Articles
- Page Path
- HOME > J Pathol Transl Med > Volume 50(2); 2016 > Article
-
Original Article
Prognostic Implication of Semi-quantitative Immunohistochemical Assessment of CD20 Expression in Diffuse Large B-Cell Lymphoma - Chang Hwan Choia,*, Young Hoon Park1,*, Joo Han Lim1, Suk Jin Choi, Lucia Kim, In Suh Park, Jee Young Han, Joon Mee Kim, Young Chae Chu
-
Journal of Pathology and Translational Medicine 2016;50(2):96-103.
DOI: https://doi.org/10.4132/jptm.2016.01.12
Published online: February 15, 2016
Department of Pathology, Inha University Hospital, Inha University School of Medicine, Incheon, Korea
1Department of Hematology-Oncology, Inha University Hospital, Inha University School of Medicine, Incheon, Korea
- Corresponding Author: Suk Jin Choi, MD Department of Pathology, Inha University Hospital, Inha University School of Medicine, 27 Inhang-ro, Jung-gu, Incheon 22332, Korea Tel: +82-32-890-3972 Fax: +82-32-890-3464 E-mail: sukjinchoi007@gmail.com
- *Chang Hwan Choi and Young Hoon Park contributed equally to this work.
aPresent address: Scientific Investigation Laboratory, Criminal Investigation Command, Ministry of National Defense, Seoul, Korea.
© 2016 The Korean Society of Pathologists/The Korean Society for Cytopathology
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/3.0/) which permits unrestricted noncommercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
Abstract
-
Background:
- Immunohistochemical demonstration of CD20 in diffuse large B-cell lymphoma (DLBCL) is prerequisite not only for the diagnosis but also for assigning patients to rituximab-containing chemotherapy. However, little is known about the impact of abundance of CD20 expression assessed by immunohistochemistry on the clinical outcome of DLBCL. We performed a semi-quantitative immunohistochemical analysis of CD20 expression in DLBCL to examine the prognostic implication of the level of CD20 expression.
-
Methods:
- Pre-treatment diagnostic tissue samples from 48 DLBCL patients who were treated with rituximab, cyclophosphamide, doxorubicin, vincristine, and prednisone (R-CHOP) regimen were represented in a tissue microarray and immunostained for CD20. The relative abundance of CD20 expression was semi-quantitatively scored using a web-based ImmunoMembrane plug-in. Receiver operating characteristic curve analysis was used to determine a prognostically relevant cut-off score in order to dichotomize the patients into CD20-high versus CD20-low groups.
-
Results:
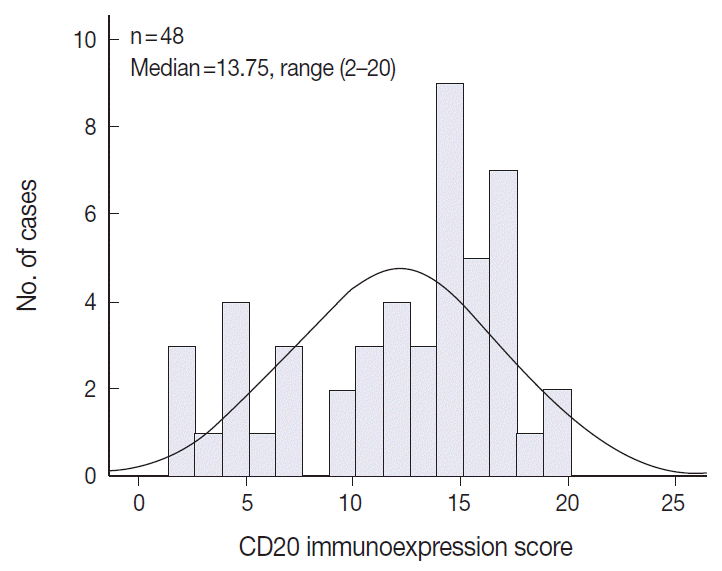
- The levels of CD20 expression were heterogeneous among the patients, with a wide and linear distribution of scores. Patients in CD20-low group showed significantly poor clinical outcome.
-
Conclusions:
- The levels of CD20 expression in DLBCL are heterogeneous among the patients with DLBCL. A subgroup of the patients with CD20 expression levels below the cut-off score showed poor clinical outcome.
- TMA cohort
- We searched the surgical pathology archives for cases diagnosed with de novo DLBCL in Inha University Hospital between January 2006 and December 2013. Among them, we included the patients who received six to eight cycles of standard R-CHOP chemotherapy. Having excluded the primary central nervous system lymphomas and cases diagnosed based on small biopsy specimens such as endoscopic biopsy or needle biopsy, a total of 48 patients with complete outcome data were eligible for the preparation of TMA cohort, including 27 male and 21 female with a median age of 58.5 years (range, 20 to 81 years). Two 1-mm duplicate cores from the diagnostic FFPET samples were represented on a TMA by using a self-made TMA and a home-made recipient block as described previously [4].
- Immunohistochemical staining of TMA sections
- Using an automated immunohistochemical stainer (Bench-MarkXT, Ventana, Tucson, AZ, USA), sections of the TMA cut in 4-μm thickness were routinely processed for immunohistochemical stains for CD20 (1:500, H1, heat-induced antigen retrieval, BD Pharmingen, San Jose, CA, USA), BCL2 (1:100, 124, heat-induced antigen retrieval, Dako, Carpinteria, CA, USA), BCL6 (1:30, PG-B6p, heat-induced antigen retrieval, Dako), CD10 (pre-diluted, SP67, heat-induced antigen retrieval, Ventana), and MUM1 (1:100, MUM1p, heat-induced antigen retrieval, Dako). Cases were designated as positive when ≥30% of the tumor cells were immuoreactive for all antibodies except for CD20 [5]. Cases were classified into germinal center B-cell versus non-germinal center B-cell type according to Hans classification [6].
- Quantification of CD20 levels
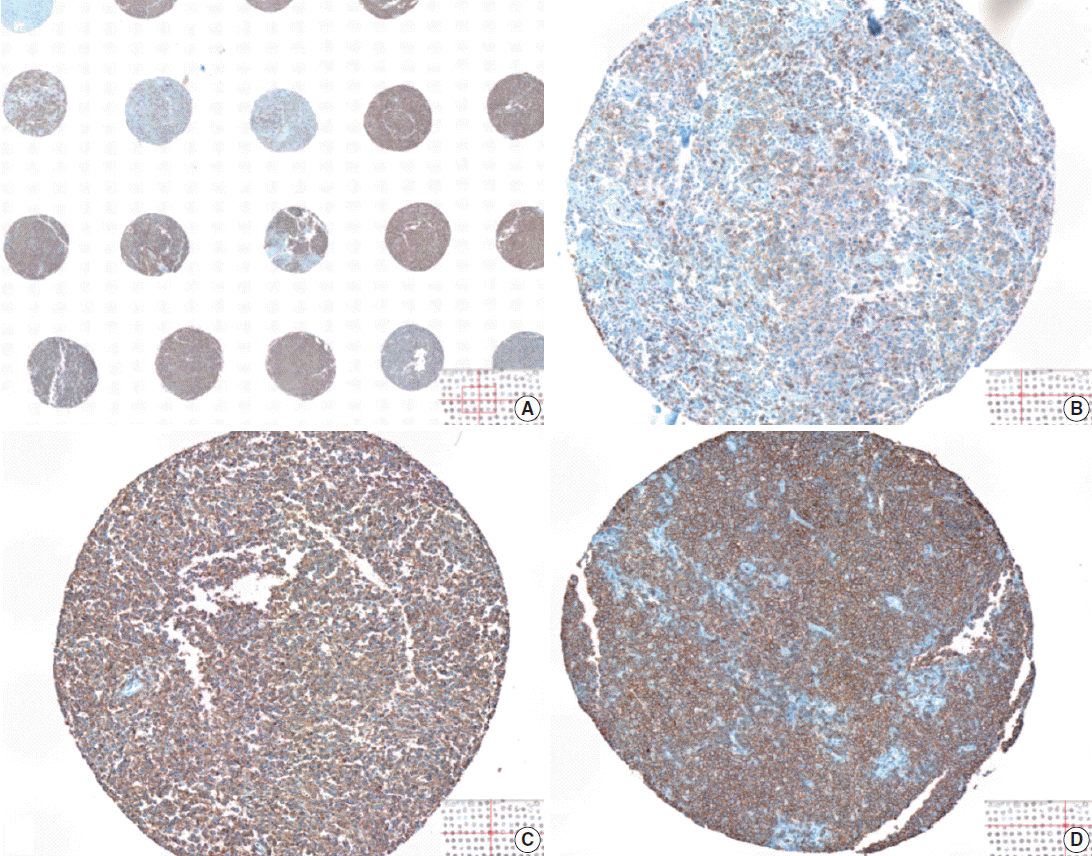
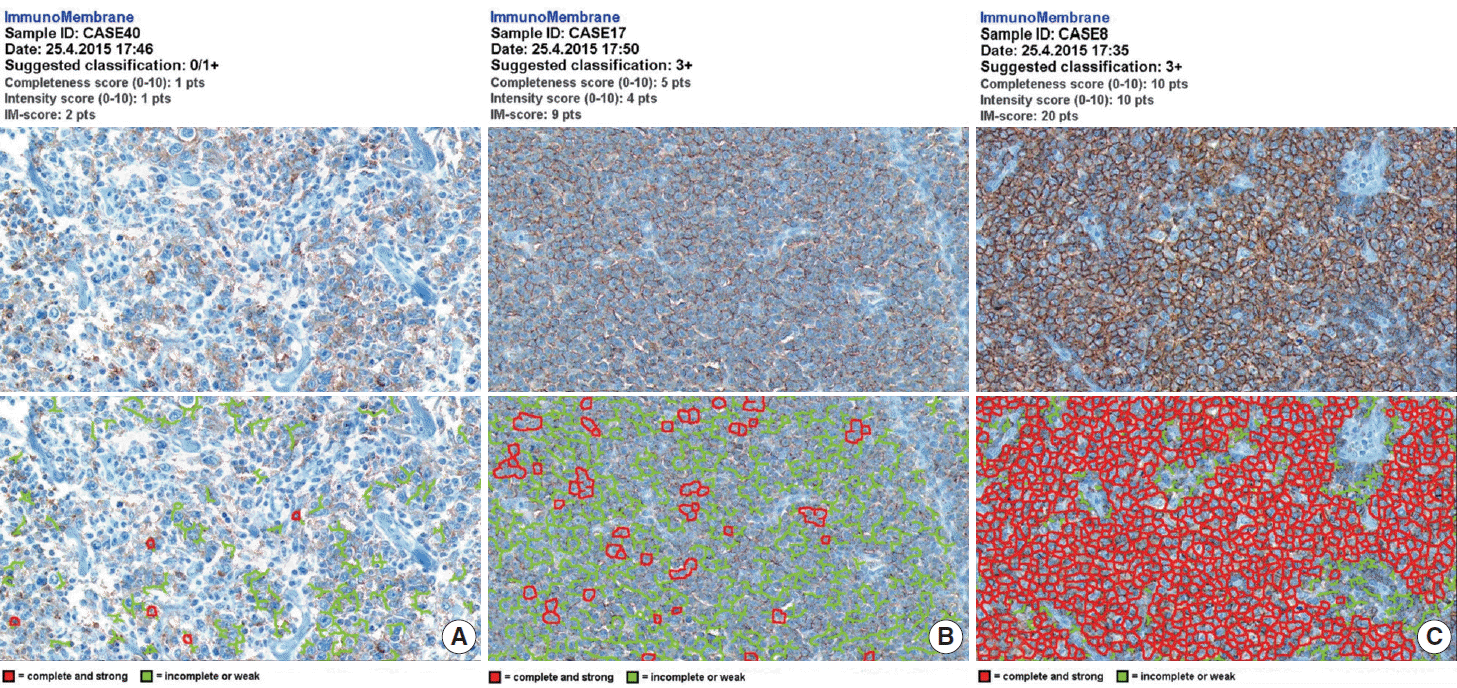
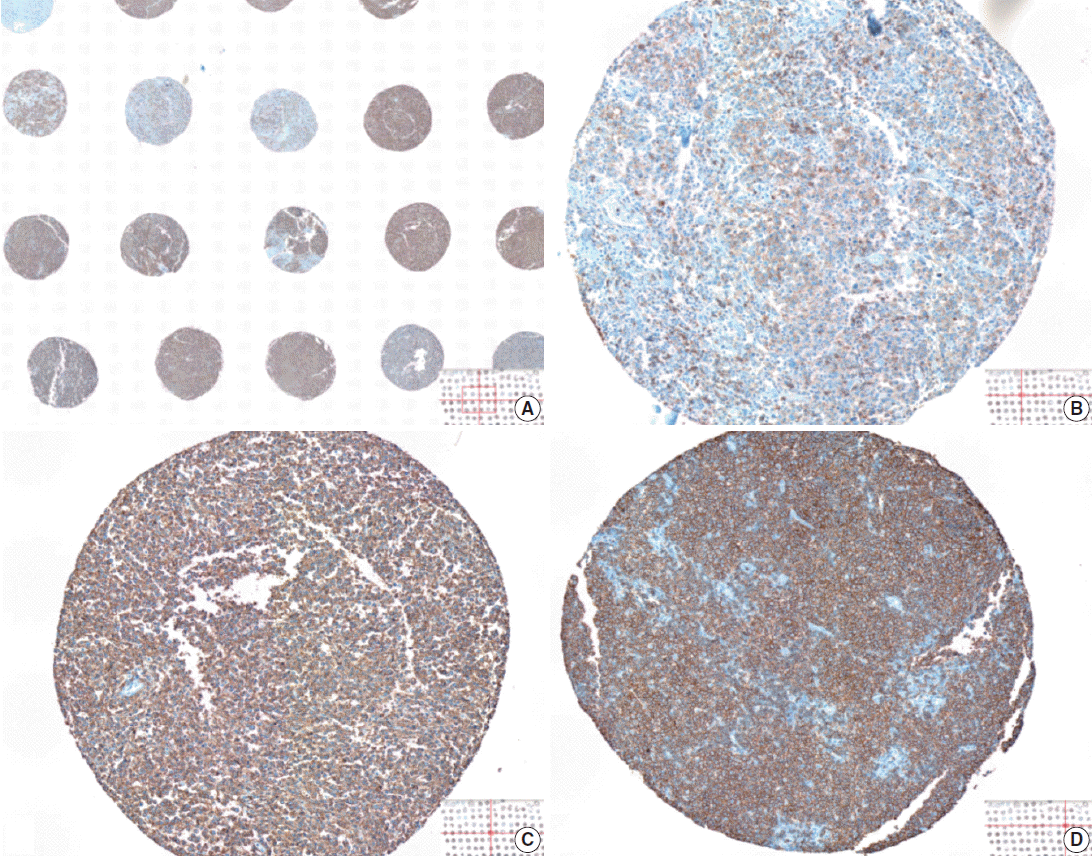
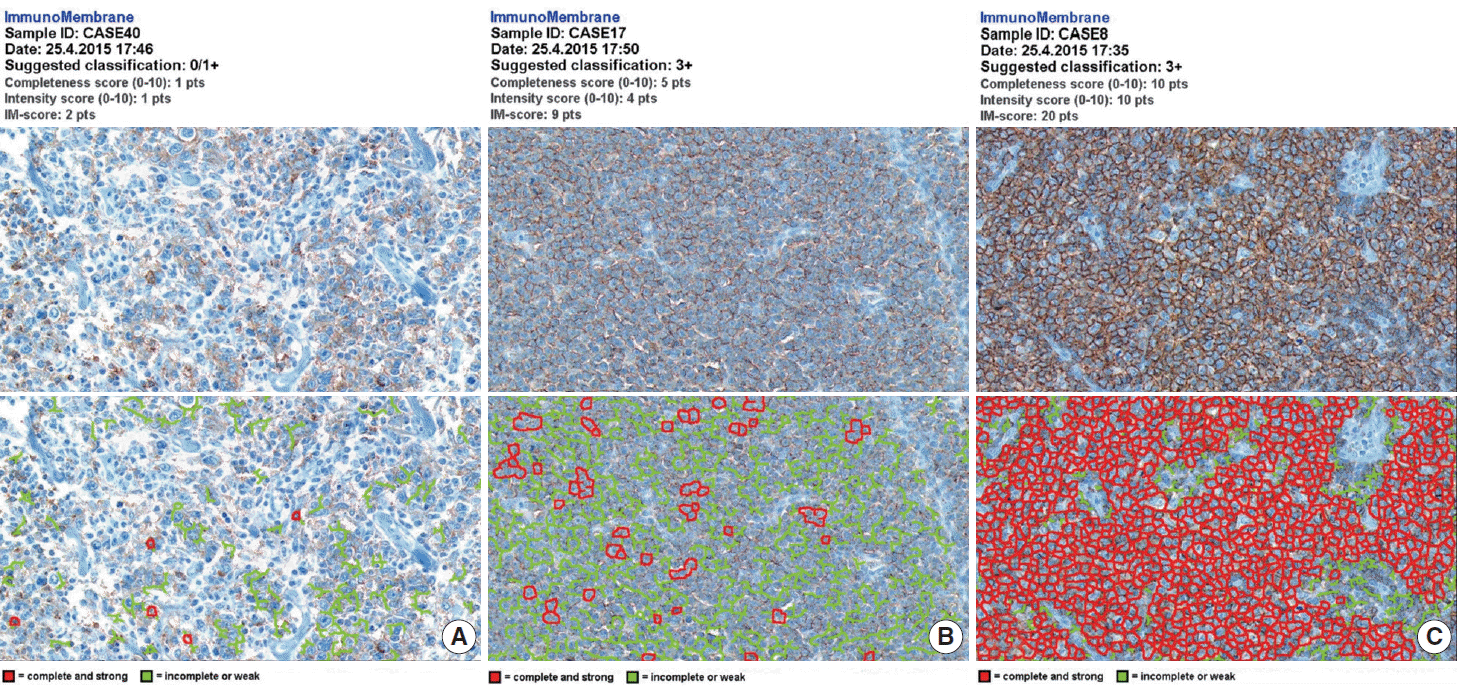
- The whole image of CD20-stained TMA section was digitalized using a digital slide scanner (VM600, Motic, Ximen, China) using a ×20 objective (Fig. 1). Then, representative JPEG images were acquired from each tissue sample (Fig. 1). CD20 immunoreactivity within each JPEG image was semi-quantitatively assessed using ImageJ software (http:/rsbweb.nih.gov/ij/) coupled with ImmunoMembrane plug-in (http://imtmicroscope.uta.fi/immunomembrane) [7]. Completeness (0–10 points) and intensity (0–10 points) of membrane staining was added for a score (0–20 points) (Fig. 2). Then, we used the average of the duplicates in analysis as the arbitrary level of CD20 for each case.
- Statistical analysis
- The primary outcomes of interest in this study were overall survival (OS) and event-free survival (EFS). OS was defined as the time interval between the date of diagnosis and the date of death by any cause. EFS was estimated from the date of diagnosis to the date of disease progression, relapse, last contact, or death. Complete response (CR) was determined according to the conventional response criteria. Receiver operating characteristic (ROC) curve analysis was used to select a prognostically relevant cut-off score of CD20 expression that can dichotomize the patients in terms of clinical otucome [8,9]. Kaplan-Meier analysis was used to estimate cumulative survivals and the differences between survival curves were analyzed using log-rank test. Chi-square test or Fisher exact test were used to compare the differences in frequency of categorical variables between two groups. Mann-Whitney U test and Kruskal-Wallis test were used to assess mean differences between the groups. Multivariate regression analysis using the Cox proportional hazards model was performed to determine the hazard ratios (HRs) of the clinicopathologic factors and CD20 levels. All p-values presented were two-sided, and p <.05 was considered statistically significant. All statistical analyses were performed using SPSS ver. 19.0 (IBM Co., Armonk, NY, USA).
MATERIALS AND METHODS
- Heterogeneity in CD20 levels among patients with DLBCL
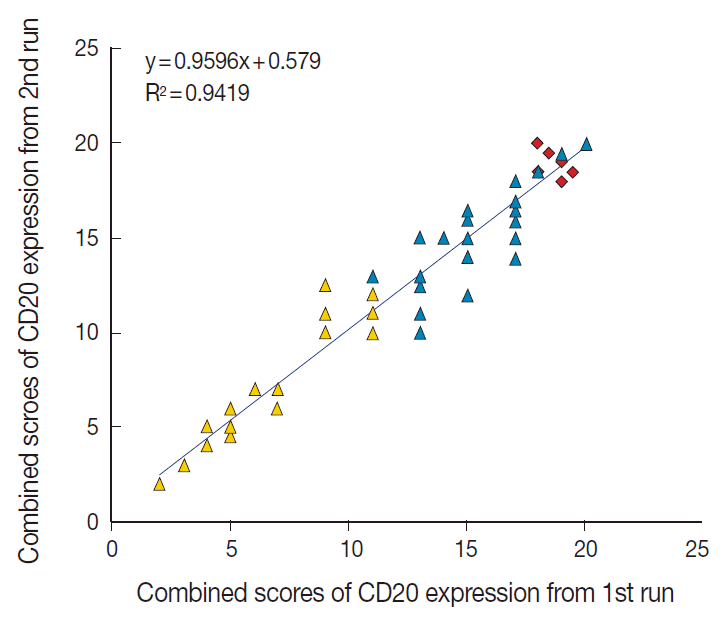
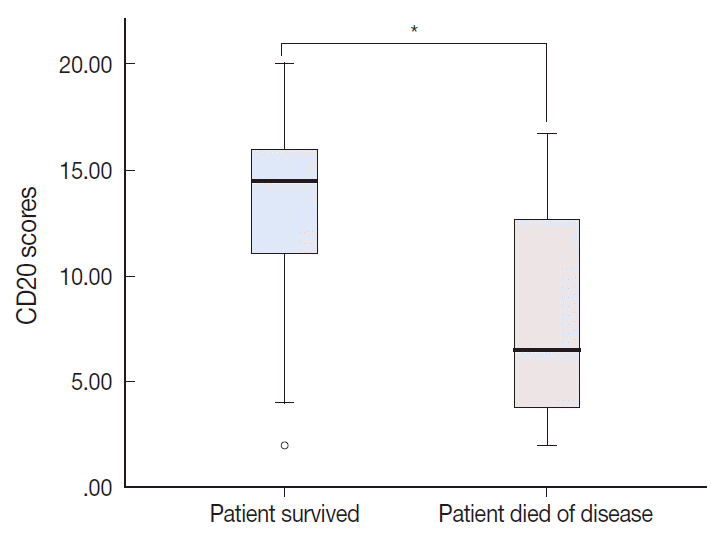
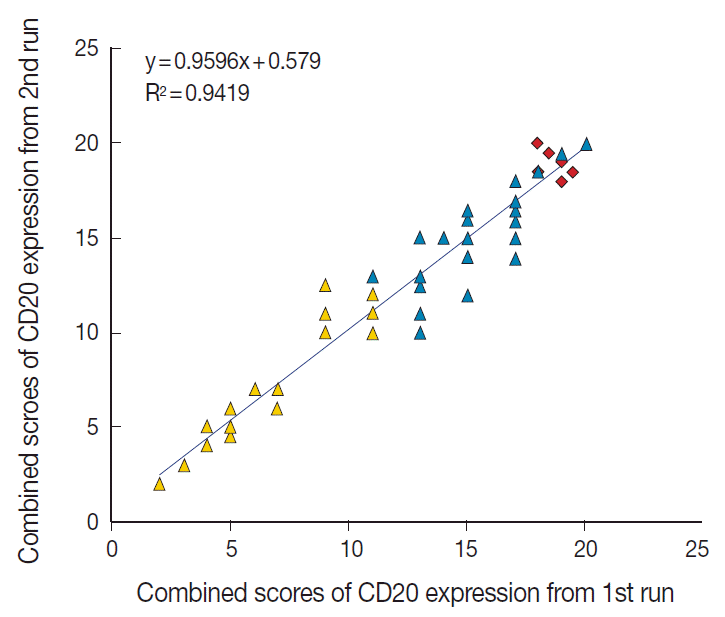
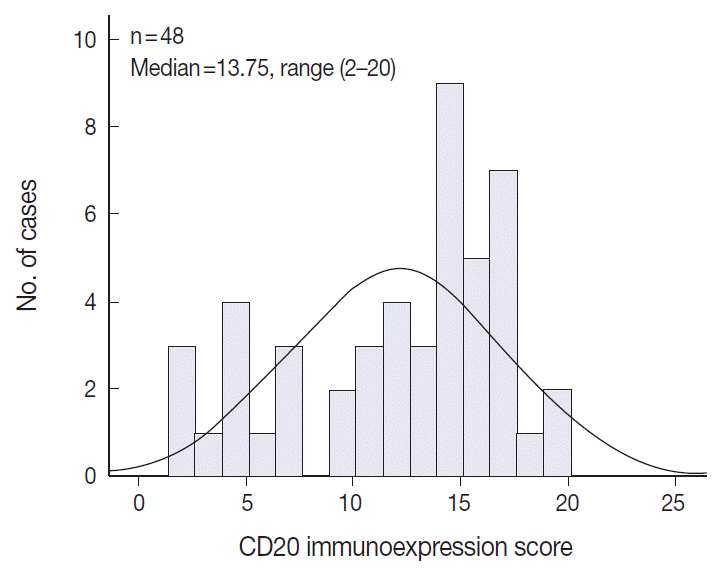
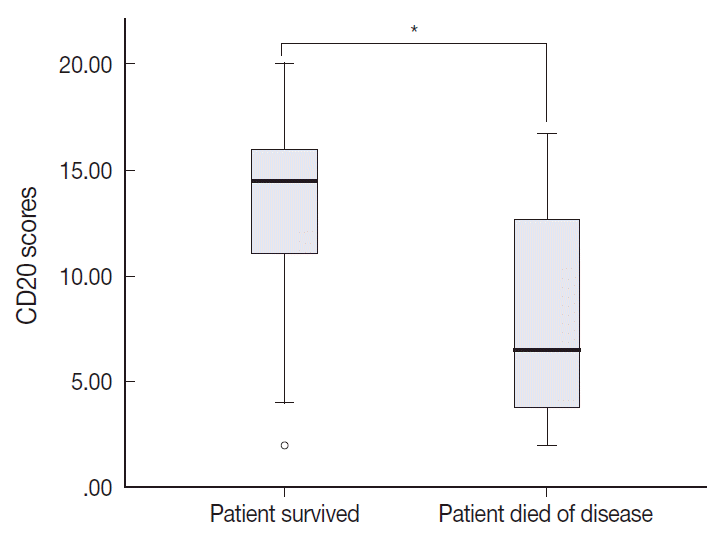
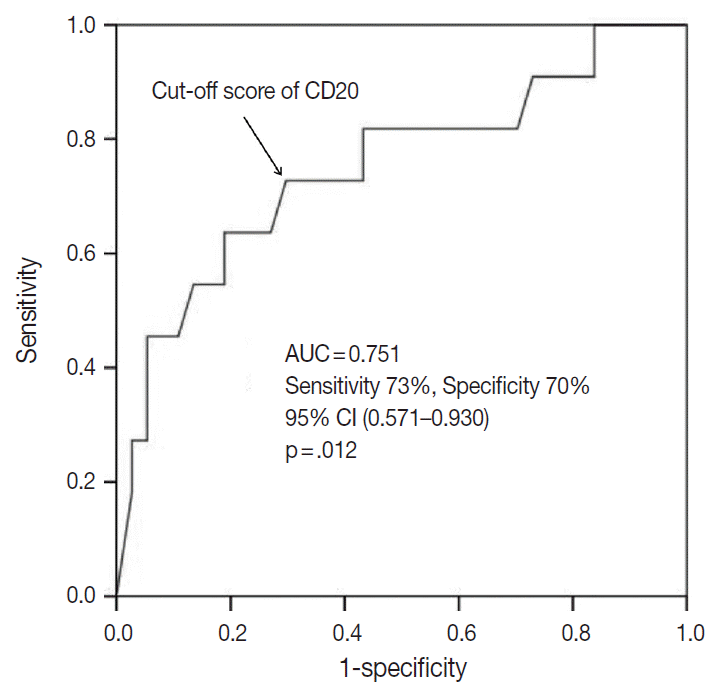
- The TMA section immunostained for CD20 exhibited heterogeneous CD20 levels among patients, with a high degree of reproducibility in scores between duplicate tissue cores to duplicate runs (Fig. 3). As indicated by a wide and linear range of the semi-quantitative scores with a negatively skewed distribution of data on the frequency histogram (Fig. 4), the level of CD20 expression was significantly lower in patients who died of disease compared to those survived (p=.012) (Fig. 5).
- Association between CD20 level and clinical outcome
- The median follow-up period was 58.5 months (range, 11 to 125 months). Overall, there were 27 male (56.3%) and 21 female (43.7%) patients, and the median age was 58.5 years (range, 20 to 81 years). After six to eight cycles of R-CHOP chemotherapy, CR was achieved in 41 patients (85.4%) and 11 (11/41, 26.8%) of them experienced relapse.
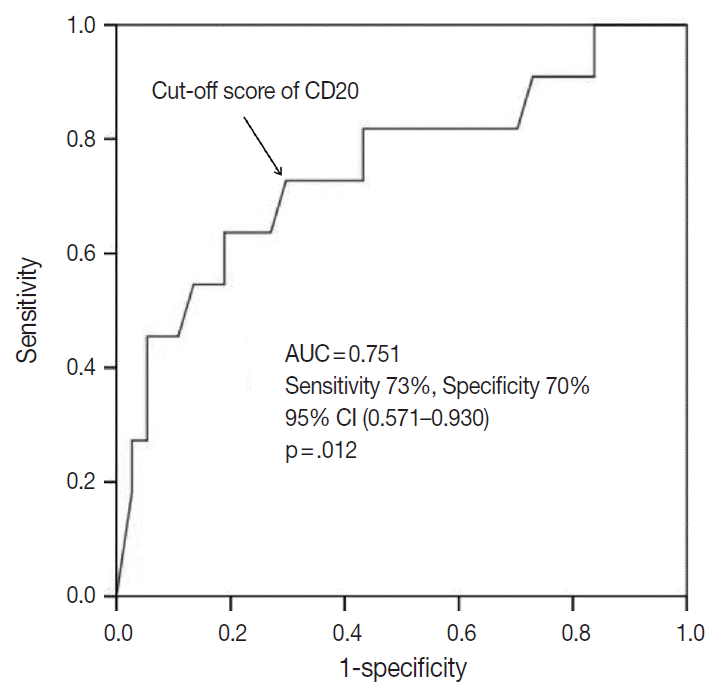
- The outcome-based ROC curve analysis produced a predictive cut-off score of 11.75 in CD20 levels (with an area under the ROC curve of 0.751; 95% confidence interval [CI], 0.571 to 0.930; sensitivity, 72.7%; specificity, 70.3%; p=.012) that could predict poor prognosis in terms of OS (Fig. 6). Based on the predictive cut-off score, the overall patients in our cohort were dichotomized into the CD20-low (n=20, 41.7%) and CD20-high (n=28, 58.3%).
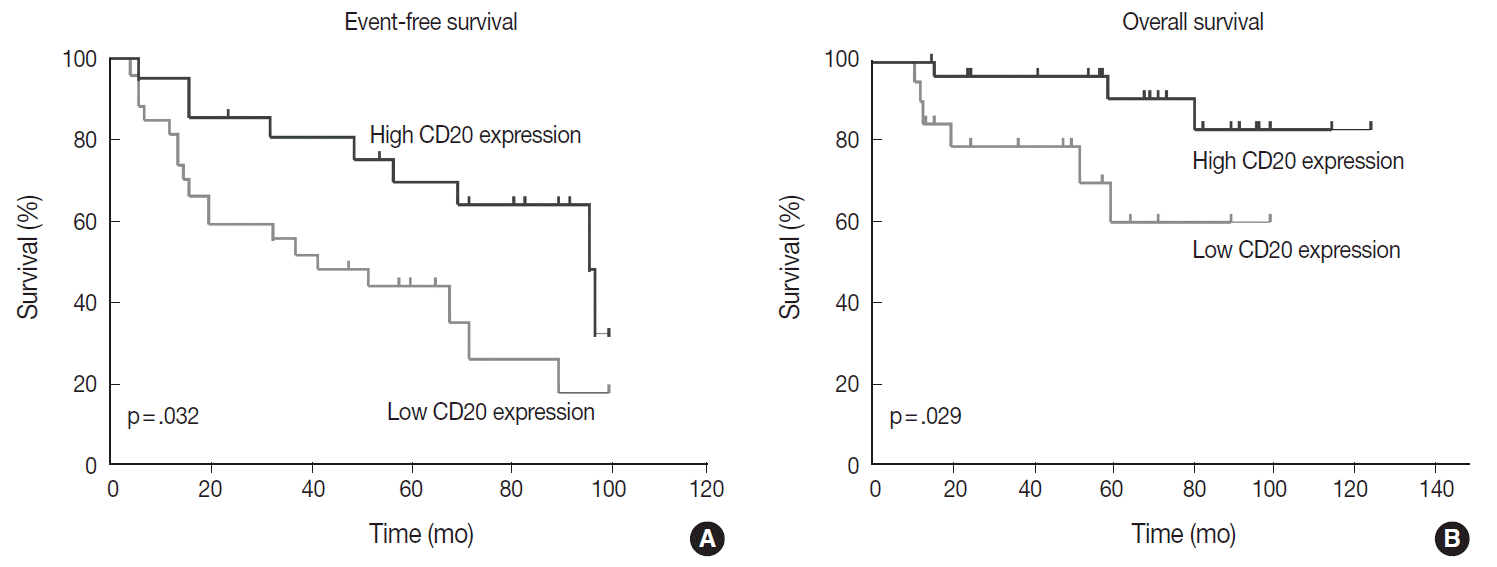
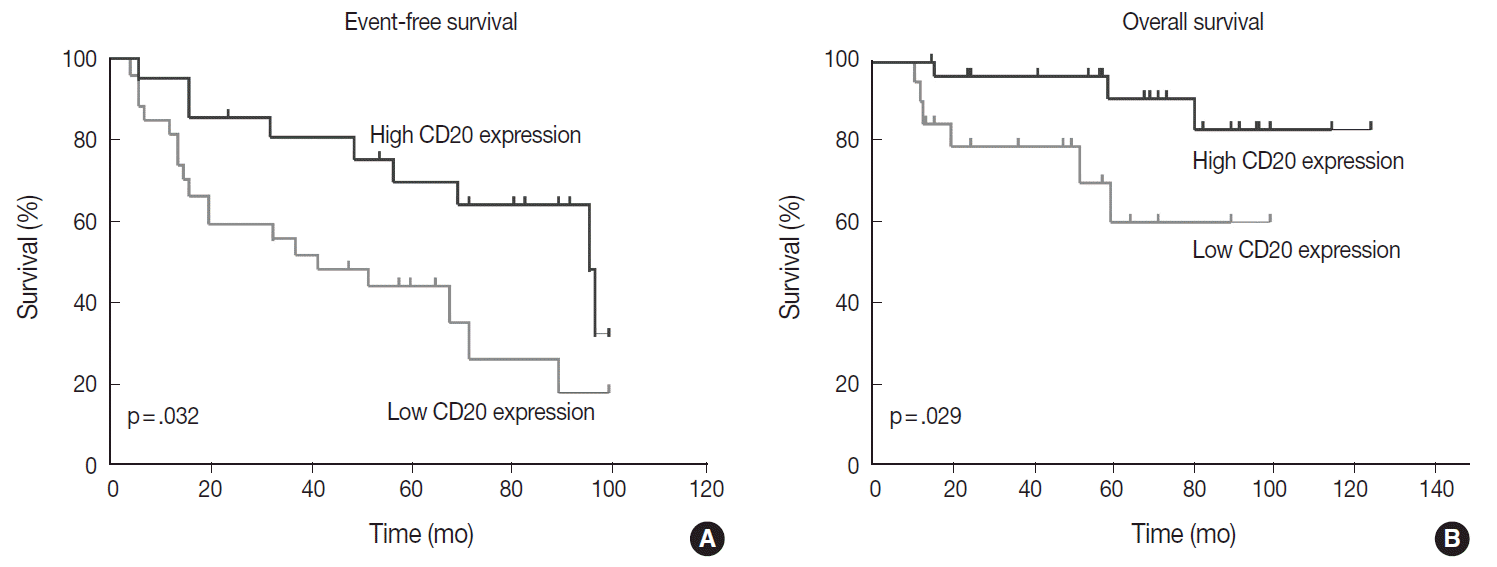
- Table 1 shows the distribution of the clinical background characteristics of the patients in the two groups divided according to the CD20 expression level. There were no significant differences in clinicopathologic parameters between the two groups. CR was achieved in 15 patients (75%) in the CD20-low group and 26 patients (92.9%) in the CD20-high group, with no significant difference in CR rate between these two groups (p= .197). Eventually, disease relapse was experienced by four out of 15 patients (26.7%) in the CD20-low group and by seven out of 26 patients (26.9%) in CD20-high group, with no significant difference in relapse rate between these two groups. Clinical events including disease progression were more frequently observed in CD20-low group (14/20, 70%) compared to CD20-high group (13/28, 46.4%), with a significant difference in EFS (median, 37 months [range, 4 to 100 month] vs 96 months [range, 6 to 100 months]) for the CD20-high group (p=.032) (Fig. 7A). The 5-year EFS were 39.4% in CD20-low group and 66.5% in CD20-high group, respectively. Overall, nine patients died, including six deaths in the CD20-low group and three deaths in the CD20-high group. Median OS was 50 months (range, 11 to 100 months) for the CD20-low group and 71 months (range, 15 to 125 months) for the CD20-high group, with a significantly shorter 5-year EFS (39.4% vs 66.5%) and OS (59.9% vs 90.6%) in CD20-low group (Fig. 7B).
- Prognostic factor analyses
- In the univariate analysis of prognostic factors, following parameters were significantly associated with worse prognosis in terms of both EFS and OS: CD20 level lower than the cut-off score (p=.039 and p=.035) and two or more sites of extranodal involvement (p=.034 and p=.044). In addition, poor performance status with Eastern Cooperative Oncology Group (ECOG) score ≥2 (p=.042) and presence of B symptoms (p=.023) were significantly associated with a poor prognosis in terms of EFS, but not in terms of OS. The stage at diagnosis and International Prognostic Index score were not significantly associated with EFS (p=.423 and p=.099) and OS (p=.576 and p=.530). When all the parameters significant in univariate analysis were included in multivariate analysis, lower CD20 level remained an independent prognostic factor for EFS (HR, 2.187; 95% CI, 1.015 to 4.745; p=.048) and for OS (HR, 4.291; 95% CI, 1.012 to 17.724; p= .043) (Table 2). Two or more sites of extranodal involvement was an independent prognostic factor (HR, 2.768; 95% CI, 1.028 to 7.571; p=.047) for EFS but it was not of prognostic relevance with regard to OS (p=.098). ECOG performance status (ECOG PS) ≥2 (p=.125) and presence of B symptoms (p=.095) were not found to be in correlation with OS in multivariate analysis.
RESULTS
- The prognostic significance of the traditional clinicopathologic parameters of DLBCL seems to be overshadowed by the introduction of R-CHOP as a standard regimen for DLBCL [8-10]. Bcl-2 expression assessed by immunohistochemical staining seems to be no longer associated with a poor outcome in DLBCL patients treated with R-CHOP [11]. In addition, the prognostic value of immunophenotypic subtype determined by immunohistochemistry for CD10, Bcl-6, and MUM1 is questioned in rituximab era [12,13]. Furthermore, the predictive significance of International Prognostic Index that has been the most important prognostic factor for DLBCL in pre-rituximab era also seems to be overridden by the addition of rituximab to CHOP regimen [14].
- When assessed semi-quantitatively using flow cytometry (FCM) analysis, the levels of CD20 expression in the fresh tissue sample were quite heterogeneous both among and within different types of non-Hodgkin B-cell lymphomas [15,16]. In addition, the level of CD20 expression at the onset of disease seems to be an independent predictor for poor outcomes in patients treated with R-CHOP [15,17,18]. FCM analysis of fresh tissue sample, however, is not a routine practice in many institutions with limited resources. Furthermore, fresh tissue samples for FCM analysis are not always available. Accordingly, immunohistochemical assessment of CD20 expression in diagnostic tissue samples in forms of paraffin blocks should be a more feasible method both in terms of low cost and convenience in sample recruitment.
- There has been no previous literature specific to the distribution of immunohistochemical levels of CD20 expression in the diagnostic FFPET samples of DLBCL and their association with the prognosis of the patients. In this study, we used a free web-based image analysis tool that enabled us to acquire objective semi-quantitative scores of CD20 expression levels based on intensity and completeness of CD20-immunostaining of the FFPET sections. As indicated by the previous investigations using FCM analysis of fresh tissue samples, the levels of CD20 expression assessed by semi-quantitative immunohistochemistry were quite heterogeneous among the patients, with scores spread over a wide and linear range. By correlating the expression level of CD20 with the clinical outcomes, we were able to identify a subgroup of DLBCL patients with CD20 expression levels below the cut-off score, who showed poor EFS and OS independent of other known clinical and pathologic parameters. Further studies are warranted to validate the prognostic role of the relative abundance of CD20 expression in a larger cohort.
- Interestingly, Shimizu et al. [19] recently reported that histone deacetylase (HDAC) inhibitors augment cytotoxic effect of rituximab by increasing the level of CD20 in the cell lines of DLBCL. Accordingly, the patients with lower levels of CD20 expression who could be less amenable to rituximab therapy could probably benefit from pretreatment of HDAC inhibitors such as valproic acid prior to the standard R-CHOP treatment [20]. Alternatively, they could more likely benefit from the treatment with novel anti-CD20 monoclonal antibodies which show more anti-cancer activity in CD20 low-expression B-cell lymphomas. For example, ofatumumab (a fully human anti-CD20 monoclonal antibody) and obinutuzumab (a humanized type II antibody targeted against CD20) have been shown to be active and safe in clinical trials that had included patients with chronic lymphocytic leukemia which is known to have relatively lower levels of CD20 expression [21,22].
- In conclusion, a level of CD20 expression is heterogeneous in DLBCL and lower level of CD20 expression is a poor prognostic marker for survival in patients with DLBCL who are treated with R-CHOP chemotherapy. Semi-quantitative immunohistochemical estimation of CD20 expression can be used to identify a subgroup of DLBCL patients with poor outcome who might possibly benefit from the pretreatment with agents that can increase the level of CD20 expression in the cell membranes of the tumor cells.
DISCUSSION
Acknowledgments
- 1. Stein H, Warnke RA, Chan WC, et al. Diffuse large B-cell lymphoma, not otherwise specified. In: Swerdlow SH, Campo E, Harris NL, eds. WHO classification of tumours of haematopoietic and lymphoid tissues. 4th ed. Lyon: IARC Press, 2008; 233-7.
- 2. Coiffier B. State-of-the-art therapeutics: diffuse large B-cell lymphoma. J Clin Oncol 2005; 23: 6387-93. ArticlePubMed
- 3. Boross P, Leusen JH. Mechanisms of action of CD20 antibodies. Am J Cancer Res 2012; 2: 676-90. PubMedPMC
- 4. Kim KH, Choi SJ, Choi YI, et al. In-house manual construction of high-density and high-quality tissue microarrays by using homemade recipient agarose-paraffin blocks. Korean J Pathol 2013; 47: 238-44. ArticlePubMedPMC
- 5. Berglund M, Thunberg U, Amini RM, et al. Evaluation of immunophenotype in diffuse large B-cell lymphoma and its impact on prognosis. Mod Pathol 2005; 18: 1113-20. ArticlePubMedPDF
- 6. Hans CP, Weisenburger DD, Greiner TC, et al. Confirmation of the molecular classification of diffuse large B-cell lymphoma by immunohistochemistry using a tissue microarray. Blood 2004; 103: 275-82. ArticlePubMed
- 7. Tuominen VJ, Tolonen TT, Isola J. ImmunoMembrane: a publicly available web application for digital image analysis of HER2 immunohistochemistry. Histopathology 2012; 60: 758-67. ArticlePubMed
- 8. Salles G, de Jong D, Xie W, et al. Prognostic significance of immunohistochemical biomarkers in diffuse large B-cell lymphoma: a study from the Lunenburg Lymphoma Biomarker Consortium. Blood 2011; 117: 7070-8. PubMed
- 9. Culpin RE, Sieniawski M, Angus B, et al. Prognostic significance of immunohistochemistry-based markers and algorithms in immunochemotherapy-treated diffuse large B cell lymphoma patients. Histopathology 2013; 63: 788-801. ArticlePubMed
- 10. Coiffier B. Rituximab therapy in malignant lymphoma. Oncogene 2007; 26: 3603-13. ArticlePubMedPDF
- 11. Mounier N, Briere J, Gisselbrecht C, et al. Rituximab plus CHOP (R-CHOP) overcomes bcl-2-associated resistance to chemotherapy in elderly patients with diffuse large B-cell lymphoma (DLBCL). Blood 2003; 101: 4279-84. ArticlePubMed
- 12. Nyman H, Adde M, Karjalainen-Lindsberg ML, et al. Prognostic impact of immunohistochemically defined germinal center phenotype in diffuse large B-cell lymphoma patients treated with immunochemotherapy. Blood 2007; 109: 4930-5. ArticlePubMedPDF
- 13. Winter JN, Weller EA, Horning SJ, et al. Prognostic significance of Bcl-6 protein expression in DLBCL treated with CHOP or R-CHOP: a prospective correlative study. Blood 2006; 107: 4207-13. ArticlePubMedPMC
- 14. Sehn LH, Berry B, Chhanabhai M, et al. The revised International Prognostic Index (R-IPI) is a better predictor of outcome than the standard IPI for patients with diffuse large B-cell lymphoma treated with R-CHOP. Blood 2007; 109: 1857-61. ArticlePubMedPDF
- 15. Johnson NA, Boyle M, Bashashati A, et al. Diffuse large B-cell lymphoma: reduced CD20 expression is associated with an inferior survival. Blood 2009; 113: 3773-80. ArticlePubMedPMCPDF
- 16. Olejniczak SH, Stewart CC, Donohue K, Czuczman MS. A quantitative exploration of surface antigen expression in common B-cell malignancies using flow cytometry. Immunol Invest 2006; 35: 93-114. ArticlePubMed
- 17. Horvat M, Kloboves Prevodnik V, Lavrencak J, Jezersek Novakovic B. Predictive significance of the cut-off value of CD20 expression in patients with B-cell lymphoma. Oncol Rep 2010; 24: 1101-7. PubMed
- 18. Suzuki Y, Yoshida T, Wang G, et al. Association of CD20 levels with clinicopathological parameters and its prognostic significance for patients with DLBCL. Ann Hematol 2012; 91: 997-1005. ArticlePubMedPDF
- 19. Shimizu R, Kikuchi J, Wada T, Ozawa K, Kano Y, Furukawa Y. HDAC inhibitors augment cytotoxic activity of rituximab by upregulating CD20 expression on lymphoma cells. Leukemia 2010; 24: 1760-8. ArticlePubMedPDF
- 20. Damm JK, Gordon S, Ehinger M, et al. Pharmacologically relevant doses of valproate upregulate CD20 expression in three diffuse large B-cell lymphoma patients in vivo. Exp Hematol Oncol 2015; 4: 4.ArticlePubMedPMCPDF
- 21. Coiffier B, Lepretre S, Pedersen LM, et al. Safety and efficacy of ofatumumab, a fully human monoclonal anti-CD20 antibody, in patients with relapsed or refractory B-cell chronic lymphocytic leukemia: a phase 1-2 study. Blood 2008; 111: 1094-100. ArticlePubMedPDF
- 22. Goede V, Fischer K, Busch R, et al. Obinutuzumab plus chlorambucil in patients with CLL and coexisting conditions. N Engl J Med 2014; 370: 1101-10. ArticlePubMed
REFERENCES
Figure & Data
References
Citations

- The Expression Levels of CD20 as a Prognostic Value in Feline B-Cell Nasal Lymphoma: A Pilot Study
Kravee Chaipoca, Theerapol Sirinarumitr, Supreeya Srisampan, Charuwan Wongsali, Attawit Kovitvadhi, Tassanee Jaroensong
Animals.2024; 14(7): 1043. CrossRef - Prognostic molecular biomarkers in diffuse large B-cell lymphoma in the rituximab era and their therapeutic implications
Sotirios G. Papageorgiou, Thomas P. Thomopoulos, Ioannis Katagas, Anthi Bouchla, Vassiliki Pappa
Therapeutic Advances in Hematology.2021;[Epub] CrossRef - Novel tumour–infiltrating lymphocyte-related risk stratification based by flow cytometry for patients with de novo angioimmunoblastic T cell lymphoma
Qiqi Zhu, Xueqin Deng, Wenqing Yao, Zihang Chen, Yunxia Ye, Limin Gao, Wenyan Zhang, Weiping Liu, Sha Zhao
Annals of Hematology.2021; 100(3): 715. CrossRef - Induced CD20 Expression on B-Cell Malignant Cells Heightened the Cytotoxic Activity of Chimeric Antigen Receptor Engineered T Cells
Yingxi Xu, Saisai Li, Ying Wang, Jia Liu, Xinhe Mao, Haiyan Xing, Zheng Tian, Kejing Tang, Xiaolong Liao, Qing Rao, Dongsheng Xiong, Min Wang, Jianxiang Wang
Human Gene Therapy.2019; 30(4): 497. CrossRef - Characterization of head and neck squamous cell carcinoma arising in young patients: Particular focus on molecular alteration and tumor immunity
Hyang Joo Ryu, Eun Kyung Kim, Byoung Chul Cho, Sun Och Yoon
Head & Neck.2019; 41(1): 198. CrossRef - Immunoglobulin D (IgD) and IgD receptor expression in diffuse large B-cell lymphoma
Xing Dai, Yu-Jing Wu, Xiao-Yi Jia, Yan Chang, Hua-Xun Wu, Chun Wang, Wei Wei
Hematology.2019; 24(1): 544. CrossRef - The implications of TrkA and MET aberrations in de novo salivary duct carcinoma
Hyang Joo Ryu, Yoon Woo Koh, Sun Och Yoon
Human Pathology.2018; 81: 18. CrossRef - Prognostic stratification improvement by integrating ID1/ID3/IGJ gene expression signature and immunophenotypic profile in adult patients with B-ALL
Nataly Cruz-Rodriguez, Alba L. Combita, Leonardo J. Enciso, Lauren F. Raney, Paula L. Pinzon, Olga C. Lozano, Alba M. Campos, Niyireth Peñaloza, Julio Solano, Maria V. Herrera, Jovanny Zabaleta, Sandra Quijano
Journal of Experimental & Clinical Cancer Research.2017;[Epub] CrossRef - Implications of infiltrating immune cells within bone marrow of patients with diffuse large B-cell lymphoma
Juhyeon Jeong, Eun Ji Oh, Woo Ick Yang, Soo Jeong Kim, Sun Och Yoon
Human Pathology.2017; 64: 222. CrossRef - Architectural patterns of p16 immunohistochemical expression associated with cancer immunity and prognosis of head and neck squamous cell carcinoma
Hyang Joo Ryu, Eun Kyung Kim, Su Jin Heo, Byoung Chul Cho, Hye Ryun Kim, Sun Och Yoon
APMIS.2017; 125(11): 974. CrossRef - New developments in the pathology of malignant lymphoma. A review of the literature published from January–April 2016
J. Han van Krieken
Journal of Hematopathology.2016; 9(2): 73. CrossRef - Diffuse large B-cell lymphoma: R-CHOP failure—what to do?
Bertrand Coiffier, Clémentine Sarkozy
Hematology.2016; 2016(1): 366. CrossRef
 PubReader
PubReader ePub Link
ePub Link-
 Cite this Article
Cite this Article
- Cite this Article
-
- Close
- Download Citation
- Close
- Figure







Fig. 1.
Fig. 2.
Fig. 3.
Fig. 4.
Fig. 5.
Fig. 6.
Fig. 7.
| Characteristic | CD20 level above the cut-off score | CD20 level below the cut-off score | p-value |
|---|---|---|---|
| No. of patients | 28 | 20 | |
| Age, median (range, yr) | 54 (20–75) | 61 (34–81) | .446 |
| > 60 yr | 11 (45.8) | 11 (57.9) | .175 |
| Sex | .169 | ||
| Male | 13 (46.4) | 14 (70) | |
| Female | 15 (53.6) | 6 (30) | |
| ECOG PS | .212 | ||
| < 2 | 18 (64.3) | 10 (50) | |
| ≥ 2 | 10 (35.7) | 10 (50) | |
| Stage | .583 | ||
| I/II | 16 (57.1) | 13 (65) | |
| III/IV | 12 (42.9) | 7 (35) | |
| Serum LDH level | .299 | ||
| Normal | 4 (14.3) | 1 (5) | |
| Elevated | 24 (85.7) | 19 (95) | |
| No. of extranodal sites | 1 | ||
| < 2 | 24 (85.7) | 17 (85) | |
| ≥ 2 | 4 (14.3) | 3 (15) | |
| IPI risk stratification | .269 | ||
| Low/Low-intermediate | 21 (75) | 12 (60) | |
| High-intermediate/High | 7 (25) | 8 (40) | |
| Presence of B symptom | .176 | ||
| Yes | 5 (17.9) | 7 (35) | |
| No | 23 (82.1) | 13 (65) | |
| BM involvement | .369 | ||
| Negative | 25 (89.3) | 16 (80) | |
| Positive | 3 (10.7) | 4 (20) | |
| Subtype by IHC | .175 | ||
| GCB-like | 9 (32.1) | 3 (15) | |
| Non-GCB-like | 19 (67.9) | 17 (85) | |
| BCL2 IHC | .212 | ||
| Negative | 9 (32.1) | 10 (50) | |
| Positive | 19 (67.9) | 10 (50) | |
| Response | .197 | ||
| CR | 26 (92.9) | 15 (75) | |
| No CR | 2 (7.1) | 5 (25) |
| Variable | Event-free survival |
Overall survival |
||||||
|---|---|---|---|---|---|---|---|---|
| Univariate |
Multivariate |
Univariate |
Multivariate |
|||||
| HR (95% CI) | p-value | HR (95% CI) | p-value | HR (95% CI) | p-value | HR (95% CI) | p-value | |
| Age | ||||||||
| < 60 yr vs > 60 yr | 0.884 (0.413-1.890) | .750 | - | - | 0.599 (0.150-2.397) | .464 | - | - |
| Serum LDH level | ||||||||
| Elevated vs normal | 1.698 (0.268-3.699) | .562 | - | - | 1.487 (0.258-5.614) | .658 | - | - |
| ECOG PS | ||||||||
| ≥ 2 vs < 2 | 2.228 (1.030–4.819) | .042 | 1.421 (0.598-3.581) | .125 | 2.802 (0.730–10.752) | .071 | - | - |
| Stage | ||||||||
| III/IV vs I/II | 1.372 (0.633-2.976) | .423 | - | - | 1.453 (0.389-5.422) | .576 | - | - |
| Extranodal site | ||||||||
| ≥ 2 vs < 2 | 2.953 (1.087-8.024) | .034 | 2.768 (1.028-7.571) | .047 | 3.58 (1.485-20.974) | .044 | 2.35 (0.798-12.257) | .098 |
| IPI score | ||||||||
| ≥ 3 vs < 3 | 1.943 (0.883-4.276) | .099 | - | - | 1.560 (0.385-6.318) | .530 | - | - |
| B symptoms | ||||||||
| Positive vs negative | 3.090 (1.404-6.810) | .023 | 2.255 (0.825–4.287) | .095 | 2.268 (0.892-17.258) | .085 | - | - |
| BCL2 | ||||||||
| Positive vs negative | 0.612 (0.284-1.321) | .204 | - | - | 1.036 (0.257-4.169) | .960 | - | - |
| BCL6 | ||||||||
| Negative vs positive | 1.128 (0.515–2.472) | .763 | - | - | 0.969 (0.259-3.624) | .962 | - | - |
| Subtype by IHC | ||||||||
| Non-GCB vs GCB | 1.324 (0.569-3.080) | .509 | - | - | 1.298 (0.326-5.218) | .712 | - | - |
| CD20 level | ||||||||
| Below vs above the cut-off | 2.261 (1.047-4.875) | .039 | 2.187 (1.015–4.745) | .048 | 4.89 (1.043-17.857) | .035 | 4.291 (1.012-17.724) | .043 |
Values are presented as number (%). ECOG PS, Eastern Cooperative Oncology Group Performance status; LDH, lactate dehydrogenase; IPI, International Prognostic Index; BM, bone marrow; IHC, immunohistochemistry; GCB, germinal center B cell; CR, complete response.

 E-submission
E-submission