Articles
- Page Path
- HOME > J Pathol Transl Med > Volume 46(6); 2012 > Article
-
Original Article
Expression of c-Met Is Different along the Location and Associated with Lymph Node Metastasis of Head and Neck Carcinoma - Ji-Young Choe, Ji Yun Yun, Soo-Jeong Nam, Ji Eun Kim
-
Korean Journal of Pathology 2012;46(6):515-522.
DOI: https://doi.org/10.4132/KoreanJPathol.2012.46.6.515
Published online: December 26, 2012
Department of Pathology, Seoul National University Boramae Hospital, Seoul National University College of Medicine, Seoul, Korea.
- Corresponding Author: Ji Eun Kim, M.D. Department of Pathology, Seoul National University Boramae Hospital, Seoul National University College of Medicine, 20 Boramae-ro 5-gil, Dongjak-gu, Seoul 156-707, Korea. Tel: +82-2-870-2642, Fax: +82-2-831-0261, npol181@snu.ac.kr
© 2012 The Korean Society of Pathologists/The Korean Society for Cytopathology
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/3.0/) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
Abstract
-
Background
- Activation of the c-Met pathway is involved in cancer progression and the prognosis. We aimed to identify any association of c-Met protein expression with a number of clinicopathologic variables including infection of human papillomavirus and Epstein-Barr virus (EBV) in head and neck carcinomas (HNCa).
-
Methods
- Eighty-two cases were enrolled in this study. Expression of c-Met and p16 was investigated immunohistochemically. EBV was detected by in situ hybridization and amplification of the c-Met gene by fluorescence in situ hybridization.
-
Results
- The c-Met protein was expressed in 41.5% (34/82), and gene amplification was found in 1.4% (1/71). High expression of c-Met was associated with the primary location of the tumor; the hypopharynx showed the highest expression, followed by the oral cavity, larynx, and nasal cavity. Squamous cell carcinoma expressed c-Met more frequently than undifferentiated carcinoma. Also, p16 immunoreactivity or EBV infection was associated with the tumor location and well-differentiated histologic type, but were not linked to c-Met expression. The patients with positive c-Met expression showed frequent lymph node metastasis.
-
Conclusions
- Activation of the c-Met pathway might be involved in a subset of HNCa. Cases showing positive c-Met expression should be carefully monitored because of the high probability of lymph node metastasis.
- Patients
- This study enrolled a total of 82 patients who had been diagnosed with SCC or undifferentiated carcinoma in the head and neck area at the Seoul National University Boramae Hospital (SNUH-B) between 2006 and 2010. The patients were categorized into five groups according to the locations of the tumor: 1) the nasopharynx and nasal cavity, 2) the oropharynx and palatine tonsil, 3) the oral cavity including the tongue, 4) the hypopharynx, and 5) the larynx. The histopathologic features were reviewed by two pathologists, and clinical information such as clinical stage, treatment methods, response after treatment, recurrence, and follow-up were obtained from the electronic medical records. This study was approved by the Institutional Review Board (IRB) of SNUH-B.
- Tissue microarray (TMA) and immunohistochemistry (IHC)
- After reviewing the tumor sections of HNCa, one of the representative areas of each donor block was punched-out with a 2 mm diameter core from the formalin-fixed paraffin-embedded blocks and arranged in the new recipient TMA blocks using a trephine apparatus (Superbiochips Laboratories, Seoul, Korea). IHC was performed using an automated immunostainer (Ventana BenchMark XT, Ventana Medical Systems Inc., Tucson, AZ, USA) according to the manufacturer's protocol. In brief, 5 µm-thick sections from the TMA blocks were deparaffinized and rehydrated. Heat-induced epitope retrieval was performed using the CC1 standard protocol, a combination reagent of disodium ethylenediaminetetraacetic acid and boric acid in a Tris buffer for 60 minutes. The endogenous peroxidase was blocked by incubation with 0.3% hydrogen peroxide. After treatment with 10% normal goat serum to block the nonspecific antibody binding, the slides were incubated with primary antibodies, a monoclonal rabbit antibody against c-Met (Ventana Medical Systems Inc.) and a monoclonal mouse antibody against p16 (CINtec, Heidelberg, Germany), for 30 minutes at room temperature. Detection was performed using the Ventana DAB detection kit (Ventana Medical Systems Inc.).
- Immunoreactivity was evaluated semi-quantitatively by multiplication of the frequency and the intensity of the positive tumor cells to obtain a final semi-quantitative H-score.8 The frequency of positivity was proportionally scored (0 if <1%, 0.1 if 1-9%, 0.5 if 10-49%, and 1 if 50-100%). The staining intensity was graded from 0 to 3.
- EBV in situ hybridization (ISH)
- ISH was performed using a fluorescein-conjugated EBV RNA probe (Y5200, DakoCytomation, Glostrup, Denmark). In brief, 5 µm-thick sections from the TMA blocks were deparaffinized and rehydrated, and then the sections were digested with a proteolytic enzyme (proteinase K at 37℃ for 25 minutes). Thereafter, the slides were incubated with the probe at 55℃ for 1.5 hours and washed with a stringent solution. The slides were labeled with an anti-alkaline phosphatase-conjugated antibody to fluorescein. A chromogen (5-bromo-4-chloro-3-indolylphosphate and nitroblue tetrazolium) was then added and counterstained with Mayer's hematoxylin.
- Fluorescence in situ hybridization (FISH) assay
- The FISH assay was performed on the previously described TMA blocks using a MET/CEP7 dual-color probe (Vysis LSI D7S522 Spectrum Orange/CEP7 Spectrum Green Probes, Abbott Molecular Inc., Chicago, IL, USA). After the pretreatment procedure, the DNA probe kit was applied to the slides, and incubated in humidified air at 73℃ for 5 minutes to denature the target DNA and probes, and subsequently at 37℃ for 19 hours to achieve hybridization. After washing, the slides were counterstained with 4', 6-diamidino-2-phenylindole (DAPI) and p-phenylenediamine.
- Statistical analysis
- Statistical analyses were performed using statistical software SPSS ver. 19.0 (IBM SPSS Inc., Chicago, IL, USA). The correlations between c-Met expression and the clinicopathological variables were evaluated using the Pearson's χ2 test or Fisher's exact test and multiple logistic regression analysis. Overall survival was defined as the interval between the date of diagnosis and the last follow-up date or death. The progression-free survival was calculated from the end of primary treatment to the last follow-up date without relapse or metastasis. The Kaplan-Meier methods were used to compare the survivals. All of the statistical tests were two-sided, and statistical significance was defined as p<0.05.
MATERIALS AND METHODS
- Most of the patients were men (male:female=76:6), aged from 11 to 84 years old (mean, 59 years), including 19 smokers. The patients were grouped as follows: 1) 19 patients with a tumor in the nasopharynx or nasal cavity, 2) 17 with a tumor in the oropharynx or palatine tonsil, 3) 13 with a tumor in the oral cavity including the tongue, 4) 3 with a tumor in the hypopharynx, and 5) 29 with a tumor in the larynx. The histologic types of HNCa cases were 66 SCC including 23 well-differentiated (WD), 32 moderately differentiated (MD), 10 poorly differentiated (PD) type, 1 undetermined and 16 nasopharyngeal undifferentiated carcinomas. According to the clinical tumor-node-metastasis (TNM) stage, 40 patients belonged to the advanced status (stage III-IV) and 30 to the limited stage (stage I-II). Lymph node metastasis was found in 44 (58.7%) cases, and distant metastasis was found in 3 (3.9%) at the time of diagnosis. The treatment modalities were extremely variable including laryngomicrosurgery, laser excision, curative radical resection, primary radiotherapy, systemic chemotherapy, concomitant chemotherapy with radiation, and follow-up without treatment. Expression of p16 protein was present in 34 of the 82 (41.5%) patients, and EBV infection was present in 11 of the 82 (13.4%). Other details of the patient characteristics and marker status are summarized in Table 1.
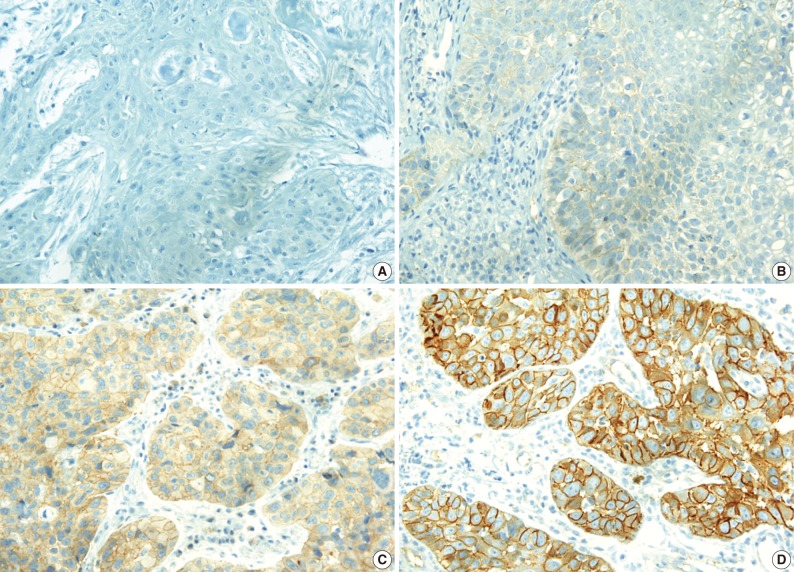
- The c-Met protein was expressed in the cytoplasm or cytoplasmic membrane of the HNCa cells in 34 of the 82 cases (41.5%) in variable degrees. In contrast to the tumor cells, the normal squamous epithelial cells showed a very weak or no expression of c-Met. Expression of c-Met was evaluated as either c-Met negative when the H score was ≤1 or c-Met positive when the H score was >1 (Fig. 1).
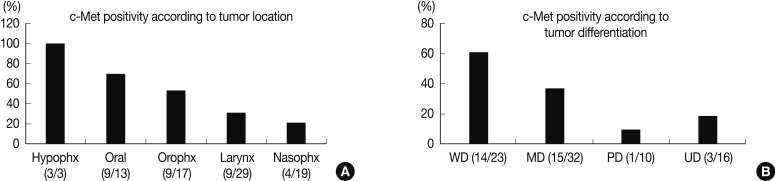
- Expression of c-Met was significantly different between the anatomic tumor groups (p=0.007) in the Pearson's χ2 test. All 3 of the hypopharyngeal SCC were positive for c-Met (100%). SCC of the oral cavity group showed the second highest rate of c-Met positivity (9/13, 69.2%), followed by the oropharynx group (9/17, 52.9%), larynx group (9/29, 41.0%), and the nasal cavity group (4/19, 21.1%). Expression of c-Met was associated with the histologic type of the tumor, and SCC showed a higher positivity than undifferentiated carcinoma (35/66, 53.0% vs. 3/16, 18.8%, respectively). Moreover, in SCC, the WD tumors and MD tumors expressed a stronger c-Met positivity than the PD tumors (14/23, and 15/32 vs. 1/10, respectively) (p=0.023) (Fig. 2). In multiple logistic regression analysis, the degree of squamous differentiation was associated with c-Met expression (p=0.007).
- Expression of p16 was strongly associated with the primary location of the tumor (p=0.001), showing the highest expression in the oropharynx group (10/17, 58.8%) and the lowest expression in the nasopharynx group (1/19, 5.3%). Among the clinical variables, p16 positivity correlated with lymph node metastasis (p=0.034). EBV was detected in 11 cases, most of them being nasopharyngeal undifferentiated carcinoma (10/11, 90.9%); however, one SCC of the nasal cavity was also positive for EBV (1/11, 9.1%). EBV positivity was not associated with any clinical parameters.
- Expression of p16 or presence of EBV was not linked to c-Met expression. The c-Met was more highly expressed in the cases with lymph node metastasis (24/44, 54.5%) than the node negative tumors (9/31, 29%). In the cases with distant metastasis, 1 case out of the 3 was positive for c-Met, and the other two cases were negative for c-Met. Expression of c-Met was not associated with age, sex, clinical TNM stage, tumor recurrence, or history of smoking.
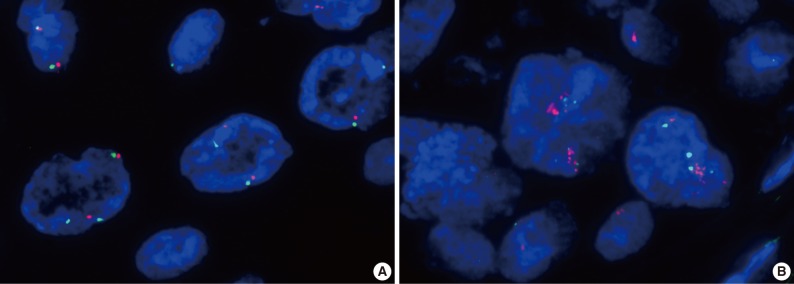
- Amplification of the c-Met gene using FISH analysis was observed in only one of the 71 cases (1.4%), and the other 70 cases showed negative results (Fig. 3). High polysomy was not detected. In the case with c-Met amplification, most of the tumor cells showed clustered red signals, which was interpreted as gene amplification based on the University of Colorado Cancer Center (UCCC) criteria for epidermal growth factor receptor FISH assay (MET to CEP7 ratio≥2 or red signal clusters in ≥10% of the tumor cells) and also the Cappuzzo scoring system for the MET gene (mean≥5 copies/cell in 50 counted cells).9-11 Strong expression of c-Met was also found in this patient showing an H score of 1.5 (a grade 3 of staining intensity in more than 30% of the tumor cells). The patient was a 68-year-old, non-smoking male, who had WD SCC of the larynx with clinical stage I, and no recurrent tumor was found for 33 months after curative resection.
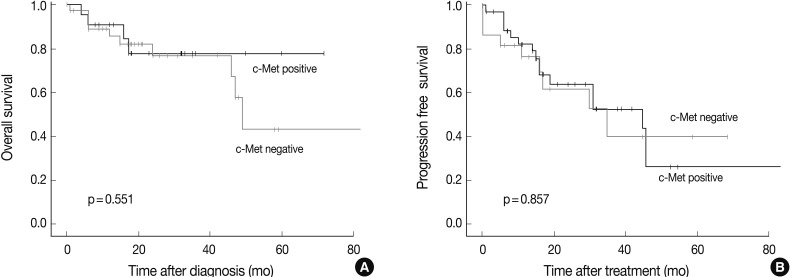
- Survival analysis was performed in the 61 patients who could be reached for follow-up data collection at least 6 months after the surgery or biopsy. The median follow-up period was 47 months (range, 6 to 173 months), and the follow-up showed that 41 patients had died and 12 had recurred tumors. There was no prognostic significance measured by the overall survival or progression free survival between the c-Met positive and the c-Met negative groups (Fig. 4).
RESULTS
- This study aimed to identify the overall expression pattern of c-Met in HNCa according to the anatomic sites and the clinical significance of c-Met expression. Expression of c-Met in HNCa has been mostly investigated in cases limited to oral cavity, tongue, or nasopharynx tumors, which were reported to have a positivity of 42-82%.12-19 We focused on the differences of the c-Met expression pattern along the tumor locations and histologic features of HNCa.
- Regarding the anatomic location of HNCa, c-Met was highly expressed in the oral cavity and oropharynx, but was relatively low in the nasopharynx and nasal cavity. This raises a suspicion that the two important etiologic agents of HNCa, HPV and EBV, can be involved in these differences of aberrant expression of c-Met according to the tumor location. It has been well recognized that HPV is frequently detected in oropharyngeal carcinoma, and that EBV is found in the nasopharynx and nasal cavity. These two viruses are thought to participate in the pathogenesis of HNCa and also to have significant clinical impacts, but the relationship with the c-Met pathway has not been thoroughly elucidated. Walker et al.20 suggested that overexpression of c-Met might be involved in tumor progression in HPV-positive uterine cervical carcinoma. Regarding EBV, one study revealed that latent membrane protein-1 can induce c-Met in nasopharyngeal carcinoma,21 but a contradictory result was also reported by another researcher.22 In this study, we aimed to analyze the effect of HPV or EBV status on c-Met expression. Although there are several methods for detecting an HPV infection, IHC for p16 can be very specific and even more sensitive than HPV polymerase chain reaction assay, especially in high-risk HPV infection.23,24 Therefore, we used p16 positivity as a surrogate marker of HPV infection in HNCa. However, we could not find any significant correlation between c-Met expression and p16 expression or EBV. Further studies should be performed in this regard; however, at least we found regional propensity toward c-Met expression in HNCa.
- In this study, we demonstrated that c-Met expression was associated with histologic differentiation of HNCa. However, this can be partly affected by the prevalence of certain histologic types according to the anatomic sites. Notably, undifferentiated carcinoma arises most frequently in the nasopharynx and nasal cavity. To clarify the interaction of the anatomic location, histologic type, degree of squamous differentiation, and EBV and HPV on c-Met expression, we performed multiple logistic regression analysis, which revealed a significant association between the degree of differentiation and c-Met expression. However, our results are not in agreement with former studies, which showed a lack of correlation between c-Met expression and histologic differentiation in nasopharyngeal carcinoma,15,16 or in SCC in the head and neck.16,17 These findings suggest that differential expression of c-Met along the anatomic sites of HNCa may have other interacting factors that we could not yet find.
- Regarding the positive IHC pattern of c-Met, there have been only a few studies published up to the present. Lo Muzio et al.14 observed a tendency of strong membranous positivity in WD oral SCC instead of a diffuse cytoplasmic reaction in less differentiated tumors. We also observed two distinct patterns of c-Met positivity in HNCa, but no significant difference was found between the staining pattern of c-Met and the clinicopathologic parameters.
- A strong correlation of c-Met expression with lymph node metastasis was noted in our study. However, other variables indicative of invasiveness such as distant metastasis, tumor recurrence, advanced stages, overall survival, and progression free survival did not show a significant association with c-Met expression. This could be partly explained by several factors: 1) our cases contained relatively fewer cases of advanced status and 2) there were many options for the treatment of HNCa. Because of the extreme variety of treatment modalities in HNCa, there have been many contradictory reports on c-Met expression and the prognosis of HNCa.12-14,16,19 However, the only constant result that can be gathered from the previous studies is the correlation of c-Met expression and lymph node metastasis, as shown in this study.
- There are several mechanisms involved in the activation of the c-Met pathway. One of them is gene amplification, which has been reported in non-small cell lung cancer (NSCLC) and other cancers.10 We found c-Met amplification in only one case out of 71, which was a much lower incidence than a study by Seiwert et al.25 who reported a c-Met gene amplification of 13% (3/23) in HNCa. Taken together with the low frequency of c-Met amplification in NSCLC among Koreans,11 this low incidenceof c-Met amplification can reflect a real frequency of c-Met amplification in HNCa in Korea. Although c-Met was highly expressed in HNCa, gene amplification might not contribute highly to the aberrancy of c-Met pathway regulation. Among the other mechanisms of c-Met regulation, ligand-mediated paracrine stimulation has been reported in HNCa by some researchers.4,6
- In conclusion, we found that c-Met was differentially expressed among the various locations of HNCa, and was associated with WD histologic features and frequent lymph node metastases. Based on these findings, positive expression of c-Met can be an indicator of the likelihood of lymph node metastasis; therefore, a more meticulous data gathering and aggressive treatment should be applied in such cases. Although c-Met overexpression was frequently detected in HNCa, gene amplification rarely occurred in our study.
DISCUSSION
Acknowledgments
Acknowledgments
- 1. The Korea Central Cancer Registry, National Cancer Center. Annual report of cancer statistics in Korea in 2009. 2011; Seoul: Ministry of Health and Welfare.
- 2. Siegel R, Ward E, Brawley O, Jemal A. Cancer statistics, 2011: the impact of eliminating socioeconomic and racial disparities on premature cancer deaths. CA Cancer J Clin 2011; 61: 212-236. PubMed
- 3. Szabó B, Nelhubel GA, Kárpáti A, et al. Clinical significance of genetic alterations and expression of epidermal growth factor receptor (EGFR) in head and neck squamous cell carcinomas. Oral Oncol 2011; 47: 487-496. ArticlePubMed
- 4. Lau PC, Chan AT. Novel therapeutic target for head and neck squamous cell carcinoma: HGF-MET signaling pathway. Anticancer Drugs 2011; 22: 665-673. PubMed
- 5. Organ SL, Tsao MS. An overview of the c-MET signaling pathway. Ther Adv Med Oncol 2011; 3(1 Suppl): S7-S19. ArticlePubMedPMCPDF
- 6. Birchmeier C, Birchmeier W, Gherardi E, Vande Woude GF. Met, metastasis, motility and more. Nat Rev Mol Cell Biol 2003; 4: 915-925. ArticlePubMedPDF
- 7. Eder JP, Vande Woude GF, Boerner SA, LoRusso PM. Novel therapeutic inhibitors of the c-Met signaling pathway in cancer. Clin Cancer Res 2009; 15: 2207-2214. ArticlePubMedPDF
- 8. Olaussen KA, Dunant A, Fouret P, et al. DNA repair by ERCC1 in non-small-cell lung cancer and cisplatin-based adjuvant chemotherapy. N Engl J Med 2006; 355: 983-991. ArticlePubMed
- 9. Varella-Garcia M, Diebold J, Eberhard DA, et al. EGFR fluorescence in situ hybridisation assay: guidelines for application to non-small-cell lung cancer. J Clin Pathol 2009; 62: 970-977. ArticlePubMedPMC
- 10. Cappuzzo F, Marchetti A, Skokan M, et al. Increased MET gene copy number negatively affects survival of surgically resected non-small-cell lung cancer patients. J Clin Oncol 2009; 27: 1667-1674. ArticlePubMedPMC
- 11. Go H, Jeon YK, Park HJ, Sung SW, Seo JW, Chung DH. High MET gene copy number leads to shorter survival in patients with non-small cell lung cancer. J Thorac Oncol 2010; 5: 305-313. ArticlePubMed
- 12. Kim CH, Koh YW, Han JH, et al. c-Met expression as an indicator of survival outcome in patients with oral tongue carcinoma. Head Neck 2010; 32: 1655-1664. ArticlePubMed
- 13. Endo K, Shirai A, Furukawa M, Yoshizaki T. Prognostic value of cell motility activation factors in patients with tongue squamous cell carcinoma. Hum Pathol 2006; 37: 1111-1116. ArticlePubMed
- 14. Lo Muzio L, Farina A, Rubini C, et al. Effect of c-Met expression on survival in head and neck squamous cell carcinoma. Tumour Biol 2006; 27: 115-121. ArticlePubMed
- 15. Xie LQ, Bian LJ, Li Z, Li Y, Li ZX, Li B. Altered expression of E-cadherin by hepatocyte growth factor and effect on the prognosis of nasopharyngeal carcinoma. Ann Surg Oncol 2010; 17: 1927-1936. ArticlePubMedPDF
- 16. Qian CN, Guo X, Cao B, et al. Met protein expression level correlates with survival in patients with late-stage nasopharyngeal carcinoma. Cancer Res 2002; 62: 589-596. PubMed
- 17. Sawatsubashi M, Sasatomi E, Mizokami H, Tokunaga O, Shin T. Expression of c-Met in laryngeal carcinoma. Virchows Arch 1998; 432: 331-335. ArticlePubMedPDF
- 18. Kim CH, Moon SK, Bae JH, et al. Expression of hepatocyte growth factor and c-Met in hypopharyngeal squamous cell carcinoma. Acta Otolaryngol 2006; 126: 88-94. ArticlePubMed
- 19. Knowles LM, Stabile LP, Egloff AM, et al. HGF and c-Met participate in paracrine tumorigenic pathways in head and neck squamous cell cancer. Clin Cancer Res 2009; 15: 3740-3750. ArticlePubMedPMCPDF
- 20. Walker F, Kermorgant S, Daraī E, et al. Hepatocyte growth factor and c-Met in cervical intraepithelial neoplasia: overexpression of proteins associated with oncogenic human papillomavirus and human immunodeficiency virus. Clin Cancer Res 2003; 9: 273-284. PubMed
- 21. Horikawa T, Sheen TS, Takeshita H, Sato H, Furukawa M, Yoshizaki T. Induction of c-Met proto-oncogene by Epstein-Barr virus latent membrane protein-1 and the correlation with cervical lymph node metastasis of nasopharyngeal carcinoma. Am J Pathol 2001; 159: 27-33. ArticlePubMedPMC
- 22. Kijima Y, Hokita S, Yoshinaka H, et al. Amplification and overexpression of c-met gene in Epstein-Barr virus-associated gastric carcinomas. Oncology 2002; 62: 60-65. ArticlePubMedPDF
- 23. Schlecht NF, Brandwein-Gensler M, Nuovo GJ, et al. A comparison of clinically utilized human papillomavirus detection methods in head and neck cancer. Mod Pathol 2011; 24: 1295-1305. ArticlePubMedPMCPDF
- 24. Singhi AD, Westra WH. Comparison of human papillomavirus in situ hybridization and p16 immunohistochemistry in the detection of human papillomavirus-associated head and neck cancer based on a prospective clinical experience. Cancer 2010; 116: 2166-2173. ArticlePubMed
- 25. Seiwert TY, Jagadeeswaran R, Faoro L, et al. The MET receptor tyrosine kinase is a potential novel therapeutic target for head and neck squamous cell carcinoma. Cancer Res 2009; 69: 3021-3031. ArticlePubMedPMCPDF
REFERENCES
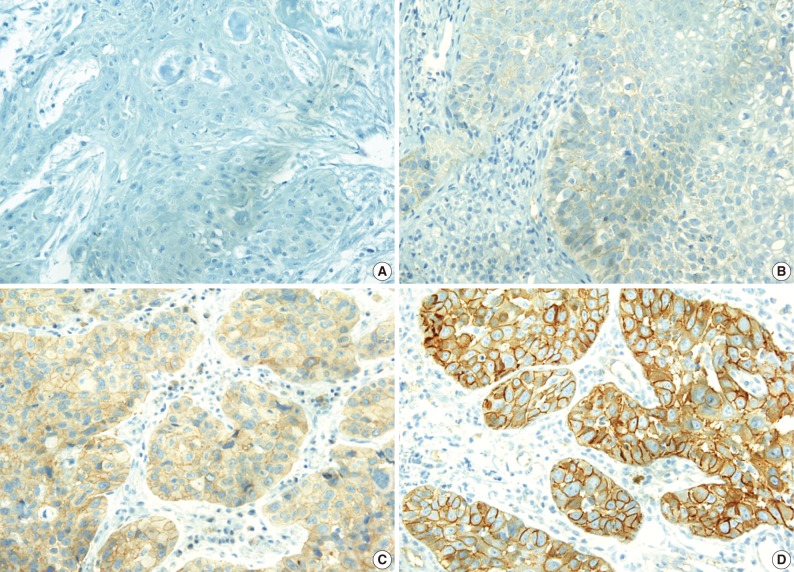
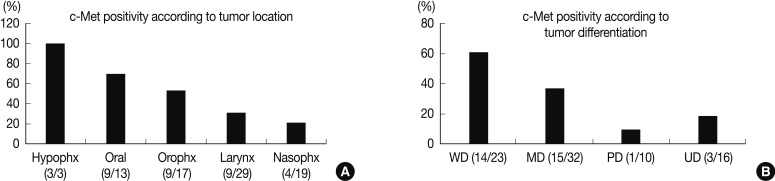
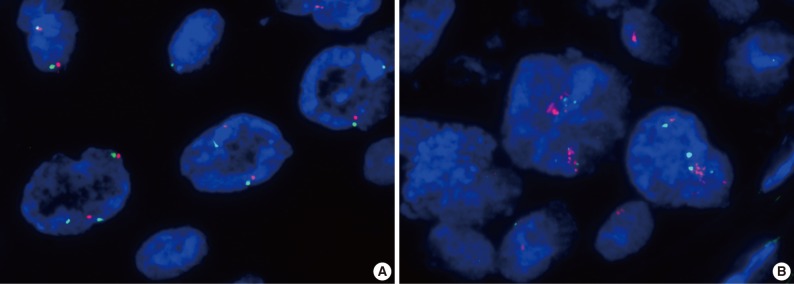
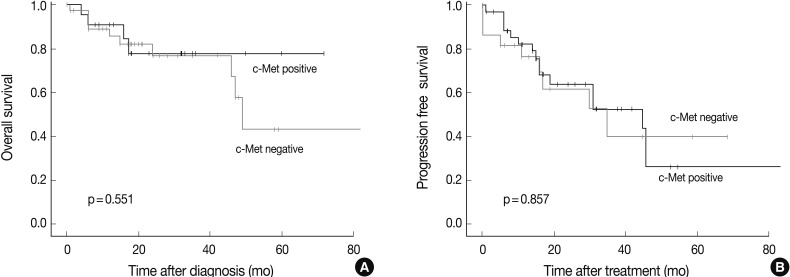
Figure & Data
References
Citations

- Case Report: A rare case of MET-amplified gastric cancer with systemic metastasis: remarkable efficacy of crizotinib and the role of precision medicine
Yan Shen, Yaxin Xu, Jing Sun
Frontiers in Oncology.2025;[Epub] CrossRef - c-MET pathway in human malignancies and its targeting by natural compounds for cancer therapy
Chakrabhavi Dhananjaya Mohan, Muthu K Shanmugam, Siddegowda Gopalapura Shivanne Gowda, Arunachalam Chinnathambi, Kanchugarakoppal S. Rangappa, Gautam Sethi
Phytomedicine.2024; 128: 155379. CrossRef - Nadir paranazal sinüs kanserlerinde yeni tanımlanan reseptör tirozin kinaz mutasyonları ve potansiyel fonksiyonel etkileri
Bakiye GÖKER BAGCA, Sercan GÖDE, Göksel TURHAL, Neslihan Pınar ÖZATEŞ, Ali VERAL, Cumhur GÜNDÜZ, Çığır Biray AVCI
Ege Tıp Dergisi.2023; 62(1): 139. CrossRef - The Emerging Role of c-Met in Carcinogenesis and Clinical Implications as a Possible Therapeutic Target
Antonio Faiella, Ferdinando Riccardi, Giacomo Cartenì, Martina Chiurazzi, Livia Onofrio, Rengyun Liu
Journal of Oncology.2022; 2022: 1. CrossRef - NK4 Regulates Laryngeal Squamous Cell Carcinoma Cell Properties and Inhibits Tumorigenicity by Modulating the DKK1/Wnt/β-Catenin Axis
Shoukai Zhang, Hulai Wei, Xiaoqin Ha, Yueyu Zhang, Yufen Guo
Frontiers in Oncology.2021;[Epub] CrossRef - Epstein-Barr Virus-Associated Carcinoma of the Larynx: A Systematic Review with Meta-Analysis
Marcos Antonio Pereira de Lima, Álife Diêgo Lima Silva, Antônio Carlos Silva do Nascimento Filho, Thiago Lima Cordeiro, João Pedro de Souza Bezerra, Maria Aline Barroso Rocha, Sally de França Lacerda Pinheiro, Roberto Flávio Fontenelle Pinheiro Junior, Ma
Pathogens.2021; 10(11): 1429. CrossRef - The roles of PTEN, cMET, and p16 in resistance to cetuximab in head and neck squamous cell carcinoma
Alexandre A. B. A. da Costa, Felipe D’Almeida Costa, Daniel Vilarim Araújo, Marcos Pedro Guedes Camandaroba, Victor Hugo Fonseca de Jesus, Audrey Oliveira, Ana Caroline Fonseca Alves, Carlos Stecca, Larissa Machado, Andrea Cruz Feraz de Oliveira, Thiago B
Medical Oncology.2019;[Epub] CrossRef - Role of c‐Met expression on prognosis of head and neck cancer: A literature review and meta‐analysis
Lei Li, Zhijun Sun, Xin Huang, Xiao Li, Lihua Sun, Lei Zhang, Xiaodan Zhang, Longwei Ye, Jie Yuan, Limin Mao, Guolin Li
Head & Neck.2019; 41(6): 1999. CrossRef - MET Genomic Alterations in Head and Neck Squamous Cell Carcinoma (HNSCC): Rapid Response to Crizotinib in a Patient with HNSCC with a Novel MET R1004G Mutation
Lisa Pei Chu, Debra Franck, Christine A. Parachoniak, Jeffrey P. Gregg, Michael G. Moore, D. Gregory Farwell, Shyam Rao, Andreas M. Heilmann, Rachel L. Erlich, Jeffrey S. Ross, Vincent A. Miller, Siraj Ali, Jonathan W. Riess
The Oncologist.2019; 24(10): 1305. CrossRef - Understanding c-MET signalling in squamous cell carcinoma of the head & neck
P. Szturz, E. Raymond, C. Abitbol, S. Albert, A. de Gramont, S. Faivre
Critical Reviews in Oncology/Hematology.2017; 111: 39. CrossRef - Prognostic value of c-MET in head and neck cancer: A systematic review and meta-analysis of aggregate data
Petr Szturz, Marie Budíková, Jan B. Vermorken, Ivana Horová, Břetislav Gál, Eric Raymond, Armand de Gramont, Sandrine Faivre
Oral Oncology.2017; 74: 68. CrossRef - Activated HGF-c-Met Axis in Head and Neck Cancer
Levi Arnold, Jonathan Enders, Sufi Thomas
Cancers.2017; 9(12): 169. CrossRef - Clinicopathological impacts of high c-Met expression in head and neck squamous cell carcinoma: a meta-analysis and review
Jung Han Kim, Bum Jun Kim, Hyeong Su Kim
Oncotarget.2017; 8(68): 113120. CrossRef - High expression of c‑Met and EGFR is associated with poor survival of patients with glottic laryngeal squamous cell carcinoma
Mei Jiang, Hui Zhang, He Xiao, Zhimin Zhang, Dan Que, Jia Luo, Jian Li, Bijing Mao, Yuanyuan Chen, Meilin Lan, Ge Wang, Hualiang Xiao
Oncology Letters.2017;[Epub] CrossRef - Profiling of cMET and HER Family Receptor Expression in Pancreatic Ductal Adenocarcinomas and Corresponding Lymph Node Metastasis to Assess Relevant Pathways for Targeted Therapies
Alexander Muckenhuber, Galina Babitzki, Marlene Thomas, Gabriele Hölzlwimmer, Magdalena Zajac, Moritz Jesinghaus, Frank Bergmann, Jens Werner, Albrecht Stenzinger, Wilko Weichert
Pancreas.2016; 45(8): 1167. CrossRef - Absent and abundant MET immunoreactivity is associated with poor prognosis of patients with oral and oropharyngeal squamous cell carcinoma
Maria J. De Herdt, Stefan M. Willems, Berdine van der Steen, Rob Noorlag, Esther I. Verhoef, Geert J.L.H. van Leenders, Robert J.J. van Es, Senada Koljenović, Robert J. Baatenburg de Jong, Leendert H.J. Looijenga
Oncotarget.2016; 7(11): 13167. CrossRef - Biological, diagnostic and therapeutic relevance of the MET receptor signaling in head and neck cancer
Lluís Nisa, Daniel Matthias Aebersold, Roland Giger, Yitzhak Zimmer, Michaela Medová
Pharmacology & Therapeutics.2014; 143(3): 337. CrossRef - Frequent hepatocyte growth factor overexpression and low frequency of c-Met gene amplification in human papillomavirus–negative tonsillar squamous cell carcinoma and their prognostic significances
Mi Jung Kwon, Dong Hoon Kim, Hye-Rim Park, Hyung Sik Shin, Ji Hyun Kwon, Dong Jin Lee, Jin Hwan Kim, Seong Jin Cho, Eun Sook Nam
Human Pathology.2014; 45(7): 1327. CrossRef - Distinct c-Met activation mechanisms induce cell rounding or invasion through pathways involving integrins, RhoA and HIP1
Anja Mai, Ghaffar Muharram, Rachel Barrow-McGee, Habib Baghirov, Juha Rantala, Stéphanie Kermorgant, Johanna Ivaska
Journal of Cell Science.2014; 127(9): 1938. CrossRef




Fig. 1
Fig. 2
Fig. 3
Fig. 4
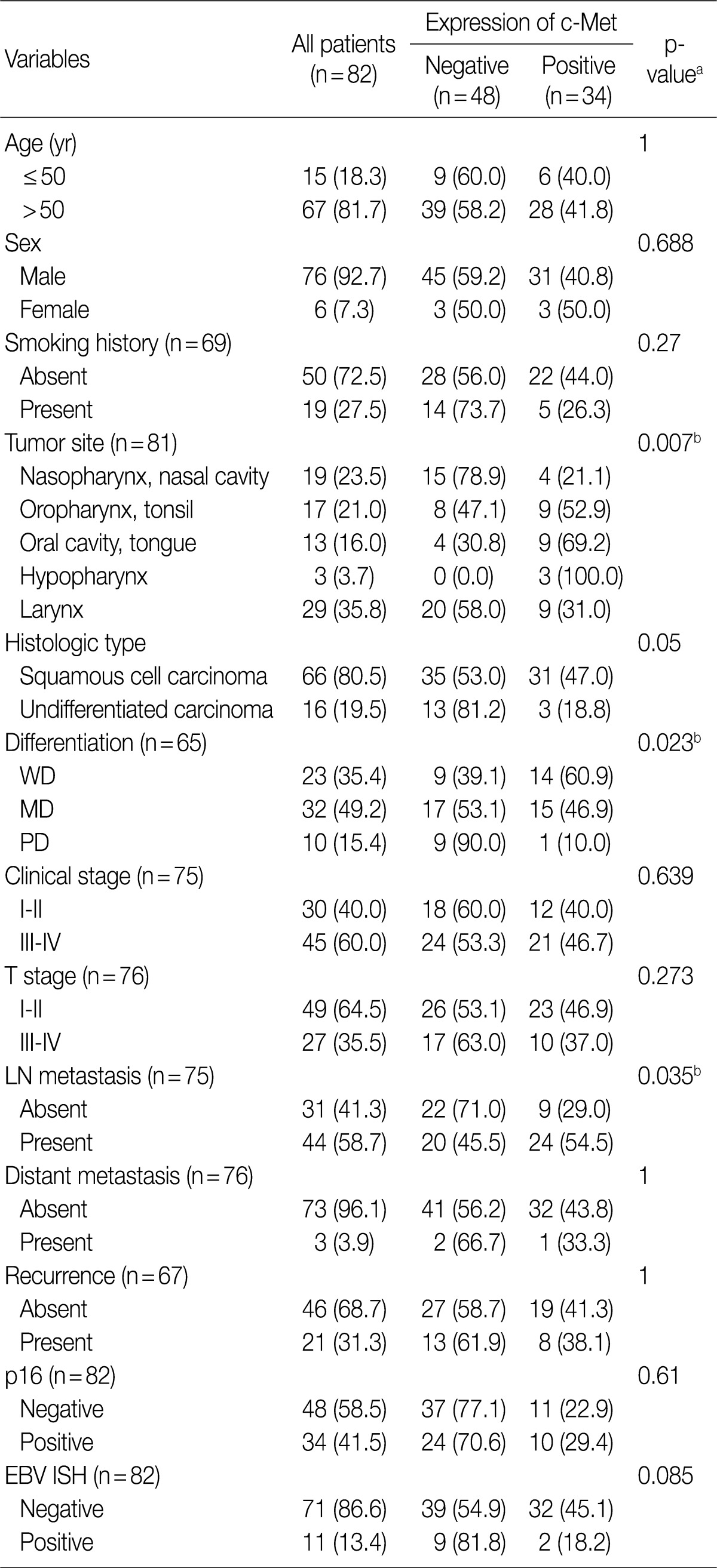
Values are presented as number (%). WD, well-differentiated; MD, moderately differentiated; PD, poorly differentiated; LN, lymph node; EBV, Epstein-Barr virus; ISH, aBy chi-square test; bStatistically significant.

 E-submission
E-submission

 PubReader
PubReader Cite this Article
Cite this Article