Articles
- Page Path
- HOME > J Pathol Transl Med > Volume 48(2); 2014 > Article
-
Case Study
Follicular Dendritic Cell Sarcoma of the Inflammatory Pseudotumor-like Variant Presenting as a Colonic Polyp - Shien-Tung Pan1, Chih-Yuan Cheng2, Nie-Sue Lee2, Peir-In Liang3, Shih-Sung Chuang3,4
-
Korean Journal of Pathology 2014;48(2):140-145.
DOI: https://doi.org/10.4132/KoreanJPathol.2014.48.2.140
Published online: April 28, 2014
1Department of Pathology, Miao-Li General Hospital, Miaoli City, Taiwan.
2Department of Surgery, Miao-Li General Hospital, Miaoli City, Taiwan.
3Department of Pathology, Chi-Mei Medical Center, Tainan, Taiwan.
4Department of Pathology, Taipei Medical University, Taipei, Taiwan.
- Corresponding Author: Shih-Sung Chuang, M.D. Department of Pathology, Chi-Mei Medical Center, 901 Chung-Hwa Road, Yung-Kang District, Tainan 710, Taiwan. Tel: +886-6-281-2811 (ext. 53686), Fax: +886-6-251-1235, cmh5301@mail.chimei.org.tw
© 2014 The Korean Society of Pathologists/The Korean Society for Cytopathology
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/3.0/) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
Abstract
- Follicular dendritic cell (FDC) sarcoma is rare and is classified either as conventional type or inflammatory pseudotumor (IPT)-like variant. Extranodal presentation is uncommon and nearly all gastrointestinal FDC tumors are of the conventional type. IPT-like variant tumors occur almost exclusively in the liver and spleen and are consistently associated with Epstein-Barr virus (EBV). Here we report the case of a 78-year-old woman with an IPT-like FDC sarcoma presenting as a pedunculated colonic polyp. Histologically, scanty atypical ovoid to spindle cells were mixed with a background of florid lymphoplasmacytic infiltrate, which led to an initial misdiagnosis of pseudolymphoma. These atypical cells expressed CD21, CD23, CD35, and D2-40, and were positive for EBV by in situ hybridization, confirming the diagnosis. The patient was free of disease five months after polypectomy without adjuvant therapy. Although extremely rare, the differential diagnosis for colonic polyp should include FDC sarcoma to avoid an erroneous diagnosis. A review of the 24 cases of IPT-like FDC sarcoma reported in the literature reveal that this tumor occurs predominantly in females with a predilection for liver and spleen, and has a strong association with EBV.
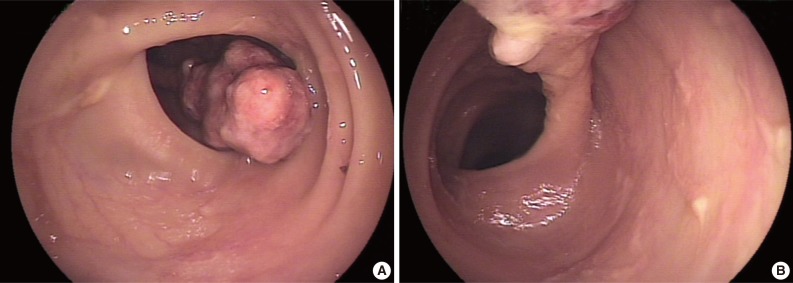
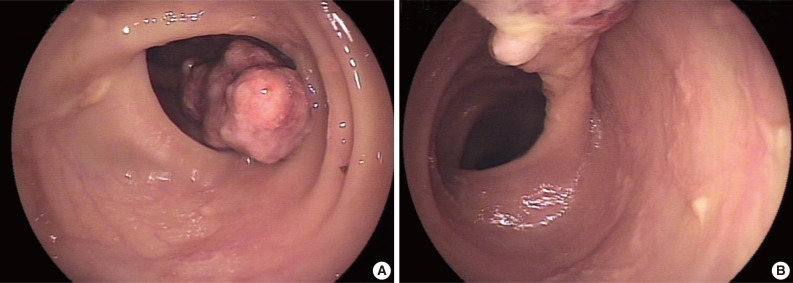
- A 78-year-old female presented with several months of abdominal discomfort and bloody stool. The patient also reported irregular bowel movements. There was no fever, body weight loss, night sweating or malaise. Physical examination showed no evidence of lymphadenopathy. Laboratory data showed mild anemia with hemoglobin at 10 g/dL and normal carcinoembryonic antigen level. Barium enema performed one month prior to endoscopy revealed an oval-shaped filling defect in the colon. Colonoscopic examination revealed a 3 cm pedunculated polyp in the transverse colon, 50 cm from the anal verge (Fig. 1). An endoscopic biopsy showed granulation tissue with inflammatory exudates. Two weeks later, the patient underwent polypectomy. The resected tumor, 3.9×1.9×1.7 cm, was polypoid with ulceration. The cut surface was solid and tan without necrosis or hemorrhage. An initial diagnosis of pseudolymphoma was made, however, subsequent pathological review with additional ancillary studies lead to a revised diagnosis of FDC sarcoma. Abdominal computerized tomography and chest X-ray were negative. The patient was free of disease five months after the polypectomy without adjuvant chemotherapy.
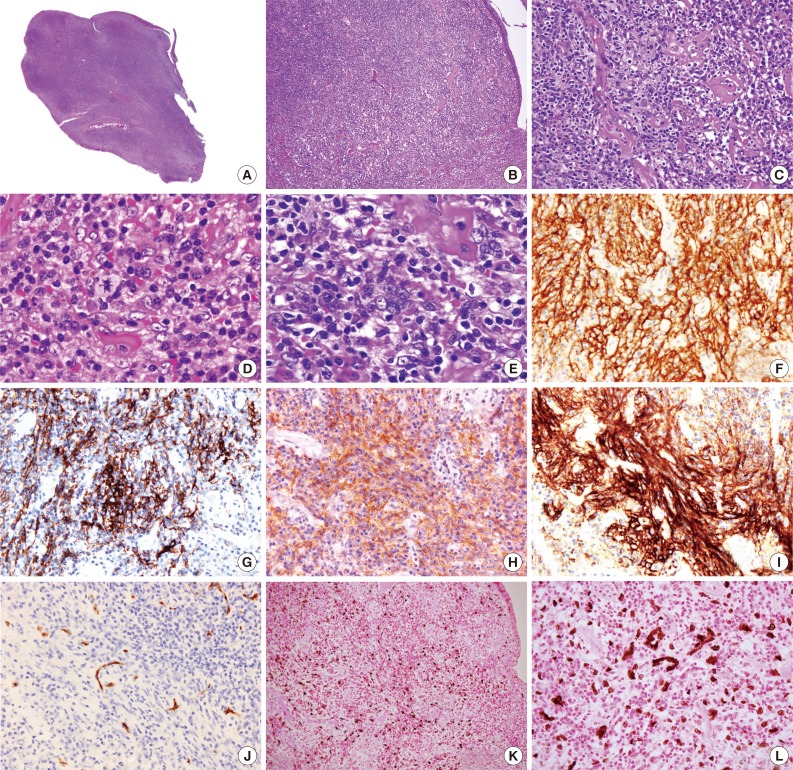
- Microscopically, the polypoid tumor showed focal ulceration and a florid lymphocytic infiltration with germinal center formation and a vascular stroma (Fig. 1). There were no epithelioid granulomas or hyalinized blood vessels entering the germinal centers. The interfollicular areas were extended by small lymphocytes and numerous plasma cells and included some large ovoid to spindled cells with vesicular nuclei and open chromatin with lightly eosinophilic syncytial cytoplasm. There were essentially no eosinophils visualized in the stroma. Occasionally, multinucleated giant cells and rare mitotic figures were discernible (Fig. 2). These atypical cells were dispersed and were more prominent beneath the area of ulceration. They were often accompanied by small sclerotic bands and were mostly scattered individually without forming intersecting spindle cell fascicles. Immunohistochemical studies showed that these large cells expressed CD21, CD23, CD35, CD45, p53, D2-40, and epithelial membrane antigen and weakly expressed estrogen receptors, but not CD34, CD68, CD117, DOG-1, S-100 protein, pan-cytokeratin, and progesterone receptors. Staining for smooth muscle actin was strongly positive in the spindle cells, but less intense in the areas where FDC markers such as CD21 were strongly expressed. The spindle cells were also positive for EBV-encoded mRNA (EBER) by in situ hybridization. The background lymphocytes were mixed small T and B cells; and the plasma cells were polytypic for light chain expression. The polymerase chain reaction-based clonality study for B-cell receptor gene rearrangement showed no amplification using IGH/FR3 primers. A diagnosis of IPT-like FDC sarcoma was made based on these results, while GI stromal tumor was excluded based on the histopathology and lack of staining for CD34, CD117, and DOG-1.
- The pertinent clinicopathological features of cases with IPT-like FDC sarcoma in the English-language literature are summarized in Table 1. Overall there have been 20 women and 4 men (female:male ratio of 5:1) diagnosed with IPT-like FDC sarcoma, with a median age of 50 (range, 19 to 78). Excepting two cases, including the current one, all cases (92%) presented as hepatic or a splenic tumor. All the cases expressed at least one FDC marker with of the most frequent being CD21 (100%) and CAN.42 (100%), followed by CD23 (72%) and CD35 (67%). EBV-latent membrane protein 1 (LMP-1) was detected immunohistochemically in 75% of cases while all cases were positive for EBV by in situ hybridization. All cases, except the one described here, were treated with surgery and one case received additional adjuvant chemotherapy. Recurrence occurred in 4 (17%) patients with follow-up times ranging from 0.2 to 7.9 years. Only one patient (4%) died of disease at 7.9 years. In summary, IPT-like FDC sarcoma occurs predominantly in females, most commonly in the liver and spleen, displays a strong association with EBV, and generally has an indolent course.
CASE REPORT
- Extranodal FDC sarcoma remains under-recognized due to its rarity, and the diagnosis of the IPT-like variant is even more challenging. Although the polypoid presentation has never been reported previously in FDC sarcoma and the tumor cells were obscured by lymphoplasmacytic and stromal cells, some atypical cells with spindled or ovoid nuclei were still discernible in our case. Most importantly, the tumor cells could be highlighted using FDC markers (CD21, CD23, and CD35) and were diffusely positive for EBER. Our case demonstrated that when FDC sarcoma was included in the differential diagnostic list, a correct diagnosis could be reached even in a rare clinical setting using detailed morphological evaluation, immunohistochemistry and ancillary study with EBER.
- Most FDC sarcomas present as lymphadenopathy, with the neck being the most common affected site.2 First reported by Chan et al.2 in 1994, extranodal FDC sarcomas are extremely rare with only around 100 cases in the English-language literature.4,5 They occur at various sites including the palate, tonsil, oral cavity, soft tissue, skin, mediastinum, liver, spleen, and GI tract. Extranodal FDC sarcomas involving the GI tract are extremely rare and the vast majority of these tumors are of the conventional type, circumscribed with yellowish white, fleshy cutting surfaces, ranging from 1 to 20 cm.17 Microscopically they comprise spindled to ovoid cells forming fascicles, a storiform growth pattern pattern, whorls, diffuse sheets or vague nodules. Scattered small lymphocytes are always present. The tumor cells show various degrees of nuclear atypia from benign to high-grade malignant features. Most commonly, the tumor cells form a syncytial pattern with indistinct cell borders containing oval or elongated nuclei and vesicular or granular finely dispersed chromatin and distinct nucleoli. Hemorrhage and necrosis are often present.18
- The IPT-like variant of FDC sarcoma differs from the conventional type by a marked female predominance, a selective localization in the liver and spleen, frequent systemic symptoms, indolent behavior despite an intra-abdominal location and a dispersed distribution of tumor cells among prominent lymphoplasmacytic cells. Many of these tumors were initially reported in the literature as inflammatory pseudotumors. Most notably, this variant is consistently associated with EBV, in contrast to the conventional type, which is only very rarely associated with EBV.3 The universal association of EBV with IPT-like variant of FDC sarcomas is strongly suggestive of a pathogenetic role; EBV-induced cytokines and monokines are known to lead to vascular proliferation, inflammation and damage of the blood vessels,19 which may explain the unusual presentations of prominent vascular changes in our case.
- All GI and IPT-like FDC sarcomas involving liver/spleen present as circumscribed fleshy masses with frequent hemorrhage and necrosis. Furthermore, previously reported GI tumors involved the intestinal wall or mesentery, and were nearly all of the conventional type. The exception is a single case of IPT-like variant reported by Agaimy and Wünsch6 in which the main tumor was identified in the mesentery (6 cm) of the terminal ileum with a submucosal spreading (1.5 cm).20 Interestingly, this tumor exhibited neoplastic cells with bizarre lobulated nuclei and prominent eosinophilia mimicking Hodgkin lymphoma. As in all the other previously reported GI tumors, this case was also EBER-negative.6 The tumor in our patient was unique as the first case of GI FDC sarcoma with a polypoid growth and an association with EBV.
- A wide age range and equal sex distribution have been reported for FDC sarcoma, except for a marked female predilection in cases of the IPT-like variant. The clinical course is usually indolent, much like a low or intermediate-grade soft tissue sarcoma. Most patients are treated by complete surgical resection, with or without adjuvant radiotherapy or chemotherapy, though the local recurrence rate is more than 50% and metastases occur in about 25% of patients.2 In a series of 11 patients with IPT-like FDC sarcoma treated by surgical excision alone, the overall mortality rate was 11% and the recurrence rate was 33%.3 A recent review of 106 cases of extranodal FDC sarcomas showed that extranodal FDC tumors behave like soft tissue sarcomas and could be classified histologically into low- and high-grade lesions. Tumor recurrence was associated with tumors greater than or equal to 5 cm in size, having high-grade histology and a mitotic count greater than or equal to 5 per 10 high power field were associated with tumor recurrence.5 In the 18 cases of GI FDC sarcoma reviewed by Agaimy and Wunsch,6 33% of the patients developed recurrence in 1-84 months and 11% died of disease within a short mean follow-up time, suggesting that some patients might develop recurrence or metastasis if extended follow-up were obtained. Our patient received polypectomy alone without adjuvant therapy and was free of recurrence in 5 months. A longer follow-up is clearly necessary in this case.
- The most important alternate diagnosis to consider in our patient is inflammatory fibroid polyp (IFP). Occurring most commonly in the terminal ileum, IFPs are rare mesenchymal tumors of the GI tract that consist of edematous spindle-shaped stromal cells and an inflammatory infiltrate rich in eosinophils.21 Pantanowitz et al.22 investigated 16 IFPs and found that stromal cells were diffusely positive for CD34 and fascin in all cases, and that these stromal cells were, in addition, immunoreactive for calponin and smooth muscle actin in 88% and 25% of cases, respectively. CD35 was also found to be focally reactive in the stromal cells.12 They were negative for CD21, CD23, and EBER. Based on these findings, they considered IFP to be a reactive lesion and suggested that the proliferating stromal cells were of dendritic cell origin, with some cases also exhibiting myofibroblastic features. In brief, IFP can be distinguished from IPT-like variant of FDC sarcoma by the abundance of eosinophils, CD34 immunoreactivity, and the absence of FDC markers other than CD35 (such as CD21 and CD23) and EBER-negativity. In our case, in addition to EBER-positivity, there were essentially no eosinophils and the tumor cells expressed all FDC markers including CD21, CD23, CD35, and D2-40. These results are more in keeping with FDC sarcoma than with IFP.
- The other item on the differential diagnosis is IPT. Makhlouf and Sobin23 compared IPT (or inflammatory myofibroblastic tumors [IMFT]) and IFP of the GI tract. They found that in four of the 20 IPT/IMFT and five of 21 IFP, the spindle cell population were positive for EBV by in situ hybridization and immunohistochemistry for LMP-1.23 They considered IMFT synonymous with IPT and concluded that the lesions in the GI tract were extremely rare and differ clinically, histologically, and immunohistochemically from IFP. However, they did not address the significance of EBV in these lesions. In the current World Helath Organization (WHO) classification of GI tumors, IPT and IPT-like FDC sarcoma can be distinguished by the expression of FDC markers and EBER-positivity in the later tumors.21 The EBV-positive tumors described earlier in the literature might well be classified as IPT-like FDC sarcomas using the current WHO classification.
- In summary, we presented a rare case of primary IPT-like FDC sarcoma in the GI tract. This tumor presented as a colonic polyp and was misdiagnosed initially as pseudolymphoma due to the heavy inflammatory background obscuring the scanty tumor cells. To avoid misdiagnosis, a high index of suspicion is warranted and FDC sarcoma should be considered in the differential diagnosis of a colonic polyp with a heavy inflammatory background on histological examination.
DISCUSSION
- 1. Monda L, Warnke R, Rosai J. A primary lymph node malignancy with features suggestive of dendritic reticulum cell differentiation: a report of 4 cases. Am J Pathol 1986; 122: 562-572. PubMedPMC
- 2. Chan JK, Pileri SA, Delsol G, et al. Follicular dendritic cell sarcoma. In: Swerdlow SH, Campo E, Harris NL, eds. WHO classification of tumours of haemtopoietic and lymphoid tissues. Lyon: IARC Press, 2008; 363-364.
- 3. Cheuk W, Chan JK, Shek TW, et al. Inflammatory pseudotumor-like follicular dendritic cell tumor: a distinctive low-grade malignant intra-abdominal neoplasm with consistent Epstein-Barr virus association. Am J Surg Pathol 2001; 25: 721-731. PubMed
- 4. Shia J, Chen W, Tang LH, et al. Extranodal follicular dendritic cell sarcoma: clinical, pathologic, and histogenetic characteristics of an underrecognized disease entity. Virchows Arch 2006; 449: 148-158. ArticlePubMedPDF
- 5. Li L, Shi YH, Guo ZJ, et al. Clinicopathological features and prognosis assessment of extranodal follicular dendritic cell sarcoma. World J Gastroenterol 2010; 16: 2504-2519. ArticlePubMedPMC
- 6. Agaimy A, Wünsch PH. Follicular dendritic cell tumor of the gastrointestinal tract: report of a rare neoplasm and literature review. Pathol Res Pract 2006; 202: 541-548. ArticlePubMed
- 7. Arber DA, Kamel OW, van de Rijn M, et al. Frequent presence of the Epstein-Barr virus in inflammatory pseudotumor. Hum Pathol 1995; 26: 1093-1098. ArticlePubMed
- 8. Selves J, Meggetto F, Brousset P, et al. Inflammatory pseudotumor of the liver: evidence for follicular dendritic reticulum cell proliferation associated with clonal Epstein-Barr virus. Am J Surg Pathol 1996; 20: 747-753. PubMed
- 9. Shek TW, Ho FC, Ng IO, Chan AC, Ma L, Srivastava G. Follicular dendritic cell tumor of the liver: evidence for an Epstein-Barr virus-related clonal proliferation of follicular dendritic cells. Am J Surg Pathol 1996; 20: 313-324. PubMed
- 10. Shek TW, Liu CL, Peh WC, Fan ST, Ng IO. Intra-abdominal follicular dendritic cell tumour: a rare tumour in need of recognition. Histopathology 1998; 33: 465-470. ArticlePubMedPDF
- 11. Chen TC, Kuo TT, Ng KF. Follicular dendritic cell tumor of the liver: a clinicopathologic and Epstein-Barr virus study of two cases. Mod Pathol 2001; 14: 354-360. ArticlePubMedPDF
- 12. Brittig F, Ajtay E, Jaksó P, Kelényi G. Follicular dendritic reticulum cell tumor mimicking inflammatory pseudotumor of the spleen. Pathol Oncol Res 2004; 10: 57-60. ArticlePubMedPDF
- 13. Horiguchi H, Matsui-Horiguchi M, Sakata H, et al. Inflammatory pseudotumor-like follicular dendritic cell tumor of the spleen. Pathol Int 2004; 54: 124-131. ArticlePubMed
- 14. Bai LY, Kwang WK, Chiang IP, Chen PM. Follicular dendritic cell tumor of the liver associated with Epstein-Barr virus. Jpn J Clin Oncol 2006; 36: 249-253. ArticlePubMed
- 15. Laurent C, Meggetto F, de Paiva GR, et al. Follicular dendritic cell tumor of the spleen associated with diffuse large B-cell lymphoma. Hum Pathol 2008; 39: 776-780. ArticlePubMed
- 16. Granados R, Aramburu JA, Rodríguez JM, Nieto MA. Cytopathology of a primary follicular dendritic cell sarcoma of the liver of the inflammatory pseudotumor-like type. Diagn Cytopathol 2008; 36: 42-46. ArticlePubMed
- 17. Chan JK, Tsang WY, Ng CS, Tang SK, Yu HC, Lee AW. Follicular dendritic cell tumors of the oral cavity. Am J Surg Pathol 1994; 18: 148-157. ArticlePubMed
- 18. Chan JK, Fletcher CD, Nayler SJ, Cooper K. Follicular dendritic cell sarcoma: clinicopathologic analysis of 17 cases suggesting a malignant potential higher than currently recognized. Cancer 1997; 79: 294-313. ArticlePubMed
- 19. Teruya-Feldstein J, Jaffe ES, Burd PR, et al. The role of Mig, the monokine induced by interferon-gamma, and IP-10, the interferon-gamma-inducible protein-10, in tissue necrosis and vascular damage associated with Epstein-Barr virus-positive lymphoproliferative disease. Blood 1997; 90: 4099-4105. PubMed
- 20. Li Z, Jin K, Yu X, et al. Extranodal follicular dendritic cell sarcoma in mesentery: a case report. Oncol Lett 2011; 2: 649-652. ArticlePubMedPMC
- 21. Miettinen M, Fletcher CD, Kindblom LG, et al. Mesenchymal tumors of the small intestine. In: Bosman FT, Carneiro F, Hruban RH, Theise ND, eds. WHO classification of tumours of the digestive system. Lyon: IARC Press, 2010; 241-250.
- 22. Pantanowitz L, Antonioli DA, Pinkus GS, Shahsafaei A, Odze RD. Inflammatory fibroid polyps of the gastrointestinal tract: evidence for a dendritic cell origin. Am J Surg Pathol 2004; 28: 107-114. PubMed
- 23. Makhlouf HR, Sobin LH. Inflammatory myofibroblastic tumors (inflammatory pseudotumors) of the gastrointestinal tract: how closely are they related to inflammatory fibroid polyps? Hum Pathol 2002; 33: 307-315. ArticlePubMed
REFERENCES
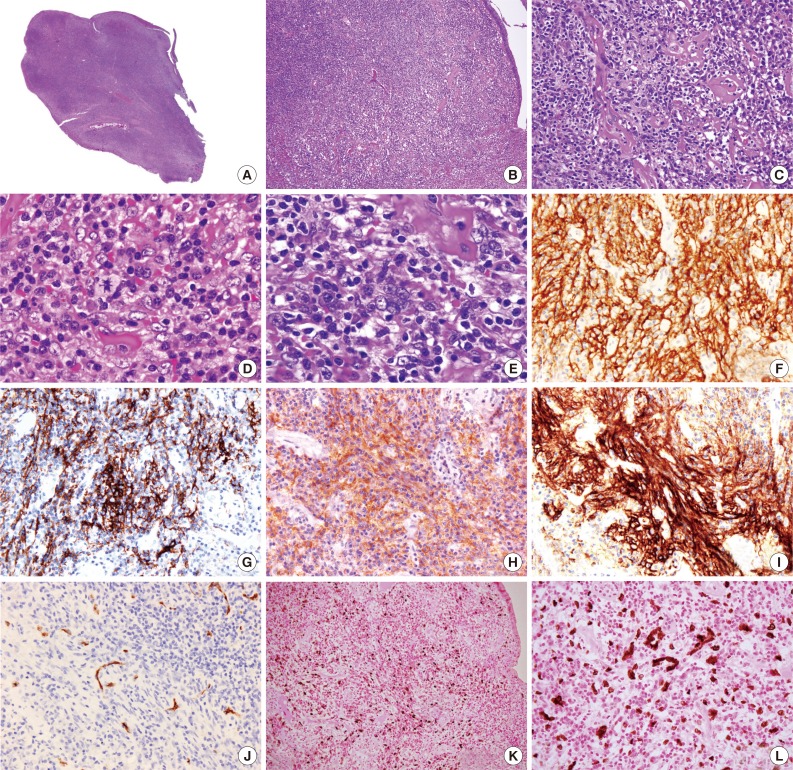
| Case No. | Sex/Age (yr) | Site | Size (cm) |
Immunohistochemistry |
EBER | Tx | Outcome (yr) | Reference | |||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| CD21 | CD23 | CD35 | CNA.42 | Other | LMP1 | ||||||||
| 1 | M/70 | Spleen | 5.5 | ND | ND | ND | ND | FDC+ | ND | + | Surgery | NR | Arber et al. [7] |
| SMA- | |||||||||||||
| 2 | F/68 | Liver | 12 | + | ND | + | ND | R4/23+ | + | + | Surgery | NR | Selves et al. [8] |
| CT | |||||||||||||
| 3 | F/35 | Liver | 20 | + | - | + | ND | R4/23+ | + | + | Surgery | Recurrence | Shek et al. [9] |
| SMA+ | NED, 2.5 | ||||||||||||
| 4 | M/37 | Liver | 15 | + | + | + | ND | ND | + | Surgery | NED, 2 | Shek et al. [10] | |
| 5 | F/19 | Liver | 12 | -/+a | - | -/+a | + | HHF-35+ | +/- | + | Surgery | NED, 3.5 | Cheuk et al. [3] |
| 6 | F/56 | Liver | 15 | +a | + | +a | + | HHF-35- | + | + | Surgery | Recurrence, alive | Cheuk et al. [3] |
| with disease, 4.5 | |||||||||||||
| 7 | F/40 | Liver | 12.5 | -/+a | - | -/+a | + | HHF-35- | + | + | Surgery | Recurrence, 9 | Cheuk et al. [3] |
| 8 | F/49 | Liver | 4.2 | +a | + | +a | + | +/- | + | Surgery | NED, 0.75 | Cheuk et al. [3] | |
| 9 | M/37 | Liver | 15 | +a | +/- | +a | + | HHF-35+/- | - | + | Surgery | NED, 3.5 | Cheuk et al. [3] |
| 10 | F/35 | Liver | 20 | +* | + | +* | + | HHF-35+/- | +/- | + | Surgery | Recurrence | Cheuk et al. [3] |
| DOD, 7.9 | |||||||||||||
| 11 | F/31 | Liver | 15 | -/+a | - | -/+a | + | HHF-35+/- | - | + | Surgery | NED, 5 | Cheuk et al. [3] |
| 12 | F/58 | Spleen | 22 | +a | - | +a | + | +/- | + | Surgery | NED, 0.3 | Cheuk et al. [3] | |
| 13 | F/39 | Spleen | 7.5 | +a | + | +a | + | + | + | Surgery | NED, 0.2 | Cheuk et al. [3] | |
| 14 | F/61 | Spleen | 3.5 | +a | + | +a | + | HHF-35+/- | +/- | + | Surgery | NR | Cheuk et al. [3] |
| 15 | F/49 | Peripancreas | 9.5 | +a | + | +a | + | HHF-35- | +/- | + | Surgery | NR | Cheuk et al. [3] |
| 16 | F/51 | Liver | 15 | + | + | - | ND | ND | + | Surgery | NED, 3 | Chen et al. [11] | |
| 17 | F/57 | Liver | 12 | + | + | - | ND | ND | + | Surgery | NED, 1 | Chen et al. [11] | |
| 18 | M/54 | Spleen | 12 | + | + | ND | ND | SMA+ | +/- | + | Surgery | NED, 4 | Brittig et al. [12] |
| 19 | F/77 | Spleen | 11 | ND | ND | ND | + | + | + | Surgery | NED, 3 | Horiguchi et al. [13] | |
| 20 | F/30 | Liver | 5.5 | + | ND | + | ND | ND | + | Surgery | NED, 2 | Bai et al. [14] | |
| 21 | F/50 | Spleen | 5 | + | ND | + | + | SMA+ | ND | + | Surgery | NED, 0.3 | Laurent et al. [15] |
| 22 | F/57 | Liver | 13 | + | + | ND | ND | ND | + | Surgery | NED, 2 | Granados et al. [16] | |
| 23 | M/45 | Liver | 14.5 | + | + | - | ND | ND | + | Surgery | NED | Li et al. [5] | |
| 24 | F/78 | Colon | 3.9 | + | + | + | ND | ND | + | Polypectomy | NED, 0.5 | Present case | |
| Subtotal (%) | NA | NA | 11/11 (100) | 13/18 (72) | 6/9 (67) | 13/13 (100) | 6/8 (75) | 24/24 (100) | NA | NA | NA | ||
NACase 1-3 and 11 are reported as inflammatory pseudotumor.
IPT, inflammatory pseudotumor; FDC, follicular dendritic cell; LMP-1, latent membrane protein 1; EBER, Epstein-Barr virus-encoded mRNA; Tx, treatment; M, male; ND, not done; SMA, smooth muscle actin; NR, not recorded; F, female; CT, chemotherapy; NED, no evidence of disease; DOD, die of disease; NA, not applicable.
aStaining results from using combined CD21 and CD35 antibodies. These cases are not included in the calculation for the percentage of markers expressed by IPT-like FDC sarcomas.
Figure & Data
References
Citations

- The fifth edition of the WHO classification of mature T cell, NK cell and stroma-derived neoplasms
Ayoma D Attygalle, Kennosuke Karube, Yoon Kyung Jeon, Wah Cheuk, Govind Bhagat, John K C Chan, Kikkeri N Naresh
Journal of Clinical Pathology.2025; 78(4): 217. CrossRef - Genomic and Transcriptomic Landscape of Epstein-Barr Virus-Positive Inflammatory Follicular Dendritic Cell Sarcoma: A Multicenter Study
Yan Li, Ze-Lin Weng, Han-Xiao Fei, Hai-Feng Li, Yi-Na Liu, Le-Le Zhang, Qiong Zhang, Xin Weng, Yuan-Yuan Wang, Wen-Yong Huang, Zhi-Xing Cao, Kai-Yan Yang, Xi-Liang Chen, Jie Gao, Wen-Sheng Yang, Fang Liu, Juan-Juan Yong, Jing-Ping Yun, Hua Zhang, Yu-Hua H
Modern Pathology.2025; 38(10): 100864. CrossRef - What is new in the 5th edition of the World Health Organization classification of mature B and T/NK cell tumors and stromal neoplasms?
Ayoma D. Attygalle, John K. C. Chan, Sarah E. Coupland, Ming-Qing Du, Judith A. Ferry, Daphne de Jong, Dita Gratzinger, Megan S. Lim, Alina Nicolae, German Ott, Andreas Rosenwald, Anna Schuh, Reiner Siebert
Journal of Hematopathology.2024; 17(2): 71. CrossRef - Pathologic characteristics of histiocytic and dendritic cell neoplasms
Sun Och Yoon
Blood Research.2024;[Epub] CrossRef - Epstein-barr virus (EBV)-positive inflammatory pseudotumor-like follicular dendritic cell sarcoma (IPT-like FDCS) presenting as thrombocytopenia: A case report and literature review
Jiawei Jin, Xiaolong Zhu, Yi Wan, Yang Shi
Heliyon.2024; 10(12): e32997. CrossRef - EBV-positive inflammatory follicular dendritic cell sarcoma of the colon with clonal immunoglobulin gene rearrangement: A case report and literature review
Xia Xu, Xiuzhen Li, Qun Deng, Kaihang Yu, Jinfan Li
Heliyon.2024; 10(11): e31947. CrossRef - Challenges in the Diagnosis of Epstein-Barr Virus-positive Inflammatory Follicular Dendritic Cell Sarcoma
Yan Li, Xia Yang, Lili Tao, Weimei Zeng, Min Zuo, Shuo Li, Liyan Wu, Yanshong Lin, Ziying Zhang, Jingping Yun, Yuhua Huang
American Journal of Surgical Pathology.2023; 47(4): 476. CrossRef - Epstein-Barr Virus-Positive Inflammatory Follicular Dendritic Cell Sarcoma Presenting as a Colonic Polyp: Report of a Case with a Literature Review
Jiahui Hu, Dongdong Huang, Chengfu Xu, Yi Chen, Han Ma, Zhe Shen
Medicina.2023; 59(7): 1341. CrossRef - A Clinicopathology Review and Update of Epstein–Barr Virus-Associated Mesenchymal Tumors
Oswald Zhao Jian Lee, Noorjehan Omar, Joshua K. Tay, Victor Kwan Min Lee
Cancers.2023; 15(23): 5563. CrossRef - Granulomatous splenic mass with necrosis revealing an EBV-positive inflammatory follicular dendritic cell sarcoma
Irena Antonia Ungureanu, Renato Micelli Lupinacci, Marie Parrens, Jean-François Emile
Journal of Surgical Case Reports.2022;[Epub] CrossRef - Case report: Hepatic inflammatory pseudotumor-like follicular dendritic cell sarcoma: A rare case and minireview of the literature
Fan Ding, Chao Wang, Chi Xu, Hui Tang
Frontiers in Medicine.2022;[Epub] CrossRef - Follicular dendritic cell sarcoma of gastrointestinal tract with two emerging distinct subtypes: a case report and systemic review
Hongxing Gui, Jigisha Chaudhari, Rifat Mannan
Diagnostic Pathology.2022;[Epub] CrossRef - Surgical treatment of liver inflammatory pseudotumor-like follicular dendritic cell sarcoma: A case report
Li-Yue Fu, Jiu-Liang Jiang, Meng Liu, Jun-Jun Li, Kai-Ping Liu, Hai-Tao Zhu
World Journal of Gastrointestinal Oncology.2022; 14(11): 2288. CrossRef - Inflammatory pseudotumor-like follicular/fibroblastic dendritic cell sarcoma: focus on immunohistochemical profile and association with Epstein-Barr virus
Francesca Pagliuca, Andrea Ronchi, Annamaria Auricchio, Eva Lieto, Renato Franco
Infectious Agents and Cancer.2022;[Epub] CrossRef - Recent Advances in Digestive Tract Tumors: Updates From the 5th Edition of the World Health Organization “Blue Book”
Raul S. Gonzalez, Anwar Raza, Robert Propst, Oyedele Adeyi, Justin Bateman, Sabrina C. Sopha, Janet Shaw, Aaron Auerbach
Archives of Pathology & Laboratory Medicine.2021; 145(5): 607. CrossRef - Hepatic inflammatory pseudotumor-like follicular dendritic cell tumor: a case report
Ana Daniela Pascariu, Andreea Ioana Neagu, Andrei Valentin Neagu, Alexandru Băjenaru, Cezar Iulian Bețianu
Journal of Medical Case Reports.2021;[Epub] CrossRef - Inflammatory pseudotumor-like follicular dendritic cell sarcoma: Literature review of 67 cases
Hao Wu, Peng Liu, Xiao-Ran Xie, Jing-Shu Chi, Huan Li, Can-Xia Xu
World Journal of Meta-Analysis.2021; 9(1): 1. CrossRef - New Clinicopathologic Scenarios of EBV+ Inflammatory Follicular Dendritic Cell Sarcoma
Xiang-Nan Jiang, Yan Zhang, Tian Xue, Jie-Yu Chen, Alex C.L. Chan, Wah Cheuk, John K.C. Chan, Xiao-Qiu Li
American Journal of Surgical Pathology.2021; 45(6): 765. CrossRef - Select Epstein-Barr Virus–Associated Digestive Tract Lesions for the Practicing Pathologist
Zainab I. Alruwaii, Elizabeth A. Montgomery
Archives of Pathology & Laboratory Medicine.2021; 145(5): 562. CrossRef - Overview of Gastrointestinal Lymphoproliferative disorders✰
Aaron Auerbach, Nadine S. Aguilera
Seminars in Diagnostic Pathology.2021; 38(4): 1. CrossRef - Follicular dendritic cell sarcoma
Fabio Facchetti, Matteo Simbeni, Luisa Lorenzi
Pathologica.2021; 113(5): 316. CrossRef - Hepatic inflammatory pseudotumor-like follicular dendritic cell tumor with hepatic lymphoma history
Jiang Li, Hai-su Tao, Dong Chen, Zhi-yong Huang, Er-lei Zhang
Medicine.2021; 100(39): e27392. CrossRef - Clinicopathological characteristics of extranodal follicular dendritic cell sarcoma: A report of two cases
Xing Zhao, Dayong Sun, Gang Zhang
Oncology Letters.2021;[Epub] CrossRef - Inflammatory pseudotumour-like follicular dendritic cell tumour of the colon with plasmacytosis mimicking EBV-positive lymphoproliferative disorder
Ying-Ren Chen, Chi-Lin Lee, Yen-Chien Lee, Kung-Chao Chang
Pathology.2020; 52(4): 484. CrossRef - Beware the inflammatory cell-rich colonic polyp: a rare case of EBV-positive inflammatory pseudotumour-like follicular dendritic cell sarcoma with increased IgG4-positive plasma cells
Lynne Goh, Nan Zun Teo, Lai Mun Wang
Pathology.2020; 52(6): 713. CrossRef - Epstein–Barr virus‐positive inflammatory follicular dendritic cell sarcoma presenting as a solitary colonic mass: two rare cases and a literature review
Xiaokang Ke, Huihua He, Qingping Zhang, Jingping Yuan, Qilin Ao
Histopathology.2020; 77(5): 832. CrossRef - Inflammatory pseudotumor-like follicular dendritic cell sarcoma: A brief report of two cases
Bi-Xi Zhang, Zhi-Hong Chen, Yu Liu, Yuan-Jun Zeng, Yan-Chun Li
World Journal of Gastrointestinal Oncology.2019; 11(12): 1231. CrossRef - Epstein-Barr virus (EBV)–associated lymphoid proliferations, a 2018 update
Sherif A. Rezk, Xiaohui Zhao, Lawrence M. Weiss
Human Pathology.2018; 79: 18. CrossRef - A Rare Case of Epstein-Barr Virus Negative Inflammatory Pseudotumor-like Follicular Dendritic Cell Sarcoma Presenting as a Solitary Colonic Mass in a 53-Year-Old Woman; Case Report and Review of Literature
Rossana Kazemimood, Farid Saei Hamedani, Asma Sharif, Sujata Gaitonde, Elizabeth Wiley, Pier Cristoforo Giulianotti, John Vincent Groth
Applied Immunohistochemistry & Molecular Morphology.2017; 25(5): e30. CrossRef - A Case of Inflammatory Pseudotumor-like Follicular Dendritic Cell Sarcoma of the Lymph Node in the Small Bowel Mesentery Accompanied by Myasthenia Gravis
Daichi KITAGUCHI, Katsuji HISAKURA, Taiki SATO, Masanao KURATA, Tatsuya ODA, Nobuhiro OHKOHCHI
Nihon Rinsho Geka Gakkai Zasshi (Journal of Japan Surgical Association).2017; 78(3): 527. CrossRef - Clinicopathological features of inflammatory pseudotumour‐like follicular dendritic cell tumour of the abdomen
Yanyang Chen, Huijuan Shi, Hui Li, Tiantian Zhen, Anjia Han
Histopathology.2016; 68(6): 858. CrossRef - A Rare Case of Follicular Dendritic Cell Sarcoma with Pseudochylous Effusion and Review of Literature From India
Kamal Kant Sahu, Gaurav Prakash, Sandeep Rao, Amanjit Bal, Pankaj Malhotra, Jasmina Ahluwalia, Rakesh K. Vashistha
Indian Journal of Hematology and Blood Transfusion.2015; 31(2): 307. CrossRef - Epstein-Barr virus–associated inflammatory pseudotumor presenting as a colonic mass
Shunyou Gong, Iwona Auer, Rajan Duggal, Stefania Pittaluga, Mark Raffeld, Elaine S. Jaffe
Human Pathology.2015; 46(12): 1956. CrossRef - Response of follicular dendritic cell sarcoma to gemcitabine and docetaxel: report of two cases and literature review
Robert M Conry
Clinical Sarcoma Research.2014;[Epub] CrossRef
 PubReader
PubReader ePub Link
ePub Link-
 Cite this Article
Cite this Article
- Cite this Article
-
- Close
- Download Citation
- Close
- Figure


Fig. 1
Fig. 2
| Case No. | Sex/Age (yr) | Site | Size (cm) | Immunohistochemistry |
EBER | Tx | Outcome (yr) | Reference | |||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| CD21 | CD23 | CD35 | CNA.42 | Other | LMP1 | ||||||||
| 1 | M/70 | Spleen | 5.5 | ND | ND | ND | ND | FDC+ | ND | + | Surgery | NR | Arber et al. [7] |
| SMA- | |||||||||||||
| 2 | F/68 | Liver | 12 | + | ND | + | ND | R4/23+ | + | + | Surgery | NR | Selves et al. [8] |
| CT | |||||||||||||
| 3 | F/35 | Liver | 20 | + | - | + | ND | R4/23+ | + | + | Surgery | Recurrence | Shek et al. [9] |
| SMA+ | NED, 2.5 | ||||||||||||
| 4 | M/37 | Liver | 15 | + | + | + | ND | ND | + | Surgery | NED, 2 | Shek et al. [10] | |
| 5 | F/19 | Liver | 12 | -/+ |
- | -/+ |
+ | HHF-35+ | +/- | + | Surgery | NED, 3.5 | Cheuk et al. [3] |
| 6 | F/56 | Liver | 15 | + |
+ | + |
+ | HHF-35- | + | + | Surgery | Recurrence, alive | Cheuk et al. [3] |
| with disease, 4.5 | |||||||||||||
| 7 | F/40 | Liver | 12.5 | -/+ |
- | -/+ |
+ | HHF-35- | + | + | Surgery | Recurrence, 9 | Cheuk et al. [3] |
| 8 | F/49 | Liver | 4.2 | + |
+ | + |
+ | +/- | + | Surgery | NED, 0.75 | Cheuk et al. [3] | |
| 9 | M/37 | Liver | 15 | + |
+/- | + |
+ | HHF-35+/- | - | + | Surgery | NED, 3.5 | Cheuk et al. [3] |
| 10 | F/35 | Liver | 20 | +* | + | +* | + | HHF-35+/- | +/- | + | Surgery | Recurrence | Cheuk et al. [3] |
| DOD, 7.9 | |||||||||||||
| 11 | F/31 | Liver | 15 | -/+ |
- | -/+ |
+ | HHF-35+/- | - | + | Surgery | NED, 5 | Cheuk et al. [3] |
| 12 | F/58 | Spleen | 22 | + |
- | + |
+ | +/- | + | Surgery | NED, 0.3 | Cheuk et al. [3] | |
| 13 | F/39 | Spleen | 7.5 | + |
+ | + |
+ | + | + | Surgery | NED, 0.2 | Cheuk et al. [3] | |
| 14 | F/61 | Spleen | 3.5 | + |
+ | + |
+ | HHF-35+/- | +/- | + | Surgery | NR | Cheuk et al. [3] |
| 15 | F/49 | Peripancreas | 9.5 | + |
+ | + |
+ | HHF-35- | +/- | + | Surgery | NR | Cheuk et al. [3] |
| 16 | F/51 | Liver | 15 | + | + | - | ND | ND | + | Surgery | NED, 3 | Chen et al. [11] | |
| 17 | F/57 | Liver | 12 | + | + | - | ND | ND | + | Surgery | NED, 1 | Chen et al. [11] | |
| 18 | M/54 | Spleen | 12 | + | + | ND | ND | SMA+ | +/- | + | Surgery | NED, 4 | Brittig et al. [12] |
| 19 | F/77 | Spleen | 11 | ND | ND | ND | + | + | + | Surgery | NED, 3 | Horiguchi et al. [13] | |
| 20 | F/30 | Liver | 5.5 | + | ND | + | ND | ND | + | Surgery | NED, 2 | Bai et al. [14] | |
| 21 | F/50 | Spleen | 5 | + | ND | + | + | SMA+ | ND | + | Surgery | NED, 0.3 | Laurent et al. [15] |
| 22 | F/57 | Liver | 13 | + | + | ND | ND | ND | + | Surgery | NED, 2 | Granados et al. [16] | |
| 23 | M/45 | Liver | 14.5 | + | + | - | ND | ND | + | Surgery | NED | Li et al. [5] | |
| 24 | F/78 | Colon | 3.9 | + | + | + | ND | ND | + | Polypectomy | NED, 0.5 | Present case | |
| Subtotal (%) | NA | NA | 11/11 (100) | 13/18 (72) | 6/9 (67) | 13/13 (100) | 6/8 (75) | 24/24 (100) | NA | NA | NA | ||
NACase 1-3 and 11 are reported as inflammatory pseudotumor. IPT, inflammatory pseudotumor; FDC, follicular dendritic cell; LMP-1, latent membrane protein 1; EBER, Epstein-Barr virus-encoded mRNA; Tx, treatment; M, male; ND, not done; SMA, smooth muscle actin; NR, not recorded; F, female; CT, chemotherapy; NED, no evidence of disease; DOD, die of disease; NA, not applicable. Staining results from using combined CD21 and CD35 antibodies. These cases are not included in the calculation for the percentage of markers expressed by IPT-like FDC sarcomas.

 E-submission
E-submission