Articles
- Page Path
- HOME > J Pathol Transl Med > Volume 47(2); 2013 > Article
-
Case Study
Silent Colonic Malakoplakia in a Living-Donor Kidney Transplant Recipient Diagnosed during Annual Medical Examination - Go Eun Bae, Nara Yoon, Ha Young Park, Sang Yun Ha, Junhun Cho, Yunkyung Lee, Kyoung-Mee Kim, Cheol Keun Park
-
Korean Journal of Pathology 2013;47(2):163-166.
DOI: https://doi.org/10.4132/KoreanJPathol.2013.47.2.163
Published online: April 24, 2013
Department of Pathology, Samsung Medical Center, Sungkyunkwan University School of Medicine, Seoul, Korea.
- Corresponding Author: Yunkyung Lee, M.D. Department of Pathology, Samsung Medical Center, Sungkyunkwan University School of Medicine, 81 Irwon-ro, Gangnam-gu, Seoul 135-710, Korea. Tel: +82-2-3410-2801, Fax: +82-2-3410-6396, yk427.lee@samsung.com
• Received: June 14, 2012 • Revised: July 25, 2012 • Accepted: July 26, 2012
© 2013 The Korean Society of Pathologists/The Korean Society for Cytopathology
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/3.0/) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
Abstract
- Malakoplakia is a characteristic inflammatory condition, which is usually seen in the urogenital tract, and less frequently in the gastrointestinal tract. We present a case of colonic malakoplakia in an immunocompromised patient. A 55-year-old female visited the outpatient clinic for routine cancer surveillance. Her past medical history was significant for kidney transplantation 11 years ago, and she had been taking immunosuppressants. A colonoscopy revealed several depressed flat lesions and elevated polyps, which were 0.3 to 0.4 cm in size and accompanied by whitish exudates. A biopsy revealed an infiltration of histiocytes with ample granular eosinophilic cytoplasm, with some lymphocytes and plasma cells. Many histiocytes had the characteristic morphology, described as Michaelis-Gutmann bodies: one or several round basophilic structures of approximately 1 to 10 µm in size with some being laminated, some appearing homogeneous, and others having a dense central core with a targetoid appearance. These Michaelis-Gutmann bodies were positively stained on von Kossa stain, and were diagnostic for malakoplakia.
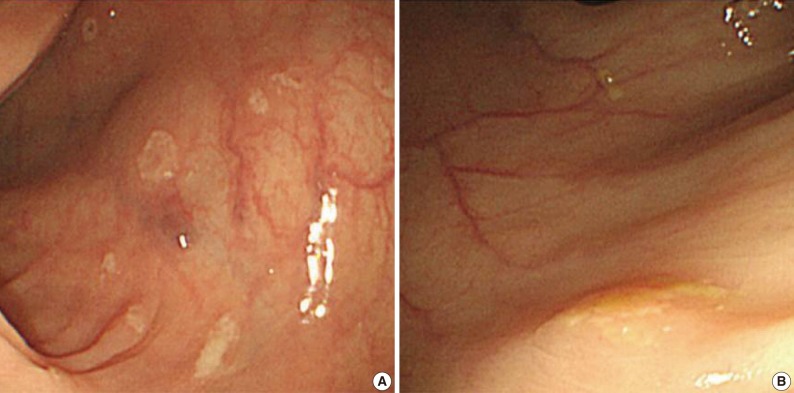
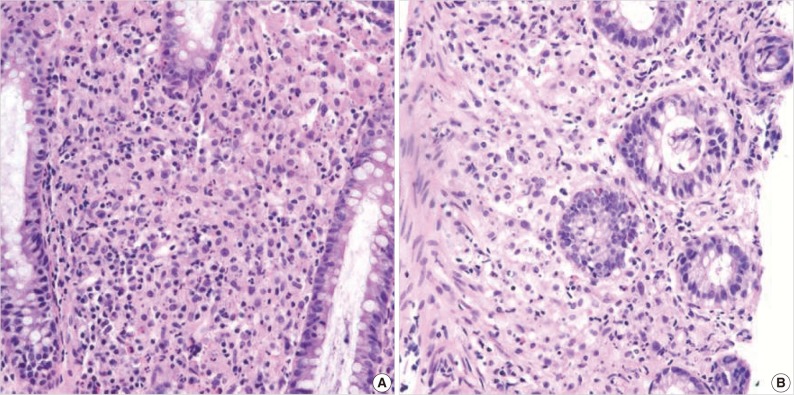
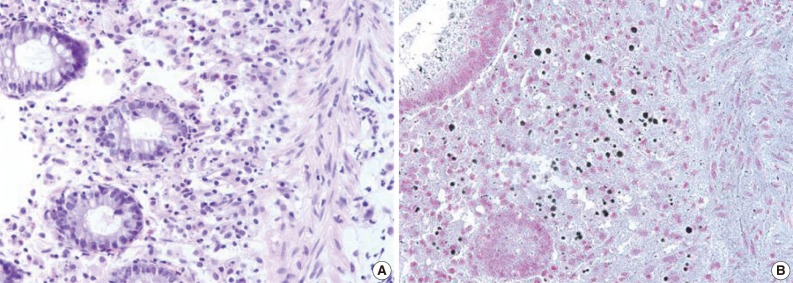
- A 55-year-old female visited the outpatient clinic of the Samsung Medical Center for routine cancer surveillance in May 2011. The patient received a living-donor kidney transplant eleven years ago for end-stage renal disease secondary to IgA nephropathy at Kyunghee University Medical Center. She was on stable doses of immunosuppressive drugs, including prednisone, tacrolimus, and mycophenolate, at the time of the current visit. She had no rejection or other complications of transplantation to date, and was asymptomatic. Her routine cancer surveillance included complete blood counts, a liver function test, abdominal sonogram, gynecologic exam, upper gastrointestinal endoscopy, and colonoscopy. Results were all unremarkable, except or the colonoscopy findings. The colonoscopy revealed several depressed flat lesions and elevated polyps, which were 0.3 to 0.4 cm in size and accompanied by whitish exudates (Fig. 1). The differential diagnosis for the endoscopic lesions included graft-versus-host disease, cytomegalovirus colitis, and amoebiasis. Multiple biopsies were obtained from several sites. Microscopic findings from all biopsy materials revealed the infiltration of many histiocytes with ample granular eosinophilic cytoplasm with some lymphocytes and plasma cells. Many histiocytes had the characteristic morphology described as Michaelis-Gutmann bodies (Fig. 2): one or several round basophilic structures of approximately 1 to 10 µm in size, with some being laminated, some appearing homogeneous, and others having a dense central core with a targetoid appearance. These Michaelis-Gutmann bodies were positively stained on von Kossa stain, and were diagnostic for malakoplakia (Fig. 3). The patient did not receive any treatment. After 1 year, she underwent an annual health examination at Samsung Medical Center in May 2012. She still did not have any symptoms, and had been taking immune suppressive medication. This time she did not receive a colonoscopy. Aspects of the check-up showed results within normal ranges.
CASE REPORT
- Malakoplakia is a chronic granulomatous inflammatory disorder frequently involving the genitourinary tract. The gastrointestinal tract is the second most commonly involved system of malakoplakia.3 The etiology of malakoplakia is idiopathic, but is thought to be associated with dysfunctional bacterial clearance by neutrophils and macrophages,7 and thus has been closely associated with states of immunodeficiency.8 Diagnosis is established histologically by the presence of large "Hansemann macrophages," containing laminated calcific spherules, known as Michaelis-Gutmann inclusions.9 The Michaelis-Gutmann body is derived from phagolysosomes containing incompletely destroyed bacteria, and is pathognomonic for malakoplakia.10 Escherichia coli is the most commonly associated bacterial pathogen, and is found in over 90% of affected patients.2
- Malakoplakia has been described in post-transplant patients. We performed a Medline search and analyzed all cases reporting colonic malakoplakia in transplant recipients. Only four cases were previously reported in kidney transplant recipients from 1994 to 2010. Other transplant-associated colonic malakoplakia included three cases with liver transplants and one with a cardiac transplant.8,11-13 All patients in the reported cases presented some symptoms, including diarrhea, abdominal pain, fever, and intestinal perforation. Precedent colonic malakoplakia cases associated with kidney transplant are compared in Table 1, which shows patient ages ranging from 40 to 52 years,2,4-6 and all were male. Unlike the previous cases, our case is unique for its demographics, particularly the age and female sex, and for its asymptomatic presentation. This is the fourth case report of colonic malakoplakia identified subsequent to kidney transplantation, and the only case of its asymptomatic presentation.
- There are two strategies for the treatment of malakoplakia: antibiotics for the bacterial infection and a cholinergic agonist to improve macrophage function.8 Antibiotics that penetrate and concentrate in macrophages, such as quinolone or trimethoprim-sulfamethoxazole, are currently used with a high cure rate for malakoplakia.14 Antibiotic therapy directed against E. coli in combination with surgery provides the best chance of cure.14 Van Furth et al.15 also reported on two patients with malakoplakia, who were treated with long-term ciprofloxacin and experienced a regression of their lesions. The second approach is an attempt to correct the lysosomal defect by using a cholinergic agonist.14 A cholinergic agonist may correct the decreased cGMP levels that are believed to interfere with bactericidal activity.16 Discontinuation of immunosuppressants is also usually needed to effectively treat malakoplakia.9 However, unlike other previously reported cases, this case did not reveal any symptoms and antecedent infection with gram-negative bacteria despite extensive colonic involvement. This case is unprecedented to date, and therefore there are no available treatment guidelines. Our patient did not require any of the above therapies, given her indolent and asymptomatic presentation.
DISCUSSION
- 1. Puerto IM, Mojarrieta JC, Martinez IB, Navarro S. Renal malakoplakia as a pseudotumoral lesion in a renal transplant patient: a case report. Int J Urol 2007; 14: 655-657. ArticlePubMed
- 2. Berney T, Chautems R, Ciccarelli O, Latinne D, Pirson Y, Squifflet JP. Malakoplakia of the caecum in a kidney-transplant recipient: presentation as acute tumoral perforation and fatal outcome. Transpl Int 1999; 12: 293-296. ArticlePubMed
- 3. Fenoglio-Preiser CM, Noffsinger AE, Stemmermann GN, Lantz PE, Isaacson PG. Gastrointestinal pathology: an atlas and text. 2008; 3rd ed. Philadelphia: Wolters Kluwer/Lippincott Williams & Wilkins.
- 4. Ourahma S, Barrou B, Bitker MO, Sylla C, Mouquet C, Chatelain C. Rectal malacoplakia in a renal transplant recipient. Transplant Proc 1996; 28: 2824.PubMed
- 5. Shah MB, Sundararajan S, Crawford CV, Hartono C. Malakoplakia-induced diarrhea in a kidney transplant recipient. Transplantation 2010; 90: 461-462. ArticlePubMed
- 6. Yousif M, Abbas Z, Mubarak M. Rectal malakoplakia presenting as a mass and fistulous tract in a renal transplant patient. J Pak Med Assoc 2006; 56: 383-385. PubMed
- 7. Streem SB. Genitourinary malacoplakia in renal transplant recipients: pathogenic, prognostic and therapeutic considerations. J Urol 1984; 132: 10-12. ArticlePubMed
- 8. Kim PT, Davis JE, Erb SR, Yoshida EM, Steinbrecher UP. Colonic malakoplakia in a liver transplant recipient. Can J Gastroenterol 2007; 21: 753-755. ArticlePubMedPMCPDF
- 9. Biggar WD, Crawford L, Cardella C, Bear RA, Gladman D, Reynolds WJ. Malakoplakia and immunosuppressive therapy: reversal of clinical and leukocyte abnormalities after withdrawal of prednisone and azathioprine. Am J Pathol 1985; 119: 5-11. PubMedPMC
- 10. Dobyan DC, Truong LD, Eknoyan G. Renal malacoplakia reappraised. Am J Kidney Dis 1993; 22: 243-252. ArticlePubMed
- 11. Teeters JC, Betts R, Ryan C, et al. Rectal and cutaneous malakoplakia in an orthotopic cardiac transplant recipient. J Heart Lung Transplant 2007; 26: 411-413. ArticlePubMed
- 12. Weinrach DM, Wang KL, Cisler JJ, Diaz LK. Pathologic quiz case: a 54-year-old liver transplant recipient with diffuse thickening of the sigmoid colon. Malakoplakia of the colon associated with liver transplant. Arch Pathol Lab Med 2004; 128: e133-e134. PubMed
- 13. Rull R, Grande L, García-Valdecasas JC, et al. Malakoplakia in the gastrointestinal tract of a liver transplant recipient. Transplantation 1995; 59: 1492-1494. ArticlePubMed
- 14. Yousef GM, Naghibi B, Hamodat MM. Malakoplakia outside the urinary tract. Arch Pathol Lab Med 2007; 131: 297-300. ArticlePubMedPDF
- 15. van Furth R, van't Wout JW, Wertheimer PA, Zwartendijk J. Ciprofloxacin for treatment of malakoplakia. Lancet 1992; 339: 148-149. ArticlePubMed
- 16. Abdou NI, NaPombejara C, Sagawa A, et al. Malakoplakia: evidence for monocyte lysosomal abnormality correctable by cholinergic agonist in vitro and in vivo. N Engl J Med 1977; 297: 1413-1419. ArticlePubMed
REFERENCES
Fig. 1Endoscopic features of the ascending (A) and transverse (B) colon. A colonoscopy reveals several depressed flat lesions and elevated polyps, which are measuring up to 4 mm in size with whitish exudates.
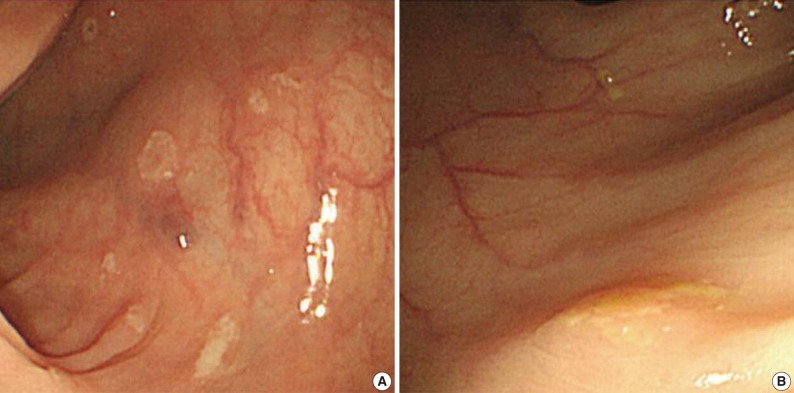

Fig. 2(A) There is an infiltration of many histiocytes with ample granular eosinophilic cytoplasm with some lymphocytes and plasma cells (ascending colon). (B) Round basophilic structures are identified, measuring approximately between 1 µm and 10 µm in size within histiocytes, so-called Michaelis-Gutmann bodies (transverse colon).
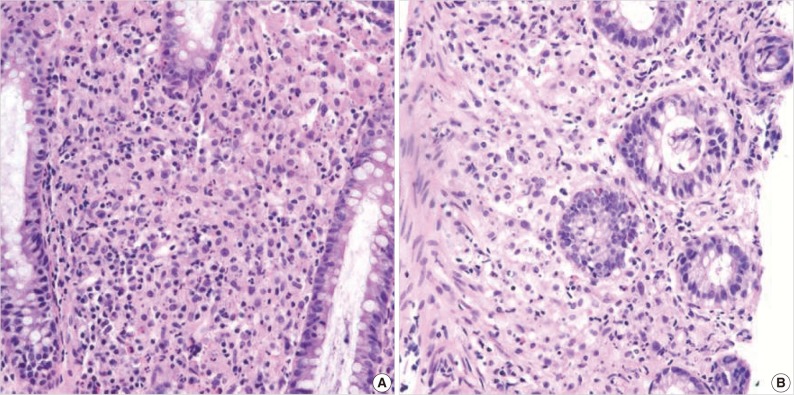

Fig. 3Michaelis-Gutmann bodies are positively stained on von Kossa stain. (A) Hematoxylin and eosin stain. (B) von Kossa stain.
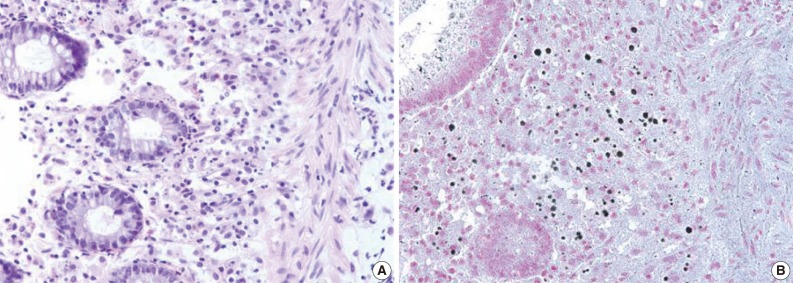

Table 1.Clinical features of previously reported colonic malakoplakia in a kidney transplant patient
| Reference | Site of involvement | Complaints | Presentation | Age (yr)/Sex |
|---|---|---|---|---|
| Shah et al. [5] | Cecum ascending and sigmoid colon | Diarrhea | 3-4 mm polypoid nodule | 45/M |
| Berney et al. [2] | Cecum and rectum | Acute intestinal perforation | Mass forming | 52/M |
| Yousif et al. [6] | Rectum | Painful defecation | Mass and fistulous tract forming | 40/M |
| Present case | From cecum to descending colon | No symptom | 3-4 mm multiple polypoid nodules and depressed lesions | 55/F |
Figure & Data
References
Citations
Citations to this article as recorded by 

- Malakoplakia in kidney transplant recipients: Three case reports
Prathap Kumar Simhadri, Renish Contractor, Deepak Chandramohan, Matthew McGee, Udit Nangia, Mohammad Atari, Syed Bushra, Sanjana Kapoor, Ramya Krishna Velagapudi, Pradeep K Vaitla
World Journal of Nephrology.2025;[Epub] CrossRef - Caecal malakoplakia: a rare mimic of malignancy
Jeffrey Li Voon Chong, Noor Ali
BMJ Case Reports.2024; 17(1): e257130. CrossRef - A Surgical Challenge Generated by Colonic Malakoplakia in Disguise as a Locally Advanced Colonic Malignancy—A Case Report
Cristina Șerban, Alexandra Toma, Dragoș Cristian Voicu, Constantin Popazu, Dorel Firescu, George Țocu, Raul Mihailov, Laura Rebegea
Medicina.2023; 59(1): 156. CrossRef - Colonic malakoplakia in a cardiac transplant recipient: A case report
Sadiya Shafijan
Indian Journal of Pathology and Microbiology.2020; 63(2): 322. CrossRef - Immunosuppressive drugs and the gastrointestinal tract in renal transplant patients
Merel M. Tielemans, Gerben A.J. van Boekel, Teun van Gelder, Eric T. Tjwa, Luuk B. Hilbrands
Transplantation Reviews.2019; 33(2): 55. CrossRef - Malakoplakia of the colon following renal transplantation in a 73 year old woman: report of a case presenting as intestinal perforation
Andrew Mitchell, Alexandre Dugas
Diagnostic Pathology.2019;[Epub] CrossRef - Colonic malakoplakia in a liver transplant recipient: A case report
Rana Ajabnoor, Mohammad Mawardi, Abdulmonem Almutawa
Human Pathology: Case Reports.2019; 18: 200323. CrossRef - Malakoplakia after kidney transplantation: Case report and literature review
John Fredy Nieto‐Ríos, Isabel Ramírez, Mónica Zuluaga‐Quintero, Lina María Serna‐Higuita, Federico Gaviria‐Gil, Alejandro Velez‐Hoyos
Transplant Infectious Disease.2017;[Epub] CrossRef - Megalocytic Interstitial Nephritis Following Acute Pyelonephritis with Escherichia coli Bacteremia: A Case Report
Hee Jin Kwon, Kwai Han Yoo, In Young Kim, Seulkee Lee, Hye Ryoun Jang, Ghee Young Kwon
Journal of Korean Medical Science.2015; 30(1): 110. CrossRef
 PubReader
PubReader ePub Link
ePub Link-
 Cite this Article
Cite this Article
- Cite this Article
-
- Close
- Download Citation
- Close
- Figure
Silent Colonic Malakoplakia in a Living-Donor Kidney Transplant Recipient Diagnosed during Annual Medical Examination



Fig. 1 Endoscopic features of the ascending (A) and transverse (B) colon. A colonoscopy reveals several depressed flat lesions and elevated polyps, which are measuring up to 4 mm in size with whitish exudates.
Fig. 2 (A) There is an infiltration of many histiocytes with ample granular eosinophilic cytoplasm with some lymphocytes and plasma cells (ascending colon). (B) Round basophilic structures are identified, measuring approximately between 1 µm and 10 µm in size within histiocytes, so-called Michaelis-Gutmann bodies (transverse colon).
Fig. 3 Michaelis-Gutmann bodies are positively stained on von Kossa stain. (A) Hematoxylin and eosin stain. (B) von Kossa stain.
Fig. 1
Fig. 2
Fig. 3
Silent Colonic Malakoplakia in a Living-Donor Kidney Transplant Recipient Diagnosed during Annual Medical Examination
| Reference | Site of involvement | Complaints | Presentation | Age (yr)/Sex |
|---|---|---|---|---|
| Shah et al. [5] | Cecum ascending and sigmoid colon | Diarrhea | 3-4 mm polypoid nodule | 45/M |
| Berney et al. [2] | Cecum and rectum | Acute intestinal perforation | Mass forming | 52/M |
| Yousif et al. [6] | Rectum | Painful defecation | Mass and fistulous tract forming | 40/M |
| Present case | From cecum to descending colon | No symptom | 3-4 mm multiple polypoid nodules and depressed lesions | 55/F |
Table 1. Clinical features of previously reported colonic malakoplakia in a kidney transplant patient

 E-submission
E-submission