Articles
- Page Path
- HOME > J Pathol Transl Med > Volume 46(3); 2012 > Article
-
Case Report
Squamoid Eccrine Ductal Carcinoma of the Scalp - Yong-Han Jung, Hye-Jung Jo, Mi-Seon Kang
-
Korean Journal of Pathology 2012;46(3):278-281.
DOI: https://doi.org/10.4132/KoreanJPathol.2012.46.3.278
Published online: June 22, 2012
Department of Pathology, Pusan Paik Hospital, Inje University College of Medicine, Busan, Korea.
- Corresponding Author: Mi Seon Kang, M.D. Department of Pathology, Pusan Paik Hospital, Inje University College of Medicine, 75 Bokji-ro, Busanjin-gu, Busan 614-735, Korea. Tel: +82-51-890-6043, Fax: +82-51-891-1834, pathmsk@hanmail.net
• Received: April 7, 2011 • Revised: May 24, 2011 • Accepted: May 27, 2011
© 2012 The Korean Society of Pathologists/The Korean Society for Cytopathology
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/3.0) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
- 13,353 Views
- 126 Download
- 13 Crossref
Abstract
- Squamoid eccrine ductal carcinoma (SEDC) is an exceedingly rare tumor that shows both squamous and adnexal ductal differentiation. We report a case of this unusual tumor occurring on the occiput of a 53-year-old man. A histopathological examination revealed a nodular lesion infiltrating the dermis and subcutaneous tissue with numerous duct-like structure and squamoid differentiation foci. Five months later, the patient presented with a palpable mass at the site of the previous excision and the right side of the neck. Sono-guided fine needle aspiration of the right neck mass was performed and was diagnosed as a metastastasis of a lymph node. A right neck node dissection and re-excision of the occiput was performed. The histopathological findings were similar, but squamoid differentiation was more prominent than that in the previous lesion. Because of the rarity of SEDC, little is known about its biological behavior and optimal treatment.
- A 53-year-old man presented with a 10-year history of a nodule that had recently started growing and producing an itching sensation on the right occiput. His medical history was unremarkable. A large 2.3×1.5 cm nodular mass was seen, and focal ulceration was noted. A neck computed tomography scan suggested a benign soft tissue mass in the subcutaneous layer, so a local excision was performed. The tumor measured 2.6×1.7×1.5 cm, and there was no evidence of erosion or ulcer on the skin surface. The cut surface showed a relatively well demarcated white solid mass, which was located in the dermis and subcutaneous fat tissue. A focal fibrotic infiltrative border and connection with the epidermis were also noted.
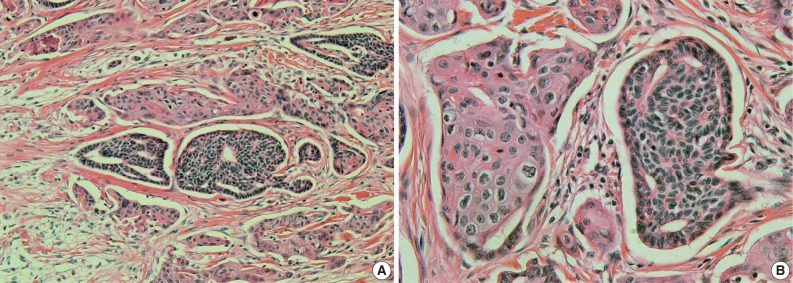
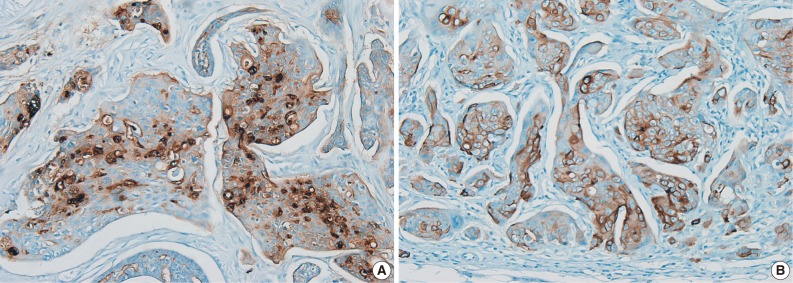
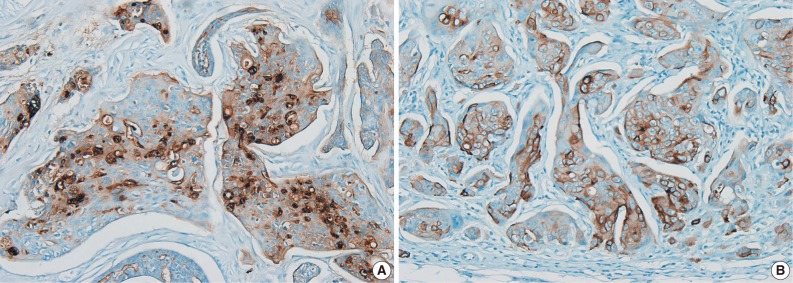
- A histopathological examination revealed a nodular lesion composed of duct-like structures lined by cuboidal epithelium (Fig. 1A) in the upper portion of the dermis and numerous nests of squamoid cells with amphophilic cytoplasm and keratinization in the deeper portion (Fig. 1B). The periphery of the lesion revealed more prominent squamoid differentiation and showed plump eosinophilic cytoplasm and keratinization with dense lymphocytic infiltration. Most of the tumor cells were slightly pleomorphic (variation of cellular and nuclear size, high N/C ratio, and hyperchromatic nucleoli), but focally more pleomorphism was seen in the infiltrating lesion. Tumor necrosis foci, lymphatic emboli, and mitotic figures were present. Immunohistochemistry showed positive staining for cytokeratin (CK), CK5/6, CK7, carcinoembryonic antigen (CEA) (Fig. 2A), and epithelial membrane antigen (EMA) (Fig. 2B) but was negative for S-100 protein, gross cystic disease fluid protein-15, and CD15. A diagnosis of SEDC was made. The lateral resection margin was free, but the deep resection margin was very close to the tumor. No further treatments were administered.
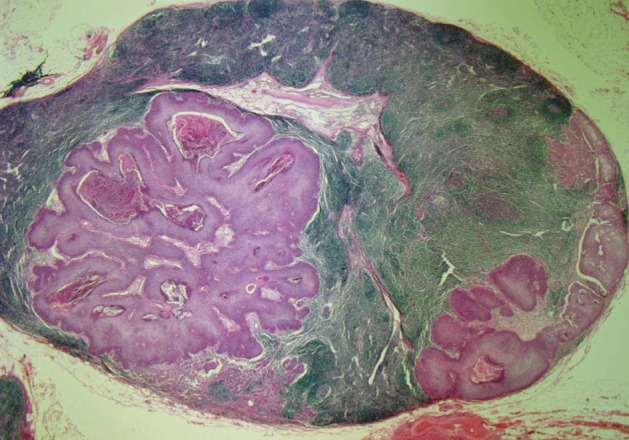
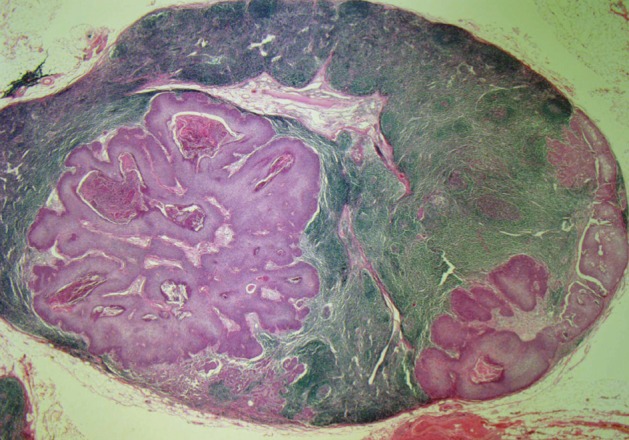
- After 5 months, the patient returned with a palpable mass at the previous excision site and on the right side of the neck. Sono-guided fine needle aspiration of the right neck mass revealed a metastatic adenocarcinoma probably from an eccrine ductal adenocarcinoma of the skin. A right neck node dissection and wide local excision of the occiput was performed. The dissected neck lymph nodes were involved with tumor cells and showed ductal structure and squamous differentiation (Fig. 3).
CASE REPORT
- Eccrine carcinomas uncommonly originate from eccrine glands. A review at the Mayo Clinic, covering more than 75 years of skin tumors, reported 14 cases of eccrine carcinoma.9 These carcinomas have a slow growth rate and a high potential for recurrence. Diverse variants of eccrine carcinoma exist, including a variant with abundant fibromyxoid stroma, a spindle cell variant with myoepithelial differentiation, a basal cell carcinoma-like variant, and a SEDC variant characterized by squamous metaplasia.1,7 Classification of eccrine neoplasms is problematic because no universally accepted classification scheme exists, and the variable clinical appearance and histological features are often not predictive of clinical behavior.10 Therefore, the diagnostic identification of sweat gland carcinomas is hampered by their rarity and their histological resemblance to various visceral tumors, leading to confusion with metastatic lesions.
- SEDC is an exceedingly rare variant of eccrine carcinoma. Wong et al.5 reported the first three cases of SEDC in 1997. Since then, only four additional cases have been reported. SEDC shows a predilection for the head, neck, and extremities and presents most often in the middle-aged and elderly as a hard, usually nonulcerated cutaneous nodule. The histological features of SEDC are duct-like structures resembling the dermal part of the eccrine duct and atypical squamous cell clusters. Follicular or sebaceous differentiation can be seen.11
- The histological differential diagnosis for SEDC includes microcystic adnexal carcinoma (MAC) with squamous features. MAC is poorly circumscribed and usually deeply infiltrating. It expresses a constellation of features, including numerous keratinous cysts. Keratinous cysts are often superficially located in the dermis without connection to the epidermis. Deeper components include smaller nests and strands of cells. A dense fibrous stroma surrounds all components. The tumor cells express CEA and EMA.10,12,13 In our case, the absence of a keratinous cyst and a dense fibrous stroma surrounding tumor cells made it less likely that it was a MAC. The differential diagnosis could also be SCC. Immunohistochemical staining with CEA could resolve this problem, as SCCs are typically negative. But if ductal differentiation is not obvious in shave or punch biopsy specimens, SEDC may be mistaken for a SCC.
- Recent studies have reported that positive p63 and CK5/6 staining is relatively useful for distinguishing primary adnexal neoplasm with sweat gland differentiation from metastatic adenocarcinoma at various sites.14 p63 is a member of the p53 gene family and may be essential for regulating the differentiation of cells in normal human skin. p63 is normally expressed in basal and spinous cell nuclei of the epidermis, peripheral cells of the eccrine dermal ducts, germinative cells of sebaceous glands, and myoepithelial cells of the terminal portion of the eccrine and apocrine glands.15 CK5/6 is a basic high molecular weight CK. CK5/6 are normally expressed in stratified squamous epithelium, the basal-myoepithelial cell layer of the prostate, the salivary gland, and the breasts, as well as in myoepithelial cells of secretory glands and both layers of the epithelial cells of excretory ducts of eccrine and apocrine glands.16 In our case, the tumor cells were p63 negative but CK5/6 positive.
- Limited information is available to manage SEDC because of its rarity. However, a wide surgical excision with a wide negative margin (with or without Mohs's technique) is the treatment of choice. A high rate of local recurrence and subsequent metastasis after conventional surgical excision of eccrine carcinoma is observed. In our case, the SEDC recurred and showed lymph node metastasis 5 months after the first excision.
- The significance of squamous differentiation in eccrine carcinoma is unclear.17 Some believe that it shows more aggressive biological behavior, whereas others believe that it is an incidental finding and has no impact on disease biology. A study by Kohda et al.17 reported that both benign and malignant eccrine neoplasms had foci of squamous differentiation.
- Our case was interesting for a variety of reasons. First, the relative rarity of SEDC was reported. Second, the tumor recurred and metastasized to a lymph node.
DISCUSSION
- 1. Wick MR, Swanson PE. Cutaneous adnexal tumors: a guide to pathological diagnosis. 1991; Chicago: American Society Of Clinical Pathologists Press, 10.
- 2. Alessi E, Caputo R. Syringomatous carcinoma of the scalp presenting as a slowly enlarging patch of alopecia. Am J Dermatopathol 1993; 15: 503-505. ArticlePubMed
- 3. Gregurek-Novak T, Talan-Hranilović J, Troskot N, Vucić M, Kruslin B. Syringoid eccrine carcinoma. J Eur Acad Dermatol Venereol 2001; 15: 143-146. ArticlePubMed
- 4. Goto M, Sonoda T, Shibuya H, et al. Digital syringomatous carcinoma mimicking basal cell carcinoma. Br J Dermatol 2001; 144: 438-439. ArticlePubMed
- 5. Wong TY, Suster S, Mihm MC. Squamoid eccrine ductal carcinoma. Histopathology 1997; 30: 288-293. ArticlePubMedPDF
- 6. Kim YJ, Kim AR, Yu DS. Mohs micrographic surgery for squamoid eccrine ductal carcinoma. Dermatol Surg 2005; 31(11 Pt 1): 1462-1464. ArticlePubMed
- 7. Chhibber V, Lyle S, Mahalingam M. Ductal eccrine carcinoma with squamous differentiation: apropos a case. J Cutan Pathol 2007; 34: 503-507. ArticlePubMed
- 8. Herrero J, Monteagudo C, Jordá E, Llombart-Bosch A. Squamoid eccrine ductal carcinoma. Histopathology 1998; 32: 478-480. ArticlePDF
- 9. Wick MR, Goellner JR, Wolfe JT 3rd, Su WP. Adnexal carcinomas of the skin. I. Eccrine carcinomas. Cancer 1985; 56: 1147-1162. ArticlePubMed
- 10. Wildemore JK, Lee JB, Humphreys TR. Mohs surgery for malignant eccrine neoplasms. Dermatol Surg 2004; 30(12 Pt 2): 1574-1579. ArticlePubMed
- 11. Kavand S, Cassarino DS. "Squamoid eccrine ductal carcinoma": an unusual low-grade case with follicular differentiation. Are these tumors squamoid variants of microcystic adnexal carcinoma? Am J Dermatopathol 2009; 31: 849-852. ArticlePubMed
- 12. Antley CA, Carney M, Smoller BR. Microcystic adnexal carcinoma arising in the setting of previous radiation therapy. J Cutan Pathol 1999; 26: 48-50. ArticlePubMed
- 13. Ongenae KC, Verhaegh ME, Vermeulen AH, Naeyaert JM. Microcystic adnexal carcinoma: an uncommon tumor with debatable origin. Dermatol Surg 2001; 27: 979-984. ArticlePubMed
- 14. Qureshi HS, Ormsby AH, Lee MW, Zarbo RJ, Ma CK. The diagnostic utility of p63, CK5/6, CK 7, and CK 20 in distinguishing primary cutaneous adnexal neoplasms from metastatic carcinomas. J Cutan Pathol 2004; 31: 145-152. ArticlePubMedPDF
- 15. Dotto JE, Glusac EJ. p63 is a useful marker for cutaneous spindle cell squamous cell carcinoma. J Cutan Pathol 2006; 33: 413-417. ArticlePubMed
- 16. Chu PG, Weiss LM. Expression of cytokeratin 5/6 in epithelial neoplasms: an immunohistochemical study of 509 cases. Mod Pathol 2002; 15: 6-10. ArticlePubMedPDF
- 17. Kohda M, Manabe T, Ueki H. Squamous islands in eccrine neoplasms. Am J Dermatopathol 1990; 12: 344-349. ArticlePubMed
REFERENCES
Figure & Data
References
Citations
Citations to this article as recorded by 

- Surgical Treatment of Squamoid Eccrine Ductal Carcinoma: A Systematic Review
Nehaa Sohail, Hannah R. Riva, Mojahed Mohammad K Shalabi, Benjamin Kelley, Stanislav N. Tolkachjov
Dermatologic Surgery.2026; 52(3): 290. CrossRef - Squamoid Eccrine Ductal Carcinoma: A Systematic Review
Baraa Saifi, Maria Maalouf, Areeba Hasan, Doha Fadel, Marita Kassab, Nancy Emmanuel
Current Dermatology Reports.2025;[Epub] CrossRef - Aggressive Squamoid Eccrine Ductal Carcinoma of the Face: A Rare and Challenging Diagnosis—Case Report and Literature Review
Bruno Špiljak, Damir Sauerborn, Matej Tomas, Brankica Gregorić Butina, Ivana Mahovne, Suzana Erić, Bruno Vidaković, Stjepanka Lešić
Medicina.2025; 61(4): 612. CrossRef - Squamoid Eccrine Ductal Carcinoma: Treatment and Outcomes
Megan M. Lim, Jillian A. Macdonald
The American Journal of Dermatopathology.2022; 44(4): 249. CrossRef - Recurrent Squamoid Eccrine Ductal Carcinoma After Mohs Micrographic Surgery
Cardwell Gavin, Brinker Alyson
Dermatologic Surgery.2021; 47(8): 1108. CrossRef - Carcinoma ecrino: dos casos de una neoplasia inusual y revisión de la literatura
Clara Matas-Nadal, Josep Manel Fernández-Armenteros, Felip Vilardell, Josep Manel Casanova, Rafael S. Aguayo Ortiz
Piel.2020; 35(3): 159. CrossRef - Squamoid eccrine ductal carcinoma: a case report
Zeynep Bayramoğlu, Betül Ünal
The European Research Journal.2020; 6(2): 173. CrossRef - Squamoid eccrine ductal carcinoma of the ear helix
Sunmin Yim, Yun Ho Lee, Seoung Wan Chae, Won‐Serk Kim
Clinical Case Reports.2019; 7(7): 1409. CrossRef - A Case of Eccrine Carcinoma Presenting with Neurological Manifestations
Lynne Michelle B Soldivillo, Rosalina Espiritu Picar, Allen Evaristo
Philippine Journal of Internal Medicine.2017; 55(1): 46. CrossRef - Squamoid eccrine ductal carcinoma of the scalp
Hsien Chan, Vicki Howard, Denis Moir, Delwyn Dyall‐Smith
Australasian Journal of Dermatology.2016;[Epub] CrossRef - Squamoid Eccrine Ductal Carcinoma
Michiel P.J. van der Horst, Adriana Garcia-Herrera, Dorota Markiewicz, Blanca Martin, Eduardo Calonje, Thomas Brenn
American Journal of Surgical Pathology.2016; 40(6): 755. CrossRef - Squamoid eccrine ductal carcinoma
Maria Isabel Ramos Saraiva, Marcella Amaral Horta Barbosa Vieira, Larissa Karine Leite Portocarrero, Rafael Cavanellas Fraga, Priscila Kakizaki, Neusa Yuriko Sakai Valente
Anais Brasileiros de Dermatologia.2016; 91(6): 799. CrossRef - Anatomoclinical study of 30 cases of sclerosing sweat duct carcinomas (microcystic adnexal carcinoma, syringomatous carcinoma and squamoid eccrine ductal carcinoma)
E. Frouin, M.D. Vignon‐Pennamen, B. Balme, B. Cavelier‐Balloy, U. Zimmermann, N. Ortonne, A. Carlotti, L. Pinquier, J. André, B. Cribier
Journal of the European Academy of Dermatology and Venereology.2015; 29(10): 1978. CrossRef
Squamoid Eccrine Ductal Carcinoma of the Scalp



Fig. 1 A nodular lesion composed of duct-like structures lined by cuboidal epithelium (A) and numerous nests of squamoid cells with amphophilic cytoplasm and keratinization (B).
Fig. 2 The tumor cells are immunoreactively positive for carcinoembryonic antigen (A) and epithelial membrane antigen (B).
Fig. 3 Dissected neck lymph nodes are involved with tumor cells and show ductal structure and squamous differentiation.
Fig. 1
Fig. 2
Fig. 3
Squamoid Eccrine Ductal Carcinoma of the Scalp

 E-submission
E-submission
 PubReader
PubReader Cite this Article
Cite this Article