Articles
- Page Path
- HOME > J Pathol Transl Med > Volume 47(1); 2013 > Article
-
Original Article
Finding and Characterizing Mammary Analogue Secretory Carcinoma of the Salivary Gland - Min Jung Jung, Joon Seon Song, Sang Yoon Kim1, Soon Yuhl Nam1, Jong-Lyel Roh1, Seung-Ho Choi1, Sung-Bae Kim2, Kyung-Ja Cho
-
Korean Journal of Pathology 2013;47(1):36-43.
DOI: https://doi.org/10.4132/KoreanJPathol.2013.47.1.36
Published online: February 25, 2013
Department of Pathology, Asan Medical Center, University of Ulsan College of Medicine, Seoul, Korea.
1Department of Head and Neck Surgery, Asan Medical Center, University of Ulsan College of Medicine, Seoul, Korea.
2Department of Medical Oncology, Asan Medical Center, University of Ulsan College of Medicine, Seoul, Korea.
- Corresponding Author: Kyung-Ja Cho, M.D. Department of Pathology, Asan Medical Center, University of Ulsan College of Medicine, 88 Olympic-ro 43-gil, Songpa-gu, Seoul 138-736, Korea. Tel: +82-2-3010-4545, Fax: +82-2-472-7898, kjc@amc.seoul.kr
© 2013 The Korean Society of Pathologists/The Korean Society for Cytopathology
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/3.0/) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
Abstract
-
Background
- A new tumor entity of the salivary glands, mammary analogue secretory carcinoma (MASC) with ETV6-NTRK3 translocation, has recently been proposed. MASC was originally diagnosed as adenocarcinoma, not otherwise specified (ANOS), or acinic cell carcinoma (AciCC) by the current World Health Organization classification. We aimed to identify MASC cases by molecular tests, and to characterize their clinical, histological, and immunohistochemical features.
-
Methods
- Thirty cases of MASC candidates were selected after review of 196 salivary gland tumors, and subjected to break-apart ETV6 fluorescence in situ hybridization (FISH), and immunohistochemical study for S100 protein, gross cystic disease fluid protein 15, DOG1, estrogen receptor, and progesterone receptor.
-
Results
- Valid FISH results were obtained in 23 cases, and 13 positive cases were retrieved. MASCs were histologically varied, and the most frequent features observed in 10 cases were low-grade papillary/cystic/glandular patterns, intraluminal secretory materials, ovoid/wrinkled nuclei, and relatively abundant granular eosinophilic cytoplasms, corresponding to papillary-cystic or follicular types of AciCC. All cases showed diffuse immunopositivity for S100 protein. Three cases developed recurrences, but all patients remained alive.
-
Conclusions
- MASC could be a molecularly well-defined salivary gland neoplasm, encompassing some portions of AciCC and ANOS, but its histological spectrum and clinical implication require further investigation.
- We selected excised salivary gland tumors, excluding biopsy or consultation cases, from the surgical pathology files of the Asan Medical Center from 1990 to 2012. For retrieval of MASC candidates, 196 salivary gland tumors (MEC, n=137; AciCC, n=43; ANOS, n=11; CAC, n=2; other unusual salivary gland neoplasm, n=3) were retrospectively reviewed by 2 pathologists. The selection of study candidates was built on the histological features described by recent literature1,3-5 and sufficient amount of tissue available to construct a tissue microarray (TMA). The criteria for selection among the above tumors included the presence of glandular formation or secretory activity. The final specimens comprised 30 cases initially diagnosed as AciCC (n=16), ANOS (n=6), MEC (n=3), CAC (n=2), salivary duct carcinoma (SDC; n=1), squamous cell carcinoma (n=1), and oncocytic carcinoma (n=1). Additionally, 6 conventional AciCC cases, morphologically showing unequivocal serous acinar differentiation, and 2 SDC cases were selected for reference. For all 38 cases, TMAs were generated using a manual tissue arrayer (Pathology Devices, Westminster, MD, USA). Two 1.0- or 1.5-mm cores were taken from donor blocks and arrayed into recipient blocks. Clinical data were obtained through review of medical records and pathologic staging was based on the Cancer Staging Manual of American Joint Committee.8 Disease-free survival was assessed from the time of histological diagnosis to recurrence or death, based on review of the patients' medical records and information from the National Health Insurance. A t-test was employed to characterize the relationships between quantitative variables and the Fisher's exact test was used to characterize the relationship between categorical variables. Disease-free survival periods with 95% confidence intervals (CI) were estimated using the Kaplan-Meier method, with statistical significance of differences between groups estimated by log-rank test. A p-value of <0.05 was defined as statistically significant. Statistical analysis was performed using SPSS ver. 18 (SPSS Inc., Chicago, IL, USA).
- Immunohistochemistry and ETV6 FISH
- From TMA blocks, 4-µm sectioned slides were obtained and prepared for immunohistochemistry and FISH. Immunohistochemical stainings were performed using the Ventana autostainer and ultra view DAB detection kit (Ventana, Tucson, AZ, USA) according to manufacturer's instructions. The antibody sources and dilutions are listed in Table 1. FISH was performed using a break-apart probe for the ETV6 gene (Abbott Molecular, Des Plaines, IL, USA) according to the manufacturer's recommendations. FISH slides were examined with an Olympus BX51 fluorescence microscope (Olympus, Tokyo, Japan) using a 100× objective. Through FITC and Texas Red Band pass filters, each image was obtained with an Olympus DP70 camera using DP Controller (ver. 3.3.1.292) acquisition software. Fifty to 100 cells were examined in each case. A single green (or red) signal without a corresponding red (or green) signal in addition to a fused signal was considered negative (non-rearranged) in the present study. Red and green signals that were less than 2 signal diameters apart were considered as a single fused signal. The average percentage of split signal in 6 referential AciCCs, showing unequivocal serous acinar differentiation (conventional AciCC) was 4.565 and standard deviation (SD) 3.042. Thus, we considered cases, if more than 15% (mean+3 SD, rounded up) of examined nuclei showed a split signal, as positive for translocation which is the similar cut-off value used in previous literature.1 The slides were independently interpreted by 2 observers. If 10-20% of the analyzed cells showed a split signal, more cells were enumerated and the first and second cell count readings by the 2 observers were added together and a percentage was calculated. Valid FISH results were obtained in 31 out of 38 cases. The laboratory used breast secretory carcinoma as a positive control.
MATERIALS AND METHODS
- Verifying MASC with ETV6 translocation and its clinicopathologic characteristics
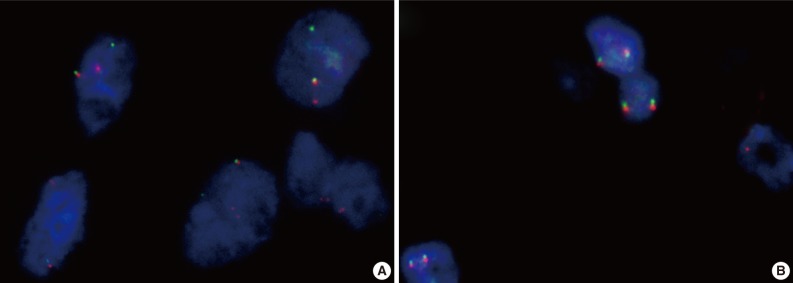
- Among the 31 technically successful cases, 13 cases showed ETV6 translocation, reclassified as MASC, and 10 cases showed intact ETV6, categorized as mimic MASC (Fig. 1). The other 8 cases served as a reference, including 6 conventional AciCCs and 2 SDCs, and showed intact ETV6. On average, 57.65% of examined cells showed a split signal with ETV6 translocation (MASC) in 13 cases, while 9.55% of examined cells showed a split signal in 10 cases with intact ETV6 (mimic MASC). The percentage of cells with a split signal ranged between 20.75% and 78.57% in cases with ETV6 translocation, and between 3.10% and 12.96% in cases with intact ETV6.
- The clinicopathologic features of 23 MASC candidates with ETV6 translocation (MASC, n=13) or with intact ETV6 (mimic MASC, n=10), are summarized in Table 2. For referential comparison, conventional AciCCs (n=15) surgically treated in our institution are also summarized in Table 2.
- The 13 MASC cases consisted of 8 males and 5 females. Although no differences in gender distribution were found between MASC and mimic MASC cases, a noticeable male predilection was observed compared with conventional AciCCs, which affected females more frequently than males (F:M=12:3). The average age in the MASCs, mimic MASCs, and conventional AciCCs was in the fifth decade. Both MASCs and mimic MASCs showed relatively even age distribution from the second to the eighth decade. The parotid gland was the most frequently involved site in MASCs, mimic MASCs, and conventional AciCCs and the tumor size in MASCs ranged from 0.7 cm to 2.5 cm (mean, 1.77 cm). The average tumor size was similar to mimic MASC and smaller than conventional AciCC. Only 4 cases of MASC showed extraparenchymal extension, and any lymph node involvement at the time of surgery was not observed. All patients were initially treated by surgery, and 3 MASC, 3 mimic MASC, and 2 conventional AciCC patients received postoperative radiotherapy and all are currently alive. Three MASC patients developed local recurrence (3/13, 23.1%) with median recurrence time of 44 months after diagnosis (range, 10 to 101 months) and 2 mimic MASC patients developed metastases to neck lymph nodes (2/10, 20.0%) at 62 months and 70 months after diagnosis, while an AciCC patient developed local recurrence (1/15, 6.7%) at 39 months after diagnosis. The mean follow-up period was 50 months. In summary, MASC, mimic MASC, and conventional AciCC showed no statistically significant differences in gender, age, site, pathologic T stage, and disease-free survival.
- Defining the histological MASC properties and comparison with non-MASC
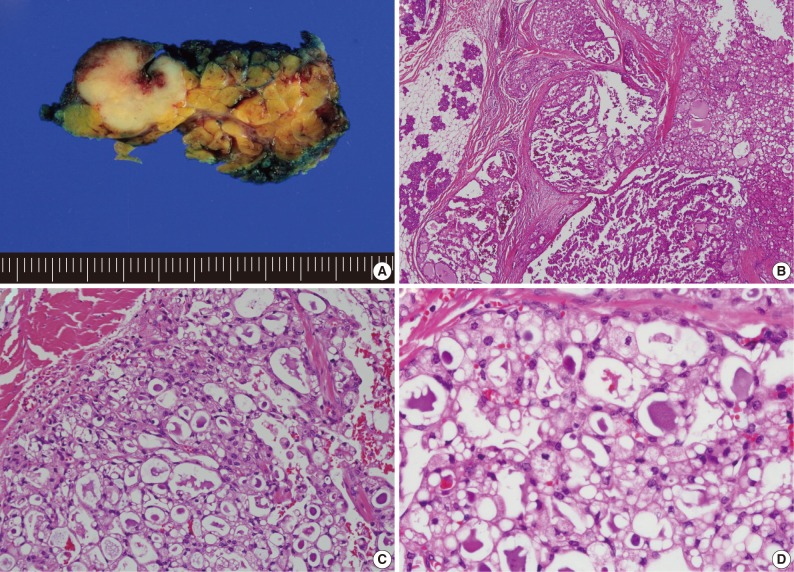
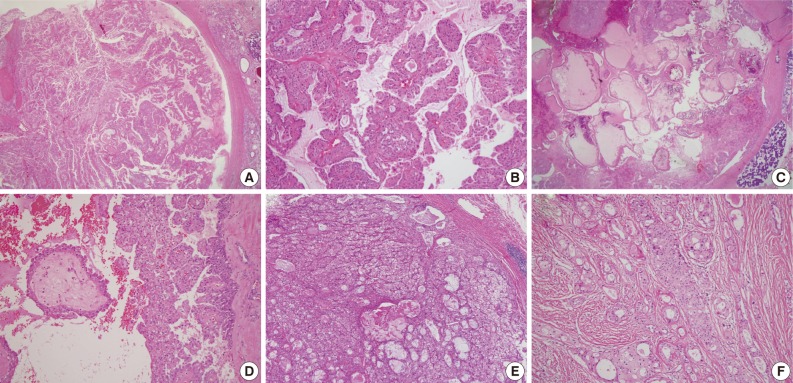
- Histologically, the most frequent features in the 13 MASC cases were well circumscribed, encapsulated, or multinodular masses, showing papillary-cystic, microcystic, solid, follicular or their combination growth pattern. Cytomorphologically, most tumors showed an admixture of variable cell types. Some cells were smaller than typical acinar cells appearing to have an increased nuclear to cytoplasmic ratio, and showed eosinophilic or finely granular cytoplasm and ovoid basophilic nuclei, known as intercalated duct-like cells. Other cells showed a more abundant or vacuolated cytoplasm and low-grade wrinkled nuclei. A small number of cells appeared clear or mucin-containing. Numerous variable-sized spaces were frequently observed and the spaces were usually clear but sometimes contained pinkish- to grayish blue-colored amorphous materials, which were positive for periodic acid-Schiff. As stated in previous literature, the above-mentioned characteristics were predominant features of MASC and corresponded to microcystic, papillary-cystic, or follicular type of AciCC.1-5 These usual morphological MASC features are illustrated in Fig. 2.
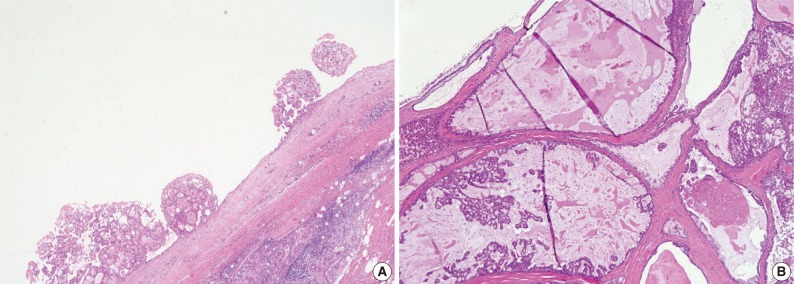
- However, 3 MASC cases showed unusual histological features. One case was macrocystic, measuring up to 2.5 cm, and partially filled with intraluminal papillary proliferation. A macrocystic tumor with sparse projection could be misidentified as CAC. Nevertheless, epithelial configuration of the cyst lining and papillary projection was similar to the mentioned MASC. Another case showed large but variable-sized cysts interspersed among the fibrous stroma. The tumor showed focal proliferation of the epithelial lining and even cribriform appearance in some relatively small islands. These features were also reminiscent of CAC, however, epithelial lining cells were similar to MASC rather than the cuboidal, clear, tall columnar, or oncocytic features of CAC. Notably, the thin epithelial lining cells often exhibited mucinous cytoplasm (Fig. 3).
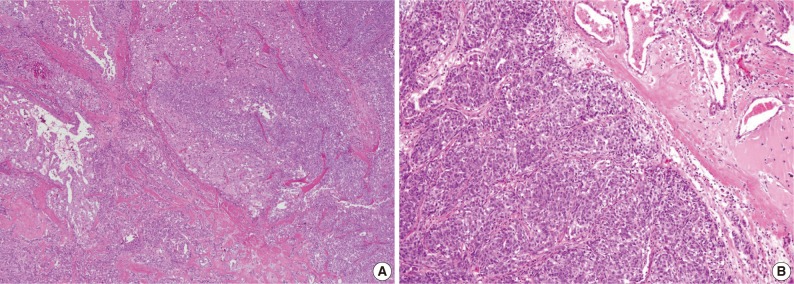
- The third case showed a distinct morphologic change previously referred to as dedifferentiation or high-grade transformation of AciCC.9 The tumor was composed of 2 distinct areas with an obvious transformation zone. Histological features of one area corresponded to microcystic and papillary-cystic MASC and the other area showed poor zymogen granule containing-basophilic polymorphous cells, forming solid nests with comedonecrosis (Fig. 4).
- A diversity of morphological features with intact ETV6 was also obtained. Among 10 mimic MASCs, 3 cases could be interpreted as CAC. Though the cytomorphologic features were not uniform, the tumor cells were slightly different from MASC, showing cuboidal, tall columnar, clear to oncocytic features. Additionally, the epithelial lining of cysts was more evenly thin in CAC than MASC. Additionally, when papillary projections were present, they were mostly large papillae covered with columnar or tall cuboidal cells. Others showed nonspecific morphologies, corresponding to ANOS. Infiltrating nests of clear cells, glandular growth in the prominent sclerosing background, or solid nests surrounded by very thin fibrovascular septa with reticular and abundant cytoplasm were other examples of mimic MASC. These architectural patterns were obviously different from MASC, but cytomorphological features were similar. Two cases of MECs lacking mucous and epidermoid cells were initially similar to MASC, but careful examination can help in distinguishing MEC from MASC. Simultaneously, oncocytoma lacking characteristic cytoplasm features could also mimic MASC. The above morphological features mimicking MASC are illustrated in Fig. 5.
- Immunohistochemical findings
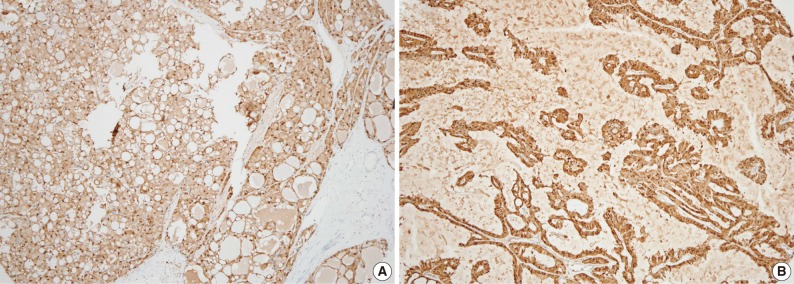
- Immunohistochemically, all 13 MASC cases were positive for S100 protein. Most staining patterns were strong and diffuse, except for a case showing dedifferentiation (Fig. 6). Three out of 10 mimic MASCs expressed S100 protein and no expression was observed in 6 conventional AciCCs. Gross cystic disease fluid protein 15 (GCDFP-15) immunostains were focally positive for 2 out of 13 MASCs, 3 out of 10 mimic MASCs, and none of the conventional AciCCs. DOG1 was expressed in 3 conventional AciCCs and 1 MASC, with weak apical or apicoluminal membranous staining. Estrogen receptor (ER) and progesterone receptor (PR) were all negative for MASC, mimic MASC and conventional AciCCs (Table 3).
RESULTS
- In this study we investigated the new salivary gland tumor, known as MASC, to clarify the clinical, histological, and immunohistochemical characteristics, and to determine any pathognomonic features helpful in diagnosing MASCs.
- The first MASC report focused on features reminiscent of breast secretory carcinoma.1 In recent years, there have been several studies extending the clinical, histological, and immunohistochemical properties of MASCs.3,5 However, MASC clinicopathologic features appear to be indistinctive and the molecular demonstration of ETV6 rearrangement is the only diagnostic method. Regardless of the originality of this entity, a salivary gland tumor with ETV6 translocation has yet to be documented in Korea.
- The present study compared MASC candidate groups with an intact ETV6 and ETV6 translocation. To evaluate MASCs with an emphasis on easy identification of specific characteristics, consideration of the ideal cut-off value to assess the ETV6 FISH results was necessary. In this study, a 15% cut-off value was determined as acceptable. The negative control cut-off value (mean+3 SD, 13.688) showed a 99.7% CI for intact ETV6. The cut-off value used (15%) was slightly higher than the negative control cut-off value. Moreover, the interpretation of a single colored signal without a corresponding colored signal in addition to a fused signal as negative could decrease the percentage of positive cells for ETV6 translocation. The highest negative value for ETV6 translocation was 12.96% and the lowest positive value was 20.75%. This result shows the importance of determining an accurate cut-off value in a study.
- Most MASC features overlapped with subtypes of AciCC. Overall, papillary-cystic, microcystic, or their mixture types were predominantly observed. AciCCs, according to the current World Health Organization classification, are comprised of heterogeneous tumors. Many cell types, including acinar, intercalated duct type, vacuolated, clear, and nonspecific glandular cells, and variable growth patterns such as solid, microcystic, papillary-cystic, and follicular, are described as constituents of AciCC. ANOS is also a heterogeneous tumor group, and their diagnosis is based on the lack of features that characterize other salivary gland adenocarcinomas. According to prior studies and our results, a considerable number of ANOS and zymogen granule-poor AciCCs belong to the MASC group. The concept that MASC is a distinctive entity of salivary gland tumors requires more consideration, however, no zymogen granule-rich AciCC or SDC has shown an ETV6 rearrangement. Our study revealed previously undescribed histology in ETV6-rearranged tumors, such as a solid pattern resembling dedifferentiated AciCC and a large cyst with sparse papillary proliferation. Chiosea et al.4 reported that the results of ETV6 FISH in 4 cases of AciCC with high-grade malformation were all negative for ETV6 translocation. Our results were contrary to the previous result, even in one case.
- Apart for a few exceptions, the histological outline of MASC appears evident, and mimic MASC cases without an ETV6 translocation could be differentiated from MASC by careful histological examination. However, MASC diagnosis in routine practice remains difficult by histological examination only. We attempted to identify an immunohistochemical marker for MASC. DOG1, known as a novel marker of salivary acinar and intercalated duct differentiation, was not helpful.10 GCDFP-15, ER, and PR were considered a possibility due to their mammary-like character, but were not expressed in MASC. The S100 protein was highly sensitive, but showed low specificity to differentiate MASC from mimic MASC. Thus, new immunohistochemical markers that could substitute ETV6 FISH are required.
- In conclusion, MASC could be a molecularly well-defined salivary gland neoplasm, encompassing some portions of AciCC and ANOS, but its histological spectrum and clinical implication need further investigation.
DISCUSSION
- 1. Skálová A, Vanecek T, Sima R, et al. Mammary analogue secretory carcinoma of salivary glands, containing the ETV6-NTRK3 fusion gene: a hitherto undescribed salivary gland tumor entity. Am J Surg Pathol 2010; 34: 599-608. ArticlePubMed
- 2. Griffith C, Seethala R, Chiosea SI. Mammary analogue secretory carcinoma: a new twist to the diagnostic dilemma of zymogen granule poor acinic cell carcinoma. Virchows Arch 2011; 459: 117-118. ArticlePubMedPDF
- 3. Connor A, Perez-Ordoñez B, Shago M, Skálová A, Weinreb I. Mammary analog secretory carcinoma of salivary gland origin with the ETV6 gene rearrangement by FISH: expanded morphologic and immunohistochemical spectrum of a recently described entity. Am J Surg Pathol 2012; 36: 27-34. PubMed
- 4. Chiosea SI, Griffith C, Assaad A, Seethala RR. The profile of acinic cell carcinoma after recognition of mammary analog secretory carcinoma. Am J Surg Pathol 2012; 36: 343-350. ArticlePubMed
- 5. Chiosea SI, Griffith C, Assaad A, Seethala RR. Clinicopathological characterization of mammary analogue secretory carcinoma of salivary glands. Histopathology 2012; 61: 387-394. ArticlePubMed
- 6. Chi HT, Ly BT, Kano Y, Tojo A, Watanabe T, Sato Y. ETV6-NTRK3 as a therapeutic target of small molecule inhibitor PKC412. Biochem Biophys Res Commun 2012; 429: 87-92. ArticlePubMed
- 7. Tognon CE, Somasiri AM, Evdokimova VE, et al. ETV6-NTRK3-mediated breast epithelial cell transformation is blocked by targeting the IGF1R signaling pathway. Cancer Res 2011; 71: 1060-1070. ArticlePubMedPDF
- 8. Edge SB, Byrd DR, Compton CC, Fritz AG, Greene FL, Trotti A. AJCC cancer staging manual. 2010; 7th ed. New York: Springer.
- 9. Skálová A, Sima R, Vanecek T, et al. Acinic cell carcinoma with high-grade transformation: a report of 9 cases with immunohistochemical study and analysis of TP53 and HER-2/neu genes. Am J Surg Pathol 2009; 33: 1137-1145. PubMed
- 10. Chenevert J, Duvvuri U, Chiosea S, et al. DOG1: a novel marker of salivary acinar and intercalated duct differentiation. Mod Pathol 2012; 25: 919-929. ArticlePubMedPDF
REFERENCES
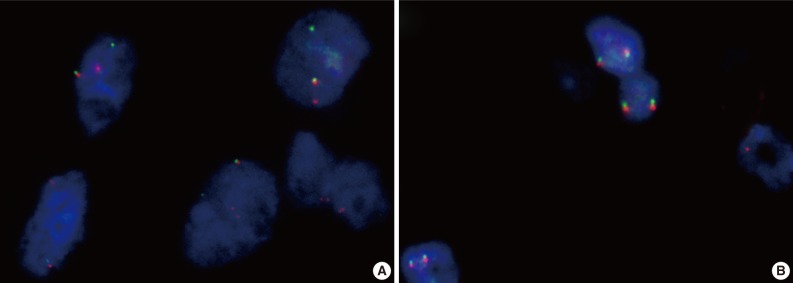
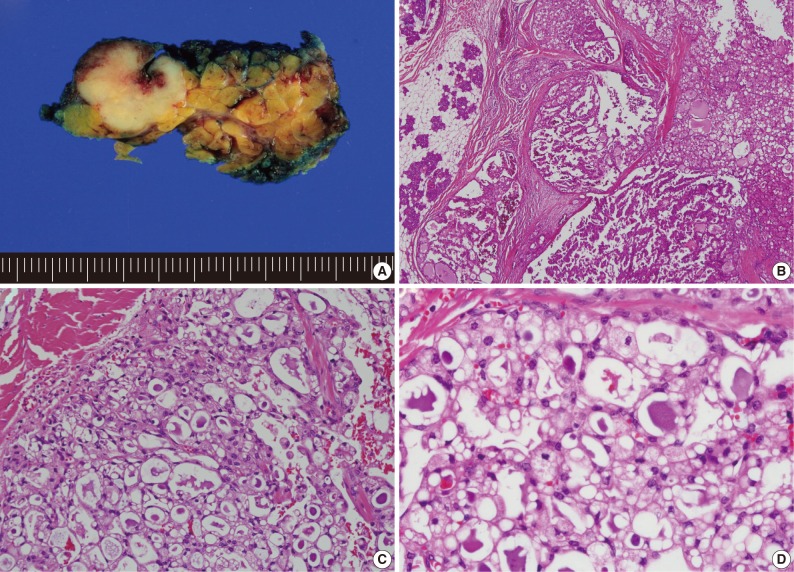
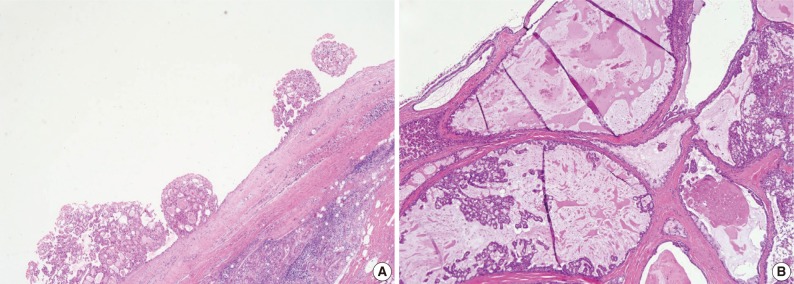
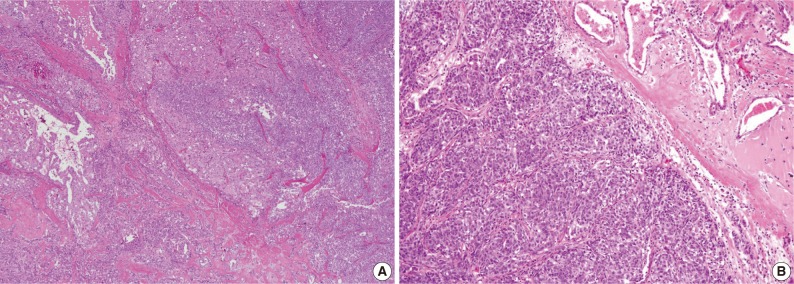
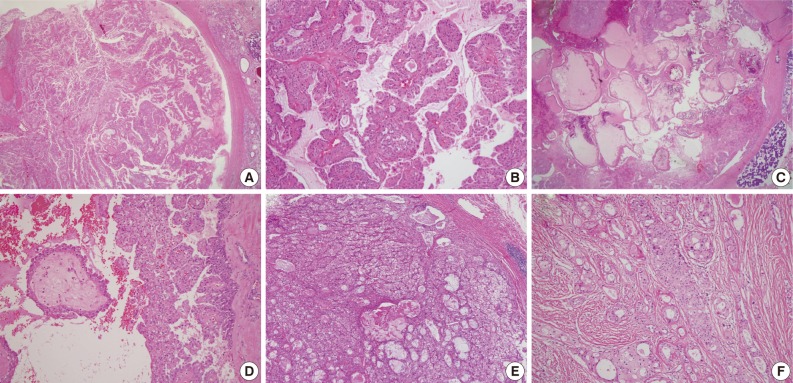
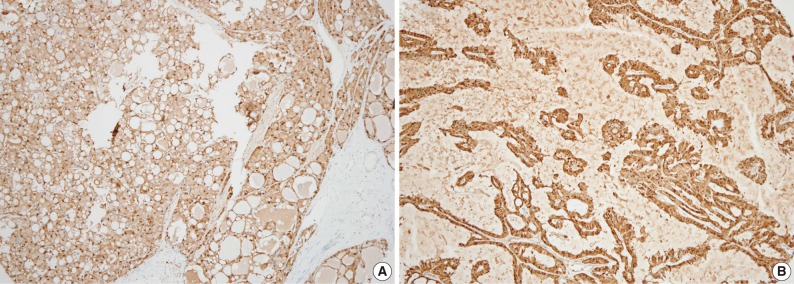
Figure & Data
References
Citations

- Rare Tumors of Oral Cavity: A Case Report and Literature Review on Secretory Carcinoma of Minor Salivary Glands
Emilio Salerno, Silvio Abati, Gianluigi Arrigoni, Daniela Finocchiaro, Giorgio Gastaldi, Alessandra Lissoni, Andrea Galli, Matteo Trimarchi
Clinical and Experimental Dental Research.2025;[Epub] CrossRef - Salivary Gland Secretory Carcinoma; Review of 13 Years World‐Wide Experience and Meta‐Analysis
Eyal Yosefof, Tomer Boldes, Daniel Dan, Eyal Robenshtok, Yulia Strenov, Gideon Bachar, Thomas Shpitzer, Aviram Mizrachi
The Laryngoscope.2024; 134(4): 1716. CrossRef - Salivary gland secretory carcinoma presenting as a cervical soft tissue mass: a case report
Parisa Mokhles, Alireza Sadeghipour, Pegah Babaheidarian, Saleh Mohebbi, Zahra Keshtpour Amlashi, Mohammad Hadi Gharib, Mohammad Saeid Ahmadi, Zeinab Khastkhodaei
Journal of Medical Case Reports.2024;[Epub] CrossRef - Mammary analogue secretory carcinoma of the head and neck — Clinicopathological, imaging features and prognosis analysis
Runjia Liu, Chuanzheng Sun, Likang Zhao, Shiyu Zhou, Tao Xie, Ji Zhang, Dengpeng Tang, Lei Li, Yan Xi
Journal of Radiation Research and Applied Sciences.2024; 17(2): 100914. CrossRef - Mammary analogue secretory carcinoma involving submandibular gland: Diagnostic pitfall with review of literature
Nimisha Dhankar, Nidhi Verma, Abhinav Agarwal, Ravi Mehar, Sunil Pasricha
Journal of Cancer Research and Therapeutics.2024; 20(5): 1658. CrossRef - Metastatic salivary gland mammary analogue secretory carcinoma (MASC) of parotid gland – A rare case report in the literature review
Aynur Aliyeva, Ziya Karimov, Togay Muderris
Acta Oto-Laryngologica Case Reports.2023; 8(1): 38. CrossRef - Secretory carcinoma of minor salivary glands of buccal mucosa: A case report and review of the literature
Noshad Ali Langah, Abdul Ahad, Shayan Khalid Ghaloo, Muhammad Faisal, Raza Tasawar Hussain, Fareed Akbar Shah
International Journal of Surgery Case Reports.2023; 107: 108357. CrossRef - An Underappreciated Cytomorphological Feature of Secretory Carcinoma of Salivary Gland on Fine Needle Aspiration Biopsy: Case Report with Literature Review
Yinan Hua, Bing Leng, Kenneth E. Youens, Lina Liu
Head and Neck Pathology.2022; 16(2): 567. CrossRef - Clinicopathological investigation of secretory carcinoma cases including a successful treatment outcome using entrectinib for high-grade transformation: a case report
Kensuke Suzuki, Hiroshi Harada, Masayuki Takeda, Chisato Ohe, Yoshiko Uemura, Akihiko Kawahara, Shunsuke Sawada, Akira Kanda, Bhaswati Sengupta, Hiroshi Iwai
BMC Medical Genomics.2022;[Epub] CrossRef - DOG1 as an Immunohistochemical Marker of Acinic Cell Carcinoma: A Systematic Review and Meta-Analysis
Vincenzo Fiorentino, Patrizia Straccia, Pietro Tralongo, Teresa Musarra, Francesco Pierconti, Maurizio Martini, Guido Fadda, Esther Diana Rossi, Luigi Maria Larocca
International Journal of Molecular Sciences.2022; 23(17): 9711. CrossRef - Secretory carcinoma of the sinonasal cavity and pharynx: A retrospective analysis of four cases and literature review
Changli Yue, Xiaoli Zhao, Donglin Ma, Yingshi Piao
Annals of Diagnostic Pathology.2022; 61: 152052. CrossRef - Secretory carcinoma of the salivary gland: a multi‐institutional clinicopathologic study of 90 cases with emphasis on grading and prognostic factors
Bin Xu, Kartik Viswanathan, Kavita Umrau, Thair A. D. Al‐Ameri, Snjezana Dogan, Kelly Magliocca, Ronald A. Ghossein, Nicole A. Cipriani, Nora Katabi
Histopathology.2022; 81(5): 670. CrossRef - A systematic review of secretory carcinoma of the salivary gland: where are we?
Lísia Daltro Borges Alves, Andreia Cristina de Melo, Thayana Alves Farinha, Luiz Henrique de Lima Araujo, Leandro de Souza Thiago, Fernando Luiz Dias, Héliton Spíndola Antunes, Ana Lucia Amaral Eisenberg, Luiz Claudio Santos Thuler, Daniel Cohen Goldember
Oral Surgery, Oral Medicine, Oral Pathology and Oral Radiology.2021; 132(4): e143. CrossRef - A case report of surgical resection of secretory carcinoma in the maxillary and ethmoid sinus
Kurt Willis, Martin Bullock, Matthew H. Rigby
International Journal of Surgery Case Reports.2021; 81: 105750. CrossRef - High‐grade salivary gland carcinoma with the ETV6‐NTRK3 gene fusion: A case report and literature review of secretory carcinoma with high‐grade transformation
Satsuki Asai, Shinji Sumiyoshi, Yosuke Yamada, Ichiro Tateya, Toshitaka Nagao, Sachiko Minamiguchi, Hironori Haga
Pathology International.2021; 71(6): 427. CrossRef - High-grade Transformation/Dedifferentiation in Salivary Gland Carcinomas: Occurrence Across Subtypes and Clinical Significance
Alena Skalova, Ilmo Leivo, Henrik Hellquist, Abbas Agaimy, Roderick H.W. Simpson, Göran Stenman, Vincent Vander Poorten, Justin A. Bishop, Alessandro Franchi, Juan C. Hernandez-Prera, David Slouka, Stefan M. Willems, Kerry D. Olsen, Alfio Ferlito
Advances in Anatomic Pathology.2021; 28(3): 107. CrossRef - Radiological features of head and neck mammary analogue secretory carcinoma: 11 new cases with a systematic review of 29 cases reported in 28 publications
Ryo Kurokawa, Mariko Kurokawa, Akira Baba, Yoshiaki Ota, Toshio Moritani, Ashok Srinivasan
Neuroradiology.2021; 63(11): 1901. CrossRef - A case of secretory carcinoma of the salivary glands in the lower lip
Reiko OHARA, Haruki SATO, Kensuke NAGANAWA, Taihei HAYAKAWA, Tatsuya KATAOKA, Ichiro OH-IWA
Japanese Journal of Oral and Maxillofacial Surgery.2021; 67(2): 83. CrossRef - Undifferentiated and dedifferentiated head and neck carcinomas
Alessandro Franchi, Alena Skalova
Seminars in Diagnostic Pathology.2021; 38(6): 127. CrossRef - Cytopathological Findings of Secretory Carcinoma of the Salivary Gland and the Diagnostic Utility of Giemsa Staining
Yuria Egusa, Midori Filiz Nishimura, Satoko Baba, Kengo Takeuchi, Takuma Makino, Tomoyasu Tachibana, Asami Nishikori, Azusa Fujita, Hiroyuki Yanai, Yasuharu Sato
Diagnostics.2021; 11(12): 2284. CrossRef - Diagnosis and treatment of secretory carcinoma arising from the oral minor salivary gland
Masaru Ogawa, Satoshi Yokoo, Takahiro Yamaguchi, Keisuke Suzuki, Mai Seki-Soda, Takahiro Shimizu, Jun Kurihara, Takaya Makiguchi
Medicine.2021; 100(51): e28390. CrossRef - A biphasic sessile mass of the buccal mucosa
Tiffany M. Peters, James A. Phero, Brent A. Golden, Alice E. Curran
Oral Surgery, Oral Medicine, Oral Pathology and Oral Radiology.2020; 130(6): 612. CrossRef - Don't stop the champions of research now: a brief history of head and neck pathology developments
Lester D.R. Thompson, James S. Lewis, Alena Skálová, Justin A. Bishop
Human Pathology.2020; 95: 1. CrossRef - High Grade Transformation in Mucoepidermoid Carcinoma of the Minor Salivary Gland with Polyploidy of the Rearranged MAML2 Gene
Hyun Lee, Jong-Lyel Roh, Young-Jun Choi, Jene Choi, Kyung-Ja Cho
Head and Neck Pathology.2020; 14(3): 822. CrossRef - Pan‐Trk immunohistochemistry is a sensitive and specific ancillary tool for diagnosing secretory carcinoma of the salivary gland and detecting ETV6–NTRK3 fusion
Bin Xu, Mohamed R Haroon Al Rasheed, Cristina R Antonescu, Deepu Alex, Denise Frosina, Ronald Ghossein, Achim A Jungbluth, Nora Katabi
Histopathology.2020; 76(3): 375. CrossRef - Characterization of novel genetic alterations in salivary gland secretory carcinoma
Kiyong Na, Juan C. Hernandez-Prera, Jae-Yol Lim, Ha Young Woo, Sun Och Yoon
Modern Pathology.2020; 33(4): 541. CrossRef - Mammary analogue secretory carcinoma: An Indian experience of a novel entity
Zeba Nisar, JaydeepN Pol, RakhiV Jagdale, MadhuraD Phadke, GirishA Kadkol
Indian Journal of Pathology and Microbiology.2020; 63(5): 134. CrossRef - Secretory Carcinoma of Salivary Gland with High-Grade Histology Arising in Hard Palate: A Case Report
Kiyofumi Takabatake, Keisuke Nakano, Hotaka Kawai, Saori Yoshida, Haruka Omori, May Wathone Oo, Shan Qiusheng, Kenichiro Uchida, Katsuaki Mishima, Hitoshi Nagatsuka
Reports.2020; 3(2): 6. CrossRef - Secretory carcinoma of the salivary gland (mammary analogue secretory carcinoma) in children
I. V. Sidorov, I. S. Kletskaya, D. M. Konovalov
Arkhiv patologii.2020; 82(2): 43. CrossRef - Secretory carcinoma of the major salivary gland: Provincial population‐based analysis of clinical behavior and outcomes
Gareth Ayre, Martin Hyrcza, Jonn Wu, Eric Berthelet, Alena Skálová, Tom Thomson
Head & Neck.2019; 41(5): 1227. CrossRef - Higher Ki67 Index, Nodal Involvement, and Invasive Growth Were High Risk Factors for Worse Prognosis in Conventional Mammary Analogue Secretory Carcinoma
Jingjing Sun, Lizhen Wang, Zhen Tian, Yuhua Hu, Ronghui Xia, Jiang Li
Journal of Oral and Maxillofacial Surgery.2019; 77(6): 1187. CrossRef - Mammary analogue secretory carcinoma of salivary gland diagnosed on submandibular gland cytology: A case report and review of the literature
Ethar Al‐Husseinawi, Soheila Hamidpour, Evanthia Omoscharka
Cytopathology.2019; 30(3): 318. CrossRef - Secretory Carcinoma of Minor Salivary Gland in Buccal Mucosa: A Case Report and Review of the Literature
Durga Paudel, Michiko Nishimura, Bhoj Raj Adhikari, Daichi Hiraki, Aya Onishi, Tetsuro Morikawa, Puja Neopane, Sarita Giri, Koki Yoshida, Jun Sato, Masayuki Ono, Yoshitaka Kamino, Hiroki Nagayasu, Yoshihiro Abiko
Case Reports in Pathology.2019; 2019: 1. CrossRef - Estrogen Receptor, Progesterone Receptor, and HER-2 Expression in Recurrent Pleomorphic Adenoma
Ana Amélia de Souza, Albina Altemani, Ney Soares de Araujo, Lucas Novaes Texeira, Vera Cavalcanti de Araújo, Andresa Borges Soares
Clinical Pathology.2019;[Epub] CrossRef - Sinonasal Secretory Carcinoma of Salivary Gland with High Grade Transformation: A Case Report of this Under-Recognized Diagnostic Entity with Prognostic and Therapeutic Implications
Bin Xu, Ruth Aryeequaye, Lu Wang, Nora Katabi
Head and Neck Pathology.2018; 12(2): 274. CrossRef - Primary mammary analogue secretory carcinoma of the lung: a case report
Tao Huang, Jonathan B. McHugh, Gerald J. Berry, Jeffrey L. Myers
Human Pathology.2018; 74: 109. CrossRef - Salivary Secretory Carcinoma With a Novel ETV6-MET Fusion
Lisa M. Rooper, Theodoros Karantanos, Yi Ning, Justin A. Bishop, Sarah W. Gordon, Hyunseok Kang
American Journal of Surgical Pathology.2018; 42(8): 1121. CrossRef - Secretory carcinoma: The eastern Canadian experience and literature review
David Forner, Martin Bullock, Daniel Manders, Timothy Wallace, Christopher J. Chin, Liane B. Johnson, Matthew H. Rigby, Jonathan R. Trites, Mark S. Taylor, Robert D. Hart
Journal of Otolaryngology - Head & Neck Surgery.2018;[Epub] CrossRef - Newly described salivary gland tumors
Alena Skalova, Michal Michal, Roderick HW Simpson
Modern Pathology.2017; 30: S27. CrossRef - Diagnóstico, tratamiento y seguimiento de un tumor de reciente descripción: el carcinoma análogo secretor de mama (MASC) de glándula salival. A propósito de 2 nuevos casos
Marina Alexandra Gavín-Clavero, M. Victoria Simón-Sanz, Ana M. López-López, Alberto Valero-Torres, Esther Saura-Fillat
Revista Española de Cirugía Oral y Maxilofacial.2017; 39(4): 221. CrossRef - Systematic review of mammary analog secretory carcinoma of salivary glands at 7 years after description
Bacem A. Khalele
Head & Neck.2017; 39(6): 1243. CrossRef - Salivary Gland Secretory Carcinoma With High-Grade Transformation, CDKN2A/B Loss, Distant Metastasis, and Lack of Sustained Response to Crizotinib
Nicole A. Cipriani, Elizabeth A. Blair, Joshua Finkle, Jennifer L. Kraninger, Christopher M. Straus, Victoria M. Villaflor, Daniel Thomas Ginat
International Journal of Surgical Pathology.2017; 25(7): 613. CrossRef - A systematic review including an additional pediatric case report: Pediatric cases of mammary analogue secretory carcinoma
Amanda L. Ngouajio, Sarah M. Drejet, D. Ryan Phillips, Don-John Summerlin, John P. Dahl
International Journal of Pediatric Otorhinolaryngology.2017; 100: 187. CrossRef - Newly Described Entities in Salivary Gland Pathology
Alena Skálová, Douglas R. Gnepp, James S. Lewis, Jennifer L. Hunt, Justin A. Bishop, Henrik Hellquist, Alessandra Rinaldo, Vincent Vander Poorten, Alfio Ferlito
American Journal of Surgical Pathology.2017; 41(8): e33. CrossRef - Mammary Analogue Secretory Carcinoma of Salivary Glands: Diagnostic Pitfall with Distinct Immunohistochemical Profile and Molecular Features
Oliver Bissinger, Carolin Götz, Andreas Kolk, Henning A. Bier, Abbas Agaimy, Henning Frenzel, Sven Perner, Julika Ribbat-Idel, Klaus Dietrich Wolff, Wilko Weichert, Caroline Mogler
Rare Tumors.2017; 9(3): 89. CrossRef - Mammary Analogue Secretory Carcinoma of Salivary Gland. A Case Report Emphasizing its Diagnostic Histological, Immunohistochemistry and Molecular Findings
Monalisa Hui, Shantveer G Uppin, Vamshi Krishna Thamtam, Abhiram Kalle
International Journal of Head and Neck Surgery.2017; 8(4): 160. CrossRef - A case of mammary analog secretory carcinoma of the lower lip
Takako Aizawa, Taro Okui, Ken Kitagawa, Yoshikazu Kobayashi, Koji Satoh, Hideki Mizutani
Journal of Oral and Maxillofacial Surgery, Medicine, and Pathology.2016; 28(3): 277. CrossRef - Mammary analogue secretory carcinoma of parotid: Is preoperative cytological diagnosis possible?
Nikita Oza, Kintan Sanghvi, Tanuja Shet, Asawari Patil, Santosh Menon, Mukta Ramadwar, Shubhada Kane
Diagnostic Cytopathology.2016; 44(6): 519. CrossRef - Lysozyme Expression Can be Useful to Distinguish Mammary Analog Secretory Carcinoma from Acinic Cell Carcinoma of Salivary Glands
Fernanda Viviane Mariano, Camila Andrea Concha Gómez, Juliana de Souza do Nascimento, Harim Tavares dos Santos, Erika Said Egal, Victor Angelo Martins Montalli, Pablo Agustin Vargas, Oslei Paes de Almeida, Albina Altemani
Head and Neck Pathology.2016; 10(4): 429. CrossRef - Cytogenetic and immunohistochemical characterization of mammary analogue secretory carcinoma of salivary glands
Syed A. Khurram, Jemel Sultan-Khan, Neil Atkey, Paul M. Speight
Oral Surgery, Oral Medicine, Oral Pathology and Oral Radiology.2016; 122(6): 731. CrossRef - The role of DOG1 immunohistochemistry in dermatopathology
Keisuke Goto
Journal of Cutaneous Pathology.2016; 43(11): 974. CrossRef - Extended immunologic and genetic lineage of mammary analogue secretory carcinoma of salivary glands
Hao Ni, Xue-ping Zhang, Xiao-tong Wang, Qiu-yuan Xia, Jing-huan Lv, Xuan Wang, Shan-shan Shi, Rui Li, Xiao-jun Zhou, Qiu Rao
Human Pathology.2016; 58: 97. CrossRef - A New Hitherto Unreported Histopathologic Manifestation of Mammary Analogue Secretory Carcinoma: “Masked MASC” Associated With Low-grade Mucinous Adenocarcinoma and Low-grade In Situ Carcinoma Components
Fredrik Petersson, Michael Michal, Dmitry V. Kazakov, Petr Grossmann, Michal Michal
Applied Immunohistochemistry & Molecular Morphology.2016; 24(9): e80. CrossRef - Papillary-cystic pattern is characteristic in mammary analogue secretory carcinomas but is rarely observed in acinic cell carcinomas of the salivary gland
Min-Shu Hsieh, Yueh-Hung Chou, Shin-Joe Yeh, Yih-Leong Chang
Virchows Archiv.2015; 467(2): 145. CrossRef - Mammary analog secretory carcinoma, low-grade salivary duct carcinoma, and mimickers: a comparative study
Todd M Stevens, Andra O Kovalovsky, Claudia Velosa, Qiuying Shi, Qian Dai, Randall P Owen, Walter C Bell, Shi Wei, Pamela A Althof, Jennifer N Sanmann, Larissa Sweeny, William R Carroll, Gene P Siegal, Martin J Bullock, Margaret Brandwein-Gensler
Modern Pathology.2015; 28(8): 1084. CrossRef - Aspiration cytology of mammary analogue secretory carcinoma of the salivary gland
Min Jung Jung, Sang Yoon Kim, Soon Yuhl Nam, Jong‐Lyel Roh, Seung‐Ho Choi, Jeong Hyun Lee, Jung Hwan Baek, Kyung‐Ja Cho
Diagnostic Cytopathology.2015; 43(4): 287. CrossRef - A Unique Case of a Cutaneous Lesion Resembling Mammary Analog Secretory Carcinoma
Jennifer Albus, Jacqueline Batanian, Bruce M. Wenig, Claudia I. Vidal
The American Journal of Dermatopathology.2015; 37(4): e41. CrossRef - Mammary analogue secretory carcinoma of salivary glands: a new entity associated with ETV6 gene rearrangement
Hanna Majewska, Alena Skálová, Dominik Stodulski, Adéla Klimková, Petr Steiner, Czesław Stankiewicz, Wojciech Biernat
Virchows Archiv.2015; 466(3): 245. CrossRef - A comparative immunohistochemistry study of diagnostic tools in salivary gland tumors: usefulness of mammaglobin, gross cystic disease fluid protein 15, and p63 cytoplasmic staining for the diagnosis of mammary analog secretory carcinoma?
Fabrice Projetti, Magali Lacroix‐Triki, Elie Serrano, Sebastien Vergez, Béatrice Herbault Barres, Julie Meilleroux, Marie‐Bernadette Delisle, Emmanuelle Uro‐Coste
Journal of Oral Pathology & Medicine.2015; 44(4): 244. CrossRef - Mammary Analogue Secretory Carcinoma of Salivary Glands
Yohei Ito, Kenichiro Ishibashi, Ayako Masaki, Kana Fujii, Yukio Fujiyoshi, Hideo Hattori, Daisuke Kawakita, Manabu Matsumoto, Satoru Miyabe, Kazuo Shimozato, Toshitaka Nagao, Hiroshi Inagaki
American Journal of Surgical Pathology.2015; 39(5): 602. CrossRef - Cytopathological features of mammary analogue secretory carcinoma—Review of literature
Maiko Takeda, Takahiko Kasai, Kohei Morita, Mao Takeuchi, Takeshi Nishikawa, Akinori Yamashita, Shinji Mikami, Hiroshi Hosoi, Chiho Ohbayashi
Diagnostic Cytopathology.2015; 43(2): 131. CrossRef - Mammary Analog Secretory Carcinoma of Salivary Glands
Justin A. Bishop
Pathology Case Reviews.2015; 20(1): 7. CrossRef - Diagnostic utility of phosphorylated signal transducer and activator of transcription 5 immunostaining in the diagnosis of mammary analogue secretory carcinoma of the salivary gland: A comparative study of salivary gland cancers
Akihiko Kawahara, Tomoki Taira, Hideyuki Abe, Yorihiko Takase, Takashi Kurita, Eiji Sadashima, Satoshi Hattori, Ichio Imamura, Shinji Matsumoto, Hitomi Fujisaki, Kazunobu Sueyoshi, Jun Akiba, Masayoshi Kage
Cancer Cytopathology.2015; 123(10): 603. CrossRef - A Case of Mammary Analogue Secretory Carcinoma Arising from Parotid Gland
Hee Tae Kim, Cha Hee Lee, Han Su Kim, Hae Sang Park
Korean Journal of Otorhinolaryngology-Head and Neck Surgery.2015; 58(8): 563. CrossRef - WITHDRAWN: A biphasic sessile mass of the buccal mucosa
Tiffany M. Peters, Jose P. Zevallos, Brent A. Golden, Alice E. Curran
Oral Surgery, Oral Medicine, Oral Pathology and Oral Radiology.2015;[Epub] CrossRef - Mammary analogue secretory carcinoma: an evaluation of its clinicopathological and genetic characteristics
Peter P. Luk, Christina I. Selinger, Timothy J. Eviston, Trina Lum, Bing Yu, Sandra A. O’Toole, Jonathan R. Clark, Ruta Gupta
Pathology.2015; 47(7): 659. CrossRef - A case of mammary analogue secretory carcinoma arising in the submandibular region
Kotaro ISHII, Koji NAKAMATSU, Koji SATO, Chikashi MINEMURA, Wataru KUMAMARU, Hiroyo YOSHIKAWA
Japanese Journal of Oral and Maxillofacial Surgery.2015; 61(11): 564. CrossRef - DOG1, p63, and S100 protein: a novel immunohistochemical panel in the differential diagnosis of oncocytic salivary gland neoplasms in fine-needle aspiration cell blocks
Alessandra C. Schmitt, Ryan McCormick, Cynthia Cohen, Momin T. Siddiqui
Journal of the American Society of Cytopathology.2014; 3(6): 303. CrossRef - Salivary Gland Tumor “Wishes” to Add to the Next WHO Tumor Classification: Sclerosing Polycystic Adenosis, Mammary Analogue Secretory Carcinoma, Cribriform Adenocarcinoma of the Tongue and Other Sites, and Mucinous Variant of Myoepithelioma
Douglas R. Gnepp
Head and Neck Pathology.2014; 8(1): 42. CrossRef - Hepatoid differentiation in renal cell carcinoma: a rare histologic pattern with clinical significance
Jungweon Shim, Heounjeong Go, Young-Suk Lim, Kyung Chul Moon, Jae Y. Ro, Yong Mee Cho
Annals of Diagnostic Pathology.2014; 18(6): 363. CrossRef - Mammary analogue secretory carcinoma: Update on a new diagnosis of salivary gland malignancy
Roshan Sethi, Elliott Kozin, Aaron Remenschneider, Josh Meier, Paul VanderLaan, William Faquin, Daniel Deschler, Robert Frankenthaler
The Laryngoscope.2014; 124(1): 188. CrossRef - Mammary analog secretory carcinoma of salivary gland in a 5 year old: Case report
Matthew Keisling, Michael Bianchi, Judy Mae Pascasio
International Journal of Pediatric Otorhinolaryngology Extra.2014; 9(4): 163. CrossRef - Fine-Needle Aspiration Cytology of Mammary Analog Secretory Carcinoma Masquerading as Low-Grade Mucoepidermoid Carcinoma: Case Report with a Review of the Literature
Jaya Bajaj, Cecilia Gimenez, Farah Slim, Mohamed Aziz, Kasturi Das
Acta Cytologica.2014; 58(5): 501. CrossRef
 PubReader
PubReader ePub Link
ePub Link-
 Cite this Article
Cite this Article
- Cite this Article
-
- Close
- Download Citation
- Close
- Figure



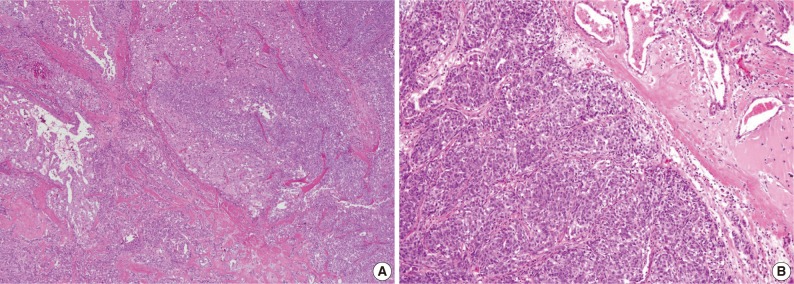


Fig. 1
Fig. 2
Fig. 3
Fig. 4
Fig. 5
Fig. 6
| Primary antibodies | Source | Dilution | Clone |
|---|---|---|---|
| S100 protein | Zymed | 1 : 200 | Mouse monoclonal |
| ER | Novo | 1 : 50 | Mouse monoclonal |
| PR | Novo | 1 : 200 | Mouse monoclonal |
| GCDFP-15 | Neomarkers | 1 : 50 | Mouse monoclonal |
| DOG1 | Spring Science | 1 : 200 | Rabbit polyclonal |
| Feature | MASC (n = 13) | Mimic MASC (n = 10) | Conventional AciCC (n = 15) | |
|---|---|---|---|---|
| Sex | Male | 8 | 8 | 3 |
| Female | 5 | 2 | 12 | |
| Average age (yr) | 46.4 (17-76) | 46.3 (15-75) | 44.1 (24-65) | |
| Site | Parotid gland | 11 | 6 | 13 |
| Non-parotid gland | 2 | 4 | 2 | |
| Tumor size (cm) | 1.77 (0.7-2.5) | 2.34 (1.0-3.3) | 2.57 (1.0-5.5) | |
| Pathologic T stage | T1 | 6 | 2 | 5 |
| T2 | 3 | 2 | 9 | |
| T3 | 4 | 6 | 1 | |
| Lymph node metastasis | 0 | 2 | 0 | |
| Treatment | Surgery | 11 | 7 | 13 |
| Surgery+radiation | 2 | 3 | 2 | |
| Follow-up | Died of disease | 0 | 0 | 0 |
| Local recurrence | 3 | 0 | 1 | |
| Metastasis | 0 | 2 | 0 | |
| No evidence of disease | 6 | 7 | 11 | |
| Loss | 4 | 1 | 3 |
| S100 protein | GCDFP-15 | ER | PR | DOG1 | |
|---|---|---|---|---|---|
| MASC | 13/13 | 2/13 | 0/13 | 0/13 | 1/13 |
| Mimic MASC | 3/10 | 3/10 | 0/10 | 0/13 | 0/10 |
| Conventional AciCC | 0/6 | 0/6 | 0/6 | 0/6 | 3/6 |
ER, estrogen receptor; PR, progesterone receptor; GCDFP-15, gross cystic disease fluid protein 15.
MASC, mammary analogue secretory carcinoma; AciCC, acinic cell carcinoma.
MASC, mammary analogue secretory carcinoma; AciCC, acinic cell carcinoma; GCDEP-15, gross cystic disease fluid protein 15; ER, estrogen receptor; PR, progesterone receptor.

 E-submission
E-submission