Articles
- Page Path
- HOME > J Pathol Transl Med > Volume 51(1); 2017 > Article
-
Case Study
A Rare Case of Recurrent Metastatic Solid Pseudopapillary Neoplasm of the Pancreas - Hye Seung Lee, Han Kyeom Kim, Bong Kyung Shin, Jin Hyuk Choi, Yoo Jin Choi, Ha Yeon Kim
-
Journal of Pathology and Translational Medicine 2017;51(1):87-91.
DOI: https://doi.org/10.4132/jptm.2016.06.16
Published online: August 6, 2016
Department of Pathology, Korea University Guro Hospital, Seoul, Korea
- Corresponding Author Bong Kyung Shin, MD, PhD Department of Pathology, Korea University Guro Hospital, Korea University College of Medicine, 148 Gurodong-ro, Guro-gu, Seoul 08308, Korea Tel: +82-2-2626-1472 Fax: +82-2-2626-1486 E-mail: tabrega@korea.ac.kr
• Received: March 30, 2016 • Revised: May 21, 2016 • Accepted: June 16, 2016
© 2017 The Korean Society of Pathologists/The Korean Society for Cytopathology
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/3.0/) which permits unrestricted noncommercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
Abstract
- A 61-year-old woman visited our hospital for bilateral multiple lung nodules and a mass in her thorax. She had a long history of multiple metastatic recurrences of solid pseudopapillary neoplasm (SPN); 24 years previously, the patient had undergone pylorus-preserving pancreaticoduodenectomy for a 9.9 × 8.6 cm mass in the pancreatic head. The tumor was diagnosed as an SPN. Nine years later, metastatic nodules were found on computed tomography in the patient’s liver and peritoneum and were excised. She subsequently underwent an additional eight metastatectomy procedures in diverse organs. For the presented event, the lung nodules were removed. The prevalence of malignant SPN in the general population is 5%–15%. However, multiple metastatic recurrence of malignant SPN is rare; the lung is a particularly rare site of metastasis, found in only three cases in the literature. Here, we describe this exceptional case and provide a literature review.
- Clinical summary
- A 61-year-old woman visited Korea University Guro Hospital (KUGH) for bilateral multiple lung nodules and a mass in the intrathoracic soft tissue. Twenty-four years prior, she had presented to KUGH with epigastric pain; computed tomography (CT) found a 9.9×8.6 cm heterogeneous mass in the pancreatic head. The tumor was removed and histologically diagnosed as SPN. After tumor removal, the patient underwent two choledochojejunostomy procedures but showed no sign of tumor recurrence. Nine years passed before her next visit for this same condition, when some newly developed nodules in her liver and peritoneum were biopsied and confirmed as metastatic SPN. During the next 15 years, the patient suffered eight additional metastatic recurrences in her liver, peritoneum, omentum, spleen, ovary, uterus, colon, mesocolon, and retroperitoneum before finally developing the present multiple lung metastases. A metastatectomy was performed to remove the lung nodules, and the patient is currently alive (Fig. 1). This study was approved by the Institutional Review Board (KUGH 15363-001) of KUGH.
- Pathologic findings
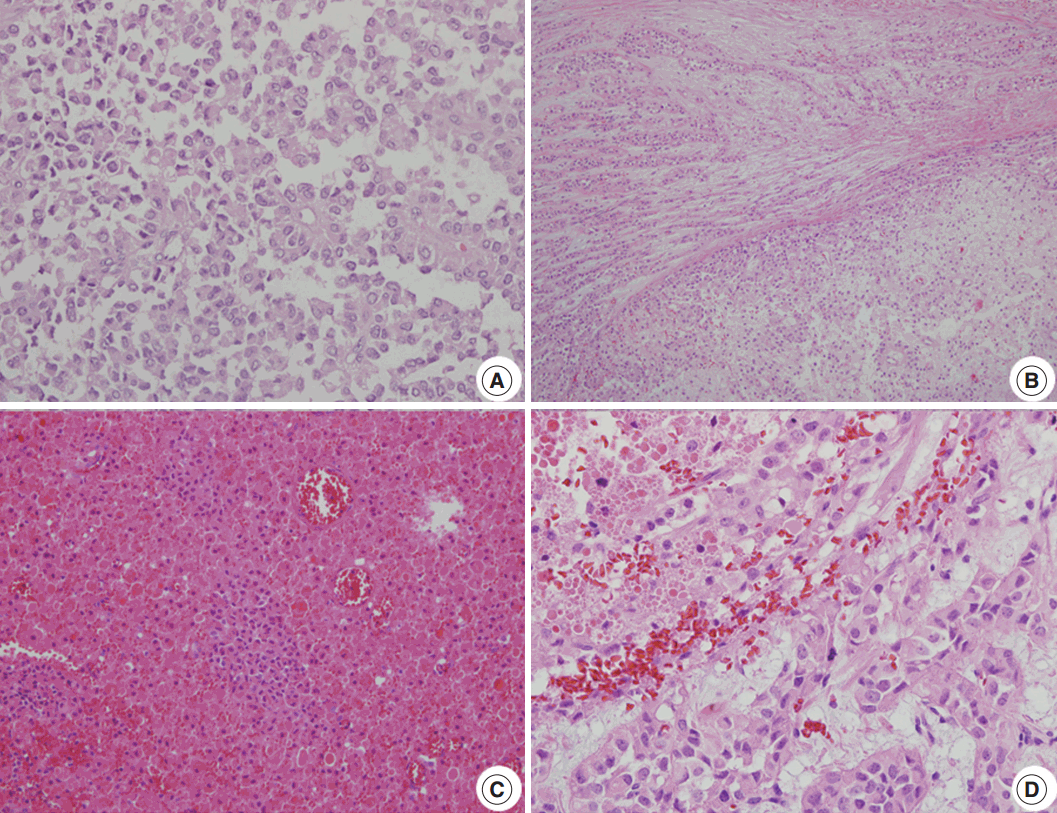
- The clinical and pathologic features of the primary tumor were reviewed in the patient’s electronic medical record. The glass slides and paraffin blocks that had been made from the recurrent tumors were also available for review. Overall, the tumors showed a pseudopapillary pattern with relatively monomorphic nuclei and abundant eosinophilic cytoplasm. However, we noted a certain degree of change over time, as summarized in Table 1. In accordance with the histological changes, we subdivided the lesions into early (first to third operations), intermediate (fourth to seventh), and late (eighth to 11th) lesions. In the early metastatic lesions, the tumor showed frequent cystic changes with bland nuclei and indistinct necrobiotic nests without capsular invasion. The Ki-67 labeling index was also very low (<1%). In the intermediate lesions, however, frequent necrobiotic nests (from the fourth to seventh) and capsular invasions (from the fifth) were noted. The Ki-67 labeling index, which was assessed using an image analysis program, increased to 5% (sixth). In the late lesions, malignant histological features, such as distinct tumor necrosis beyond necrobiotic nests (from the seventh), peritumoral infiltration of the soft tissue or organ (from the seventh), prominent solid architecture (from the eighth), nuclear atypia (from the ninth), and lymphatic emboli (11th), were seen (Fig. 2A–D). The average Ki-67 labeling index increased gradually (0.03%, 2.7%, and 5.6% in early, intermediate, and late metastatic lesions, respectively). Mitosis was usually absent with exceptions of 5/10 high-power field (HPF) (fifth) and 2/10 HPF (seventh) but did not show a significant change. All of the tumors showed identical immunohistochemical results and were diffusely positive for β-catenin, vimentin, neuron-specific enolase, CD10, cyclin D1, progesterone receptor, CD56, and synaptophysin. Interestingly, they were also positive for chromogranin A, which is very rare in SPN.
CASE REPORT
- Generally, the prognosis of SPN is good. Over 95% of patients with a solitary pancreatic lesion are cured by complete excision. Even with local spread, recurrences, or metastases, long periods of disease-free survival have been recorded [1]. The general prevalence of metastatic SPN is 5%–15% [1,2]. Debulking is recommended for these lesions in order to increase the chances of survival [6]. The lung is a very rare metastatic site and was found in only three cases in the literature [3-5]. The first was reported in 2005 by Takahashi et al. [5] and showed a lung lesion following three liver metastases. Hosokawa et al. [3] reported the second case in 2014, which demonstrated concurrent portal vein and superior mesenteric arterial invasion. The third case was described by Xu et al. in 2015 [4], with both liver and bone involvement. Two of the three cases showed a good survival outcome (Table 2). Our case also reveals a long overall survival period despite many recurrences.
- After reviewing the eight large studies in the literature, the most commonly quoted malignant SPN characteristics were nuclear atypia (5/8), angioinvasion (5/8), lymph node metastasis (4/8), mitotic count (3/8), advanced age (2/8), size (2/8), capsular invasion (2/8), perineural invasion (2/8), extra-pancreatic invasion (2/8), significant MIB-1 index (2/8), and prominent necrobiotic nests (1/8) [1,3,5-10]. Of the four cases with lung metastases (including ours), three showed liver metastasis, three exhibited capsular invasion, and two demonstrated necrobiotic nests (Table 2). Our case initially presented with a bland-looking tumor; however, most of the malignant features including nuclear atypia, lymph node metastasis (upon CT), lymphatic invasion, large tumor size, capsular invasion, and necrobiotic nests developed in later metastases.
- Though many studies have claimed that nuclear atypia is a malignant prognostic factor, there is still some debate on this topic. For example, Kim et al. [11] recently suggested that some subsets of SPN with nuclear pleomorphism exist with stronger p53 immunoreactivity but do not appear to be more aggressive than conventional SPN. The 2010 World Health Organization classification for tumors of the pancreas also claims that nuclear atypia of pancreas SPN cannot predict malignant tumor behavior [12]. In this study, nuclear atypia accompanied other malignant features. However, due to the patient’s relatively good prognosis, the atypia is still a predictor of metastasis but should not be considered a poor prognostic indicator of overall survival. More studies of SPN focusing on nuclear atypia will be needed to further explain this connection.
- In conclusion, this case depicts the aggressive progression of tumor cells by repeated peritoneal seeding and lymphatic invasion that still showed an indolent clinical course despite multiple recurrences, including the lung.
DISCUSSION
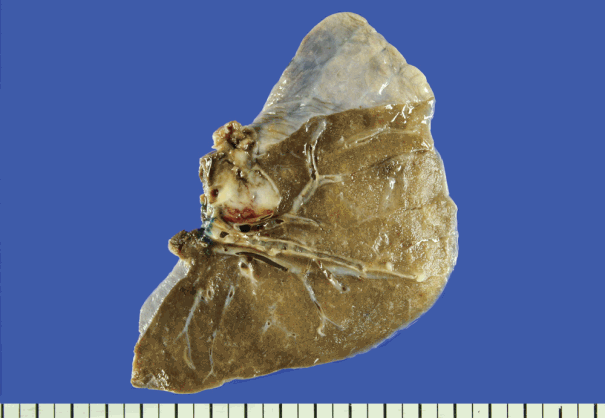
Fig. 1.Gross specimen of the patient’s lung metastasis. A solid, well-demarcated and cystic mass with hemorrhage is seen.
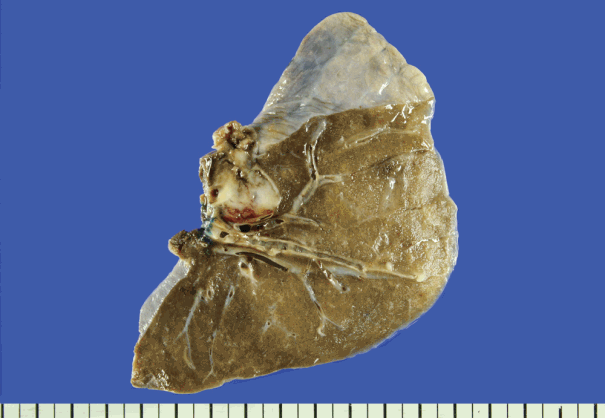

Fig. 2.Microscopic findings. (A) A loosely cohesive pseudopapillary pattern. (B) Capsular invasion with a cord or glandular pattern is visible in the intermediate and late metastatic lesions. (C) Necrobiotic nests are present in the intermediate lesions. (D) Distinct nuclear atypia is seen in the late lesions.
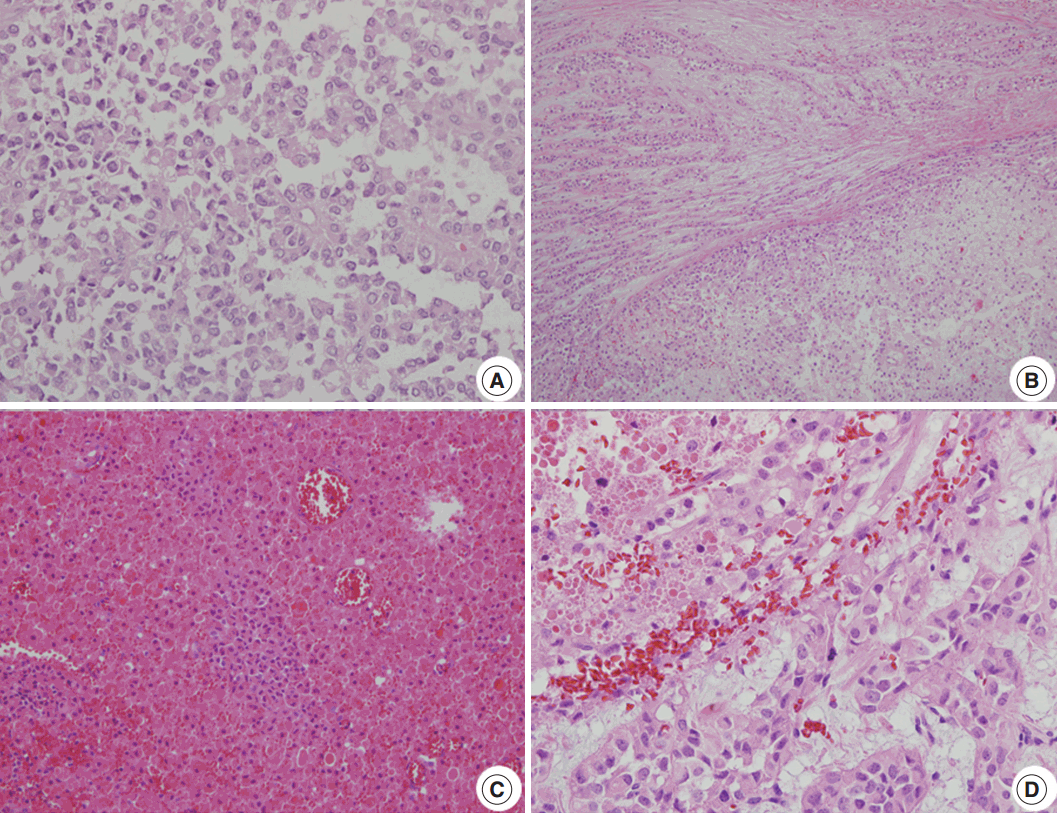

Table 1.Clinicopathological data of the patient
Table 2.Cases of solid pseudopapillary neoplasm with lung metastasis
| Reference | Sex | Age (yr) | Primary site | Size (cm) | Malignant histologic features | Metastasis | Operation | RI (mo) | Recurrence site | Follow-up (mo) | Survival | Mitoses (/10HPF) | Ki-67 (%) |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Takahashi et al. (2005) [5] | F | 41 | Tail | 12 | Capsule invasion, necrobiotic nests, venous invasion | Liver (multiple) | Distal pancreatectomy, metastatectomy | 14 | Liver, multiple (14 mo); liver, multiple (27 mo); liver, multiple (38 mo); lung, single (41 mo) | 60 | Alive | 5 | 10 |
| Hosokawa et al. (2014) [3] | F | 24 | Head | 6 | Capsule invasion, per pancreatic tissue | PV, SMA, lung | PPPD, PV, resection, pulmonary resection | None | None | 34 | Alive | - | 1.5 |
| Xu et al. (2015) [4] | F | 42 | Body | 13 | NE | Liver, lung, bone | NE | NE | NE | NE | NE | NE | NE |
| Our case (2016) | F | 37 | Head | 8.6 | Capsule invasion, necrobiotic nests, lymphatic invasion, lymph node invasion | None | PPPD | 107 | Liver, spleen, abdominal mesentery, mesocolon, retroperitoneum, ovary, uterus, lung, thoracic cavity | 284 | Alive | 2 | 5 |
- 1. Papavramidis T, Papavramidis S. Solid pseudopapillary tumors of the pancreas: review of 718 patients reported in English literature. J Am Coll Surg 2005; 200: 965-72. ArticlePubMed
- 2. Yu PF, Hu ZH, Wang XB, et al. Solid pseudopapillary tumor of the pancreas: a review of 553 cases in Chinese literature. World J Gastroenterol 2010; 16: 1209-14. ArticlePubMedPMC
- 3. Hosokawa I, Shimizu H, Ohtsuka M, et al. Preoperative diagnosis and surgical management for solid pseudopapillary neoplasm of the pancreas. J Hepatobiliary Pancreat Sci 2014; 21: 573-8. ArticlePubMed
- 4. Xu M, Shi X, Wan T, et al. Treatment and prognosis of solid pseudopapillary neoplasms with metastases or recurrence. Zhonghua Wai Ke Za Zhi 2015; 53: 685-9. PubMed
- 5. Takahashi Y, Fukusato T, Aita K, et al. Solid pseudopapillary tumor of the pancreas with metastases to the lung and liver. Pathol Int 2005; 55: 792-6. ArticlePubMed
- 6. El Nakeeb A, Abdel Wahab M, Elkashef WF, Azer M, Kandil T. Solid pseudopapillary tumour of the pancreas: incidence, prognosis and outcome of surgery (single center experience). Int J Surg 2013; 11: 447-57. ArticlePubMed
- 7. Nishihara K, Nagoshi M, Tsuneyoshi M, Yamaguchi K, Hayashi I. Papillary cystic tumors of the pancreas. Assessment of their malignant potential. Cancer 1993; 71: 82-92. ArticlePubMed
- 8. Tipton SG, Smyrk TC, Sarr MG, Thompson GB. Malignant potential of solid pseudopapillary neoplasm of the pancreas. Br J Surg 2006; 93: 733-7. ArticlePubMedPDF
- 9. Yu P, Cheng X, Du Y, et al. Solid pseudopapillary neoplasms of the pancreas: a 19-year multicenter experience in China. J Gastrointest Surg 2015; 19: 1433-40. ArticlePubMedPDF
- 10. Ansari D, Elebro J, Tingstedt B, et al. Single-institution experience with solid pseudopapillary neoplasm of the pancreas. Scand J Gastroenterol 2011; 46: 1492-7. ArticlePubMed
- 11. Kim SA, Kim MS, Kim MS, et al. Pleomorphic solid pseudopapillary neoplasm of the pancreas: degenerative change rather than high-grade malignant potential. Hum Pathol 2014; 45: 166-74. ArticlePubMed
- 12. Kloppel G, Hruban RH, Klimstra DS, et al. Solid-pseudopapillary tumor of pancreas. In: Bosman FT, Carneiro F, Hruban RH, Theise ND, eds. World Health Organization classification of tumours of the digestive system. Lyon: IARC Press, 2010; 327-30.
REFERENCES
Figure & Data
References
Citations
Citations to this article as recorded by 

- Solid Pseudopapillary Neoplasms (SPNs) of the Pancreas: Appraisal of Contemporary Clinicopathologic Features ‐ Clinical Implications and Surgical Outcomes of Patients Treated and Followed at a Single Institution
Subhadra Acharya, Luke T. Meredith, Zachary Kaplan, Rehana Persaud, David Moskal, Brandon Wummer, Wei Jiang, Richard Zheng, Haresh Naringrekar, Harish Lavu, Charles J. Yeo, Avinoam Nevler, Wilbur B. Bowne
Journal of Surgical Oncology.2026;[Epub] CrossRef - A rare case of a large solid pseudopapillary neoplasm with extensive liver metastasis
Jun Hyung Kim, Hyung Sun Kim, Jung Min Lee, Ji Hae Nahm, Joon Seong Park
Annals of Hepato-Biliary-Pancreatic Surgery.2025; 29(1): 83. CrossRef - Curative Resection for Metastatic Solid Pseudopapillary Neoplasm of Pancreas—a Case Series
Aparna M. Jagannathan, Manbha L. Rymbai, Abhilasha Anand, Anoop Paul, Borna Das, Thomas Alex Kodiatte, Frederick L. Vyas, Ravish Sanghi Raju, Philip Joseph
Indian Journal of Surgical Oncology.2024; 15(S2): 232. CrossRef - Malignant Solid Pseudopapillary Neoplasm of the Pancreas: An Orthogonal Analysis
Andrew M. Fleming, Leah E. Hendrick, Danny Yakoub, Hafeez Abdelhafeez, Jeremiah L. Deneve, Max R. Langham, Evan S. Glazer, Andrew M. Davidoff, Nipun B. Merchant, Paxton V. Dickson, Andrew J. Murphy
Annals of Surgical Oncology.2024; 31(1): 475. CrossRef - A Unique Presentation of Solid Pseudopapillary Neoplasm of the Pancreas Requiring Pancreaticoduodenectomy Without Pancreatojejunostomy: A Case Report and Literature Review
Alexis L Carmona, Sameh A Fayek
Cureus.2024;[Epub] CrossRef - Case report: Clinical analysis and literature review of five cases of metastatic solid pseudopapillary tumor of the pancreas
Run Hu, Renjie Gui, Xi Nie, Huaxin Duan
Frontiers in Oncology.2024;[Epub] CrossRef - Case of Treatment of Solid Pseudopapillary Pancreatic Tumor
F. S. Rakhimova, N. D. Mamashev, O. A. Shimkina, B. Kh. Bebezov
Creative surgery and oncology.2023; 13(2): 178. CrossRef - Diagnosis and treatment of solid pseudopapillary neoplasm of the pancreas in children: A report of 18 cases
Ayiguzaili Maimaijiang, Haiyun Wang, Wanfu Li, Yaqi Wang
Frontiers in Pediatrics.2022;[Epub] CrossRef - Large tumor size, lymphovascular invasion, and synchronous metastasis are associated with the recurrence of solid pseudopapillary neoplasms of the pancreas
Goeun Lee, You-Na Sung, Sung Joo Kim, Jae Hoon Lee, Ki-Byung Song, Dae Wook Hwang, Jihun Kim, Sang Soo Lee, Song Cheol Kim, Seung-Mo Hong
HPB.2021; 23(2): 220. CrossRef - Solid-Pseudopapillary Neoplasm of the Pancreas: A 63-Case Analysis of Clinicopathologic and Immunohistochemical Features and Risk Factors of Malignancy
Hongchun Chen, Yuchen Huang, Ningning Yang, Wentian Yan, Ruxue Yang, Shan Zhang, Panpan Yang, Nan Li, Zhenzhong Feng
Cancer Management and Research.2021; Volume 13: 3335. CrossRef - Minimally Invasive Approach for Pancreatic Solid Pseudopapillary Neoplasm and Initially Undiagnosed Slowly Progressing Liver Tumor
Shohei Takaichi, Yoshifumi Iwagami, Shogo Kobayashi, Yoshito Tomimaru, Hirofumi Akita, Tadafumi Asaoka, Takehiro Noda, Kunihito Gotoh, Masaki Mori, Yuichiro Doki, Hidetoshi Eguchi
Pancreas.2020; 49(8): e70. CrossRef - Rare Solid Tumor of the Exocrine Pancreas: A Pictorial Review
Marco Dioguardi Burgio, Maxime Ronot, Valérie Vilgrain
Seminars in Ultrasound, CT and MRI.2019; 40(6): 483. CrossRef - The Stomach: a Rare Site for Metastatic Solid Pseudopapillary Neoplasm of the Pancreas
Prajwala S. Prakash, Dexter Yak Seng Chan, Krishnakumar Madhavan
Journal of Gastrointestinal Surgery.2018; 22(4): 759. CrossRef - European evidence-based guidelines on pancreatic cystic neoplasms
Gut.2018; 67(5): 789. CrossRef
 PubReader
PubReader ePub Link
ePub Link-
 Cite this Article
Cite this Article
- Cite this Article
-
- Close
- Download Citation
- Close
- Figure
A Rare Case of Recurrent Metastatic Solid Pseudopapillary Neoplasm of the Pancreas


Fig. 1. Gross specimen of the patient’s lung metastasis. A solid, well-demarcated and cystic mass with hemorrhage is seen.
Fig. 2. Microscopic findings. (A) A loosely cohesive pseudopapillary pattern. (B) Capsular invasion with a cord or glandular pattern is visible in the intermediate and late metastatic lesions. (C) Necrobiotic nests are present in the intermediate lesions. (D) Distinct nuclear atypia is seen in the late lesions.
Fig. 1.
Fig. 2.
A Rare Case of Recurrent Metastatic Solid Pseudopapillary Neoplasm of the Pancreas
| Operation No. | Recurrence interval (mo) | Site | Size (largest diameter) (cm) | Necrobiotic nests | Necrosis | Capsular invasion | Ki-67 labeling index (%) | Solid architecture | Nuclear atypia | Mitotic count (/10HPF) |
|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 0 | Pancreas | 8 | NE | NE | NE | NE | NE | NE | NE |
| 2 | 107 | Liver, peritoneum | 5.5 (liver) | N | N | N | < 0.1 | N | N | < 1 |
| 3 | 131 | Omentum, peritoneum | 4.3 (peritoneum) | N | N | N | < 0.1 | N | N | < 1 |
| 4 | 135 | Peritoneum | 1.3 | Y | N | N | < 0.1 | N | N | < 1 |
| 5 | 180 | Peritoneum | 3.5 | N | N | Y | 0.2 | N | N | 5 |
| 6 | 197 | Peritoneum | 5 | N | N | Y | 5 | N | N | < 1 |
| 7 | 205 | Liver, spleen, peritoneum, ovary | 10 (ovary) | Y | Y | N | 2.9 | N | N | 2 |
| 8 | 214 | Uterus, peritoneum, retroperitoneum, omentum, mesocolon, intrathoracic | 3.8 (intrathoracic) | N | Y | N | 12.4 | Y | N | < 1 |
| 9 | 240 | Peritoneum, mesocolon, colon | 1.5 (colon) | N | Y | Y | 6.6 | Y | Y | < 1 |
| 10 | 272 | Spleen, retroperitoneum | 3.5 (retroperitoneum) | N | Y | Y | < 0.1 | Y | Y | < 1 |
| 11 | 284 | Lung, internal mammary soft tissue | 2.8 (lung) | N | N | N | 3.4 | Y | Y | < 1 |
| Reference | Sex | Age (yr) | Primary site | Size (cm) | Malignant histologic features | Metastasis | Operation | RI (mo) | Recurrence site | Follow-up (mo) | Survival | Mitoses (/10HPF) | Ki-67 (%) |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Takahashi et al. (2005) [5] | F | 41 | Tail | 12 | Capsule invasion, necrobiotic nests, venous invasion | Liver (multiple) | Distal pancreatectomy, metastatectomy | 14 | Liver, multiple (14 mo); liver, multiple (27 mo); liver, multiple (38 mo); lung, single (41 mo) | 60 | Alive | 5 | 10 |
| Hosokawa et al. (2014) [3] | F | 24 | Head | 6 | Capsule invasion, per pancreatic tissue | PV, SMA, lung | PPPD, PV, resection, pulmonary resection | None | None | 34 | Alive | - | 1.5 |
| Xu et al. (2015) [4] | F | 42 | Body | 13 | NE | Liver, lung, bone | NE | NE | NE | NE | NE | NE | NE |
| Our case (2016) | F | 37 | Head | 8.6 | Capsule invasion, necrobiotic nests, lymphatic invasion, lymph node invasion | None | PPPD | 107 | Liver, spleen, abdominal mesentery, mesocolon, retroperitoneum, ovary, uterus, lung, thoracic cavity | 284 | Alive | 2 | 5 |
Table 1. Clinicopathological data of the patient
HPF, high power field; NE, not evaluable; N, absent; Y, present.
Table 2. Cases of solid pseudopapillary neoplasm with lung metastasis
RI, recurrence interval; HPF, high power field; F, female; PV, portal vein; SMA, superior mesenteric artery; PPPD, pylorus-preserving pancreaticoduodenectomy; NE, not evaluable.

 E-submission
E-submission