Articles
- Page Path
- HOME > J Pathol Transl Med > Volume 46(1); 2012 > Article
-
Case Report
Fine Needle Aspiration Cytology of Metastatic Adenocarcinoma of the Gingiva from the Lung: A Case Report - Tack Kune You, So Ri Kim1, Ho Sung Park, Kyu Yun Jang, Woo Sung Moon, Myoung Ja Chung, Dong Geun Lee, Myoung Jae Kang
-
Korean Journal of Pathology 2012;46(1):101-104.
DOI: https://doi.org/10.4132/KoreanJPathol.2012.46.1.101
Published online: February 23, 2012
Department of Pathology, Chonbuk National University Medical School, Jeonju, Korea.
1Department of Internal Medicine, Chonbuk National University Medical School, Jeonju, Korea.
- Corresponding Author: Myoung Jae Kang, M.D. Department of Pathology, Chonbuk National University Medical School, San 2-20 Geumam-dong, Deokjin-gu, Jeonju 561-180, Korea. Tel: +82-63-270-3075, Fax: +82-63-270-3135, mjkang@jbnu.ac.kr
• Received: October 19, 2011 • Revised: December 27, 2011 • Accepted: January 12, 2012
© 2012 The Korean Society of Pathologists/The Korean Society for Cytopathology
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/3.0) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
- 8,864 Views
- 44 Download
- 2 Scopus
Abstract
- Metastases of malignant tumors to the oral region from distant sites are uncommon. A 45-year-old man with painless gingival swelling was diagnosed with adenocarcinoma of the lung. On cytology, clusters of tumor cells on mucous background revealed enlarged nuclei, indistinct cell borders, and irregular nuclear membranes. Some cells showed nuclear inclusions, nuclear grooves and small nucleoli. These findings are indicative of metastatic adenocarcinoma. We present a case of gingival metastasis from a lung adenocarcinoma.
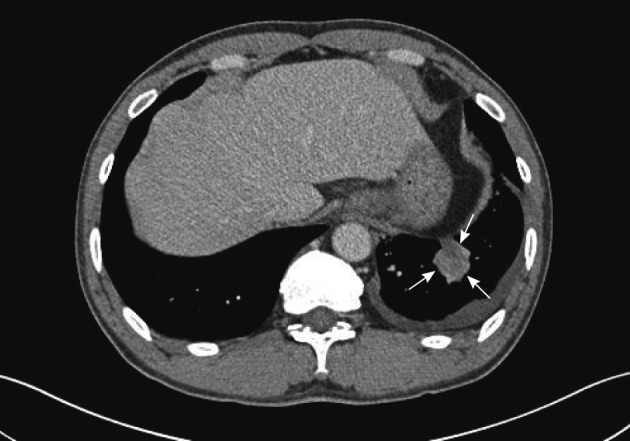
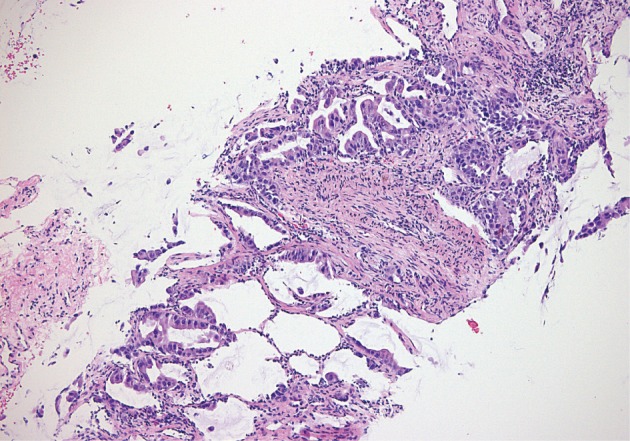
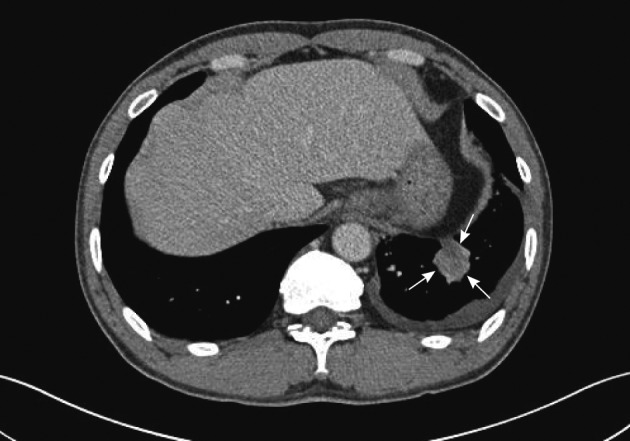
- A 45-year-old male complaining of chronic cough and sputum was referred to our hospital in October, 2010. Chest computed tomography (CT) scan demonstrated a left lower lobe mass of the lung (Fig. 1). Percutaneous transthoracic needle biopsy was performed on the left lower lobe mass of the lung. Histological features showed alveoli lined by large cuboidal to columnar tumor cells with moderate amount of eosinophilic cytoplasm and high nucleocytoplasmic ratio and large hyperchromatic nuclei (Fig. 2). Positron emission tomography-CT of the whole body revealed lung malignancy in the left lower lobe with multiple lymph nodes metastasis, hematogenous metastasis to both adrenal glands and multiple bones. This patient received chemotherapy and radiotherapy.
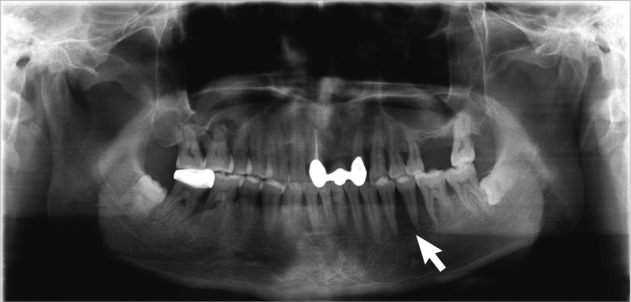
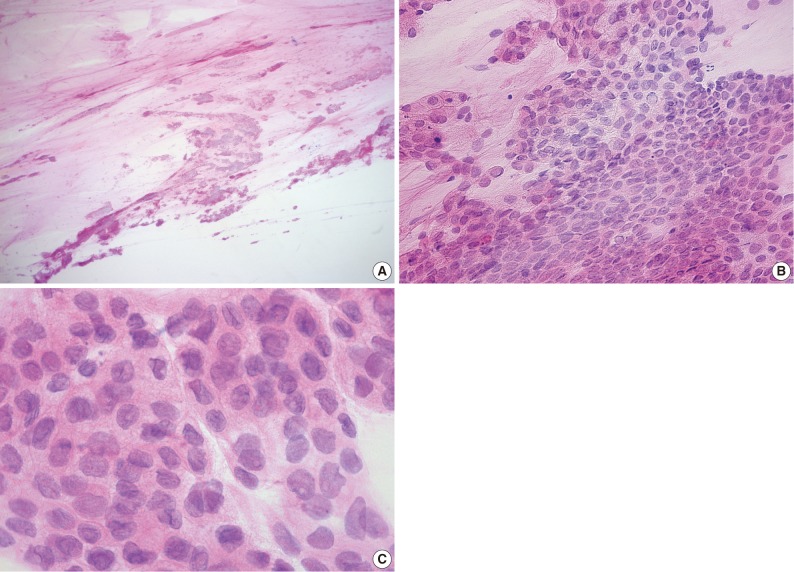
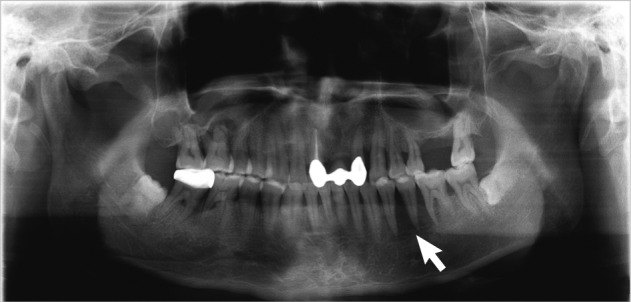
- In January, 2011, the patient had noticed painless gingival swelling in the left lower mandible for the past 3 weeks. Intraoral findings revealed an irregular-shaped reddish-pink soft to firm exophytic mass in the right lower mandibular gingiva measuring about 2×1.5 cm. A panoramic radiograph showed diffuse radiolucency at No. 34, 35, 36 regions (Fig. 3). Fine needle aspiration was performed on the gingival lesion, that was alcohol-fixed and hematoxylin and eosin stained. Cytological features showed moderate to high cellularity on mucous background (Fig. 4A). There were many cohesive clusters of epithelial tumor cells with a moderate amount of vacuolated eosinophilic cytoplasm, indistinct cell border, and increased nucleocytoplasmic ratio. Those cells had enlarged nuclei with irregular nuclear membranes, occasional nuclear inclusion and linear groove, and small nucleoli (Fig. 4B, C). The above cytologic findings are consistent with metastatic adenocarcinoma of the gingiva. After fine needle aspiration, the patient was referred to another hospital in January, 2011 and died one month later. Biopsy was not performed.
CASE REPORT
- Metastases of malignant tumors to the oral cavity are relatively uncommon. Only 25 of over 2,400 malignant oral cavity tumors are metastatic (approximately 1%), as reported by Meyer and Shklar.1 This report also showed that those tumors metastatic only to the oral mucosa were extremely rare, occupying only about 0.1% of all oral malignancy. The major primary tumors presenting oral metastases were the lung, kidney, liver, and prostate in men, breast, female genital organs, kidney and colorectum in women.5 Metastasis to oral cavity often have indistinct symptoms that mimic dental infections such as epulis, pyogenic granuloma, peripheral giant cell granuloma and usually have an undiagnosed primary malignancy at some other site.5
- Six-hundred and seventy-three cases of oral metastasis were reviewed by Hirshberg et al.5 in 2008. They reported that the most common primary site of oral cavity metastasis was in the lung (112 cases). In the 112 cases of oral metastasis, 54 were noted in the oral mucosa and another 58 in the jaw bones.5
- The pathogenesis of oral metastases is yet to be elucidated. Some authors suggested that circulating tumor cells may be entrapped in the rich capillary network of local chronic inflammation in the gingiva once the cells have reached the oral region. Because the patient had previously complained of poor oral hygiene such as ill-fitting denture and poorly fitting dental appliances suspected to be an inducing factor of local chronic inflammation in the mandibular gingiva.2 Five other reported cases of gingival metastasis did not explain if the patients had dental appliances or chronic inflammation, but interestingly, four of five cases were located in the soft tissue of mandibular gingiva. There may be some association with localization, chronic inflammation and blood supply.2 Another report proposed that the pathogenic mechanism of cervicofacial metastases would be the role of the prevertebral venous network without valves called by Batson's plexus, authorizing the retrograde crossing of tumoral cells from the lung toward the face.6 Finally, in the case of metastases to oral cavity of lung cancers, the direct bronchotracheal way could be accused.7
- Squamous cell carcinoma and adenocarcinoma of the lung have peculiar cytomorphological detail. Squmous cell carcinoma have cells arranged as sheet and syncytial groups, tumor diathesis, tadpole cell, bizarre cell shapes, dense and orangeophilic cytoplasm, and sharp cell borders. Three dimensional clusters, papillae and acini containing vacuolated cytoplasm are compatible with adenocarcinoma. Poorly differentiated carcinoma may lack peculiar cytologic features and can be misdiagnosed.8-10 Another study evaluated the cytomorphological features of small cell lung carcinoma and adenocarcinoma of the lung. In this study abundant cytoplasm and absence of nuclear smearing was the most distinct finding in adenocarcinoma compared to small cell carcinoma. Necrotic background, mostly single cells architecture, inconspicuous nucleoli, nuclear molding and hyperchromasia were prominent in small cell carcinoma compared to adenocarcinoma.11 In our case cohesive clusters of tumor cells with moderate amounts of vacuolated eosinophilic cytoplasm and indistinct cell border are noted. Those cells had small nucleoli without nuclear smearing and molding. These findings are consistent with adenocarcinoma.
- Gingival metastases often reveals a benign appearance and a misdiagnosis can be made, interfering with the treatment.12 Therefore any suspicious lesion in the oral cavity in a patient with a history of malignancy should be thoroughly evaluated including histopathological examination.
DISCUSSION
- 1. Meyer I, Shklar G. Malignant tumors metastatic to mouth and jaws. Oral Surg Oral Med Oral Pathol 1965; 20: 350-362. ArticlePubMed
- 2. Hirshberg A, Leibovich P, Buchner A. Metastases to the oral mucosa: analysis of 157 cases. J Oral Pathol Med 1993; 22: 385-390. ArticlePubMed
- 3. Lee KS, Lee YS, Kwon SJ, et al. A case of small cell lung cancer metastasis to the gingiva. Tuberc Respir Dis 2001; 51: 65-69. Article
- 4. Woo JS, Maeng CH, Lee JJ. A case of non-small cell lung cancer that metastasized to the gingiva. Korean J Med 2007; 73: 661-665.
- 5. Hirshberg A, Shnaiderman-Shapiro A, Kaplan I, Berger R. Metastatic tumours to the oral cavity: pathogenesis and analysis of 673 cases. Oral Oncol 2008; 44: 743-752. ArticlePubMed
- 6. Auerbach R, Lu WC, Pardon E, Gumkowski F, Kaminska G, Kaminski M. Specificity of adhesion between murine tumor cells and capillary endothelium: an in vitro correlate of preferential metastasis in vivo. Cancer Res 1987; 47: 1492-1496. PubMed
- 7. Chossegros C, Blanc JL, Cheynet F, Bataille JF, Tessier H. Metastatic localization in the buccal cavity: case report and literature review. Rev Stomatol Chir Maxillofac 1991; 92: 160-164. PubMed
- 8. Vazquez MF, Koizumi JH, Henschke CI, Yankelevitz DF. Reliability of cytologic diagnosis of early lung cancer. Cancer 2007; 111: 252-258. ArticlePubMed
- 9. Kim WS. Cytological diagnosis of lung cancer: the diagnostic accuracy and pitfalls. Korean J Cytopathol 2008; 19: 1-8. Article
- 10. Hong SW, Lee KG. A cytopathologic study of fine needle aspiration biopsy of lung cancer. Korean J Pathol 1990; 24: 465-475.
- 11. Choi YH, Koh JS, Park S, et al. Differential diagnosis between small cell carcinoma and adenocarcinoma of lung in fine needle aspiration cytology. Korean J Cytopathol 2006; 17: 120-125.
- 12. Ellis GL, Jensen JL, Reingold IM, Barr RJ. Malignant neoplasms metastatic to gingivae. Oral Surg Oral Med Oral Pathol 1977; 44: 238-245. ArticlePubMed
REFERENCES
Fig. 4(A) Many cohesive clusters of epithelial tumor cells with moderate to high cellularity on mucous background. (B) Tumor cells reveal cohesive cluster with a moderate amount of eosinophilic cytoplasm, pleomorphic nuclei, and small nucleoli. (C) Tumor cells reveal vacuolated eosinophilic cytoplasm, enlarged nuclei with high nuclear-cytoplasmic ratio, indistinct cell borders, irregular nuclear membranes, nuclear groove, and nuclear inclusion.
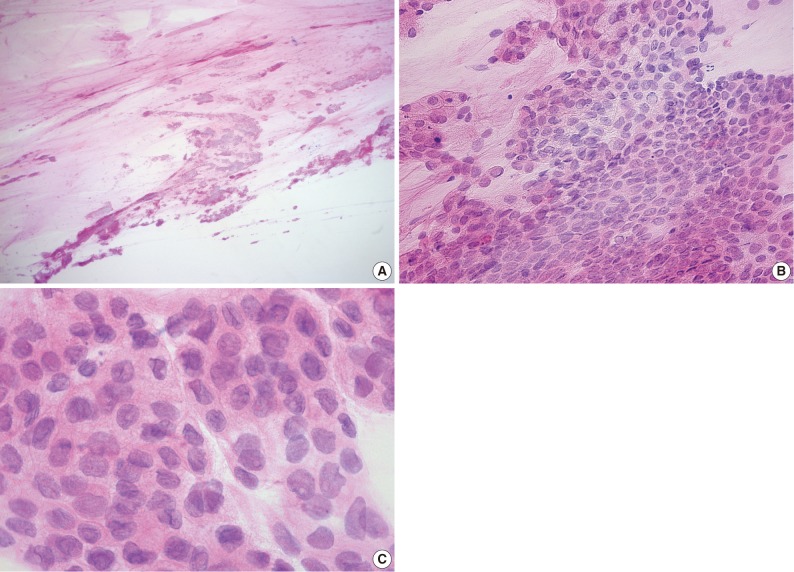

Figure & Data
References
Citations
Citations to this article as recorded by 

Fine Needle Aspiration Cytology of Metastatic Adenocarcinoma of the Gingiva from the Lung: A Case Report




Fig. 1 Computed tomography of the chest shows a mass in the left lower lobe (arrows).
Fig. 2 Alveoli lined by atypical cuboidal to columnar cells with large hyperchromatic nuclei.
Fig. 3 Panoramic radiograph shows diffuse radiolucency at the No. 34, 35, 36 regions (arrow).
Fig. 4 (A) Many cohesive clusters of epithelial tumor cells with moderate to high cellularity on mucous background. (B) Tumor cells reveal cohesive cluster with a moderate amount of eosinophilic cytoplasm, pleomorphic nuclei, and small nucleoli. (C) Tumor cells reveal vacuolated eosinophilic cytoplasm, enlarged nuclei with high nuclear-cytoplasmic ratio, indistinct cell borders, irregular nuclear membranes, nuclear groove, and nuclear inclusion.
Fig. 1
Fig. 2
Fig. 3
Fig. 4
Fine Needle Aspiration Cytology of Metastatic Adenocarcinoma of the Gingiva from the Lung: A Case Report

 E-submission
E-submission

 PubReader
PubReader Cite this Article
Cite this Article