Articles
- Page Path
- HOME > J Pathol Transl Med > Volume 46(1); 2012 > Article
-
Original Article
Evaluation of Urine Cytology in Urothelial Carcinoma Patients: A Comparison of CellprepPlus® Liquid-Based Cytology and Conventional Smear - Seung-Myoung Son,, Ji Hae Koo,, Song-Yi Choi, Ho-Chang Lee, Yong-Moon Lee, Hyung Geun Song, Hae-Kyung Hwang, Hye-Suk Han1, Seok-Joong Yun2, Wun-Jae Kim2, Eun-Joong Kim3, Ok-Jun Lee
-
Korean Journal of Pathology 2012;46(1):68-74.
DOI: https://doi.org/10.4132/KoreanJPathol.2012.46.1.68
Published online: February 23, 2012
Department of Pathology, Chungbuk National University College of Medicine, Cheongju, Korea.
1Department of Internal Medicine, Chungbuk National University College of Medicine, Cheongju, Korea.
2Department of Urology, Chungbuk National University College of Medicine, Cheongju, Korea.
3Department of Clinical Laboratory Science, Juseong University, Cheongwon, Korea.
- Corresponding Author: Ok-Jun Lee, M.D. Department of Pathology, Chungbuk National University College of Medicine, 410 Seongbong-ro, Heungdeok-gu, Cheongju 361-711, Korea. Tel: +82-43-269-6260, Fax: +82-43-269-6269, ok5218@hanmail.net
- *Seung-Myoung Son and Ji Hae Koo contributed equally to this work.
© 2012 The Korean Society of Pathologists/The Korean Society for Cytopathology
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/3.0) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
Abstract
-
Background
- Urine cytology is an important test in the screening of urothlelial neoplasms. The conventional smear (CS) method of testing urine samples has a low sensitivity, approximately 50% result accuracy for detecting urothelial carcinomas, while liquid-based cytology (LBC) has much improved diagnostic accuracy, sensitivity, and specificity. The aim of this study was to compare the morphologic features and diagnostic efficacy of CellprepPlus® LBC with those of CS for urine cytology.
-
Methods
- A total of 713 cases of urine specimens collected from November 2009 to September 2010 were included. All specimens were divided equally for the preparation of CellprepPlus® LBC and CS for each case.
-
Results
- CellprepPlus® revealed more cellularity, a cleaner background and better cytomorphologic features, but it showed a less intact architectural pattern compared to that of CS. Of the 88 histologically confirmed cases, the diagnostic sensitivity for CellprepPlus® was 50% and higher than the 37.5% for CS. The specificity of both preparations was 100%.
-
Conclusions
- The CellprepPlus® showed an improved quality of slides and provided better diagnostic accuracy, thus CellprepPlus® could be a first-line screening tool in urinary tract cytology.
- The study included 713 cases of voided urine specimens that were collected from November 2009 to September 2010 and submitted to the department of Pathology of Chungbuk National University Hospital. The amount of urine per case was approximately 50 mL and all specimens were divided equally for the preparation of one CellprepPlus® (BIODYNE, Seongnam, Korea) LBC and one CS slide. The CS slides were prepared by cytocentrifugation for 10 minutes at 2,000 rpm. After centrifugation, the CS slides were stained on an autostainer and coverslipped. The LBC slides were prepared by cytocentrifugation for 10 minutes at 2,000 rpm. The supernatant was decanted, the remaining pellet was vortexed and added to preservation liquid, and then the bottle of preservation liquid was placed onto the CellprepPlus® device. After that, CellprepPlus® filtered out the cells and blew them from the filter to the slide using blowing method. Once smearing was completed, the slide transferred automatically to fixing alcohol. The whole process only takes 26 seconds in the device.
- All of the LBC and CS slides were screened by two cytotechnicians and then independently confirmed by two pathologists. After all reviewers examined the LBC slides, the CS slides were diagnosed with no information about the results of the LBC slides. The final diagnosis was made by putting the two results together. The cytological findings were categorized into adequate or inadequate specimen, and then the adequate specimens were classified according to four categories: 1) negative, 2) atypical, 3) suspicious, and 4) malignant. Eighty-eight out of 713 cases (12.3%) underwent transurethral resection or cystoscopic biopsy in the urinary tract, and the cytologic diagnosis was confirmed by histologic diagnosis. The histological diagnoses were divided into two groups, benign and malignant, and the malignant category was classified as low or high grade urothelial carcinoma. In 51 out of the 88 histologically confirmed cases (58.0%), 19 cases diagnosed as more than atypical on both LBC and CS slides were included to compare the general cytologic features of urothelial carcinomas including cellularity, cell distribution, cohesiveness, cell size, nuclear size, nuclear hyperchromasia, and nuclear detail. The sensitivity, specificity, positive predictive value, and negative predictive value were also compared according to the two methods. A statistical analysis including sensitivity, specificity, and positive and negative predictive values was performed using SPSS ver. 12.0 (IBM Inc., Somers, NY, USA).
MATERIALS AND METHODS
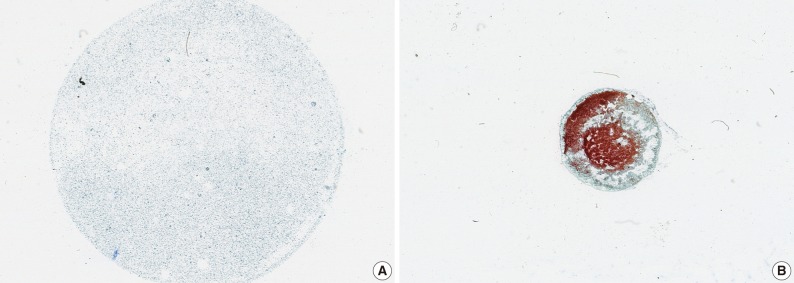
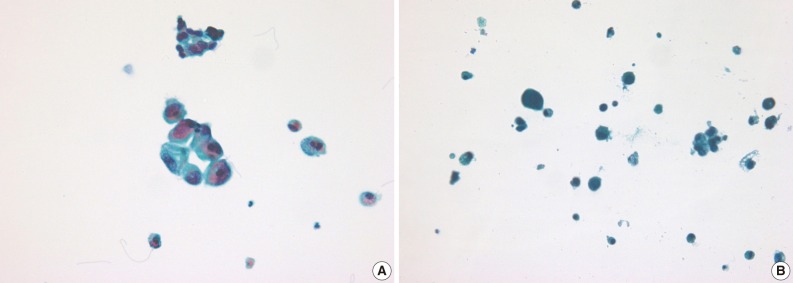
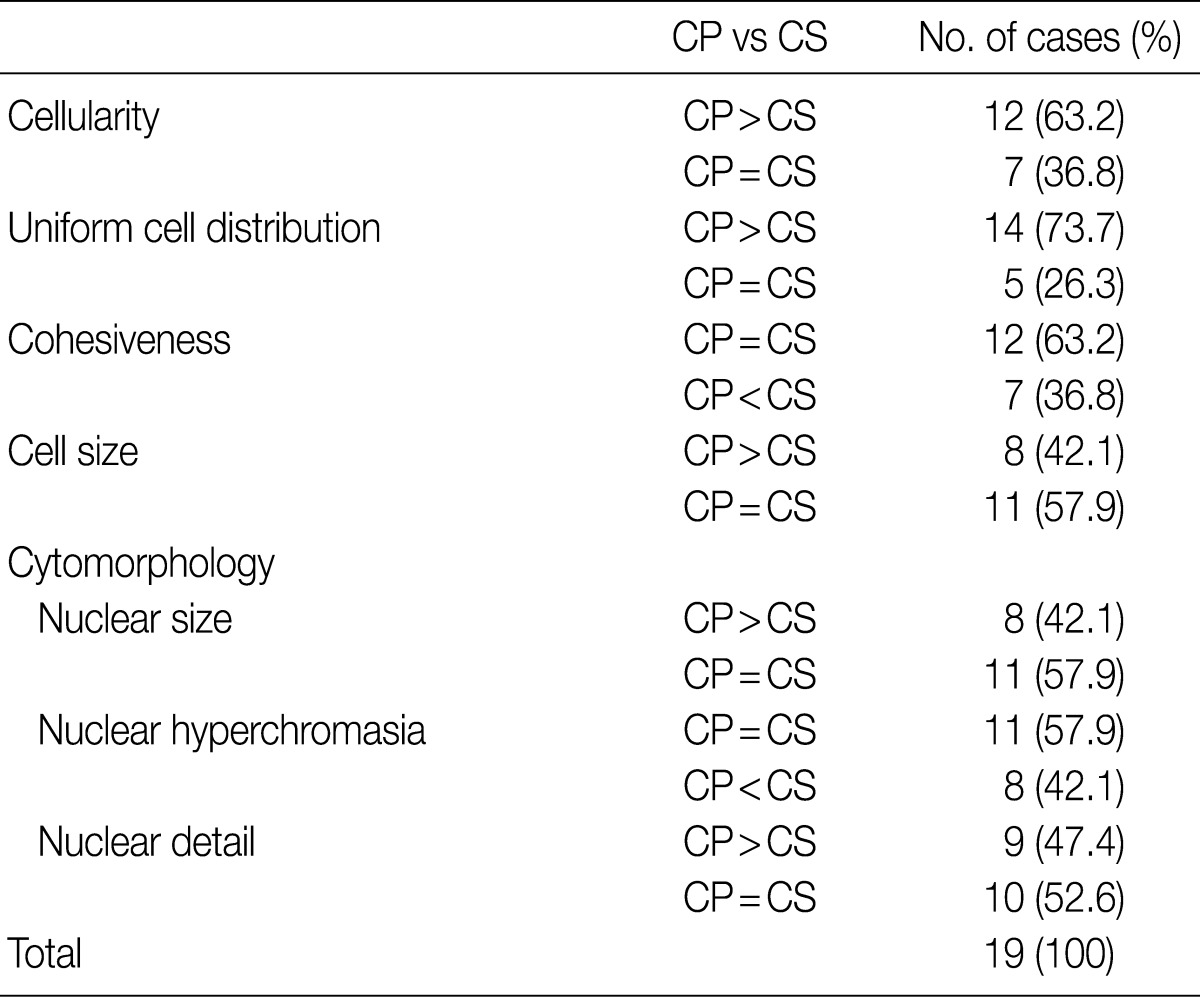
- LBC produced a well-defined circular area measuring 20 mm in diameter which consists of uniformly distributed cells in a thin layer (Fig. 1). All the LBC slides had greater or equivalent cellularity and uniformity compared to those of CS. The architectural features from LBC were less intact and cohesive than those of CS in 7 of 19 cases (36.8%). In two cases, CS preserved the papillary structures of urothelial carcinoma, whereas in the same specimen processed with LBC, there were only loose clusters or isolated single cells instead of the papillary structures. These findings probably resulted from the cell filtration step during processing the specimen (Fig. 2). Cells and nuclei appeared larger on LBC than CS in 8 cases, and the remaining 11 cases had no differences. Nuclear hyperchromasia was present in all 19 cases on both LBC and CS slides but was more pronounced on CS slides. LBC preserved nuclear details such as pleomorphism, membrane irregularity, chromatin changes, and nucleoli better than CS in 9 cases (Table 1, Fig. 3).
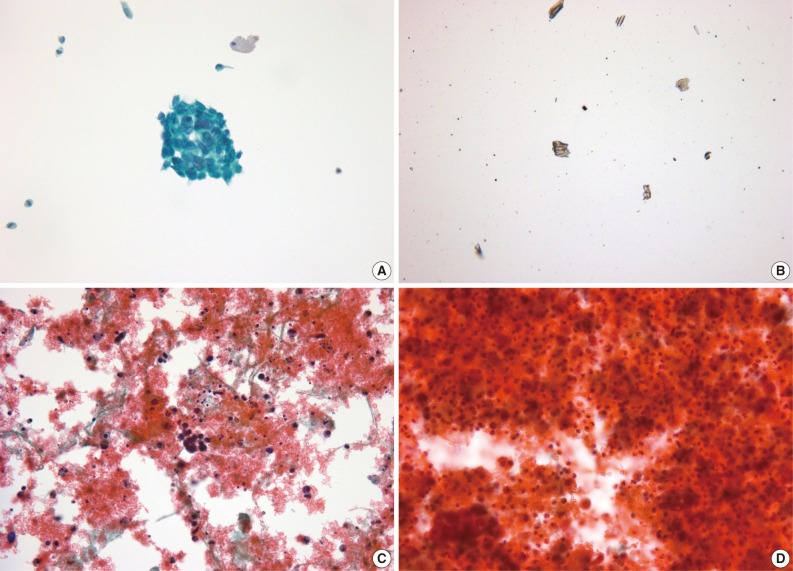
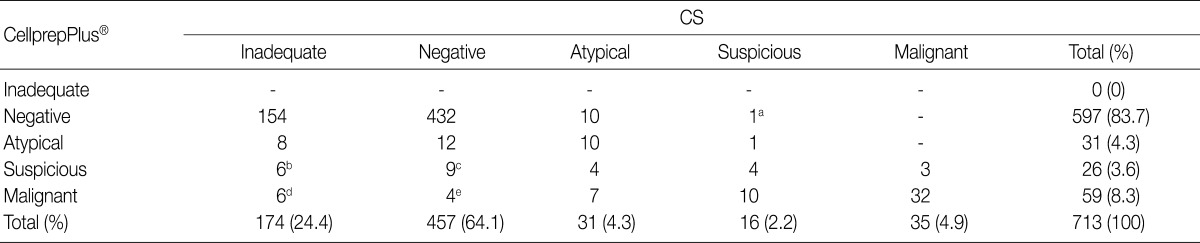
- All 713 LBC slides were similar in quality and appropriate for diagnosis. However, 174 of 713 cases (24.4%) were inadequate for diagnosis from CS. Of the 174 cases, 27 were bloody, 5 were interfered with by inflammatory cells, 140 showed a significant loss of cells, and 2 were smeared too thick (Fig. 4, Table 2).
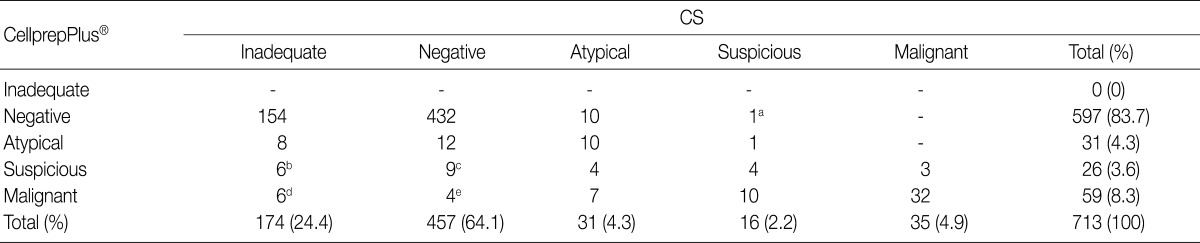
- In the 174 cases of inadequate specimens from CS, 6 were interpreted as suspicious, and 6 were malignant by LBC. Of these 12 cases, 3 were histologically confirmed to be malignant. Four hundred and fifty-seven cases were diagnosed as negative by CS. Of these cases, 12 were atypical, and 13 were suspicious or malignant by LBC. Three of these 13 cases were histologically confirmed to be malignant. Thirty-one cases were atypical by CS, whereas only 10 of them were atypical by LBC. Of the remaining 21 cases, 11 were suspicious or malignant by LBC. In the 16 cases of suspicious specimen from CS, 1 was atypical and 1 revealed negative by LBC. In 35 cases of malignant specimen from CS, 3 were suspicious, and 32 were also malignant by LBC (Table 3).
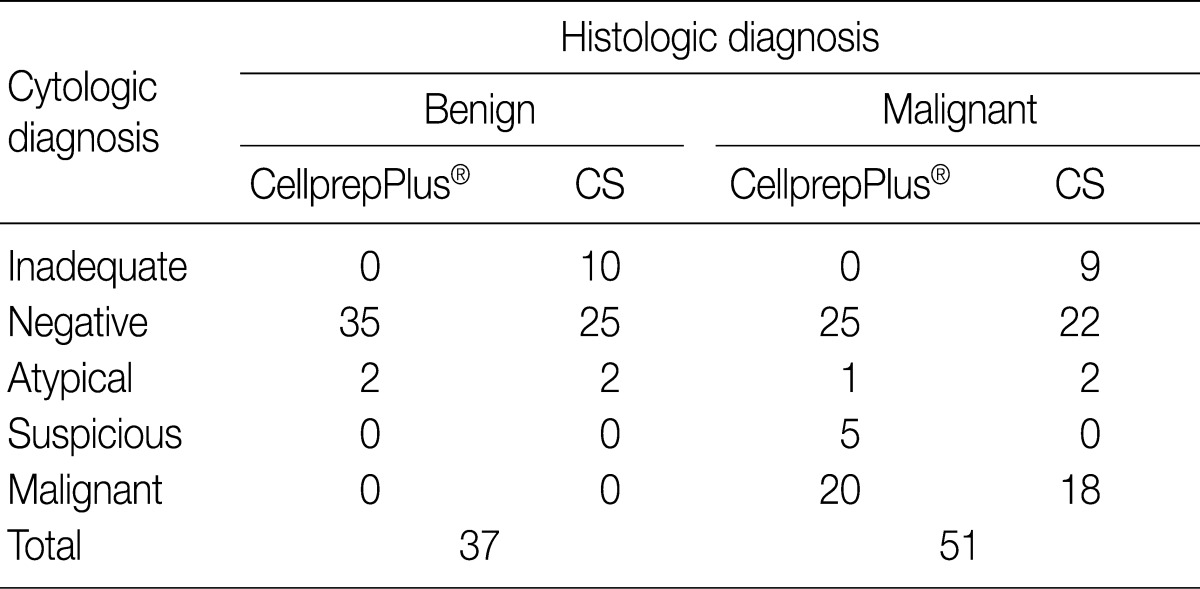
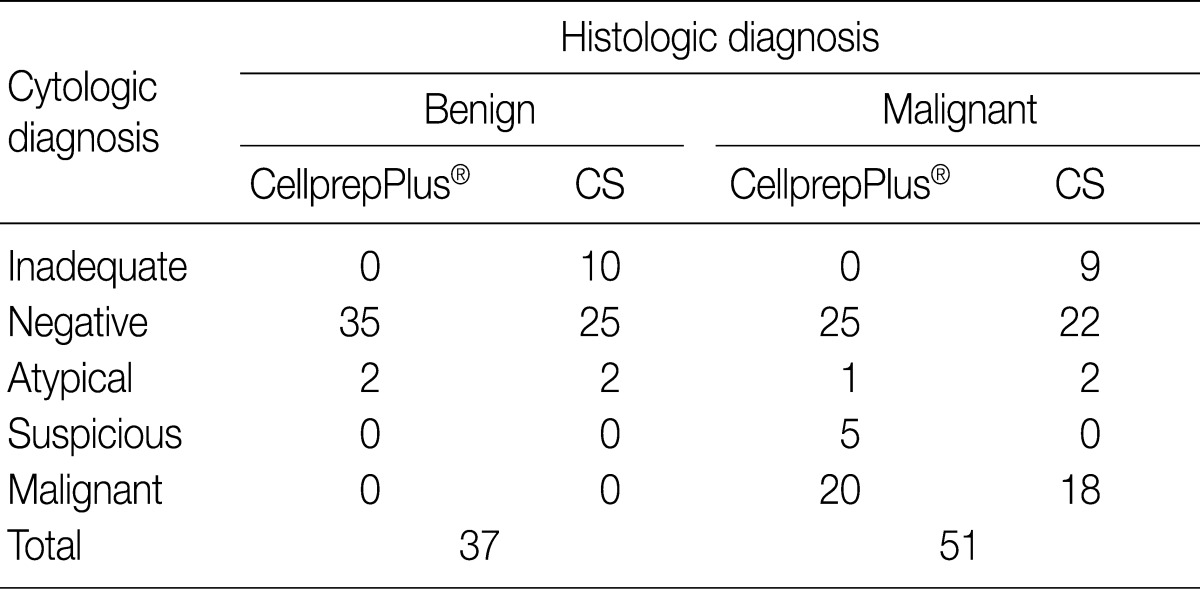
- A histological diagnosis was made in 88 cases. Fifty-one out of the 88 cases (58.0%) were histologically confirmed to be malignant. Of these 51 cases, CS diagnosed 9 as inadequate, 22 as negative, 2 as atypical and 18 as malignant. On the contrary, 25 cases were negative, 1 was atypical, 5 were suspicious, and 20 were malignant by LBC. In the remaining 37 cases (42.0%) were histologically benign lesions. Two out of 37 cases were cytologically atypical both on the LBC and CS slides (Table 4).
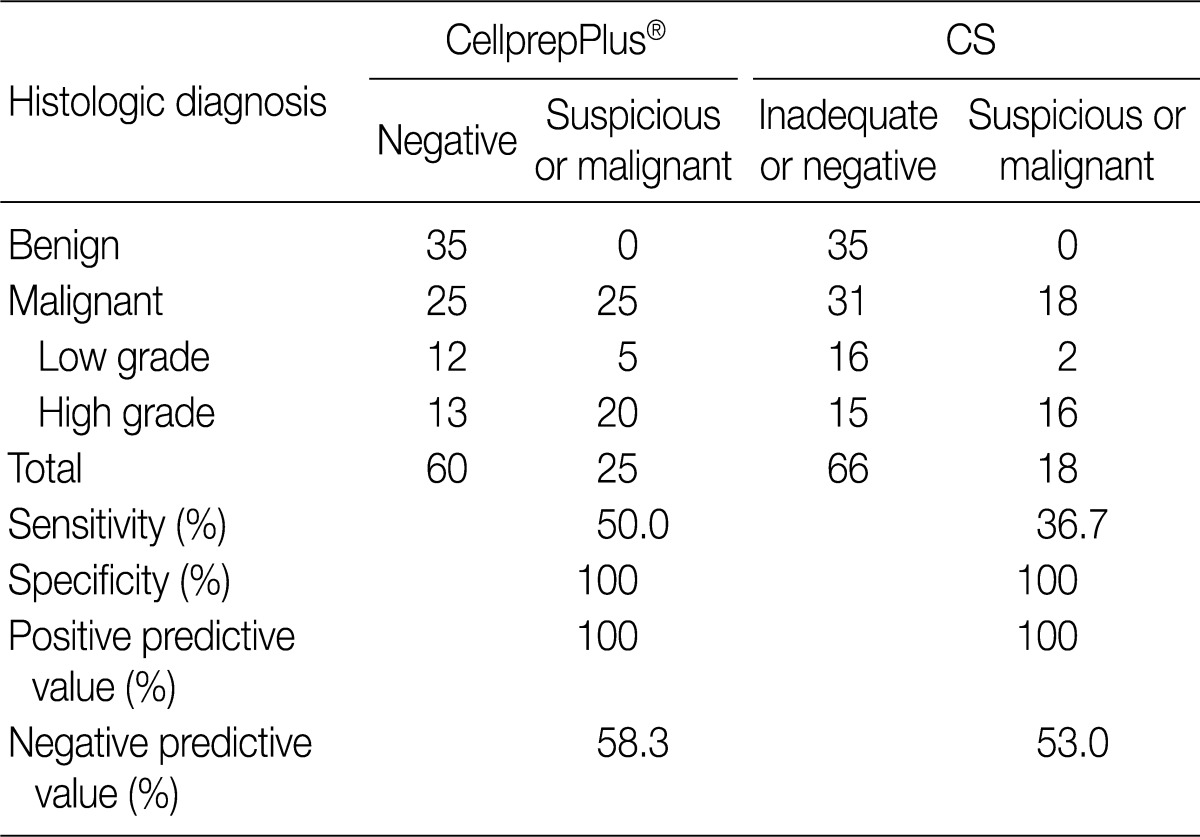
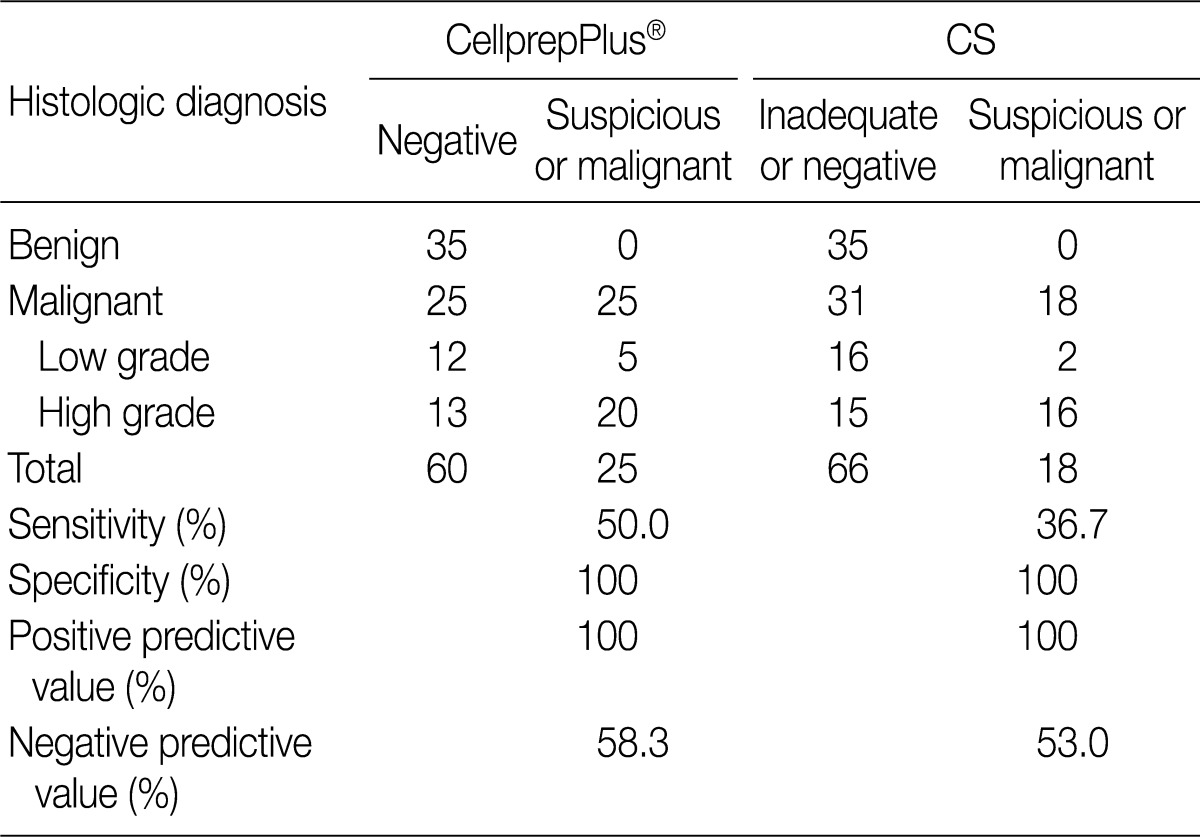
- Except the cases diagnosed as atypical, cytologic diagnosis were reclassified. The "negative" and "inadequate" slides went into a "benign" category, and the "suspicious" and "malignant" ones were placed under a "malignant" category. The sensitivity and specificity of each preparation were 50.0% and 100% by LBC, and 36.7% and 100% by CS, respectively. The positive and negative predictive values were 100% and 58.3% by LBC, and 100% and 53.0% by CS, respectively. The malignant category was also divided into low and high grade urothelial carcinoma. The sensitivity of the former was 29.4% by LBC and 11.1% by CS, and the latter was 60.6% by LBC and 51.6% by CS (Table 5).
RESULTS
- Since 1939, the screening of malignant neoplasms has relied in large part on cytology. However, the obstacles of CS such as thick smear, overlapping cellular areas, low cellularity, obscuring inflammatory cells, blood, and air-drying artifacts have made diagnosis difficult and resulted in a low diagnostic sensitivity. Thus, LBC was introduced as a replacement for the conventional method, and it has been increasingly used over the past two decades.5,6
- There have been several studies comparing LBC with CS for non-gynecologic cytology. In the analysis of 236 urine samples, Lee et al.7 demonstrated that the use of ThinPrep® liquid-based preparations method was beneficial to improve the quality of the slides and reduced the duration of a microscopic examination, but did not show better sensitivity, accuracy and predictive values. Koo et al.6 noted that using CellprepPlus® LBC for body fluid showed better sensitivity and negative predictive values, and it produced a higher quality of slide than CS, making it suitable in screening body fluid as a cytologic diagnostic tool.
- Conventional methods of urine cytology have included cytocentrifugation, millipore filtration, and direct smearing. In this technique, cytocentrifuge processing may result in a low cell yield and non-uniform and thickly smeared cells with poor cellular preservation. Unlike urine, other gynecologic and non-gynecologic conventional preparations generally have no problems with low cellularity. On the contrary, their high cellular contents require more screening time because of the increased number of slides per specimen and the non-uniform screening area.6,9-10 With LBC, instead of being smeared, cells are rinsed into a liquid collection medium and processed automatically. When compared with CS, LBC shows a higher cell yield and it reduces obscuring elements, therefore, LBC solves one of the big issues of low cellularity in urine cytology.3
- Two different liquid-based preparatory techniques were developed. One is based on filtration and a computer-assisted thin-layer deposition of cells, and the other is a sedimentation process. CellprepPlus® used in this study is based on a filtration process and has its own special blowing technology that was developed in Korea. CellprepPlus® transfer cells from a preservation liquid to a slide in a two-step process: 1) once the preservation liquid bottle was placed into the CellprepPlus® device, it filters out cells with its own pressure, and 2) it blows cells from the filter to the slide using air pressure, resulting in the cells being transferred to a 20 mm circular area in a thin layer. In the study that compared CellprepPlus® with SurePath™, Kim et al.8 reported that CellprepPlus® gave non-inferior results to those of SurePath™, and 1 case was diagnosed as atypical by SurePath™ turned out to be malignant by CellprepPlus®. Koh et al.9 also reported that Cellprep® gave comparable results to those of ThinPrep® in terms of smear quality and the cytological diagnostic efficacy, and Cellprep® was available on immunocytochemistry.
- In this study, the sensitivity of CellprepPlus® LBC and CS was 29.4% and 11.1% in low grade urothelial carcinoma, and 60.6% and 51.6% in high grade urothelial carcinoma, respectively. When the low and high grade carcinomas were put together under one "malignant" category, the sensitivity of CellprepPlus® LBC and CS was 50.0% and 36.7%, respectively, and the specificity of both techniques was 100%. The negative predictive value was 58.3% and 53.0%, respectively, and the positive predictive value was 100% for both. The sensitivity and negative predictive values of CellprepPlus® LBC are superior to those of CS in detecting urothelial tumors.
- As malignant urothelial cells were distributed in a thin layer with less overlapping on CellprepPlus® LBC, there was no inadequacy due to low cellularity or thick preparation compared with CS processing. This is consistent with the earlier studies reported in the literature.2-10 CellprepPlus® LBC also shows a cleaner background with only a small amount of blood, inflammatory cells, and debris as reported in previous studies;5,6,9 therefore, CellprepPlus® LBC can identify malignant cells easier. In terms of the architectural pattern, LBC was less cohesive than CS, and in some cases, CS preserves papillary pattern, while CellprepPlus® LBC shows ball-shaped loose clusters of cells or scattered single cells. It is thought that the cell filtration during processing influenced the less cohesive pattern of CellprepPlus® LBC. However, it does not affect the accuracy of the diagnosis. This study also demonstrated that CellprepPlus® LBC generally reveals enlarged cells compared to those of CS. Both CellprepPlus® LBC and CS show nuclear features characteristics of urothelial carcinomas. However, CellprepPlus® LBC displays more enlarged and translucent nuclei, making it easier to recognize nuclear details such as nucleoli or chromatin changes.
- To summarize, CellprepPlus® LBC is notable for its increased cellularity, clean background, and increased maintenance of cytomorphologic features, thus it provides a more definitive diagnosis and could replace the conventional preparations. LBC is more cost-effective than CS in that the increased quality of the slides reduces the needs for repeated examinations. Furthermore, it has been revealed that the residual samples could be used to process multiple slides for ancillary tests for immunocytochemistry and molecular tests.
DISCUSSION
Acknowledgments
Acknowledgments
- 1. Eble JN, Sauter G, Epstein JI, Sesterhenn I. World Health Organization classification of tumors. Pathology and genetics of tumours of the urinary system and male genital organs. 2005; Lyon: IARC Press.
- 2. Shin BK, Lee YS, Jeong H, et al. Detecting malignant urothelial cells by morphometric analysis of ThinPrep® liquid-based urine cytology specimens. Korean J Cytopathol 2008; 19: 136-143. Article
- 3. Hoda RS. Non-gynecologic cytology on liquid-based preparations: a morphologic review of facts and artifacts. Diagn Cytopathol 2007; 35: 621-634. ArticlePubMed
- 4. Lu DY, Nassar A, Siddiqui MT. High-grade urothelial carcinoma: comparison of SurePath™ liquid-based processing with cytospin processing. Diagn Cytopathol 2009; 37: 16-20. ArticlePubMed
- 5. Koo JH, Lee SY, Lee HC, et al. CellprepPlus® Liquid-based Smear in Sono-guided Thyroid Fine Needle Aspiration: A Comparison of Conventional Method and CellprepPlus® Liquid-based Cytology. Korean J Pathol 2011; 45: 182-187. Article
- 6. Koo JH, Lee HC, Song HG, et al. Comparison of cytologic evaluation between conventional method and CellprepPlus® liquid-based cytology in body fluid. Korean J Pathol 2009; 43: 448-452. Article
- 7. Lee S, Park JH, Do SI, et al. Diagnostic value of urine cytology in 236 cases: a comparison of liquid-based preparation and conventional cytospin method. Korean J Cytopathol 2007; 18: 119-125.
- 8. Kim H, Hwang DH, Park SY, Chung JH, Choe G. A pilot comparative study: Cellprep®Plus LBC system and SurePath™ in cervicovaginal and urine cytology 2009; The 21st Annual Fall Meetings of the Korean Society for Cytopathology; 2009 Nov 6-7; Hoengseong. Seoul: The Korean Society for Cytopathology, -52.
- 9. Koh JS, Cho SY, Ha HJ, Kim JS, Shin MS. Cytologic evaluation of CellPrep® liquid-based cytology in cervicovaginal, body fluid, and urine specimens: comparison with ThinPrep®. Korean J Cytopathol 2007; 18: 29-35.
- 10. Yim H, Joo HJ, Kim YB, Hong SW. Comparison of conventional smear, cell block and liquid-based preparation in the evaluation of bronchial washing specimen in lung cancer patients. Korean J Pathol 2011; 45: 296-302. Article
REFERENCES
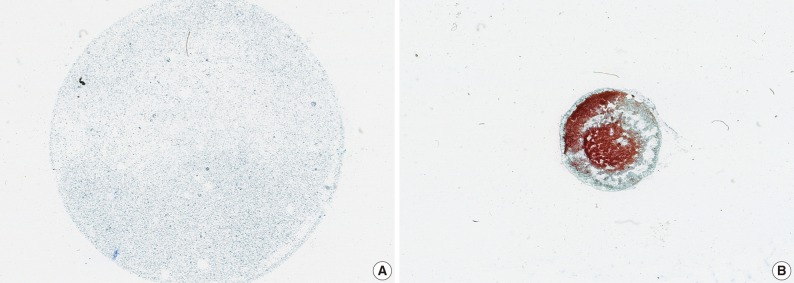
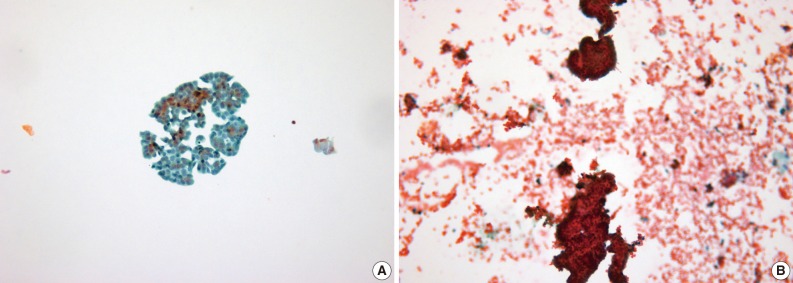

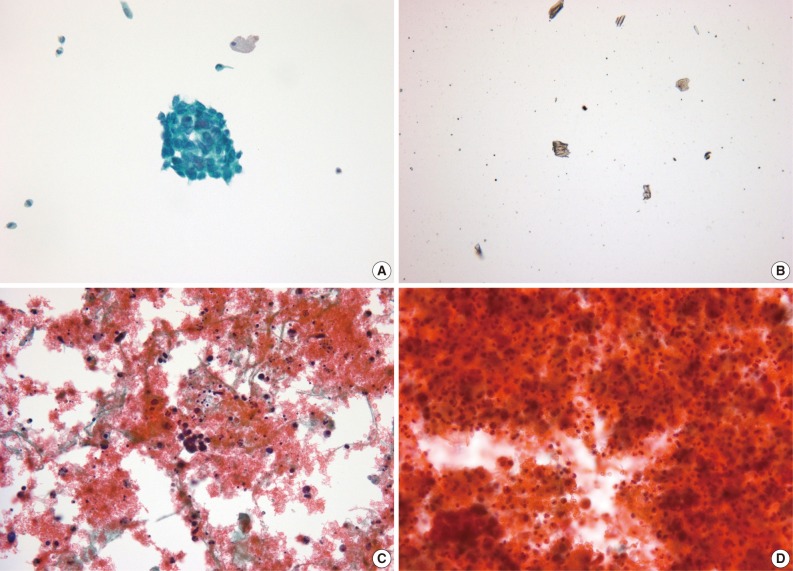
Figure & Data
References
Citations

- Comparison of Conventional Sediment Smears and Liquid‐Based Cytology and Utility of Cell Block Preparation in Urinary Cytology
Jagriti Singh Fartiyal, Meenakshi Rao, Poonam Abhay Elhence, Aasma Nalwa, Divya Aggarwal, Taruna Yadav, Gautam Ram Choudhary
Diagnostic Cytopathology.2026; 54(4): 285. CrossRef - A Study on the Workload of Cytotechnologists: Focus on Commercial Laboratories
Eun-Suk PARK
Korean Journal of Clinical Laboratory Science.2025; 57(2): 228. CrossRef - A Comparative Analysis of Diagnostic Performance and Cytomorphologic Features Between Newly Developed WellPrep® and SurePath™ in Serous Effusion Cytology
Ji Eun Choi, Min-Sun Jin, Da Sol Kim, Ilias P. Nikas, Han Suk Ryu
Journal of Cytology.2025; 42(3): 142. CrossRef - Advances in diagnostic liquid‐based cytology
Hideyuki Abe, Akihiko Kawahara, Jun Akiba, Rin Yamaguchi
Cytopathology.2024; 35(6): 682. CrossRef - Impact of implementing the first edition of the Paris system for reporting: A systematic review and meta‐analysis
Sahar J. Farahani, Joshua Li, Beatrice Minder, Philippe Vielh, Marija Glisic, Taulant Muka
Cytopathology.2024; 35(5): 616. CrossRef - Body fluids
Shyam H. Nemade, Meherbano M. Kamal
Indian Journal of Pathology and Microbiology.2023; 66(1): 75. CrossRef - Deep Learning-Based Screening of Urothelial Carcinoma in Whole Slide Images of Liquid-Based Cytology Urine Specimens
Masayuki Tsuneki, Makoto Abe, Fahdi Kanavati
Cancers.2022; 15(1): 226. CrossRef - Diagnostic efficacy of smear plus liquid-based cytology for EUS-FNA of solid pancreatic lesions
Masahiro Itonaga, Shin-Ichi Murata, Keiichi Hatamaru, Takashi Tamura, Junya Nuta, Yuki Kawaji, Takao Maekita, Mikitaka Iguchi, Jun Kato, Fumiyoshi Kojima, Hiroki Yamaue, Manabu Kawai, Ken-Ichi Okada, Seiko Hirono, Toshio Shimokawa, Kensuke Tanioka, Masayu
Medicine.2019; 98(19): e15575. CrossRef - Evaluation and application of Cellprep for cervical cytology
Rinko OZEKI, Keiichi IWAYA, Yuko UMAYAHARA, Yuka MORITA, Mie ARAI, Yukari TAKASUGI, Ryoko KIKUCHI, Kiyohiko MIYAKE, Atsuhiko SAKAMOTO, Masaru SAKAMOTO
The Journal of the Japanese Society of Clinical Cytology.2018; 57(3): 159. CrossRef - Quantitative Proteomic Analysis Identifies AHNAK (Neuroblast Differentiation-associated Protein AHNAK) as a Novel Candidate Biomarker for Bladder Urothelial Carcinoma Diagnosis by Liquid-based Cytology
Hyebin Lee, Kwangsoo Kim, Jongmin Woo, Joonho Park, Hyeyoon Kim, Kyung Eun Lee, Hyeyeon Kim, Youngsoo Kim, Kyung Chul Moon, Ji Young Kim, In Ae Park, Bo Bae Shim, Ji Hye Moon, Dohyun Han, Han Suk Ryu
Molecular & Cellular Proteomics.2018; 17(9): 1788. CrossRef - Reliability of Estrogen Receptor and Human Epidermal Growth Factor Receptor 2 Expression on Breast Cancer Cells Stored in Cellprep® Vials
Ayumi Ryu, Jyun-ichi Ashimura, Takahiro Nakayama, Yasuhiro Tamaki, Shin-ichi Nakatsuka, Yasuhiko Tomita
Acta Cytologica.2018; 62(5-6): 360. CrossRef - Current status of urine cytology: what should the urologist know?
Kristýna Pivovarčíková, Tomáš Pitra, Milan Hora, Marián Švajdler, Ondřej Hes
Czech Urology.2018; 22(4): 242. CrossRef - Morphologic Analysis of Cytomegalovirus Infected Cells in Bronchial Washing Cytology: Comparison of Liquid-Based Preparation and Conventional Smear
Jae Yeon Seok, Jungsuk An, Seung Yeon Ha, Dong Hae Chung, Sangho Lee, Hyunchul Kim
Journal of Pathology and Translational Medicine.2016; 50(2): 147. CrossRef - Romanowsky staining using liquid‐based cytology: A pilot study using Cytolyt®/HESPANDER® processing solution for ThinPrep® preparations
Yuichi Kinoshita, Takashi Yuri, Katsuhiko Yoshizawa, Kosho Takasu, Yuko Emoto, Airo Tsubura, Nobuaki Shikata
Diagnostic Cytopathology.2015; 43(12): 960. CrossRef - Diagnostic Value of Liquid-Based Cytology in Urothelial Carcinoma Diagnosis: A Systematic Review and Meta-Analysis
You Luo, Dong-Li She, Hu Xiong, Li Yang, Sheng-Jun Fu, Francisco X. Real
PLOS ONE.2015; 10(8): e0134940. CrossRef - Comparison of diagnostic accuracy between CellprepPlus® and ThinPrep® liquid‐based preparations in effusion cytology
Yong‐Moon Lee, Ji‐Yong Hwang, Seung‐Myoung Son, Song‐Yi Choi, Ho‐Chang Lee, Eun‐Joong Kim, Hye‐Suk Han, Jin young An, Joung‐Ho Han, Ok‐Jun Lee
Diagnostic Cytopathology.2014; 42(5): 384. CrossRef - Diagnostic Efficacy of Cell Block Immunohistochemistry, Smear Cytology, and Liquid-Based Cytology in Endoscopic Ultrasound-Guided Fine-Needle Aspiration of Pancreatic Lesions: A Single-Institution Experience
Shan-yu Qin, You Zhou, Ping Li, Hai-xing Jiang, Robert L. Schmidt
PLoS ONE.2014; 9(9): e108762. CrossRef




Fig. 1
Fig. 2
Fig. 3
Fig. 4
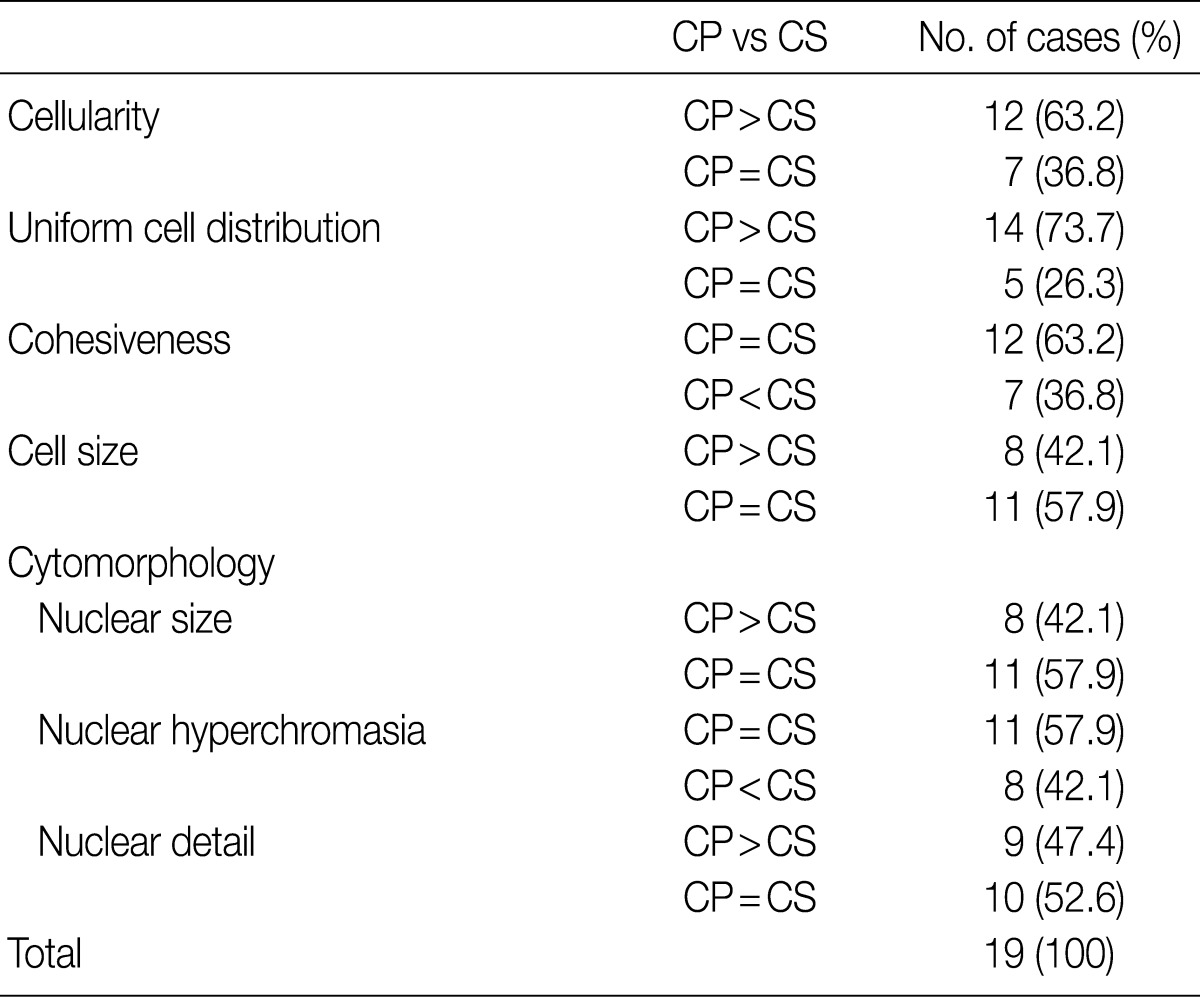
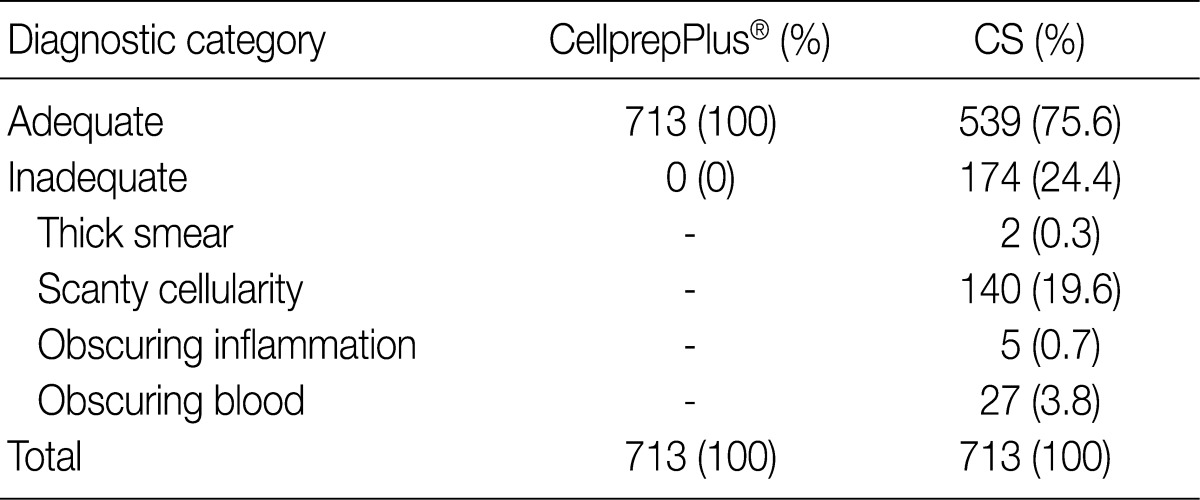
CP, CellprepPlus®; CS, Conventional smear.
CS, conventional smear.
CS, conventional smear. aHistologic confirmation had not made; bUrothelial carcinoma confirmed in 2 out of 6 cases; cUrothelial carcinoma confirmed in 2 out of 9 cases; dUrothelial carcinoma confirmed in 1 out of 6 cases; eUrothelial carcinoma confirmed in 1 out of 4 cases.
CS, conventional smear.
CS, conventional smear.

 E-submission
E-submission

 PubReader
PubReader Cite this Article
Cite this Article