Articles
- Page Path
- HOME > J Pathol Transl Med > Volume 56(6); 2022 > Article
-
Original Article
A clinicopathologic and immunohistochemical study of primary and secondary breast angiosarcoma -
Evi Abada1
 , Hyejeong Jang2
, Hyejeong Jang2 , Seongho Kim2
, Seongho Kim2 , Rouba Ali-Fehmi1, Sudeshna Bandyopadhyay1
, Rouba Ali-Fehmi1, Sudeshna Bandyopadhyay1 -
Journal of Pathology and Translational Medicine 2022;56(6):342-353.
DOI: https://doi.org/10.4132/jptm.2022.08.31
Published online: October 27, 2022
1Department of Pathology, Wayne State University School of Medicine/Detroit Medical Center, Detroit, MI, USA
2Biostatistics and Bioinformatics Core, Karmanos Cancer Institute, Department of Oncology, Wayne State University School of Medicine, Detroit, MI, USA
- Corresponding Author: Evi Abada, MD, MS, Department of Pathology, Wayne State University School of Medicine/Detroit Medical Center, 3990 John R Street, Detroit, MI 48201, USA Tel: +1-313-577-1102, Fax: +1-313-577-0057, E-mail: gs5839@wayne.edu
© 2022 The Korean Society of Pathologists/The Korean Society for Cytopathology
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/4.0) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
Figure & Data
References
Citations

- Prognostic significance of clinicopathological parameters, margin width and locoregional recurrences on outcome of primary and radiation associated breast angiosarcoma- results from a large UK sarcoma regional service
Samar Ali, Salena Bains, Emily Fox, Anant Desai, Mike Hallissey, Alaa El-Ghobashy, Robert Warner, Abeer M. Shaaban
Breast Cancer Research.2026;[Epub] CrossRef - Angiosarcoma: a systematic review of biomarkers in diagnosis, prognosis, and therapeutic strategies
Huyen Thuc Tran Luong, Sofie Vercammen, Ario de Marco, Hilde de Rooster, Antonio Cosma
Frontiers in Oncology.2025;[Epub] CrossRef - Etiology, pathogenesis, and management of angiosarcoma associated with implants and foreign body: Clinical cases and research updates
Ramy Samargandi
Medicine.2024; 103(18): e37932. CrossRef - Ovarian angiosarcoma: A systematic review of literature and survival analysis
Shafi Rehman, Arya Harikrishna, Amisha Silwal, B.R. Sumie, Safdar Mohamed, Nisha Kolhe, Meghana Maddi, Linh Huynh, Jesus Gutierrez, Yoshita Rao Annepu, Ameer Mustafa Farrukh
Annals of Diagnostic Pathology.2024; 73: 152331. CrossRef - Neoadjuvant chemotherapy for radiation associated angiosarcoma (RAAS) of the breast: A retrospective single center study
Stijn J.C. van der Burg, Sophie J.M. Reijers, Anke Kuijpers, Lotte Heimans, Astrid N. Scholten, Rick L.M. Haas, Hester van Boven, Willemijn M. Kolff, Marie-Jeanne T.F.D. Vrancken Peeters, Martijn Kerst, Beatrijs A. Seinstra, Neeltje Steeghs, Winette T.A.
The Breast.2024; 78: 103825. CrossRef - Lymph node involvement in secondary breast angiosarcoma – a case presentation
Adriana Irina Ciuvică, Tiberiu Augustin Georgescu , Andrei Dennis Voichiţoiu , Angela Arsene , Luchian Marinescu , George Ionuţ Bucur , Livia Iordache , Nahedd Saba
Romanian Journal of Morphology and Embryology.2024; 65(3): 523. CrossRef - Primary ovarian angiosarcoma: Two case reports and review of literature
Ying Zhou, Yi-Wen Sun, Xiao-Yang Liu, Dan-Hua Shen
World Journal of Clinical Cases.2023; 11(21): 5122. CrossRef
 PubReader
PubReader ePub Link
ePub Link-
 Cite this Article
Cite this Article
- Cite this Article
-
- Close
- Download Citation
- Close
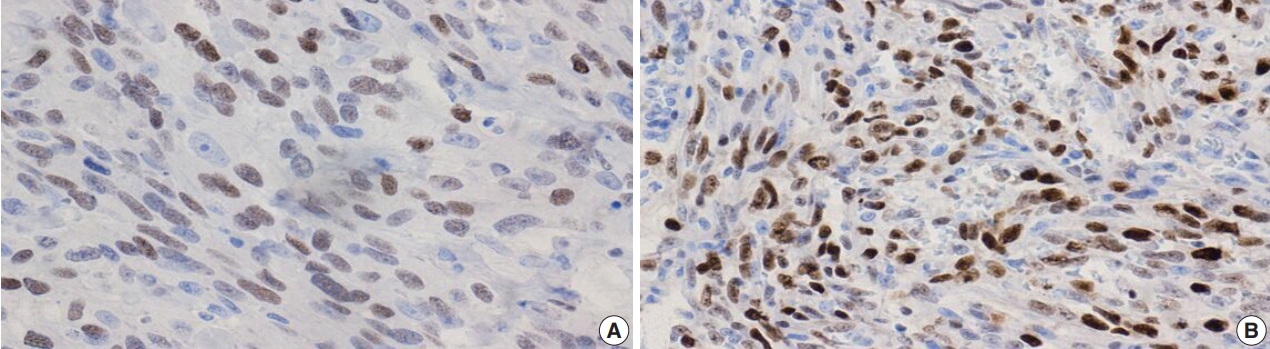
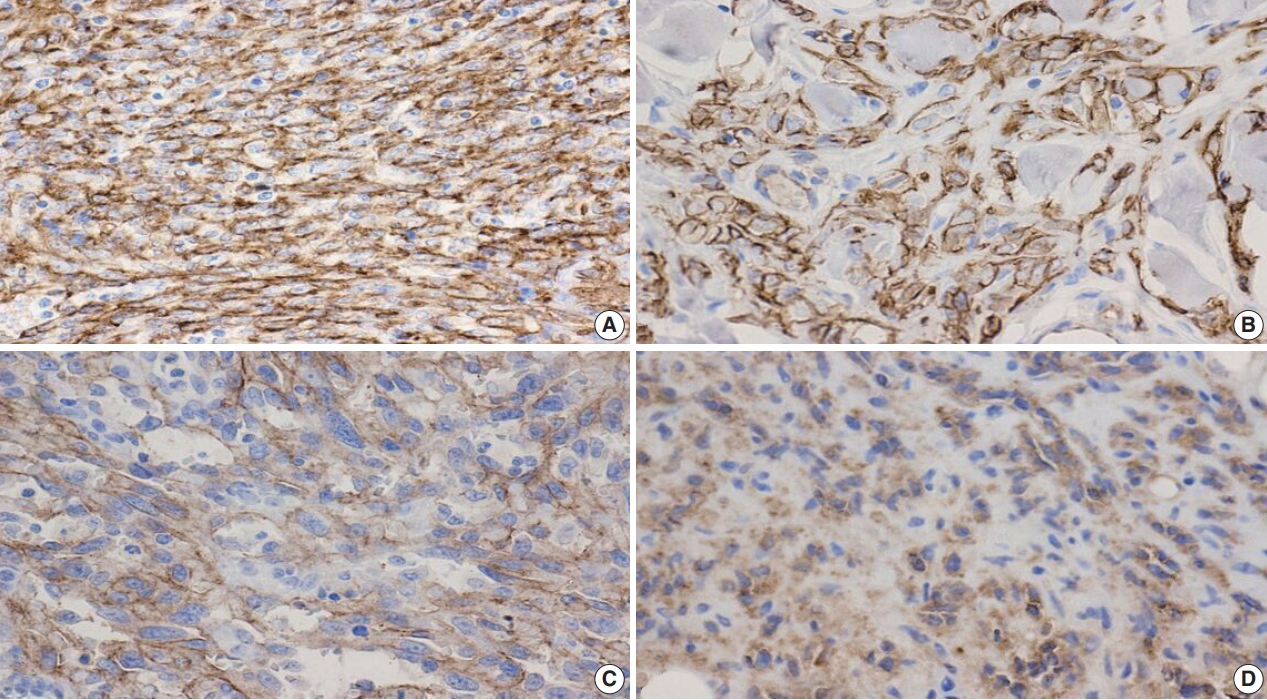
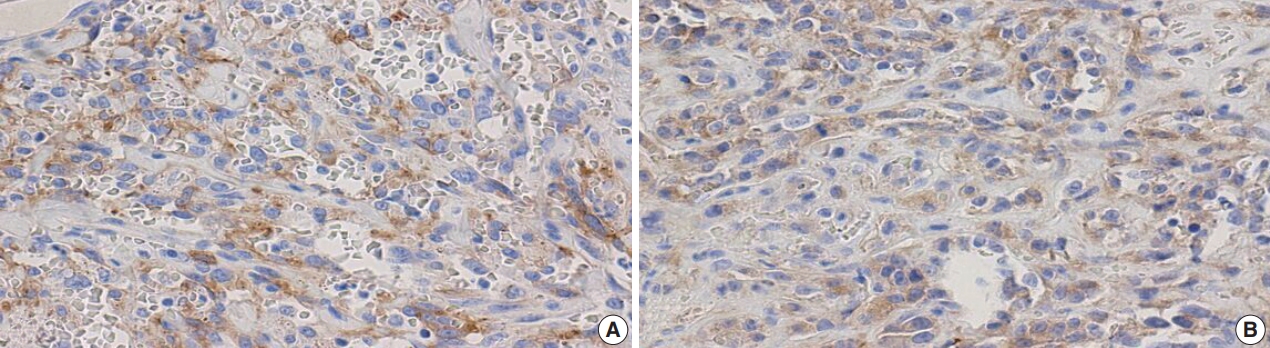
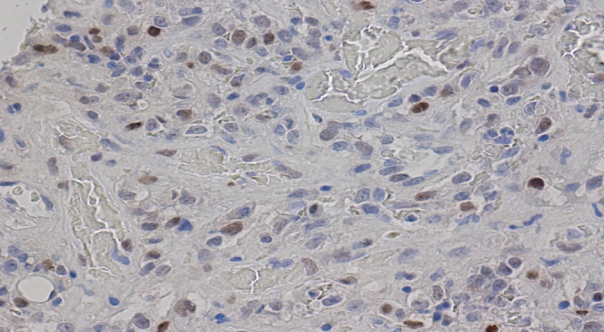
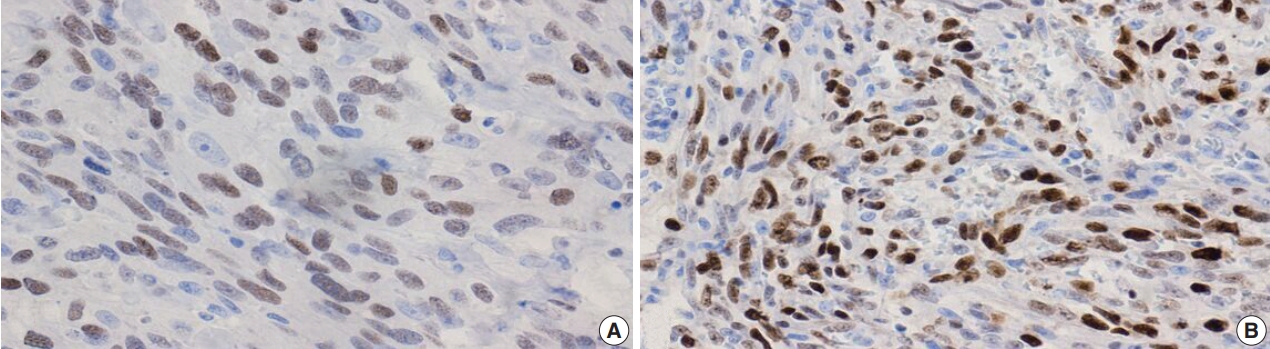
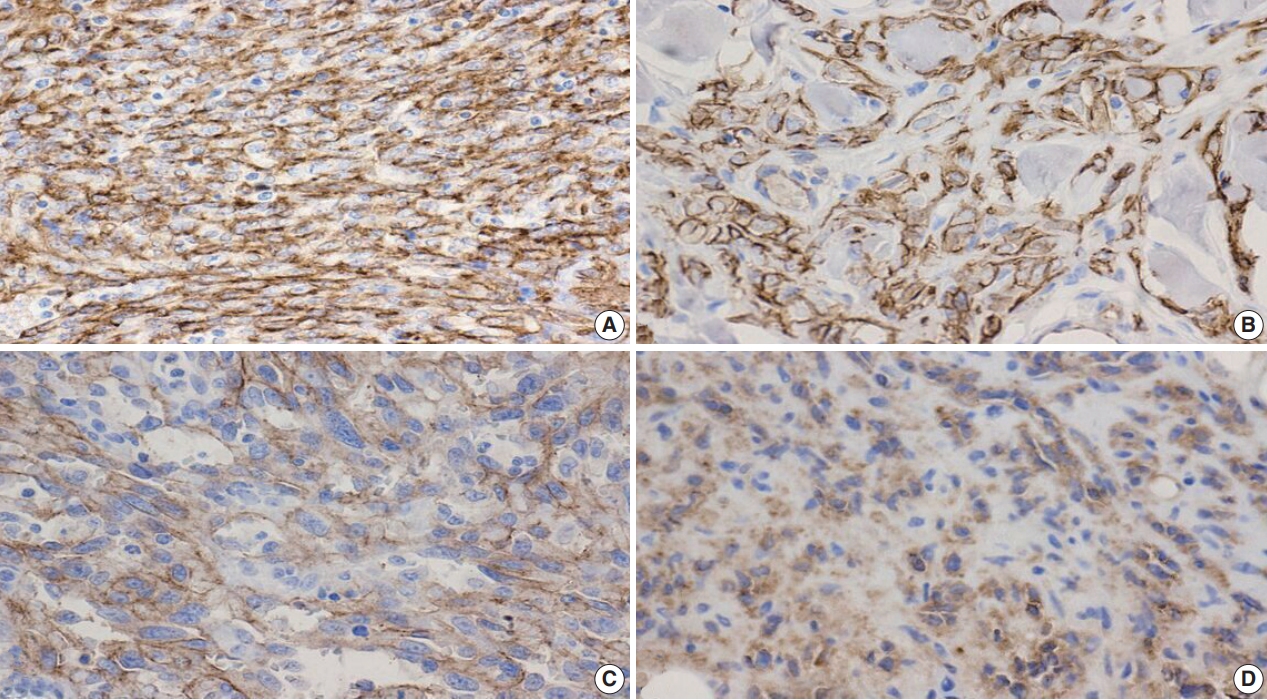
- Figure















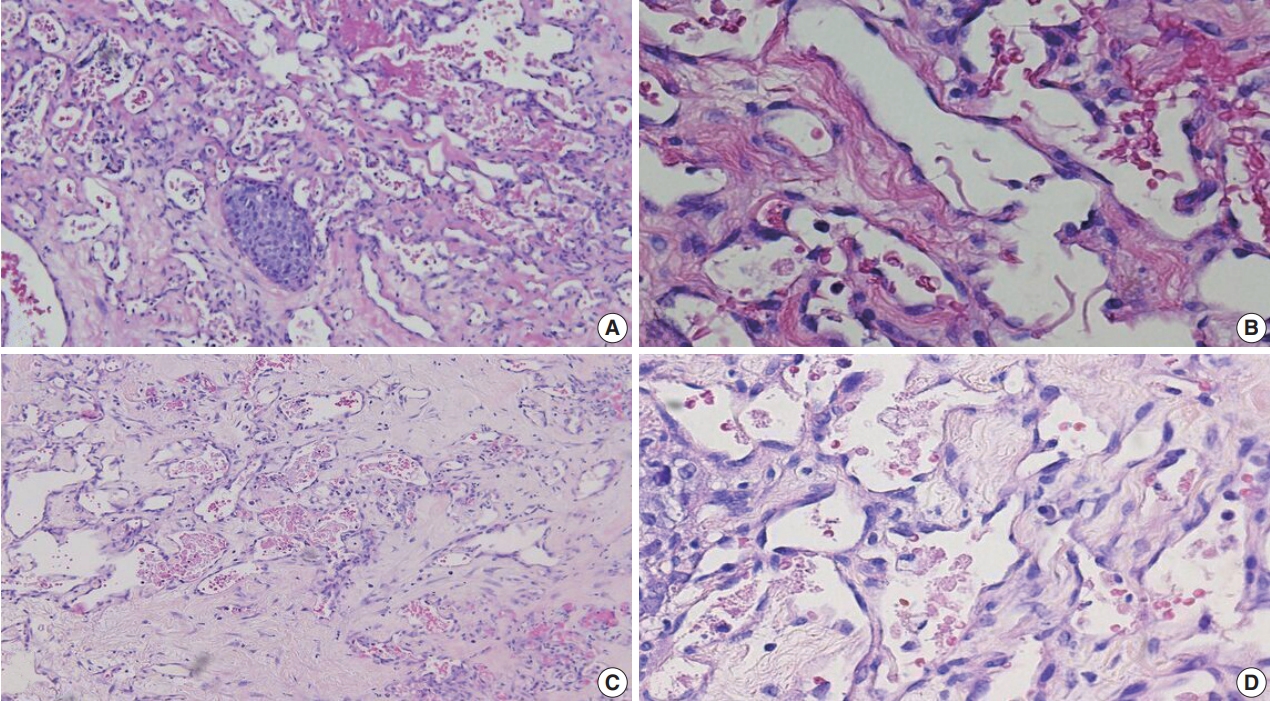
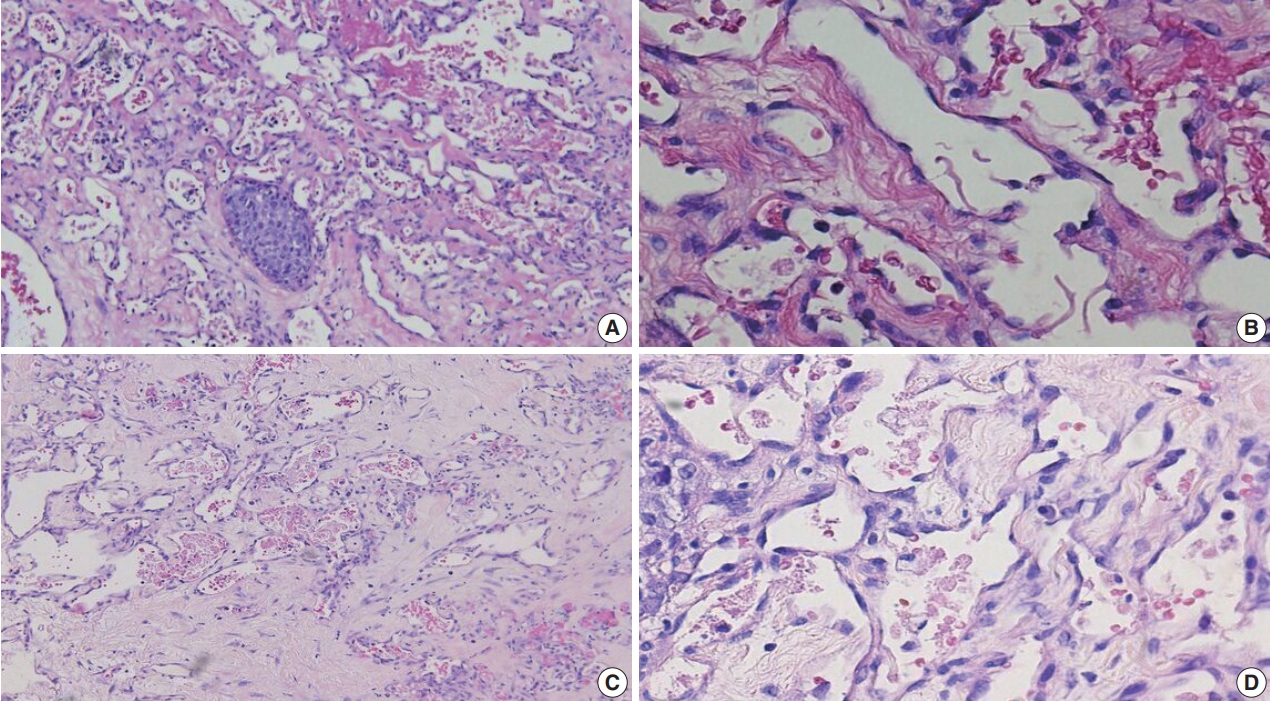
Fig. 1.
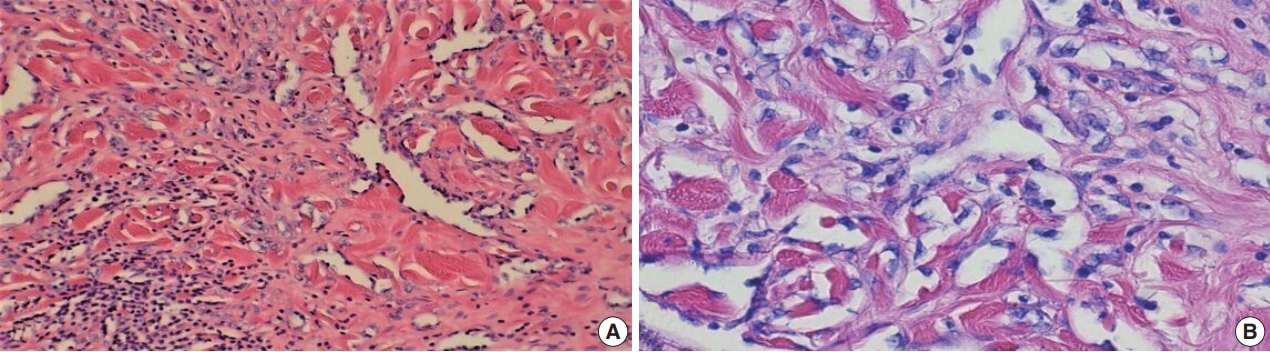
Fig. 2.
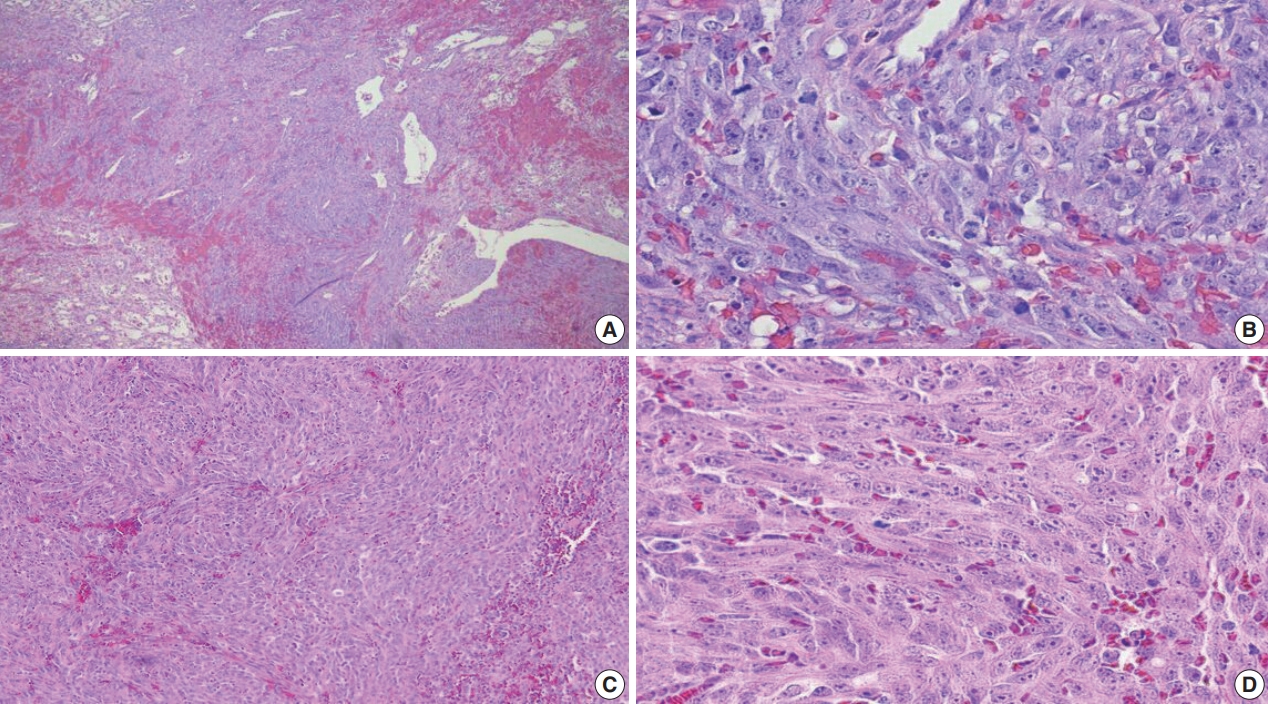
Fig. 3.
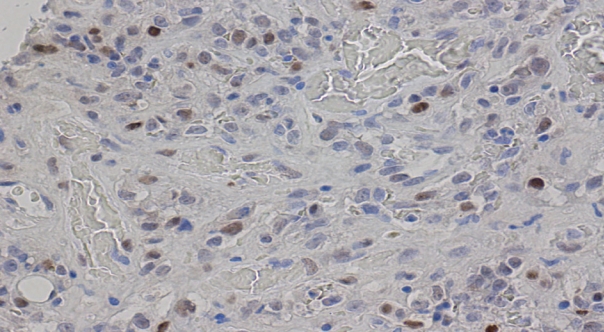
Fig. 4.
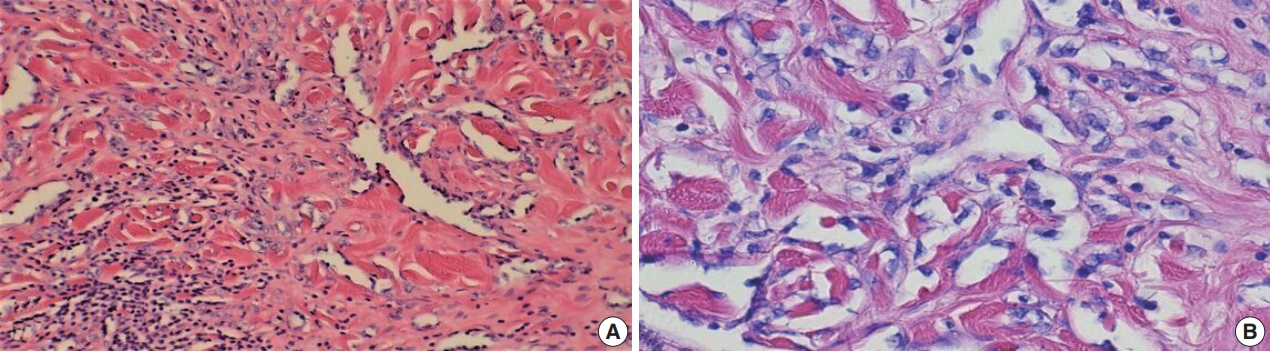
Fig. 5.
Fig. 6.
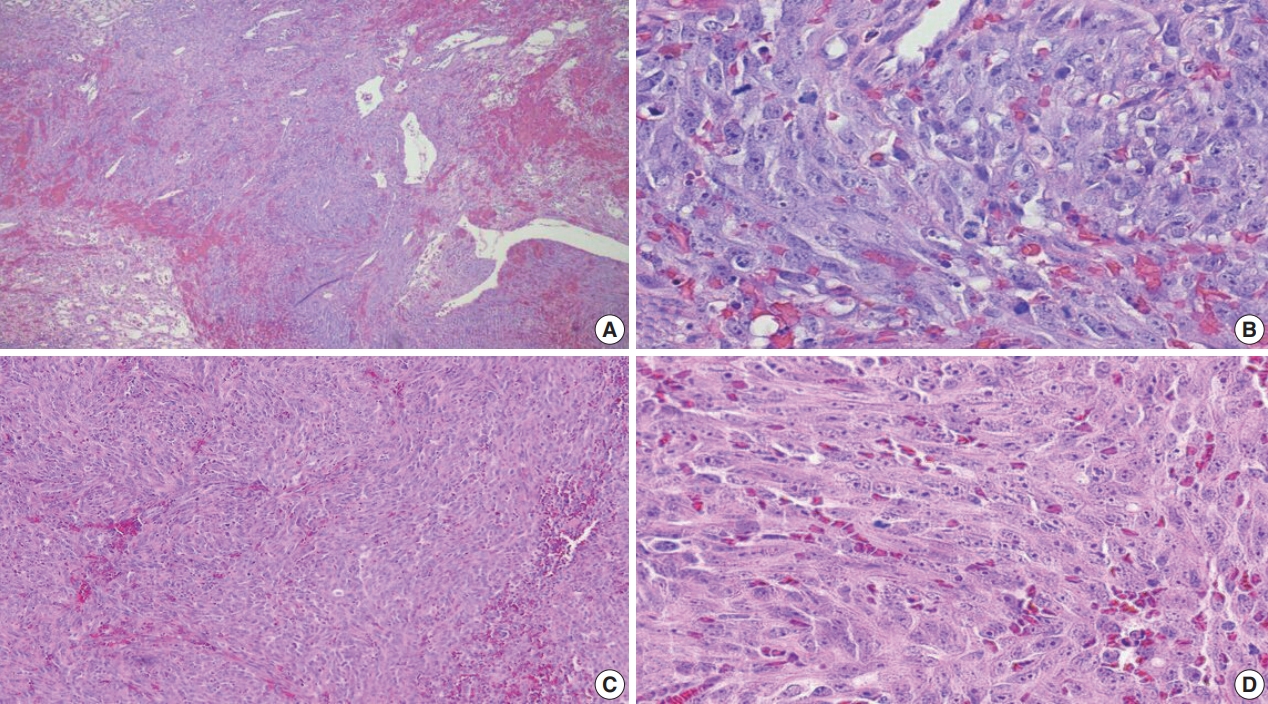
Fig. 7.
Fig. 8.
Fig. 9.
Fig. 10.
Fig. 11.
Fig. 12.
Fig. 13.
Fig. 14.
Fig. 15.
| Antibody | Clone | Dilution | Vendor | Retrieval (HIER) (min) | Antibody incubation (min) |
|---|---|---|---|---|---|
| c-kit (CD117) | Rabbit polyclonal | 1:250 | Dako | CC1: 36 | 60 |
| p53 | DO-7 mouse monoclonal | RTU | Ventana Medical Systems | CC1: 36 | 32 |
| c-Myc | Y69 (rabbit) | RTU | Ventana Medical Systems | CC1: 64 | 32 |
| CD31 | JC70 (mouse) | RTU | Cell Marque | CC1: 36 | 40 |
| D2-40 (podoplanin) | D2-40 (mouse) | RTU | Cell Marque | CC1: 36 | 32 |
| Variable | Total (n = 11) | Primary BAS (n = 3) | Secondary BAS (n = 8) | p-value |
|---|---|---|---|---|
| CD117 | > .99 | |||
| Positive | 7 (63.6) | 2 (66.7) | 5 (62.5) | |
| Negative | 4 (36.4) | 1 (33.3) | 3 (37.5) | |
| p53 | .236 | |||
| Positive | 4 (36.4) | 0 | 4 (50.0) | |
| Negative | 7 (63.6) | 3 (100) | 4 (50.0) | |
| D-240 | > .99 | |||
| Positive | 7 (63.6) | 2 (66.7) | 5 (62.5) | |
| Negative | 4 (36.4) | 1 (33.3) | 3 (37.5) | |
| CD31 | > .99 | |||
| Positive | 11 (100) | 3 (100) | 8 (100) | |
| Negative | 0 | 0 | 0 | |
| C-MYC | > .99 | |||
| Positive | 8 (72.7) | 2 (66.7) | 6 (75.0) | |
| Negative | 3 (27.3) | 1 (33.3) | 2 (25.0) |
| Variable | Total (n = 17) | Primary BAS (n = 5) | Secondary BAS (n = 12) | p-value |
|---|---|---|---|---|
| Age at diagnosis (yr) | 66 (23–76) | 36 (23–71) | 67 (33–76) | .246 |
| Race | > .99 | |||
| Caucasian | 12 (70.6) | 4 (80.0) | 8 (66.7) | |
| African American | 5 (29.4) | 1 (20.0) | 4 (33.3) | |
| Tumor size (cm) | 1.1 (0.4–28) | 2 (0.5–28) | 0.95 (0.4–2.5) | .437 |
| Missing | 8 | 2 | 6 | |
| Tumor grade | .087 | |||
| Low | 6 (35.3) | 2 (40.0) | 4 (33.3) | |
| Intermediate | 2 (11.8) | 2 (40.0) | 0 | |
| High | 9 (52.9) | 1 (20.0) | 8 (66.7) | |
| Tumor necrosis | .538 | |||
| Yes | 4 (23.5) | 2 (40.0) | 2 (16.7) | |
| No | 13 (76.5) | 3 (60.0) | 10 (83.3) | |
| Mitotic count | .593 | |||
| > 10/10 HPF | 7 (41.2) | 3 (60.0) | 4 (33.3) | |
| < 10/10 HPF | 10 (58.8) | 2 (40.0) | 8 (66.7) | |
| Lymph node metastasis | .191 | |||
| Yes | 3 (17.6) | 2 (40.0) | 1 (8.3) | |
| No | 14 (82.4) | 3 (60.0) | 11 (91.7) | |
| Positive margins | > .99 | |||
| Yes | 8 (47.1) | 2 (40.0) | 6 (50.0) | |
| No | 9 (52.9) | 3 (60.0) | 6 (50.0) | |
| Tumor site | .294 | |||
| Right | 9 (52.9) | 4 (80.0) | 5 (41.7) | |
| Left | 8 (47.1) | 1 (20.0) | 7 (58.3) | |
| Multifocal tumors | > .99 | |||
| Yes | 4 (23.5) | 1 (20.0) | 3 (25.0) | |
| No | 13 (76.5) | 4 (80.0) | 9 (75.0) | |
| Skin involved | .044 | |||
| Yes | 7 (41.2) | 0 | 7 (58.3) | |
| No | 10 (58.8) | 5 (100) | 5 (41.7) | |
| Obesity | .117 | |||
| Yes | 12 (70.6) | 2 (40.0) | 10 (83.3) | |
| No | 5 (29.4) | 3 (60.0) | 2 (16.7) | |
| Hypertension | .102 | |||
| Yes | 6 (35.3) | 0 | 6 (50.0) | |
| No | 11 (64.7) | 5 (100) | 6 (50.0) | |
| Smoking | > .99 | |||
| Yes | 4 (23.5) | 1 (20.0) | 3 (25.0) | |
| No | 13 (76.5) | 4 (80.0) | 9 (75.0) | |
| Diabetes | .338 | |||
| Yes | 7 (41.2) | 1 (20.0) | 6 (50.0) | |
| No | 10 (58.8) | 4 (80.0) | 6 (50.0) | |
| Treatment received | .353 | |||
| Surgery only | 14 (82.4) | 4 (80.0) | 10 (83.3) | |
| Surgery + chemotherapy | 2 (11.8) | 0 | 2 (16.7) | |
| Surgery + radiation + chemotherapy | 1 (5.9) | 1 (20.0) | 0 |
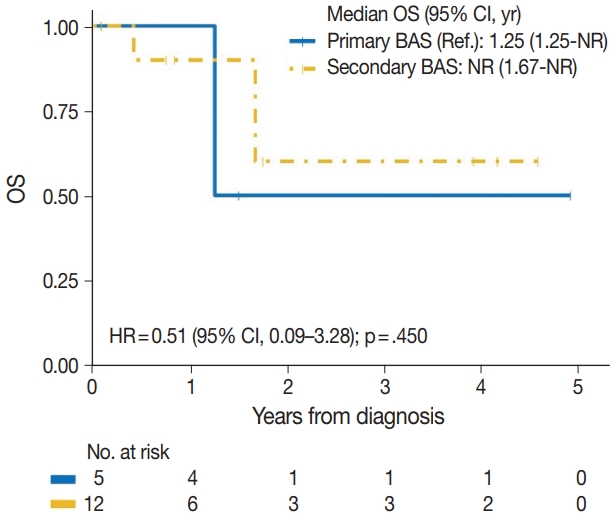
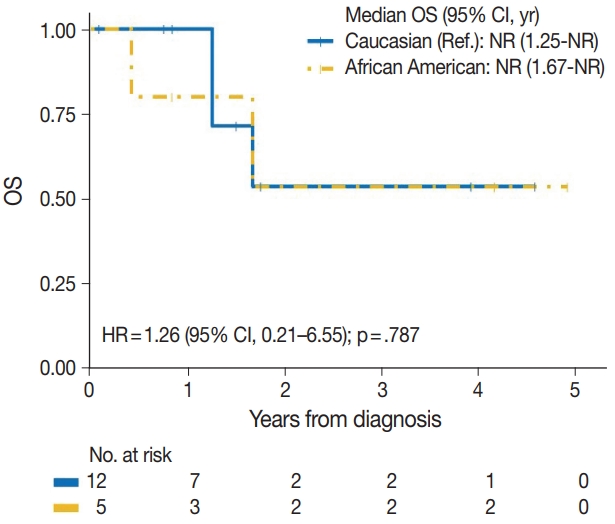
| Variable | Event/No. | HR (95% CI) | p-value |
|---|---|---|---|
| Age at diagnosis | 5/17 | 1.01 (0.96–1.07) | .845 |
| Race | |||
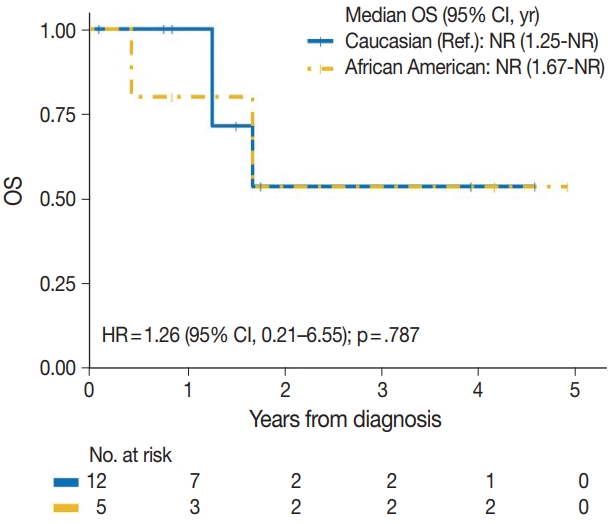
| Caucasian | 3/12 | Reference | |
| African American | 2/5 | 1.26 (0.21–6.55) | .787 |
| BAS type | |||
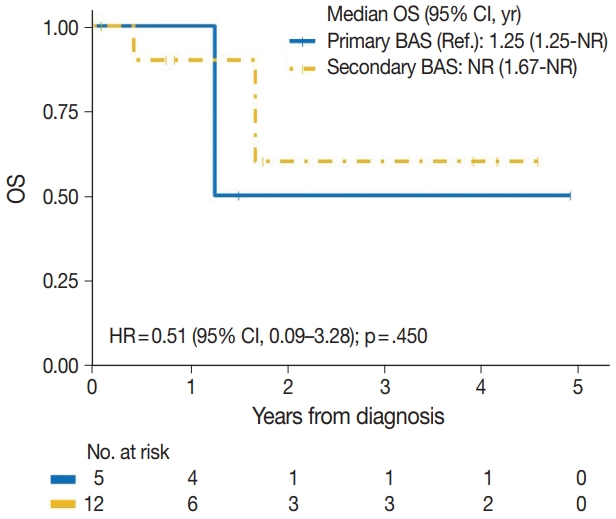
| Primary BAS | 2/5 | Reference | |
| Secondary BAS | 3/12 | 0.51 (0.09–3.28) | .450 |
| Tumor size (cm) | 3/9 | 1.04 (0.95–1.13) | .307 |
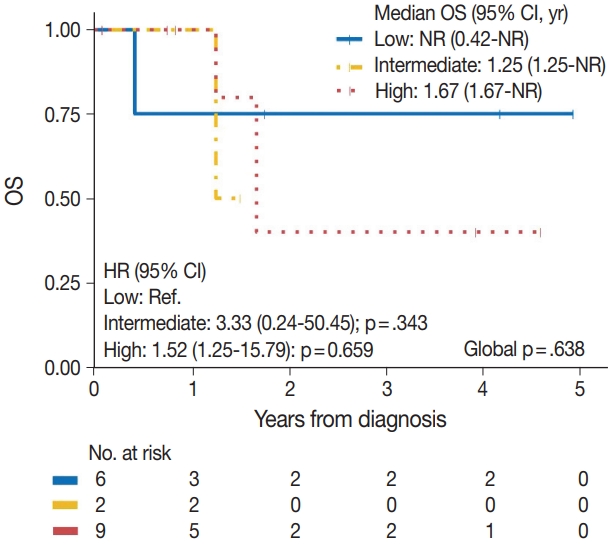
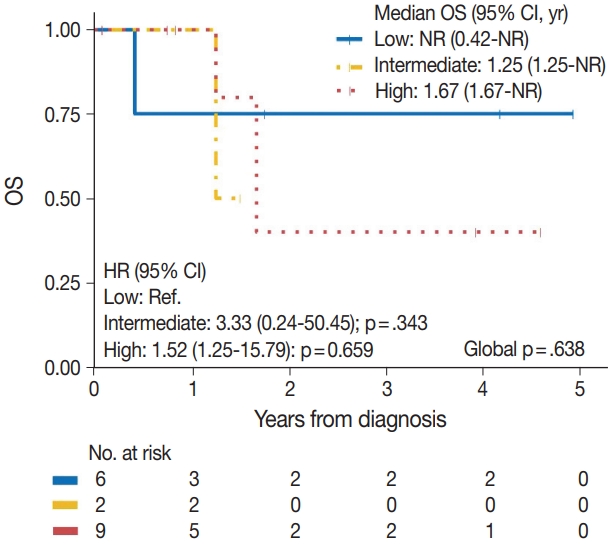
| Tumor grade | .638 |
||
| Low | 1/6 | Reference | |
| Intermediate | 1/2 | 3.33 (0.24–50.45) | .343 |
| High | 3/9 | 1.52 (0.25–15.79) | .659 |
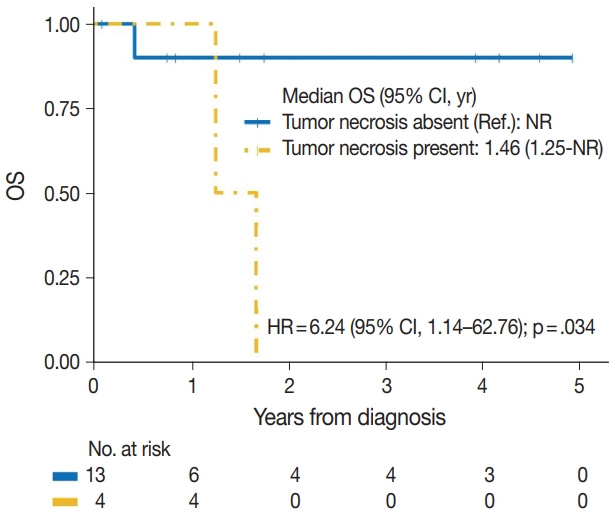
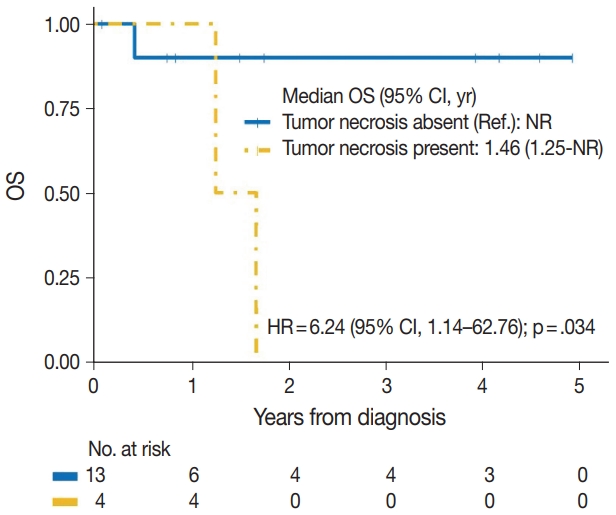
| Tumor necrosis | |||
| No | 1/13 | Reference | |
| Yes | 4/4 | 6.24 (1.14–62.76) | .034 |
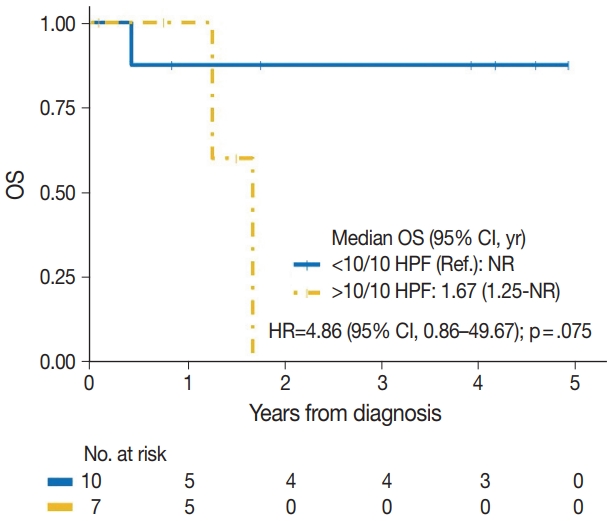
| Mitotic count | |||
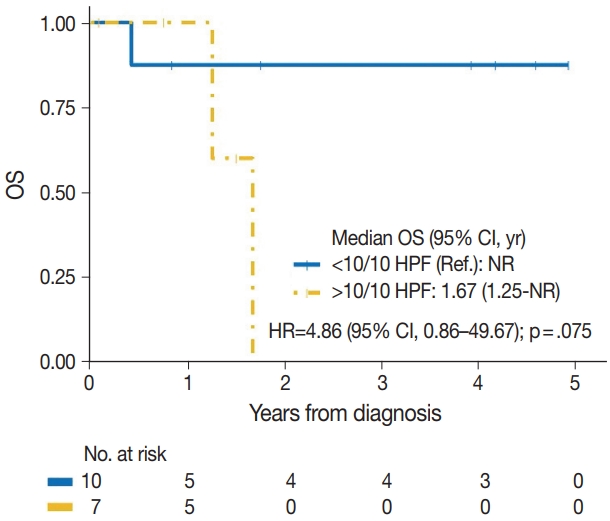
| < 10/10 HPF | 1/10 | Reference | |
| > 10/10 HPF | 4/7 | 4.86 (0.86–49.67) | .075 |
| Lymph node metastasis | |||
| No | 3/14 | Reference | |
| Yes | 2/3 | 2.61 (0.42–14.00) | .278 |
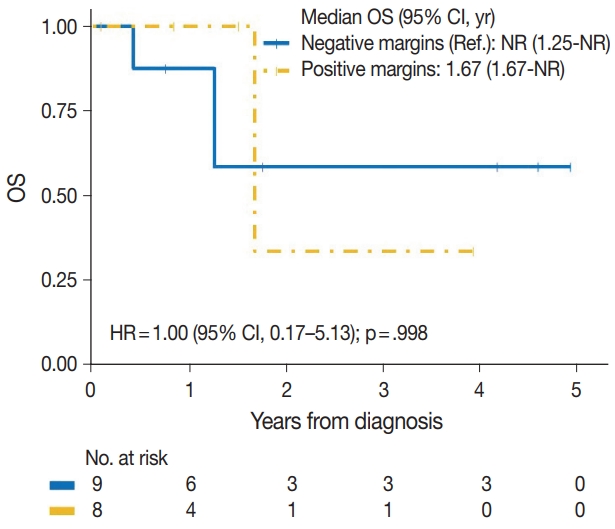
| Positive margins | |||
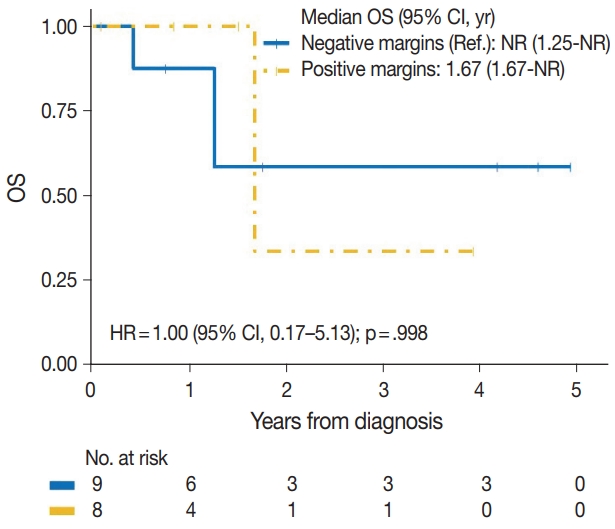
| No | 3/9 | Reference | |
| Yes | 2/8 | 1.00 (0.17–5.13) | .998 |
| Tumor site | |||
| Left | 2/8 | Reference | |
| Right | 3/9 | 1.35 (0.26–8.26) | .719 |
| Multifocal tumors | |||
| No | 5/13 | Reference | |
| Yes | 0/4 | 0.37 (0.003–3.32) | .439 |
| Skin involved | |||
| No | 3/10 | Reference | |
| Yes | 2/7 | 0.78 (0.13–4.14) | .773 |
| Obesity | |||
| No | 3/5 | Reference | |
| Yes | 2/12 | 0.20 (0.03–1.10) | .063 |
| Hypertension | |||
| No | 4/11 | Reference | |
| Yes | 1/6 | 0.99 (0.10–5.40) | .990 |
| Smoking | |||
| No | 5/13 | Reference | |
| Yes | 0/4 | 0.45 (0.003–4.04) | .551 |
| Diabetes | |||
| No | 4/10 | Reference | |
| Yes | 1/7 | 0.58 (0.06–3.17) | .548 |
| Treatment received | |||
| Surgery only | 4/14 | Reference | |
| Surgery + chemotherapy/radiotherapy | 1/3 | 1.52 (0.15–8.29) | .671 |
| CD117 | |||
| Negative | 1/4 | Reference | |
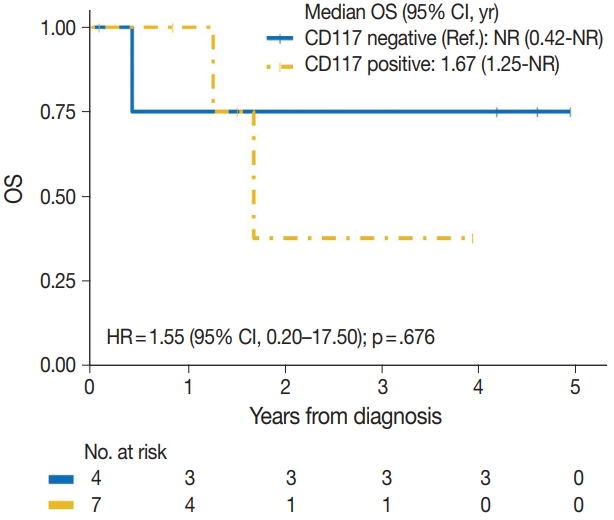
| Positive | 2/7 | 1.55 (0.20–17.50) | .676 |
| p53 | |||
| Negative | 2/7 | Reference | |
| Positive | 1/4 | 1.23 (0.11–9.37) | .847 |
| D2-40 | |||
| Negative | 1/4 | Reference | |
| Positive | 2/7 | 0.95 (0.12–10.49) | .960 |
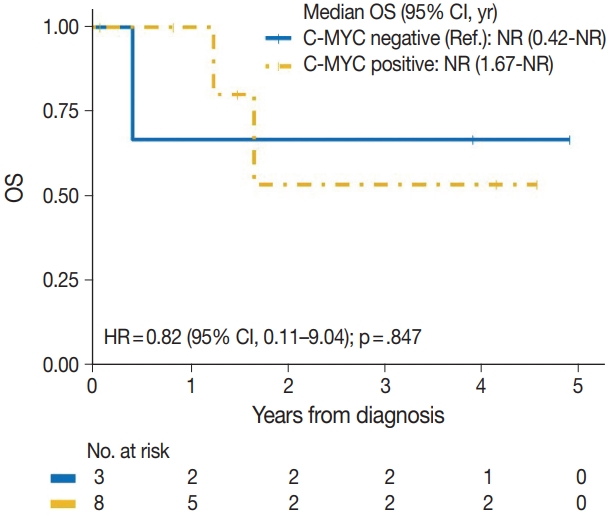
| c-Myc | |||
| Negative | 1/3 | Reference | |
| Positive | 2/8 | 0.82 (0.11–9.04) | .847 |
HIER, heat-induced epitope retrieval; RTU, ready to use reagent. CC1 is Ventana Medical Systems retrieval solution, RTU, at pH 8.0.
BAS, breast angiosarcoma. Fisher exact test.
Values are presented as median (range) or number (%). BAS, breast angiosarcoma; HPF, high-power field. Fisher exact test or Wilcox rank-sum test as appropriate.
Event/n, the number of events and patients; HR, hazard ratio; CI, confidence interval; BAS, breast angiosarcoma; HPF, high-power field. Global p-value calculated by likelihood ratio test.

 E-submission
E-submission