Articles
- Page Path
- HOME > J Pathol Transl Med > Volume 56(3); 2022 > Article
-
Brief Case Report
Vascular Ehlers-Danlos syndrome with distinct histopathologic features -
Hee Sang Hwang1
 , Jin Woo Song2
, Jin Woo Song2 , Se Jin Jang1
, Se Jin Jang1
-
Journal of Pathology and Translational Medicine 2022;56(3):167-169.
DOI: https://doi.org/10.4132/jptm.2021.03.24
Published online: May 11, 2021
1Department of Pathology, Asan Medical Center, University of Ulsan College of Medicine, Seoul, Korea
2Department of Internal Medicine, Asan Medical Center, University of Ulsan College of Medicine, Seoul, Korea
- Corresponding Author: Hee Sang Hwang, MD, PhD Department of Pathology, Asan Medical Center, University of Ulsan College of Medicine, 88 Olympic-ro 43-gil, Songpa-gu, Seoul 05505, Korea Tel: +82-2-3010-1869, Fax: +82-2-472-7898, E-mail: hshwang0908@amc.seoul.kr
© 2022 The Korean Society of Pathologists/The Korean Society for Cytopathology
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/4.0) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
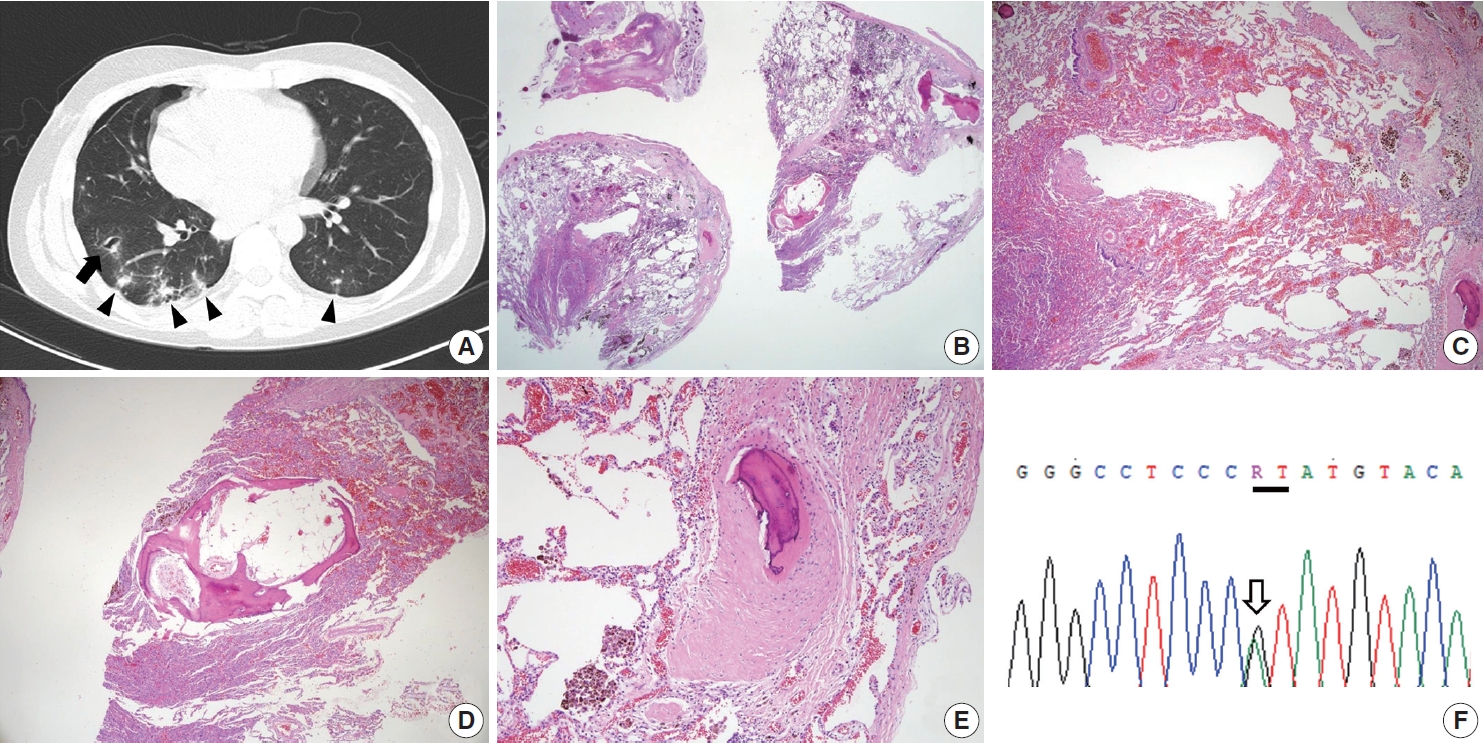
- A 19-year-old male was admitted to our hospital for recurrent episodes of spontaneous pneumothorax (6 times), which started about 8 months ago. His father had a history of recurrent pneumothorax and also suffered from spontaneous intraabdominal bleeding due to aneurysmal rupture of the common hepatic artery 7 years ago. The patient had undergone wedge resection of the lung at another hospital, after which he experienced a secondary pneumothorax, but no specific pathologic diagnosis was made. Thoracic computed tomography at our hospital showed multiple parenchymal cystic lesions with surrounding ground-glass opacity and several small nodular lesions in both lungs (Fig. 1A), suggesting vasculitis with hemorrhage, infected cystic lung disease, or paragonimiasis. However, neither serum anti-nuclear antibody nor anti-Paragonimus Westermani antibody titer was elevated. No abnormality in the thoracic greater vessels was identified. Abnormalities of the extrathoracic organs were not evaluated at the time of the admission. Pathological examination of the wedge-resected lung specimen slides sent from an external pathology laboratory revealed intra-alveolar accumulation of red blood cells and hemosiderin-laden macrophages (Fig. 1B, C), indicating recurrent episodes of intra-alveolar hemorrhage. The most diagnostic histologic finding was the presence of several round fibro-osseous nodules scattered in a hemorrhagic background of lung parenchyma (Fig. 1D, E). Disruption of the vascular wall was not identified, even in the examination under van Gieson elastic staining. Due to suspicion of vEDS based on the patient’s familial history and the histopathologic findings, COL3A1 gene sequencing was performed. The sequencing test revealed a heterozygous germline mutation in the spliceosome acceptor site in intron 23 of the COL3A1 gene (COL3A1 c. 1662 + 1G > A [p. Gly537_Pro554del]) (Fig. 1F), a known pathogenic mutation in the ClinVar database (https://www.ncbi.nlm.nih.gov/clinvar/variation/101269/). Hence, the patient was diagnosed with vEDS.
CASE REPORT
- vEDS comprises about 4%–5% of Ehlers-Danlos syndrome cases and has a high rate of fatal complications related to cardiovascular and gastrointestinal systems, such as cerebrovascular hemorrhage or intestinal rupture. In addition, pregnancy can be fatal in patients with vEDS who might develop ruptures in the uterus or uterine vessels [1]. Therefore, prompt diagnosis is the mainstay of medical management of vEDS to lower the rate of such fatal complications [2]. However, the rarity of vEDS makes its diagnosis challenging, frequently leading to delayed diagnosis. The pathologic diagnosis of vEDS is challenging because the characteristic histopathological findings of vEDS are not widely recognized. Abrupt caliber changes in the muscularis propria with diverticular formation have been identified in colon resection specimens of vEDS patients [3]. Kawabata et al. [4] reported that fragile lung parenchyma with spontaneous laceration, acute hemorrhage and/or hematoma, and fibro-osseous nodules, which are hypothesized to be a consequence of the organization of hematomas, are frequently identified in surgical lung biopsy specimens of vEDS patients, findings which serve as clues for pathologic diagnosis. In line with these findings, a previous report described a case of vEDS that was diagnosed based on identification of fibro-osseous nodules and vascular disruption in the lung parenchyma, suggesting the potential of histopathologic features in clinical diagnosis of vEDS [5]. Therefore, the distinct histopathologic features of vEDS should be considered when assessing surgical lung biopsy specimens from patients with recurrent spontaneous pneumothorax and/or hemorrhagic lung parenchymal cysts.
- In conclusion, identifying characteristic histopathologic features in surgical lung biopsy samples, such as parenchymal fragility, spontaneous laceration, parenchymal hemorrhage, and fibroosseous nodules, can contribute to early clinical diagnosis and proper management of vEDS.
DISCUSSION
Ethics Statement
This case study was approved by the Institutional Review Board of Asan Medical Center (2020-1889), which waived the requirement for informed consent.
Availability of Data and Material
Data sharing not applicable to this article as no datasets were generated or analysed during the study.
Code Availability
Not applicable.
Author Contributions
Conceptualization: SJJ, JWS, HSH. Data curation: HSH. Formal analysis: HSH, SJJ. Investigation: HSH. Methodology: HSH, JWS. Resources: SJJ, JWS. Supervision: SJJ. Writing—original draft: HSH. Writing—review & editing: HSH, SJJ. Approval of final manuscript: all authors.
Conflicts of Interest
The authors declare that they have no potential conflicts of interest.
Funding Statement
No funding to declare.

- 1. Pepin M, Schwarze U, Superti-Furga A, Byers PH. Clinical and genetic features of Ehlers-Danlos syndrome type IV, the vascular type. N Engl J Med 2000; 342: 673-80. ArticlePubMed
- 2. Shalhub S, Black JH 3rd, Cecchi AC, et al. Molecular diagnosis in vascular Ehlers-Danlos syndrome predicts pattern of arterial involveent and outcomes. J Vasc Surg 2014; 60: 160-9. PubMedPMC
- 3. Blaker H, Funke B, Hausser I, Hackert T, Schirmacher P, Autschbach F. Pathology of the large intestine in patients with vascular type Ehlers-Danlos syndrome. Virchows Arch 2007; 450: 713-7. ArticlePubMedPDF
- 4. Kawabata Y, Watanabe A, Yamaguchi S, et al. Pleuropulmonary pathology of vascular Ehlers-Danlos syndrome: spontaneous laceration, haematoma and fibrous nodules. Histopathology 2010; 56: 944-50. ArticlePubMed
- 5. Berezowska S, Christe A, Bartholdi D, Koch M, von Garnier C. Pulmonary fibrous nodule with ossifications may indicate vascular Ehlers-Danlos syndrome with missense mutation in COL3A1. Am J Respir Crit Care Med 2018; 197: 661-2. ArticlePubMed
REFERENCES
Figure & Data
References
Citations

- Variante no descrita previamente en el gen COL3A1: causalidad del Síndrome de Ehlers-Danlos tipo Vascular
Estrella Gutiérrez Romero, Nuria Padilla Apuntate, Silvia Izquierdo Álvarez
Advances in Laboratory Medicine / Avances en Medicina de Laboratorio.2025; 6(4): 504. CrossRef - A de novo variant of the COL3A1 gene: causality of vascular Ehlers-Danlos syndrome
Estrella Gutiérrez Romero, Nuria Padilla Apuntate, Silvia Izquierdo Álvarez
Advances in Laboratory Medicine / Avances en Medicina de Laboratorio.2025; 6(4): 500. CrossRef - Uncommon association between vascular Ehlers–Danlos syndrome and ocular complications
Matei Popa Cherecheanu, Mihaela Oana Romanitan, Ruxandra Pirvulescu, Raluca Iancu, Gerhard Garhöfer, George Iancu, Alina Popa Cherecheanu, Mihail Zemba, Victor Vasile, Andrei Simonov, Daniel Branisteanu
Frontiers in Medicine.2023;[Epub] CrossRef
 PubReader
PubReader ePub Link
ePub Link-
 Cite this Article
Cite this Article
- Cite this Article
-
- Close
- Download Citation
- Close
- Figure


 E-submission
E-submission