Articles
- Page Path
- HOME > J Pathol Transl Med > Volume 58(5); 2024 > Article
-
Case Study
Intravascular schwannoma as an extremely unusual cause of vein obstruction: a case report -
Luis Miguel Chinchilla-Tábora1,2,3
 , Beatriz Segovia Blázquez4
, Beatriz Segovia Blázquez4 , José María Sayagués1,2,3
, José María Sayagués1,2,3 , Marta Rodríguez González1,2,3
, Marta Rodríguez González1,2,3 , Joaquín González-Rivero1,3
, Joaquín González-Rivero1,3 , José Antonio Muñoz León1,3
, José Antonio Muñoz León1,3 , Andrea Beatriz Jiménez Pérez1,3
, Andrea Beatriz Jiménez Pérez1,3 , Idalia González Morais1
, Idalia González Morais1 , Diego Bueno-Sacristán1
, Diego Bueno-Sacristán1 , María Dolores Ludeña1,2,3
, María Dolores Ludeña1,2,3
-
Journal of Pathology and Translational Medicine 2024;58(5):249-254.
DOI: https://doi.org/10.4132/jptm.2024.05.15
Published online: July 3, 2024
1Pathology Department, University Hospital of Salamanca, Salamanca, Spain
2University of Salamanca, Salamanca, Spain
3Institute for Biomedical Research of Salamanca (IBSAL), Salamanca, Spain
4Pathology Department, Nuestra Señora de Sonsoles Hospital, Ávila, Spain
- Corresponding Author: Luis Miguel Chinchilla-Tábora, MD, PhD, Servicio de Anatomía Patológica, planta cero, bloque G, Complejo Asistencial Universitario de Salamanca, Paseo de la Transición Española S/N, CP: 37007, Salamanca, Salamanca, Spain Tel: +34-923291100 (ext. 56738), Fax: +34-923291131, E-mail: lmct@usal.es
© The Korean Society of Pathologists/The Korean Society for Cytopathology
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/4.0) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
- 4,937 Views
- 289 Download
Abstract
- The blood vessel lumen is an extremely rare location for a benign peripheral nerve sheath tumor like schwannoma. Less than 10 cases have been previously reported. In this report, we present a case of a 68-year-old woman who had a soft tissue nodule at the posterior calf of her left leg during a physical examination. Pathological examination was performed after complete surgical excision. The patient underwent follow-up for 12 months after surgery without evidence of recurrence or any other complication. This is the first case of intravascular schwannoma reported as a cause of vein obstruction. Microscopically, the tumor was composed of Schwann spindle cells that were immunoreactive for S100 protein and SOX10. This tumor was surrounded by a well-defined vascular smooth muscle wall. Prospective series are required to improve the knowledge on the underlying mechanisms of intravascular schwannoma development.
- Case presentation
- We present the case of a 68-year-old woman who had a three-year history of non-radiating pain on the medial side of her left leg, 5 cm distal to the popliteal fossa. No CNS disorders were identified at physical examination, and no other related symptoms were observed.
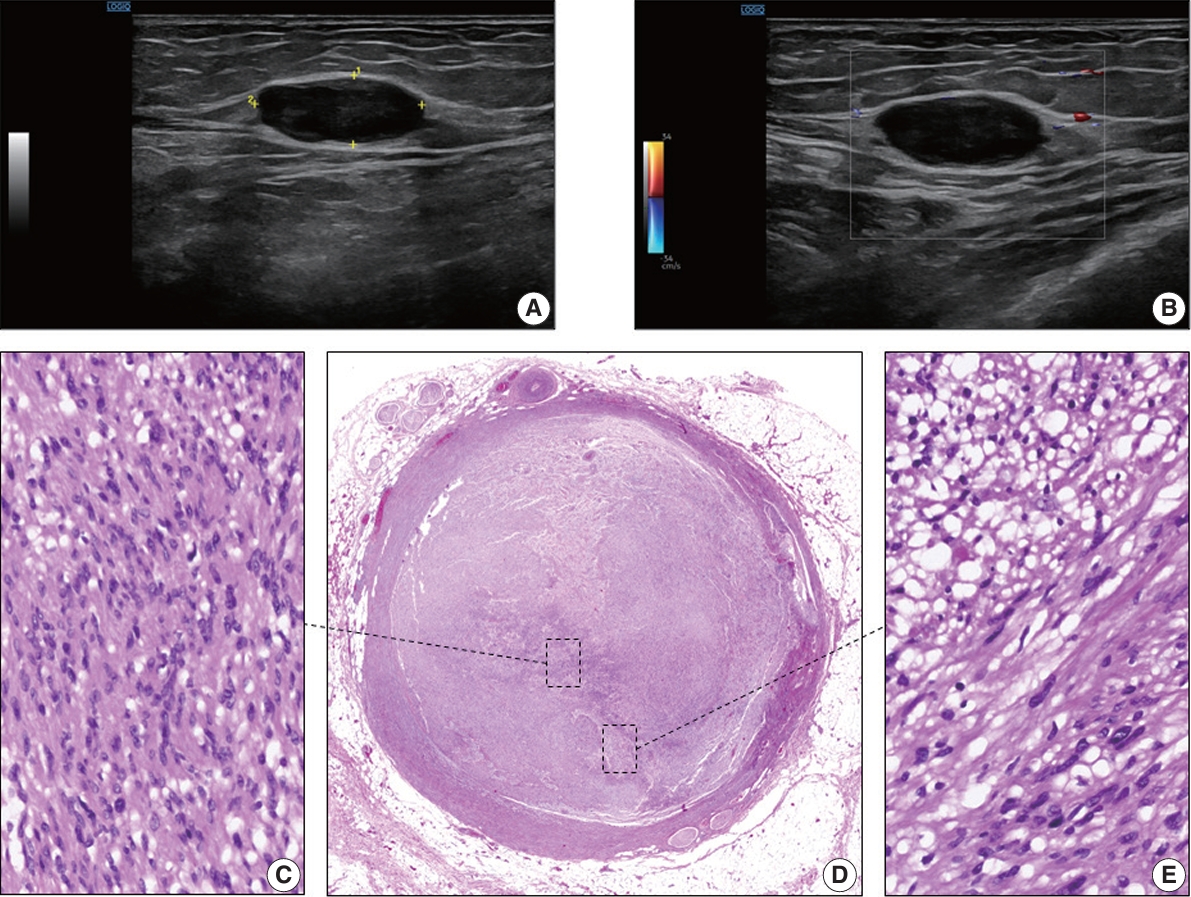
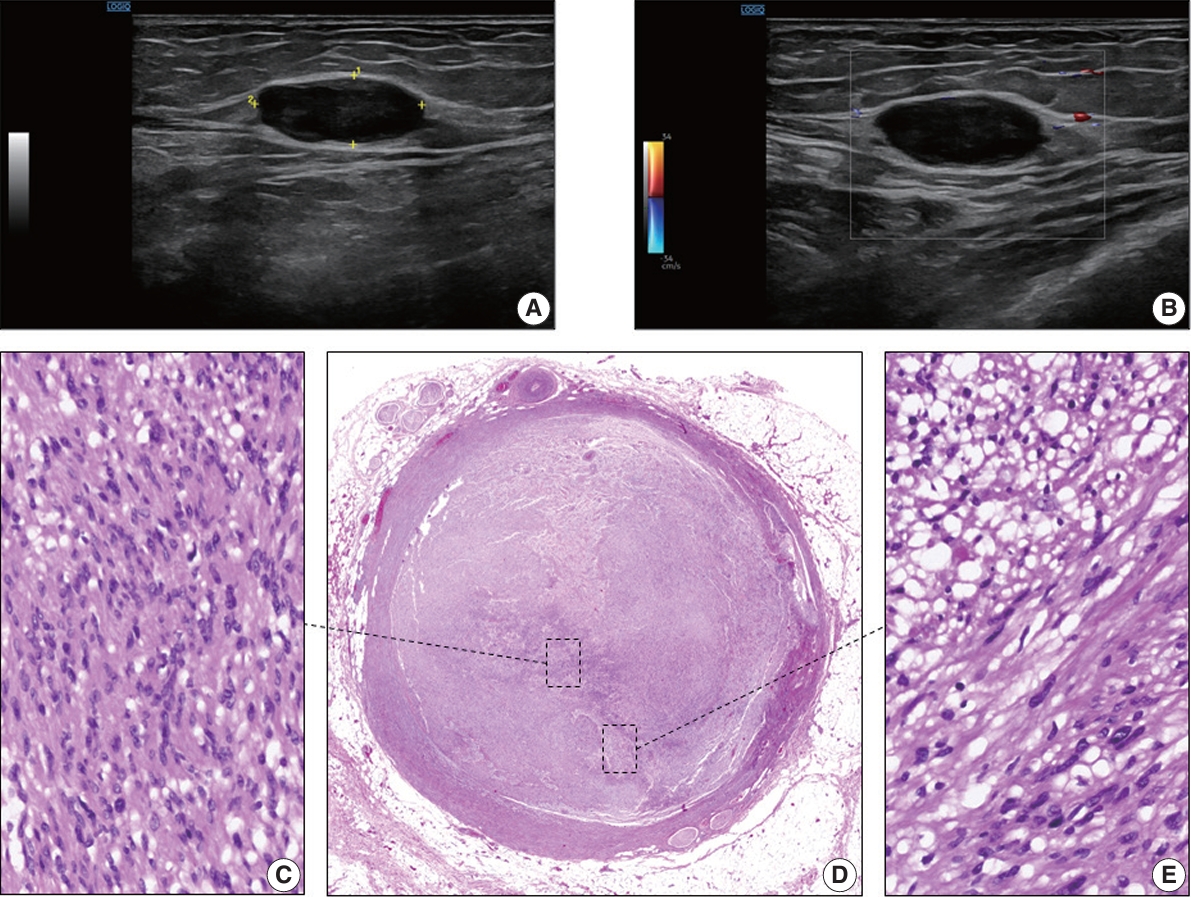
- A soft tissue Doppler ultrasound was performed and showed a spindle-shaped to ovoid lesion, the largest diameter of which was 2.3 cm. The mass was in the deep subcutaneous tissue, near the muscular fascia (Fig. 1A, B) and did not display marked internal blood flow (Fig. 1B). The radiological characteristics of the lesion on ultrasonographic examination suggested the presence of a soft tissue tumor of probable neural origin. The lesion was completely surgically removed.
- Pathologic characteristics
- The macroscopic examination of the resected area revealed the presence of a soft tissue nodule, largest diameter 2.3 cm, with a vaguely spindle morphology and smooth external surface. The cut surface of the mass was grayish brown and had the consistency of rubber.
- Microscopically, the tumor was nodular and composed of spindle cells. This tumor occupied the entire lumen of a venous vessel that had a thickened smooth muscle wall with focal myxoid degeneration and no elastic fibers (Fig. 1D). The lesion was composed of at least two intermingled cellular areas, at least one of which shared features with the normal peripheral nerve sheath cells. Within these areas, some of the cells had elongated, fusiform, or oval hyperchromatic nuclei with sharp edges and scarce cytoplasm (Antoni A zones) (Fig. 1C); these were arranged to form parallel palisades with interposed filaments (Verocay bodies).
- In addition, the tumor had areas composed of less cohesive cells with small, rounded nuclei and large pale cytoplasm in a loose myxoid stroma (Antoni B zones) (Fig. 1E). The lesion was not necrotic or hemorrhagic. Neither mitosis nor atypia was present. The venous vessel within which the tumor expanded was surrounded by tiny mature vessels, the vasa vasorum, and nerves, the nervi vasorum (Fig. 1D).
- Immunohistochemistry
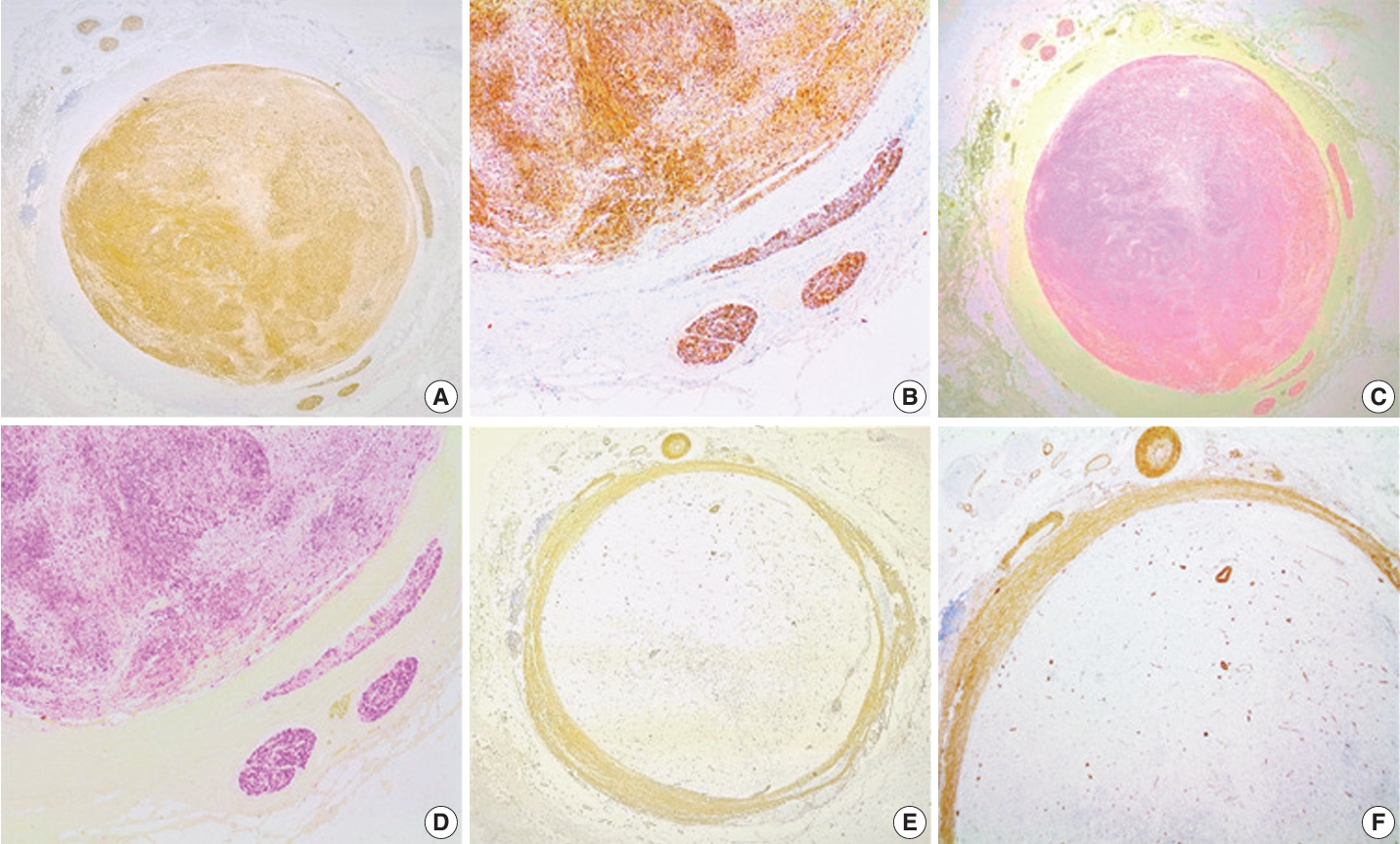
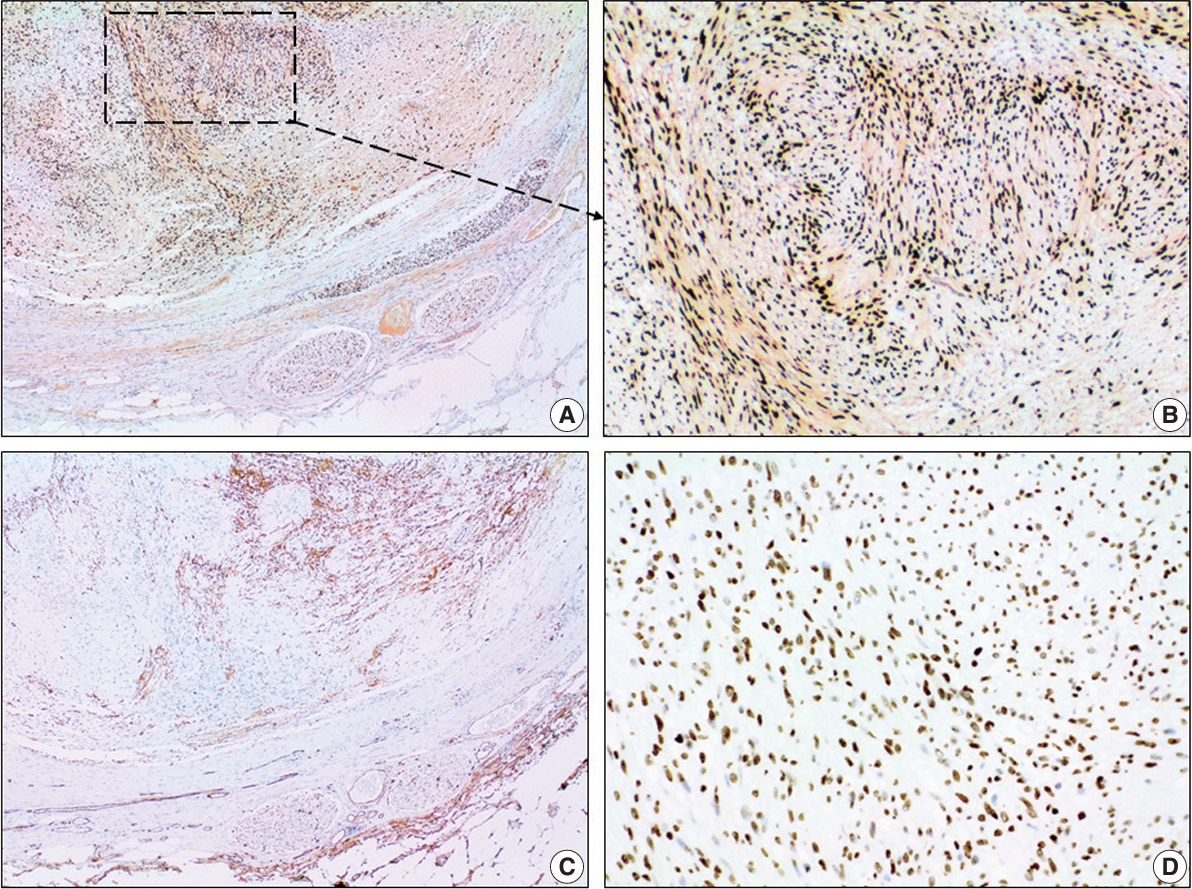
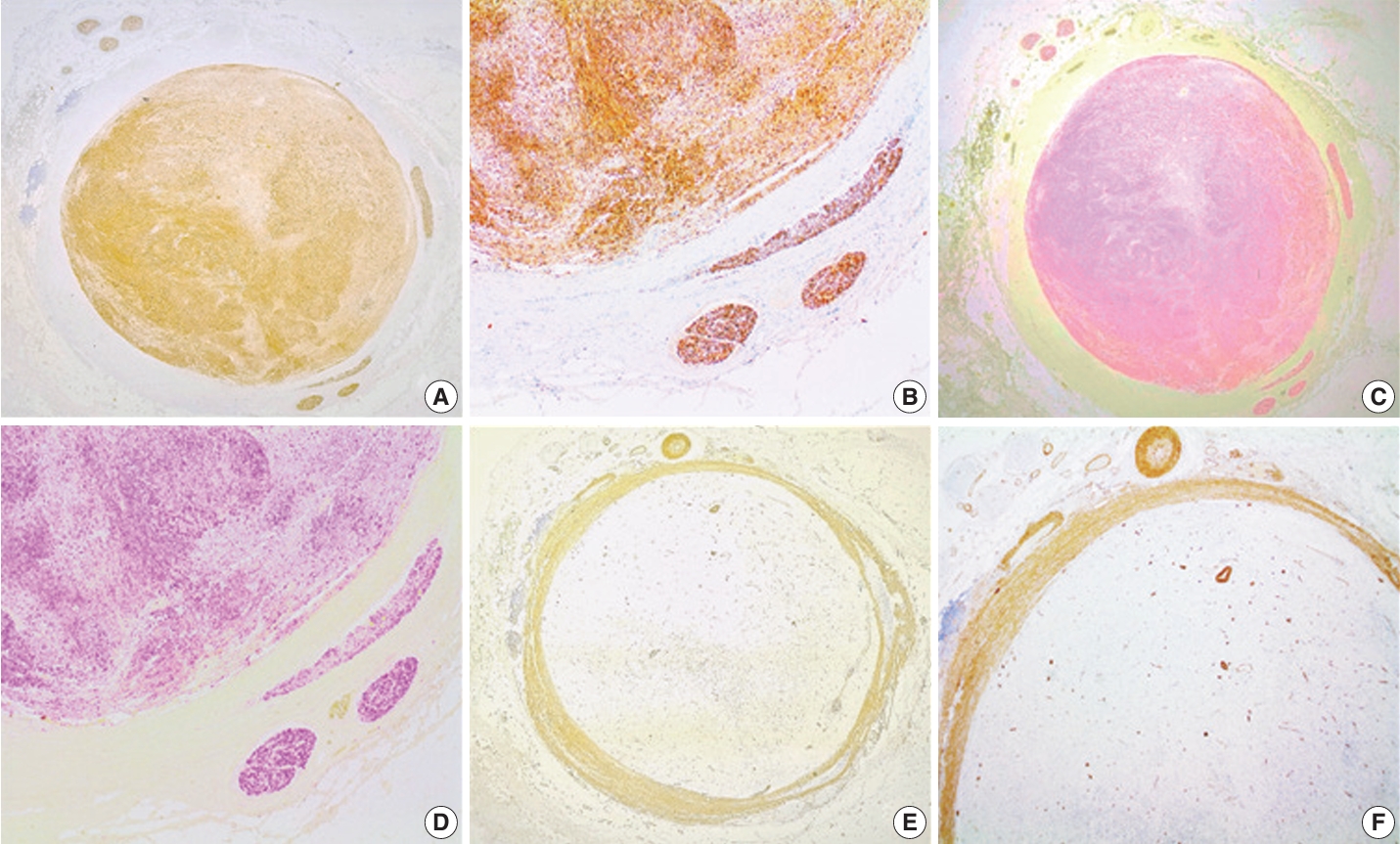
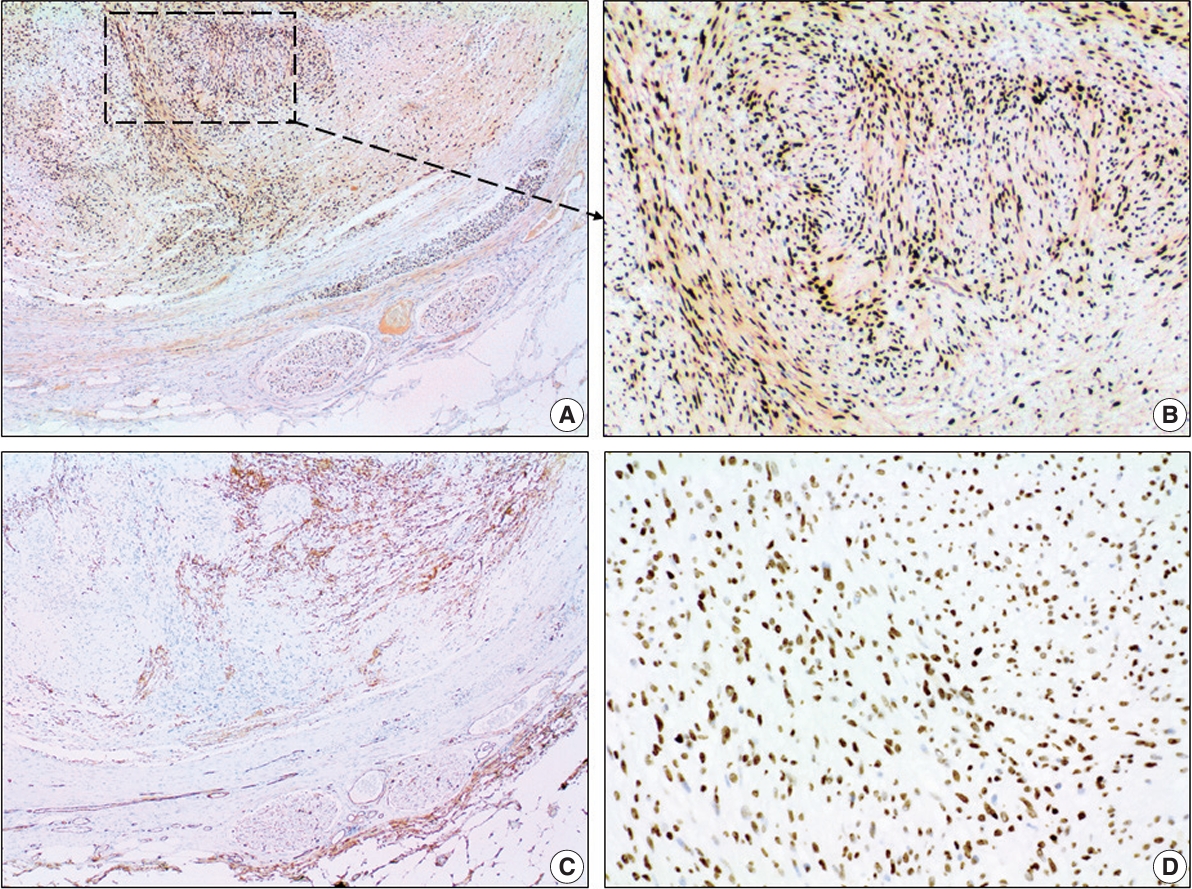
- The tumor had typical immunohistochemical characteristics of a schwannoma. A diffuse and strong positivity for S100 protein (clone EP32, monoclonal antibody, ready-to-use on Leica BOND III system, Leica Biosystems, Newcastle Upon Tyne, UK) (Fig. 2A–D) and SOX10 (clone EP268, monoclonal antibody, ready-to-use on Leica BOND III system, Master Diagnóstica, Grenada, Spain) (Fig. 3A, B) was found in the tumor spindle cells. We also observed a normal nuclear expression level of H3K27me3 (clone C36B11, monoclonal antibody, 1:400 on Leica BOND III system, Cell Signaling Technology, Danvers, MA, USA) in the tumor cells (Fig. 3D). These markers were also positive in the nervi vasorum inside and around the vein wall. The tumor cells were negative for CD34 (clone QBEnd/10, monoclonal antibody, ready-to-use on Leica BOND III system, Leica Biosystems) (Fig. 3C), smooth muscle actin (SMA) (clone Alpha sm-1, monoclonal antibody, ready-to-use on Leica BOND III system, Leica Biosystems) (Fig. 2E, F), and CD31 (clone JC70A, monoclonal antibody, ready-to-use on Leica BOND III system, Leica Biosystems). CD34 (Fig. 3C) and CD31 highlight the endothelial elements inside the tumor and the vein endothelium surrounding the tumor. CD34 was also positive in some fibroblastic cells of the tumor stroma (Fig. 3C). SMA was positive in the smooth muscle cells of the vein wall, small vessels present in the vasa vasorum, and vessels inside the tumor (Fig. 2E, F).
CASE REPORT
- Schwannoma is a nerve sheath tumor entirely or nearly entirely composed of differentiated neoplastic Schwann cells that are associated in some cases with different conditions such as type 2 neurofibromatosis and schwannomatosis [1].
- The incidence of intravascular schwannomas is unknown. The first published case of a schwannoma in a vein lumen was reported in 2011 by Gaudi et al. [5,9].
- Venous obstruction is most commonly seen in the form of deep venous thrombosis [8,13]. To the best of our knowledge, a complete obstruction of a deep vein by an intravascular schwannoma has not been previously reported. This is the third reported case of an intravascular schwannoma located in the posterior calf and the first case of a schwannoma in a deep vein of the left leg. Our case showed some clinical similarities with the cases previously reported by other authors, such as the presence of localized pain, a long evolution period (3 years in our case), and recent size enlargement [9-12]. However, unlike previously reported cases in which the age range was 44–56 years and the tumor size range was 0.3–1.5 cm, our patient was older, 68 years, and the tumor was larger, 2.3 cm.
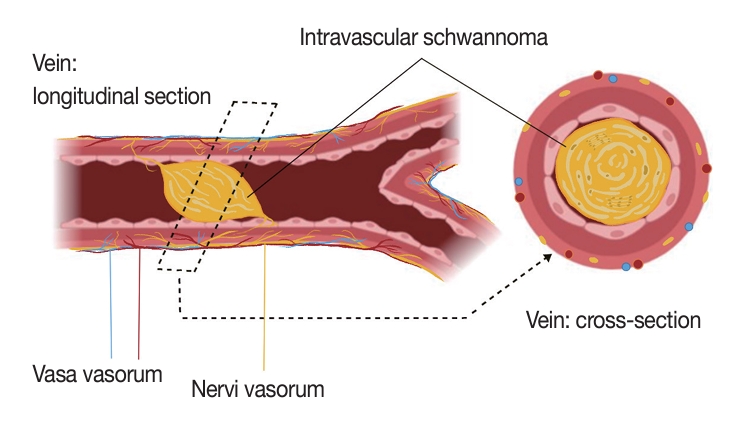
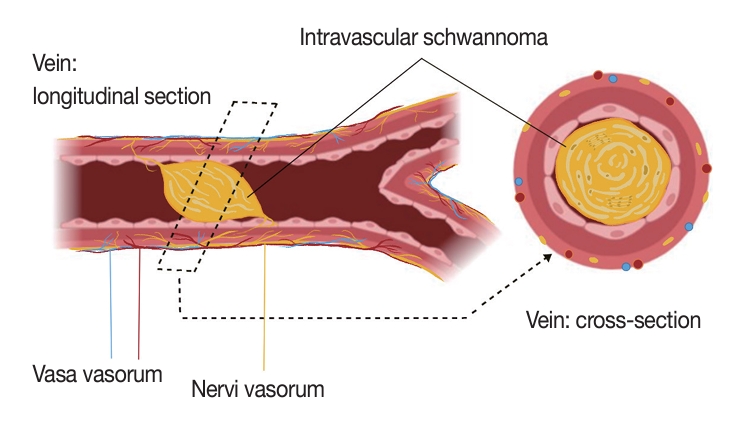
- Various theories have been proposed to explain intravascular schwannoma onset. Some of these theories include the nerve origin being native to the vessel wall, tumor embolization, or direct tumor growth extension [9-11]. In some cases, the schwannomas show vascular alterations that display reactive vessel proliferation and tumor vessel permeability that allow for the schwannoma extension to vascular territories [14]. We theorize a nervi vasorum origin due to the proximity of the intravascular tumor to the nervi vasorum nerves (Fig. 4) seen in our case (Fig. 2A–D). Nervi vasorum, or “vessel nerves,” are small nerves organized as a network and located in the outer and middle layers of the blood vessels [15,16]. Nervi vasorum play an important role in the maintenance of an appropriate tonus of vascular smooth muscle cells and regulate local blood pressure in doing so [17]. This vascular nerve system participates in the regulation of vasoconstriction and vasodilation and is generally composed of sympathetic fibers. Some of these trigger vasodilation and others induce vasoconstriction depending on the nature of the neurotransmitter and receptors located on the target cell, while parasympathetic stimulation triggers vasodilation.
- The correct interpretation of histopathologic features by the pathologist is key to distinguish schwannomas from other tumors on the differential diagnosis list that includes neurofibroma, intravascular leiomyoma/leiomyosarcoma, intravascular lobular capillary hemangioma, intravascular fasciitis, and malignant peripheral nerve sheath tumor.
- The treatment of choice for schwannomas is surgery in the vast majority of symptomatic cases when the integrity of near organs is not compromised; but, in some cases, the surgical option depends on the tumor location [18,19]. There are no widely accepted treatment guidelines for this tumor. In all previously reported cases, surgical removal in these benign cases, as with conventional cases of schwannomas in other locations, has been the treatment of choice.
- The patient is symptom-free after 12 months of follow-up from the surgery and continues to be monitored.
- Recognizing the histopathologic features of this rare entity in an intravascular location is important to prevent misdiagnosis [11].
- More studies with a large prospective series and molecular studies are required to elucidate the mechanisms underlying the development and the clinical and biological behavior of intravascular schwannomas.
DISCUSSION
Ethics Statement
Informed written consent was obtained from the patient. This case report was approved by the ethics committee of the University Hospital of Salamanca (2023 02 387).
Availability of Data and Material
The datasets generated or analyzed during the study are available from the corresponding author on reasonable request.
Code Availability
Not applicable.
Author Contributions
Conceptualization: LMCT, BSB. Funding acquisition: MDLC, JMS. Investigation: LMCT, BSB. Resources: LMCT, BSB, JMS, MRG, JAML, ABJP, IGM, DBS, MDL. Writing—original draft: LMCT. Writing—review & editing: BSB, LMCT, JGR, ABJP, DBS, IGM, MRG, JAML, JMS, MDLC. Approval of final manuscript: all authors.
Conflicts of Interest
The authors declare that they have no potential conflicts of interest.
Funding Statement
No funding to declare.
- 1. Schwannoma Perry A. In: WHO Classification of Tumours Editorial Board, ed. WHO classification of tumours: soft tissue and bone tumours. 5th ed. Lyon: IARC Press, 2020; 226-31.
- 2. Evans DG. Neurofibromatosis type 2 (NF2): a clinical and molecular review. Orphanet J Rare Dis 2009; 4: 16.ArticlePubMedPMCPDF
- 3. Casadei GP, Komori T, Scheithauer BW, Miller GM, Parisi JE, Kelly PJ. Intracranial parenchymal schwannoma: a clinicopathological and neuroimaging study of nine cases. J Neurosurg 1993; 79: 217-22. PubMed
- 4. Voltaggio L, Murray R, Lasota J, Miettinen M. Gastric schwannoma: a clinicopathologic study of 51 cases and critical review of the literature. Hum Pathol 2012; 43: 650-9. ArticlePubMedPMC
- 5. Belakhoua SM, Rodriguez FJ. Diagnostic pathology of tumors of peripheral nerve. Neurosurgery 2021; 88: 443-56. ArticlePubMedPMCPDF
- 6. Guedes-Correa JF, Cardoso RS. Immunohistochemical markers for schwannomas, neurofibromas and malignant peripheral nerve sheath tumors: what can the recent literature tell us? Arq Bras Neurocir 2018; 37: 105-12. Article
- 7. Magro G, Broggi G, Angelico G, et al. Practical approach to histological diagnosis of peripheral nerve sheath tumors: an update. Diagnostics (Basel) 2022; 12: 1463.ArticlePubMedPMC
- 8. Packer CF, Bickel S, Dattilo JB. Vein obstruction [Internet]. Treasure Island: StatPearls Publishing, 2023 [cited 2023 Dec 12]. Available from: http://www.ncbi.nlm.nih.gov/books/NBK537255/.
- 9. Gaudi S, Mills O, Goyette EF, Morgan MB. Intravascular schwannoma. Am J Dermatopathol 2011; 33: 850-4. ArticlePubMed
- 10. Park J, Lee D. Intraarterial schwannoma in horizontal segment of middle cerebral artery causing subarachnoid hemorrhage. J Neurosurg 2013; 118: 1069-71. ArticlePubMed
- 11. Ward RE, Shah JA, Mehregan DR, Ali L. A rare case of intravascular schwannoma. J Cutan Pathol 2018; 45: 731-3. ArticlePubMedPDF
- 12. Dai A, Cai JP. Intravascular schwannoma: a review of a rare diagnosis. J Cutan Pathol 2021; 48: 314-7. ArticlePubMedPDF
- 13. Stubbs MJ, Mouyis M, Thomas M. Deep vein thrombosis. BMJ 2018; 360: k351.ArticlePubMed
- 14. Papiez J, Rojiani MV, Rojiani AM. Vascular alterations in schwannoma. Int J Clin Exp Pathol 2014; 7: 4032-8. PubMedPMC
- 15. Fernandez-Flores A, Saeb-Lima M, Cassarino DS. Intramural schwannoma involving a vein. J Cutan Pathol 2019; 46: 211-5. ArticlePubMedPDF
- 16. Taylor AM, Bordoni B. Histology, blood vascular system [Internet]. Treasure Island: StatPearls Publishing, 2023 [cited 2024 Jan 22]. Available from: http://www.ncbi.nlm.nih.gov/books/NBK553217/.
- 17. Cabrera-Fischer EI, Bia D, Zocalo Y, Wray S, Armentano R. The adventitia layer modulates the arterial wall elastic response to intra-aortic counterpulsation: in vivo studies. Artif Organs 2013; 37: 1041-8. ArticlePubMed
- 18. Halliday J, Rutherford SA, McCabe MG, Evans DG. An update on the diagnosis and treatment of vestibular schwannoma. Expert Rev Neurother 2018; 18: 29-39. ArticlePubMed
- 19. Neurilemmoma (schwannoma) treatment and management [Internet]. Newark: Medscape, 2022 [cited 2023 Dec 15]; Available from: https://emedicine.medscape.com/article/1256405-treatment?form=fpf.
REFERENCES
Figure & Data
References
Citations

 PubReader
PubReader ePub Link
ePub Link-
 Cite this Article
Cite this Article
- Cite this Article
-
- Close
- Download Citation
- Close
- Figure





 E-submission
E-submission