Search
- Page Path
- HOME > Search
- Incidental serrated lesions of the appendix: analysis of 2,137 appendectomy specimens
- Ömer Atmış, Ecem Dokuzlu Küçük, Hanife Seda Mavili, Fatma Seher Pehlivan, Ayça Tan, Semin Ayhan
- Received December 5, 2025 Accepted February 9, 2026 Published online May 4, 2026
- DOI: https://doi.org/10.4132/jptm.2026.02.08 [Epub ahead of print]
- 188 View
- 8 Download
-
 Abstract
Abstract
 PDF
PDF - Background
Serrated lesions of the appendix are rare, often incidental findings in routine appendectomy specimens. Their true frequency, histopathologic spectrum, and anatomic distribution remain incompletely characterized, partly due to variability in sampling practices. Methods: We retrospectively reviewed 2,137 appendectomy specimens (2015–2025) from a single tertiary pathology center. Cases with histologically confirmed serrated lesions were reexamined, classified as hyperplastic polyp (HP) or sessile serrated lesion/polyp (SSL/P), and assessed for clinicopathologic parameters including lesion size, location, and associated pathologies. Nonparametric tests were used, with statistical significance defined as p < .05. Results: Serrated lesions were identified in 34 cases (1.6%), comprising 17 HPs (0.8%) and 19 SSL/Ps (0.9%). SSL/Ps were significantly larger than HPs (median 10.0 vs. 2.7 mm, p < .001) and were more frequently located in the distal appendix (68.4% vs. 33.3%, p = .045, one-tailed Fisher’s exact test). No dysplasia or traditional serrated adenoma was detected. Acute appendicitis was present in 88% of cases, and associated neoplasms in 9%. Conclusions: Appendiceal serrated lesions are uncommon and often incidental. In this large appendectomy series, SSL/Ps differed from HPs by larger size and distal predilection. These findings primarily support diagnostic awareness and optimized sampling/ grossing practices—particularly careful evaluation of the distal appendix—rather than clinical risk stratification. Further studies incorporating systematic clinical correlation and molecular/immunohistochemistry analyses are warranted.
- Solitary Peutz-Jeghers type harmartomatous polyp in duodenum with gastric foveolar epithelium: a case report
- Eugene Choi, Junghwan Lee, Youngsoo Park
- J Pathol Transl Med. 2023;57(2):128-131. Published online January 10, 2023
- DOI: https://doi.org/10.4132/jptm.2022.11.07
- 5,976 View
- 197 Download
- 1 Web of Science
- 1 Crossref
-
 Abstract
Abstract
 PDF
PDF - Peutz-Jeghers type hamartomatous polyp is known to be associated with Peutz-Jeghers syndrome, which shows characteristic multiple hamartomatous polyp involvement in the gastrointestinal tract, combined with mucocutaneous symptom, familial history of Peutz- Jeghers syndrome or STK11/LTB1 mutation. However, some cases showing histologic appearance of the polyps discovered in Peutz- Jeghers syndrome while lacking other diagnostic criteria of the syndrome have been reported, and these are called solitary Peutz- Jeghers type polyps. Herein, we report a case of solitary Peutz-Jeghers type polyp covered with heterotopic epithelium. The patient was 47-year-old female without any mucocutaneous symptoms nor familial history of Peutz-Jeghers syndrome. Microscopic examination revealed Peutz-Jeghers type hamartomatous polyp in duodenum covered with gastric type foveolar epithelium. Considering the definition of hamartomatous polyp, which is, the abnormal overgrowth of the indigenous epithelial component, the histological feature of current case is noteworthy in a point that it shows proliferation of heterotopic component, rather than the indigenous component.
-
Citations
Citations to this article as recorded by- A Solitary Peutz-Jeghers Hamartomatous Polyp in the Gastric Body: A Case Report
Noelia Madera, Noemí Acevedo, Carmen González-Peralta, Rafael Castro, Vismelis Mezquita-Luna
Cureus.2024;[Epub] CrossRef
- A Solitary Peutz-Jeghers Hamartomatous Polyp in the Gastric Body: A Case Report
- Evolving pathologic concepts of serrated lesions of the colorectum
- Jung Ho Kim, Gyeong Hoon Kang
- J Pathol Transl Med. 2020;54(4):276-289. Published online June 26, 2020
- DOI: https://doi.org/10.4132/jptm.2020.04.15
- 20,029 View
- 868 Download
- 37 Web of Science
- 36 Crossref
-
 Abstract
Abstract
 PDF
PDF Supplementary Material
Supplementary Material - Here, we provide an up-to-date review of the histopathology and molecular pathology of serrated colorectal lesions. First, we introduce the updated contents of the 2019 World Health Organization classification for serrated lesions. The sessile serrated lesion (SSL) is a new diagnostic terminology that replaces sessile serrated adenoma and sessile serrated polyp. The diagnostic criteria for SSL were revised to require only one unequivocal distorted serrated crypt, which is sufficient for diagnosis. Unclassified serrated adenomas have been included as a new category of serrated lesions. Second, we review ongoing issues concerning the morphology of serrated lesions. Minor morphologic variants with distinct molecular features were recently defined, including serrated tubulovillous adenoma, mucin-rich variant of traditional serrated adenoma (TSA), and superficially serrated adenoma. In addition to intestinal dysplasia and serrated dysplasia, minimal deviation dysplasia and not otherwise specified dysplasia were newly suggested as dysplasia subtypes of SSLs. Third, we summarize the molecular features of serrated lesions. The critical determinant of CpG island methylation development in SSLs is patient age. Interestingly, there may be ethnic differences in BRAF/KRAS mutation frequencies in SSLs. The molecular pathogenesis of TSAs is divided into KRAS and BRAF mutation pathways. SSLs with MLH1 methylation can progress into favorable prognostic microsatellite instability-positive (MSI+)/CpG island methylator phenotype-positive (CIMP+) carcinomas, whereas MLH1-unmethylated SSLs and BRAF-mutated TSAs can be precursors of poor-prognostic MSI−/CIMP+ carcinomas. Finally, based on our recent data, we propose an algorithm for stratifying risk subgroups of non-dysplastic SSLs.
-
Citations
Citations to this article as recorded by- Predominant Serrated Molecular Signature in Postcolonoscopy Colorectal Cancer: A Systematic Review and Meta-Analysis
Jen-Hao Yeh, Sin-Hua Moi, Chia-Chi Chen, Chao-Wen Hsu, Wen-Shuo Yeh, Tzu-Ning Tseng, Chuan-Pin Lin, Yu-Peng Liu, Jaw-Yuan Wang
American Journal of Gastroenterology.2026; 121(1): 122. CrossRef - Re-evaluating post-polypectomy surveillance: The role of non-invasive modalities in colorectal cancer prevention
Ethna McFerran, Damian McKay, Maurice B. Loughrey, Mark Lawler, Stephen T. McSorley
Best Practice & Research Clinical Gastroenterology.2026; 80: 102092. CrossRef - Clinical and endoscopic characteristics of colorectal traditional serrated adenomas with dysplasia/adenocarcinoma in a Korean population
Ki-Hyun Kim, Eun Myung, Hyung Hoon Oh, Chan-Muk Im, Young-Eun Seo, Je-Seong Kim, Chae-June Lim, Ga-Ram You, Sung-Bum Cho, Wan-Sik Lee, Myung-Giun Noh, Kyung-Hwa Lee, Young-Eun Joo
World Journal of Gastrointestinal Oncology.2025;[Epub] CrossRef - MicroRNA: role in macrophage polarisation and colorectal cancer pathogenesis
Haihong Lin, Jun Zhou, Ying He, Yifan Zhu, Puwen Chen, Hongwei Yan, Junyun Huang, Ersheng Gong, Xiaoling Wang
Frontiers in Cell and Developmental Biology.2025;[Epub] CrossRef - Submucosal fibrosis in large colorectal serrated lesions in cases receiving endoscopic submucosal dissection
Erik Manriquez-Alegria, Naohisa Yoshida, Reo Kobayashi, Naoto Iwai, Ken Inoue, Osamu Dohi, Lucas Cardoso, Hideyuki Konishi
Therapeutic Advances in Gastroenterology.2025;[Epub] CrossRef - Navigating the Colorectal Cancer Maze: Unveiling Pathways To Diagnosis, Management, Pathophysiology and Prevention
Khalid Ali Mohammed Al Kamzari, Constantina Constantinou
Current Oncology Reports.2025; 27(10): 1115. CrossRef - Fosl1 is a transcriptional effector of BRAFV600E-driven intestinal tumorigenesis
Zakia Alam, Rebecca Nightingale, Analia Lesmana, Cheng Liu, Laura J. Jenkins, Mark F. Richardson, Lawrence Croft, Ian Y. Luk, Camilla M. Reehorst, Fiona Chionh, Natalia Vukelic, Faiza Basheer, Eugene Tulchinsky, Joshua Badshah, Troy Dumenil, Latifa Bakiri
iScience.2025; 28(11): 113875. CrossRef - Sessile Serrated Lesions in Inflammatory Bowel Disease: Hidden Players in Colitis-Associated Colorectal Cancer?
Roberto de Sire, Diletta De Deo, Miriana Mercurio, Gianluca Franchellucci, Giulio Calabrese, Livio Bonacci, Mauro Sollai Pinna, Cristina Bezzio, Alessandro Armuzzi, Cesare Hassan, Alessandro Repici, Fabiana Castiglione, Sandro Ardizzone, Roberta Maselli
Journal of Clinical Medicine.2025; 14(22): 8042. CrossRef - Histologic Reappraisal and Evaluation of MLH1 Protein Expression in Sessile Serrated Lesions of the Proximal Colon
Priscilla de Sene Portel Oliveira, Miriam Aparecida da Silva Trevisan, Rita Barbosa de Carvalho, Rita de Cássia Perina Martins, João José Fagundes, Claudio Saddy Rodrigues Coy, Ashwini Esnakula
Gastroenterology Research and Practice.2025;[Epub] CrossRef - Superiority of excellent over good bowel preparation for proximal serrated polyp detection in a fecal immunochemical test-based screening cohort
Stefano Fantasia, Stefano Kayali, Pablo Cortegoso Valdivia, Stefano Andreotti, Daniele Macchi, Giorgio Nervi, Nico Pagano, Luigi Laghi
Gastrointestinal Endoscopy.2025;[Epub] CrossRef - Impact of AI-aided colonoscopy in clinical practice: a prospective randomised controlled trial
Johanna Schöler, Marko Alavanja, Thomas de Lange, Shunsuke Yamamoto, Per Hedenström, Jonas Varkey
BMJ Open Gastroenterology.2024; 11(1): e001247. CrossRef - The histologic features, molecular features, detection and management of serrated polyps: a review
Jin-Dong Wang, Guo-Shuai Xu, Xin-Long Hu, Wen-Qiang Li, Nan Yao, Fu-Zhou Han, Yin Zhang, Jun Qu
Frontiers in Oncology.2024;[Epub] CrossRef - Serrated polyps <10 mm cannot reliably be characterized by i-Scan without magnification at routine colonoscopy
Sabrina G.G. TESTONI, Chiara NOTARISTEFANO, Giuliano F. BONURA, Maria NAPOLITANO, Dario ESPOSITO, Edi VIALE, Lorella FANTI, Francesco AZZOLINI, Giulia M. CAVESTRO, PierAlberto TESTONI
Minerva Gastroenterology.2024;[Epub] CrossRef - Interobserver variability in the histopathological classification and grading of dysplasia in elevated colon lesions in the city of Lima
Guido Gallegos-Serruto, Aldo Gutiérrez, César Chian García, Isthvan Torres Perez
Revista de Gastroenterología del Perú.2024; 44(3): 239. CrossRef - Comparison of adenoma detection rate and proximal serrated polyp detection rate and their effect on post-colonoscopy colorectal cancer mortality in screening patients
Jasmin Zessner-Spitzenberg, Elisabeth Waldmann, Lena Jiricka, Lisa-Maria Rockenbauer, Anna Hinterberger, Jeremy Cook, Arno Asaturi, Aleksandra Szymanska, Barbara Majcher, Michael Trauner, Monika Ferlitsch
Endoscopy.2023; 55(05): 434. CrossRef - The yield of dysplasia and serrated lesions in a single-centre tertiary inflammatory bowel disease cohort
Fiona Yeaman, Lena Thin
Therapeutic Advances in Gastroenterology.2023;[Epub] CrossRef -
The BEETS (JACCRO CC-18) Trial: An Observational and Translational Study of
BRAF
-Mutated Metastatic Colorectal Cancer
Chiaki Inagaki, Ryo Matoba, Satoshi Yuki, Manabu Shiozawa, Akihito Tsuji, Eisuke Inoue, Kei Muro, Wataru Ichikawa, Masashi Fujii, Yu Sunakawa
Future Oncology.2023; 19(17): 1165. CrossRef - A retrospective analysis of the histology of resected polyps and colonoscopy quality parameters in Belgium
E Macken, S Van Dongen, G Van Hal
Acta Gastro Enterologica Belgica.2023; 86(2): 277. CrossRef - Prognostic Biomarkers of Cell Proliferation in Colorectal Cancer (CRC): From Immunohistochemistry to Molecular Biology Techniques
Aldona Kasprzak
Cancers.2023; 15(18): 4570. CrossRef - Assimilating Epigenetics and Transcriptomics for the Identification
of Prognostic Novel Biomarkers and Imminent Targets in
Colorectal Carcinoma with Therapeutic Potential
Suman Kumar Ray, Sukhes Mukherjee
Current Molecular Medicine.2023; 23(8): 784. CrossRef - Multitarget Stool RNA Test for Colorectal Cancer Screening
Erica K. Barnell, Elizabeth M. Wurtzler, Julie La Rocca, Thomas Fitzgerald, Jessica Petrone, Yansheng Hao, Yiming Kang, Faith L. Holmes, David A. Lieberman
JAMA.2023; 330(18): 1760. CrossRef - Microbiome in Colonic Carcinogenesis
Jun Sun, Yinglin Xia
Comprehensive Physiology.2023; 13(3): 4685. CrossRef - Impact of comprehensive optical diagnosis training using Workgroup serrAted polypS and Polyposis classification on detection of adenoma and sessile serrated lesion
Jooyoung Lee, Jung Ho Bae, Su Jin Chung, Hae Yeon Kang, Seung Joo Kang, Min‐Sun Kwak, Ji Yeon Seo, Ji Hyun Song, Sun Young Yang, Jong In Yang, Seon Hee Lim, Jeong Yoon Yim, Joo Hyun Lim, Goh Eun Chung, Eun Hyo Jin, Ji Min Choi, Yoo Min Han, Joo Sung Kim
Digestive Endoscopy.2022; 34(1): 180. CrossRef - Clinicopathological and molecular analyses of hyperplastic lesions including microvesicular variant and goblet cell rich variant hyperplastic polyps and hyperplastic nodules—Hyperplastic nodule is an independent histological entity
Noriyuki Uesugi, Yoichi Ajioka, Tomio Arai, Yoshihito Tanaka, Tamotsu Sugai
Pathology International.2022; 72(2): 128. CrossRef - Comprehensive clinicopathologic, molecular, and immunologic characterization of colorectal carcinomas with loss of three intestinal markers, CDX2, SATB2, and KRT20
Ji Ae Lee, Mi-Kyoung Seo, Seung-Yeon Yoo, Nam-Yun Cho, Yoonjin Kwak, Kyoungbun Lee, Jung Ho Kim, Gyeong Hoon Kang
Virchows Archiv.2022; 480(3): 543. CrossRef - Serrated Colorectal Lesions: An Up-to-Date Review from Histological Pattern to Molecular Pathogenesis
Martino Mezzapesa, Giuseppe Losurdo, Francesca Celiberto, Salvatore Rizzi, Antonio d’Amati, Domenico Piscitelli, Enzo Ierardi, Alfredo Di Leo
International Journal of Molecular Sciences.2022; 23(8): 4461. CrossRef - Arterial stiffness is associated with high-risk colorectal adenomas and serrated lesions: A cross-sectional study in a Taiwanese population
Hung-Yu Chen, Wen-Huang Lee, Hung-Lung Hsu, Yu-Tsung Chou, Fei-Lin Su, I-Hsuan Wu, Ting-Hsing Chao
Journal of Cardiology.2022; 80(2): 139. CrossRef - Morphological and molecular characterization of colorectal sessile serrated lesions with dysplasia
Filippo Cappello, Valentina Angerilli, Luca Dal Santo, Giada Munari, Marianna Sabbadin, Marcello Lo Mele, Gianmaria Pennelli, Claudio Luchini, Paola Parente, Stefano Lazzi, Matteo Fassan
Pathology - Research and Practice.2022; 240: 154214. CrossRef - Serrated polyposis: an overview
Jonathan Fawkes
Gastrointestinal Nursing.2022; 20(9): 24. CrossRef - Sessile serrated lesion presenting as large pedunculated polyp in the rectum: A case report
Shin Ju Oh, Jung-Wook Kim, Chi Hyuk Oh
Medicine.2022; 101(51): e32287. CrossRef - WHICH LESIONS ARE AT HIGHER RISK OF DEVELOPING COLORECTAL CARCINOMAS: SUPERFICIALLY ELEVATED SERRATED LESIONS OR DEPRESSED LESIONS?
Artur Adolfo PARADA, Filadelfio Euclydes VENCO, Miguel Reynaldo VARCA-NETO, Roberto EL IBRAHIM, Paula Bechara POLETTI, Helcio Pedrosa BRITO, Heloisa de Fátima SARE, Osvaldo MALAFAIA
ABCD. Arquivos Brasileiros de Cirurgia Digestiva (São Paulo).2022;[Epub] CrossRef - WNT5a in Colorectal Cancer: Research Progress and Challenges
Guangshun Sun, Liangliang Wu, Guoqiang Sun, Xuesong Shi, Hongyong Cao, Weiwei Tang
Cancer Management and Research.2021; Volume 13: 2483. CrossRef - Endoscopic diagnosis for colorectal sessile serrated lesions
Toshihiro Nishizawa, Shuntaro Yoshida, Akira Toyoshima, Tomoharu Yamada, Yoshiki Sakaguchi, Taiga Irako, Hirotoshi Ebinuma, Takanori Kanai, Kazuhiko Koike, Osamu Toyoshima
World Journal of Gastroenterology.2021; 27(13): 1321. CrossRef - NTRK oncogenic fusions are exclusively associated with the serrated neoplasia pathway in the colorectum and begin to occur in sessile serrated lesions
Jung Ho Kim, Jeong Hoon Hong, Yoon‐La Choi, Ji Ae Lee, Mi‐kyoung Seo, Mi‐Sook Lee, Sung Bin An, Min Jung Sung, Nam‐Yun Cho, Sung‐Su Kim, Young Kee Shin, Sangwoo Kim, Gyeong Hoon Kang
The Journal of Pathology.2021; 255(4): 399. CrossRef - Differential pre-malignant programs and microenvironment chart distinct paths to malignancy in human colorectal polyps
Bob Chen, Cherie’ R. Scurrah, Eliot T. McKinley, Alan J. Simmons, Marisol A. Ramirez-Solano, Xiangzhu Zhu, Nicholas O. Markham, Cody N. Heiser, Paige N. Vega, Andrea Rolong, Hyeyon Kim, Quanhu Sheng, Julia L. Drewes, Yuan Zhou, Austin N. Southard-Smith, Y
Cell.2021; 184(26): 6262. CrossRef - Molecular Insights Into Colorectal Carcinoma
Domenika Ortiz Requena, Monica Garcia-Buitrago
Archives of Medical Research.2020; 51(8): 839. CrossRef
- Predominant Serrated Molecular Signature in Postcolonoscopy Colorectal Cancer: A Systematic Review and Meta-Analysis
- Colorectal epithelial neoplasm associated with gut-associated lymphoid tissue
- Yo Han Jeon, Ji Hyun Ahn, Hee Kyung Chang
- J Pathol Transl Med. 2020;54(2):135-145. Published online January 29, 2020
- DOI: https://doi.org/10.4132/jptm.2019.11.06
- 10,807 View
- 259 Download
- 3 Web of Science
- 3 Crossref
-
 Abstract
Abstract
 PDF
PDF - Background
Colorectal epithelial neoplasm extending into the submucosal gut-associated lymphoid tissue (GALT) can cause difficulties in the differential diagnosis. Regarding GALT-associated epithelial neoplasms, a few studies favor the term “GALT carcinoma” while other studies have mentioned the term “GALT-associated pseudoinvasion/epithelial misplacement (PEM)”.
Methods
The clinicopathologic characteristics of 11 cases of colorectal epithelial neoplasm associated with submucosal GALT diagnosed via endoscopic submucosal dissection were studied.
Results
Eight cases (72.7%) were in males. The median age was 59 years, and age ranged from 53 to 73. All cases had a submucosal tumor component more compatible with GALT-associated PEM. Eight cases (72.7%) were located in the right colon. Ten cases (90.9%) had a non-protruding endoscopic appearance. Nine cases (81.8%) showed continuity between the submucosal and surface adenomatous components. Nine cases showed (81.8%) focal defects or discontinuation of the muscularis mucosae adjacent to the submucosal GALT. No case showed hemosiderin deposits in the submucosa or desmoplastic reaction. No case showed single tumor cells or small clusters of tumor cells in the submucosal GALT. Seven cases (63.6%) showed goblet cells in the submucosa. No cases showed oncocytic columnar cells lining submucosal glands.
Conclusions
Our experience suggests that pathologists should be aware of the differential diagnosis of GALT-associated submucosal extension by colorectal adenomatous neoplasm. Further studies are needed to validate classification of GALT-associated epithelial neoplasms. -
Citations
Citations to this article as recorded by- Redefining GALT-associated carcinoma: a distinct subtype of colorectal adenocarcinoma
Jennifer Fallas, Marianna Arvanitaki, Sophie Lecomte, Jean-Yves Bonnet, Sarah De Clercq, Audrey Verrellen, Nicky D’Haene, María Gómez Galdón, Laurine Verset
Virchows Archiv.2026; 488(3): 695. CrossRef - Family adenomatous polyposis come across dome type adenocarcinoma: a case report and literature review
Ying-Ying Chang, Xiao-Long Zhang, Yao-Hui Wang, Ting-Sheng Ling
Diagnostic Pathology.2025;[Epub] CrossRef - Radiation-induced injury and the gut microbiota: insights from a microbial perspective
Qiaoli Wang, Guoqiang Xu, Ouying Yan, Shang Wang, Xin Wang
Therapeutic Advances in Gastroenterology.2025;[Epub] CrossRef
- Redefining GALT-associated carcinoma: a distinct subtype of colorectal adenocarcinoma
- Rectal Invasion by Prostatic Adenocarcinoma That Was Initially Diagnosed in a Rectal Polyp on Colonoscopy
- Ghilsuk Yoon, Man-Hoon Han, An Na Seo
- J Pathol Transl Med. 2019;53(4):266-269. Published online April 11, 2019
- DOI: https://doi.org/10.4132/jptm.2019.03.25
- 10,039 View
- 130 Download
- 9 Web of Science
- 9 Crossref
-
 Abstract
Abstract
 PDF
PDF - Despite anatomical proximity, prostatic adenocarcinoma with rectal invasion is extremely rare. We present a case of rectal invasion by prostatic adenocarcinoma that was initially diagnosed from a rectal polyp biopsied on colonoscopy in a 69-year-old Korean man. He presented with dull anal pain and voiding discomfort for several days. Computed tomography revealed either prostatic adenocarcinoma with rectal invasion or rectal adenocarcinoma with prostatic invasion. His tumor marker profile showed normal prostate specific antigen (PSA) level and significantly elevated carcinoembryonic antigen level. Colonoscopy was performed, and a specimen was obtained from a round, 1.5 cm, sessile polyp that was 1.5 cm above the anal verge. Microscopically, glandular tumor structures infiltrated into the rectal mucosa and submucosa. Immunohistochemically, the tumor cells showed alpha-methylacyl-CoA-racemase positivity, PSA positivity, and caudal-related homeobox 2 negativity. The final diagnosis of the rectal polyp was consistent with prostatic adenocarcinoma. Here, we present a rare case that could have been misdiagnosed as rectal adenocarcinoma.
-
Citations
Citations to this article as recorded by- An Extremely Rare Metastatic Prostate Tumor From Rectal Cancer With Characteristic MRI Findings Due to Necrosis
Sohei Iwagami, Shoji Oura, Haruka Miyai, Naoki Kataoka, Masaya Nishihata
Cureus.2025;[Epub] CrossRef - Prostate cancer invading rectal serosa and anal sphincter treated with definitive radiation therapy: Case report and review of the literature
Mi-Jo Lee
Journal of Cancer Research and Therapeutics.2024; 20(3): 1081. CrossRef - Metastatic Adenocarcinoma of the Prostate Masquerading as a Splenic Flexure Colonic Polyp: A Diagnostic Conundrum
Zakaria W Shkoukani, Alaa Chamsin, Mohamed I Abdulmajed
Cureus.2024;[Epub] CrossRef - An Interesting Case of Prostate Cancer Presenting With Colonic Metastasis
Shawn Keating, Ayesha Imtiaz, Kenneth Nahum, Ankita Prasad, Pramil Cheriyath
Cureus.2023;[Epub] CrossRef - Metastase d’un adenocarcinome prostatique au sein d’un polype colique. À propos d’un cas et revue de la littérature
Guillaume Abitbol, Clémence Barthomeuf, Olivier Varennes, Marine Clement, Sami Hakim, Denis Chatelain
Annales de Pathologie.2023; 43(4): 342. CrossRef - Isolated Rectal Metastases from Locally Advanced Carcinoma Prostate Detected by 18F-PSMA-1007 PET/CT
Shashank Shekhar Singh, Rani Kunti Randhir Singh, Narvesh Kumar, Harshvardhan Atrey
World Journal of Nuclear Medicine.2022; 21(03): 248. CrossRef - Rectal Invasion by Metastatic Prostate Adenocarcinoma
Anshu Wadehra, Samer Alkassis, Aliza Rizwan, Omid Yazdanpanah
Cureus.2021;[Epub] CrossRef - Metastatic Prostate Cancer Presenting as a Rectal Polyp: A Rare Occurrence
Ese Uwagbale, Ifeanyichukwu Onukogu, Vimal Bodiwala, Solomon Agbroko, Niket Sonpal
Cureus.2021;[Epub] CrossRef - Local Staging of Prostate Cancer with Multiparametric MRI
Nandan Keshav, Mark D. Ehrhart, Steven C. Eberhardt, Martha F. Terrazas
Seminars in Roentgenology.2021; 56(4): 366. CrossRef
- An Extremely Rare Metastatic Prostate Tumor From Rectal Cancer With Characteristic MRI Findings Due to Necrosis
- CpG Island Methylation in Sessile Serrated Adenoma/Polyp of the Colorectum: Implications for Differential Diagnosis of Molecularly High-Risk Lesions among Non-dysplastic Sessile Serrated Adenomas/Polyps
- Ji Ae Lee, Hye Eun Park, Seung-Yeon Yoo, Seorin Jeong, Nam-Yun Cho, Gyeong Hoon Kang, Jung Ho Kim
- J Pathol Transl Med. 2019;53(4):225-235. Published online March 19, 2019
- DOI: https://doi.org/10.4132/jptm.2019.03.12
- 10,431 View
- 247 Download
- 9 Web of Science
- 8 Crossref
-
 Abstract
Abstract
 PDF
PDF Supplementary Material
Supplementary Material - Background
Although colorectal sessile serrated adenomas/polyps (SSA/Ps) with morphologic dysplasia are regarded as definite high-risk premalignant lesions, no reliable grading or risk-stratifying system exists for non-dysplastic SSA/Ps. The accumulation of CpG island methylation is a molecular hallmark of progression of SSA/Ps. Thus, we decided to classify non-dysplastic SSA/Ps into risk subgroups based on the extent of CpG island methylation.
Methods
The CpG island methylator phenotype (CIMP) status of 132 non-dysplastic SSA/Ps was determined using eight CIMP-specific promoter markers. SSA/Ps with CIMP-high and/or MLH1 promoter methylation were regarded as a high-risk subgroup.
Results
Based on the CIMP analysis results, methylation frequency of each CIMP marker suggested a sequential pattern of CpG island methylation during progression of SSA/P, indicating MLH1 as a late-methylated marker. Among the 132 non-dysplastic SSA/Ps, 34 (26%) were determined to be high-risk lesions (33 CIMP-high and 8 MLH1-methylated cases; seven cases overlapped). All 34 high-risk SSA/Ps were located exclusively in the proximal colon (100%, p = .001) and were significantly associated with older age (≥ 50 years, 100%; p = .003) and a larger histologically measured lesion size (> 5 mm, 100%; p = .004). In addition, the high-risk SSA/Ps were characterized by a relatively higher number of typical base-dilated serrated crypts.
Conclusions
Both CIMP-high and MLH1 methylation are late-step molecular events during progression of SSA/Ps and rarely occur in SSA/Ps of young patients. Comprehensive consideration of age (≥ 50), location (proximal colon), and histologic size (> 5 mm) may be important for the prediction of high-risk lesions among non-dysplastic SSA/Ps. -
Citations
Citations to this article as recorded by- Distinguishing colorectal sessile serrated lesions from hyperplastic polyps: development of a prediction model based on logistic regression
Dian Zhang, Xiao Tan, Weiling Hu
BMC Gastroenterology.2026;[Epub] CrossRef - MLH1 Methylation Status and Microsatellite Instability in Patients with Colorectal Cancer
Manuel Alejandro Rico-Méndez, Miguel Angel Trujillo-Rojas, María de la Luz Ayala-Madrigal, Jesús Arturo Hernández-Sandoval, Anahí González-Mercado, Melva Gutiérrez-Angulo, José Geovanni Romero-Quintana, Jesús Alonso Valenzuela-Pérez, Ruth Ramírez-Ramírez,
Genes.2025; 16(2): 182. CrossRef - Histologic Reappraisal and Evaluation of MLH1 Protein Expression in Sessile Serrated Lesions of the Proximal Colon
Priscilla de Sene Portel Oliveira, Miriam Aparecida da Silva Trevisan, Rita Barbosa de Carvalho, Rita de Cássia Perina Martins, João José Fagundes, Claudio Saddy Rodrigues Coy, Ashwini Esnakula
Gastroenterology Research and Practice.2025;[Epub] CrossRef - Immune microenvironmental heterogeneity according to tumor DNA methylation phenotypes in microsatellite instability-high colorectal cancers
Jung Ho Kim, Jiyun Hong, Ji Ae Lee, Minsun Jung, Eunwoo Choi, Nam-Yun Cho, Gyeong Hoon Kang, Sangwoo Kim
Cancer Immunology, Immunotherapy.2024;[Epub] CrossRef - How to "pick up" colorectal serrated lesions and polyps in daily histopathology practice: From terminologies to diagnostic pitfalls
Thai H Tran, Vinh H Nguyen, Diem TN Vo
World Journal of Clinical Oncology.2024; 15(9): 1157. CrossRef - Serrated Colorectal Lesions: An Up-to-Date Review from Histological Pattern to Molecular Pathogenesis
Martino Mezzapesa, Giuseppe Losurdo, Francesca Celiberto, Salvatore Rizzi, Antonio d’Amati, Domenico Piscitelli, Enzo Ierardi, Alfredo Di Leo
International Journal of Molecular Sciences.2022; 23(8): 4461. CrossRef - NTRK oncogenic fusions are exclusively associated with the serrated neoplasia pathway in the colorectum and begin to occur in sessile serrated lesions
Jung Ho Kim, Jeong Hoon Hong, Yoon‐La Choi, Ji Ae Lee, Mi‐kyoung Seo, Mi‐Sook Lee, Sung Bin An, Min Jung Sung, Nam‐Yun Cho, Sung‐Su Kim, Young Kee Shin, Sangwoo Kim, Gyeong Hoon Kang
The Journal of Pathology.2021; 255(4): 399. CrossRef - Evolving pathologic concepts of serrated lesions of the colorectum
Jung Ho Kim, Gyeong Hoon Kang
Journal of Pathology and Translational Medicine.2020; 54(4): 276. CrossRef
- Distinguishing colorectal sessile serrated lesions from hyperplastic polyps: development of a prediction model based on logistic regression
- A Case of Giant Colonic Muco-submucosal Elongated Polyps Associated with Intussusception
- Joo Heon Kim, Seung Yun Lee, Je Ho Jang, Hyun Young Han, Dong Wook Kang
- J Pathol Transl Med. 2016;50(6):474-478. Published online May 23, 2016
- DOI: https://doi.org/10.4132/jptm.2016.04.27
- 12,270 View
- 134 Download
- 6 Web of Science
- 8 Crossref
-
 Abstract
Abstract
 PDF
PDF - Colonic muco-submucosal elongated polyp (CMSEP), a newly categorized non-neoplastic colorectal polyp, is a pedunculated and elongated polyp composed of normal mucosal and submucosal layers without any proper muscle layer. We herein report a giant variant of CMSEP associated with intussusception in the rectosigmoid colon, with a review of the literature. A 48-year-old woman underwent a laparoscopic low anterior resection due to multiple large submucosal polypoid masses associated with intussusception. Grossly, the colonic masses were multiple pedunculated polyps with a long stalk and branches ranging in size from a few millimeters to 14.0 cm in length. Microscopically, there was no evidence of hyperplasia, atypia, or active inflammation in the mucosa. The submucosal layers were composed of edematous and fibrotic stroma with fat tissue, dilated vessels, and lymphoid follicles.
-
Citations
Citations to this article as recorded by- Unusually rapid growth of a duodenal muco-submucosal elongated polyp: A case report
Yi Yang, Ding-Fu Zhong
World Journal of Gastrointestinal Surgery.2025;[Epub] CrossRef - Multiple enteric muco-submucosal elongated polyps causing intussusception
Atsuki Taniguchi, Izuru Endo, Takeyoshi Nishiyama, Nobuyuki Watanabe, Osamu Yoshida, Hiroaki Asano, Masatoshi Kubo, Tetsunobu Udaka
Clinical Journal of Gastroenterology.2024; 17(1): 41. CrossRef - Intussusception due to a Muco-submucosal Elongated Polyp in the Small Intestine—A Case Report—
Hiroki ISHIGE, Ken IMAIZUMI, Takumu FUKASAWA, Keiichiro ITO, Hiroyuki KASAJIMA, Satoru MUNAKATA, Norihiko SHIMOYAMA, Kazuaki NAKANISHI
Nihon Rinsho Geka Gakkai Zasshi (Journal of Japan Surgical Association).2024; 85(6): 744. CrossRef - Jejunal Intussusception Caused by Enteric Muco-submucosal Elongated Polyp: A Case Report
Young Min Jo
Soonchunhyang Medical Science.2024; 30(2): 60. CrossRef - Jejunal intussusception and perforation due to enteric muco-submucosal elongated polyp: a case report and literature review
Ryosuke Kikuchi, Shigenobu Emoto, Hiroaki Nozawa, Kazuhito Sasaki, Koji Murono, Shinya Abe, Hirofumi Sonoda, Aya Shinozaki-Ushiku, Soichiro Ishihara
Surgical Case Reports.2023;[Epub] CrossRef - A stalk with no polyp—A muco‐submucosal elongated polyp in the duodenum
Neil O’Morain, Ciaran McCloskey, Sinead Flanagan, Glen Doherty
United European Gastroenterology Journal.2023; 11(4): 392. CrossRef - Duodenal Worm-Like Polyp
Pan Pan, Guoshan Zhang, Xiao Cui, Liang Liu
Digestive Diseases and Sciences.2023; 68(12): 4275. CrossRef - Colonic Mucosubmucosal Elongated Polyp in the Sigmoid Colon on Surveillance Colonoscopy
Xiaowen Fan, Melissa Hershman, Gabriel Levi, Ilan Weisberg
ACG Case Reports Journal.2019; 6(6): e00110. CrossRef
- Unusually rapid growth of a duodenal muco-submucosal elongated polyp: A case report
- Investigation of the Roles of Cyclooxygenase-2 and Galectin-3 Expression in the Pathogenesis of Premenopausal Endometrial Polyps
- Esin Kasap, Serap Karaarslan, Esra Bahar Gur, Mine Genc, Nur Sahin, Serkan Güclü
- J Pathol Transl Med. 2016;50(3):225-230. Published online April 16, 2016
- DOI: https://doi.org/10.4132/jptm.2016.03.08
- 9,766 View
- 84 Download
- 4 Web of Science
- 5 Crossref
-
 Abstract
Abstract
 PDF
PDF - Background
The pathogenesis and etiology of endometrial polyps has not been elucidated. In this study, we aimed to examine the pathogenic mechanisms of endometrial polyp development using immunohistochemistry. We evaluated the expression of galectin-3 and cyclooxgenase-2 (COX-2) during the menstrual cycle in premenopausal women with endometrial polyps or normal endometrium.
Methods
Thirty-one patients with endometrial polyps and 50 healthy control patients were included in this study. The levels of expression of COX-2 and galectin-3 were studied by immunohistochemistry.
Results
The percentage of COX-2–positive cells and the intensity of COX-2 staining in the endometrium did not vary during the menstrual cycle either in the control group or in patients with endometrial polyps. However, expression of galectin-3 was significantly lower in endometrial polyps and during the proliferative phase of the endometrium compared with the secretory phase.
Conclusions
Our data suggests that the pathogenesis of endometrial polyps does not involve expression of COX-2 or galectin-3. -
Citations
Citations to this article as recorded by- Abnormal expression of Hippo–YAP1 signalling pathway and progesterone resistance mechanism in endometrial polyps
Xinyu Yu, Weijia Kong, Kaiyue Shang, Hongxin Xing, Wenjing Sun, Qianqian Li, Hui Zhang
Journal of Obstetrics and Gynaecology.2025;[Epub] CrossRef - Research Progress in the Treatment of Endometrial Polyps
秀芬 蔡
Advances in Clinical Medicine.2024; 14(01): 1772. CrossRef - ER and COX2 expression in endometrial hyperplasia processes
Nataliia Tsyndrenko, Mykola Lyndіn, Kateryna Sikora, Andrew Awuah Wireko, Toufik Abdul-Rahman, Nataliia Hyriavenko, Anatolii Romaniuk
Medicine.2023; 102(33): e34864. CrossRef - Novel microarchitecture of human endometrial glands: implications in endometrial regeneration and pathologies
Nicola Tempest, Christopher J Hill, Alison Maclean, Kathleen Marston, Simon G Powell, Hannan Al-Lamee, Dharani K Hapangama
Human Reproduction Update.2022; 28(2): 153. CrossRef - Variances in the Level of COX-2 and iNOS in Different Grades of Endometrial Cancer
Marcin Oplawski, Konrad Dziobek, Nikola Zmarzły, Beniamin O. Grabarek, Robert Kiełbasiński, Przemysław Kieszkowski, Piotr Januszyk, Karol Talkowski, Michał Schweizer, Piotr Kras, Andrzej Plewka, Dariusz Boroń
Current Pharmaceutical Biotechnology.2020; 21(1): 52. CrossRef
- Abnormal expression of Hippo–YAP1 signalling pathway and progesterone resistance mechanism in endometrial polyps
- Ménétrier’s Disease: Its Mimickers and Pathogenesis
- Won Jae Huh, Robert J. Coffey, Mary Kay Washington
- J Pathol Transl Med. 2016;50(1):10-16. Published online December 18, 2015
- DOI: https://doi.org/10.4132/jptm.2015.09.15
- 28,482 View
- 397 Download
- 34 Web of Science
- 35 Crossref
-
 Abstract
Abstract
 PDF
PDF - Ménétrier’s disease is a rare protein-losing hypertrophic gastropathy. Histologically, it can be mistaken for other disorders showing hypertrophic gastropathy. The pathogenesis of Ménétrier’s disease is not fully understood; however, it appears that the epidermal growth factor receptor (EGFR) ligand, transforming growth factor alpha, contributes to the pathogenesis of this disorder. In this review, we will discuss disease entities that can mimic Ménétrier’s disease and the role of EGFR signaling in Ménétrier’s disease.
-
Citations
Citations to this article as recorded by- Transcriptomic profiling reveals the role of Hedgehog signaling as a biomarker and in the pathogenesis of Ménétrier's disease
Miyoung Shin, Tryston T Gabriel, Fred Kwame Ofosu, Jiali Zhang, Tricia T Wang, Matthew E Bechard, Robert J Coffey, Won Jae Huh
The Journal of Pathology.2026; 268(1): 65. CrossRef - Ménétrier’s disease: a narrative review of molecular pathogenesis, clinical spectrum and evolving therapeutic strategies
Tai Zhang, Ting Chen, Xudong Tang
QJM: An International Journal of Medicine.2026; 119(2): 85. CrossRef - Cobblestone-like Gastric Mucosal Changes on Endoscopy in Dogs with a History of Prolonged Proton Pump Inhibitor Therapy
Martine Dominique Didier, Laura Zagnoli, Deborah Cattaneo, Silvia Lucia Benali, Enrico Bottero
Animals.2026; 16(3): 406. CrossRef - Clinicopathological Features of Biopsy-Proven Foveolar Hyperplasia in the Absence of Hypertrophic Gastropathy
Omer Kucukdemırcı, Merve Soylu, Ufuk Avcıoğlu, Ahmet Bektaş
Uludağ Üniversitesi Tıp Fakültesi Dergisi.2026; 52: 1885379. CrossRef - Co-existence of Ménétrier’s Disease and Collagenous Colitis: A Diagnostic and Therapeutic Challenge
Shigeto Mashiko, Daiki Murata, Ryohei Takahashi, Daisuke Fukushi, Takahiro Ohara, Katsutoshi Furukawa
Journal of General Internal Medicine.2026;[Epub] CrossRef - Ménétrier-like disease in a dog without glandular atrophy: expanding the morphologic spectrum
Mario Pultrone, Dyana Erba, Margherita Orlandi, Michela Pugliese
BMC Veterinary Research.2026;[Epub] CrossRef - Ménétrier's disease presenting with melena and severe anemia in the absence of hypoalbuminemia: A case report
Bipneet Singh, Jahnavi Ethakota, Palak Grover, Gurleen Kaur, Sruthi Ramanan
Medical Reports.2025; 13: 100290. CrossRef - Ménétrier disease: A clinical review
Ana R Barros, Sara Monteiro, Jorge Silva
World Journal of Gastroenterology.2025;[Epub] CrossRef - Robotic-assisted total gastrectomy for refractory hypoproteinemia in Menetrier’s disease: a case report with operative video and literature review
Lang Wang, Ziping Liu, Jing Zhang, Xianglin Zhu, Shijun Zhao, Cheng Zhao, Hao Liang, Jie Zhang, Tian Gao, Yinlu Ding
Frontiers in Medicine.2025;[Epub] CrossRef - RAB25 modulates pit cell commitment by coordinating transforming growth factor-alpha secretion from gastric epithelial cells
Haengdueng Jeong, Yura Lee, Chanyang Uhm, Seok Young Hwang, Sumin Hur, Minsoo Noh, Robert J. Coffey, James R. Goldenring, Ki Taek Nam
Cell Death & Disease.2025;[Epub] CrossRef - Dynamic tuft cell expansion during gastric metaplasia and dysplasia
Bogun Jang, Hyesung Kim, Su‐Hyung Lee, Yoonkyung Won, Izumi Kaji, Robert J Coffey, Eunyoung Choi, James R Goldenring
The Journal of Pathology: Clinical Research.2024;[Epub] CrossRef - Update S2k-Guideline Helicobacter pylori and gastroduodenal ulcer disease of the German Society of Gastroenterology, Digestive and Metabolic Diseases (DGVS)
Wolfgang Fischbach, Jan Bornschein, Jörg C. Hoffmann, Sibylle Koletzko, Alexander Link, Lukas Macke, Peter Malfertheiner, Kerstin Schütte, Dieter-Michael Selgrad, Sebastian Suerbaum, Christian Schulz
Zeitschrift für Gastroenterologie.2024; 62(02): 261. CrossRef - Enfermedad de Menetrier y su asociación a pólipos gástricos hiperplásicos
Romario Ruiz, Luis Huaman, Jacqueline Abad, Irene Cárdenas
Revista de Gastroenterología del Perú.2024; 44(1): 79. CrossRef - Role of gastric ultrasound in pediatric Menetrier's disease: Report of two cases
Francesco Pellegrino, Anna Opramolla, Antonio Pizzol, Caterina Rigazio, Laura Giugliano, Michele Pinon, Pier Luigi Calvo
JPGN Reports.2024; 5(4): 508. CrossRef - Beyond Regulation of Acid Secretion: A Novel Role for Histamine in Gastric Macrophage Differentiation and Function
Diane Bimczok
Cellular and Molecular Gastroenterology and Hepatology.2023; 15(1): 277. CrossRef - Upper gastrointestinal bleeding as an unusual manifestation of localized Ménétrier’s disease with an underlying lipoma: A case report
Michal Kmiecik, Aleksandra Walczak, Pawel Samborski, Jacek Paszkowski, Agnieszka Dobrowolska, Jacek Karczewski, Ewelina Swora-Cwynar
World Journal of Gastrointestinal Endoscopy.2023; 15(1): 10. CrossRef - Chronic Ménétrier disease leading to gastric cancer in youth
Bruno Salomão Hirsch, Silvia R. Cardoso, Elisa R. Baba, Diogo T. H. de Moura, Manoel Ernesto P. Gonçalves, Rodrigo S. de P. Rocha, Eduardo G. H. de Moura
Clinical Endoscopy.2023; 56(1): 125. CrossRef - Aktualisierte S2k-Leitlinie Helicobacter
pylori und gastroduodenale Ulkuskrankheit der Deutschen Gesellschaft für Gastroenterologie, Verdauungs- und Stoffwechselkrankheiten (DGVS) – Juli 2022 – AWMF-Registernummer: 021–001
Zeitschrift für Gastroenterologie.2023; 61(05): 544. CrossRef - Clinical Manifestation of Cytomegalovirus-Associated Protein-Losing Enteropathy in Children
Claire Ferrua, Anais Lemoine, Alexis Mosca, Anne-Aurélie Lopes
Nutrients.2023; 15(13): 2844. CrossRef - Guidelines for diagnosis and treatment of chronic gastritis inChina(2022,Shanghai)
Journal of Digestive Diseases.2023; 24(3): 150. CrossRef - A rare case of Menetrier's disease‐induced gastric outlet obstruction
Yi‐An Chen, Hsiang‐Lin Tsai, Peir‐In Liang, Jaw‐Yuan Wang
The Kaohsiung Journal of Medical Sciences.2023; 39(10): 1054. CrossRef - Biliary gastritis. Pathomorphological features and differential diagnosis
R. A. Nasyrov, Yu. A. Fominykh, E. Yu. Kalinina, O. A. Kizimova, D. P. Kovtun, E. V. Shcherbakova, O. S. Minina, A. P. Zavitaeva
Experimental and Clinical Gastroenterology.2023; (8): 134. CrossRef - Case report: Fundic gland polyps caused by long-term omeprazole use in a Maltese dog
Haemin Lee, Sanggu Kim, Dohee Lee, Yeon Chae, Taesik Yun, Mhan-Pyo Yang, Byeong-Teck Kang, Soochong Kim, Hakhyun Kim
Frontiers in Veterinary Science.2023;[Epub] CrossRef - Menetrier disease and Cytomegalovirus infection in paediatric age: report of three cases and a review of the literature
Federica Barbati, Edoardo Marrani, Giuseppe Indolfi, Paolo Lionetti, Sandra Trapani
European Journal of Pediatrics.2021; 180(3): 679. CrossRef - Ménétrier-like disease in a Pointer with concurrent granulomatous gastritis, helicobacteriosis and leishmaniosis: a case report
E. Lagerstedt, T. Spillmann, N. Airas, L. Solano-Gallego, S. Kilpinen
BMC Veterinary Research.2021;[Epub] CrossRef - EGF and BMPs Govern Differentiation and Patterning in Human Gastric Glands
Sarah Wölffling, Alice Anna Daddi, Aki Imai-Matsushima, Kristin Fritsche, Christian Goosmann, Jan Traulsen, Richard Lisle, Monika Schmid, Maria del Mar Reines-Benassar, Lennart Pfannkuch, Volker Brinkmann, Jan Bornschein, Peter Malfertheiner, Jürgen Ordem
Gastroenterology.2021; 161(2): 623. CrossRef - Diagnosis and Management of Ménétrier Disease in Children: A Case Series Review
Jasmina Krikilion, Elvira Ingrid Levy, Yvan Vandenplas
Pediatric Gastroenterology, Hepatology & Nutrition.2021; 24(1): 109. CrossRef - Gastric Mucosal Hypertrophy Masquerading as Metastasis From Neuroendocrine Tumor of the Pancreas Detected on 68Ga-DOTANOC PET/CT
Dharmender Malik, Ramkumar Elumalai, Ritu Verma, Ethel Shangne Belho, Nikhil Seniaray, Harsh Mahajan
Clinical Nuclear Medicine.2020; 45(1): 74. CrossRef - Unusual case of adult familial Menetrier disease in siblings
Emily Brownson, Adrian J Stanley, Prakash Konanahalli, John P Seenan
BMJ Case Reports.2019; 12(10): e231175. CrossRef - Enfermedad de Ménétrier, reporte de caso (con video)
Martín Alonso Gómez Zuleta, Oscar Fernando Ruíz Morales, Javier Riveros
Revista Colombiana de Gastroenterología.2019; 34(2): 190. CrossRef - Atrophic gastritis and chronic diarrhea due to Helicobacter pylori infection in early infancy
Toshihiko Kakiuchi, Aiko Nakayama, Ryo Shimoda, Muneaki Matsuo
Medicine.2019; 98(47): e17986. CrossRef - The causes of upper gastrointestinal bleeding and urgent gastroscopy at the Center for Emergency Medicine of the Clinical Center Kragujevac
Biljana Milojković-Kicevska, Zoran Kovačević, Mirjana Janićijević-Petrović, Katarina Janićijević
Timocki medicinski glasnik.2019; 44(4): 151. CrossRef - Enfermedad de Ménétrier como causa de síndrome edematoso en pediatría
Magalí Squitín Tasende, María Daniela Frangi, Carolina Silva
Revista del Hospital Italiano de Buenos Aires.2018; 38(1): 34. CrossRef - Wnt/β-catenin promotes gastric fundus specification in mice and humans
Kyle W. McCracken, Eitaro Aihara, Baptiste Martin, Calyn M. Crawford, Taylor Broda, Julie Treguier, Xinghao Zhang, John M. Shannon, Marshall H. Montrose, James M. Wells
Nature.2017; 541(7636): 182. CrossRef - A SMAD4 mutation indicative of juvenile polyposis syndrome in a family previously diagnosed with Menetrier's disease
James K. Burmester, Lauren N. Bell, Deanna Cross, Patrick Meyer, Steven H. Yale
Digestive and Liver Disease.2016; 48(10): 1255. CrossRef
- Transcriptomic profiling reveals the role of Hedgehog signaling as a biomarker and in the pathogenesis of Ménétrier's disease
- Follicular Dendritic Cell Sarcoma of the Inflammatory Pseudotumor-like Variant Presenting as a Colonic Polyp
- Shien-Tung Pan, Chih-Yuan Cheng, Nie-Sue Lee, Peir-In Liang, Shih-Sung Chuang
- Korean J Pathol. 2014;48(2):140-145. Published online April 28, 2014
- DOI: https://doi.org/10.4132/KoreanJPathol.2014.48.2.140
- 12,443 View
- 108 Download
- 35 Crossref
-
 Abstract
Abstract
 PDF
PDF Follicular dendritic cell (FDC) sarcoma is rare and is classified either as conventional type or inflammatory pseudotumor (IPT)-like variant. Extranodal presentation is uncommon and nearly all gastrointestinal FDC tumors are of the conventional type. IPT-like variant tumors occur almost exclusively in the liver and spleen and are consistently associated with Epstein-Barr virus (EBV). Here we report the case of a 78-year-old woman with an IPT-like FDC sarcoma presenting as a pedunculated colonic polyp. Histologically, scanty atypical ovoid to spindle cells were mixed with a background of florid lymphoplasmacytic infiltrate, which led to an initial misdiagnosis of pseudolymphoma. These atypical cells expressed CD21, CD23, CD35, and D2-40, and were positive for EBV by
in situ hybridization, confirming the diagnosis. The patient was free of disease five months after polypectomy without adjuvant therapy. Although extremely rare, the differential diagnosis for colonic polyp should include FDC sarcoma to avoid an erroneous diagnosis. A review of the 24 cases of IPT-like FDC sarcoma reported in the literature reveal that this tumor occurs predominantly in females with a predilection for liver and spleen, and has a strong association with EBV.-
Citations
Citations to this article as recorded by- Distinct subgroups of follicular dendritic cell sarcoma: insights from clinical, histologic and immunophenotypic characterization
Hsin-Ni Li, Ren Ching Wang, Chuan-Han Chen, Jun-Peng Chen, Chiann-Yi Hsu, Chang-Tsu Yuan, Yi-Hsiang Liu, Chiu-Hsuan Cheng, Yung-Fang Chia, Yun-Chih Jung, Shih-Sung Chuang
Virchows Archiv.2026;[Epub] CrossRef - The fifth edition of the WHO classification of mature T cell, NK cell and stroma-derived neoplasms
Ayoma D Attygalle, Kennosuke Karube, Yoon Kyung Jeon, Wah Cheuk, Govind Bhagat, John K C Chan, Kikkeri N Naresh
Journal of Clinical Pathology.2025; 78(4): 217. CrossRef - Genomic and Transcriptomic Landscape of Epstein-Barr Virus-Positive Inflammatory Follicular Dendritic Cell Sarcoma: A Multicenter Study
Yan Li, Ze-Lin Weng, Han-Xiao Fei, Hai-Feng Li, Yi-Na Liu, Le-Le Zhang, Qiong Zhang, Xin Weng, Yuan-Yuan Wang, Wen-Yong Huang, Zhi-Xing Cao, Kai-Yan Yang, Xi-Liang Chen, Jie Gao, Wen-Sheng Yang, Fang Liu, Juan-Juan Yong, Jing-Ping Yun, Hua Zhang, Yu-Hua H
Modern Pathology.2025; 38(10): 100864. CrossRef - What is new in the 5th edition of the World Health Organization classification of mature B and T/NK cell tumors and stromal neoplasms?
Ayoma D. Attygalle, John K. C. Chan, Sarah E. Coupland, Ming-Qing Du, Judith A. Ferry, Daphne de Jong, Dita Gratzinger, Megan S. Lim, Alina Nicolae, German Ott, Andreas Rosenwald, Anna Schuh, Reiner Siebert
Journal of Hematopathology.2024; 17(2): 71. CrossRef - Pathologic characteristics of histiocytic and dendritic cell neoplasms
Sun Och Yoon
Blood Research.2024;[Epub] CrossRef - Epstein-barr virus (EBV)-positive inflammatory pseudotumor-like follicular dendritic cell sarcoma (IPT-like FDCS) presenting as thrombocytopenia: A case report and literature review
Jiawei Jin, Xiaolong Zhu, Yi Wan, Yang Shi
Heliyon.2024; 10(12): e32997. CrossRef - EBV-positive inflammatory follicular dendritic cell sarcoma of the colon with clonal immunoglobulin gene rearrangement: A case report and literature review
Xia Xu, Xiuzhen Li, Qun Deng, Kaihang Yu, Jinfan Li
Heliyon.2024; 10(11): e31947. CrossRef - Challenges in the Diagnosis of Epstein-Barr Virus-positive Inflammatory Follicular Dendritic Cell Sarcoma
Yan Li, Xia Yang, Lili Tao, Weimei Zeng, Min Zuo, Shuo Li, Liyan Wu, Yanshong Lin, Ziying Zhang, Jingping Yun, Yuhua Huang
American Journal of Surgical Pathology.2023; 47(4): 476. CrossRef - Epstein-Barr Virus-Positive Inflammatory Follicular Dendritic Cell Sarcoma Presenting as a Colonic Polyp: Report of a Case with a Literature Review
Jiahui Hu, Dongdong Huang, Chengfu Xu, Yi Chen, Han Ma, Zhe Shen
Medicina.2023; 59(7): 1341. CrossRef - A Clinicopathology Review and Update of Epstein–Barr Virus-Associated Mesenchymal Tumors
Oswald Zhao Jian Lee, Noorjehan Omar, Joshua K. Tay, Victor Kwan Min Lee
Cancers.2023; 15(23): 5563. CrossRef - Granulomatous splenic mass with necrosis revealing an EBV-positive inflammatory follicular dendritic cell sarcoma
Irena Antonia Ungureanu, Renato Micelli Lupinacci, Marie Parrens, Jean-François Emile
Journal of Surgical Case Reports.2022;[Epub] CrossRef - Case report: Hepatic inflammatory pseudotumor-like follicular dendritic cell sarcoma: A rare case and minireview of the literature
Fan Ding, Chao Wang, Chi Xu, Hui Tang
Frontiers in Medicine.2022;[Epub] CrossRef - Follicular dendritic cell sarcoma of gastrointestinal tract with two emerging distinct subtypes: a case report and systemic review
Hongxing Gui, Jigisha Chaudhari, Rifat Mannan
Diagnostic Pathology.2022;[Epub] CrossRef - Surgical treatment of liver inflammatory pseudotumor-like follicular dendritic cell sarcoma: A case report
Li-Yue Fu, Jiu-Liang Jiang, Meng Liu, Jun-Jun Li, Kai-Ping Liu, Hai-Tao Zhu
World Journal of Gastrointestinal Oncology.2022; 14(11): 2288. CrossRef - Inflammatory pseudotumor-like follicular/fibroblastic dendritic cell sarcoma: focus on immunohistochemical profile and association with Epstein-Barr virus
Francesca Pagliuca, Andrea Ronchi, Annamaria Auricchio, Eva Lieto, Renato Franco
Infectious Agents and Cancer.2022;[Epub] CrossRef - Recent Advances in Digestive Tract Tumors: Updates From the 5th Edition of the World Health Organization “Blue Book”
Raul S. Gonzalez, Anwar Raza, Robert Propst, Oyedele Adeyi, Justin Bateman, Sabrina C. Sopha, Janet Shaw, Aaron Auerbach
Archives of Pathology & Laboratory Medicine.2021; 145(5): 607. CrossRef - Hepatic inflammatory pseudotumor-like follicular dendritic cell tumor: a case report
Ana Daniela Pascariu, Andreea Ioana Neagu, Andrei Valentin Neagu, Alexandru Băjenaru, Cezar Iulian Bețianu
Journal of Medical Case Reports.2021;[Epub] CrossRef - Inflammatory pseudotumor-like follicular dendritic cell sarcoma: Literature review of 67 cases
Hao Wu, Peng Liu, Xiao-Ran Xie, Jing-Shu Chi, Huan Li, Can-Xia Xu
World Journal of Meta-Analysis.2021; 9(1): 1. CrossRef - New Clinicopathologic Scenarios of EBV+ Inflammatory Follicular Dendritic Cell Sarcoma
Xiang-Nan Jiang, Yan Zhang, Tian Xue, Jie-Yu Chen, Alex C.L. Chan, Wah Cheuk, John K.C. Chan, Xiao-Qiu Li
American Journal of Surgical Pathology.2021; 45(6): 765. CrossRef - Select Epstein-Barr Virus–Associated Digestive Tract Lesions for the Practicing Pathologist
Zainab I. Alruwaii, Elizabeth A. Montgomery
Archives of Pathology & Laboratory Medicine.2021; 145(5): 562. CrossRef - Overview of Gastrointestinal Lymphoproliferative disorders✰
Aaron Auerbach, Nadine S. Aguilera
Seminars in Diagnostic Pathology.2021; 38(4): 1. CrossRef - Follicular dendritic cell sarcoma
Fabio Facchetti, Matteo Simbeni, Luisa Lorenzi
Pathologica.2021; 113(5): 316. CrossRef - Hepatic inflammatory pseudotumor-like follicular dendritic cell tumor with hepatic lymphoma history
Jiang Li, Hai-su Tao, Dong Chen, Zhi-yong Huang, Er-lei Zhang
Medicine.2021; 100(39): e27392. CrossRef - Clinicopathological characteristics of extranodal follicular dendritic cell sarcoma: A report of two cases
Xing Zhao, Dayong Sun, Gang Zhang
Oncology Letters.2021;[Epub] CrossRef - Inflammatory pseudotumour-like follicular dendritic cell tumour of the colon with plasmacytosis mimicking EBV-positive lymphoproliferative disorder
Ying-Ren Chen, Chi-Lin Lee, Yen-Chien Lee, Kung-Chao Chang
Pathology.2020; 52(4): 484. CrossRef - Beware the inflammatory cell-rich colonic polyp: a rare case of EBV-positive inflammatory pseudotumour-like follicular dendritic cell sarcoma with increased IgG4-positive plasma cells
Lynne Goh, Nan Zun Teo, Lai Mun Wang
Pathology.2020; 52(6): 713. CrossRef - Epstein–Barr virus‐positive inflammatory follicular dendritic cell sarcoma presenting as a solitary colonic mass: two rare cases and a literature review
Xiaokang Ke, Huihua He, Qingping Zhang, Jingping Yuan, Qilin Ao
Histopathology.2020; 77(5): 832. CrossRef - Inflammatory pseudotumor-like follicular dendritic cell sarcoma: A brief report of two cases
Bi-Xi Zhang, Zhi-Hong Chen, Yu Liu, Yuan-Jun Zeng, Yan-Chun Li
World Journal of Gastrointestinal Oncology.2019; 11(12): 1231. CrossRef - Epstein-Barr virus (EBV)–associated lymphoid proliferations, a 2018 update
Sherif A. Rezk, Xiaohui Zhao, Lawrence M. Weiss
Human Pathology.2018; 79: 18. CrossRef - A Rare Case of Epstein-Barr Virus Negative Inflammatory Pseudotumor-like Follicular Dendritic Cell Sarcoma Presenting as a Solitary Colonic Mass in a 53-Year-Old Woman; Case Report and Review of Literature
Rossana Kazemimood, Farid Saei Hamedani, Asma Sharif, Sujata Gaitonde, Elizabeth Wiley, Pier Cristoforo Giulianotti, John Vincent Groth
Applied Immunohistochemistry & Molecular Morphology.2017; 25(5): e30. CrossRef - A Case of Inflammatory Pseudotumor-like Follicular Dendritic Cell Sarcoma of the Lymph Node in the Small Bowel Mesentery Accompanied by Myasthenia Gravis
Daichi KITAGUCHI, Katsuji HISAKURA, Taiki SATO, Masanao KURATA, Tatsuya ODA, Nobuhiro OHKOHCHI
Nihon Rinsho Geka Gakkai Zasshi (Journal of Japan Surgical Association).2017; 78(3): 527. CrossRef - Clinicopathological features of inflammatory pseudotumour‐like follicular dendritic cell tumour of the abdomen
Yanyang Chen, Huijuan Shi, Hui Li, Tiantian Zhen, Anjia Han
Histopathology.2016; 68(6): 858. CrossRef - A Rare Case of Follicular Dendritic Cell Sarcoma with Pseudochylous Effusion and Review of Literature From India
Kamal Kant Sahu, Gaurav Prakash, Sandeep Rao, Amanjit Bal, Pankaj Malhotra, Jasmina Ahluwalia, Rakesh K. Vashistha
Indian Journal of Hematology and Blood Transfusion.2015; 31(2): 307. CrossRef - Epstein-Barr virus–associated inflammatory pseudotumor presenting as a colonic mass
Shunyou Gong, Iwona Auer, Rajan Duggal, Stefania Pittaluga, Mark Raffeld, Elaine S. Jaffe
Human Pathology.2015; 46(12): 1956. CrossRef - Response of follicular dendritic cell sarcoma to gemcitabine and docetaxel: report of two cases and literature review
Robert M Conry
Clinical Sarcoma Research.2014;[Epub] CrossRef
- Distinct subgroups of follicular dendritic cell sarcoma: insights from clinical, histologic and immunophenotypic characterization
- Cellular Pseudosarcomatous Fibroepithelial Stromal Polyp of the Vagina during Pregnancy: A Lesion That Is Overdiagnosed as a Malignant Tumor
- Joon Seon Song, Dong Eun Song, Kyu-Rae Kim, Jae Y. Ro
- Korean J Pathol. 2012;46(5):494-498. Published online October 25, 2012
- DOI: https://doi.org/10.4132/KoreanJPathol.2012.46.5.494
- 13,103 View
- 115 Download
- 12 Crossref
-
 Abstract
Abstract
 PDF
PDF Fibroepithelial stromal polyp (FSP) is a benign lesion that can occur at various sites, including the lower female genital tract. In rare cases, however, it may exhibit hypercellularity, bizarre cytomorphological features, and atypical mitoses resulting in an overdiagnosis as a malignant tumor despite its benign clinical course. Recently, we experienced one case of a 30-year-old pregnant woman with cellular pseudosarcomatous FSP that was initially diagnosed as a malignant fibrous histiocytoma at a primary clinic. In addition to describing the rare features of this case, we wish to increase awareness about this benign lesion which will be essential for avoiding unnecessary radical surgery or chemoradiation treatment.
-
Citations
Citations to this article as recorded by- A Giant Fibroepithelial Polyp of the Nipple: A Case Report of a Rare Benign Tumor
Saif S Al Sahee, Jaideep Rait, Layloma Hamidi-Latifi
Cureus.2026;[Epub] CrossRef - Giant hypopharyngeal fibroepithelial polyp: A case report and literature review
Muhammad Nour Alabdullah, Nagham Halaweek, Yasser Al Ghabra, Mohammad Hamdi, Mhd Ayham Abo Trab, Faysal Hajjar
Ear, Nose & Throat Journal.2025;[Epub] CrossRef - A Rare Case of A Pseudosarcomatous Stromal Tumor: A Unique Benign Ampullary Tumor
Jacob C. Jones, Penelope I. Quiles, John G. Quiles, Jerome G. Edelson
ACG Case Reports Journal.2025; 12(7): e01767. CrossRef - Fibroepithelial Polyp of the Vagina With Torsion: A Difficult Diagnosis Based on Clinical and Morphological Findings of the Vaginal Lesion
Efthymia Thanasa, Anna Thanasa, Gerasimos Kontogeorgis, Ektoras-Evangelos Gerokostas, Ioannis-Rafail Antoniou, Athanasios Chasiotis, Emmanouil M Xydias, Apostolos C Ziogas, Evangelos Kamaretsos, Ioannis Thanasas
Cureus.2024;[Epub] CrossRef - Recurrent fibroepithelial vaginal polyp in a 2-year-old girl: a case report and review of the literature
Mohammad Hakam Shehadeh, Ahmad M. Abualrub, Waleed M. Malhes, Amar Msarweh, Wael Amro
Annals of Medicine & Surgery.2024; 86(4): 2335. CrossRef - A vaginal fibroepithelial stromal polyp: a case report with magnetic resonance images
Naoko Ogura, Mieko Inagaki, Ritsuko Yasuda, Shigeki Yoshida, Tetsuo Maeda
BJR|case reports.2022;[Epub] CrossRef - Fast-growing fibroepithelial stromal vaginal polyp
Ana Marta Pinto, Maria Boia Martins, Isabel Ferreira, Clara Moreira
BMJ Case Reports.2022; 15(6): e250076. CrossRef - Mesenchymal lesions of the vulva
David B. Chapel, Nicole A. Cipriani, Jennifer A. Bennett
Seminars in Diagnostic Pathology.2021; 38(1): 85. CrossRef - Giant Fibroepithelial Stromal Polyp of the Vulva: Diffusion-Weighted and Conventional Magnetic Resonance Imaging Features and Pathologic Correlation
Joonghyun Yoo, Bo-Kyung Je, Suk Keu Yeom, Ye Sul Park, Kyung-Jin Min, Joo Han Lee
Journal of Pediatric and Adolescent Gynecology.2019; 32(1): 93. CrossRef - Cellular Pseudosarcomatous Fibroepithelial Stromal Polyp of the Cervix: A Lesion Mimicking as Sarcoma
Ruquiya Afrose
Advances in Cytology & Pathology.2018;[Epub] CrossRef - A Large Vaginal Bulge Might Not Be a Genital Prolapse
Natasha Ferreira Teixeira Meletti, Susana Cristina Aidé Viviani Fialho, Fabiana Resende Rodrigues, Carlos Augusto Faria
Open Journal of Obstetrics and Gynecology.2018; 08(04): 362. CrossRef - Pseudosarcomatous Vaginal Polyp
Alexis Heller, Adanna Ukazu, Qing Wang
International Journal of Surgical Pathology.2017; 25(1): 54. CrossRef
- A Giant Fibroepithelial Polyp of the Nipple: A Case Report of a Rare Benign Tumor
- Multifocal Adenocarcinomas Arising within a Gastric Inverted Hyperplastic Polyp
- Hyun-Soo Kim, Eun-Jung Hwang, Jae-Young Jang, Juhie Lee, Youn Wha Kim
- Korean J Pathol. 2012;46(4):387-391. Published online August 23, 2012
- DOI: https://doi.org/10.4132/KoreanJPathol.2012.46.4.387
- 9,825 View
- 66 Download
- 14 Crossref
-
 Abstract
Abstract
 PDF
PDF We present herein the occurrence of multifocal adenocarcinomas with a minute signet ring cell carcinoma that arose within a gastric inverted hyperplastic polyp (IHP) in a 40-year-old woman. Endoscopic ultrasonography demonstrated a heterogeneous hypoechoic mass in the third layer of the gastric wall. The endoscopic submucosal dissection specimen measuring 3.5×3.2×1.8 cm was a well-circumscribed protruding lesion that had a slit-shaped cavity. Histologically, the lesion consisted mainly of endophytic proliferation of hyperplastic columnar cells resembling normal foveolar epithelium. In addition, six foci of adenocarcinomas and a minute focus of signet ring cell carcinoma were randomly distributed in the superficial and deep regions. The adenocarcinoma was gradually transitioning from dysplasia, while the signet ring cell carcinoma was surrounded by hyperplastic foveolar epithelium. This is the first report of a gastric IHP with multifocal intramucosal adenocarcinomas and a signet ring cell carcinoma, and endoscopic submucosal dissection is used to completely resect it.
-
Citations
Citations to this article as recorded by- Case report and literature analysis of ectopic gastric glands combined with intestinal-type gastric cancer in an HP-negative background
Shiyu Peng, Shuxin Tian
Frontiers in Oncology.2025;[Epub] CrossRef - Clinicopathologic and endoscopic characteristics of ten patients with gastric hamartomatous inverted polyp: a single center case series
Ningning Dong, Fandong Meng, Bing Yue, Junzhen Hou
BMC Gastroenterology.2024;[Epub] CrossRef - Gastric hamartomatous inverted polyp: Report of three cases with a review of the endoscopic and clinicopathological features
Takuya Ohtsu, Yu Takahashi, Mitsuo Tokuhara, Tomomitsu Tahara, Mitsuaki Ishida, Chika Miyasaka, Koji Tsuta, Makoto Naganuma
DEN Open.2023;[Epub] CrossRef - Gastric Inverted Hyperplastic Polyp Removed Using Endoscopic Submucosal Dissection
Jee Won Boo, Joon Sung Kim, Byung-Wook Kim
The Korean Journal of Helicobacter and Upper Gastrointestinal Research.2023; 23(1): 63. CrossRef - The Many Faces of Gastric Inverted Polyps: a case report
S.I. Kim, M.Y. Agapov, T.F. Savostyanov, A.A. Paratovskaya, I.A. Sokolova
Russian Journal of Evidence-Based Gastroenterology.2023; 12(2): 88. CrossRef - Large gastric hamartomatous inverted polyp accompanied by advanced gastric cancer: A case report
Gyerim Park, Jihye Kim, Sung Hak Lee, Younghoon Kim
World Journal of Clinical Cases.2023; 11(28): 6967. CrossRef - Activating KRAS and GNAS mutations in heterotopic submucosal glands of the stomach
Hourin Cho, Taiki Hashimoto, Tomoaki Naka, Yasushi Yatabe, Ichiro Oda, Yutaka Saito, Takaki Yoshikawa, Shigeki Sekine
Journal of Gastroenterology.2022; 57(5): 333. CrossRef - Gastric Inverted Polyps—Distinctive Subepithelial Lesions of the Stomach
Ji-Ye Kim, Soomin Ahn, Kyoung-Mee Kim, Sun Hee Chang, Han Seong Kim, Jun Haeng Lee, Jae J. Kim, Tae Sung Sohn, Hye Ju Kang, Mee Joo
American Journal of Surgical Pathology.2021; 45(5): 680. CrossRef - Signet-ring Cell Carcinoma in Hyperplastic Polyp of the Stomach
Kenta Yoshida, Tatsuya Mikami, Takao Oyama, Yuki Sato, Taro Saito, Takafumi Mikami, Chieko Itabashi, Yasushi Soma, Shinsaku Fukuda
Internal Medicine.2019; 58(24): 3531. CrossRef - Inverted Hyperplastic Polyp in Stomach: A Case Report and Literature Review
Yeon Ho Lee, Moon Kyung Joo, Beom Jae Lee, Ji-Ae Lee, Taehyun Kim, Jin Gu Yoon, Jung Min Lee, Jong-Jae Park
The Korean Journal of Gastroenterology.2016; 67(2): 98. CrossRef - Inflammatory myofibroblastic tumor‐like stromal proliferation within gastric inverted hyperplastic polyp
Byeong‐Joo Noh, Ji Won Min, Ji‐Youn Sung, Yong‐Koo Park, Juhie Lee, Youn Wha Kim
Pathology International.2016; 66(3): 180. CrossRef - Gastric inverted hyperplastic polyp: A rare cause of iron deficiency anemia
Jin Tak Yun, Seung Woo Lee, Dong Pil Kim, Seung Hwa Choi, Seok-Hwan Kim, Jun Kyu Park, Sun Hee Jang, Yun Jung Park, Ye Gyu Sung, Hae Jung Sul
World Journal of Gastroenterology.2016; 22(15): 4066. CrossRef - Gastric Inverted Hyperplasic Polyp Composed Only of Pyloric Glands
Minsun Jung, Kyueng-Whan Min, Young-Joon Ryu
International Journal of Surgical Pathology.2015; 23(4): 313. CrossRef - A Pedunculated Submucosal Lesion in the Stomach with Inverted Downgrowth
Yoshihiko Miyamoto, Naoki Muguruma, Seisuke Okamura, Yasuyuki Okada, Shinji Kitamura, Koichi Okamoto, Akiko Yoneda, Noriko Kagawa, Tetsuji Takayama
Internal Medicine.2014; 53(15): 1625. CrossRef
- Case report and literature analysis of ectopic gastric glands combined with intestinal-type gastric cancer in an HP-negative background
- Clinicopathologic Characteristics of Left-Sided Colon Cancers with High Microsatellite Instability.
- Sang Kyum Kim, Junjeong Choi, Hyun Ki Kim, Young Nyun Park, Si Young Song, Hoguen Kim
- Korean J Pathol. 2009;43(5):428-434.
- DOI: https://doi.org/10.4132/KoreanJPathol.2009.43.5.428
- 4,661 View
- 43 Download
- 1 Crossref
-
 Abstract
Abstract
 PDF
PDF - BACKGROUND
High microsatellite instability (MSI-H) colorectal carcinomas (CRCs) with numerous mutations in the microsatellite sequence are characterized by a right-sided preponderance, frequent peritumoral and intratumoral lymphocytic infiltration, and frequent mucin production. However, no study has correlated anatomic site and type of genetic changes with clinicopathologic changes.
METHODS
We analyzed the histopathologic features of 135 MSI-H CRCs and compared them to 140 microsatellite stable (MSS) CRCs. Histopathologic changes in MSI-H were further analyzed according to anatomic sites and genetic changes.
RESULTS
MSI-H CRCs showed previously reported clinicopathologic findings; a right-sided preponderance, an increased number of mucinous carcinomas, and peritumoral lymphoid reactions (p<0.001 for each variable). Increased serum CEA levels showed an MSS CRC preponderance (p=0.013). We further analyzed the histologic differences between right- and left-sided MSI-H tumors. We found that MSI-H CRCs on both sides had similar clinicopathologic findings, except for higher tumor stage (p=0.048) and less frequent abnormal CEA levels in left-sided MSI-H tumors (p=0.027). We found that not all clinicopathologic features were different between hereditary nonpolyposis colorectal cancers (HNPCCs) and sporadic MSI-H CRCs.
CONCLUSIONS
These findings indicate that MSI-H CRCs of the left colon have similar clinicopathologic characteristics as right-sided MSI-H CRCs. We did not find any significant clinicopathological difference between HNPCCs and sporadic MSI-H CRCs. -
Citations
Citations to this article as recorded by- Fibroblast Growth Factor Receptor 1 Gene Copy Number and mRNA Expression in Primary Colorectal Cancer and Its Clinicopathologic Correlation
Yoonjin Kwak, Soo Kyung Nam, An Na Seo, Duck-Woo Kim, Sung-Bum Kang, Woo Ho Kim, Hye Seung Lee
Pathobiology.2015; 82(2): 76. CrossRef
- Fibroblast Growth Factor Receptor 1 Gene Copy Number and mRNA Expression in Primary Colorectal Cancer and Its Clinicopathologic Correlation
- Gastrointestinal Stromal Tumor of the Colon Mimicking Inflammatory Fibroid Polyp with a Novel 63 bp c-kit Deletion Mutation: A Case Report.
- In Gu Do, Cheol Keun Park, Sung Hyun Yoon, John Goldblum, Kyoung Mee Kim
- Korean J Pathol. 2009;43(4):374-377.
- DOI: https://doi.org/10.4132/KoreanJPathol.2009.43.4.374
- 3,768 View
- 26 Download
-
 Abstract
Abstract
 PDF
PDF - Colonic gastrointestinal stromal tumors (GISTs) are rare and behave aggressively compared to GISTs in other parts of the gastrointestinal tract. Therefore, accurate diagnosis of GISTs and their distinction from other mesenchymal tumors is important for proper patient management and follow-up. Herein, we present an unusual case of a colonic GIST mimicking an inflammatory fibroid polyp with a novel 63 bp deletion mutation in exon 11 of the c-kit gene, which has not previously been reported. The tumor consisted of loosely arranged spindle cells and many inflammatory cells scattered throughout the tumor. Immunohistochemically, the tumor cells were focally and weakly positive for c-kit and diffusely positive for CD34, but were negative for PKC-theta, SMA, S-100 protein, ALK-1, and desmin. Our case re-emphasizes the broad morphologic spectrum of GISTs.
- Familial Juvenile Polyposis.
- Sun Hee Chang, Shi Nae Lee, Hea Soo Koo, Ok Kyung Kim, Sun Sub Jung, Eung Bum Park
- Korean J Pathol. 1997;31(2):185-188.
- 1,986 View
- 14 Download
-
 Abstract
Abstract
 PDF
PDF - Familial juvenile polyposis is a rare intestinal polyposis characterized by the occurrence of multiple juvenile polyps in the gastrointestinal tract. We report a case of familial juvenile polyposis in a 17-year-old man with a review of the literature. This patient underwent total colectomy due to a 6 years history of rectal bleeding. Grossly, the colon showed 36 variable sized pedunculated polyps, measuring 2.5cm x 2cm from the largest size and 0.2cm x 0.2cm to the smallest size. Histologically, the polyps consisted of cystically dilated glands, lined by normal colonic epithelial cells, scattered in loose, edematous stroma containing inflammatory cell infiltration. There were no areas of tubular adenoma or malignancy in any of the polyp.
- A Case of Pseudoinvasion in Peutz-Jeghers Polyposis.
- Sang Pyo Kim, Sang Sook Lee, Eun Sook Chang
- Korean J Pathol. 1993;27(1):72-74.
- 2,377 View
- 49 Download
-
 Abstract
Abstract
 PDF
PDF - Peutz-Jeghers polyp with pseudoinvasion is characterized by the presence of mucosal islands within muscle layer often associated with mucinous cysts. It occurs when mucosal components completely penetrate the muscularis propria, and shows no cytologic signs of malignancy. In most instances, the lesion may be the result of forces exerted during intussusception in the involved small intestine. We describe a case of Peutz-Jeghers polyposis with pseudoinvasion in a 24-year-old female patient. This patient presented with intussusception and a segmental resection of the ileum was performed. Microscopically, it disclosed a hamartomatous polyp with pseudoinvasion. There were glandular islands and mucinous cysts embedded within the bundle of muscle, layer, extending, to the subserosa. There was o evidence of cellular dysplasia or desmoplasia.
- Development of Desmoid and Mesenteric Fibromas following Total Colectomy for Adenomatous Polyposis Coli in Gardner's syndrome.
- Jung Hee Cho, Yong Il Kim
- Korean J Pathol. 1989;23(4):465-469.
- 1,865 View
- 11 Download
-
 Abstract
Abstract
 PDF
PDF - We describe a case of polyposis coli, which was followed by development of desmoid in the rectus adbominis muscle and fibromas in the mesentery during an interval of two years. This case supports the hypothesis that, in Garder's syndrome, the traumatic injury by colectomy triggers an unusual fibrous proliferation in the peritoneal cavity and incision site under the possible genetic basis.
- Crohn's Disease Involving Small Intestine and Colon: 2 cases report.
- Shi Nae Lee, Sun Hee Chang, Hee Soo Yoon, Hea Soo Koo, Ok Kyung Kim, Ryung Ah Lee, Eung Beum Park
- Korean J Pathol. 1997;31(4):379-382.
- 2,222 View
- 13 Download
-
 Abstract
Abstract
 PDF
PDF - Crohn's disease was originally described as a small bowel disorder and has been known to involve the large bowel in approximately 40% of all cases with or without concomitant ileal component. We describe two cases of Crohn's diseas of small intestine and colon with a summary of differential diagnosis with ulcerative colitis. Both cases were originally diagnosed and treated as ileal tuberculosis. Grossly, there were skip lesions in both cases with prominent pseudopolyps and ulcerations in colon. Also noted were typical serpentine lesions in ileum as well as in colon. Microscopically, transmural inflammation was confirmed and one case showed scattered noncaseating granulomas in the wall. Submucosal edema and fibrosis with thickening of the wall was not prominent in colon. Polymerase chain reaction performed on paraffin block for the demonstration of Mycobacterium tuberculosis in one case showed negative reaction.
- Giant Fibrovascular Polyp of the Esophagus: A Case Report.
- Ilseon Hwang, Jong lyel Roh, Young Hee Kim, Kyung Ja Cho
- Korean J Pathol. 2007;41(6):409-411.
- 2,185 View
- 29 Download
-
 Abstract
Abstract
 PDF
PDF - Giant fibrovascular polyps are very rare benign esophageal lesions arising in the cervical esophagus. We present a case of a 70-year old man with a history of dysphagia. On esophagogram and computed tomogram, a large, sausage-like polyp (7.0 cm in greatest dimension) was identified in the cervical esophagus. Surgical resection of the polyp was successfully performed. Histologically, the polyp was composed of a mixture of highly vascularized fibrous tissue and benign adipose tissue. The fibrous tissue was loosely and densely collagenized and the surface was covered by squamous epithelium. The lesion was diagnosed as a giant fibrovascular polyp and the patient has shown no evidence of recurrence for 8 months after surgery.
- Gastroduodenal Adenomas and Carcinoma in Patients with Familial Polyposis Coli.
- Seung Sook Lee, Woo Ho Kim, Yong Il Kim
- Korean J Pathol. 1993;27(3):263-267.
- 2,069 View
- 20 Download
-
 Abstract
Abstract
 PDF
PDF - Gastric lesion in familial polyposis coli is commonly presented with fundic gland hyperplastic polyps, but duodenal and gastric adenomas together with their carcinomatous transformation have been rarely described in familial polyposis coli mostly by case reports. We present three cases of gastric adenomas in familial polyposis coli with one in synchronous development of gastric adenocarcinoma in Korea. All three cases had the family history related to familial polyosis coli and received proctocolectomy because of synchronous development of colonic adenocarcinoma. One patient developed gastric polyposis and adenocarcinoma 8 years after colectomy, and the remaining two presented with multiple polyps either in the stomach or duodenum synchronously at the time of the diagnosis of familial polyposis coli with colonic adenocarcinoma. None disclosed any evidence of Gardner's syndrome. We conclude that association of gastric adenomas in familial oplyposis coli is not uncommon and gastric adenoma-carcinoma sequence is an another important participating mechanism to understand the histogenesis of gastric carcinoma in Korea.
- Isolated Polypoid Ganglioneuroma in the Rectum.
- Se Hoon Kim, Chang Hwan Choi, Yong Han Paik, Won Ho Kim, Hoguen Kim
- Korean J Pathol. 2001;35(4):344-346.
- 2,471 View
- 42 Download
-
 Abstract
Abstract
 PDF
PDF - Gastrointestinal ganglioneuroma is a rare benign neoplasm, composed of ganglion cells, nerve fibers, and supporting cells. Ganglioneuromas are presented as isolated polypoid ganglioneuroma, ganglioneuromatous polyposis, and diffuse ganglioneuromas. We have experienced a case of an isolated ganglioneuromatous polyp in the rectum. The patient was a 58-year-old female who had experienced low abdominal discomfort and tenesmus for 6 to 7 months. Colonoscopic examination revealed a polypoid tumor in the rectum. Microscopically, the tumor showed cystic glands, expanded lamina propria, and smooth surface epithelium. Many proliferated ganglion cells with nerve fibers were evident in the lamina propria which was extended to the submucosa.
- Fibrovascular Polyp of the Hypopharynx: A Case Report.
- Sunhee Chang, Sang Hwa Shim, Ji Eun Kwak, Mee Joo, Hanseong Kim, Bum Jo Jung, Joong Wook Shin, See Young Park, Kyung Ja Cho, Je G Chi
- Korean J Pathol. 2008;42(4):226-228.
- 2,302 View
- 19 Download
-
 Abstract
Abstract
 PDF
PDF - We report here on a case of fibrovascular polyp arising in the hypopharynx of a 62-year-old man. Laryngomicroscopic surgery with laser ablation was performed to excise the mass. Histopathologically, the surface of the polyp was covered with mature squamous epithelium. The polyp showed a characteristic lobular proliferation of mature adipose tissue that was separated by myxoid or collagenous connective tissue. Some scattered skeletal muscle bundles were seen in the central portions of the polyp and these bundles were surrounded by a concentric proliferation of the spindle cells; this was reminiscent of Pacinian corpuscles. Regarding their location and the intermingled pattern of proliferating tissues, it is more plausible that the skeletal muscle is a hamartomatous component rather than entrapped, preexisting tissue.
- Myxoma of the Larynx Presenting As a Nodule.
- Young Soo Song, Si Hyong Jang, Kyueng Whan Min, Woong Na, Se Min Jang, Young Jin Jun, Seung Sam Paik
- Korean J Pathol. 2008;42(5):306-307.
- 2,367 View
- 22 Download
-
 Abstract
Abstract
 PDF
PDF - We describe herein a rare case of a laryngeal myxoma presenting as a nodule. Laryngeal myxomas involving the neck region, especially the laryngeal area, are quite rare. A 36-year-old male patient presented with a 2 month history of hoarseness. On laryngoscopic examination, there was a myxoid homogeneous transparent mass on the right vocal cord. On microscopic examination, the lesion was hypocellular and myxoid. The lesion showed stellate or spindle cells which were evenly dispersed in the poorly vascularized myxoid stroma. Although the incidence is extremely rare, a laryngeal myxoma should be considered in the differential diagnosis of laryngeal masses.
- An Unusual Meningothelial Element in a Hairy Polyp of the Hard Palate.
- Si Hyong Jang, Kyueng Whan Min, Woong Na, Se Min Jang, Seung Sam Paik
- Korean J Pathol. 2008;42(5):311-313.
- 2,139 View
- 17 Download
-
 Abstract
Abstract
 PDF
PDF - Hairy polyps are a rare malformations of bigerminal origin that comprise of both ectodermal and mesodermal elements. Meningothelial elements are an extremely rare pathologic finding in hairy polyps. Here we report a case of a hairy polyp with a meningothelial element, which originated from the hard palate. A 1-year-old boy was evaluated for an intraoral mass accompanied by multiple congenital anomalies. A small polypoid mass was noted at the midline of the hard palate. The lesion had central fibroconnective tissue with an unusual stromal component showing reticulated anastomosing pseudovascular patterns. Immunohistochemical staining of the cells lining the pseudovascular spaces and the interstitial cells revealed vimentin and epithelial membrane antigen positivity.
- Localized Pseudopolyposis of the Ascending Colon Associated with Granulomatous Colitis: A case report.
- Jeong Ja Kwak, Kye Hyun Kwon, So Young Jin, Dong Wha Lee
- Korean J Pathol. 1994;28(1):82-85.
- 2,197 View
- 17 Download
-
 Abstract
Abstract
 PDF
PDF - Pseudopolyps represent discrete areas of mucosal inflammation and regeneration that are seen in a variety of inflammatory bowel disease including ulcerative colitis and Crohn's disease. These polyps are typically short, measuring less than 1.5 cm in height. Rarely, localized giant pseudopolyposis can occur, i.e., a collection of larger inflammatory pseudopolyps giving rise to a mass lesion within the colon. The most serious problem concerned with pseudopolyposis is a confusion with carcinoma. We experienced a case of localized giant pseudopolyposis causing partial large bowel obstruction. Right hemicolectomy was done for a preoperative diagnosis of ascending colon carcinoma. The resected specimen contained a circumferential lesion, which was composed of numerous interconnecting cylindrical villi, measuring 12 cm in length and 3 cm in height. Microscopically, these polypoid lesions were inflammatory pseudopolyps. Several deep fissure-like ulcerations were noted with multifocal microabscess, lymphoid hyperplasia and an area of noncaseating granuloma.
- roded Polypoid Hyperplasia of the Rectosigmoid Colon: Report of 2 cases with special reference to its relation to mucosal prolapse syndrome.
- Nam Hoon Cho, Hee Jeong Ahn, Chan Il Park
- Korean J Pathol. 1994;28(3):297-301.
- 2,376 View
- 10 Download
-
 Abstract
Abstract
- Polypoid prolapse of mucosal folds can occur at various sites and in various conditions predominantly associated with strain during defecation. There are two well known types of mucosal prolapse syndrome(MPS), the inflammatory cloacogenic polyp(ICP) and the mucosal redundant polyp associated with diverticular disease(N4RPD). ICP is a mucosal prolapse of the anorectal junction and MRPD is a proximal analogue involving the sigmoid colon. We experienced two cases of eroded polypoid hyperplasia(EPH) of the rectosigmoid colon which manifested as a huge gyriform mass simulating the gross features of gastrointestinal lymphomas or other malignant tumors. The EPH consisted of confluent polypoid mucosal folds with rolled-up submucosa to form stalk, The polypoid lesion represented hyperplastic epithelium, erosion of the mucosal surface and congestive vascular ectasia of lamina propria and submucosa. To explain the whole morphologic features, the initial phenomenon should be the mucosal prolapse. Vascular stretching with ischemic erosion of the mucosal surface and compensatory epithelial hyperplasia ensue as the result. The ominous endoscopic and gross features of EPH should be kept in mind to avoid erroneous radical surgery.
- Ureteral Fibroepithelial Polyp: A report of four cases (One case with nephrogenic adenoma).
- Won Mee Lee, Seung Sam Paik, Eun Kyung Hong, Moon Hyang Park, Jung Dal Lee
- Korean J Pathol. 1996;30(8):715-720.
- 2,281 View
- 37 Download
-
 Abstract
Abstract
 PDF
PDF - Fibroepithelial polyps of the ureter are usually acquired rather than congenital. Most polyps are hamartomatous growths which tend to arise in the proximal portion of the left ureter. Most patients exibit either hematuria or persistent flank pain secondary to partial ureteral obstruction. Because of false positivity of urine cytology, as well as intravenous pyelogram, the correct diagnosis is confused with malignancy. Herein we report four cases of ureteral fibroepithelial polyp, which are associated with stones resulting in partial obstruction of the ureter. One of the four cases is associated with nephrogenic adenoma in the lamina propria of the adjacent ureter. The following report describes clinicopathologic findings of fibroepithelial polyp with review of literatures.
- Gastrointestinal Polyposis in Koreans: A Nationwide Survey of Clinicopathologic Analysis of 112 Surgically Resected Cases.
- Mee Soo Chang, Hoguen Kim, Woo Ho Kim, Chan Il Park, Eun Kyung Hong, Han Kyeom Kim, In Soo Suh, Byung Kee Kim, Ja June Jang, Woon Sub Han, Hyung Sik Shin, So Young Jin, Dae Young Kang, Yong Il Kim
- Korean J Pathol. 1998;32(6):404-412.
- 2,472 View
- 11 Download
-
 Abstract
Abstract
- Gastrointestinal polyposis (GIP) is a rare disease characterized by formation of the numerous polyps in the gastrointestinal tract and presenting several extraintestinal manifestations. Most of the diseases are transmitted in an autosomal dominant pattern. In Korea, the epidemiological study as well as the pathological analysis of the GIP is not well established. We therefore analysed 38 items of GIP using surgically resected specimens. The materials in this study were collected from the 12 institutions and case reports in Korean literature between 1980 and 1991. The clinicopathologic findings were reevaluated by several members of the study group for gastrointestinal pathology. The results are as follows: (1) A total of 112 cases were included in this study: 83 cases were collected from 12 institutions and 29 cases were collected from Korean literature. The cases were classified as familial adenomatous polyposis (FAP), 59 cases; Gardner's syndrome, 3 cases; juvenile polyposis, 12 cases; Peutz-Jeghers syndrome, 35 cases; multiple colonic adenomas, 3 cases. (2) Among 59 cases of FAP, the range of age at operation was 14 to 61 years, and a family history was positive in 25 cases. The number of polyps in colorectum was 100~8,000. Of the 37 cases in which the examination of polyp density was available, 16 cases (43%) showed the highest density in the rectum and the sigmoid colon. The carcinomatous change within polyp(s) was present in 18 cases (31%), and associated advanced single or multiple colonic carcinomas existed in 37 cases (63%). Twenty-six (45%) tumors out of total 58 carcinomas were in the rectum. Twenty-five patients were evaluated for the upper gastrointestinal lesions, and 11 patients (44%) had pathologic lesions; multiple fundic gland polyps in 3 cases (12%), gastric and duodenal adenomas in 2 cases (8%), gastric adenomas in 2 cases (8%), duodenal adenomas in 2 cases (8%), gastric carcinoma and adenoma in 1 case (4%), gastric carcinoma in 1 case (4%). (3) Among 3 cases of Gardner's syndrome, the range of age at operation was 25 to 31 years, a family history was identified in 2 cases. One case was associated with an advanced colonic carcinoma and carcinomatous change within polyp. Extra gastrointestinal lesions were sebaceous cyst, epidermal cyst, osteoma and desmoid tumor. (4) Among 12 juvenile polyposis, the range of age at operation was 8 to 51 years and 5 patients had a family history. The carcinomatous change within polyp was found in 2 cases (17%) and associated advanced colonic carcinoma was in 4 cases (33%). The associated different type of polyps was tubular adenomas in 9 cases (75%), hyperplastic polyps in 4 cases (33%) and villous adenomas in 2 cases (17%). (5) Among 35 Peutz-Jeghers syndrome, the range of age at first operation was 6 to 42 years, family history was positive in 11 cases. The carcinomatous change within polyp was found in 1 case (3%), and associated advanced colonic carcinoma in 1 case (3%). The epithelial misplacement was observed in 4 cases (11%), and tubular or villous adenomatous feature in 4 cases (11%). In summary, the most frequent GIP for the surgical resection in Korea is FAP and the FAP is associated with high incidence of coexisting advanced and intramucosal carcinomas. Hamartomatous polyposis syndromes, such as juvenile polyposis and Peutz-Jeghers syndrome are another frequent disease for the surgical resection and are also associated with an increased risk of cancer.
- Clinicopathologic Analysis of Gastrointestinal Polyps.
- Hye Rim Park, In Sun Kim, Seung Yong Paik
- Korean J Pathol. 1988;22(3):232-243.
- 1,975 View
- 18 Download
-
 Abstract
Abstract
 PDF
PDF - Pathologists play an important role in proper evaluation of endoscopically removed polyps of the gastrointestinal tract. This study is purposed to reclassify the polyps and review the clinicopathologic features of each histologic subtypes and their malignant potential. Our material consists of total 345 gastrointestinal polyps obtained from Jan. 1986 to Dec. 1987. The results are as follows: 1) A total of 345 gastrointestinal polyps was removed from stomach is 151 cases, from colon in 180 cases, and from small intestine in 14 cases. 2) Hyperplastic polyps were the most common type of polyps I stomach (53.6%) whereas neoplastic polyps were the most common in colon (56.1%). 3) Hyperplastic polyps of the stomach occur in any age after the 3rd decade of life and neoplastic polyps predominantly developed between the 5th and 8th decades. Juvenile retention polyps were frequently noted before the 3rd decade of age. 4) Approximately 267 cases (77.4%) of patients had a single polyp and the remainders had multiple polyps. The gastric polyps were usually located at the antrum and the colonic polyps were at the sigmoid colon and rectum. 5) Epithelial atypia was exclusively noted in the neoplastic polyps of stomach (72.7%) and colon (72.3%). Malignancy in the polyp was observed in the neoplastic polyps only (13 cases). 6) Different types of polyp may occur in the same organ.
- Tonsillar Lymphangiomatous Polyp: Report of Two Cases.
- Han Suk Ryu, Soo Young Jung, Jae Soo Koh, Seung Sook Lee
- Korean J Pathol. 2006;40(5):381-384.
- 2,448 View
- 23 Download
-
 Abstract
Abstract
 PDF
PDF - Tonsillar lymphangiomatous polyp is an uncommon hamartomatous lesion that generally arises from the tonsillar surface, and it has rarely been reported in the medical literature. Because of the uncommon clinical and pathological features of these polyps, pathologists and clinicians may experience difficulty in correctly classifying them. We report herein two cases of lymphangiomatous polyp of the tonsil in a 49 year-old man and a 30 year-old man who both presented with a tonsillar mass of the palatine tonsil. Microscopically, there were protruding polypoid lesions that had a core of fibroadipose tissue with dilated multiple lymphatics and aggregated lymphoid tissue. Both patients have been stable since resection.
- Fibroepithelial Polyp of Vagina with Atypical Stromal Cells: A case report.
- Mi Ok Park, Yong Jin Kim, Jae Bok Park
- Korean J Pathol. 1998;32(8):619-921.
- 2,415 View
- 10 Download
-
 Abstract
Abstract
- A case of fibroepithelial polyp with stromal atypia in a 25-year-old female is described. The tumor arose from the vaginal wall and measured 3.5 cm in maximal diameter. It was composed of hypercellular connective tissue stroma and focal myxoid area containing numerous atypical mono- and multinucleated stromal cells. These cells may mislead to make a diagnosis of sarcoma, particulary of botryoid rhabdomyosarcoma.
- Atypical Polypoid Adenomyomas of the Endometrium: 2 case reports.
- Hee Jeong Ahn, Kyu Rae Kim, Yoon Jung Choi, Bok Soo Kim
- Korean J Pathol. 1996;30(11):1034-1039.
- 2,175 View
- 20 Download
-
 Abstract
Abstract
 PDF
PDF - Atypical polypoid adenomyoma(APA) is a rare benign polypoid tumor arising in the uterine endometrium which was first designated by Mazur in 1981. Microscopic examination reveals architecturally and cytologically atypical endometrial glands separated by intersecting fascicles of smooth muscle cells. The tumor can be misdiagnosed as endometrial adenocarcinoma with myometrial invasion, especially on microscopic examination of a curettage specimen. However a granulation tissue-like stromal response is absent and the smooth muscle in APA is more cellular than that of normal myometrium. We are reporting 2 cases of APA of the uterine endometrium which are 1.5 cm and 1.7 cm in size, respectively, in a 30 and a 22 year-old women. This is the first report in Korean literature using the immunohistochemical staining for smooth muscle actin and desmin.
- Gastrointestinal Adenomatous Polyposis Associated with Small Cell Neuroendocrine Carcinoma of the Rectosigmoid: A case report.
- Wan Seop Kim, Eun Kyung Hong, Kang Sik Kim, Kwang Soo Lee, Jung Dal Lee
- Korean J Pathol. 1996;30(11):1040-1044.
- 2,048 View
- 16 Download
-
 Abstract
Abstract
 PDF
PDF - In adenomatous polyposis coli there are many colonic and extracolonic manifestations, and various combinations of these induce different clinical presentations and syndromes. We experienced a unique case of adenomatous polyposis of the large intestine and stomach in a 39-year-old man. In the colon, small cell neuroendocrine carcinoma rather than adenocarcinoma had developed, which did not contain adenomatous or carcinomatous foci. The adenomatous polyps in the colon were all small and sessile with no cancerous or precancerous change two years after the resection of the symptomatic gastric adenomas, even though the gastric adenomas were larger and showed dysplastic change. We think this case is another variant of adenomatous polyposis syndrome.
- Appendiceal Polyp: A report of two cases.
- Ki Hwa Yang, Jung Min Lee, Mi Sook Lee, Sang Ho Park, Young Gun Yoon, Choong Gu Kang
- Korean J Pathol. 1996;30(11):1045-1049.
- 2,006 View
- 15 Download
-
 Abstract
Abstract
 PDF
PDF - Varying types of polyps could occur in the vermiform appendix. However, it is very unusual. Collins found 57 cases (0.08%) of benign mucosal polyps in the 71,000 cases of appendectomy specimens. There has been no reported case of appendiceal polyp in the literature in Korea. The authors experienced two cases of polyp in the vermiform appendix. The first case was a 51 year-old male patient who received a left hemicolectomy due to colonic polyposis. The second case was a 71 year-old male patient who was treated by appendectomy under the clinical diagnosis of acute appendicitis. The microscopic type of both cases were hyperplastic polyp.
- Methylation Patterns of Small Nuclear Ribonucleoprotein Polypeptide N (SNRPN) Related to the Germ Cell Differentiation of Human Germ Cell Tumors.
- Sun Young Jun, Kyu Rae Kim, Jene Choi, Jae Y Ro
- Korean J Pathol. 2007;41(1):21-29.
- 2,232 View
- 20 Download
-
 Abstract
Abstract
 PDF
PDF - BACKGROUND
The histogenesis and interrelationship of the various types of germ cell tumors (GCTs) have been proposed. Dysgerminoma/seminoma (D/S) is a primitive GCT that has not acquired the potential for further differentiation, whereas other types of GCTs are in a dynamic process of differentiation towards a somatic or extraembryonal direction. A primordial germ cell giving rise to a GCT undergoes a developmentally regulated erasure and resetting of imprinted genes, but changes in the imprinting pattern in GCTs as the tumor differentiates have not been well defined. We aimed to investigate the changes of the SNRPN methylation pattern between the germinomas and non-germinomatous GCTs, as compared with the somatic methylation pattern.
METHODS
We used formalin-fixed paraffin-embedded tissue sections of 97 GCTs (18 Ds, 21 Ss, 17 yolk sac tumors (YSTs), 19 immature teratomas, and 22 mature teratomas). DNA methylation was evaluated after bisulfite modification, PCR amplification, and restriction enzyme digestion.
RESULTS
The SNRPN methylation pattern was changed in 53/74 (71.6%) of GCTs as non-somatic patterns. There were significant differences in the methylation pattern between the germinomas and non-germinomatous GCTs, the GCTs being frequently hypo- methylated in Ds/Ss (73.3%), in contrast to the frequent hypermethylation seen in the YSTs and teratomas (47.7%, p<0.05).
CONCLUSIONS
The methylation status of an imprinting gene may be involved in the mechanism causing cellular differentiation and tumorigenesis of GCTs.
- A Pathological and Immunohistochemical Study of 9 Cases of Inflammatory Fibroid Polyp.
- Nam Hoon Cho, Hyeon Joo Jeong, Ho Guen Kim
- Korean J Pathol. 1989;23(1):20-28.
- 2,217 View
- 16 Download
-
 Abstract
Abstract
 PDF
PDF - We aimed to study the clinicopathologic features of inflammatory fibroid polyp by histological and immunohistochemical methods. The materials used in this study consisted of 9 cases of inflammatory fibroid polyp: 4 in the stomach, 4 in the small intestine and 1 in the cecum. The results were as follows: Females were affected more frequently than males and the average age was 45 years(range:27-61). In cases of gastric lesion, the size tended to be smaller, the mass was mainly located along the greater curvature side of antrum, and confined to the submucosa. However inflammatory fibroid polyp of the small intestine was over 2.5 cm in size, located along the antemesenteric border, and involved the proper muscle layer. In addition, intussusception was accompanied by polyp in 2 cases of small intestinal lesions. Histologically inflammatory fibroid polyps of the stomach were characterized by prominent lymphocytic infiltration and occasional onion-skinning of stromal cells, whereas plasmocytic infiltration was prominent in those of the small intestine. Main component cells comprising this lesion were confirmed to be fibroblasts by immunohistochemistry which revealed strong reactivity to vimentin in the cytoplasm of slindle cells.
- A Case of Gastric Inverted Hyperplastic Polyp Associated with Gastritis Cystica Profunda and Early Gastric Carcinoma.
- Min Sung Choi, So Young Jin, Dong Won Kim, Dong Wha Lee, Sang Mo Park
- Korean J Pathol. 2007;41(1):55-58.
- 2,587 View
- 40 Download
-
 Abstract
Abstract
 PDF
PDF - A gastric inverted hyperplastic polyp is characterized by downward growth of the hyperplastic mucosal components into the submucosa. Lesions are composed of hyperplastic foveolar-type glands, and sometimes coexist with gastritis cystica profunda (GCP). Adenocarcinoma frequen- tly can coexist, but the relationship is not clear. A 71-year-old male was admitted to hospital because of dyspepsia for one month. He underwent a wedge resection of the stomach, after endoscopic biopsies. The gross finding showed a slightly elevated papillary lesion with central depression. Microscopically, the elevated lesion was composed of hyperplastic fundic glands and foveolar cells, and the central depressed lesion showed a nodular inverted proliferation of normal appearing gastric epithelium and glands in the submucosa. An additional proximal gastrectomy specimen exhibited marked GCP and a minute adenocarcinoma at the proximal margin with p53 protein overexpression.
- Hairy Polyp of Soft Palate in an Infant.
- Kyeongmee Park, Yeonmee Kim, Hoonyoung Woo
- Korean J Pathol. 1998;32(12):1101-1103.
- 2,176 View
- 10 Download
-
 Abstract
Abstract
- Hairy polyp is an uncommon lesion of the nasopharynx and oropharynx. It is most frequently seen as a pedunculated mass at birth or in the first year of life. It generally has been classified as dermoid derived from the ectoderm and mesoderm. The symptoms depend on the size and location of the lesion. We report the clinico-pathological features of a hairy polyp arising from the right nasopharyngeal side of soft palate in a 21-day-old girl. Grossly, a well demarcated brownish yellow solid mass, measuring 2.7x1.5x1.2 cm, showed fine hairs on the surface. The cut surface was a yellowish brown solid appearance. Microscopically, it was covered with keratinizing squamous epithelium and was composed of mature adipose tissue, skeletal muscle, a few peripheral nerve bundles, and blood vessels in the center and mature pilosebaceous units in the periphery. Knowledge of this type of malformation facilitates early intervention and avoids significant morbidity.
- Multiple Localized Hyperplastic Gastropathy: Report of A Case with A Special Reference to its Growth Pattern.
- Jung Ran Kim, Yong Il Kim
- Korean J Pathol. 1989;23(1):154-159.
- 2,016 View
- 16 Download
-
 Abstract
Abstract
 PDF
PDF - We present a case of localized mucosal hyperplasia of the stomach. The resected stomach contained four large, short stalked polyps, three of which were located in the anterior wall of body and the other in the posterior wall. In addition, numerous small sessile polyps were also scattered in the anterior and posterior fundic walls. Microscopically, the abnormally thick mucosa, carrying with it the muscularis mucosae and a thin core of loose fibrous tissue comprised the polyps by intraluminal infolding of widening of mucosal area. Abundant vasculature of the rugal pattern was prominent in the submucosa. The above findings suggest that the histogenesis of the polyps is related to both hyperplastic thickening and widening of mucosal areas in rugal pattern in the background of inverted distribution pattern of intestinal metaplasia.

 E-submission
E-submission



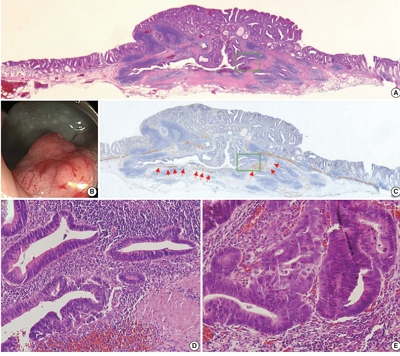
 First
First Prev
Prev



