Articles
- Page Path
- HOME > J Pathol Transl Med > Volume 49(4); 2015 > Article
-
Original Article
Cancers with Higher Density of Tumor-Associated Macrophages Were Associated with Poor Survival Rates - Kyong Yeun Jung1,2, Sun Wook Cho1,3, Young A Kim4, Daein Kim3, Byung-Chul Oh5, Do Joon Park1, Young Joo Park1
-
Journal of Pathology and Translational Medicine 2015;49(4):318-324.
DOI: https://doi.org/10.4132/jptm.2015.06.01
Published online: June 17, 2015
1Department of Internal Medicine, Seoul National University College of Medicine, Seoul, Korea
2Department of Internal Medicine, Eulji University School of Medicine, Seoul, Korea
3Department of Internal Medicine, National Medical Center, Seoul, Korea
4Department of Pathology, SMG-SNU Boramae Medical Center, Seoul, Korea
5Lee Gil Ya Cancer and Diabetes Institute, Gachon University Graduate School of Medicine, Incheon, Korea
- Corresponding Author Young Joo Park, MD, PhD Department of Internal Medicine, Seoul National University College of Medicine, 101 Daehak-ro, Jongno-gu, Seoul 110-799, Korea Tel: +82-2-2072-4183 Fax: +82-2-764-2199 E-mail: yjparkmd@snu.ac.kr
© 2015 The Korean Society of Pathologists/The Korean Society for Cytopathology
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/3.0/) which permits unrestricted noncommercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
Abstract
-
Background:
- Macrophages are a component of a tumor’s microenvironment and have various roles in tumor progression and metastasis. This study evaluated the relationships between tumor-associated macrophage (TAM) density and clinical outcomes in 14 different types of human cancers.
-
Methods:
- We investigated TAM density in human tissue microarray sections from 14 different types of human cancers (n = 266) and normal thyroid, lung, and breast tissues (n = 22). The five-year survival rates of each cancer were obtained from the 2011 Korea Central Cancer Registry.
-
Results:
- Among 13 human cancers, excluding thyroid cancer, pancreas, lung, and gallbladder cancers had the highest density of CD163-positive macrophages (7.0±3.5%, 6.9±7.4%, and 6.9 ± 5.5%, respectively). The five-year relative survival rates of these cancers (pancreas, 8.7%; lung, 20.7%; gallbladder, 27.5%) were lower than those of other cancers. The histological subtypes in thyroid cancer exhibited significantly different CD163-positive macrophages densities (papillary, 1.8 ± 1.6% vs anaplastic, 22.9 ± 17.1%; p < .001), but no significant difference between histological subtypes was detected in lung and breast cancers. Moreover, there was no significant difference in CD163-positive macrophages densities among the TNM stages in lung, breast, and thyroid cancers.
-
Conclusions:
- Cancers with higher TAM densities (pancreas, lung, anaplastic thyroid, and gallbladder) were associated with poor survival rate.
- Study subjects and tissue microarrays
- We purchased 14 different type of human cancer human tissue microarray sections (lung, 49; breast, 49; thyroid, 10; pancreas, 10; gallbladder, 9; larynx, 9; esophagus, 10; liver, 10; cervix, 10; ovary, 10; stomach, 10; prostate, 9; kidney, 9; and endometrium, 9 sections) and normal tissues (lung, 9; breast, 8 sections) from SuperBioChips Laboratories (Seoul, Korea). The supplier provided the clinical information associated with each section, including age at surgery; gender; pathologic diagnosis; TNM staging for cancers; survival/death follow-up result for lung and breast cancers; and estrogen receptor (ER), progesterone receptor (PR), p53, and C-erbB2 expression for breast cancers. Thyroid microarrays contained normal thyroid (n=5), papillary thyroid cancer (PTC; n=35), and anaplastic thyroid cancer (ATC; n=18), and one of the thyroid microarray slides was used in a previous study [14]. Clinical and pathologic data for the thyroid samples were obtained from medical records at Seoul National University Hospital and SMG-SNU Boramae Medical Center. The five-year cancer relative survival rates were obtained from the 2011 annual report of cancer statistics in Korea [15].
- Immunohistochemistry on tissue array blocks
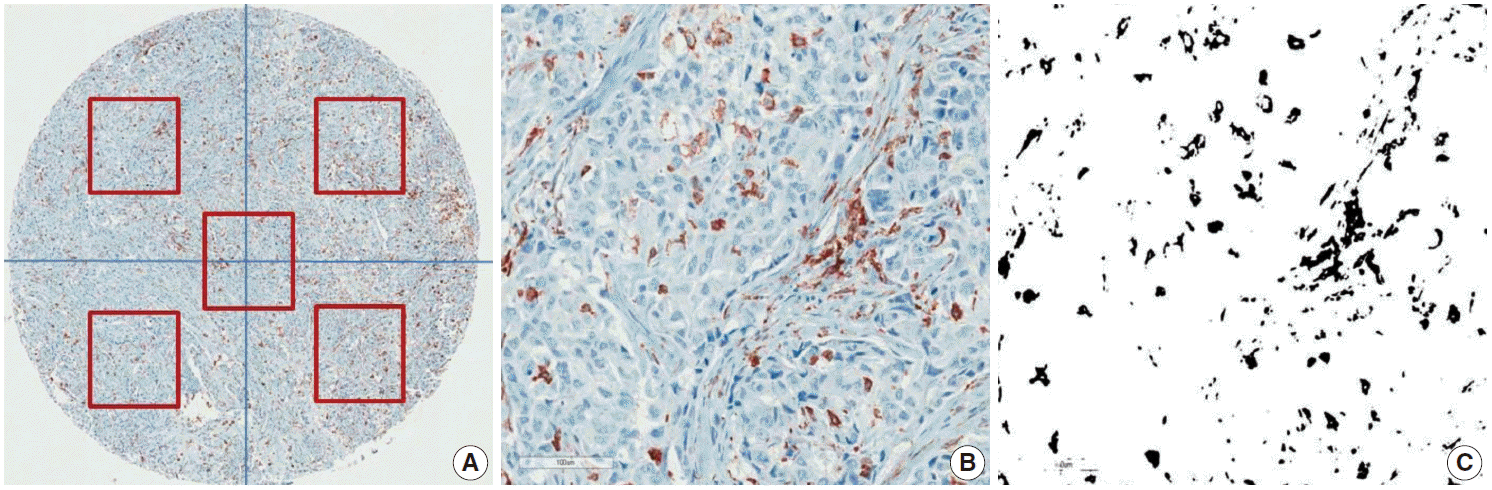
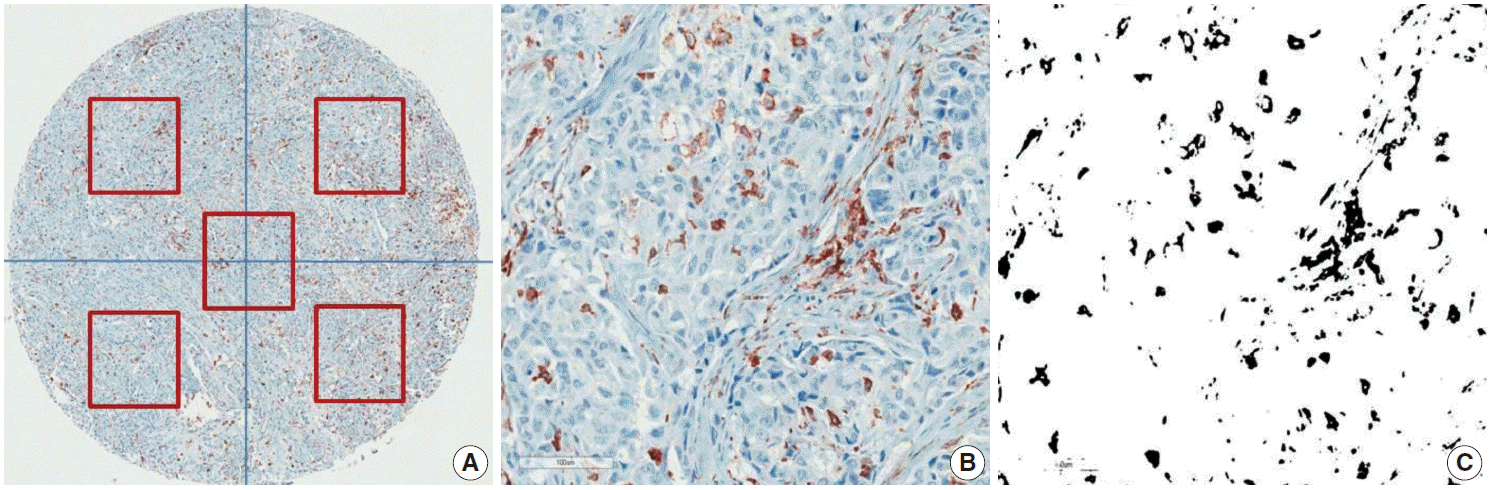
- CD68 and CD163 were used as TAM markers. Immunohistochemical (IHC) staining for CD68 and CD163 was performed using the BenchMark XT Slide Preparation System (Ventana Medical Systems, Tucson, AZ, USA) and CD68 (ready-to-use, 514H12, Novocastra, Newcastle upon Tyne, UK) and CD163 (1:200, 10D6, ER2, Novocastra). The proportion of CD163-positive area in each tumor was evaluated after IHC staining (Fig. 1). We divided the area of each tissue core into quarters and a central area and randomly chose all five fragments to determine the positive stain proportion (Fig. 1A). Areas of fibrosis or tumor necrosis among the randomly chosen fragments were excluded. We used a color deconvolution plug-in for Image J software to identify the positive stains and to calculate the percent CD163-positive area (Fig. 1B, C). TAM density was determined by calculating the average CD163-positive area (%) at a minimum of four different sites in each tissue.
- Statistical analysis
- The CD163-positive macrophages density (%) is presented as mean±standard deviation. Differences in CD163-positive macrophages densities among TNM cancer stages, histological subtypes, and pathological characteristics were determined using one-way analysis of variance (ANOVA) or Student’s t test. Statistical significance is indicated by a p-value less than .05. All data were analyzed with IBM SPSS Statistics ver. 20.0 (SPSS Inc., Chicago, IL, USA).
- Ethics statement
- This study was approved by the Institutional Review Boards of Seoul National University Hospital (1107-060-369) and SMG-SNU Boramae Medical Center (06-2010-176). The need for informed consent was waived by those boards.
MATERIALS AND METHODS
- CD163-positive macrophage densities and prognoses in human cancers
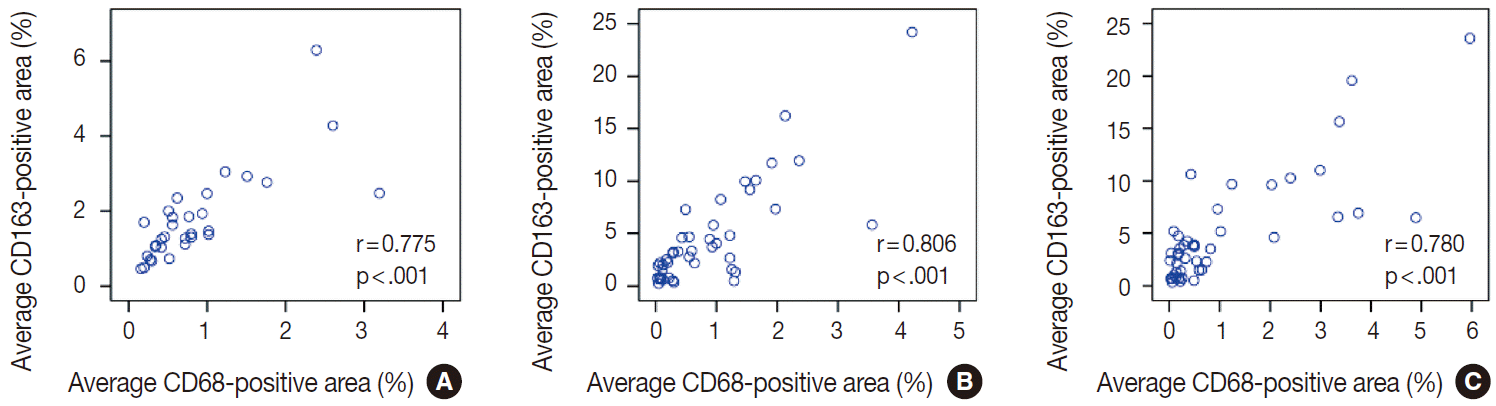
- The average CD163-positive area was significantly correlated with the average CD68-positive area (%) in thyroid (r=0.775, p<.001) (Appendix 1A), breast (r=0.806, p<.001) (Appendix 1B), and lung (r=0.780, p<.001) (Appendix 1C) cancers. Therefore, our remaining IHC assessments were performed with CD163 data.
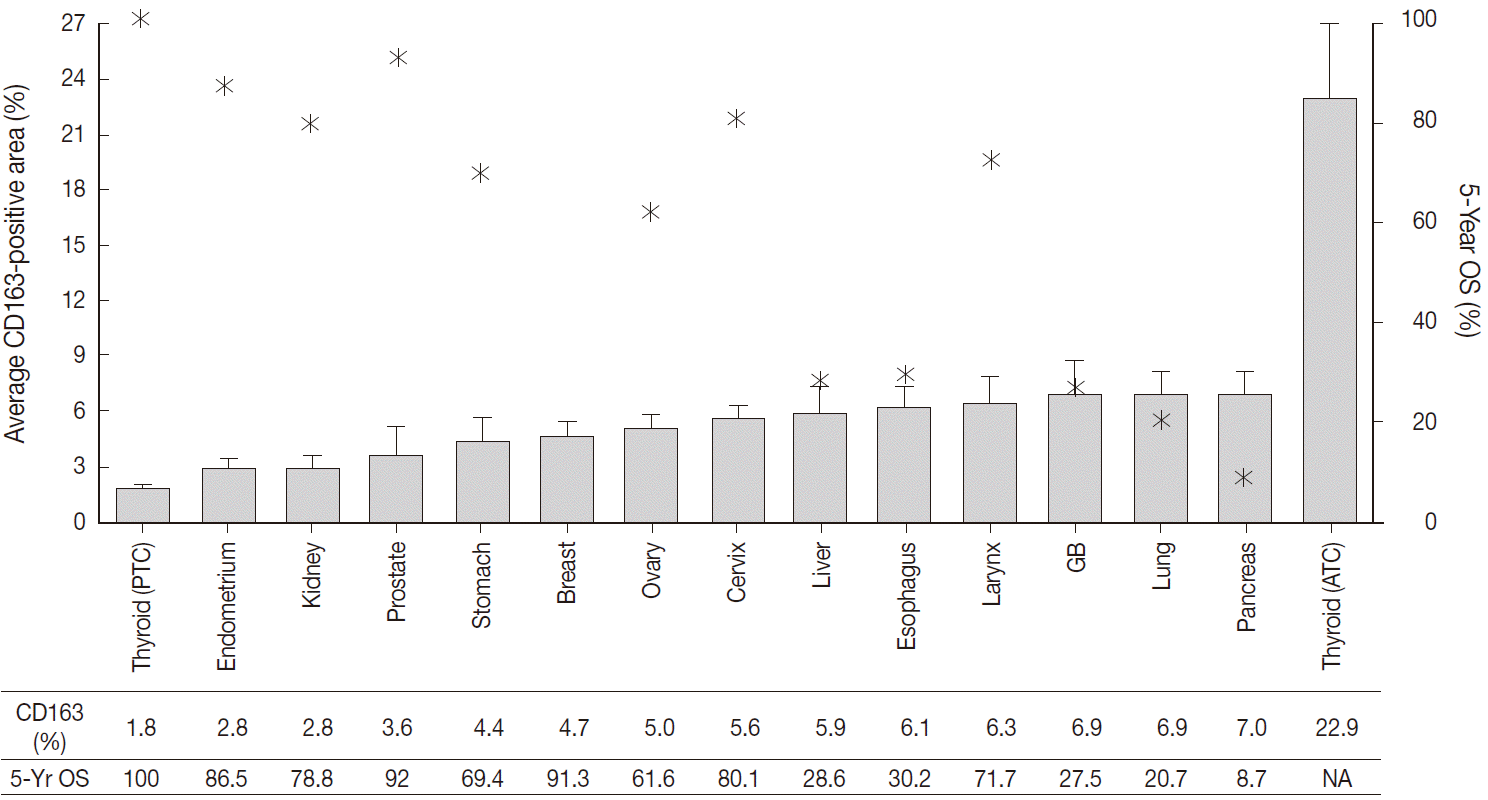
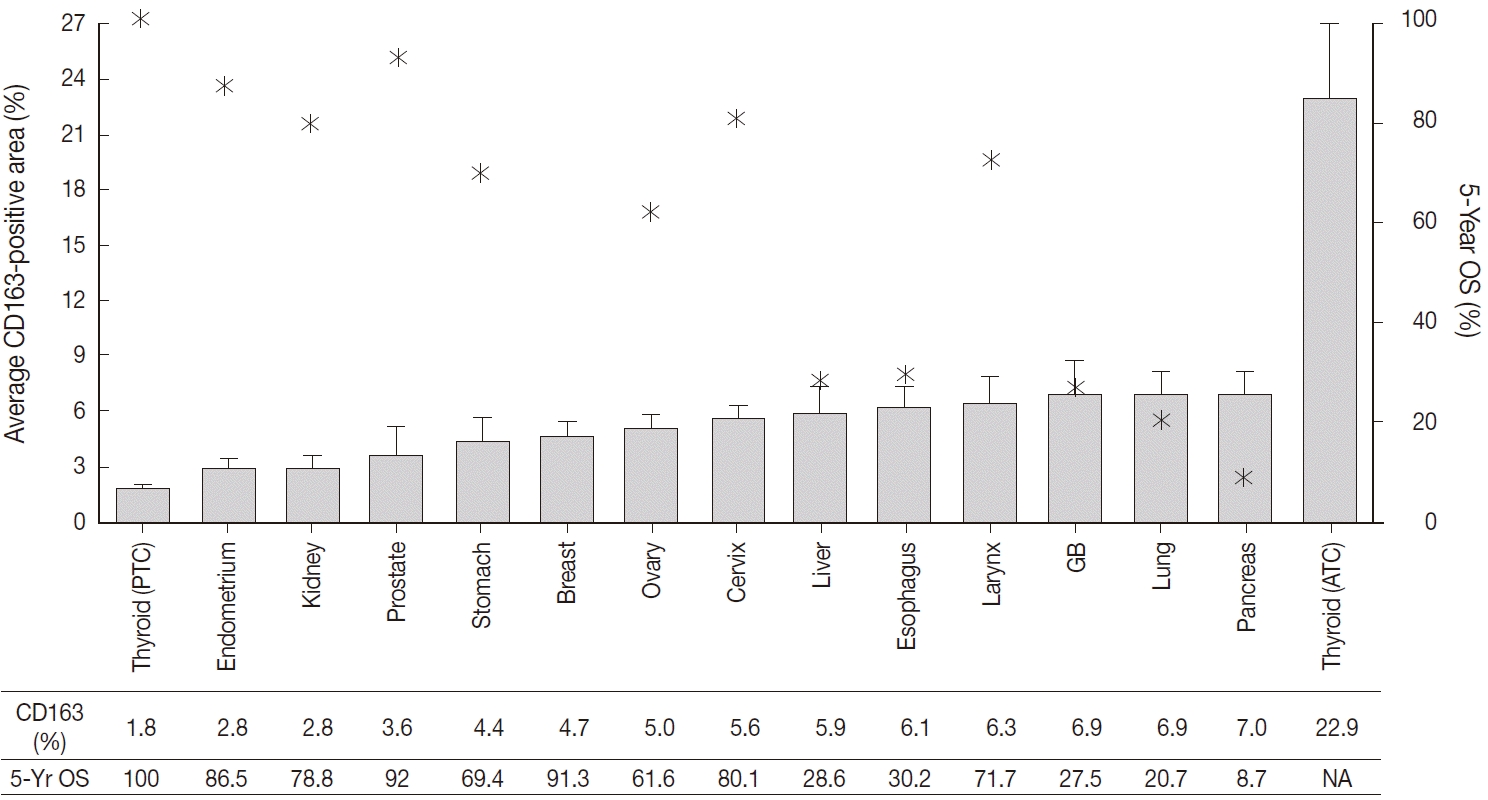
- The CD163-positive macrophages densities and the five-year relative survival rates in the 14 different types of human cancers are summarized in Fig. 2. Excluding thyroid cancer, of the 13 other human cancers, pancreas, lung, and gallbladder cancers had the highest density of CD163-positive macrophages (7.0±3.5%, 6.9±7.4%, and 6.9±5.5%, respectively). In contrast, endometrium, prostate, and kidney cancers had the lowest densities of CD163-positive macrophages (3.6±4.6%, 2.8±2.5%, and 2.8±1.8%, respectively). Interestingly, among the tested cancers, five-year relative survival rate (%) was inversely correlated with CD163-positive macrophages density. The five-year overall survival rates of the cancers with highest TAM densities (pancreas, 8.7%; lung, 20.7%; gallbladder and biliary tract, 27.5%) were lower than those of the cancers with the lowest TAM densities (endometrial, 86.5%; kidney, 78.8%; prostate, 92%).
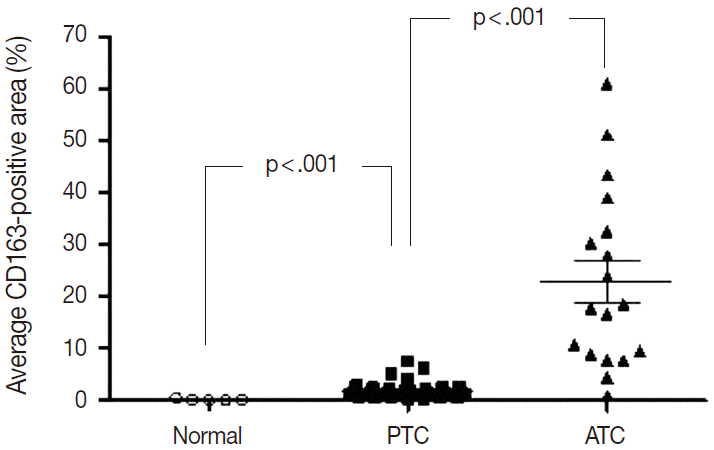
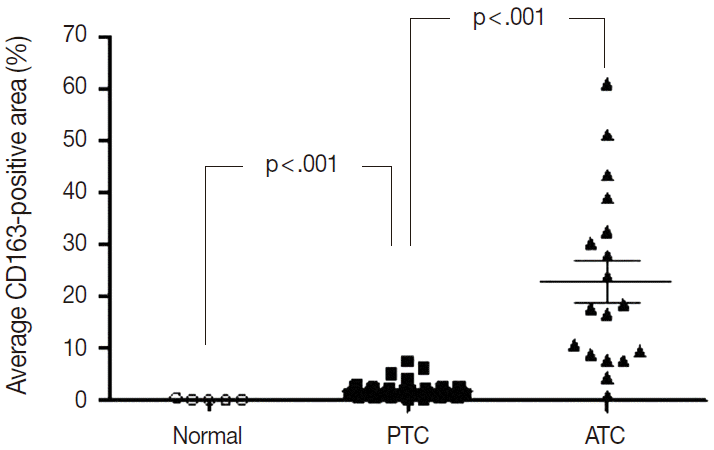
- Of the 14 cancers assessed, thyroid cancer exhibited an extremely wide range of TAM densities that varied according to pathologic subtype (Fig. 3). The ATC cases had the highest density of CD163-positive macrophages (22.9±17.1%), whereas the PTC cases had the lowest CD163-positive macrophage density (1.8±1.3%).
- Clinicopathological correlations with TAM density in thyroid, lung, and breast cancers
- To investigate the role of TAMs in human cancers, we evaluated the correlations between TAM density and clinicopathological features in lung, breast, and PTC cancers. The CD163-positive macrophage density was significantly higher in cancer tissues than normal tissues in breast (4.7±4.5% vs 0.6±0.2%; p<.001), lung (6.9±7.4% vs 1.6±1.6%; p<.001), and thyroid (PTC, 1.8±1.3% vs normal, 0.2±0.2%; p<.001) tissues (Fig. 3). Among the histological subtypes of lung (adenocarcinoma, 5.8±4.4% vs squamous cell carcinoma, 5.9±4.5% vs large cell carcinoma, 8.7±7.3%; p=.428) and breast cancers (infiltrating ductal carcinoma, 4.0±3.4% vs medullary carcinoma, 9.3±3.4%; p=.055), there were no significant differences in CD163-positive macrophages density (Appendix 2). Differences among TNM stages were evaluated by assessing adenocarcinoma and squamous cell carcinoma of lung cancer, infiltrating ductal carcinoma of breast cancer, and PTC. There were no significant differences in CD163-positive macrophage density associated with TNM stage in lung (p=.821), breast (p=.060), or thyroid (PTC, p=.943) cancer (Appendix 2). Within the breast cancer samples, several prognostic markers including ER, PR, p53, and C-erbB2 were analyzed. The ER-positive (3.4±4.5% vs 5.6±4.8%; p=.182) and PR-positive (3.7±4.4% vs 5.4±4.9%; p=.319) breast cancers showed non-significant trends toward lower CD163-positive macrophages density than those in the ER- or PR-negative breast cancers. In contrast, p53-positive (5.9±5.9% vs 4.2±3.6%; p=.269) and C-erbB2-positive (6.4±6.8% vs 4.4±3.8%; p=.233) breast cancer showed nonsignificant trends toward higher CD163-positive macrophage density than those in p53- or C-erbB2-negative breast cancers.
RESULTS
- Our results demonstrate that CD163-positive macrophage density is inversely correlated with five-year cancer survival rate in a variety of human cancers. This finding suggests the presence of a potential pro-tumorigenic role of TAMs in some human cancers. Among the 14 different types of human cancers assessed, ATC, the most aggressive cancer in humans [16,17], had the highest CD163-positive macrophages density (23%), which was markedly higher than the second highest TAM density (7%) detected in the pancreas cancer samples. ATC accounts for up to 2% of thyroid cancers and is characterized by an extremely poor survival rate with a one-year average survival rate of 20% and a median survival duration of five months [18,19]. In contrast, the most common well-differentiated thyroid cancer, PTC, had the lowest CD163-positive macrophages density (1.8%). PTC is associated with lower recurrence and mortality rates [20] than other solid and relatively indolent tumors and has a five-year survival rate higher than 90% [15]. Since the differentiation of pathologic subtypes is distinctive and survival rates markedly differ among subtypes, our observation of a striking difference in CD163-positive macrophages density between two thyroid cancer subtypes led us to hypothesize that TAM density is a potential prognostic factor in human cancers.
- In contrast, histological subtypes of lung and breast cancers did not exhibit significant differences in CD163-positive macrophage density. Correlations between TAM density and clinicopathological features have been reported in ductal and lobular carcinomas in breast cancer [21,22] and in adenocarcinoma [23,24] and non-small cell [25,26] lung cancers. Nonetheless, only a few reports have compared TAM density based on histological subtype. Although our data did not detect a significant difference related to histological subtype in lung or breast cancers, the number of each subtype in our sample population was limited. Therefore, we recommend a larger-scale study of TAM density in a variety of histological subtypes.
- In this study, there were no significant differences in CD163-positive macrophage density among TNM stages in lung, breast, or thyroid cancers. In addition, cancer tissues showed higher CD163-positive macrophage densities than those in normal tissues in lung, breast, and thyroid samples. In breast cancer, there were trends toward negative correlations of TAM density with ER and PR expression, whereas there were trends toward positive correlations of TAM density with p53 and C-erbB2 expression. Previous reports indicate that TAM infiltration is inversely correlated with ER expression in breast cancer [7,27]. However, this correlation might vary with TAM location [22]. Medrek et al. [22] reported that a dense infiltration of CD163-positive macrophages in tumor stroma was associated with ER- and PR-negativity, but there was no such association in tumor nest. Our study results show a non-significant trend toward negative correlations between ER and PR expressions and density of CD163-positive macrophages. Due to data limitations, we could not assess such correlations according to infiltration location.
- Selecting the most appropriate marker for TAM assessment can be challenging. Originally, a TAM was seen as a residual macrophage within the tumor microenvironment, and the biological characteristics of TAMs have been shown to be anti-tumorigenic, cytotoxic, or pro-tumorigenic [1,28]. Furthermore, macrophages can undergo polarization and change from an M1-macrophage (classically activated, pro-inflammatory) to an M2-macrophage (alternatively activated, anti-inflammatory or regenerative) [29]. Collectively, these findings indicate the difficulty in choosing a cancer-specific TAM marker. In the beginning of this study, we performed IHC investigations using both CD68, a pan macrophage marker [29], and CD163, a more specific marker for M2-macrophages [30]. Those investigations revealed that the CD163-positive areas were significantly positively correlated with the CD68-positive areas in lung, breast, and thyroid cancers; thus, our remaining IHC assessments were performed with CD163 data.
- Previous clinical studies have shown a correlation between TAM density and cancer prognosis. Most studies have shown a significant correlation between TAM density and poor prognosis, especially among breast, thyroid, and lung cancers [5,7,8,22]. However, several studies have reported that TAM density is associated with good prognosis. Such contradictory results might be due to differences in the number, grade, stage, or size of tumors among the studies. In addition, previous clinical studies have used various methods to assess TAM infiltration. The use of different approaches could have also contributed to the inconsistent results. Although the sample size for each cancer subtype in our study population was small, the results in this study comparing TAM density in various human cancers are significant; therefore, further studies with larger sample sizes are warranted.
- In summary, we detected a trend toward an inverse correlation between CD163-positive macrophage density and five-year survival rate in 14 different types of human cancers. In particular, PTC and ATC clearly showed an inverse correlation between TAM density and prognosis. Our results suggest that TAM density is a potential biomarker of poor prognosis in human cancers.
DISCUSSION
Acknowledgments
- 1. Coussens LM, Werb Z. Inflammation and cancer. Nature 2002; 420: 860-7. ArticlePubMedPMCPDF
- 2. Cho SW. Interactions between immune cells and tumor cells. J Korean Thyroid Assoc 2013; 6: 96-100. Article
- 3. Zhang QW, Liu L, Gong CY, et al. Prognostic significance of tumor-associated macrophages in solid tumor: a meta-analysis of the literature. PLoS One 2012; 7: e50946. Article
- 4. Bingle L, Brown NJ, Lewis CE. The role of tumour-associated macrophages in tumour progression: implications for new anticancer therapies. J Pathol 2002; 196: 254-65. ArticlePubMed
- 5. Kim S, Cho SW, Min HS, et al. The expression of tumor-associated macrophages in papillary thyroid carcinoma. Endocrinol Metab (Seoul) 2013; 28: 192-8. ArticlePubMedPMC
- 6. Zeni E, Mazzetti L, Miotto D, et al. Macrophage expression of interleukin-10 is a prognostic factor in nonsmall cell lung cancer. Eur Respir J 2007; 30: 627-32. ArticlePubMed
- 7. Campbell MJ, Tonlaar NY, Garwood ER, et al. Proliferating macrophages associated with high grade, hormone receptor negative breast cancer and poor clinical outcome. Breast Cancer Res Treat 2011; 128: 703-11. ArticlePubMedPDF
- 8. Sato S, Hanibuchi M, Kuramoto T, et al. Macrophage stimulating protein promotes liver metastases of small cell lung cancer cells by affecting the organ microenvironment. Clin Exp Metastasis 2013; 30: 333-44. ArticlePubMedPDF
- 9. Forssell J, Oberg A, Henriksson ML, Stenling R, Jung A, Palmqvist R. High macrophage infiltration along the tumor front correlates with improved survival in colon cancer. Clin Cancer Res 2007; 13: 1472-9. ArticlePubMedPDF
- 10. Ohno S, Inagawa H, Dhar DK, et al. The degree of macrophage infiltration into the cancer cell nest is a significant predictor of survival in gastric cancer patients. Anticancer Res 2003; 23: 5015-22. PubMed
- 11. Welsh TJ, Green RH, Richardson D, Waller DA, O’Byrne KJ, Bradding P. Macrophage and mast-cell invasion of tumor cell islets confers a marked survival advantage in non-small-cell lung cancer. J Clin Oncol 2005; 23: 8959-67. ArticlePubMed
- 12. Kawai O, Ishii G, Kubota K, et al. Predominant infiltration of macrophages and CD8(+) T cells in cancer nests is a significant predictor of survival in stage IV nonsmall cell lung cancer. Cancer 2008; 113: 1387-95. ArticlePubMed
- 13. Ohno S, Ohno Y, Suzuki N, et al. Correlation of histological localization of tumor-associated macrophages with clinicopathological features in endometrial cancer. Anticancer Res 2004; 24: 3335-42. PubMed
- 14. Cho SW, Kim YA, Sun HJ, et al. Therapeutic potential of Dickkopf-1 in wild-type BRAF papillary thyroid cancer via regulation of betacatenin/E-cadherin signaling. J Clin Endocrinol Metab 2014; 99: E1641-9. PubMed
- 15. Jung KW, Won YJ, Kong HJ, Oh CM, Lee DH, Lee JS. Cancer statistics in Korea: incidence, mortality, survival, and prevalence in 2011. Cancer Res Treat 2014; 46: 109-23. ArticlePubMedPMCPDF
- 16. Cornett WR, Sharma AK, Day TA, et al. Anaplastic thyroid carcinoma: an overview. Curr Oncol Rep 2007; 9: 152-8. ArticlePubMedPDF
- 17. Are C, Shaha AR. Anaplastic thyroid carcinoma: biology, pathogenesis, prognostic factors, and treatment approaches. Ann Surg Oncol 2006; 13: 453-64. ArticlePubMedPDF
- 18. Smallridge RC, Copland JA. Anaplastic thyroid carcinoma: pathogenesis and emerging therapies. Clin Oncol (R Coll Radiol) 2010; 22: 486-97. ArticlePubMedPMC
- 19. Haymart MR, Banerjee M, Yin H, Worden F, Griggs JJ. Marginal treatment benefit in anaplastic thyroid cancer. Cancer 2013; 119: 3133-9. ArticlePubMedPMC
- 20. Lin JD, Chen ST, Hsueh C, Chao TC. A 29-year retrospective review of papillary thyroid cancer in one institution. Thyroid 2007; 17: 535-41. ArticlePubMed
- 21. Murri AM, Hilmy M, Bell J, et al. The relationship between the systemic inflammatory response, tumour proliferative activity, T-lymphocytic and macrophage infiltration, microvessel density and survival in patients with primary operable breast cancer. Br J Cancer 2008; 99: 1013-9. ArticlePubMedPMCPDF
- 22. Medrek C, Pontén F, Jirström K, Leandersson K. The presence of tumor associated macrophages in tumor stroma as a prognostic marker for breast cancer patients. BMC Cancer 2012; 12: 306.ArticlePubMedPMCPDF
- 23. Ohtaki Y, Ishii G, Nagai K, et al. Stromal macrophage expressing CD204 is associated with tumor aggressiveness in lung adenocarcinoma. J Thorac Oncol 2010; 5: 1507-15. ArticlePubMed
- 24. Zhang BC, Gao J, Wang J, Rao ZG, Wang BC, Gao JF. Tumor-associated macrophages infiltration is associated with peritumoral lymphangiogenesis and poor prognosis in lung adenocarcinoma. Med Oncol 2011; 28: 1447-52. ArticlePubMedPDF
- 25. Ma J, Liu L, Che G, Yu N, Dai F, You Z. The M1 form of tumor-associated macrophages in non-small cell lung cancer is positively associated with survival time. BMC Cancer 2010; 10: 112.ArticlePubMedPMCPDF
- 26. Al-Shibli K, Al-Saad S, Donnem T, Persson M, Bremnes RM, Busund LT. The prognostic value of intraepithelial and stromal innate immune system cells in non-small cell lung carcinoma. Histopathology 2009; 55: 301-12. ArticlePubMed
- 27. Steele RJ, Eremin O, Brown M, Hawkins RA. Oestrogen receptor concentration and macrophage infiltration in human breast cancer. Eur J Surg Oncol 1986; 12: 273-6. PubMed
- 28. Pollard JW. Tumour-educated macrophages promote tumour progression and metastasis. Nat Rev Cancer 2004; 4: 71-8. ArticlePubMedPDF
- 29. Heusinkveld M, van der Burg SH. Identification and manipulation of tumor associated macrophages in human cancers. J Transl Med 2011; 9: 216.ArticlePubMedPMCPDF
- 30. Ambarus CA, Krausz S, van Eijk M, et al. Systematic validation of specific phenotypic markers for in vitro polarized human macrophages. J Immunol Methods 2012; 375: 196-206. ArticlePubMed
REFERENCES
Appendix
Appendix 1.
Appendix 2.
| Charaacteristic | No. | CD163 (%) | p-valuea |
|---|---|---|---|
| Lung | |||
| Histology | |||
| Adenocarcinoma | 19 | 5.8 ± 4.4 | .428 |
| SQCC | 20 | 5.9 ± 4.5 | |
| Large cell carcinoma | 4 | 8.7 ± 7.3 | |
| SCC | 2 | 1.0 ± 0.5 | |
| Mixed carcinoma | 4 | 5.0 ± 2.3 | |
| Stageb | |||
| I | 16 | 5.7 ± 4.4 | .821 |
| II | 12 | 6.6 ± 5.2 | |
| III | 10 | 5.5 ± 3.8 | |
| IV | 1 | 2.4 | |
| Survival | |||
| Alive | 15 | 3.6 ± 3.4 | .104 |
| Death | 15 | 5.9 ± 3.9 | |
| Breast | |||
| Infiltrating ductal | 45 | 4.0 ± 3.4 | .055 |
| Medullary | 2 | 9.3 ± 3.4 | |
| Sarcomatoid | 1 | 1.9 | |
| Metaplastic | 1 | 10.1 | |
| Stageb | |||
| IIA | 13 | 5.3 ± 4.9 | .060 |
| IIB | 10 | 4.2 ± 2.4 | |
| IIIA | 12 | 1.8 ± 1.4 | |
| IIIB | 10 | 4.5 ± 2.3 | |
| Alive | 30 | 4.2 ± 3.7 | .726 |
| Death | 6 | 3.7 ± 2.5 | |
| Thyroid | |||
| PTC | 35 | 1.8 ± 1.3 | < .001 |
| ATC | 18 | 22.9 ± 17.1 | |
| Stageb | |||
| I | 3 | 1.4 ± 0.6 | .943 |
| II | 3 | 1.7 ± 1.1 | |
| III | 21 | 1.8 ± 1.2 | |
| IV | 4 | 1.8 ± 0.8 | |
| Alive | 15 | 1.8 ± 1.5 | .241 |
| Death | 2 | 3.3 ± 2.8 |
SQCC, squamous cell carcinoma; SCC; small cell carcinoma, PTC; papillary thyroid carcinoma, ATC; anaplastic thyroid carcinoma.
ap-value by ANOVA or student t test;
bTNM stage and survival was classified in adenocarcinoma and squamous cell carcinoma (lung), infiltrating ductal carcinoma (breast) and papillary thyroid carcinoma (thyroid).
Figure & Data
References
Citations

- Caveolin-1 drives immunosuppression in esophageal squamous cell carcinoma by enhancing exosome secretion and macrophage M2 polarization via inhibition of MVB autophagic degradation
Ke-Rong Zhai, Pei-Lin Zhao, Zi-Han Wang, Hai-Ming Feng, Zhen-Qing Li, Hui-Rong Huang, Ning Yang, Zhi-Peng Su, Bai-Qiang Cui, Tie-Niu Song, Bin Li
Journal of Translational Medicine.2026;[Epub] CrossRef - Cancer Stemness and Dedifferentiation in Anaplastic Thyroid Carcinoma: Insights into a Multigenic, Microenvironmental Network and the Role of CD44
Benny Mosoane, Brandon S. Jackson, Michelle McCabe, Tebogo Marutha, Zodwa Dlamini
Biomedicines.2026; 14(2): 453. CrossRef - The double-edged sword role of tumor-associated macrophages: preventing or causing resistance to immunotherapy
Raheleh Roudi, Behnaz Beikzadeh, Babak Beikzadeh, Sima Kalantari, Navid Sobhani
Journal of Experimental & Clinical Cancer Research.2026;[Epub] CrossRef - Innovative immunotherapeutic strategies for thyroid cancer: challenges and opportunities
Jinpeng Cui, Huali Fang, Changhua Qu
Clinical and Translational Oncology.2026;[Epub] CrossRef - The role of lenvatinib in different types of thyroid cancer
Catarina REGALA, Tiago N. SILVA, Valeriano LEITE
Minerva Endocrinology.2026;[Epub] CrossRef - A MERTK-Targeting Antibody–Drug Conjugate Selectively Depletes M2 Tumor-Associated Macrophages and MERTK-Expressing Cancer Cells
Shugaku Takeda, Subhasree Sridhar, Daniel Schefer, Celia Andreu-Agullo, Pui C. Lo, Minhee Lee, Robert Busby, David M. Darst, Anne Assmus, Suresh Anaganti, Nils Halberg, Benjamin N. Ostendorf, Ivo C. Lorenz, Sohail F. Tavazoie, Masoud F. Tavazoie, Isabel K
Cancer Research.2026; 86(8): 2024. CrossRef - Immune cells in thyroid adenoma and carcinoma: uncovering a hidden value of assessing tumor-host interplay and its potential application in thyroid cytopathology
Iryna Omelianenko, Nazarii Kobyliak, Tetyana Falalyeyeva, Oleksii Seleznov, Pavlina Botsun, Lyudmila Ostapchenko, Oleksandr Korotkyi, Liudmyla Domylivska, Olena Tsyryuk, Galyna Mykhalchyshyn, Tetiana Shapochka, Oksana Sulaieva
Frontiers in Molecular Biosciences.2025;[Epub] CrossRef - Research Progress on the Immunological Correlation Between Papillary Thyroid Carcinoma and Hashimoto’s Thyroiditis
Digui Fang, Limei Zhou, Biao Zheng, Ran Wang
Journal of Immunology Research.2025;[Epub] CrossRef - Tumour-associated macrophages in human meningiomas
Rahmina Meta, Henrik Sahlin Pettersen, Sofie Eline Tollefsen, Borgny Ytterhus, Øyvind Olav Salvesen, Wenche Sjursen, Sverre Helge Torp, Jianhong Zhou
PLOS One.2025; 20(5): e0319960. CrossRef - Turning tumor microenvironmental foes to friends: A new opportunity for thyroid cancer therapy and redifferentiation
Liya Zhu, Xiuli Jing, Byeong-Cheol Ahn
Oral Oncology.2025; 168: 107513. CrossRef - Unveiling Macrophage Content as a Predictive Biomarker for Intraoperative ICG Imaging Efficacy in Lung Cancer
Yue Yan, Jiahui Mi, Yun Li, Guanchao Jiang, Yimeng Zhang, Yanguo Liu, Hui Zhao, Jianfeng Li, Xing Yang, Jun Wang, Fan Yang, Kezhong Chen, Yiguang Wang, Jian Zhou
Advanced Science.2025;[Epub] CrossRef - Imaging Through Scattering Tissue Based on NIR Multispectral Image Fusion Technique
Nisan Atiya, Amir Shemer, Ariel Schwarz, Yevgeny Beiderman, Yossef Danan
Sensors.2025; 25(16): 4977. CrossRef - Molecular landscape of colorectal cancer liver metastasis: Tumor microenvironment heterogeneity and driver inference
Ailing Zhang, Yi Zhang, Jiayue Xu, Rui Zhu, Tingming Liang, Li Guo
Critical Reviews in Oncology/Hematology.2025; 216: 104946. CrossRef - Precision engineering of macrophage reprogramming with RNA interference-loaded lipid nanoparticles: a game-changer in cancer immunotherapy
Sezen Gül, Juliette Vergnaud, François Fay, Elias Fattal
Drug Delivery and Translational Research.2025;[Epub] CrossRef - Oncogenic mutation-driven metabolism-immunity regulatory axis: Potential prospects for thyroid cancer precision therapy
Tingting Zhang, Hengtong Han, Tianying Zhang, Yating Zhang, Libin Ma, Ze Yang, Yong-xun Zhao
Biochimica et Biophysica Acta (BBA) - Reviews on Cancer.2025; 1880(6): 189459. CrossRef - Impact of the tumor microenvironment on survival in anaplastic thyroid carcinoma
Chanchal Rana, Prabhakar Mishra, Kulranjan Singh, Pooja Ramakant, Anand Mishra, Sumaira Qayoom, Gulshan Kumar, Jyoti Mishra, Komal Verma
Scientific Reports.2025;[Epub] CrossRef - Chimeric antigen receptor cells as a tool for localized delivery of TNFα in solid cancer treatment
Zhenqiang Yao, Chutamath Sittplangkoon
Human Vaccines & Immunotherapeutics.2025;[Epub] CrossRef - Defining Gene Signature of Tumor-Associated Macrophages in Intrahepatic Cholangiocarcinoma as Target for Immunotherapy Using Single Cell and Bulk RNA Sequencing
Joshua S. Badshah, Ryan M. Lee, Andrea Reitsma, Marc L. Melcher, Olivia M. Martinez, Sheri M. Krams, Daniel J. Delitto, Varvara A. Kirchner
Livers.2025; 5(4): 53. CrossRef - Therapeutic Strategies Targeting CD163 and CD169 in Macrophages for Cancer
Yukio Fujiwara, Yoshihiro Komohara
Pathology International.2025; 75(11): 551. CrossRef - Induction of trained immunity in myeloid cells and their progenitors from thyroid cancer patients
Pepijn van Houten, Prashant Changoer, Liesbeth van Emst, Han J. Bonenkamp, Johannes H. W. de Wilt, Willemijn Hobo, Manita E. J. Bremmers, Mieke W. H. Roeven, Janneke E. W. Walraven, Petronella B. Ottevanger, Willem J. M. Mulder, Mihai G. Netea, Martin Jae
Frontiers in Immunology.2025;[Epub] CrossRef - Circulating immunophenotypes are potentially prognostic in follicular cell-derived thyroid cancer
Anupam Kotwal, Michael P. Gustafson, Svetlana Bornschlegl, Allan B. Dietz, Danae Delivanis, Mabel Ryder
Frontiers in Immunology.2024;[Epub] CrossRef - Nano-enhanced immunotherapy: Targeting the immunosuppressive tumor microenvironment
Yuzhi Jin, Yangyue Huang, Hui Ren, Huanhuan Huang, Chunyu Lai, Wenjun Wang, Zhou Tong, Hangyu Zhang, Wei Wu, Chuan Liu, Xuanwen Bao, Weijia Fang, Hongjun Li, Peng Zhao, Xiaomeng Dai
Biomaterials.2024; 305: 122463. CrossRef - Tumor microenvironment in thyroid cancer: Immune cells, patterns, and novel treatments
Beatriz Febrero, Juan José Ruiz‐Manzanera, Inmaculada Ros‐Madrid, Antonio Miguel Hernández, Esteban Orenes‐Piñero, José Manuel Rodríguez
Head & Neck.2024; 46(6): 1486. CrossRef - Research progress of immunotherapy against anaplastic thyroid cancer
Jiaqian Chen, Zuixuan Xiao, Hongyan Wu
Frontiers in Oncology.2024;[Epub] CrossRef - Tumor‐associated macrophages impair NK cell IFN‐γ production and contribute to tumor progression in clear cell renal cell carcinoma
Sol Yanel Núñez, Aldana Trotta, María Victoria Regge, María Sofía Amarilla, Florencia Secchiari, Jessica Mariel Sierra, María Cecilia Santilli, Mariana Gantov, Agustín Rovegno, Nicolás Richards, Carlos Ameri, Hernando Ríos Pita, Luis Rico, Mauro Mieggi, G
European Journal of Immunology.2024;[Epub] CrossRef - Monocytes in leukapheresis products affect the outcome of CD19–targeted CAR T-cell therapy in patients with lymphoma
Cristiana Carniti, Nicole M. Caldarelli, Luca Agnelli, Tommaso Torelli, Silva Ljevar, Sadhana Jonnalagadda, Giada Zanirato, Eugenio Fardella, Federico Stella, Daniele Lorenzini, Silvia Brich, Flavio Arienti, Anna Dodero, Annalisa Chiappella, Martina Magni
Blood Advances.2024; 8(8): 1968. CrossRef - Clinicopathological Features and Molecular Signatures of Lateral Neck Lymph Node Metastasis in Papillary Thyroid Microcarcinoma
Jinsun Lim, Han Sai Lee, Jin-Hyung Heo, Young Shin Song
Endocrinology and Metabolism.2024; 39(2): 324. CrossRef - Relaxin-2 is a novel biomarker for differentiated thyroid carcinoma in humans
Anupam Kotwal, Ronda Simpson, Nicholas Whiteman, Benjamin Swanson, Ana Yuil-Valdes, Madelyn Fitch, Joshua Nguyen, Salma Elhag, Oleg Shats, Whitney Goldner, Robert Bennett
Biochemical Pharmacology.2024; 225: 116323. CrossRef - Emerging therapeutic options for follicular-derived thyroid cancer in the era of immunotherapy
Naimah Turner, Sarah Hamidi, Rim Ouni, Rene Rico, Ying C. Henderson, Maria Puche, Sayan Alekseev, Jocelynn G. Colunga-Minutti, Mark E. Zafereo, Stephen Y. Lai, Sang T. Kim, Maria E. Cabanillas, Roza Nurieva
Frontiers in Immunology.2024;[Epub] CrossRef - Deciphering the performance of macrophages in tumour microenvironment: a call for precision immunotherapy
Belén Toledo, Linrui Zhu Chen, María Paniagua-Sancho, Juan Antonio Marchal, Macarena Perán, Elisa Giovannetti
Journal of Hematology & Oncology.2024;[Epub] CrossRef - Characterization of Immune Infiltrate Along the Leading Edge of Differentiated Thyroid Cancer
Anupam Kotwal, Krysten Vance, Kemal Hajric, Ana Yuil-Valdes, Benjamin Swanson, Ernesto Martinez Duarte, Oleg Shats, Michael Hollingsworth, Hamid Band, Whitney Goldner
Thyroid®.2024; 34(8): 999. CrossRef - Leveraging molecular targeted drugs and immune checkpoint inhibitors treat advanced thyroid carcinoma to achieve thyroid carcinoma redifferentiation
Jing-Yang Su
American Journal of Cancer Research.2024; 14(2): 407. CrossRef - Harnessing the tumor microenvironment to boost adoptive T cell therapy with engineered lymphocytes for solid tumors
Martina Spiga, Elisa Martini, Maria Chiara Maffia, Fabio Ciceri, Eliana Ruggiero, Alessia Potenza, Chiara Bonini
Seminars in Immunopathology.2024;[Epub] CrossRef - Anaplastic thyroid cancer cell-secreted TGFβ1 plays a key role in inducing macrophage polarization of human monocytes
Agustina Jaroszewski
American Journal of Cancer Research.2024; 14(7): 3626. CrossRef - Therapeutic treatments targeting communication between angiogenic and immune microenvironments in thyroid cancers
Alessandro Prete, Carmelo Nucera
Current Opinion in Endocrine and Metabolic Research.2024; 37: 100544. CrossRef - Immune microenvironment in papillary thyroid carcinoma: roles of immune cells and checkpoints in disease progression and therapeutic implications
Xun Zheng, Ruonan Sun, Tao Wei
Frontiers in Immunology.2024;[Epub] CrossRef - Impact of Hashimoto’s thyroiditis on the tumor microenvironment in papillary thyroid cancer: insights from single-cell analysis
Hongzhe Ma, Guoqi Li, Diwei Huo, Yangguang Su, Qing Jin, Yangxu Lu, Yanyan Sun, Denan Zhang, Xiujie Chen
Frontiers in Endocrinology.2024;[Epub] CrossRef - Unconventional p65/p52 NF-κB module regulates key tumor microenvironment-related genes in breast tumor-associated macrophages (TAMs)
Veronica De Paolis, Virginia Troisi, Antonella Bordin, Francesca Pagano, Viviana Caputo, Chiara Parisi
Life Sciences.2024; 357: 123059. CrossRef - Revolutionizing prognostic predictions in colorectal cancer: Macrophage‑driven transcriptional insights from single‑cell RNA sequencing and gene co‑expression network analysis
Yang Feng, Zhuo Cheng, Jingyuan Gao, Tao Huang, Jun Wang, Qian Tang, Ke Pu, Chang Liu
Oncology Letters.2024;[Epub] CrossRef - Aggressive Types of Malignant Thyroid Neoplasms
Maria Boudina, Eleana Zisimopoulou, Persefoni Xirou, Alexandra Chrisoulidou
Journal of Clinical Medicine.2024; 13(20): 6119. CrossRef - Unravelling the Reasons Behind Limited Response to Anti-PD Therapy in ATC: A Comprehensive Evaluation of Tumor-Infiltrating Immune Cells and Checkpoints
Monikongkona Boruah, Shipra Agarwal, Riyaz Ahmad Mir, Saumitra Dey Choudhury, Kapil Sikka, Sameer Rastogi, Nishikant Damle, Mehar C. Sharma
Endocrine Pathology.2024; 35(4): 419. CrossRef - Imaging tumor and ascites-associated macrophages in a mouse model of metastatic ovarian cancer
Catherine A. Foss, Flonné Wildes, Delia Mezzanzanica, Franca Podo, Chien-Fu Hung, Santosh Yadav, Marie-France Penet Vidaver
EJNMMI Research.2024;[Epub] CrossRef - From ductal carcinoma in situ to invasive breast cancer: the prognostic value of the extracellular microenvironment
Taylor S. Hulahan, Peggi M. Angel
Journal of Experimental & Clinical Cancer Research.2024;[Epub] CrossRef - Combinatory actions of cytokines induce M2-like macrophages in anaplastic thyroid cancer
Takahito Kimura
American Journal of Cancer Research.2024; 14(12): 5812. CrossRef - The ratio of adaptive to innate immune cells differs between genders and associates with improved prognosis and response to immunotherapy
Johanne Ahrenfeldt, Ditte S. Christensen, Andreas B. Østergaard, Judit Kisistók, Mateo Sokač, Nicolai J. Birkbak, Albert Rübben
PLOS ONE.2023; 18(2): e0281375. CrossRef - Current Landscape of Immunotherapy for Advanced Sarcoma
Víctor Albarrán, María Luisa Villamayor, Javier Pozas, Jesús Chamorro, Diana Isabel Rosero, María San Román, Patricia Guerrero, Patricia Pérez de Aguado, Juan Carlos Calvo, Coral García de Quevedo, Carlos González, María Ángeles Vaz
Cancers.2023; 15(8): 2287. CrossRef - Iron-mediated oxidative stress induces PD-L1 expression via activation of c-Myc in lung adenocarcinoma
Anna Martina Battaglia, Alessandro Sacco, Ilenia Aversa, Gianluca Santamaria, Camillo Palmieri, Cirino Botta, Roberto De Stefano, Maurizio Bitetto, Lavinia Petriaggi, Emanuele Giorgio, Concetta Maria Faniello, Francesco Costanzo, Flavia Biamonte
Frontiers in Cell and Developmental Biology.2023;[Epub] CrossRef - Molecular mechanisms of immunotherapy resistance in triple-negative breast cancer
Yiwen Zheng, Shujin Li, Hongchao Tang, Xuli Meng, Qinghui Zheng
Frontiers in Immunology.2023;[Epub] CrossRef - Modeling the tumor microenvironment of anaplastic thyroid cancer: an orthotopic tumor model in C57BL/6 mice
Zhen Xu, Hyo Shik Shin, Yoo Hyung Kim, Seong Yun Ha, Jae-Kyung Won, Su-jin Kim, Young Joo Park, Sareh Parangi, Sun Wook Cho, Kyu Eun Lee
Frontiers in Immunology.2023;[Epub] CrossRef - A new approach to overcoming resistance to immunotherapy: nanotechnology
Jiangbo Shao, Ying Jin, Chunxiang Jin
Frontiers in Oncology.2023;[Epub] CrossRef - Integrative single-cell transcriptome analysis reveals immune suppressive landscape in the anaplastic thyroid cancer
Chao Feng, Yujia Tao, Chao Yu, Lirui Wang, Xiao Liu, Yuan Cao
Cancer Gene Therapy.2023; 30(12): 1598. CrossRef - Tumor-associated macrophages as a potential therapeutic target in thyroid cancers
Liya Zhu, Xiu Juan Li, Prakash Gangadaran, Xiuli Jing, Byeong-Cheol Ahn
Cancer Immunology, Immunotherapy.2023; 72(12): 3895. CrossRef - Influence of Macrophages on Vascular Invasion of Inflammatory Breast Cancer Emboli Measured Using an In Vitro Microfluidic Multi-Cellular Platform
Manasa Gadde, Melika Mehrabi-Dehdezi, Bisrat G. Debeb, Wendy A. Woodward, Marissa Nichole Rylander
Cancers.2023; 15(19): 4883. CrossRef - The Prognosis of Cancer Depends on the Interplay of Autophagy, Apoptosis, and Anoikis within the Tumor Microenvironment
Shweta Gulia, Prakash Chandra, Asmita Das
Cell Biochemistry and Biophysics.2023; 81(4): 621. CrossRef - Comprehensive analysis of BTNL9 as a prognostic biomarker correlated with immune infiltrations in thyroid cancer
Luyao Zhang, Shuang Yu, Shubin Hong, Xi Xiao, Zhihong Liao, Yanbing Li, Haipeng Xiao
BMC Medical Genomics.2023;[Epub] CrossRef - FF-10850, a Novel Liposomal Topotecan Achieves Superior Antitumor Activity via Macrophage- and Ammonia-Mediated Payload Release in the Tumor Microenvironment
Susumu Shimoyama, Ken Okada, Toshifumi Kimura, Yasushi Morohashi, Shinji Nakayama, Sayaka Kemmochi, Keiko Makita-Suzuki, Ursula A. Matulonis, Mikinaga Mori
Molecular Cancer Therapeutics.2023; 22(12): 1454. CrossRef - Estrogen-related genes for thyroid cancer prognosis, immune infiltration, staging, and drug sensitivity
Leiying Zhang, Man Zhou, Xiaoni Gao, Yang Xie, Junqi Xiao, Tao Liu, Xiangtai Zeng
BMC Cancer.2023;[Epub] CrossRef - Harnessing Immunity to Treat Advanced Thyroid Cancer
Hiroki Komatsuda, Michihisa Kono, Risa Wakisaka, Ryosuke Sato, Takahiro Inoue, Takumi Kumai, Miki Takahara
Vaccines.2023; 12(1): 45. CrossRef - Are third-generation active-targeting nanoformulations definitely the best? In vitro and in vivo comparisons of pixantrone-loaded liposomes modified with different sialic acid derivatives
Yanzhi Song, Zhennan She, Zhenjun Huang, Shuo Wang, Xinrong Liu, Qi Zhang, Jing Sun, Donghua Di, Yihui Deng
Drug Delivery and Translational Research.2022; 12(3): 647. CrossRef - Synergistic nanoassemblies constructed from a STAT3 inhibitor and a cabazitaxel prodrug with enhanced cancer chemo-immunotherapy
X. Shi, L. Shu, Y. Qiao, J. Yao, H. Xie, L. Zhou, H. Wang, S. Zheng
Materials Today Nano.2022; 17: 100155. CrossRef - Polypharmacologic Reprogramming of Tumor-Associated Macrophages toward an Inflammatory Phenotype
Nao Nishida-Aoki, Taranjit S. Gujral
Cancer Research.2022; 82(3): 433. CrossRef - Role of Suprabasin in the Dedifferentiation of Follicular Epithelial Cell-Derived Thyroid Cancer and Identification of Related Immune Markers
Hao Tan, Lidong Wang, Zhen Liu
Frontiers in Genetics.2022;[Epub] CrossRef - Macrophage C/EBPδ Drives Gemcitabine, but Not 5-FU or Paclitaxel, Resistance of Pancreatic Cancer Cells in a Deoxycytidine-Dependent Manner
C. Arnold Spek, Hella L. Aberson, JanWillem Duitman
Biomedicines.2022; 10(2): 219. CrossRef - Anaplastic Thyroid Carcinoma: An Update
Arnaud Jannin, Alexandre Escande, Abir Al Ghuzlan, Pierre Blanchard, Dana Hartl, Benjamin Chevalier, Frédéric Deschamps, Livia Lamartina, Ludovic Lacroix, Corinne Dupuy, Eric Baudin, Christine Do Cao, Julien Hadoux
Cancers.2022; 14(4): 1061. CrossRef - LC3-associated phagocytosis in bone marrow macrophages suppresses acute myeloid leukemia progression through STING activation
Jamie A. Moore, Jayna J. Mistry, Charlotte Hellmich, Rebecca H. Horton, Edyta E. Wojtowicz, Aisha Jibril, Matthew Jefferson, Thomas Wileman, Naiara Beraza, Kristian M. Bowles, Stuart A. Rushworth
Journal of Clinical Investigation.2022;[Epub] CrossRef - Integration of chemokine signaling with non-coding RNAs in tumor microenvironment and heterogeneity in different cancers
Shweta Arora, Salman Khan, Almaz Zaki, Gulnaz Tabassum, Mohd Mohsin, Humaira Naaz Bhutto, Tanveer Ahmad, Tasneem Fatma, Mansoor Ali Syed
Seminars in Cancer Biology.2022; 86: 720. CrossRef - BRAFV600E Induction in Thyrocytes Triggers Important Changes in the miRNAs Content and the Populations of Extracellular Vesicles Released in Thyroid Tumor Microenvironment
Ophélie Delcorte, Catherine Spourquet, Pascale Lemoine, Jonathan Degosserie, Patrick Van Der Smissen, Nicolas Dauguet, Axelle Loriot, Jeffrey A. Knauf, Laurent Gatto, Etienne Marbaix, James A. Fagin, Christophe E. Pierreux
Biomedicines.2022; 10(4): 755. CrossRef - Inflammatory Tumor Microenvironment in Cranial Meningiomas: Clinical Implications and Intraindividual Reproducibility
Johannes Wach, Tim Lampmann, Ági Güresir, Hartmut Vatter, Ulrich Herrlinger, Albert Becker, Marieta Toma, Michael Hölzel, Erdem Güresir
Diagnostics.2022; 12(4): 853. CrossRef - Roles and new Insights of Macrophages in the Tumor Microenvironment of Thyroid Cancer
Qi Liu, Wei Sun, Hao Zhang
Frontiers in Pharmacology.2022;[Epub] CrossRef - Fucoxanthin Is a Potential Therapeutic Agent for the Treatment of Breast Cancer
Tsz-Ying Lau, Hiu-Yee Kwan
Marine Drugs.2022; 20(6): 370. CrossRef - Dissecting Immunosuppressive Cell Communication Patterns Reveals JunB Proto-Oncogene (JUNB) Shaping a Non-Inflamed Tumor Microenvironment
Hualin Chen, Gang Chen
Frontiers in Genetics.2022;[Epub] CrossRef - LIMK1: A promising prognostic and immune infiltration indicator in colorectal cancer
Xin Liu, Qiang Song, Daohan Wang, Yubiao Liu, Zhixiang Zhang, Weihua Fu
Oncology Letters.2022;[Epub] CrossRef - Unusual Association of NF-κB Components in Tumor-Associated Macrophages (TAMs) Promotes HSPG2-Mediated Immune-Escaping Mechanism in Breast Cancer
Veronica De Paolis, Fabio Maiullari, Maila Chirivì, Marika Milan, Chiara Cordiglieri, Francesca Pagano, Alessandra Rita La Manna, Elena De Falco, Claudia Bearzi, Roberto Rizzi, Chiara Parisi
International Journal of Molecular Sciences.2022; 23(14): 7902. CrossRef - Crosstalk between angiogenesis and immune regulation in the tumor microenvironment
Hei Jung Kim, Young Rae Ji, You Mie Lee
Archives of Pharmacal Research.2022; 45(6): 401. CrossRef - Cancer Resistance to Immunotherapy: Molecular Mechanisms and Tackling Strategies
Son Hai Vu, Preethi Vetrivel, Jongmin Kim, Myeong-Sok Lee
International Journal of Molecular Sciences.2022; 23(18): 10906. CrossRef - Transcription Factor MAFB as a Prognostic Biomarker for the Lung Adenocarcinoma
Omar Samir, Naohiro Kobayashi, Teppei Nishino, Mennatullah Siyam, Manoj Kumar Yadav, Yuri Inoue, Satoru Takahashi, Michito Hamada
International Journal of Molecular Sciences.2022; 23(17): 9945. CrossRef - Construction and Validation of a CNV-Driven Ferroptosis-Related Gene Signature for Predicting the Prognosis of Lung Adenocarcinoma
Yanqing Wang, Yi Zhao, Yong Li, Zemin Luo, Hongzhu Chen, Yuan Li
Journal of Sensors.2022; 2022: 1. CrossRef - CXCL12 derived from CD248-expressing cancer-associated fibroblasts mediates M2-polarized macrophages to promote nonsmall cell lung cancer progression
Jieheng Wu, Xinlei Liu, Jiangwei Wu, Chunju Lou, Qiaoling Zhang, Huiping Chen, Zeyang Yang, Shiqi Long, Yun Wang, Zhenling Shang, Zuquan Hu, Rui Zhang, Jian Zhang, Zhu Zeng
Biochimica et Biophysica Acta (BBA) - Molecular Basis of Disease.2022; 1868(11): 166521. CrossRef - Cell Component and Function of Tumor Microenvironment in Thyroid Cancer
Eunah Shin, Ja Seung Koo
International Journal of Molecular Sciences.2022; 23(20): 12578. CrossRef - TIM3 Expression in Anaplastic-Thyroid-Cancer-Infiltrating Macrophages: An Emerging Immunotherapeutic Target
Luz Maria Palacios, Victoria Peyret, María Estefania Viano, Romina Celeste Geysels, Yair Aron Chocobar, Ximena Volpini, Claudia Gabriela Pellizas, Juan Pablo Nicola, Claudia Cristina Motran, María Cecilia Rodriguez-Galan, Laura Fozzatti
Biology.2022; 11(11): 1609. CrossRef - Potential diagnostic of lymph node metastasis and prognostic values of TM4SFs in papillary thyroid carcinoma patients
Kun Wang, Haomin Li, Junyu Zhao, Jinming Yao, Yiran Lu, Jianjun Dong, Jie Bai, Lin Liao
Frontiers in Cell and Developmental Biology.2022;[Epub] CrossRef - A distinct M2 macrophage infiltrate and transcriptomic profile decisively influence adipocyte differentiation in lipedema
Stefan Wolf, Jenna H. Rannikko, Reetta Virtakoivu, Paolo Cinelli, Gunther Felmerer, Anna Burger, Pietro Giovanoli, Michael Detmar, Nicole Lindenblatt, Maija Hollmén, Epameinondas Gousopoulos
Frontiers in Immunology.2022;[Epub] CrossRef - Macrophage targeting in cancer
Martha Lopez‐Yrigoyen, Luca Cassetta, Jeffrey W. Pollard
Annals of the New York Academy of Sciences.2021; 1499(1): 18. CrossRef - Immune Landscape of Thyroid Cancers: New Insights
Elisa Menicali, Martina Guzzetti, Silvia Morelli, Sonia Moretti, Efisio Puxeddu
Frontiers in Endocrinology.2021;[Epub] CrossRef - Targeting tumor-associated macrophages to synergize tumor immunotherapy
Xiaonan Xiang, Jianguo Wang, Di Lu, Xiao Xu
Signal Transduction and Targeted Therapy.2021;[Epub] CrossRef - FOXE1-Dependent Regulation of Macrophage Chemotaxis by Thyroid Cells In Vitro and In Vivo
Sara Credendino, Marta De Menna, Irene Cantone, Carmen Moccia, Matteo Esposito, Luigi Di Guida, Mario De Felice, Gabriella De Vita
International Journal of Molecular Sciences.2021; 22(14): 7666. CrossRef - Hidden Treasures: Macrophage Long Non-Coding RNAs in Lung Cancer Progression
Annika Karger, Rajender Nandigama, Albrecht Stenzinger, Friedrich Grimminger, Soni Savai Pullamsetti, Werner Seeger, Rajkumar Savai
Cancers.2021; 13(16): 4127. CrossRef - A Prognostic Role for Circulating microRNAs Involved in Macrophage Polarization in Advanced Non-Small Cell Lung Cancer
Alexia Monastirioti, Chara Papadaki, Konstantinos Rounis, Despoina Kalapanida, Dimitrios Mavroudis, Sofia Agelaki
Cells.2021; 10(8): 1988. CrossRef - Hyperglycemia-Induced miR-467 Drives Tumor Inflammation and Growth in Breast Cancer
Jasmine Gajeton, Irene Krukovets, Santoshi Muppala, Dmitriy Verbovetskiy, Jessica Zhang, Olga Stenina-Adognravi
Cancers.2021; 13(6): 1346. CrossRef - Targeting MARCO and IL37R on Immunosuppressive Macrophages in Lung Cancer Blocks Regulatory T Cells and Supports Cytotoxic Lymphocyte Function
Linnéa La Fleur, Johan Botling, Fei He, Catarina Pelicano, Chikai Zhou, Chenfei He, Giorgia Palano, Artur Mezheyeuski, Patrick Micke, Jeffrey V. Ravetch, Mikael C. I. Karlsson, Dhifaf Sarhan
Cancer Research.2021; 81(4): 956. CrossRef - Absent in melanoma 2-mediating M1 macrophages facilitate tumor rejection in renal carcinoma
Dafei Chai, Zichun Zhang, Shang yuchen Shi, Dong Qiu, Chen Zhang, Gang Wang, Lin Fang, Huizhong Li, Hui Tian, Hailong Li, Junnian Zheng
Translational Oncology.2021; 14(4): 101018. CrossRef - Hampering Stromal Cells in the Tumor Microenvironment as a Therapeutic Strategy to Destem Cancer Stem Cells
Katherine Po Sin Chung, Rainbow Wing Hei Leung, Terence Kin Wah Lee
Cancers.2021; 13(13): 3191. CrossRef - Thyroid Cancer Stem-Like Cells: From Microenvironmental Niches to Therapeutic Strategies
Elisa Stellaria Grassi, Viola Ghiandai, Luca Persani
Journal of Clinical Medicine.2021; 10(7): 1455. CrossRef - Enhanced Drug Delivery to Solid Tumors via Drug-Loaded Nanocarriers: An Image-Based Computational Framework
Farshad Moradi Kashkooli, M. Soltani, Mohammad Masoud Momeni, Arman Rahmim
Frontiers in Oncology.2021;[Epub] CrossRef - Mechanisms of primary and acquired resistance to PD-1/PD-L1 blockade and the emerging role of gut microbiome
R. Zou, Y. Wang, F. Ye, X. Zhang, M. Wang, S. Cui
Clinical and Translational Oncology.2021; 23(11): 2237. CrossRef - Role of Tumor-Associated Macrophages in Sarcomas
Tomohiro Fujiwara, John Healey, Koichi Ogura, Aki Yoshida, Hiroya Kondo, Toshiaki Hata, Miho Kure, Hiroshi Tazawa, Eiji Nakata, Toshiyuki Kunisada, Toshiyoshi Fujiwara, Toshifumi Ozaki
Cancers.2021; 13(5): 1086. CrossRef - Secreted Factors by Anaplastic Thyroid Cancer Cells Induce Tumor-Promoting M2-like Macrophage Polarization through a TIM3-Dependent Mechanism
Cinthia Carolina Stempin, Romina Celeste Geysels, Sunmi Park, Luz Maria Palacios, Ximena Volpini, Claudia Cristina Motran, Eva Virginia Acosta Rodríguez, Juan Pablo Nicola, Sheue-yann Cheng, Claudia Gabriela Pellizas, Laura Fozzatti
Cancers.2021; 13(19): 4821. CrossRef - Papillary Thyroid Carcinoma Landscape and Its Immunological Link With Hashimoto Thyroiditis at Single-Cell Resolution
Jun Pan, Fang Ye, Chengxuan Yu, Qinsheng Zhu, Jiaqi Li, Yaohui Zhang, Hedi Tian, Yunjin Yao, Minjie Zhu, Yibin Shen, Feng Zhu, Yingying Wang, Xinhui Zhou, Guoji Guo, Yijun Wu
Frontiers in Cell and Developmental Biology.2021;[Epub] CrossRef - Evaluation of solid tumor response to sequential treatment cycles via a new computational hybrid approach
Farshad Moradi Kashkooli, M. Soltani
Scientific Reports.2021;[Epub] CrossRef - Assessment of outcomes and novel immune biomarkers in metaplastic breast cancer: a single institution retrospective study
Evan Morgan, Anupama Suresh, Akaansha Ganju, Daniel G. Stover, Robert Wesolowski, Sagar Sardesai, Anne Noonan, Raquel Reinbolt, Jeffrey VanDeusen, Nicole Williams, Mathew A. Cherian, Zaibo Li, Gregory Young, Marilly Palettas, Julie Stephens, Joseph Liu, A
World Journal of Surgical Oncology.2020;[Epub] CrossRef - Differentiated agonistic antibody targeting CD137 eradicates large tumors without hepatotoxicity
Ugur Eskiocak, Wilson Guzman, Benjamin Wolf, Christine Cummings, Lauren Milling, Hsin-Jung Wu, Michael Ophir, Conner Lambden, Pearl Bakhru, Dana C. Gilmore, Samantha Ottinger, Lucy Liu, William K. McConaughy, Sunny Q. He, Chao Wang, Cheuk Lun Leung, Jason
JCI Insight.2020;[Epub] CrossRef - The Tumor Microenvironment: A Milieu Hindering and Obstructing Antitumor Immune Responses
Alireza Labani-Motlagh, Mehrnoush Ashja-Mahdavi, Angelica Loskog
Frontiers in Immunology.2020;[Epub] CrossRef - Sevoflurane depletes macrophages from the melanoma microenvironment
Isabella Sztwiertnia, Judith Schenz, Katharina Bomans, Dominik Schaack, Johanna Ohnesorge, Sandra Tamulyte, Markus A. Weigand, Florian Uhle, Roger Chammas
PLOS ONE.2020; 15(5): e0233789. CrossRef - Lung cancer aggressiveness in an intermittent hypoxia murine model of postmenopausal sleep apnea
Marta Torres, Miguel Ángel Martinez-Garcia, Francisco Campos-Rodriguez, David Gozal, Josep M. Montserrat, Daniel Navajas, Ramon Farré, Isaac Almendros
Menopause.2020; 27(6): 706. CrossRef - Neck Disability and Swallowing Function in Posttreatment Head and Neck Cancer Patients
Alexandria Harris, Lingyun Lyu, Tamara Wasserman‐Winko, Susan George, Jonas T. Johnson, Marci Lee Nilsen
Otolaryngology–Head and Neck Surgery.2020; 163(4): 763. CrossRef - Metabolic modulation via mTOR pathway and anti-angiogenesis remodels tumor microenvironment using PD-L1-targeting codelivery
Binfan Chen, Ang Gao, Bin Tu, Yonghui Wang, Xiaolu Yu, Yingshu Wang, Yanfeng Xiu, Bing Wang, Yakun Wan, Yongzhuo Huang
Biomaterials.2020; 255: 120187. CrossRef - The Thyroid Tumor Microenvironment: Potential Targets for Therapeutic Intervention and Prognostication
Laura MacDonald, Jonathan Jenkins, Grace Purvis, Joshua Lee, Aime T. Franco
Hormones and Cancer.2020; 11(5-6): 205. CrossRef - The Contribution of Race to Breast Tumor Microenvironment Composition and Disease Progression
Gina Kim, Jessica M. Pastoriza, John S. Condeelis, Joseph A. Sparano, Panagiota S. Filippou, George S. Karagiannis, Maja H. Oktay
Frontiers in Oncology.2020;[Epub] CrossRef - Prognosis of Macrophage Density in the Absence of Neutrophils in Differentiated Thyroid Cancer
Amblessed E. Onuma, Lynn Schoenfield, Chengli Shen, Charity Edwards, John E. Phay, Lawrence A. Shirley, Allan Tsung
Journal of Surgical Research.2020; 256: 458. CrossRef - Targeting of CD163+ Macrophages in Inflammatory and Malignant Diseases
Maria K. Skytthe, Jonas Heilskov Graversen, Søren K. Moestrup
International Journal of Molecular Sciences.2020; 21(15): 5497. CrossRef - Synthetic chlorin derivative self-prevented from aggregation: Behavior in homogeneous medium for PDT applications
Bianca Martins Estevão, Camila Fabiano de Freitas, Douglas Santana Franciscato, Francisco Fávaro de Assis, Kleber Thiago de Oliveira, Noboru Hioka, Wilker Caetano, Edvani Curti Muniz
Journal of Molecular Liquids.2020; 320: 114363. CrossRef - Tumor‑associated macrophages in lung cancer: Friendly or evil? (Review)
Fei Xu, Ying Wei, Zhao Tang, Baojun Liu, Jingcheng Dong
Molecular Medicine Reports.2020;[Epub] CrossRef - Macrophage-secreted MMP9 induces mesenchymal transition in pancreatic cancer cells via PAR1 activation
Cansu Tekin, Hella L Aberson, Cynthia Waasdorp, Gerrit K J Hooijer, Onno J de Boer, Frederike Dijk, Maarten F Bijlsma, C Arnold Spek
Cellular Oncology.2020; 43(6): 1161. CrossRef - Cancer Stem Cells in Thyroid Tumors: From the Origin to Metastasis
Veronica Veschi, Francesco Verona, Melania Lo Iacono, Caterina D'Accardo, Gaetana Porcelli, Alice Turdo, Miriam Gaggianesi, Stefano Forte, Dario Giuffrida, Lorenzo Memeo, Matilde Todaro
Frontiers in Endocrinology.2020;[Epub] CrossRef - The Crosstalk Between Tumor-Associated Macrophages (TAMs) and Tumor Cells and the Corresponding Targeted Therapy
Zhe Ge, Shuzhe Ding
Frontiers in Oncology.2020;[Epub] CrossRef - Immune Cell Confrontation in the Papillary Thyroid Carcinoma Microenvironment
Zhenyu Xie, Xin Li, Yuzhen He, Song Wu, Shiyue Wang, Jianjian Sun, Yuchen He, Yu Lun, Jian Zhang
Frontiers in Endocrinology.2020;[Epub] CrossRef - Early macrophage infiltrates impair pancreatic cancer cell growth by TNF-α secretion
Cansu Tekin, Hella L. Aberson, Maarten F. Bijlsma, C. Arnold Spek
BMC Cancer.2020;[Epub] CrossRef - Attenuation of CD47-SIRPα Signal in Cholangiocarcinoma Potentiates Tumor-Associated Macrophage-Mediated Phagocytosis and Suppresses Intrahepatic Metastasis
Kulthida Vaeteewoottacharn, Ryusho Kariya, Phattarin Pothipan, Sawako Fujikawa, Chawalit Pairojkul, Sakda Waraasawapati, Kazuhiko Kuwahara, Chaisiri Wongkham, Sopit Wongkham, Seiji Okada
Translational Oncology.2019; 12(2): 217. CrossRef - Circulating CEA‐dNLR score predicts clinical outcome of metastatic gallbladder cancer patient
Jing‐Hui Du, Jun Lu
Journal of Clinical Laboratory Analysis.2019;[Epub] CrossRef - The Interplay between MicroRNAs and Cellular Components of Tumour Microenvironment (TME) on Non-Small-Cell Lung Cancer (NSCLC) Progression
Sook Shien Lee, Yoke Kqueen Cheah
Journal of Immunology Research.2019; 2019: 1. CrossRef - Papillary thyroid carcinoma behavior: clues in the tumor microenvironment
Kensey Bergdorf, Donna C Ferguson, Mitra Mehrad, Kim Ely, Thomas Stricker, Vivian L Weiss
Endocrine-Related Cancer.2019; 26(6): 601. CrossRef - Interplay between thyroid cancer cells and macrophages: effects on IL-32 mediated cell death and thyroid cancer cell migration
Yvette J. E. Sloot, Katrin Rabold, Thomas Ulas, Dennis M. De Graaf, Bas Heinhuis, Kristian Händler, Joachim L. Schultze, Mihai G. Netea, Johannes W. A. Smit, Leo A. B. Joosten, Romana T. Netea-Maier
Cellular Oncology.2019; 42(5): 691. CrossRef - Iron accumulation in tumor-associated macrophages marks an improved overall survival in patients with lung adenocarcinoma
Carl Maximilian Thielmann, Milene Costa da Silva, Thomas Muley, Michael Meister, Esther Herpel, Martina U. Muckenthaler
Scientific Reports.2019;[Epub] CrossRef - The Immune Landscape of Thyroid Cancer in the Context of Immune Checkpoint Inhibition
Gilda Varricchi, Stefania Loffredo, Giancarlo Marone, Luca Modestino, Poupak Fallahi, Silvia Martina Ferrari, Amato de Paulis, Alessandro Antonelli, Maria Rosaria Galdiero
International Journal of Molecular Sciences.2019; 20(16): 3934. CrossRef - A review on the role of M2 macrophages in bladder cancer; pathophysiology and targeting
Laleh Sharifi, Mohammad Reza Nowroozi, Erfan Amini, Masoumeh Kourosh Arami, Mohsen Ayati, Monireh Mohsenzadegan
International Immunopharmacology.2019; 76: 105880. CrossRef - Tumor-associated macrophage infiltration in meningioma
Dustin T Proctor, Jordan Huang, Sanju Lama, Abdulrahman Albakr, Guido Van Marle, Garnette R Sutherland
Neuro-Oncology Advances.2019;[Epub] CrossRef - Immune and Inflammatory Cells in Thyroid Cancer Microenvironment
Silvia Martina Ferrari, Poupak Fallahi, Maria Rosaria Galdiero, Ilaria Ruffilli, Giusy Elia, Francesca Ragusa, Sabrina Rosaria Paparo, Armando Patrizio, Valeria Mazzi, Gilda Varricchi, Gianni Marone, Alessandro Antonelli
International Journal of Molecular Sciences.2019; 20(18): 4413. CrossRef - Caveolin-2 deficiency induces a rapid anti-tumor immune response prior to regression of implanted murine lung carcinoma tumors
Yajun Liu, Xiaoqiang Qi, Guangfu Li, Grzegorz Sowa
Scientific Reports.2019;[Epub] CrossRef - Paclitaxel Treatment and Proprotein Convertase 1/3 (PC1/3) Knockdown in Macrophages is a Promising Antiglioma Strategy as Revealed by Proteomics and Cytotoxicity Studies
Marie Duhamel, Mélanie Rose, Franck Rodet, Adriana Natalia Murgoci, Lea Zografidou, Anne Régnier-Vigouroux, Fabien Vanden Abeele, Firas Kobeissy, Serge Nataf, Laurent Pays, Maxence Wisztorski, Dasa Cizkova, Isabelle Fournier, Michel Salzet
Molecular & Cellular Proteomics.2018; 17(6): 1126. CrossRef - Chloroquine and nanoparticle drug delivery: A promising combination
Joe Pelt, Sara Busatto, Mauro Ferrari, E. Aubrey Thompson, Kabir Mody, Joy Wolfram
Pharmacology & Therapeutics.2018; 191: 43. CrossRef - Conditioned medium from stimulated macrophages inhibits growth but induces an inflammatory phenotype in breast cancer cells
Wenzhe Song, Parth Thakor, David A. Vesey, Glenda C. Gobe, Christudas Morais
Biomedicine & Pharmacotherapy.2018; 106: 247. CrossRef - Potential involvement of neutrophils in human thyroid cancer
Maria Rosaria Galdiero, Gilda Varricchi, Stefania Loffredo, Claudio Bellevicine, Tiziana Lansione, Anne Lise Ferrara, Raffaella Iannone, Sarah di Somma, Francesco Borriello, Eduardo Clery, Maria Triassi, Giancarlo Troncone, Gianni Marone, Fabrizio Mattei
PLOS ONE.2018; 13(6): e0199740. CrossRef - CSF1R-Expressing Tumor-Associated Macrophages, Smoking and Survival in Lung Adenocarcinoma: Analyses Using Quantitative Phosphor-Integrated Dot Staining
Kentaro Inamura, Yasuyuki Shigematsu, Hironori Ninomiya, Yasuhiro Nakashima, Maki Kobayashi, Haruyuki Saito, Katsuhiro Takahashi, Etsuko Futaya, Sakae Okumura, Yuichi Ishikawa, Hiroaki Kanda
Cancers.2018; 10(8): 252. CrossRef - Tumor heterogeneity and nanoparticle-mediated tumor targeting: the importance of delivery system personalization
K. Laxmi Swetha, Aniruddha Roy
Drug Delivery and Translational Research.2018; 8(5): 1508. CrossRef - Immune Gene Signature Delineates a Subclass of Papillary Thyroid Cancer with Unfavorable Clinical Outcomes
Kyuryung Kim, Sora Jeon, Tae-Min Kim, Chan Jung
Cancers.2018; 10(12): 494. CrossRef - Immunohistochemical Expression of TGF-Β1, SMAD4, SMAD7, TGFβRII and CD68-Positive TAM Densities in Papillary Thyroid Cancer
Koni Ivanova, Irena Manolova, Maria-Magdalena Ignatova, Maya Gulubova
Open Access Macedonian Journal of Medical Sciences.2018; 6(3): 435. CrossRef - Bone marrow-derived cells are recruited by the melanoma tumor with endothelial cells contributing to tumor vasculature
R. Bonfim-Silva, L. E. B. Souza, F. U. F. Melo, V. C. Oliveira, D. A. R. Magalhães, H. F. Oliveira, D. T. Covas, A. M. Fontes
Clinical and Translational Oncology.2017; 19(1): 125. CrossRef - Stromal contributions to the carcinogenic process
Mark Spaw, Shrikant Anant, Sufi Mary Thomas
Molecular Carcinogenesis.2017; 56(4): 1199. CrossRef - The Role of Cancer-Associated Fibroblasts and Fibrosis in Liver Cancer
Silvia Affo, Le-Xing Yu, Robert F. Schwabe
Annual Review of Pathology: Mechanisms of Disease.2017; 12(1): 153. CrossRef - PEDF increases the tumoricidal activity of macrophages towards prostate cancer cells in vitro
Dalia Martinez-Marin, Courtney Jarvis, Thomas Nelius, Werner de Riese, Olga V. Volpert, Stéphanie Filleur, Chih-Hsin Tang
PLOS ONE.2017; 12(4): e0174968. CrossRef - Anaplastic thyroid carcinoma: from clinicopathology to genetics and advanced therapies
Eleonora Molinaro, Cristina Romei, Agnese Biagini, Elena Sabini, Laura Agate, Salvatore Mazzeo, Gabriele Materazzi, Stefano Sellari-Franceschini, Alessandro Ribechini, Liborio Torregrossa, Fulvio Basolo, Paolo Vitti, Rossella Elisei
Nature Reviews Endocrinology.2017; 13(11): 644. CrossRef - Iron Induces Anti-tumor Activity in Tumor-Associated Macrophages
Milene Costa da Silva, Michael O. Breckwoldt, Francesca Vinchi, Margareta P. Correia, Ana Stojanovic, Carl Maximilian Thielmann, Michael Meister, Thomas Muley, Arne Warth, Michael Platten, Matthias W. Hentze, Adelheid Cerwenka, Martina U. Muckenthaler
Frontiers in Immunology.2017;[Epub] CrossRef - Reprogramming Tumor-Associated Macrophages To Reverse EGFRT790M Resistance by Dual-Targeting Codelivery of Gefitinib/Vorinostat
Huige Peng, Binfan Chen, Wei Huang, Yubo Tang, Yifan Jiang, Wenyuan Zhang, Yongzhuo Huang
Nano Letters.2017; 17(12): 7684. CrossRef - Numerical modeling of nanodrug distribution in tumors with heterogeneous vasculature
Cheng-Ying Chou, Wan-I Chang, Tzyy-Leng Horng, Win-Li Lin, Han-Chung Wu
PLOS ONE.2017; 12(12): e0189802. CrossRef - HPMA–Copolymer Nanocarrier Targets Tumor-Associated Macrophages in Primary and Metastatic Breast Cancer
Melissa N. Zimel, Chloe B. Horowitz, Vinagolu K. Rajasekhar, Alexander B. Christ, Xin Wei, Jianbo Wu, Paulina M. Wojnarowicz, Dong Wang, Steven R. Goldring, P. Edward Purdue, John H. Healey
Molecular Cancer Therapeutics.2017; 16(12): 2701. CrossRef - Correlation of tumor-associated macrophages and NK cells with bladder cancer size and T stage in patients with solitary low-grade urothelial carcinoma
Kristian Krpina, Emina Babarović, Josip Španjol, Gordana Đorđević, Tobias Maurer, Nives Jonjić
Wiener klinische Wochenschrift.2016; 128(7-8): 248. CrossRef - The lymphatic system and pancreatic cancer
Darci M. Fink, Maria M. Steele, Michael A. Hollingsworth
Cancer Letters.2016; 381(1): 217. CrossRef - Jacalin-Activated Macrophages Exhibit an Antitumor Phenotype
Cláudia Danella Polli, Luciana Pereira Ruas, Luciana Chain Veronez, Thais Herrero Geraldino, Fabiana Rossetto de Morais, Maria Cristina Roque-Barreira, Gabriela Pereira-da-Silva
BioMed Research International.2016; 2016: 1. CrossRef - The immune network in thyroid cancer
Maria Rosaria Galdiero, Gilda Varricchi, Gianni Marone
OncoImmunology.2016; 5(6): e1168556. CrossRef - The Expression and Relationship of CD68-Tumor-Associated Macrophages and Microvascular Density With the Prognosis of Patients With Laryngeal Squamous Cell Carcinoma
Shujun Sun, Xinliang Pan, Limin Zhao, Jianming Zhou, Hongzeng Wang, Yonghong Sun
Clinical and Experimental Otorhinolaryngology.2016; 9(3): 270. CrossRef - Role of pulmonary macrophages in initiation of lung metastasis in anaplastic thyroid cancer
Xiu Juan Li, Prakash Gangadaran, Senthilkumar Kalimuthu, Ji Min Oh, Liya Zhu, Shin Young Jeong, Sang‐Woo Lee, Jaetae Lee, Byeong‐Cheol Ahn
International Journal of Cancer.2016; 139(11): 2583. CrossRef - Perspectives for immunotherapy in endocrine cancer
S Latteyer, V Tiedje, B Schilling, D Führer
Endocrine-Related Cancer.2016; 23(10): R469. CrossRef - Macrophage Densities Correlated with CXC Chemokine Receptor 4 Expression and Related with Poor Survival in Anaplastic Thyroid Cancer
Dae In Kim, Eunyoung Kim, Young A Kim, Sun Wook Cho, Jung Ah Lim, Young Joo Park
Endocrinology and Metabolism.2016; 31(3): 469. CrossRef - High expression of C-C chemokine receptor 2 associates with poor overall survival in gastric cancer patients after surgical resection
Ruochen Li, Heng Zhang, Hao Liu, Chao Lin, Yifan Cao, Weijuan Zhang, Zhenbin Shen, Jiejie Xu
Oncotarget.2016; 7(17): 23909. CrossRef - Polycationic carbosilane dendrimer decreases angiogenesis and tumor-associated macrophages in tumor-bearing mice
Ana Judith Perisé-Barrios, María Jesús Serramia, Javier de la Mata, Rafael Gomez, Angel Luis Corbí, Ángeles Domínguez-Soto, María Ángeles Muñoz-Fernandez
RSC Advances.2015; 5(126): 104110. CrossRef
 PubReader
PubReader ePub Link
ePub Link-
 Cite this Article
Cite this Article
- Cite this Article
-
- Close
- Download Citation
- Close
- Figure



Fig. 1.
Fig. 2.
Fig. 3.
| Charaacteristic | No. | CD163 (%) | p-value |
|---|---|---|---|
| Lung | |||
| Histology | |||
| Adenocarcinoma | 19 | 5.8 ± 4.4 | .428 |
| SQCC | 20 | 5.9 ± 4.5 | |
| Large cell carcinoma | 4 | 8.7 ± 7.3 | |
| SCC | 2 | 1.0 ± 0.5 | |
| Mixed carcinoma | 4 | 5.0 ± 2.3 | |
| Stage |
|||
| I | 16 | 5.7 ± 4.4 | .821 |
| II | 12 | 6.6 ± 5.2 | |
| III | 10 | 5.5 ± 3.8 | |
| IV | 1 | 2.4 | |
| Survival | |||
| Alive | 15 | 3.6 ± 3.4 | .104 |
| Death | 15 | 5.9 ± 3.9 | |
| Breast | |||
| Infiltrating ductal | 45 | 4.0 ± 3.4 | .055 |
| Medullary | 2 | 9.3 ± 3.4 | |
| Sarcomatoid | 1 | 1.9 | |
| Metaplastic | 1 | 10.1 | |
| Stage |
|||
| IIA | 13 | 5.3 ± 4.9 | .060 |
| IIB | 10 | 4.2 ± 2.4 | |
| IIIA | 12 | 1.8 ± 1.4 | |
| IIIB | 10 | 4.5 ± 2.3 | |
| Alive | 30 | 4.2 ± 3.7 | .726 |
| Death | 6 | 3.7 ± 2.5 | |
| Thyroid | |||
| PTC | 35 | 1.8 ± 1.3 | < .001 |
| ATC | 18 | 22.9 ± 17.1 | |
| Stage |
|||
| I | 3 | 1.4 ± 0.6 | .943 |
| II | 3 | 1.7 ± 1.1 | |
| III | 21 | 1.8 ± 1.2 | |
| IV | 4 | 1.8 ± 0.8 | |
| Alive | 15 | 1.8 ± 1.5 | .241 |
| Death | 2 | 3.3 ± 2.8 |
SQCC, squamous cell carcinoma; SCC; small cell carcinoma, PTC; papillary thyroid carcinoma, ATC; anaplastic thyroid carcinoma. p-value by ANOVA or student t test; TNM stage and survival was classified in adenocarcinoma and squamous cell carcinoma (lung), infiltrating ductal carcinoma (breast) and papillary thyroid carcinoma (thyroid).

 E-submission
E-submission