Articles
- Page Path
- HOME > J Pathol Transl Med > Volume 54(5); 2020 > Article
-
Original Article
Prediction of TP53 mutations by p53 immunohistochemistry and their prognostic significance in gastric cancer -
Hye Jung Hwang1
 , Soo Kyung Nam1
, Soo Kyung Nam1 , Hyunjin Park2
, Hyunjin Park2 , Yujun Park1
, Yujun Park1 , Jiwon Koh3
, Jiwon Koh3 , Hee Young Na1
, Hee Young Na1 , Yoonjin Kwak3
, Yoonjin Kwak3 , Woo Ho Kim3
, Woo Ho Kim3 , Hye Seung Lee1
, Hye Seung Lee1
-
Journal of Pathology and Translational Medicine 2020;54(5):378-386.
DOI: https://doi.org/10.4132/jptm.2020.06.01
Published online: July 1, 2020
1Department of Pathology, Seoul National University Bundang Hospital, Seoul National University College of Medicine, Seongnam, Korea
2Department of Pathology, Gangnam Severance Hospital, Yonsei University College of Medicine, Seoul, Korea
3Department of Pathology, Seoul National University Hospital, Seoul National University College of Medicine, Seoul, Korea
- Corresponding Author: Hye Seung Lee, MD, PhD, Department of Pathology, Seoul National University Bundang Hospital, Seoul National University College of Medicine, 82 Gumi-ro 173beon-gil, Bundang-gu, Seongnam 13620, Korea Tel: +82-31-787-7714, Fax: +82-31-787-4012, E-mail: hye2@snu.ac.kr
© 2020 The Korean Society of Pathologists/The Korean Society for Cytopathology
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/4.0) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
Abstract
-
Background
- Recently, molecular classifications of gastric cancer (GC) have been proposed that include TP53 mutations and their functional activity. We aimed to demonstrate the correlation between p53 immunohistochemistry (IHC) and TP53 mutations as well as their clinicopathological significance in GC.
-
Methods
- Deep targeted sequencing was performed using surgical or biopsy specimens from 120 patients with GC. IHC for p53 was performed and interpreted as strong, weak, or negative expression. In 18 cases (15.0%) with discrepant TP53 mutation and p53 IHC results, p53 IHC was repeated.
-
Results
- Strong expression of p53 was associated with TP53 missense mutations, negative expression with other types of mutations, and weak expression with wild-type TP53 (p<.001). The sensitivity for each category was 90.9%, 79.0%, and 80.9%, and the specificity was 95.4%, 88.1%, and 92.3%, respectively. The TNM stage at initial diagnosis exhibited a significant correlation with both TP53 mutation type (p=.004) and p53 expression status (p=.029). The Kaplan-Meier survival analysis for 109 stage II and III GC cases showed that patients with TP53 missense mutations had worse overall survival than those in the wild-type and other mutation groups (p=.028). Strong expression of p53 was also associated with worse overall survival in comparison to negative and weak expression (p=.035).
-
Conclusions
- Results of IHC of the p53 protein may be used as a simple surrogate marker of TP53 mutations. However, negative expression of p53 and other types of mutations of TP53 should be carefully interpreted because of its lower sensitivity and different prognostic implications.
- Characterization of patients and sample acquisition
- The study population was composed of 120 patients treated at Seoul National University Bundang Hospital (Seongnam, Korea) from 2009 to 2019. The median age was 60 years (range, 34 to 82 years), and 85 patients (70.8%) were men. Thirty-eight of the 120 cases (31.7%) were stage II at initial diagnosis, 71 (59.2%) cases were stage III, and 11 (9.2%) were stage IV. Among them, 109 stage II and III patients (90.8%) underwent curative radical resection (R0 resection) without preoperative chemotherapy or radiotherapy. In the 11 stage IV cases, endoscopic biopsy specimen was collected in one case, metastatectomy specimens in four cases, conversion surgery specimens after chemotherapy in five patients, and gastrectomy specimen in one case for the experiments. Analysis according to the World Health Organization (WHO) classification [12] revealed that tubular adenocarcinoma accounted for 54.2% (65 cases) of diagnoses, mucinous adenocarcinoma for 3.3% (4 cases), papillary adenocarcinoma for 3.3% (4 cases), poorly cohesive carcinoma for 30.0% (36 cases), and other minor histologic types for 9.2% (11 cases). For survival analysis, 109 patients with stage II and III GC were followed up from the date of surgery to the date of death or final follow-up. The median follow-up period was 42.2 months (range of 5.4-87.7 months).
- Next-generation sequencing
- Targeted sequencing of 170 cancer-related gene panels was performed using formalin-fixed, paraffin-embedded tissue (FFPE) samples as previously described [13]. All FFPE materials had a short cold ischemic time not exceeding 2 hours, fixation time ranging from 8 to 72 hours, and were aged between 0 and 9 years.
- In brief, approximately 3 µg of genomic DNA was extracted from FFPE tumor tissues, and the sequencing library was prepared using an Agilent SureSelect Target Enrichment Kit (Agilent Technologies, Santa Clara, CA, USA) following the manufacturer’s guidelines. High-throughput sequencing was performed using the HiSeq 2500 system (Illumina, San Diego, CA, USA) (Macrogen Inc., Seoul, Korea). After quality control of the FASTQ files, sequencing reads were aligned to the reference genome (GRCh37/hg19) using Burrows-Wheeler Aligner-MEM (BWA-MEM) [14]. Single nucleotide variants and small insertions and deletions (INDELs) were detected using the MuTect2 algorithm [15]. SnpEff and SnpSift v4.3i [16] with dbNSFP v2.9.3 [17] were used for variant annotation with various databases including the OncoKB [18] and ClinVar archives [19].
- IHC staining
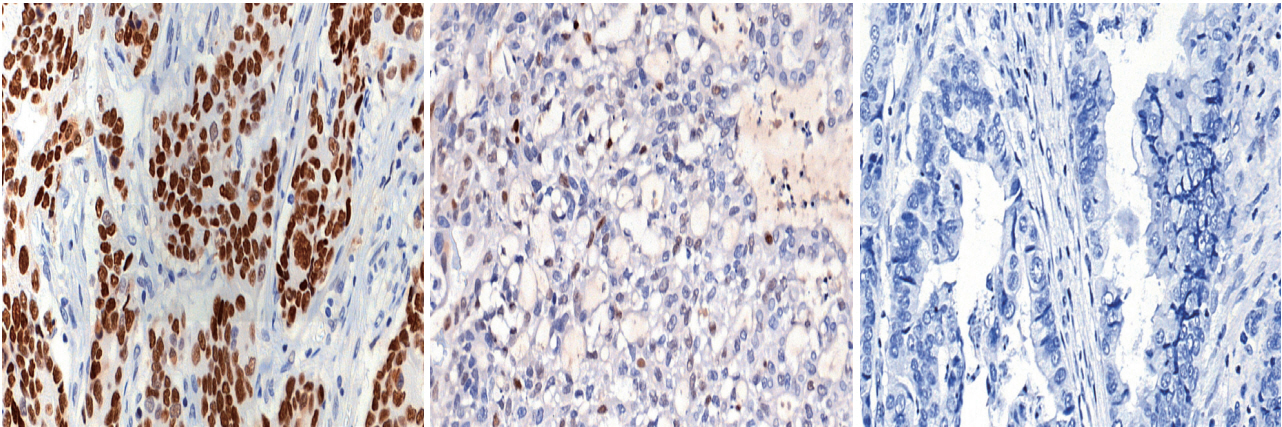
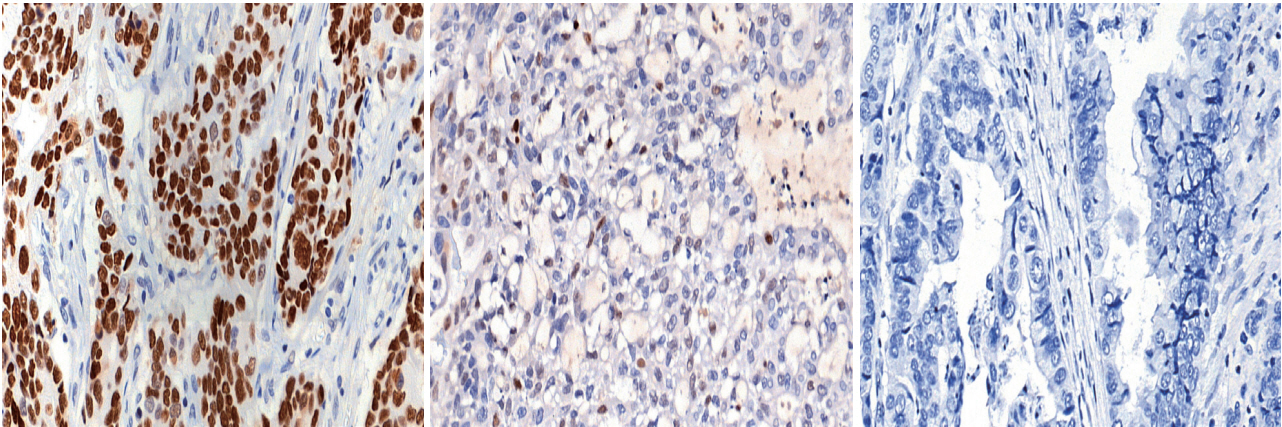
- Immunohistochemical (IHC) staining for p53 (DO7, mouse monoclonal, Dako, Agilent Technologies) was performed on 3-μm-thick slides using an automated immunostainer (BenchMark XT, Ventana Medical Systems, Tucson, AZ, USA) following the manufacturer’s protocol. The p53 IHC was interpreted in three tiers: strong nuclear staining in more than 10% of the tumor cells was considered strong positivity, samples without any nuclear staining of tumor cells (complete absence) were interpreted as negativity, and cases exhibiting weak, scattered, or patchy positivity were regarded as weak positivity. Representative images for each category are shown in Fig. 1. Cut-offs of 20% and 30% nuclear positivity were additionally applied for validation of the results.
- For cases where gene mutation and protein expression status did not match (18 cases or 15.0%), p53 IHC was repeatedly performed and interpreted. In most cases (17 out of 18 or 94.4%), repeated immunohistochemical assays did not alter the initial interpretation. Tumor heterogeneity accounted for the change in one case. Initially, strong nuclear expression of p53 was observed in some areas of the tumor (<10%) but was not sufficient to be classified as strong expression. Subsequent IHC was performed on another section of the same tumor, exhibiting overall strong expression of p53.
- EBV in-situ hybridization
- The EBV status was tested using EBV in-situ hybridization as previously described [20]. A fluorescein-conjugated EBV encoded small RNA (EBER) oligonucleotide probe (INFORM EBVencoded RNA probe, Ventana Medical Systems) was used, and positive cases were defined as diffuse nuclear reactivity for EBER in tumor cells.
- Microsatellite instability analysis
- Representative tumor tissues and matched normal gastric mucosal tissues were selected for microsatellite instability (MSI) testing. Five NCI markers (BAT-26, BAT-25, D5S346, D17S250, and S2S123) amplified through polymerase chain reaction were analyzed using an automated sequencer (ABI 3731 Genetic Analyzer; Applied Biosystems, Foster City, CA, USA). MSI-high was defined as two or more markers with unstable peaks, MSI-low was defined as one unstable marker, and microsatellite stable was defined as no unstable marker.
- Statistical analyses
- Chi-square or Fisher exact tests were used to assess significant differences in the distribution of TP53 mutations and p53 expression. For univariate survival analysis, Kaplan-Meier survival curves were plotted in 109 patients with stage II and III GC cases. The survival differences were compared using the log-rank test. For multivariate survival analysis, the Cox regression model was used. All statistical analyses were performed using SPSS Statistics ver. 22.0 (IBM Corp., Armonk, NY, USA).
MATERIALS AND METHODS
- Gene mutation and protein expression correlation
- Table 1 summarizes the p53 IHC results according to TP53 mutations. TP53 mutations were present in 52 cases (43.3%), of which missense mutations were the most common (33 of 52 cases, 63.5%). Strong expression was observed in 34 cases (28.3%) and negative expression was observed in 27 cases (22.5%). When TP53 mutations were compared with p53 IHC, 30 of the 33 missense mutation cases (90.9%) exhibited strong p53 expression, but negative expression of p53 was the dominant pattern (15 cases, 78.9%) among the 19 cases of other types of mutations (p<.001). Based on clinical significance, 37 cases (30.1%) had pathogenic or likely pathogenic TP53 mutations, of which 22 cases (59.5%) exhibited strong expression of p53, 13 cases (36.1%) negative expression, and two cases (5.4%) weak expression (p<.001). Nevertheless, most cases of uncertain significance (62.5%) and conflicting interpretations (85.7%) also showed strong expression of p53 by IHC.
- Detailed information about TP53 mutations and p53 expression status is shown in Table 2. Two mutations were observed in three cases, of which one representative mutation was included in this table. One among seven cases with TP53 mutations of conflicting interpretations regarding pathogenicity had weak expression of p53 (case No. 27 in Table 2). There have been reports suggestive of the “likely benign” and “uncertain significance” nature of this mutation. The mutations c.659A>G, c.742C>T, c.817C>T, c.796G>A, c.1024C>T, and c.375G>A were found in two cases, and c.818G>A mutation was found in three cases. The IHC results matched in cases with the same mutation. In 44 cases with single nucleotide polymorphism, C:G to T:A conversion was observed in 32 (72.7%), C:G to A:T in four (9.1%), C:G to G:C in two (4.5%), T:A to C:G in four (9.1%), and T:A to G:C in two (4.5%).
- Sensitivity, specificity, and accuracy of p53 IHC for predicting TP53 mutations
- In general, nonsynonymous mutations detected using NGS were related to strong p53 expression in IHC. Similarly, all other types of mutations tended to show negative expression, of p53 while cases with wild-type TP53 exhibited weak protein expression. The sensitivity of strong expression of p53 by IHC for predicting nonsynonymous TP53 mutations was 90.9%, sensitivity of negative expression for other types of mutations was 79.0%, and the sensitivity of weak expression for wild-type TP53 was 80.9% (Table 3). The specificity for each category was 95.4%, 88.1%, and 92.3%, respectively. The accuracy for each category was 94.2%, 86.7%, and 85.8%, respectively. In addition, the sensitivity, specificity, and accuracy of p53 IHC at 20% and 30% cut-offs are shown in Supplementary Table S1. The sensitivity of strong expression of p53 for nonsynonymous TP53 mutations was highest at the 10% cut-off.
- Clinicopathological variables and protein expression correlations
- The correlation between clinicopathological characteristics and TP53 mutations or p53 expression status is summarized in Table 4. TNM stage at initial diagnosis was the only variable that showed significant correlation with both TP53 mutation type and p53 expression status (p=.004 and p=.029, respectively). Of the 38 stage II gastric cancer cases, 27 (71.1%) did not exhibit any detectable mutations in the TP53 gene, but five nonsynonymous (13.2%) and six other types of mutations (15.8%) were found. Strong p53 expression was found in seven of the 38 stage II cases (18.4%). Among the stage III cases, which accounted for 71 cases, the proportions of nonsynonymous gene mutations and strong expression of p53 mutations increased to 39.4% (28 cases) and 38.0% (27 cases), respectively. On the other hand, the proportions of wild-type TP53 cases and weak expression cases decreased from 71.0% to 45.0% and from 55.2% to 43.6%, respectively.
- TP53 mutations were more frequently observed in intestinaltype GC (25 of 45 cases, 55.6%) compared to the non-intestinal type (27 of 75 cases, 36.0%), but with borderline statistical significance (p=.065). Other clinicopathological variables such as sex, age, tumor location, and WHO classification were not statistically significant.
- Survival analysis
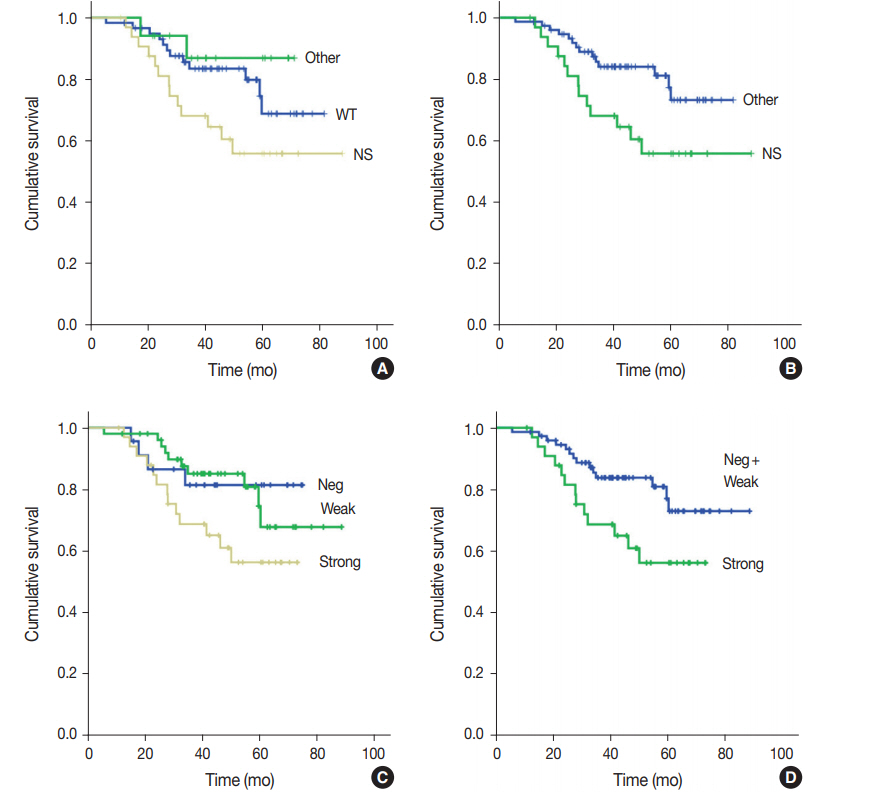
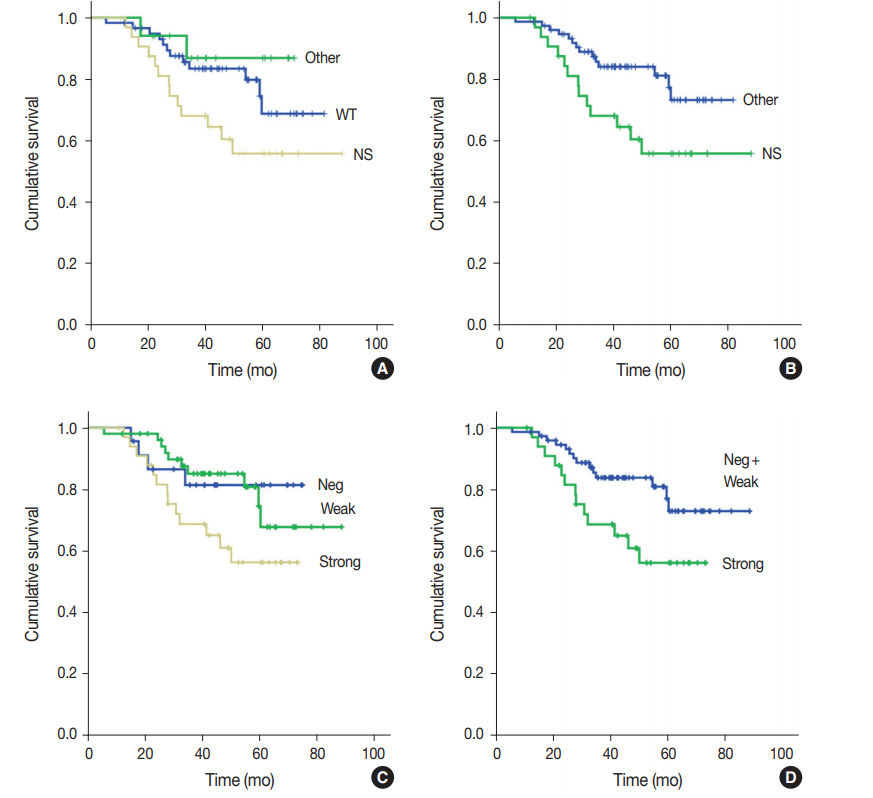
- One hundred nine patients with stage II and III GC at initial diagnosis were selected for survival analysis. The patients underwent curative surgery followed by adjuvant chemotherapy. Patients with any TP53 mutations tended to have worse OS compared to those without mutations, although the difference was not statistically significant (p=.227). When OS was analyzed based on TP53 mutation type, patients with nonsynonymous mutations had the worst OS, and the wild-type and other types of mutations exhibited similar OS (p=.074) (Fig. 2A). This trend became statistically significant when the nonsynonymous mutation group was compared to the combined wild-type and other mutation groups (p=.028) (Fig. 2B). The expression pattern of p53 was not significantly associated with patient OS (p=.107) (Fig. 2C), but it was statistically significant when strong expression of p53 was compared to the combined negative and weak expression cases (p=.035) (Fig. 2D). Patients with abnormal— negative and strong expression—expression did not exhibit a statistically significant survival difference compared to patients with weak expression (p=.208). The Kaplan-Meier survival curves of p53 expression status at 20% and 30% cut-offs were additionally plotted in Supplementary Fig. S1. The difference in survival was largest at the 30% cut-off. Multivariate Cox regression analysis showed that strong expression of p53 was associated with patient OS independent of stage with borderline significance (p=.070, data not shown). The presence of nonsynonymous missense mutations of TP53 was not an independent prognostic factor in multivariate analysis (p=.130).
RESULTS
- TP53 is the most well-known tumor suppressor gene, and p53 IHC is a method used in daily practice as a surrogate marker in various cancer patients. In this study, we performed targeted deep sequencing for detecting various TP53 mutations and IHC for p53 using a commercially available and validated primary antibody with an automatic immunostainer. Strong expression of p53 could predict nonsynonymous missense mutations of TP53 with a sensitivity of 90.9%, specificity of 95.4%, and accuracy of 94.2%. However, weak expression of p53 was less specific (80.9%) for predicting wild-type TP53, and negative expression was less sensitive (79.0%) for predicting other mutations of TP53. These results suggest that p53 IHC can be used as a surrogate marker in predicting TP53 mutations, especially for strong expression, to predict nonsynonymous mutations. There have been recent attempts to use IHC for molecular classifications, and their clinicopathological significance has been increasingly important in GC [8,10]. Our results will be helpful for these new molecular classifications although negative expression should be cautiously interpreted.
- Based on the Kaplan-Meier survival analysis, strong expression of p53 was significantly associated with worse OS compared to weak and negative expression of p53 in this study. Previous studies that investigated any relationship between p53 overexpression and survival reported p53 overexpression as a poor prognostic factor [21-23], similar to the findings from our study. Some studies did not reveal the prognostic significance of p53 overexpression in GC, but a meta-analysis demonstrated that it is a poor prognostic factor [24]. In those studies, the median cut-off value was 10% [24]. Therefore, we applied a cut-off value of 10% for defining strong expression of p53. In addition to the 10% cutoff, we applied 20% and 30% cut-offs in this study. Although the survival difference was largest at the 30% cut-off, the sensitivity of strong expression of p53 for predicting nonsynonymous TP53 mutation was highest at the 10% cut-off. Therefore, further studies are needed to validate various cut-offs.
- For interpretation of p53 IHC, Köbel et al. [11,25] proposed a three-tiered scoring system, including overexpression, complete absence, and normal or wild-type pattern in ovarian cancer. The scoring system exhibited good correlation with TP53 mutation status: overexpression with nonsynonymous mutation; complete absence with stop gain, frameshift, and splicing mutations; and a normal pattern with the wild-type TP53 gene [11]. Shin et al. [26] investigated the prognostic roles of p53 expression status in patients with GC. They defined group 0 as complete absence, group 1 as weak staining in <50%, group 2 as strong staining in 50%–90%, and group 3 as strong staining in >90%. When the Kaplan-Meier survival analysis was performed, group 1 was associated with better survival than groups 0, 2, and 3, but with borderline statistical significance. Our results showed a similar relationship between p53 IHC and TP53 mutation status to those of previous studies. If weak expression in this study was defined as a normal or wild-type pattern, the Kaplan-Meier survival curves did not show a significant difference between normal and abnormal expression patterns. Furthermore, patients with GC having nonsynonymous TP53 mutations had significantly worse prognosis compared to patients with other types of mutations and the wild-type TP53 group. Similarly, strong expression of p53, which was related to nonsynonymous TP53 mutations, was shown to be a poor prognostic factor. The complete absence of p53 expression or other types of TP53 mutations might not be significant for predicting prognosis.
- There were 17 cases (14.2%) with discrepant results between p53 IHC and TP53 mutations. Most discrepant cases had negative expression of p53 and wild-type TP53. Weak expression of p53 was observed in four missense mutation cases and one stop gain mutation case. These findings might be due to tumor heterogeneity or tissue quality issues, such as specimen ischemic time or archival age. In addition, there was one case with weak p53 expression and a missense mutation of conflicting pathogenic interpretation. Considering this case was conflicting between uncertain significance and likely benign significance, one of the possible reasons for the discrepancy is a non-pathogenic mutation. To discriminate functional and nonfunctional p53, Nenutil et al. [27] performed IHC for p53, Ki67, MDM2, and p21 in human cancers, and overexpressed p53 without increased MDM2 indicated inactivating mutations in their study. p21, a transcriptional target of p53, was considered to reflect p53 activity and could decrease false-positive results of p53 expression [28]. The ACRG considered a TP53-activity signature using p21 and MDM2 genes [7]. Therefore, in addition to p53 IHC, IHC for p21 and MDM2 would be helpful for evaluating the functional status of p53. The need for these additional analyses reflects a potential limitation of this study, necessitating further research.
- In a total of 120 gastric cancer cases, 52 (43.3%) had TP53 mutations, including nonsynonymous missense, frameshift, stop gain, in-frame deletion, and splice region mutations. Hot spot mutations within the central core (R175, G245, R248, R273, and R282) were observed in a minority of cases (10 out of 120 or 8.3%). In accordance with our results, a previous study reported hot spot mutations in 6.2% of gastric cancer cases [29]. Therefore, sequencing (Sanger or NGS) is suggested as a suitable method for detecting TP53 mutations in gastric cancer.
- In summary, we investigated the relationship between p53 expression and TP53 mutation status to predict TP53 mutations by p53 IHC and reveal their prognostic significance. TP53 mutations were observed in 43.3% of cases. Strong p53 expression could predict nonsynonymous missense mutations with high sensitivity and specificity, but only half of the p53 negative cases (55.6%) exhibited other types of TP53 mutations. Protein overexpression and nonsynonymous genetic mutations of TP53 significantly predicted worse OS. p53 IHC could be regarded as a simple surrogate marker of TP53 mutations, but negative expression of p53 and other types of TP53 mutations should be cautiously considered in daily practice or scientific research. Overall, our study results will be informative for simple molecular classification of patients with GC.
DISCUSSION
Supplementary Materials
Ethics Statement
This study was approved by the Institutional Review Board of Seoul National University Bundang Hospital (IRB number: B-2001/591-105). Written informed consent was waived by the IRB.
Author contributions
Conceptionalization: WHK, HSL. Data curation: HJH, HSL. Formal analysis: HJH, HSL. Funding Acquisition: HSL. Investigation: HJH, YP, HP, JK, HYN, YK, HSL. Methodology: HJH, SKN, HP, HSL. Supervision: HSL. Writing—original draft: HJH, HSL. Writing—review & editing: HJH, WHK, HSL. Approval of final manuscript: all authors.
Conflicts of Interest
W.H.K. and H.S.L., contributing editors of the Journal of Pathology and Translational Medicine, were not involved in the editorial evaluation or decision to publish this article. All remaining authors have declared no conflicts of interest.
Funding Statement
This work was supported by a National Research Foundation of Korea (NRF) grant funded by the Korean government (Ministry of Science and ICT) (No. 2019R1A2C1086180).
| TP53 mutation |
p53 expression by IHC |
Total | p-value | ||
|---|---|---|---|---|---|
| Strong | Negative | Weak | |||
| Mutation status | < .001 | ||||
| Wild-type | 1 (2.9) | 12 (44.4) | 55 (93.2) | 68 (56.7) | |
| Mutation present | 33 (97.1) | 15 (55.6) | 4 (6.8) | 52 (43.3) | |
| Variant summary | < .001 | ||||
| Wild-type | 1 (2.9) | 12 (44.4) | 55 (93.2) | 68 (56.7) | |
| Missense | 30 (88.2) | 0 | 3 (5.1) | 33 (27.5) | |
| Other | 3 (8.9) | 15 (55.6) | 1 (1.7) | 19 (15.8) | |
| Stop-gained | 2 (5.9) | 3 (11.1) | 1 (1.7) | 6 (5.0) | |
| Splice region | 0 | 5 (18.5) | 0 | 5 (4.2) | |
| Frameshift | 0 | 7 (25.9) | 0 | 7 (5.8) | |
| In-frame deletion | 1 (2.9) | 0 | 0 | 1 (0.8) | |
| Clinical significancea | < .001 | ||||
| Wild-type | 1 (2.9) | 12 (44.4) | 55 (93.2) | 68 (56.7) | |
| Pathogenic or likely pathogenic | 22 (64.7) | 13 (48.1) | 2 (3.4) | 37 (30.1) | |
| Uncertain significance | 5 (14.7) | 2 (7.4) | 1 (1.7) | 8 (6.7) | |
| Conflicting interpretation | 6 (17.6) | 0 | 1 (1.7) | 7 (5.8) | |
| Total | 34 | 27 | 59 | 120 | |
| Case No. | Effect | Nucleic acid alteration | Amino acid alteration | Clinical significancea |
|---|---|---|---|---|
| 1 | Missense_variant | c.422G > A | p.Cys141Tyr | Pathogenic or likely pathogenic |
| 2 | Missense_variant | c.422G > T | p.Cys141Phe | Pathogenic or likely pathogenic |
| 3 | Missense_variant | c.455C > T | p.Pro152Leu | Pathogenic or likely pathogenic |
| 4 | Missense_variant | c.524G > A | p.Arg175His | Pathogenic or likely pathogenic |
| 5 | Missense_variant | c.535C > G | p.His179Asp | Pathogenic or likely pathogenic |
| 6 | Missense_variant | c.542G > A | p.Arg181His | Pathogenic or likely pathogenic |
| 7 | Missense_variant | c.659A > G | p.Tyr220Cys | Pathogenic or likely pathogenic |
| 8 | Missense_variant | c.659A > G | p.Tyr220Cys | Pathogenic or likely pathogenic |
| 9 | Missense_variant | c.701A > G | p.Tyr234Cys | Pathogenic or likely pathogenic |
| 10 | Missense_variant | c.725G > A | p.Cys242Tyr | Pathogenic or likely pathogenic |
| 11 | Missense_variant | c.734G > A | p.Gly245Asp | Pathogenic or likely pathogenic |
| 12 | Missense_variant | c.742C > T | p.Arg248Trp | Pathogenic or likely pathogenic |
| 13 | Missense_variant | c.742C > T | p.Arg248Trp | Pathogenic or likely pathogenic |
| 14 | Missense_variant | c.743G > A | p.Arg248Gln | Pathogenic or likely pathogenic |
| 15 | Missense_variant | c.772G > A | p.Glu258Lys | Pathogenic or likely pathogenic |
| 16 | Missense_variant | c.817C > T | p.Arg273Cys | Pathogenic or likely pathogenic |
| 17 | Missense_variant | c.817C > T | p.Arg273Cys | Pathogenic or likely pathogenic |
| 18 | Missense_variant | c.818G > A | p.Arg273His | Pathogenic or likely pathogenic |
| 19 | Missense_variant | c.818G > A | p.Arg273His | Pathogenic or likely pathogenic |
| 20 | Missense_variant | c.818G > A | p.Arg273His | Pathogenic or likely pathogenic |
| 21 | Missense_variant | c.380C > T | p.Ser127Phe | Conflicting interpretations of pathogenicity |
| 22 | Missense_variant | c.473G > C | p.Arg158Pro | Conflicting interpretations of pathogenicity |
| 23 | Missense_variant | c.481G > A | p.Ala161Thr | Conflicting interpretations of pathogenicity |
| 24 | Missense_variant | c.613T > C | p.Tyr205His | Conflicting interpretations of pathogenicity |
| 25 | Missense_variant | c.796G > A | p.Gly266Arg | Conflicting interpretations of pathogenicity |
| 26 | Missense_variant | c.796G > A | p.Gly266Arg | Conflicting interpretations of pathogenicity |
| 27 | Missense_variant | c.1015G > A | p.Glu339Lys | Conflicting interpretations of pathogenicity |
| 28 | Missense_variant | c.329G > A | p.Arg110His | Uncertain significance |
| 29 | Missense_variant | c.380C > A | p.Ser127Tyr | Uncertain significance |
| 30 | Missense_variant | c.476C > T | p.Ala159Val | Uncertain significance |
| 31 | Missense_variant | c.797G > T | p.Gly266Val | Uncertain significance |
| 32 | Missense_variant | c.400T > G | p.Phe134Val | Uncertain significance |
| 33 | Missense_variant | c.470T > G | p.Val157Gly | Uncertain significance |
| 34 | Frameshift_variant | c.331_332insAG | p.Leu111fs | Pathogenic or likely pathogenic |
| 35 | Frameshift_variant | c.381_391delCCCTGCCCTCA | p.Pro128fs | Pathogenic or likely pathogenic |
| 36 | Frameshift_variant | c.635_669delTTCGACATAGTGTGGTG GTGCCCTATGAGCCGCCT | p.Phe212fs | Pathogenic or likely pathogenic |
| 37 | Frameshift_variant | c.660_661delTG | p.Tyr220fs | Pathogenic or likely pathogenic |
| 38 | Frameshift_variant | c.747delG | p.Arg249fs | Pathogenic or likely pathogenic |
| 39 | Frameshift_variant | c.1169delC | p.Pro390fs | Pathogenic or likely pathogenic |
| 40 | Frameshift_variant | c.778_779delTC | p.Ser260fs | Uncertain significance |
| 41 | Conservative_inframe_deletion | c.529_546delCCCCACCATGAGCGCTGC | p.Pro177_Cys182del | Pathogenic or likely pathogenic |
| 42 | Stop_gained | c.159G > A | p.Trp53* | Pathogenic or likely pathogenic |
| 43 | Stop_gained | c.437G > A | p.Trp146* | Pathogenic or likely pathogenic |
| 44 | Stop_gained | c.586C > T | p.Arg196* | Pathogenic or likely pathogenic |
| 45 | Stop_gained | c.637C > T | p.Arg213* | Pathogenic or likely pathogenic |
| 46 | Stop_gained | c.1024C > T | p.Arg342* | Pathogenic or likely pathogenic |
| 47 | Stop_gained | c.1024C > T | p.Arg342* | Pathogenic or likely pathogenic |
| 48 | Splice_region_variant&synonymous_variant | c.375G > A | p.Thr125Thr | Pathogenic or likely pathogenic |
| 49 | Splice_region_variant&synonymous_variant | c.375G > A | p.Thr125Thr | Pathogenic or likely pathogenic |
| 50 | Splice_region_variant&synonymous_variant | c.375G > C | p.Thr125Thr | Pathogenic or likely pathogenic |
| 51 | Splice_acceptor_variant&intron_variant | c.920 - 1G > A | Pathogenic or likely pathogenic | |
| 52 | Splice_donor_variant&intron_variant | c.96 + 1G > A | Uncertain significance (no report) |
Values are presented as number (%).
NS, nonsynonymous; Other, other type mutation; wild, wild-type; WHO, World Health Organization; WD, well-differentiated; MD, moderately differentiated; PD, poorly differentiated; PCC, poorly cohesive carcinoma; EBV, Epstein-Barr virus; MSI, microsatellite instability; MSS, microsatellite stable; MSI-L, microsatellite instability-low; MSI-H, microsatellite instability-high.
- 1. Junttila MR, Evan GI. p53: a Jack of all trades but master of none. Nat Rev Cancer 2009; 9: 821-9. ArticlePubMedPDF
- 2. Bartkova J, Horejsi Z, Koed K, et al. DNA damage response as a candidate anti-cancer barrier in early human tumorigenesis. Nature 2005; 434: 864-70. ArticlePubMedPDF
- 3. Starzynska T, Bromley M, Ghosh A, Stern PL. Prognostic significance of p53 overexpression in gastric and colorectal carcinoma. Br J Cancer 1992; 66: 558-62. ArticlePubMedPMCPDF
- 4. Kakeji Y, Korenaga D, Tsujitani S, et al. Gastric cancer with p53 overexpression has high potential for metastasising to lymph nodes. Br J Cancer 1993; 67: 589-93. ArticlePubMedPMCPDF
- 5. Maehara Y, Tomoda M, Hasuda S, et al. Prognostic value of p53 protein expression for patients with gastric cancer: a multivariate analysis. Br J Cancer 1999; 79: 1255-61. ArticlePubMedPMCPDF
- 6. Cancer Genome Atlas Research Network. Comprehensive molecular characterization of gastric adenocarcinoma. Nature 2014; 513: 202-9. ArticlePubMedPMCPDF
- 7. Cristescu R, Lee J, Nebozhyn M, et al. Molecular analysis of gastric cancer identifies subtypes associated with distinct clinical outcomes. Nat Med 2015; 21: 449-56. ArticlePubMedPDF
- 8. Setia N, Agoston AT, Han HS, et al. A protein and mRNA expressionbased classification of gastric cancer. Mod Pathol 2016; 29: 772-84. ArticlePubMedPDF
- 9. Ahn S, Lee SJ, Kim Y, et al. High-throughput Protein and mRNA Expression-based classification of gastric cancers can identify clinically distinct subtypes, concordant with recent molecular classifications. Am J Surg Pathol 2017; 41: 106-15. ArticlePubMed
- 10. Koh J, Lee KW, Nam SK, et al. Development and validation of an easy-to-implement, practical algorithm for the identification of molecular subtypes of gastric cancer: prognostic and therapeutic implications. Oncologist 2019; 24: e1321-30. ArticlePubMedPMCPDF
- 11. Kobel M, Piskorz AM, Lee S, et al. Optimized p53 immunohistochemistry is an accurate predictor of TP53 mutation in ovarian carcinoma. J Pathol Clin Res 2016; 2: 247-58. ArticlePubMedPMC
- 12. Fukayama M, Rugge M, Washington MK. Tumours of the stomach. In: Lokuhetty D, White VA, Watanabe R, Cree IA, eds. WHO classification of tumours. Lyon: International Agency for Research on Cancer (IARC), 2019; 85-103.
- 13. Koh J, Nam SK, Roh H, et al. Somatic mutational profiles of stage II and III gastric cancer according to tumor microenvironment immune type. Genes Chromosomes Cancer 2019; 58: 12-22. ArticlePubMedPDF
- 14. Martin M. Cutadapt removes adapter sequences from high-throughput sequencing reads. EMBnet J 2011; 17: 10-2. Article
- 15. Cibulskis K, Lawrence MS, Carter SL, et al. Sensitive detection of somatic point mutations in impure and heterogeneous cancer samples. Nat Biotechnol 2013; 31: 213-9. ArticlePubMedPMCPDF
- 16. Cingolani P, Patel VM, Coon M, et al. Using Drosophila melanogaster as a model for genotoxic chemical mutational studies with a new program, SnpSift. Front Genet 2012; 3: 35.ArticlePubMedPMC
- 17. Liu X, Wu C, Li C, Boerwinkle E. dbNSFP v3.0: a one-stop database of functional predictions and annotations for human nonsynonymous and splice-site SNVs. Hum Mutat 2016; 37: 235-41. ArticlePubMedPMC
- 18. Chakravarty D, Gao J, Phillips SM, et al. OncoKB: a precision oncology knowledge base. JCO Precis Oncol 2017 2017: 10.1200/PO.17.00011.
- 19. Landrum MJ, Lee JM, Benson M, et al. ClinVar: public archive of interpretations of clinically relevant variants. Nucleic Acids Res 2016; 44: D862-8. ArticlePubMedPDF
- 20. Park Y, Koh J, Na HY, et al. PD-L1 testing in gastric cancer by the combined positive score of the 22C3 PharmDx and SP263 assay with clinically relevant cut-offs. Cancer Res Treat 2020 Jan 10 [Epub]. https://doi.org/10.4143/crt.2019.718. ArticlePDF
- 21. Song KY, Jung CK, Park WS, Park CH. Expression of the antiapoptosis gene survivin predicts poor prognosis of stage III gastric adenocarcinoma. Jpn J Clin Oncol 2009; 39: 290-6. ArticlePubMedPDF
- 22. Mrena J, Wiksten JP, Kokkola A, Nordling S, Ristimaki A, Haglund C. COX-2 is associated with proliferation and apoptosis markers and serves as an independent prognostic factor in gastric cancer. Tumour Biol 2010; 31: 1-7. ArticlePubMedPDF
- 23. Zha Y, Cun Y, Zhang Q, Li Y, Tan J. Prognostic value of expression of Kit67, p53, TopoIIa and GSTP1 for curatively resected advanced gastric cancer patients receiving adjuvant paclitaxel plus capecitabine chemotherapy. Hepatogastroenterology 2012; 59: 1327-32. PubMed
- 24. Yildirim M, Kaya V, Demirpence O, Gunduz S, Bozcuk H. Prognostic significance of p53 in gastric cancer: a meta- analysis. Asian Pac J Cancer Prev 2015; 16: 327-32. ArticlePubMedPDF
- 25. Kobel M, Reuss A, du Bois A, et al. The biological and clinical value of p53 expression in pelvic high-grade serous carcinomas. J Pathol 2010; 222: 191-8. ArticlePubMed
- 26. Shin YJ, Kim Y, Wen X, et al. Prognostic implications and interaction of L1 methylation and p53 expression statuses in advanced gastric cancer. Clin Epigenetics 2019; 11: 77.ArticlePubMedPMCPDF
- 27. Nenutil R, Smardova J, Pavlova S, et al. Discriminating functional and non-functional p53 in human tumours by p53 and MDM2 immunohistochemistry. J Pathol 2005; 207: 251-9. ArticlePubMed
- 28. Watanabe G, Ishida T, Furuta A, et al. Combined immunohistochemistry of PLK1, p21, and p53 for predicting TP53 status: an independent prognostic factor of breast cancer. Am J Surg Pathol 2015; 39: 1026-34. PubMed
- 29. Tahara T, Shibata T, Okamoto Y, et al. Mutation spectrum of TP53 gene predicts clinicopathological features and survival of gastric cancer. Oncotarget 2016; 7: 42252-60. ArticlePubMedPMC
REFERENCES
Figure & Data
References
Citations

- The future is now: advancing p53 immunohistochemistry in Barrett's oesophagus and its implication for the everyday pathologist
Yevgen Chornenkyy, Monika Vyas, Vikram Deshpande
Histopathology.2026; 88(2): 380. CrossRef - Advancement in preclinical development of cancer treatment agents through modulation of Rac1: From EHop-016 to natural products
Yingyi Liu, Sze-Nga Wong, Aiping Lyu, Joshua Ka-Shun Ko
Biochimica et Biophysica Acta (BBA) - Reviews on Cancer.2026; 1881(1): 189522. CrossRef - Tumor-Associated Macrophage Infiltration and PD-L1 Expression in Gastric Cancer According to a Modified TCGA-Based Classification
Boram Song, Dong-Hoe Koo, Eo Jin Kim, In-Gu Do, Jinah Chu, Kyungeun Kim, Hyebin Lee, Min-Jung Kwon, Jung Ho Park, Byung Ho Son, Chang Hak Yoo, Seoung Wan Chae
Journal of Gastric Cancer.2026; 26(2): 247. CrossRef - Tumor microenvironment dynamics in gastric cancer pathogenesis and therapeutic resistance
Zhenhua Lu, Qinnan Zhang, Jing Han, Jiafu Ji, Xiaofang Xing
Molecular Cancer.2026;[Epub] CrossRef - Captivating Synergistic, Dose-Dependent Anticancer Effects of Tumor-Regulation Modulators Chloroquine and Ivermectin Completely Abolished by an Opposing Modulator, Deoxycholic Acid, in Hamster Fibrosarcoma: In Vivo, In Vitro, and Literature Review
Kosta J. Popović, Dušica J. Popović, Dejan Miljković, Jovan K. Popović, Mihalj Poša, Jovana Drljača Lero, Zana Dolićanin
Pharmaceuticals.2026; 19(3): 407. CrossRef - Comparative study of clinical features, pathology, and immunophenotype between HIV-related cutaneous and visceral Kaposi’s sarcoma
Jingjing Xu, Jing Zhou, Xiaoli Huang, Minmin Wu, Wenjuan Guo, Keyu Liu, Yuexiang Yang, Guangling Yang, Shu Song
BMC Cancer.2026;[Epub] CrossRef -
TP53 mutation landscape and patient survival in oral squamous cell carcinoma
Mebae Ichikawa, Yusuke Kondo, Joaquim Carreras, Masashi Sasaki, Shunsuke Nagase, Yasutaka Hoshimoto, Masashi Tamura, Masahiro Uchibori, Takayuki Aoki, Naoya Nakamura, Yohei Masugi, Yoshihide Ota
Japanese Journal of Clinical Oncology.2026;[Epub] CrossRef - Linking p53 immunostaining to TP53 mutation status in patients with non-small cell lung cancer
Taeyeong Kim, Suyeon Kim, Sangjin Lee, Soohyun Hwang, Joungho Han, Hoyeon Jeong, Yoon-La Choi
Pathology.2025; 57(7): 881. CrossRef - Correlation of TP53 Genetic Alterations with p53 Immunohistochemical Expression and Their Prognostic Significance in DLBCL
Chen Chen, Zijuan Hu, Min Ren, Longlong Bao, Ran Wei, Tian Tian, Xiaoli Zhu, Qianming Bai, Baohua Yu, Xiaoqiu Li, Xiaoyan Zhou
Current Oncology.2025; 32(9): 488. CrossRef - Immunophenotypic Panel for Comprehensive Characterization of Aggressive Thyroid Carcinomas
Mihail Ceausu, Mihai Alin Publik, Dana Terzea, Carmen Adina Cristea, Dumitru Ioachim, Dana Manda, Sorina Schipor
Cells.2025; 14(19): 1554. CrossRef - Multiple approaches revealed MGc80‐3 as a somatic hybrid with HeLa cells rather than a gastric cancer cell line
Fang Cao, Hao Sun, Zhenli Yang, Yanhua Bai, Xiao Hu, Yuhong Hou, Xiaocui Bian, Yuqin Liu
International Journal of Cancer.2024; 154(1): 155. CrossRef - In Response to p53 Immunohistochemical Staining and TP53 Gene Mutations in Endometrial Cancer: Does Null Pattern Correlate With Prognosis?
Ikuko Sakamoto, Keiko Kagami, Takahiro Nozaki, Yosuke Hirotsu, Kenji Amemiya, Toshio Oyama, Masao Omata
American Journal of Surgical Pathology.2024; 48(3): 374. CrossRef - CHEK2 germline variants identified in familial nonmedullary thyroid cancer lead to impaired protein structure and function
Carolina Pires, Inês J. Marques, Mariana Valério, Ana Saramago, Paulo E. Santo, Sandra Santos, Margarida Silva, Margarida M. Moura, João Matos, Teresa Pereira, Rafael Cabrera, Diana Lousa, Valeriano Leite, Tiago M. Bandeiras, João B. Vicente, Branca M. Ca
Journal of Biological Chemistry.2024; 300(3): 105767. CrossRef - The spectrum of TP53 mutations in Rwandan patients with gastric cancer
Augustin Nzitakera, Jean Bosco Surwumwe, Ella Larissa Ndoricyimpaye, Schifra Uwamungu, Delphine Uwamariya, Felix Manirakiza, Marie Claire Ndayisaba, Gervais Ntakirutimana, Benoit Seminega, Vincent Dusabejambo, Eric Rutaganda, Placide Kamali, François Ngab
Genes and Environment.2024;[Epub] CrossRef - Gastric cancer molecular classification based on immunohistochemistry and in‐situ hybridisation and mortality
Maarit Eskuri, Eva‐Maria Birkman, Joonas H Kauppila
Histopathology.2024; 85(2): 327. CrossRef - Redefining aberrant P53 expression of gastric cancer and its distinct clinical significance among molecular-histologic subtypes
Shih-Chiang Huang, Ian Yi-Feng Chang, Tse-Ching Chen, Hsiao-Ching Lin, Chun-Yi Tsai, Jun-Te Hsu, Chun-Nan Yeh, Shih-Cheng Chang, Ta-Sen Yeh
Asian Journal of Surgery.2024; 47(11): 4699. CrossRef - Assessment of TP53 and CDKN2A status as predictive markers of malignant transformation of sinonasal inverted papilloma
Soohyeon Kwon, Jeong-Whun Kim, Eun Sun Kim, Jin Ho Paik, Jin-Haeng Chung, Sung-Woo Cho, Tae-Bin Won, Chae-Seo Rhee, Jee Hye Wee, Hyojin Kim
Scientific Reports.2024;[Epub] CrossRef - Implementing an integrated molecular classification for gastric cancer from endoscopic biopsies using on-slide tests
Simona Costache, Adelina Baltan , Sofia Diaz McLinn , Mattia Pegoraro , Rebecca de Havilland , Matthew Porter , Ana Lerga , Teresa Thomas , Alina Elena Chefani
Romanian Journal of Morphology and Embryology.2024; 65(2): 257. CrossRef - Application of NGS molecular classification in the diagnosis of endometrial carcinoma: A supplement to traditional pathological diagnosis
Qunxian Rao, Jianwei Liao, Yangyang Li, Xin Zhang, Guocai Xu, Changbin Zhu, Shengya Tian, Qiuhong Chen, Hui Zhou, Bingzhong Zhang
Cancer Medicine.2023; 12(5): 5409. CrossRef - Predictive value of p53 and AXL immunostaining for the efficacy of immune checkpoint inhibitor-based therapy after osimertinib treatment in patients with epidermal growth factor-mutant non-small cell lung cancer
Kenji Morimoto, Tadaaki Yamada, Ryo Sawada, Koichi Azuma, Yasuhiro Goto, Taishi Harada, Shinsuke Shiotsu, Nobuyo Tamiya, Yusuke Chihara, Takayuki Takeda, Osamu Hiranuma, Isao Hasegawa, Satomi Tanaka, Akihiro Yoshimura, Masahiro Iwasaku, Shinsaku Tokuda, Y
Cancer Immunology, Immunotherapy.2023; 72(6): 1699. CrossRef - Validation of p53 Immunohistochemistry (PAb240 Clone) in Canine Tumors with Next-Generation Sequencing (NGS) Analysis
Barbara Brunetti, Dario de Biase, Giulia Dellapina, Luisa Vera Muscatello, Francesco Ingravalle, Giorgia Tura, Barbara Bacci
Animals.2023; 13(5): 899. CrossRef - Mesonephric‐like adenocarcinoma of the female genital tract: novel observations and detailed molecular characterisation of mixed tumours and mesonephric‐like carcinosarcomas
Jelena Mirkovic, Ekaterina Olkhov‐Mitsel, Yutaka Amemiya, Maysa Al‐Hussaini, Sharon Nofech‐Mozes, Bojana Djordjevic, Rachel Kupets, Arun Seth, W Glenn McCluggage
Histopathology.2023; 82(7): 978. CrossRef - Clinicopathologic characterization of cervical metastasis from an unknown primary tumor: a multicenter study in Korea
Miseon Lee, Uiree Jo, Joon Seon Song, Youn Soo Lee, Chang Gok Woo, Dong-Hoon Kim, Jung Yeon Kim, Sun Och Yoon, Kyung-Ja Cho
Journal of Pathology and Translational Medicine.2023; 57(3): 166. CrossRef - P53 in Penile Squamous Cell Carcinoma: A Pattern-Based Immunohistochemical Framework with Molecular Correlation
Isabel Trias, Adela Saco, Lorena Marimon, Ricardo López del Campo, Carolina Manzotti, Oriol Ordi, Marta del Pino, Francisco M. Pérez, Naiara Vega, Silvia Alós, Antonio Martínez, Leonardo Rodriguez-Carunchio, Oscar Reig, Pedro Jares, Cristina Teixido, Tare
Cancers.2023; 15(10): 2719. CrossRef - p53/TP53 Status Assessment in Gastroesophageal Adenocarcinoma
Elisa Boldrin, Maria Assunta Piano, Francesco Bernaudo, Rita Alfieri, Maria Raffaella Biasin, Isabella Monia Montagner, Alice Volpato, Genny Mattara, Francesco Lamacchia, Giovanna Magni, Antonio Rosato, Antonio Scapinello, Pierluigi Pilati, Matteo Curtare
Cancers.2023; 15(10): 2783. CrossRef - Genomic profiling of dedifferentiated endometrial carcinomas arising in the background of high‐grade carcinoma: a targeted next‐generation sequencing study
Ekaterina Olkhov‐Mitsel, Aurelia Busca, Carlos Parra‐Herran, Yutaka Amemiya, Sharon Nofech‐Mozes, Bojana Djordjevic, Marisa R Nucci, Arun Seth, Jelena Mirkovic
Histopathology.2023; 83(3): 366. CrossRef -
Clinicopathologic Features and Prognostic Significance of Immunohistochemistry and In Situ Hybridization Based Molecular Classification in Gastric Carcinoma
Gizem Issin, İlyas Sayar, Fatih Demir, İrem Güvendir Bakkaloğlu, Mehmet Gamsizkan, Zeliha Yildiz, Ismail Yilmaz, Sevilay Akalp Özmen, Diren Vuslat Çağatay, Itır Ebru Zemheri, Murat Demiriz, Armağan Günal
Journal of Environmental Pathology, Toxicology and Oncology.2023; 42(4): 1. CrossRef - Clinicopathologic and Molecular Characterization of Anorectal Neuroendocrine Carcinomas Reveals Human Papillomavirus, p53, and c-Myc as Alternative Mechanisms of Carcinogenesis
Allison J. Cox, William E. Crowe, Qi Yang, Bin Zhang, Zoltán N. Oltvai, Xiaoyan Liao
Modern Pathology.2023; 36(11): 100295. CrossRef - Dedifferentiated Endometrial Carcinoma: A Rare Aggressive Neoplasm-Clinical, Morphological and Immunohistochemical Features
Giovanna Giordano, Elena Ferioli, Debora Guareschi, Alessandro Tafuni
Cancers.2023; 15(21): 5155. CrossRef - Characterization on the oncogenic effect of the missense mutations of p53 via machine learning
Qisheng Pan, Stephanie Portelli, Thanh Binh Nguyen, David B Ascher
Briefings in Bioinformatics.2023;[Epub] CrossRef - Adrenal Nodules Detected at Staging CT in Patients with Resectable Gastric Cancers Have a Low Incidence of Malignancy
Hae Young Kim, Won Chang, Yoon Jin Lee, Ji Hoon Park, Jungheum Cho, Hee Young Na, Hyungwoo Ahn, Sung Il Hwang, Hak Jong Lee, Young Hoon Kim, Kyoung Ho Lee
Radiology.2022; 302(1): 129. CrossRef - Intestinal-type gastric dysplasia in Helicobacter pylori-naïve patients
Kotaro Shibagaki, Ayako Itawaki, Yoichi Miyaoka, Kenichi Kishimoto, Yusuke Takahashi, Satoshi Kotani, Tsuyoshi Mishiro, Naoki Oshima, Kousaku Kawashima, Norihisa Ishimura, Hideyuki Onuma, Makoto Nagasaki, Mamiko Nagase, Asuka Araki, Kyuichi Kadota, Ryoji
Virchows Archiv.2022; 480(4): 783. CrossRef - Dedifferentiation-like tubular and solid carcinoma of the stomach shows phenotypic divergence and association with deficient SWI/SNF complex
Shih-Chiang Huang, Kuang-Hua Chen, Kwai-Fong Ng, I-Chieh Lin, Yi-Chun Chao, Ta-Sen Yeh, Huei-Chieh Chuang, Tse-Ching Chen
Virchows Archiv.2022; 480(4): 771. CrossRef - Distinct molecular phenotype and the potential prognostic value of immune prognostic index and tumor infiltrating lymphocytes in hepatoid adenocarcinoma of stomach
Muxing Kang, Xiaojing Ma, Jifei Shi, Guofeng Chen, Xiaoli Jin, Jun Wang, Lele Lin, Zhiwei Wu, Kaibo Chen, Jinghong Xu, Pintong Huang, Jian Chen
Translational Oncology.2022; 19: 101380. CrossRef - Evaluation of Tumor DNA Sequencing Results in Patients with Gastric and Gastroesophageal Junction Adenocarcinoma Stratified by TP53 Mutation Status
Anthony C Wood, Yonghong Zhang, Qianxing Mo, Ling Cen, Jacques Fontaine, Sarah E Hoffe, Jessica Frakes, Sean P Dineen, Jose M Pimiento, Christine M Walko, Rutika Mehta
The Oncologist.2022; 27(4): 307. CrossRef - Comprehensive Clinical Analysis of Gallbladder Neuroendocrine Neoplasms: A Large-Volume Multicenter Study During One Decade
Yangyang Wang, Bingfeng Huang, Qihan Fu, Jianing Wang, Mao Ye, Manyi Hu, Kai Qu, Kai Liu, Xiao Hu, Shumei Wei, Ke Sun, Wenbo Xiao, Bo Zhang, Haijun Li, Jingsong Li, Qi Zhang, Tingbo Liang
Annals of Surgical Oncology.2022; 29(12): 7619. CrossRef - Expression of SASP, DNA Damage Response, and Cell Proliferation Factors in Early Gastric Neoplastic Lesions: Correlations and Clinical Significance
Li Liang, Yijie Chai, Fei Chai, Haijing Liu, Ningning Ma, Hong Zhang, Shuang Zhang, Lin Nong, Ting Li, Bo Zhang
Pathology and Oncology Research.2022;[Epub] CrossRef - Systems biology and OMIC data integration to understand gastrointestinal cancers
Iasmin Moreira Costa Bispo, Henry Paul Granger, Palloma Porto Almeida, Patricia Belini Nishiyama, Leandro Martins de Freitas
World Journal of Clinical Oncology.2022; 13(10): 762. CrossRef - MicroRNA-552 expression in colorectal cancer and its clinicopathological significance
Joon Im, Soo Kyung Nam, Hye Seung Lee
Journal of Pathology and Translational Medicine.2021; 55(2): 125. CrossRef - Different effects of p53 protein overexpression on the survival of gastric cancer patients according to Lauren histologic classification: a retrospective study
Ki Wook Kim, Nayoung Kim, Yonghoon Choi, Won Seok Kim, Hyuk Yoon, Cheol Min Shin, Young Soo Park, Dong Ho Lee, Young Suk Park, Sang-Hoon Ahn, Do Joong Park, Hyung-Ho Kim, Hye Seung Lee, Ji-Won Kim, Jin Won Kim, Keun-Wook Lee, Won Chang, Ji Hoon Park, Yoon
Gastric Cancer.2021; 24(4): 844. CrossRef - The association between the expression of nuclear Yes-associated protein 1 (YAP1) and p53 protein expression profile in breast cancer patients
Yoon Jin Cha, Dooreh Kim, Soong June Bae, Sung Gwe Ahn, Joon Jeong, Min Kyung Cho, Pill Sun Paik, Tae-Kyung Yoo, Woo-Chan Park, Chang Ik Yoon, Elda Tagliabue
PLOS ONE.2021; 16(5): e0250986. CrossRef
 PubReader
PubReader ePub Link
ePub Link-
 Cite this Article
Cite this Article
- Cite this Article
-
- Close
- Download Citation
- Close
- Figure

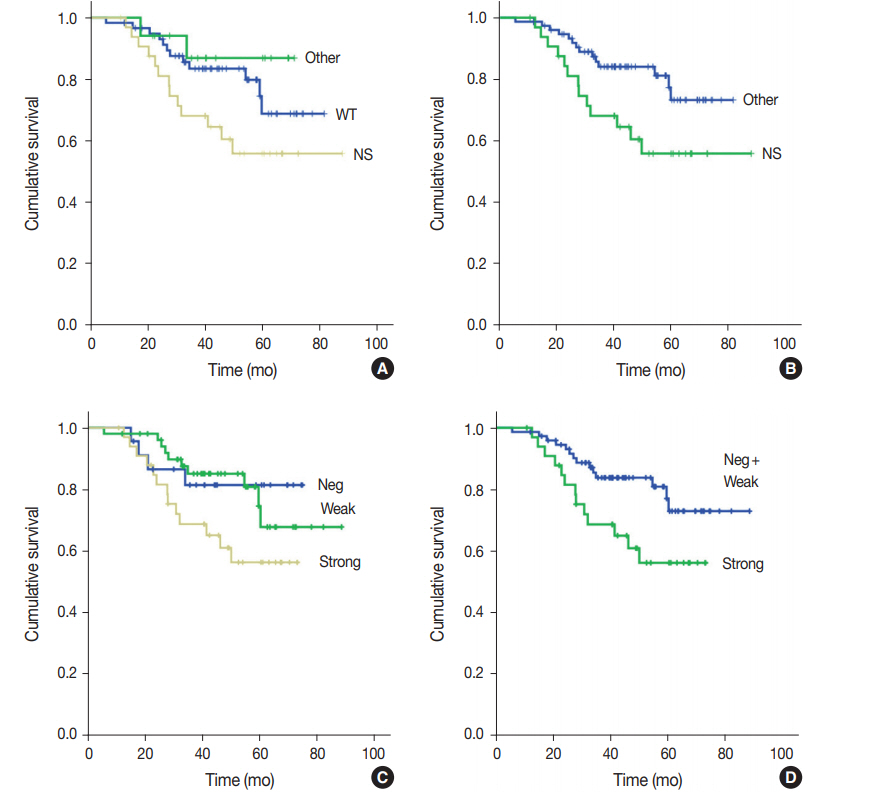
Fig. 1.
Fig. 2.
| TP53 mutation | p53 expression by IHC |
Total | p-value | ||
|---|---|---|---|---|---|
| Strong | Negative | Weak | |||
| Mutation status | < .001 | ||||
| Wild-type | 1 (2.9) | 12 (44.4) | 55 (93.2) | 68 (56.7) | |
| Mutation present | 33 (97.1) | 15 (55.6) | 4 (6.8) | 52 (43.3) | |
| Variant summary | < .001 | ||||
| Wild-type | 1 (2.9) | 12 (44.4) | 55 (93.2) | 68 (56.7) | |
| Missense | 30 (88.2) | 0 | 3 (5.1) | 33 (27.5) | |
| Other | 3 (8.9) | 15 (55.6) | 1 (1.7) | 19 (15.8) | |
| Stop-gained | 2 (5.9) | 3 (11.1) | 1 (1.7) | 6 (5.0) | |
| Splice region | 0 | 5 (18.5) | 0 | 5 (4.2) | |
| Frameshift | 0 | 7 (25.9) | 0 | 7 (5.8) | |
| In-frame deletion | 1 (2.9) | 0 | 0 | 1 (0.8) | |
| Clinical significance |
< .001 | ||||
| Wild-type | 1 (2.9) | 12 (44.4) | 55 (93.2) | 68 (56.7) | |
| Pathogenic or likely pathogenic | 22 (64.7) | 13 (48.1) | 2 (3.4) | 37 (30.1) | |
| Uncertain significance | 5 (14.7) | 2 (7.4) | 1 (1.7) | 8 (6.7) | |
| Conflicting interpretation | 6 (17.6) | 0 | 1 (1.7) | 7 (5.8) | |
| Total | 34 | 27 | 59 | 120 | |
| Case No. | Effect | Nucleic acid alteration | Amino acid alteration | Clinical significance |
|---|---|---|---|---|
| 1 | Missense_variant | c.422G > A | p.Cys141Tyr | Pathogenic or likely pathogenic |
| 2 | Missense_variant | c.422G > T | p.Cys141Phe | Pathogenic or likely pathogenic |
| 3 | Missense_variant | c.455C > T | p.Pro152Leu | Pathogenic or likely pathogenic |
| 4 | Missense_variant | c.524G > A | p.Arg175His | Pathogenic or likely pathogenic |
| 5 | Missense_variant | c.535C > G | p.His179Asp | Pathogenic or likely pathogenic |
| 6 | Missense_variant | c.542G > A | p.Arg181His | Pathogenic or likely pathogenic |
| 7 | Missense_variant | c.659A > G | p.Tyr220Cys | Pathogenic or likely pathogenic |
| 8 | Missense_variant | c.659A > G | p.Tyr220Cys | Pathogenic or likely pathogenic |
| 9 | Missense_variant | c.701A > G | p.Tyr234Cys | Pathogenic or likely pathogenic |
| 10 | Missense_variant | c.725G > A | p.Cys242Tyr | Pathogenic or likely pathogenic |
| 11 | Missense_variant | c.734G > A | p.Gly245Asp | Pathogenic or likely pathogenic |
| 12 | Missense_variant | c.742C > T | p.Arg248Trp | Pathogenic or likely pathogenic |
| 13 | Missense_variant | c.742C > T | p.Arg248Trp | Pathogenic or likely pathogenic |
| 14 | Missense_variant | c.743G > A | p.Arg248Gln | Pathogenic or likely pathogenic |
| 15 | Missense_variant | c.772G > A | p.Glu258Lys | Pathogenic or likely pathogenic |
| 16 | Missense_variant | c.817C > T | p.Arg273Cys | Pathogenic or likely pathogenic |
| 17 | Missense_variant | c.817C > T | p.Arg273Cys | Pathogenic or likely pathogenic |
| 18 | Missense_variant | c.818G > A | p.Arg273His | Pathogenic or likely pathogenic |
| 19 | Missense_variant | c.818G > A | p.Arg273His | Pathogenic or likely pathogenic |
| 20 | Missense_variant | c.818G > A | p.Arg273His | Pathogenic or likely pathogenic |
| 21 | Missense_variant | c.380C > T | p.Ser127Phe | Conflicting interpretations of pathogenicity |
| 22 | Missense_variant | c.473G > C | p.Arg158Pro | Conflicting interpretations of pathogenicity |
| 23 | Missense_variant | c.481G > A | p.Ala161Thr | Conflicting interpretations of pathogenicity |
| 24 | Missense_variant | c.613T > C | p.Tyr205His | Conflicting interpretations of pathogenicity |
| 25 | Missense_variant | c.796G > A | p.Gly266Arg | Conflicting interpretations of pathogenicity |
| 26 | Missense_variant | c.796G > A | p.Gly266Arg | Conflicting interpretations of pathogenicity |
| 27 | Missense_variant | c.1015G > A | p.Glu339Lys | Conflicting interpretations of pathogenicity |
| 28 | Missense_variant | c.329G > A | p.Arg110His | Uncertain significance |
| 29 | Missense_variant | c.380C > A | p.Ser127Tyr | Uncertain significance |
| 30 | Missense_variant | c.476C > T | p.Ala159Val | Uncertain significance |
| 31 | Missense_variant | c.797G > T | p.Gly266Val | Uncertain significance |
| 32 | Missense_variant | c.400T > G | p.Phe134Val | Uncertain significance |
| 33 | Missense_variant | c.470T > G | p.Val157Gly | Uncertain significance |
| 34 | Frameshift_variant | c.331_332insAG | p.Leu111fs | Pathogenic or likely pathogenic |
| 35 | Frameshift_variant | c.381_391delCCCTGCCCTCA | p.Pro128fs | Pathogenic or likely pathogenic |
| 36 | Frameshift_variant | c.635_669delTTCGACATAGTGTGGTG GTGCCCTATGAGCCGCCT | p.Phe212fs | Pathogenic or likely pathogenic |
| 37 | Frameshift_variant | c.660_661delTG | p.Tyr220fs | Pathogenic or likely pathogenic |
| 38 | Frameshift_variant | c.747delG | p.Arg249fs | Pathogenic or likely pathogenic |
| 39 | Frameshift_variant | c.1169delC | p.Pro390fs | Pathogenic or likely pathogenic |
| 40 | Frameshift_variant | c.778_779delTC | p.Ser260fs | Uncertain significance |
| 41 | Conservative_inframe_deletion | c.529_546delCCCCACCATGAGCGCTGC | p.Pro177_Cys182del | Pathogenic or likely pathogenic |
| 42 | Stop_gained | c.159G > A | p.Trp53* | Pathogenic or likely pathogenic |
| 43 | Stop_gained | c.437G > A | p.Trp146* | Pathogenic or likely pathogenic |
| 44 | Stop_gained | c.586C > T | p.Arg196* | Pathogenic or likely pathogenic |
| 45 | Stop_gained | c.637C > T | p.Arg213* | Pathogenic or likely pathogenic |
| 46 | Stop_gained | c.1024C > T | p.Arg342* | Pathogenic or likely pathogenic |
| 47 | Stop_gained | c.1024C > T | p.Arg342* | Pathogenic or likely pathogenic |
| 48 | Splice_region_variant&synonymous_variant | c.375G > A | p.Thr125Thr | Pathogenic or likely pathogenic |
| 49 | Splice_region_variant&synonymous_variant | c.375G > A | p.Thr125Thr | Pathogenic or likely pathogenic |
| 50 | Splice_region_variant&synonymous_variant | c.375G > C | p.Thr125Thr | Pathogenic or likely pathogenic |
| 51 | Splice_acceptor_variant&intron_variant | c.920 - 1G > A | Pathogenic or likely pathogenic | |
| 52 | Splice_donor_variant&intron_variant | c.96 + 1G > A | Uncertain significance (no report) |
| TP53 mutation | Sensitivity (%) | Specificity (%) | Accuracy (%) |
|---|---|---|---|
| Nonsynonymous mutation by p53 strong expression | 90.9 | 95.4 | 94.2 |
| Other type mutation by negative expression of p53 | 79.0 | 88.1 | 86.7 |
| Wild-type by weak expression of p53 | 80.9 | 92.3 | 85.8 |
| Characteristic | Total | TP53 mutation |
p53 expression |
||||||
|---|---|---|---|---|---|---|---|---|---|
| NS | Other | Wild | p-value | NS | Other | Wild | p-value | ||
| No. | 120 | 33 | 19 | 68 | 34 | 27 | 59 | ||
| Age (yr) | 0.248 | 0.470 | |||||||
| < 65 | 69 (57.5) | 15 (45.5) | 11 (57.9) | 43 (63.2) | 17 (50.0) | 15 (55.6) | 37 (62.7) | ||
| ≥ 65 | 51 (42.5) | 18 (54.5) | 8 (42.1) | 25 (36.8) | 17 (50.0) | 12 (44.4) | 22 (37.3) | ||
| Sex | 0.117 | 0.285 | |||||||
| Male | 85 (70.8) | 27 (81.8) | 15 (78.9) | 43 (63.2) | 27 (79.4) | 20 (74.1) | 38 (64.4) | ||
| Female | 35 (29.2) | 6 (18.2) | 4 (21.1) | 25 (36.8) | 7 (20.6) | 7 (25.9) | 21 (35.6) | ||
| Location of tumor center | 0.940 | 0.856 | |||||||
| Lower third | 53 (44.2) | 15 (45.5) | 10 (52.6) | 28 (41.2) | 16 (47.1) | 11 (40.7) | 26 (44.1) | ||
| Middle third | 33 (27.5) | 9 (27.3) | 4 (21.1) | 20 (29.4) | 7 (20.6) | 8 (29.6) | 18 (30.5) | ||
| Upper third | 34 (28.3) | 9 (27.3) | 5 (26.3) | 20 (29.4) | 11 (32.4) | 8 (29.6) | 15 (25.4) | ||
| TNM at initial diagnosis | 0.004 | 0.029 | |||||||
| II | 38 (31.7) | 5 (15.2) | 6 (31.6) | 27 (39.7) | 7 (20.6) | 10 (37.0) | 21 (35.6) | ||
| III | 71 (59.2) | 28 (84.8) | 11 (57.9) | 32 (47.1) | 27 (79.4) | 13 (48.1) | 31 (52.5) | ||
| IV | 11 (9.2) | 0 | 2 (10.5) | 9 (13.2) | 0 | 4 (14.8) | 7 (11.9) | ||
| WHO classification | 0.733 | 0.596 | |||||||
| Papillary | 4 (3.3) | 1 (3.0) | 1 (5.3) | 2 (2.9) | 1 (2.9) | 2 (7.4) | 1 (1.7) | ||
| Tubular WD/MD | 28 (23.3) | 10 (30.3) | 6 (31.6) | 12 (17.6) | 10 (29.4) | 6 (22.2) | 12 (20.3) | ||
| Tubular PD | 37 (30.8) | 9 (27.3) | 7 (36.8) | 21 (30.9) | 10 (29.4) | 8 (29.6) | 19 (32.2) | ||
| PCC | 36 (30.0) | 8 (24.2) | 3 (15.8) | 25 (36.8) | 7 (20.6) | 7 (25.6) | 22 (37.3) | ||
| Mucinous | 4 (3.3) | 2 (6.1) | 0 | 2 (2.9) | 2 (5.9) | 1 (3.7) | 1 (1.7) | ||
| Others | 11 (9.2) | 3 (9.1) | 2 (10.5) | 6 (8.8) | 4 (11.7) | 3 (11.1) | 4 (6.8) | ||
| Lauren classification | 0.065 | 0.587 | |||||||
| Intestinal | 45 (37.5) | 14 (42.4) | 11 (57.9) | 20 (29.4) | 15 (44.1) | 10 (37.0) | 20 (33.9) | ||
| Non-intestinal | 75 (62.5) | 19 (57.6) | 8 (42.1) | 48 (70.6) | 19 (55.9) | 17 (63.0) | 39 (66.1) | ||
| EBV | 0.215 | 0.036 | |||||||
| Negative | 105 (87.5) | 31 (93.9) | 18 (94.7) | 56 (82.4) | 30 (88.2) | 27 (100) | 48 (81.4) | ||
| Positive | 15 (12.5) | 2 (6.1) | 1 (5.3) | 12 (17.6) | 4 (11.8) | 0 | 11 (18.6) | ||
| MSI | 0.258 | 0.010 | |||||||
| MSS/MSI-L | 112 (93.3) | 32 (97.0) | 19 (100) | 61 (89.7) | 34 (100) | 27 (100) | 51 (86.4) | ||
| MSI-H | 8 (6.7) | 1 (3.0) | 0 | 7 (10.3) | 0 | 0 | 8 (13.6) | ||
Values are presented as number (%). According to the ClinVar and OncoKB databases accessed on March 18, 2020.
IHC, immunohistochemistry. According to the ClinVar and OncoKB databases accessed on March 18, 2020.
Values are presented as number (%). NS, nonsynonymous; Other, other type mutation; wild, wild-type; WHO, World Health Organization; WD, well-differentiated; MD, moderately differentiated; PD, poorly differentiated; PCC, poorly cohesive carcinoma; EBV, Epstein-Barr virus; MSI, microsatellite instability; MSS, microsatellite stable; MSI-L, microsatellite instability-low; MSI-H, microsatellite instability-high.

 E-submission
E-submission