Articles
- Page Path
- HOME > J Pathol Transl Med > Volume 47(2); 2013 > Article
-
Case Study
ThinPrep Cytological Findings of Desmoplastic Small Round Cell Tumor with Extensive Glandular Differentiation: A Case Study - Hyun-Jung Kim, Byeong Seok Sohn1, Ji-Eun Kwon2, Jeong Yeon Kim, Kyeongmee Park
-
Korean Journal of Pathology 2013;47(2):182-187.
DOI: https://doi.org/10.4132/KoreanJPathol.2013.47.2.182
Published online: April 24, 2013
Department of Pathology, Sanggye Paik Hospital, Inje University College of Medicine, Seoul, Korea.
1Department of Internal Medicine, Sanggye Paik Hospital, Inje University College of Medicine, Seoul, Korea.
2Department of Pathology, Ajou University Hospital, Ajou University School of Medicine, Suwon, Korea.
- Corresponding Author: Hyun-Jung Kim, M.D. Department of Pathology, Sanggye Paik Hospital, Inje University,1342 Dongil-ro, Nowon-gu, Seoul 139-707, Korea. Tel: +82-2-950-1261, Fax: +82-2-951-6964, hjkim@paik.ac.kr
© 2013 The Korean Society of Pathologists/The Korean Society for Cytopathology
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/3.0/) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
Abstract
- Desmoplastic small round cell tumor (DSRCT) is a rare and highly aggressive neoplasm. The cytological diagnosis of this tumor has only been reported in a few cases. In most of these cases, the diagnosis was made using fine-needle aspiration cytology. Most DSRCTs resemble disseminated carcinomatoses in their clinical manifestation as well as cytomorphologically, even in young-adult patients. These authors report a case of using peritoneal-washing and pleural-effusion ThinPrep cytology to diagnose DSRCT, with extensive glandular differentiation and mucin vacuoles. We found that fibrillary stromal fragment, clinical setting, and adjunctive immunocytochemical staining were most helpful for avoiding misdiagnosis.
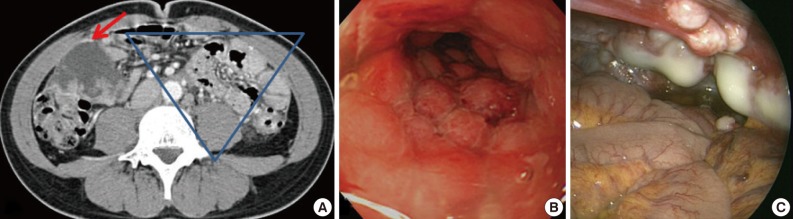
- A 31-year-old man was admitted for constipation and abdominal pain that had persisted for a month. The patient was transferred for an abdominal mass on an abdominal sonogram at local clinic. Physical examination revealed a fist-sized mass on the right lower quadrant of abdomen. The initial abdominopelvic computer-tomography images showed a 5.5 cm-sized mass at the right lower quadrant with surrounding seeding nodules and mesenteric lymphadenopathy, suggesting carcinomatosis (Fig. 1A). The colonoscopy revealed a diffuse luminal narrowing due to the extrinsic masses (Fig. 1B). The laparoscopic finding showed multiple disseminated, variable-sized, firm, whitish masses or plaques on the hepatic capsule and the serosal surface of the colon (Fig. 1C).
- Cytological findings
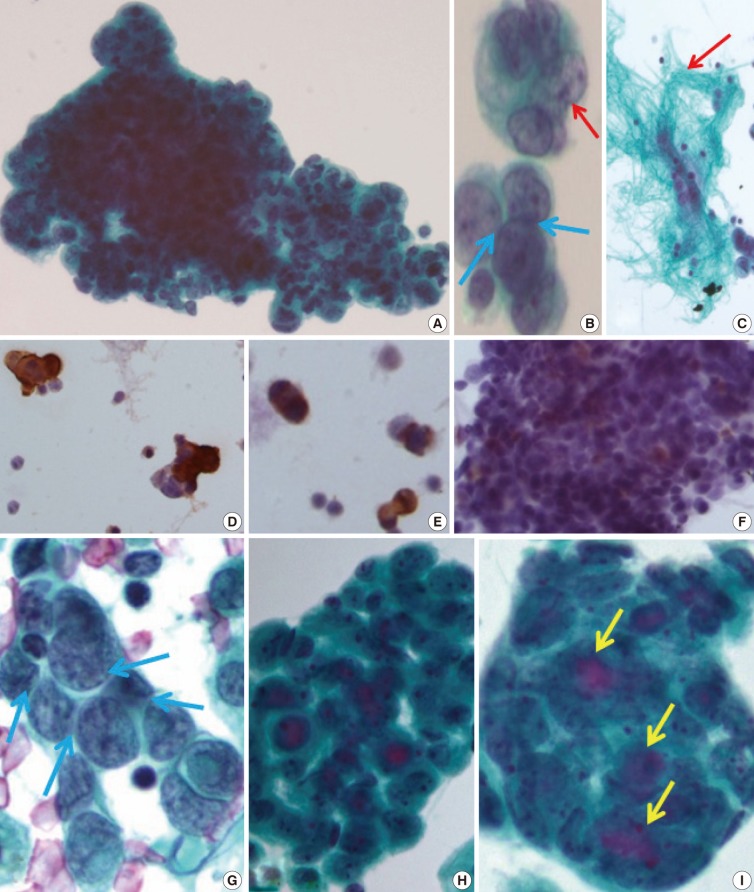
- The initial peritoneal-washing fluid (ThinPrep) diagnosed the tumors as "adenocarcinoma" because the clusters of tumor cells revealed finger-like buddings (Fig. 2A). The high power field disclosed common outer-cell borders, intracytoplasmic vacuoles, convoluted nuclei, and relatively large prominent nucleoli. However, retrospective cytological review of the distinct nuclear moldings that were also identified, favored the diagnosis of small round cell tumors (Fig. 2B). There were a few fragments of fibrillary stroma (Fig. 2C). The immunocytochemical stainings were positive for vimentin (Fig. 2D), pancytokeratin (panCK) (Fig. 2E), and vaguely positive for desmin (Fig. 2F).
- After eight months, a malignant pleural-effusion was detected. Conventional fluid cytology showed cytological features similar to those seen from the peritoneal washing, but angulated nuclei with distinct nuclear moldings were also present (Fig. 2G). In ThinPrep findings, the clusters were more rounded, with three-dimensional structures, and even extracellular mucin content, very similar to adenocarcinoma (Fig. 2H, I).
- Histologic findings
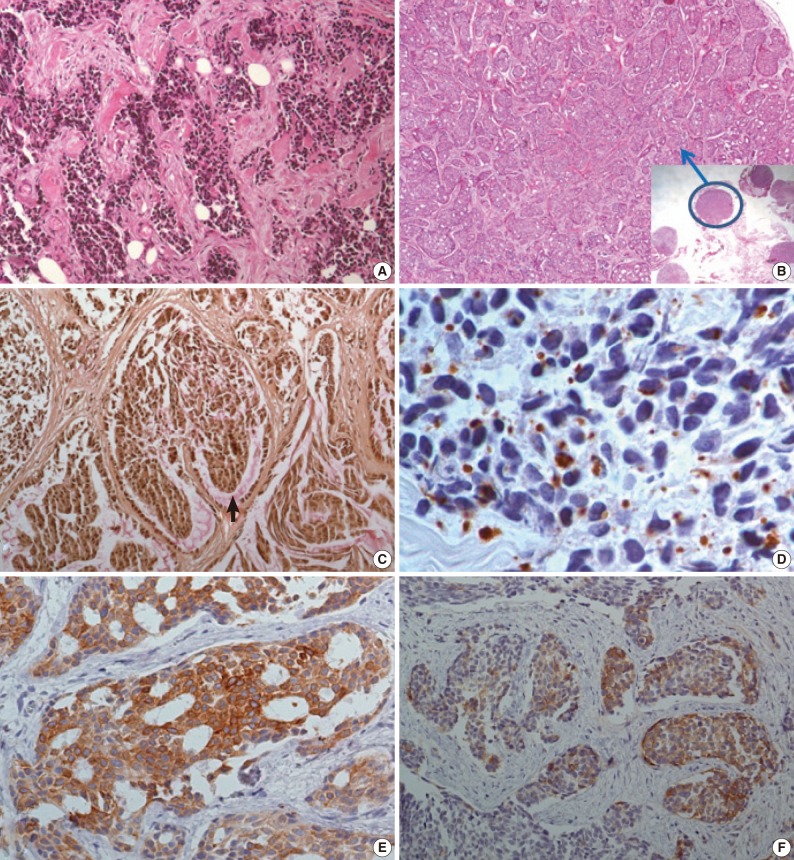
- The initial laparoscopic biopsy showed typical findings of DSRCT, desmoplastic stroma, and variable-sized nests of small round cells (Fig. 3A). Four months later, a second biopsy specimen was obtained during T-loop colostomy for a marked-intestinal-obstruction relief. There were multiple nodules on the mesentery. Each nodule had diverse morphology (Fig. 3B). The mucicarmine staining was positive for intraglandular content (Fig 3C). Immunohistochemical staining disclosed dot-like desmin positivity (Fig. 3D). Co-expression of panCK and vimentin was variable in each nodule. In the gland-forming portion, the tumor cells were strongly positive for panCK (Fig. 3E) and weakly positive for vimentin. Neuronal differentiation was also variable (Fig. 3F).
- Follow-up
- Despite the diagnosis of DSRCT, the patient refused chemotherapy and the disease rapidly progressed. The patient passed away a year after the first biopsy due to the increase in size and volume of the intra-abdominal masses and associated complications (intestinal and ureteral obstruction, hydronephrosis, and chronic renal failure).
CASE REPORT
- DSRCT is a rare neoplasm, usually affecting adolescent male patients.1 It is difficult to distinguish from other small round cell tumors, because the cytological findings consistently include the following features: loosely clustered cellular population of round, oval, or spindled blue cells with scant cytoplasm, irregular nuclear membranes, finely granular chromatin, and inconspicuous nucleoli. Differential diagnoses are mainly Ewing sarcoma/primitive neuroectodermal tumour, small cell neuroendocrine carcinoma, malignant lymphoma, embryonal/alveolar rhabdomyosarcoma, and neuroblastoma/Wilms tumor. Ultimately, cytologic differentiation is limited. To the diagnosis of DSRCT, however, can be established using the correlation of the clinical, cytological, and immunohistochemical features.2-4 Small cell neuroendocrine carcinoma is associated with a much older patient population and usually originates in the lung. Neuroblastoma and Wilms tumors, however, occur in very young children. High-grade lymphoma often demonstrates a diffuse growth pattern and does not exhibit the characteristic cohesion of DSRCT. The most distinct cytological feature of DSRCT compared to the other small round cell tumors, is its stromal fibrillary fragment, which is not a commonly found in body fluid, and can only be found using fine-needle aspiration cytology. In the case presented herein, there were occasional slender fragments of fibrillary tissue in the peritoneal-washing fluid, which were mixed with dis-cohesive single tumor cells but were not noted in the pleural fluid.
- Immunohistochemically, DSRCT may show a various differentiation, which is a striking feature of the tumor. Typically, tumor cells are immunoreactive for epithelial (keratin and epithelial-membrane antigen), mesenchymal (vimentin), myogenic (desmin), and neural (neuron-specific enolase and CD56) markers. Immunostaining for desmin frequently shows a distinct staining pattern: namely, punctuate and perinuclear positivity. In this case, desmin immunostaining was diagnostic for DSRCT.
- Attempts have been made to use immunocytochemical staining from small, round, blue cell tumor groups for the differential diagnosis of DSRCT.3 The results of the case presented herein are similar to those of the previous reported cases (vimentin [+], panCK [+], and desmin [weak+]). In this case, however, an additional interesting point is the divergent differentiation among different nodules. Each nodule showed that typical DSRCT had extended to extensive glandular differentiation, with mucicarmine-positive mucin content. The immunohistochemical profile is also quite for each nodule. The typical DSRCT nodule shows a weak epithelial marker and dot-like desmin positivity, but the extensive glandular-differentiated nodule is strongly positive for panCK and weakly positive/negative for vimentin and desmin. There is only one case report mentioning these various cytologic differentiations.5 In that paper, the same various cytologic differentiations are noted (inconspicuous to conspicuous nucleoli and with a different immunohistochemistry).
- The most well known molecular event was a specific reciprocal translocation t(11; 22)(p13; 12). Shen et al.6 and Roberts et al.7 described a variant where other chromosomes were involved in addition to chromosomes 11 and 22. The translocation t(11; 22)(p13; 12) involves the EWS gene in 22q24 and the WT1 gene in 11p13. This translocation produces the chimeric transcript EWS/WT1 and the related WT1 protein, which can be detected immunohistochemically. The case presented herein, however, was negative for WT1. There have been some trials of the molecular diagnosis of small round cell tumors via fine-needle aspiration.8 In comparison, immunocytochemical analysis resolved 19/25 Ewing sarcomas (76%), nine (82%) of the 11 rhabdomyosarcomas, six (46%) of the 13 neuroblastomas, and one (50%) of the two DSRCTs. Overall, reverse transcription polymerase chain reaction resolved 38 (86%) of 44 vs 35 (69%) of 51 cases through immunocytochemical analysis. There have been some reports of the molecular confirmation of DSRCT with a cytological sample.9
- ThinPrep smear disclosed that the cytoplasm of tumor cells was well-preserved. The clusters, however, were more rounded with a three-dimensional structure than those of a conventional smear, and were thus very similar to adenocarcinoma. In some of the literatures, liquid-based cytomorphology in serous fluid could be used to make an accurate diagnosis, as it showed excellent morphology against a clear background, with preservation of the three-dimensional configuration and a sufficient amount of extracellular material.10 In our pleural fluid case, the liquid-based cytology smear showed more glandular differentiation than a conventional smear, including enlarged nuclei, prominent nucleoli, and an extracellular mucinous material. Therefore, we should carefully examine ThinPrep smear compared to the conventional smear for cytologic differential diagnosis of DSRCT from adenocarcinoma.
- In conclusion, the cytological diagnosis of DSRCT via cytomorphology is very difficult. Clinicopathologic consideration and adjunctive immunocytochemistry and molecular study would be helpful for avoiding the misdiagnosis of "disseminated adenocarcinoma."
DISCUSSION
Acknowledgments
Acknowledgments
This subject was presented at 2011 Korean-Japanese Cytopathology meeting (Poster#16).
No potential conflict of interest relevant to this article was reported.
- 1. Fletcher CD, Unni KK, Metens F. Pathology and genetics of tumours of soft tissue and bone. 2002; Lyon: IARC Press.
- 2. Granja NM, Begnami MD, Bortolan J, Filho AL, Schmitt FC. Desmoplastic small round cell tumour: cytological and immunocytochemical features. Cytojournal 2005; 2: 6.ArticlePubMedPMC
- 3. Dave B, Shet T, Chinoy R. Desmoplastic round cell tumor of childhood: can cytology with immunocytochemistry serve as an alternative for tissue diagnosis? Diagn Cytopathol 2005; 32: 330-335. ArticlePubMed
- 4. Chang F. Desmoplastic small round cell tumors: cytologic, histologic, and immunohistochemical features. Arch Pathol Lab Med 2006; 130: 728-732. ArticlePubMedPDF
- 5. Presley AE, Kong CS, Rowe DM, Atkins KA. Cytology of desmoplastic small round-cell tumor: comparison of pre- and post-chemotherapy fine-needle aspiration biopsies. Cancer 2007; 111: 41-46. ArticlePubMed
- 6. Shen WP, Towne B, Zadeh TM. Cytogenetic abnormalities in an intraabdominal desmoplastic small cell tumor. Cancer Genet Cytogenet 1992; 64: 189-191. ArticlePubMed
- 7. Roberts P, Burchill SA, Beddow RA, Wheeldon J, Cullinane C, Lewis IJ. A combined cytogenetic and molecular approach to diagnosis in a case of desmoplastic small round cell tumor with a complex translocation (11;22;21). Cancer Genet Cytogenet 1999; 108: 19-25. ArticlePubMed
- 8. Gautam U, Srinivasan R, Rajwanshi A, Bansal D, Marwaha RK, Vasishtha RK. Reverse transcriptase-polymerase chain reaction as an ancillary molecular technique in the diagnosis of small blue round cell tumors by fine-needle aspiration cytology. Am J Clin Pathol 2010; 133: 633-645. ArticlePubMedPDF
- 9. Perez RP, Zhang PJ. Detection of EWS-WT1 fusion mRNA in ascites of a patient with desmoplastic small round cell tumor by RT-PCR. Hum Pathol 1999; 30: 239-242. PubMed
- 10. Gabriel C, Achten R, Drijkoningen M. Use of liquid-based cytology in serous fluids: a comparison with conventional cytopreparatory techniques. Acta Cytol 2004; 48: 825-835. PubMed
REFERENCES
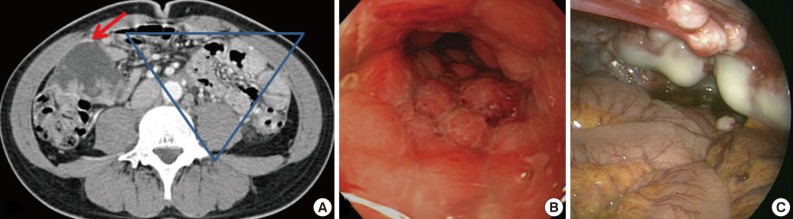
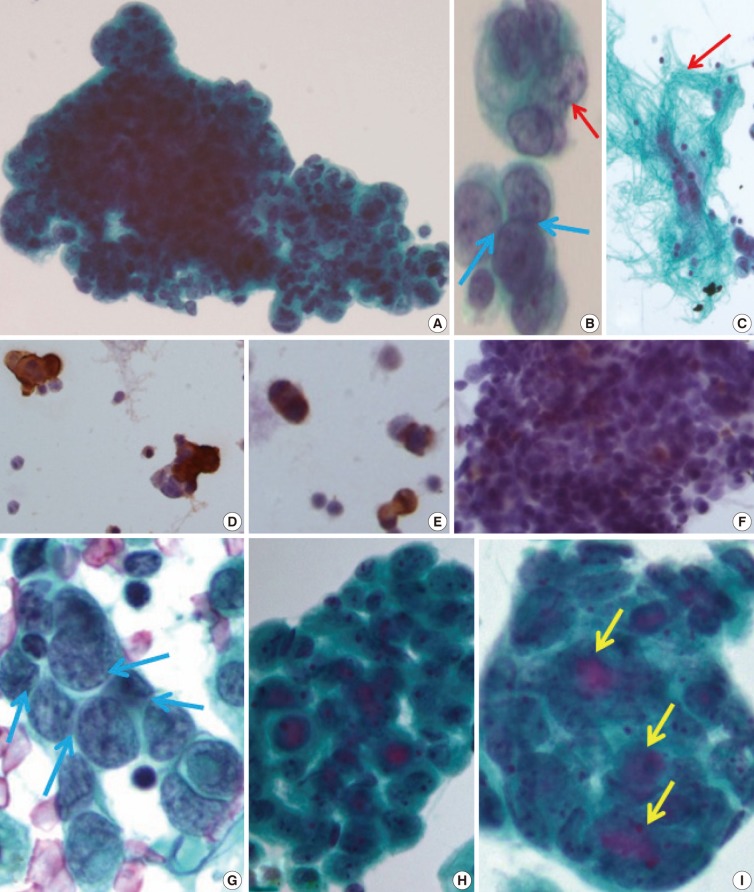
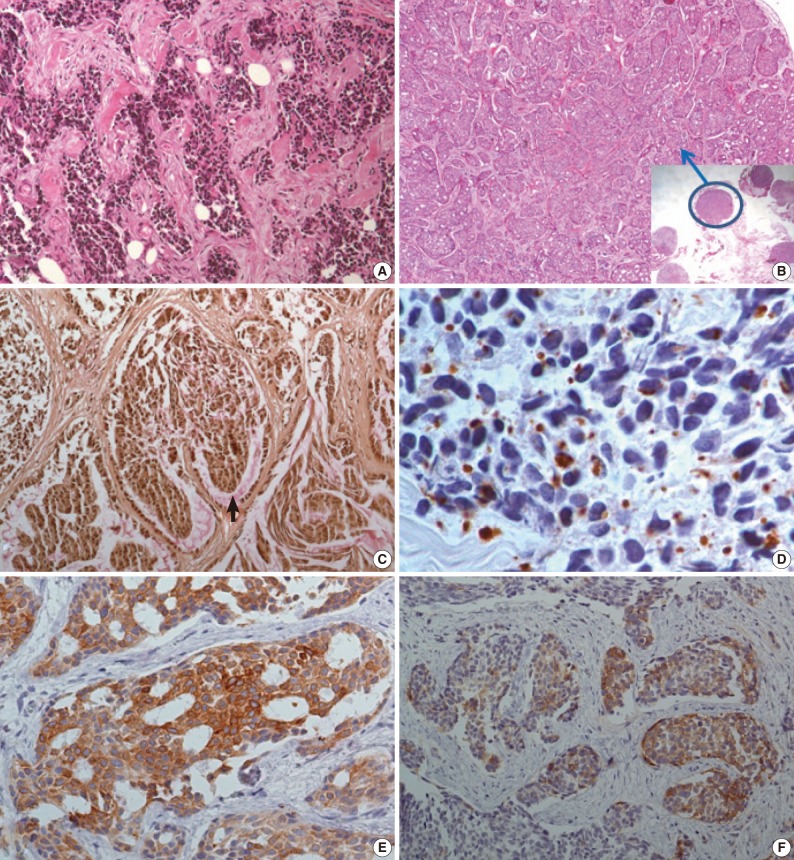
Figure & Data
References
Citations

- Desmoplastic Small Round Cell Tumor: Study of Cytomorphologic and Immunophenotypical Features in Seven Cases, One With Unusual Rhabdoid Morphology
Sameer Chhetri Aryal, Khalid Shittu, Mohamed Mustafa, Fatimah I. Alruwaii, Kyle D. Perry, Lisi Yuan
Diagnostic Cytopathology.2026;[Epub] CrossRef - A Rare and Aggressive Abdominopelvic Tumor: A Case of Desmoplastic Small Round Cell Tumor in a Young Male
Yağmur Sena Tosun, Aytan Babazade, Emine Sena Sözen, Betül Erişmiş, Enes Seyda Şahiner
Caucasian Medical Journal.2025; 3(1): 4. CrossRef - Desmoplastic Small Round Cell Tumor Involving Serous Fluid: Cytologic Features and Diagnostic Pitfalls: A Series of 8 Cases
Nibras L Fakhri, Qiong Gan
American Journal of Clinical Pathology.2023; 160(4): 417. CrossRef - A Review of Effusion Cytomorphology of Small Round Cell Tumors
Lucy M. Han, Christopher J. VandenBussche, Mads Abildtrup, Ashish Chandra, Poonam Vohra
Acta Cytologica.2022; 66(4): 336. CrossRef - Intra-abdominal desmoplastic small blue round cell tumor: A case report
Tareq Hamed Al Taei, Hasan Al Fardan, Sarah Ali Al Mail
Radiology Case Reports.2022; 17(12): 4502. CrossRef - Desmoplastic Small Round Cell Tumor of the Kidney: Report of a Case, Literature Review, and Comprehensive Discussion of the Distinctive Morphologic, Immunohistochemical, and Molecular Features in the Differential Diagnosis of Small Round Cell Tumors Affec
Carlos A. Galliani, Michele Bisceglia, Antonio Del Giudice, Giuseppe Cretì
Advances in Anatomic Pathology.2020; 27(6): 408. CrossRef - Intra-abdominal desmoplastic small round cell tumors: CT and FDG-PET/CT findings with histopathological association
JINGJING CHEN, ZENGJIE WU, BINBIN SUN, DACHENG LI, ZHENGUANG WANG, FANGJUN LIU, HUI HUA
Oncology Letters.2016; 11(5): 3298. CrossRef - Desmoplastic small round cell tumor with sphere‐like clusters mimicking adenocarcinoma
Yukinori Hattori, Akihiko Yoshida, Naoshi Sasaki, Yasuo Shibuki, Kenji Tamura, Koji Tsuta
Diagnostic Cytopathology.2015; 43(3): 214. CrossRef - Tumor intraabdominal desmoplásico de células pequeñas y redondas
Andrés Alejandro Briseño-Hernández, Deissy Roxana Quezada-López, Lilia Edith Corona-Cobián, Agar Castañeda-Chávez, Alfonso Tonatiuh Duarte-Ojeda, Michel Dassaejv Macías-Amezcua
Cirugía y Cirujanos.2015; 83(3): 243. CrossRef - Intra-abdominal desmoplastic small round cell tumour
Andrés Alejandro Briseño-Hernández, Deissy Roxana Quezada-López, Lilia Edith Corona-Cobián, Agar Castañeda-Chávez, Alfonso Tonatiuh Duarte-Ojeda, Michel Dassaejv Macías-Amezcua
Cirugía y Cirujanos (English Edition).2015; 83(3): 243. CrossRef - Diagnostic Pitfalls of Differentiating Desmoplastic Small Round Cell Tumor (DSRCT) From Wilms Tumor (WT)
Michael A. Arnold, Lynn Schoenfield, Berkeley N. Limketkai, Christina A. Arnold
American Journal of Surgical Pathology.2014; 38(9): 1220. CrossRef
 PubReader
PubReader ePub Link
ePub Link-
 Cite this Article
Cite this Article
- Cite this Article
-
- Close
- Download Citation
- Close
- Figure




 E-submission
E-submission