong TY, Hague WM. The placenta in maternal hyperhomocysteinaemia. Br J Obstet Gynaecol 1999; 106: 273-8. ArticlePubMed
- 38. Ogishima D, Matsumoto T, Nakamura Y, Yoshida K, Kuwabara Y. Placental pathology in systemic lupus erythematosus with antiphospholipid antibodies. Pathol Int 2000; 50: 224-9. ArticlePubMed
- 39. Sebire NJ, Rees H, Paradinas F, et al. Extravillus endovascular implantation site trophoblast invasion is abnormal in complete versus partial molar pregnancies. Placenta 2001; 22: 725-8. ArticlePubMed
- 40. Moldenhauer JS, Stanek J, Warshak C, Khoury J, Sibai B. The frequency and severity of placental findings in women with preeclampsia are gestational age dependent. Am J Obstet Gynecol 2003; 189: 1173-7. ArticlePubMed
- 41. Faye-Petersen O, Heller DS, Joshi VV. Handbook of placental pathology. 2nd ed. London: Taylor & Francis, 2006; 53-78.
- 42. Harsem NK, Roald B, Braekke K, Staff AC. Acute atherosis in decidual tissue: not associated with systemic oxidative stress in preeclampsia. Placenta 2007; 28: 958-64. ArticlePubMed
- 43. Fox H, Sebire NJ. Pathology of theplacenta. 3rd ed. Philadelphia: Saunders-Elsevier, 2007; 147-86.
- 44. Brosens I, Khong TY. Defective spiral artery remodeling. In: Pijnenborg R, Brosens I, Romero R, eds. Placental bed disorders. Cambridge: Cambridge University Press, 2010; 11.
- 45. Staff AC, Dechend R, Pijnenborg R. Learning from the placenta: acute atherosis and vascular remodeling in preeclampsia-novel aspects for atherosclerosis and future cardiovascular health. Hypertension 2010; 56: 1026-34. ArticlePubMed
- 46. Stevens DU, Al-Nasiry S, Bulten J, Spaanderman ME. Decidual vasculopathy in preeclampsia: lesion characteristics relate to disease severity and perinatal outcome. Placenta 2013; 34: 805-9. ArticlePubMed
- 47. Staff AC, Dechend R, Redman CW. Review: preeclampsia, acute atherosis of the spiral arteries and future cardiovascular disease: two new hypotheses. Placenta 2013; 34 Suppl: S73-8. Article
- 48. Staff AC, Johnsen GM, Dechend R, Redman CW. Preeclampsia and uteroplacental acute atherosis: immune and inflammatory factors. J Reprod Immunol 2014; 101-102: 120-6. ArticlePubMed
- 49. Staff AC, Redman CW. IFPA Award in Placentology Lecture: preeclampsia, the decidual battleground and future maternal cardiovascular disease. Placenta 2014; 35 Suppl: S26-31. Article
- 50. Mitchell RN. Graft vascular disease: immune response meets the vessel wall. Annu Rev Pathol 2009; 4: 19-47. ArticlePubMed
- 51. Kim YM, Chaemsaithong P, Romero R, et al. The frequency of acute atherosis in normal pregnancy and preterm labor, preeclampsia, small-for-gestational age, fetal death and midtrimester spontaneous abortion. J Matern Fetal Neonatal Med 2015; 28: 2001-9. ArticlePubMed
- 52. Pijnenborg R, Vercruysse L, Hanssens M. The uterine spiral arteries in human pregnancy: facts and controversies. Placenta 2006; 27: 939-58. ArticlePubMed
- 53. van Rijn BB, Nijdam ME, Bruinse HW, et al. Cardiovascular disease risk factors in women with a history of early-onset preeclampsia. Obstet Gynecol 2013; 121: 1040-8. ArticlePubMed
- 54. Veerbeek JH, Hermes W, Breimer AY, et al. Cardiovascular disease risk factors after early-onset preeclampsia, late-onset preeclampsia, and pregnancy-induced hypertension. Hypertension 2015; 65: 600-6. ArticlePubMed
- 55. Sattar N, Ramsay J, Crawford L, Cheyne H, Greer IA. Classic and novel risk factor parameters in women with a history of preeclampsia. Hypertension 2003; 42: 39-42. ArticlePubMed
- 56. Berends AL, de Groot CJ, Sijbrands EJ, et al. Shared constitutional risks for maternal vascular-related pregnancy complications and future cardiovascular disease. Hypertension 2008; 51: 1034-41. ArticlePubMed
- 57. van Rijn BB, Veerbeek JH, Scholtens LC, et al. C-reactive protein and fibrinogen levels as determinants of recurrent preeclampsia: a prospective cohort study. J Hypertens 2014; 32: 408-14. PubMed
- 58. Brosens I, Robertson WB, Dixon HG. The physiological response of the vessels of the placental bed to normal pregnancy. J Pathol Bacteriol 1967; 93: 569-79. ArticlePubMed
- 59. Espinoza J, Romero R, Kim YM, et al. Normal and abnormal transformation of the spiral arteries during pregnancy. J Perinat Med 2006; 34: 447-58. ArticlePubMedPMC
- 60. Freese UE. The uteroplacental vascular relationship in the human. Am J Obstet Gynecol 1968; 101: 8-16. ArticlePubMed
- 61. Brosens IA. Morphological changes in the utero-placental bed in pregnancy hypertension. Clin Obstet Gynaecol 1977; 4: 573-93. ArticlePubMed
- 62. Kim YM, Chaiworapongsa T, Gomez R, et al. Failure of physiologic transformation of the spiral arteries in the placental bed in preterm premature rupture of membranes. Am J Obstet Gynecol 2002; 187: 1137-42. ArticlePubMed
- 63. Kim YM, Bujold E, Chaiworapongsa T, et al. Failure of physiologic transformation of the spiral arteries in patients with preterm labor and intact membranes. Am J Obstet Gynecol 2003; 189: 1063-9. ArticlePubMed
- 64. Redman CW, Sargent IL. Latest advances in understanding preeclampsia. Science 2005; 308: 1592-4. ArticlePubMed
- 65. Steegers EA, von Dadelszen P, Duvekot JJ, Pijnenborg R. Pre-eclampsia. Lancet 2010; 376: 631-44. ArticlePubMed
- 66. Makris A, Thornton C, Thompson J, et al. Uteroplacental ischemia results in proteinuric hypertension and elevated sFLT-1. Kidney Int 2007; 71: 977-84. ArticlePubMed
- 67. Bujold E, Romero R, Chaiworapongsa T, et al. Evidence supporting that the excess of the sVEGFR-1 concentration in maternal plasma in preeclampsia has a uterine origin. J Matern Fetal Neonatal Med 2005; 18: 9-16. ArticlePubMed
- 68. Kusanovic JP, Romero R, Chaiworapongsa T, et al. A prospective cohort study of the value of maternal plasma concentrations of angiogenic and anti-angiogenic factors in early pregnancy and midtrimester in the identification of patients destined to develop preeclampsia. J Matern Fetal Neonatal Med 2009; 22: 1021-38. ArticlePubMedPMC
- 69. Maynard SE, Min JY, Merchan J, et al. Excess placental soluble fmslike tyrosine kinase 1 (sFlt1) may contribute to endothelial dysfunction, hypertension, and proteinuria in preeclampsia. J Clin Invest 2003; 111: 649-58. ArticlePubMedPMC
- 70. Lain KY, Roberts JM. Contemporary concepts of the pathogenesis and management of preeclampsia. JAMA 2002; 287: 3183-6. ArticlePubMed
- 71. Venkatesha S, Toporsian M, Lam C, et al. Soluble endoglin contributes to the pathogenesis of preeclampsia. Nat Med 2006; 12: 642-9. ArticlePubMedPDF
- 72. Libby P, Ridker PM, Maseri A. Inflammation and atherosclerosis. Circulation 2002; 105: 1135-43. ArticlePubMed
- 73. Libby P. Inflammation in atherosclerosis. Nature 2002; 420: 868-74. ArticlePubMedPDF
- 74. Young JL, Libby P, Schönbeck U. Cytokines in the pathogenesis of atherosclerosis. Thromb Haemost 2002; 88: 554-67. ArticlePubMed
- 75. Hansson GK, Libby P. The immune response in atherosclerosis: a double-edged sword. Nat Rev Immunol 2006; 6: 508-19. ArticlePubMedPDF
- 76. Libby P. Inflammatory mechanisms: the molecular basis of inflammation and disease. Nutr Rev 2007; 65(12 Pt 2): S140-6. ArticlePubMed
- 77. Rocha VZ, Libby P. Obesity, inflammation, and atherosclerosis. Nat Rev Cardiol 2009; 6: 399-409. ArticlePubMedPDF
- 78. Galkina E, Ley K. Immune and inflammatory mechanisms of atherosclerosis (*). Annu Rev Immunol 2009; 27: 165-97. ArticlePubMedPMC
- 79. Libby P, Okamoto Y, Rocha VZ, Folco E. Inflammation in atherosclerosis: transition from theory to practice. Circ J 2010; 74: 213-20. ArticlePubMed
- 80. Brosens I, Pijnenborg R, Vercruysse L, Romero R. The “Great Obstetrical Syndromes” are associated with disorders of deep placentation. Am J Obstet Gynecol 2011; 204: 193-201. ArticlePubMedPMC
- 81. Kim YM, Chaemsaithong P, Romero R, et al. Placental lesions associated with acute atherosis. J Matern Fetal Neonatal Med 2015; 28: 1554-62. ArticlePubMed
- 82. Libby P. Atherosclerosis: disease biology affecting the coronary vasculature. Am J Cardiol 2006; 98: 3Q-9Q. Article
- 83. Libby P, Ridker PM, Hansson GK; Leducq Transatantic Network on Atherothrombosis. Inflammation in atherosclerosis: from pathophysiology to practice. J Am Coll Cardiol 2009; 54: 2129-38. PubMedPMC
- 84. Packard RR, Lichtman AH, Libby P. Innate and adaptive immunity in atherosclerosis. Semin Immunopathol 2009; 31: 5-22. ArticlePubMedPMCPDF
- 85. Andersson J, Libby P, Hansson GK. Adaptive immunity and atherosclerosis. Clin Immunol 2010; 134: 33-46. ArticlePubMed
- 86. Libby P, Ridker PM, Hansson GK. Progress and challenges in translating the biology of atherosclerosis. Nature 2011; 473: 317-25. ArticlePubMedPDF
- 87. Legein B, Temmerman L, Biessen EA, Lutgens E. Inflammation and immune system interactions in atherosclerosis. Cell Mol Life Sci 2013; 70: 3847-69. ArticlePubMedPMCPDF
- 88. Libby P, Lichtman AH, Hansson GK. Immune effector mechanisms implicated in atherosclerosis: from mice to humans. Immunity 2013; 38: 1092-104. ArticlePubMedPMC
- 89. Rosenfeld ME. Inflammation and atherosclerosis: direct versus indirect mechanisms. Curr Opin Pharmacol 2013; 13: 154-60. ArticlePubMedPMC
- 90. Woollard KJ. Immunological aspects of atherosclerosis. Clin Sci (Lond) 2013; 125: 221-35. ArticlePubMedPDF
- 91. Ait-Oufella H, Sage AP, Mallat Z, Tedgui A. Adaptive (T and B cells) immunity and control by dendritic cells in atherosclerosis. Circ Res 2014; 114: 1640-60. ArticlePubMed
- 92. Kim CJ, Romero R, Kusanovic JP, et al. The frequency, clinical significance, and pathological features of chronic chorioamnionitis: a lesion associated with spontaneous preterm birth. Mod Pathol 2010; 23: 1000-11. ArticlePubMedPMCPDF
- 93. Lee J, Romero R, Xu Y, et al. Maternal HLA panel-reactive antibodies in early gestation positively correlate with chronic chorioamnionitis: evidence in support of the chronic nature of maternal anti-fetal rejection. Am J Reprod Immunol 2011; 66: 510-26. ArticlePubMedPMC
- 94. Lee J, Romero R, Xu Y, et al. A signature of maternal anti-fetal rejection in spontaneous preterm birth: chronic chorioamnionitis, anti-human leukocyte antigen antibodies, and C4d. PLoS One 2011; 6: e16806. ArticlePubMedPMC
- 95. Lee J, Kim JS, Park JW, et al. Chronic chorioamnionitis is the most common placental lesion in late preterm birth. Placenta 2013; 34: 681-9. ArticlePubMed
- 96. Mach F, Sauty A, Iarossi AS, et al. Differential expression of three T lymphocyte-activating CXC chemokines by human atheroma-associated cells. J Clin Invest 1999; 104: 1041-50. ArticlePubMedPMC
- 97. Pijnenborg R, McLaughlin PJ, Vercruysse L, et al. Immunolocalization of tumour necrosis factor-alpha (TNF-alpha) in the placental bed of normotensive and hypertensive human pregnancies. Placenta 1998; 19: 231-9. ArticlePubMed
- 98. Sacks GP, Studena K, Sargent K, Redman CW. Normal pregnancy and preeclampsia both produce inflammatory changes in peripheral blood leukocytes akin to those of sepsis. Am J Obstet Gynecol 1998; 179: 80-6. ArticlePubMed
- 99. Naccasha N, Gervasi MT, Chaiworapongsa T, et al. Phenotypic and metabolic characteristics of monocytes and granulocytes in normal pregnancy and maternal infection. Am J Obstet Gynecol 2001; 185: 1118-23. ArticlePubMed
- 100. Brenner B. Haemostatic changes in pregnancy. Thromb Res 2004; 114: 409-14. ArticlePubMed
- 101. Watts DH, Krohn MA, Wener MH, Eschenbach DA. C-reactive protein in normal pregnancy. Obstet Gynecol 1991; 77: 176-80. ArticlePubMed
- 102. Gervasi MT, Chaiworapongsa T, Naccasha N, et al. Phenotypic and metabolic characteristics of maternal monocytes and granulocytes in preterm labor with intact membranes. Am J Obstet Gynecol 2001; 185: 1124-9. ArticlePubMed
- 103. Pitiphat W, Gillman MW, Joshipura KJ, Williams PL, Douglass CW, Rich-Edwards JW. Plasma C-reactive protein in early pregnancy and preterm delivery. Am J Epidemiol 2005; 162: 1108-13. ArticlePubMedPMC
- 104. Gotsch F, Romero R, Kusanovic JP, et al. The anti-inflammatory limb of the immune response in preterm labor, intra-amniotic infection/inflammation, and spontaneous parturition at term: a role for interleukin-10. J Matern Fetal Neonatal Med 2008; 21: 529-47. ArticlePubMedPMC
- 105. Kim MA, Lee BS, Park YW, Seo K. Serum markers for prediction of spontaneous preterm delivery in preterm labour. Eur J Clin Invest 2011; 41: 773-80. ArticlePubMed
- 106. Laudanski P, Raba G, Kuc P, Lemancewicz A, Kisielewski R, Laudanski T. Assessment of the selected biochemical markers in predicting preterm labour. J Matern Fetal Neonatal Med 2012; 25: 2696-9. ArticlePubMed
- 107. Cruciani L, Romero R, Vaisbuch E, et al. Pentraxin 3 in maternal circulation: an association with preterm labor and preterm PROM, but not with intra-amniotic infection/inflammation. J Matern Fetal Neonatal Med 2010; 23: 1097-105. ArticlePubMedPMC
- 108. Stampalija T, Chaiworapongsa T, Romero R, et al. Soluble ST2, a modulator of the inflammatory response, in preterm and term labor. J Matern Fetal Neonatal Med 2014; 27: 111-21. ArticlePubMedPMC
- 109. Ismail MA, Zinaman MJ, Lowensohn RI, Moawad AH. The significance of C-reactive protein levels in women with premature rupture of membranes. Am J Obstet Gynecol 1985; 151: 541-4. ArticlePubMed
- 110. Yoon BH, Jun JK, Park KH, Syn HC, Gomez R, Romero R. Serum C-reactive protein, white blood cell count, and amniotic fluid white blood cell count in women with preterm premature rupture of membranes. Obstet Gynecol 1996; 88: 1034-40. ArticlePubMed
- 111. Gervasi MT, Chaiworapongsa T, Naccasha N, et al. Maternal intravascular inflammation in preterm premature rupture of membranes. J Matern Fetal Neonatal Med 2002; 11: 171-5. ArticlePubMed
- 112. Loukovaara MJ, Alfthan HV, Kurki MT, Hiilesmaa VK, Andersson SH. Serum highly sensitive C-reactive protein in preterm premature rupture of membranes. Eur J Obstet Gynecol Reprod Biol 2003; 110: 26-8. ArticlePubMed
- 113. Moghaddam Banaem L, Mohamadi B, Asghari Jaafarabadi M, Aliyan Moghadam N. Maternal serum C-reactive protein in early pregnancy and occurrence of preterm premature rupture of membranes and preterm birth. J Obstet Gynaecol Res 2012; 38: 780-6. ArticlePubMed
- 114. Gulati S, Agrawal S, Raghunandan C, et al. Maternal serum interleukin-6 and its association with clinicopathological infectious morbidity in preterm premature rupture of membranes: a prospective cohort study. J Matern Fetal Neonatal Med 2012; 25: 1428-32. ArticlePubMed
- 115. Vince GS, Starkey PM, Austgulen R, Kwiatkowski D, Redman CW. Interleukin-6, tumour necrosis factor and soluble tumour necrosis factor receptors in women with pre-eclampsia. Br J Obstet Gynaecol 1995; 102: 20-5. PubMed
- 116. Hamai Y, Fujii T, Yamashita T, et al. Evidence for an elevation in serum interleukin-2 and tumor necrosis factor-alpha levels before the clinical manifestations of preeclampsia. Am J Reprod Immunol 1997; 38: 89-93. PubMed
- 117. Conrad KP, Miles TM, Benyo DF. Circulating levels of immunoreactive cytokines in women with preeclampsia. Am J Reprod Immunol 1998; 40: 102-11. ArticlePubMed
- 118. Redman CW, Sacks GP, Sargent IL. Preeclampsia: an excessive maternal inflammatory response to pregnancy. Am J Obstet Gynecol 1999; 180(2 Pt 1): 499-506. ArticlePubMed
- 119. Sabatier F, Bretelle F, D’Ercole C, Boubli L, Sampol J, Dignat-George F. Neutrophil activation in preeclampsia and isolated intrauterine growth restriction. Am J Obstet Gynecol 2000; 183: 1558-63. ArticlePubMed
- 120. Gervasi MT, Chaiworapongsa T, Pacora P, et al. Phenotypic and metabolic characteristics of monocytes and granulocytes in preeclampsia. Am J Obstet Gynecol 2001; 185: 792-7. ArticlePubMed
- 121. Teran E, Escudero C, Moya W, Flores M, Vallance P, Lopez-Jaramillo P. Elevated C-reactive protein and pro-inflammatory cytokines in Andean women with pre-eclampsia. Int J Gynaecol Obstet 2001; 75: 243-9. ArticlePubMedPDF
- 122. Chaiworapongsa T, Romero R, Yoshimatsu J, et al. Soluble adhesion molecule profile in normal pregnancy and pre-eclampsia. J Matern Fetal Neonatal Med 2002; 12: 19-27. ArticlePubMed
- 123. Chaiworapongsa T, Gervasi MT, Refuerzo J, et al. Maternal lymphocyte subpopulations (CD45RA+ and CD45RO+) in preeclampsia. Am J Obstet Gynecol 2002; 187: 889-93. ArticlePubMed
- 124. Velzing-Aarts FV, Muskiet FA, van der Dijs FP, Duits AJ. High serum interleukin-8 levels in afro-caribbean women with pre-eclampsia: relations with tumor necrosis factor-alpha, duffy negative phenotype and von Willebrand factor. Am J Reprod Immunol 2002; 48: 319-22. PubMed
- 125. Serin IS, Ozcelik B, Basbug M, Kilic H, Okur D, Erez R. Predictive value of tumor necrosis factor alpha (TNF-alpha) in preeclampsia. Eur J Obstet Gynecol Reprod Biol 2002; 100: 143-5. PubMed
- 126. Belo L, Santos-Silva A, Caslake M, et al. Neutrophil activation and C-reactive protein concentration in preeclampsia. Hypertens Pregnancy 2003; 22: 129-41. ArticlePubMed
- 127. Levine RJ, Qian C, Leshane ES, et al. Two-stage elevation of cellfree fetal DNA in maternal sera before onset of preeclampsia. Am J Obstet Gynecol 2004; 190: 707-13. ArticlePubMed
- 128. Kocyigit Y, Atamer Y, Atamer A, Tuzcu A, Akkus Z. Changes in serum levels of leptin, cytokines and lipoprotein in pre-eclamptic and normotensive pregnant women. Gynecol Endocrinol 2004; 19: 267-73. ArticlePubMed
- 129. Freeman DJ, McManus F, Brown EA, et al. Short- and long-term changes in plasma inflammatory markers associated with preeclampsia. Hypertension 2004; 44: 708-14. ArticlePubMed
- 130. Kim YM, Romero R, Oh SY, et al. Toll-like receptor 4: a potential link between “danger signals,” the innate immune system, and preeclampsia? Am J Obstet Gynecol 2005; 193(3 Pt 2): 921-7. ArticlePubMed
- 131. Braekke K, Holthe MR, Harsem NK, Fagerhol MK, Staff AC. Calprotectin, a marker of inflammation, is elevated in the maternal but not in the fetal circulation in preeclampsia. Am J Obstet Gynecol 2005; 193: 227-33. ArticlePubMed
- 132. Enquobahrie DA, Williams MA, Qiu C, Woelk GB, Mahomed K. Maternal plasma transforming growth factor-beta1 concentrations in preeclamptic and normotensive pregnant Zimbabwean women. J Matern Fetal Neonatal Med 2005; 17: 343-8. PubMed
- 133. Jonsson Y, Rubèr M, Matthiesen L, et al. Cytokine mapping of sera from women with preeclampsia and normal pregnancies. J Reprod Immunol 2006; 70: 83-91. ArticlePubMed
- 134. Kusanovic JP, Romero R, Hassan SS, et al. Maternal serum soluble CD30 is increased in normal pregnancy, but decreased in preeclampsia and small for gestational age pregnancies. J Matern Fetal Neonatal Med 2007; 20: 867-78. ArticlePubMedPMC
- 135. Sharma A, Satyam A, Sharma JB. Leptin, IL-10 and inflammatory markers (TNF-alpha, IL-6 and IL-8) in pre-eclamptic, normotensive pregnant and healthy non-pregnant women. Am J Reprod Immunol 2007; 58: 21-30. PubMed
- 136. Laskowska M, Laskowska K, Leszczyn´ska-Gorzelak B, Oleszczuk J. Comparative analysis of the maternal and umbilical interleukin-8 levels in normal pregnancies and in pregnancies complicated by preeclampsia with intrauterine normal growth and intrauterine growth retardation. J Matern Fetal Neonatal Med 2007; 20: 527-32. ArticlePubMed
- 137. Sibai B, Romero R, Klebanoff MA, et al. Maternal plasma concentrations of the soluble tumor necrosis factor receptor 2 are increased prior to the diagnosis of preeclampsia. Am J Obstet Gynecol 2009; 200:630: e1-8. Article
- 138. Ogge G, Romero R, Chaiworapongsa T, et al. Leukocytes of pregnant women with small-for-gestational age neonates have a different phenotypic and metabolic activity from those of women with preeclampsia. J Matern Fetal Neonatal Med 2010; 23: 476-87. ArticlePubMedPMC
- 139. Soto E, Romero R, Richani K, et al. Preeclampsia and pregnancies with small-for-gestational age neonates have different profiles of complement split products. J Matern Fetal Neonatal Med 2010; 23: 646-57. ArticlePubMedPMC
- 140. Szarka A, Rigó J Jr, Lázár L, Beko G, Molvarec A. Circulating cytokines, chemokines and adhesion molecules in normal pregnancy and preeclampsia determined by multiplex suspension array. BMC Immunol 2010; 11: 59.ArticlePubMedPMCPDF
- 141. Xie C, Yao MZ, Liu JB, Xiong LK. A meta-analysis of tumor necrosis factor-alpha, interleukin-6, and interleukin-10 in preeclampsia. Cytokine 2011; 56: 550-9. ArticlePubMed
- 142. Twina G, Sheiner E, Shahaf G, et al. Lower circulation levels and activity of alpha-1 antitrypsin in pregnant women with severe preeclampsia. J Matern Fetal Neonatal Med 2012; 25: 2667-70. PubMed
- 143. Stampalija T, Chaiworapongsa T, Romero R, et al. Maternal plasma concentrations of sST2 and angiogenic/anti-angiogenic factors in preeclampsia. J Matern Fetal Neonatal Med 2013; 26: 1359-70. ArticlePubMedPMC
- 144. Sahin S, Ozakpinar OB, Eroglu M, et al. The impact of platelet functions and inflammatory status on the severity of preeclampsia. J Matern Fetal Neonatal Med 2015; 28: 643-8. ArticlePubMed
- 145. Labarrere C, Manni J, Salas P, Althabe O. Intrauterine growth retardation of unknown etiology. I. Serum complement and circulating immune complexes in mothers and infants. Am J Reprod Immunol Microbiol 1985; 8: 87-93. ArticlePubMedPDF
- 146. Labarrere CA, Althabe OH. Intrauterine growth retardation of unknown etiology: II. Serum complement and circulating immune complexes in maternal sera and their relationship with parity and chronic villitis. Am J Reprod Immunol Microbiol 1986; 12: 4-6. ArticlePubMedPDF
- 147. Johnston TA, Greer IA, Dawes J, Calder AA. Neutrophil activation in small for gestational age pregnancies. Br J Obstet Gynaecol 1991; 98: 105-6. ArticlePubMed
- 148. Johnson MR, Anim-Nyame N, Johnson P, Sooranna SR, Steer PJ. Does endothelial cell activation occur with intrauterine growth restriction? BJOG 2002; 109: 836-9. ArticlePubMed
- 149. Coata G, Pennacchi L, Bini V, Liotta L, Di Renzo GC. Soluble adhesion molecules: marker of pre-eclampsia and intrauterine growth restriction. J Matern Fetal Neonatal Med 2002; 12: 28-34. ArticlePubMed
- 150. Tjoa ML, van Vugt JM, Go AT, Blankenstein MA, Oudejans CB, van Wijk IJ. Elevated C-reactive protein levels during first trimester of pregnancy are indicative of preeclampsia and intrauterine growth restriction. J Reprod Immunol 2003; 59: 29-37. ArticlePubMed
- 151. Cetin I, Cozzi V, Pasqualini F, et al. Elevated maternal levels of the long pentraxin 3 (PTX3) in preeclampsia and intrauterine growth restriction. Am J Obstet Gynecol 2006; 194: 1347-53. ArticlePubMed
- 152. Girardi G, Yarilin D, Thurman JM, Holers VM, Salmon JE. Complement activation induces dysregulation of angiogenic factors and causes fetal rejection and growth restriction. J Exp Med 2006; 203: 2165-75. ArticlePubMedPMCPDF
- 153. Laskowska M, Laskowska K, Leszczyn´ska-Gorzelak B, Oleszczuk J. Maternal and umbilical sTNF-R1 in preeclamptic pregnancies with intrauterine normal and growth retarded fetus. Hypertens Pregnancy 2007; 26: 13-21. ArticlePubMed
- 154. Erez O, Romero R, Hoppensteadt D, et al. Tissue factor and its natural inhibitor in pre-eclampsia and SGA. J Matern Fetal Neonatal Med 2008; 21: 855-69. ArticlePubMedPMC
- 155. Duff P. Pyelonephritis in pregnancy. Clin Obstet Gynecol 1984; 27: 17-31. ArticlePubMed
- 156. Gilstrap LC 3rd, Lucas MJ. Urinary tract infections in women. Curr Opin Obstet Gynecol 1990; 2: 643-8. PubMed
- 157. Soto E, Richani K, Romero R, et al. Increased concentration of the complement split product C5a in acute pyelonephritis during pregnancy. J Matern Fetal Neonatal Med 2005; 17: 247-52. ArticlePubMedPMC
- 158. Rajamäki K, Lappalainen J, Oörni K, et al. Cholesterol crystals activate the NLRP3 inflammasome in human macrophages: a novel link between cholesterol metabolism and inflammation. PLoS One 2010; 5: e11765. ArticlePubMedPMC
- 159. Small DM, Shipley GG. Physical-chemical basis of lipid deposition in atherosclerosis. Science 1974; 185: 222-9. ArticlePubMed
- 160. Sattar N, Bendomir A, Berry C, Shepherd J, Greer IA, Packard CJ. Lipoprotein subfraction concentrations in preeclampsia: pathogenic parallels to atherosclerosis. Obstet Gynecol 1997; 89: 403-8. ArticlePubMed
- 161. Lorentzen B, Endresen MJ, Hovig T, Haug E, Henriksen T. Sera from preeclamptic women increase the content of triglycerides and reduce the release of prostacyclin in cultured endothelial cells. Thromb Res 1991; 63: 363-72. ArticlePubMed
- 162. Hubel CA, McLaughlin MK, Evans RW, Hauth BA, Sims CJ, Roberts JM. Fasting serum triglycerides, free fatty acids, and malondialdehyde are increased in preeclampsia, are positively correlated, and decrease within 48 hours post partum. Am J Obstet Gynecol 1996; 174: 975-82. ArticlePubMed
- 163. Belo L, Santos-Silva A, Quintanilha A, Rebelo I. Similarities between pre-eclampsia and atherosclerosis: a protective effect of physical exercise? Curr Med Chem 2008; 15: 2223-9. ArticlePubMed
- 164. Lau SY, Guild SJ, Barrett CJ, et al. Tumor necrosis factor-alpha, interleukin-6, and interleukin-10 levels are altered in preeclampsia: a systematic review and meta-analysis. Am J Reprod Immunol 2013; 70: 412-27. ArticlePubMed
- 165. Chaiworapongsa T, Chaemsaithong P, Yeo L, Romero R. Pre-eclampsia part 1: current understanding of its pathophysiology. Nat Rev Nephrol 2014; 10: 466-80. ArticlePubMedPMCPDF
- 166. Boyd EM. The lipemia of pregnancy. J Clin Invest 1934; 13: 347-63. ArticlePubMedPMC
- 167. Potter JM, Nestel PJ. The hyperlipidemia of pregnancy in normal and complicated pregnancies. Am J Obstet Gynecol 1979; 133: 165-70. ArticlePubMed
- 168. Baker AM, Klein RL, Moss KL, Haeri S, Boggess K. Maternal serum dyslipidemia occurs early in pregnancy in women with mild but not severe preeclampsia. Am J Obstet Gynecol 2009; 201: 293: e1-4. Article
- 169. Labarrere CA, Faulk WP. Antigenic identification of cells in spiral artery trophoblastic invasion: validation of histologic studies by triple-antibody immunocytochemistry. Am J Obstet Gynecol 1994; 171: 165-71. ArticlePubMed
- 170. Roberts JM, Redman CW. Pre-eclampsia: more than pregnancy-induced hypertension. Lancet 1993; 341: 1447-51. ArticlePubMed
REFERENCES
Figure & Data
References
Citations
Citations to this article as recorded by 

- Placental vascular remodeling in preeclampsia: A three-dimensional analysis of microvascular alterations across disease severity
Mingqun Li, Xiaoqiang Han, Yao Peng, Yang He, Qiangqiang You, Jiaqi Zhang
Placenta.2026; 174: 96. CrossRef - Placental aberrant inflammation and spatial-specific lipid metabolism contribute to hypertensive disorder of pregnancy susceptibility in preeclampsia offspring
Pei-ran Hu, Jing-hui Xu, Yan Shi, Ying Zhu, Gao-chen Zhang, Jie-ru Yang, Yue Xu, Ming-hao Li, Xian-hua Lin, Yu Zhang, He-feng Huang
Biochimica et Biophysica Acta (BBA) - Molecular Basis of Disease.2026; 1872(4): 168168. CrossRef - Circulating vascular endothelial growth factor receptor‐3, a pro‐lymphangiogenic and pro‐angiogenic mediator, is decreased in pre‐eclampsia
Ana C. Palei, Julyane N. S. Kaihara, Ricardo C. Cavalli, Valeria C. Sandrim
International Journal of Gynecology & Obstetrics.2025; 168(1): 210. CrossRef - ECHS1 as a Lipid Metabolism Biomarker for Pediatric Focal Segmental Glomerulosclerosis
Chao He, Wei Peng, Sheng Li, Can Xu, Xiuping Chen, Yuanhan Qin, Nasar Alwahaibi
PLOS ONE.2025; 20(3): e0319049. CrossRef - PlacEntal Acute atherosis RefLecting Subclinical systemic atherosclerosis in women up to 20 years after pre-eclampsia (PEARLS): research protocol for a cohort study
Gwyneth Jansen, Robert-Jan Alers, Emma BNJ Janssen, Laura M Jorissen, Eri Morina - Shijaku, Carmen Severens-Rijvers, Arnoud van ’t Hof, J van Drongelen, Ralph R Scholten, Salwan Al-Nasiry, Droima Stevens, Wessel Ganzevoort, Sanne Gordijn, Jérôme Cornette,
BMJ Open.2025; 15(5): e100542. CrossRef - Understanding Preeclampsia: Cardiovascular Pathophysiology, Histopathological Insights and Molecular Biomarkers
Kaltrina Kutllovci Hasani, Nurxhan Ajeti, Nandu Goswami
Medical Sciences.2025; 13(3): 154. CrossRef - Human Placenta and Evolving Insights into Pathological Changes of Preeclampsia: A Comprehensive Review of the Last Decade
Diana Maria Chiorean, Esra Cobankent Aytekin, Melinda-Ildiko Mitranovici, Sabin Gligore Turdean, Mirpooya Salehi Moharer, Ovidiu Simion Cotoi, Havva Serap Toru
Fetal and Pediatric Pathology.2024; 43(1): 33. CrossRef - Effects of hypertensive disorders of pregnancy on the complications in very low birth weight neonates
Baoquan Zhang, Xiujuan Chen, Changyi Yang, Huiying Shi, Wenlong Xiu
Hypertension in Pregnancy.2024;[Epub] CrossRef - Prevention of Pregnancy Complications Using a Multimodal Lifestyle, Screening, and Medical Model
Jim Parker, Pierre Hofstee, Shaun Brennecke
Journal of Clinical Medicine.2024; 13(15): 4344. CrossRef - Placental growth factor mediates pathological uterine angiogenesis by activating the NFAT5-SGK1 signaling axis in the endometrium: implications for preeclampsia development
Janet P. Raja Xavier, Toshiyuki Okumura, Melina Apweiler, Nirzari A. Chacko, Yogesh Singh, Sara Y Brucker, Satoru Takeda, Florian Lang, Madhuri S Salker
Biological Research.2024;[Epub] CrossRef - Genome-wide DNA methylation and gene expression in human placentas derived from assisted reproductive technology
Pauliina Auvinen, Jussi Vehviläinen, Karita Rämö, Ida Laukkanen, Heidi Marjonen-Lindblad, Essi Wallén, Viveca Söderström-Anttila, Hanna Kahila, Christel Hydén-Granskog, Timo Tuuri, Aila Tiitinen, Nina Kaminen-Ahola
Communications Medicine.2024;[Epub] CrossRef - Missing links in preeclampsia cell model systems of endothelial dysfunction
Sarah Viana-Mattioli, Miriam Helena Fonseca-Alaniz, Iguaracy Pinheiro-de-Sousa, José Eduardo Krieger, Valéria Cristina Sandrim
Trends in Molecular Medicine.2023; 29(7): 541. CrossRef - Roles of maternal HDL during pregnancy
Laura A. Woollett, Janet M. Catov, Helen N. Jones
Biochimica et Biophysica Acta (BBA) - Molecular and Cell Biology of Lipids.2022; 1867(3): 159106. CrossRef - The role of the placenta in spontaneous preterm labor and delivery with intact membranes
Sunil Jaiman, Roberto Romero, Gaurav Bhatti, Eunjung Jung, Francesca Gotsch, Manaphat Suksai, Dahiana M. Gallo, Tinnakorn Chaiworapongsa, Nicholas Kadar
Journal of Perinatal Medicine.2022; 50(5): 553. CrossRef - Gestational Antibodies to C. pneumoniae, H. pylori and CMV in Women with Preeclampsia and in Matched Controls
Abdul Wajid, David Todem, Mark R. Schleiss, David F. Colombo, Nigel S. Paneth
Maternal and Child Health Journal.2022; 26(10): 2040. CrossRef - Toward a new taxonomy of obstetrical disease: improved performance of maternal blood biomarkers for the great obstetrical syndromes when classified according to placental pathology
Roberto Romero, Eunjung Jung, Tinnakorn Chaiworapongsa, Offer Erez, Dereje W. Gudicha, Yeon Mee Kim, Jung-Sun Kim, Bomi Kim, Juan Pedro Kusanovic, Francesca Gotsch, Andreea B. Taran, Bo Hyun Yoon, Sonia S. Hassan, Chaur-Dong Hsu, Piya Chaemsaithong, Nardh
American Journal of Obstetrics and Gynecology.2022; 227(4): 615.e1. CrossRef - Preeclampsia and Fetal Growth Restriction as Risk Factors of Future Maternal Cardiovascular Disease—A Review
Sylwia Sławek-Szmyt, Katarzyna Kawka-Paciorkowska, Aleksandra Ciepłucha, Maciej Lesiak, Mariola Ropacka-Lesiak
Journal of Clinical Medicine.2022; 11(20): 6048. CrossRef - The Role of NF-κB in Uterine Spiral Arteries Remodeling, Insight into the Cornerstone of Preeclampsia
Maciej W. Socha, Bartosz Malinowski, Oskar Puk, Mateusz Wartęga, Martyna Stankiewicz, Anita Kazdepka-Ziemińska, Michał Wiciński
International Journal of Molecular Sciences.2021; 22(2): 704. CrossRef - Pathogenesis of uteroplacental acute atherosis: An update on current research
Shu Li, Yan‐Wei Hu
American Journal of Reproductive Immunology.2021;[Epub] CrossRef - Disorders of placental villous maturation are present in one-third of cases with spontaneous preterm labor
Sunil Jaiman, Roberto Romero, Percy Pacora, Offer Erez, Eunjung Jung, Adi L. Tarca, Gaurav Bhatti, Lami Yeo, Yeon Mee Kim, Chong Jai Kim, Jung-Sun Kim, Faisal Qureshi, Suzanne M. Jacques, Nardhy Gomez-Lopez, Chaur-Dong Hsu
Journal of Perinatal Medicine.2021; 49(4): 412. CrossRef - The COVID-19 Pandemic: an Appraisal of its Impact on Human Immunodeficiency Virus Infection and Pre-Eclampsia
Rowen Govender, Jagidesa Moodley, Thajasvarie Naicker
Current Hypertension Reports.2021;[Epub] CrossRef - Acute Atherosis Lesions at the Fetal-Maternal Border: Current Knowledge and Implications for Maternal Cardiovascular Health
Daniel Pitz Jacobsen, Heidi Elisabeth Fjeldstad, Guro Mørk Johnsen, Ingrid Knutsdotter Fosheim, Kjartan Moe, Patji Alnæs-Katjavivi, Ralf Dechend, Meryam Sugulle, Anne Cathrine Staff
Frontiers in Immunology.2021;[Epub] CrossRef - Aetiology, prophylaxis and management of preeclampsia
Karolina Gronkowska
Acta Universitatis Lodziensis. Folia Biologica et Oecologica.2021; 17: 111. CrossRef - HMOX1 is partly responsible for phenotypic and functional abnormalities in mesenchymal stem cells/stromal cells from placenta of preeclampsia (PE) patients
Yasser S. Basmaeil, Dana Algudiri, Reem Alenzi, Abdullah Al Subayyil, Ayodele Alaiya, Tanvir Khatlani
Stem Cell Research & Therapy.2020;[Epub] CrossRef - Analyzing Preeclampsia as the Tip of the Iceberg Represented by Women with Long-Term Cardiovascular Disease, Atherosclerosis, and Inflammation
Angélica Lemos Debs Diniz, Maria Marta Bini Martins Paes, Aline Debs Diniz
Current Atherosclerosis Reports.2020;[Epub] CrossRef - Lipids in preeclampsia: pathogenic parallels to atherosclerosis
V. I. Shcherbakov, Ya. V. Polonskaya, E. V. Kashtanova, A. V. Shirinskaya
"Arterial’naya Gipertenziya" ("Arterial Hypertension").2020; 26(2): 163. CrossRef - Transthyretin increases migration and invasion of rat placental trophoblast cells
Xiao‐Peng Ma, Chong‐Dong Liu, Guang‐Ming Cao, Zhen‐Yu Zhang
FEBS Open Bio.2020; 10(8): 1568. CrossRef - Early Onset Preeclampsia Is Associated With Glycocalyx Degradation and Reduced Microvascular Perfusion
Tracey L. Weissgerber, Oscar Garcia‐Valencia, Natasa M. Milic, Elizabeth Codsi, Hajrunisa Cubro, Meryl C. Nath, Wendy M. White, Karl A. Nath, Vesna D. Garovic
Journal of the American Heart Association.2019;[Epub] CrossRef - The immunophenotype of decidual macrophages in acute atherosis
Navleen Gill, Yaozhu Leng, Roberto Romero, Yi Xu, Bogdan Panaitescu, Derek Miller, Afrah Arif, Salma Mumuni, Faisal Qureshi, Chaur‐Dong Hsu, Sonia S. Hassan, Anne Cathrine Staff, Nardhy Gomez‐Lopez
American Journal of Reproductive Immunology.2019;[Epub] CrossRef - The potential effects of pomegranate peel extract and bee venom in improving the diabetes induced damaging of spiral artery
HIH El-Sayyad, HA El-Ghawet, AMA El-Sayed
Studies on Stem Cells Research and Therapy.2019; 5(1): 007. CrossRef - Race and risk of maternal vascular malperfusion lesions in the placenta
Vanessa Assibey-Mensah, W. Tony Parks, Alison D. Gernand, Janet M. Catov
Placenta.2018; 69: 102. CrossRef - Preclinical atherosclerosis at the time of pre‐eclamptic pregnancy and up to 10 years postpartum: systematic review and meta‐analysis
N. M. Milic, J. Milin‐Lazovic, T. L. Weissgerber, G. Trajkovic, W. M. White, V. D. Garovic
Ultrasound in Obstetrics & Gynecology.2017; 49(1): 110. CrossRef - Is an episode of suspected preterm labor that subsequently leads to a term delivery benign?
Roberto Romero, Offer Erez, Eli Maymon, Percy Pacora
American Journal of Obstetrics and Gynecology.2017; 216(2): 89. CrossRef - Establishment of the Human Uteroplacental Circulation: A Historical Perspective
Kenna Degner, Ronald R. Magness, Dinesh M. Shah
Reproductive Sciences.2017; 24(5): 753. CrossRef - Preeclampsia and coronary plaque erosion: Manifestations of endothelial dysfunction resulting in cardiovascular events in women
Saskia C.A. de Jager, John A.L. Meeuwsen, Freeke M. van Pijpen, Gerbrand A. Zoet, Arjan D. Barendrecht, Arie Franx, Gerard Pasterkamp, Bas B. van Rijn, Marie-José Goumans, Hester M. den Ruijter
European Journal of Pharmacology.2017; 816: 129. CrossRef - Placental histopathology lesions and pregnancy outcome in pregnancies complicated with symptomatic vs. non-symptomatic placenta previa
Eran Weiner, Hadas Miremberg, Ehud Grinstein, Letizia Schreiber, Shimon Ginath, Jacob Bar, Michal Kovo
Early Human Development.2016; 101: 85. CrossRef - Porphyromonas gingivalis within Placental Villous Mesenchyme and Umbilical Cord Stroma Is Associated with Adverse Pregnancy Outcome
Sizzle F. Vanterpool, Jasper V. Been, Michiel L. Houben, Peter G. J. Nikkels, Ronald R. De Krijger, Luc J. I. Zimmermann, Boris W. Kramer, Ann Progulske-Fox, Leticia Reyes, Motohiro Komaki
PLOS ONE.2016; 11(1): e0146157. CrossRef - Pregnant women with heart disease: Placental characteristics and their association with fetal adverse events
Fabio V. Lima, Paraskevi Koutrolou-Sotiropoulou, Puja B. Parikh, Cecilia Avila, Javed Butler, Kathleen Stergiopoulos
Acute Cardiac Care.2016; 18(3): 56. CrossRef
 PubReader
PubReader ePub Link
ePub Link-
 Cite this Article
Cite this Article
- Cite this Article
-
- Close
- Download Citation
- Close
- Figure
- Related articles
Acute Atherosis of the Uterine Spiral Arteries: Clinicopathologic Implications


Fig. 1. Spiral artery changes during pregnancy. (A) Normal physiologic transformation of spiral arteries in a normal pregnancy. The lumen of the spiral artery (asterisks) is dilated. Trophoblastic cells are infiltrating the wall of the spiral artery. (B) Failure of physiologic transformation of spiral arteries in a patient with preeclampsia. The lumen of the arteries (asterisk) is not dilated. The medial layers of the spiral arteries are intact. Although many interstitial trophoblasts surround the spiral artery, trophoblasts have not invaded the vessel wall.
Fig. 2. Acute atherosis in decidual spiral arteries. (A) Many lipid-laden macrophages (arrows) are seen in the spiral arteries. (B) Acute atherosis on oil-red O staining. Fat droplets (arrows) in the non-transformed spiral artery are stained red.
Fig. 1.
Fig. 2.
Acute Atherosis of the Uterine Spiral Arteries: Clinicopathologic Implications

 E-submission
E-submission