Articles
- Page Path
- HOME > J Pathol Transl Med > Volume 48(6); 2014 > Article
-
Brief Case Report
A Case of Mixed Adenoneuroendocrine Carcinoma of the Common Bile Duct: Initially Diagnosed as Cholangiocarcinoma - Soon Wook Lee, In Seok Lee, Yu Kyung Cho, Jae Myung Park, Sang Woo Kim, Myung-Gyu Choi, Kyu Yong Choi, Myung Ah Lee, Tae Ho Hong1, Young Kyoung You1, Eun-Sun Jung2
-
Korean Journal of Pathology 2014;48(6):445-448.
DOI: https://doi.org/10.4132/KoreanJPathol.2014.48.6.445
Published online: December 31, 2014
Departments of Internal Medicine, Seoul St. Mary’s Hospital, The Catholic University of Korea, Seoul, Korea
1Departments of Surgery, Seoul St. Mary’s Hospital, The Catholic University of Korea, Seoul, Korea
2Departments of Pathology, Seoul St. Mary’s Hospital, The Catholic University of Korea, Seoul, Korea
- Corresponding Author: In Seok Lee, M.D. Department of Internal Medicine, Seoul St. Mary’s Hospital, The Catholic University of Korea, 222 Banpo-daero, Seocho-gu, Seoul 137-701, Korea Tel: +82-2-2258-6022, Fax: +82-2-2258-2055, E-mail: isle@catholic.ac.kr
• Received: December 12, 2013 • Revised: January 29, 2014 • Accepted: March 10, 2014
© 2014 The Korean Society of Pathologists/The Korean Society for Cytopathology
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/3.0) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
- A 75-year-old man was admitted to the hospital with painless jaundice that had begun the previous week. He had undergone laparoscopic cholecystectomy four months previously due to gallbladder empyema. He had a history of prostate cancer which had been cured after radical prostatectomy 10 years prior. The patient was taking antihypertensive medication. He was neither a drinker nor a smoker and did not have any specific family history of any other diseases.
- Physical examinations on admission were unremarkable, except for icteric scleras. Laboratory tests showed abnormal liver function tests with aspartate aminotransferase 196 U/L, alanine transaminase 428 U/L, total bilirubin 4.62 mg/dL, direct bilirubin 3.62 mg/dL, and gamma glutamyl transpeptidase 946 IU/L. The cancer antigen 19-9 level was 68.89 U/mL.
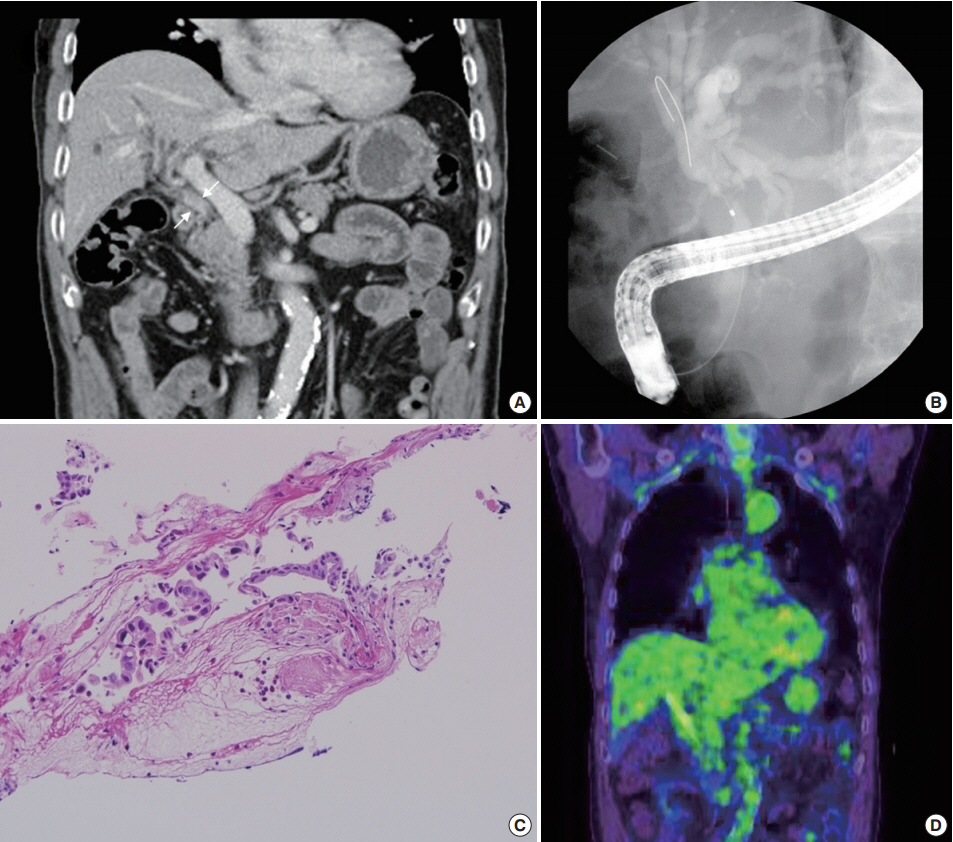
- A abdominal computed tomography revealed intrahepatic duct dilatation and luminal narrowing of the mid CBD with diffuse wall thickening and enhancement (Fig. 1A). Upon endoscopic retrograde cholangiopancreatography (ERCP), there was a luminal narrowing (2 cm in length) in the proximal CBD with proximal ductal dilatation (Fig. 1B). Biopsy with forcep and brush cytology during ERCP revealed a few atypical cells with ulcer detritus that were suggestive of adenocarcinoma (Fig. 1C). On positron emission tomography, no significant abnormal fludeoxyglucose uptake was seen (Fig. 1D).
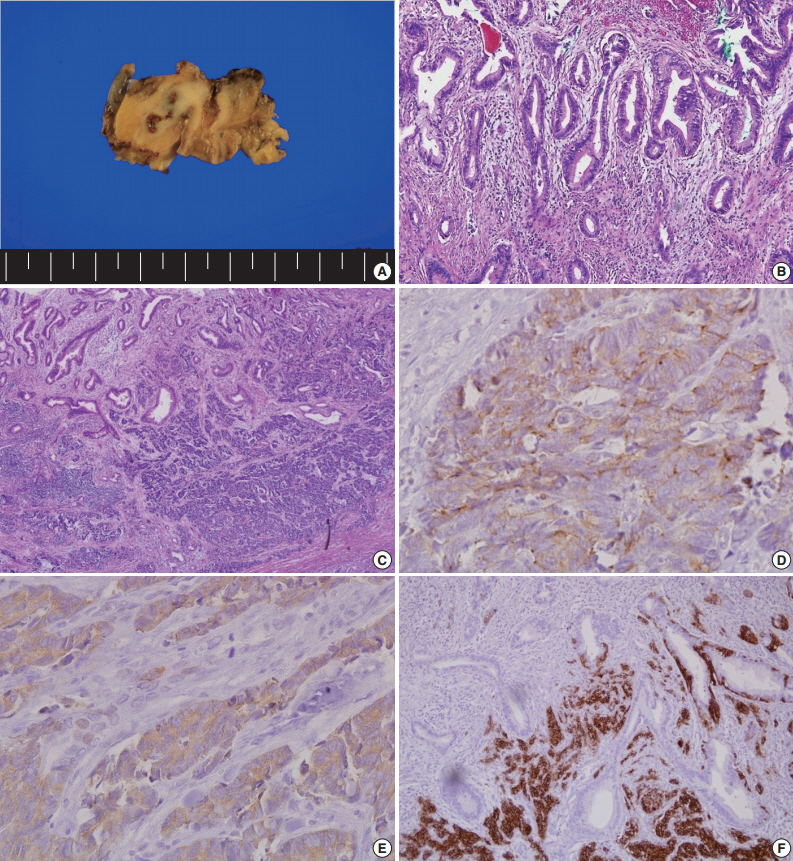
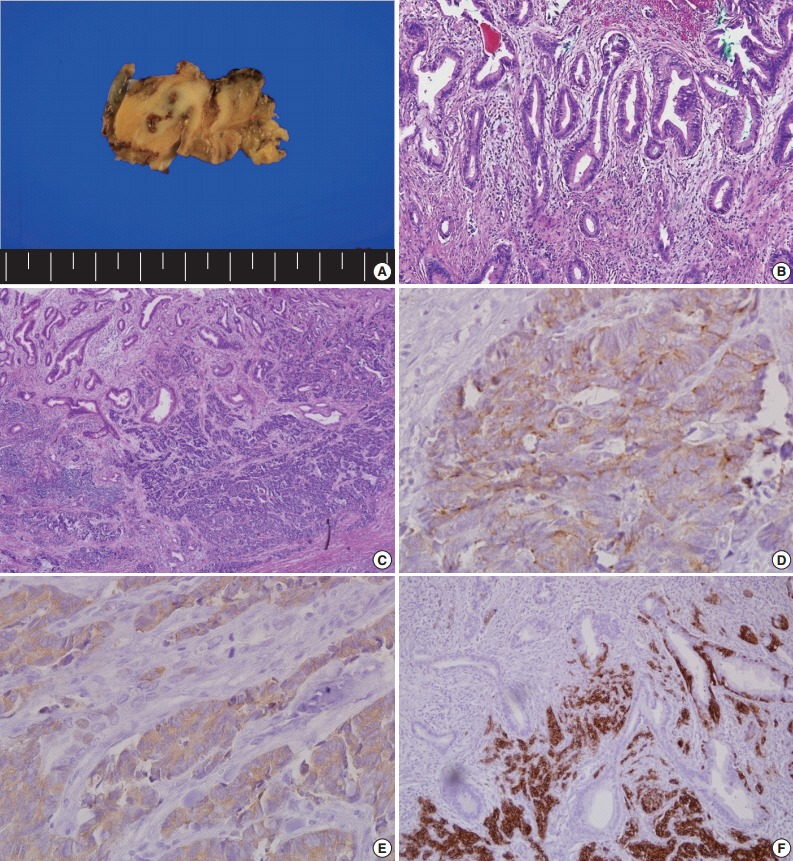
- The patient underwent CBD resection with Roux-en Y choledocojejunostomy and liver wedge resection. On the gross-section after fixation, an infiltrative, whitish tumor was noted at the CBD (Fig. 2A). Microscopically, the surface of the tumor was composed of moderately differentiated adenocarcinoma (Fig. 2B). In contrast, the tumor cells in the deep portion showed an infiltrative growth pattern and were composed of small round cells with hyperchromatic nuclei and scant cytoplasm (Fig. 2C). These infiltrative tumor cells stained positive for chromogranin A (Fig. 2D), synaptophysin (Fig. 2E), and CD56 (Fig. 2F), the markers for NET. Both adenocarcinoma and NET components occupied more than 30% of the tumor volume. The final diagnosis of the patient was MANEC (composite of moderately differentiated adenocarcinoma and small cell carcinoma). There was no lymphatic or vascular invasion.
- Five months after the procedure, the patient was readmitted to the hospital due to recurrent liver abscess. After percutaneous abscess drainage and antibiotic therapy, his condition improved. He continues to be followed-up and has shown no evidence of recurrence for 11 months after the operation.
CASE REPORT
- MANEC is a subtype of NET that contains both gland-forming epithelial tumor cells and neuroendocrine cells [1]. By definition, both components must represent at least 30% of the tumor [1]. NETs that arise from the extrahepatic bile duct are very rare, occupying less than 1% of all NETs [6-8]. MANECs arising from the extrahepatic bile duct are extremely rare [7,8]. These types of tumor have a characteristic histology that includes an adenocarcinoma component at the surface and a neuroendocrine component in the areas of vascular and perineural invasion [3-5,9].
- Because of their rarity and complexity, most MANECs of the bile duct are initially thought to be adenocarcinomas or NETs [3-5]. Our patient was also first diagnosed with cholangiocarcinoma by biopsy via ERCP, and the adenocarcinoma components were localized in the superficial layer.
- An initial pathologic diagnosis of malignant biliary stricture is frequently made following a biopsy of the site. However, due to the limitation involved with obtaining adequate biopsy specimens, ERCP may fail to reach the neuroendocrine component, which is embedded in a deeper portion of the tumor. Therefore, the pathologic diagnosis of MANEC using a surgically resected specimen with immunohistochemical stainining for neuroendocrine markers (chromogranin, synaptophysin, and CD56) may be useful for the correct diagnosis [3].
- Even in surgical specimens of biliary tract cancer, NETs and MANECs may be hidden inside the adenocarcinomatous components. A previous study examined 274 cases of surgically resected biliary tract cancer specimens and reported that 13 of 53 extrahepatic bile duct cancer cases contained neuroendocrine cells and two were newly diagnosed as MANEC [9]. Therefore careful attention and thorough examinations are needed for pathologists to accurately diagnose surgically resected biliary tumors, even when the tumor has been initially determined to be a cholangiocarcinoma via ERCP biopsy.
- In general, the prognosis of biliary MANEC is poor. While the five-year survival rates of NET G1 and NET G2 of the gastroenteropancreatic tract are greater than 70%, the same survival rate of MANEC is about 40% [2]. Disease-related causes of death are most commonly located in the biliary tract (62.2%), while the rarest arise in the rectum (5.2%) [2].
- The treatment algorithm of MANEC is not well established. As with other types of cancer, surgery may be the mainstay of the treatment of MANEC, and adjunctive therapy with chemotherapy, radiotherapy and somatostatin analogues can be considered according to the NEC type. It is also reasonable to treat MANEC in accordance with a more aggressive histology of the tumor [10]. For example, treat like adenocarcinoma in a MANEC with a well differentiated neuroendocrine component and an adenocarcinomatous component, and treat as poorly differentiated NET in a MANEC with poorly differentiated neuroendocrine component [10].
- In conclusion, we present a case of MANEC of the CBD. Most MANEC cases, including this one, are initially diagnosed as cholangiocarcinoma. Therefore, the acquisition of a surgical specimen and thorough investigations by the pathologist are crucial to make the correct diagnosis in order to determine the best treatment and estimate the prognosis in extra-hepatic bile duct cancer.
DISCUSSION
Fig. 1.Study findings before surgery. (A) A portal phase image of a dynamic computed tomography scan shows luminal narrowing of the mid common bile duct (CBD) with diffuse wall thickening and enhancement (white arrows). (B) A filling defect in the CBD with proximal dilatation is noted upon endoscopic retrograde cholangiopancreatography (ERCP). (C) ERCP biopsy of the tumor reveals a few atypical cells with ulcer detritus, suggestive of adenocarcinoma. (D) Positron emission tomography shows no significant abnormal uptake.
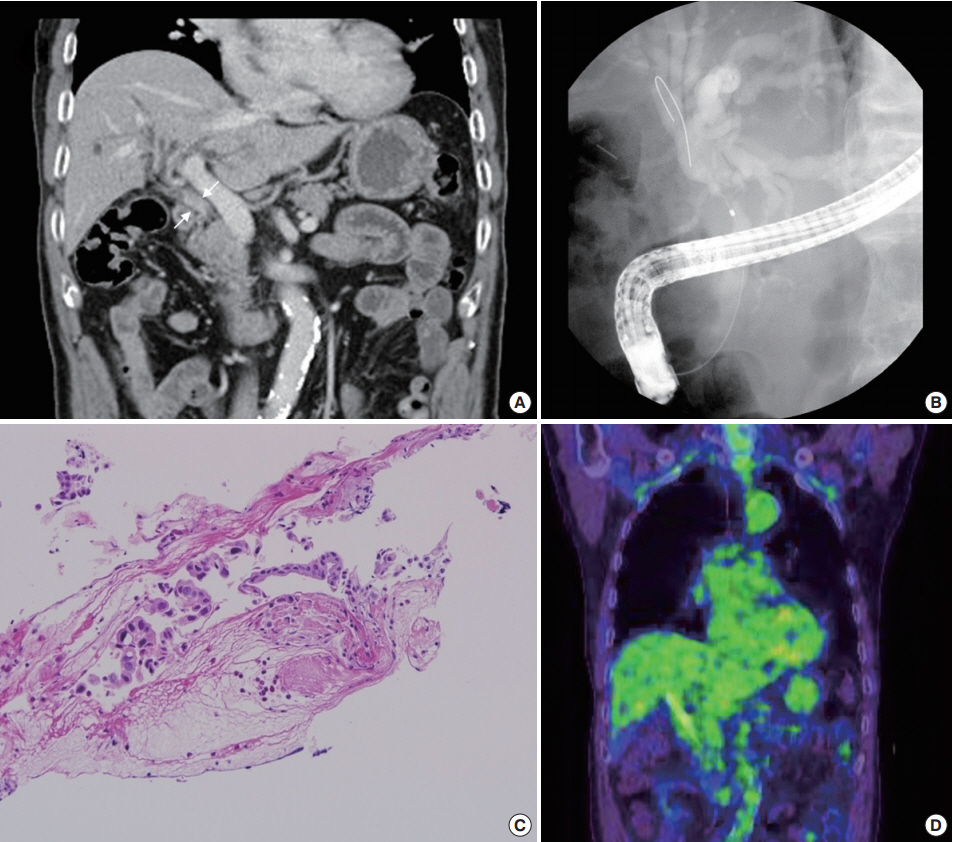

Fig. 2.Pathologic findings after surgery. (A) Gross cross-sectional view of the tumor after fixation. (B) On the surface of the tumor, the tumor cells are composed of moderately differentiated adenocarcinoma. (C) The infiltrative tumor cells are small and round with hyperchromatic nuclei and scant cytoplasm. Immunohistochemical analysis for chromogranin (D), synaptophysin (E), and CD56 (F).

- 1. Bosman FT, Carneiro F, Hruban RH, Theise ND. WHO classification of tumours of the digestive system. 4th ed. Lyon: IARC Press, 2010.
- 2. Gastrointestinal Pathology Study Group of Korean Society of Pathologists, Cho MY, Kim JM, et al. Current trends of the incidence and pathological diagnosis of gastroenteropancreatic neuroendocrine tumors (GEP-NETs) in Korea 2000-2009: multicenter study. Cancer Res Treat 2012; 44: 157-65. ArticlePubMedPMC
- 3. Linder R, Dorfman T, Ben-Ishay O, Kakiashvili E, Velodavsky E, Kluger Y. Mixed neuroendocrine tumor of the common bile duct. JOP 2013; 14: 71-3. PubMed
- 4. Kim J, Lee WJ, Lee SH, et al. Clinical features of 20 patients with curatively resected biliary neuroendocrine tumours. Dig Liver Dis 2011; 43: 965-70. ArticlePubMed
- 5. Baek IY, Chung YJ, Park MK, et al. A case of mixed adenoneuroendocrine carcinoma in common bile duct. Korean J Pancreas Biliary Tract 2012; 17: 56-60.
- 6. Modlin IM, Lye KD, Kidd M. A 5-decade analysis of 13,715 carcinoid tumors. Cancer 2003; 97: 934-59. ArticlePubMed
- 7. Chamberlain RS, Blumgart LH. Carcinoid tumors of the extrahepatic bile duct: a rare cause of malignant biliary obstruction. Cancer 1999; 86: 1959-65. ArticlePubMed
- 8. Sato K, Waseda R, Tatsuzawa Y, et al. Composite large cell neuroendocrine carcinoma and adenocarcinoma of the common bile duct. J Clin Pathol 2006; 59: 105-7. ArticlePubMedPMC
- 9. Harada K, Sato Y, Ikeda H, et al. Clinicopathologic study of mixed adenoneuroendocrine carcinomas of hepatobiliary organs. Virchows Arch 2012; 460: 281-9. ArticlePubMedPDF
- 10. Hervieu V, Scoazec JY. Mixed endocrine tumors. Ann Pathol 2005; 25: 511-28. PubMed
REFERENCES
Figure & Data
References
Citations
Citations to this article as recorded by 

- Long-term survival after chemotherapy combined immunotherapy for recurrent mixed neuroendocrine–non-neuroendocrine neoplasms of the common bile duct
Jia Li, Chunyan Yuan, Yulin Pan, Yuanyuan Yang, Nannan Lai, Xia Sheng
Clinical Journal of Gastroenterology.2025; 18(4): 653. CrossRef - Neuroendocrine carcinoma of the common hepatic duct coexisting with distal cholangiocarcinoma: A case report and review of literature
Fei Chen, Wei-Wei Li, Juan-Fen Mo, Min-Jie Chen, Su-Hang Wang, Shu-Ying Yang, Zheng-Wei Song
World Journal of Gastrointestinal Surgery.2024; 16(5): 1449. CrossRef - Comparison of Metastatic Patterns Among Neuroendocrine Tumors, Neuroendocrine Carcinomas, and Nonneuroendocrine Carcinomas of Various Primary Organs
Hyung Kyu Park, Ghee Young Kwon
Journal of Korean Medical Science.2023;[Epub] CrossRef - Mixed adenoneuroendocrine carcinoma of the distal bile duct: a case report
Takashi Maeda, Kyohei Yugawa, Nao Kinjo, Hiroto Kayashima, Daisuke Imai, Koto Kawata, Shinichiro Ikeda, Keitaro Edahiro, Kazuki Takeishi, Tomohiro Iguchi, Noboru Harada, Mizuki Ninomiya, Shohei Yamaguchi, Kozo Konishi, Shinichi Tsutsui, Hiroyuki Matsuda
Surgical Case Reports.2020;[Epub] CrossRef - The clinical profiles, management, and prognostic factors of biliary mixed neuroendocrine nonneuroendocrine neoplasms
Li-Jia Wen, Jun-Hong Chen, Hong-Ji Xu, Qiong Yu, Yu Deng, Kai Liu
Medicine.2020; 99(50): e23271. CrossRef - Rapidly progressed neuroendocrine carcinoma in the extrahepatic bile duct: a case report and review of the literature
Mariko Kamiya, Naoto Yamamoto, Yuto Kamioka, Hirohide Inoue, Hirokazu Yotsumoto, Masaaki Murakawa, Toru Aoyama, Kota Washimi, Kae Kawachi, Takashi Oshima, Makoto Ueno, Norio Yukawa, Yasushi Rino, Munetaka Masuda, Soichiro Morinaga
Surgical Case Reports.2020;[Epub] CrossRef - Mixed adenoneuroendocrine carcinoma of the hepatic bile duct: a case report and review of the literature
Sulai Liu, Zhendong Zhong, Meng Xiao, Yinghui Song, Youye Zhu, Bo Hu, Zengpeng Sun, Weimin Yi, Chuang Peng
BMC Gastroenterology.2020;[Epub] CrossRef - Mixed adenoendocrine carcinoma in the extrahepatic biliary tract: A case report and literature review
Liang Zhang, Zhengtao Yang, Qing Chen, Mengxia Li, Xiaolu Zhu, Dalong Wan, Haiyang Xie, Shusen Zheng
Oncology Letters.2019;[Epub] CrossRef - Large Cell Neuroendocrine Carcinoma Coexisting with Adenocarcinoma in the Extrahepatic Bile Duct
Masami Yuda, Teruyuki Usuba, Shin Hagiwara, Masahisa Okuma, Hisatoshi Asano, Hitoshi Sakuda, Hiroaki Katagi, Yoshiyuki Furukawa
The Japanese Journal of Gastroenterological Surgery.2018; 51(3): 187. CrossRef - Mixed Adenoneuroendocrine Carcinoma of the Distal Bile Duct
Chiaki Uchida, Yoshikazu Toyoki, Keinosuke Ishido, Daisuke Kudo, Norihisa Kimura, Shinji Tsutsumi, Takuji Kagiya, Toshiro Kimura, Kenichi Hakamada
The Japanese Journal of Gastroenterological Surgery.2017; 50(1): 43. CrossRef - Mixed adenoneuroendocrine carcinoma of the distal bile duct: A case report
Toshiaki Komo, Toshihiko Kohashi, Akira Nakashima, Ichiro Ohmori, Jun Hihara, Hidenori Mukaida, Mayumi Kaneko, Naoki Hirabayashi
International Journal of Surgery Case Reports.2017; 39: 203. CrossRef - Common Hepatic Duct Mixed Adenoneuroendocrine Carcinoma Masquerading as Cholangiocarcinoma
Sali Priyanka Akhilesh, Yadav Kamal Sunder, Tampi Chandralekha, Parikh Samir, Wagle Prasad Kashinath
Case Reports in Gastrointestinal Medicine.2016; 2016: 1. CrossRef
 PubReader
PubReader ePub Link
ePub Link-
 Cite this Article
Cite this Article
- Cite this Article
-
- Close
- Download Citation
- Close
- Figure
A Case of Mixed Adenoneuroendocrine Carcinoma of the Common Bile Duct: Initially Diagnosed as Cholangiocarcinoma


Fig. 1. Study findings before surgery. (A) A portal phase image of a dynamic computed tomography scan shows luminal narrowing of the mid common bile duct (CBD) with diffuse wall thickening and enhancement (white arrows). (B) A filling defect in the CBD with proximal dilatation is noted upon endoscopic retrograde cholangiopancreatography (ERCP). (C) ERCP biopsy of the tumor reveals a few atypical cells with ulcer detritus, suggestive of adenocarcinoma. (D) Positron emission tomography shows no significant abnormal uptake.
Fig. 2. Pathologic findings after surgery. (A) Gross cross-sectional view of the tumor after fixation. (B) On the surface of the tumor, the tumor cells are composed of moderately differentiated adenocarcinoma. (C) The infiltrative tumor cells are small and round with hyperchromatic nuclei and scant cytoplasm. Immunohistochemical analysis for chromogranin (D), synaptophysin (E), and CD56 (F).
Fig. 1.
Fig. 2.
A Case of Mixed Adenoneuroendocrine Carcinoma of the Common Bile Duct: Initially Diagnosed as Cholangiocarcinoma

 E-submission
E-submission