Articles
- Page Path
- HOME > J Pathol Transl Med > Volume 47(3); 2013 > Article
-
Case Study
Colonic Adenocarcinoma Arising from Gastric Heterotopia: A Case Study - Hyoungsuk Ko, Shin Young Park, Eun Jung Cha, Jang Sihn Sohn
-
Korean Journal of Pathology 2013;47(3):289-292.
DOI: https://doi.org/10.4132/KoreanJPathol.2013.47.3.289
Published online: June 25, 2013
Department of Pathology, Konyang University School of Medicine, Daejeon, Korea.
- Corresponding Author: Jang Sihn Sohn, M.D. Department of Pathology, Konyang University School of Medicine, 158 Gwanjeodong-ro, Seo-gu, Daejeon 302-718, Korea. Tel: +82-42-600-9283, Fax: +82-42-600-9280, jsohn@kyuh.ac.kr
• Received: July 11, 2012 • Revised: September 7, 2012 • Accepted: September 11, 2012
© 2013 The Korean Society of Pathologists/The Korean Society for Cytopathology
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/3.0/) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
Abstract
- Heterotopic gastric mucosa occurs in all areas of the gastrointestinal tract including the nasopharynx, tongue, esophagus, small intestine, colon, and rectum. Gastric heterotopia of the large bowel is infrequent, and most cases have been reported in the rectum. Review of the literature has revealed only eight cases involving the colon proximal to the rectum. Little is known of the natural history of gastric heterotopias, except that. It usually presents with gastrointestinal bleeding, though other serious complications such as bowel perforation, intussusceptions, and fistula formation, are possible. Further, it is unclear whether heterotopic gastric mucosa progresses to malignancy. Herein, we describe a case of adenocarcinoma of the transverse colon arising from gastric heterotopia. To the best of our knowledge, this is the first report of adenocarcinoma arising from heterotopic gastric mucosa in the colon.
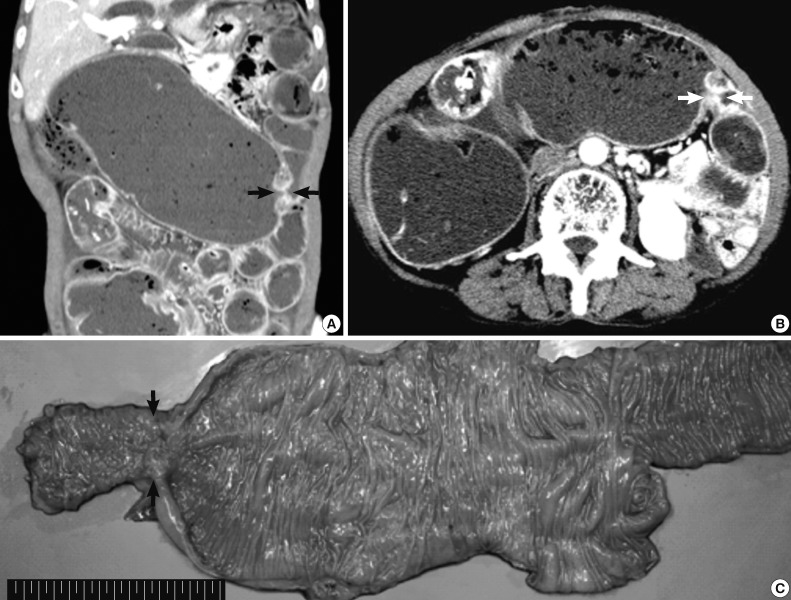
- A 70-year-old female patient was hospitalized because of known cholangitis. She had previously undergone cholecystectomy because of the cholelithiasis. Physical examination revealed tenderness of the lower quadrants of the abdomen. As part of the clinical work-up, the patient underwent an abdominal computed tomography, which demonstrated colonic distension and small bowel ileus with focal narrowing and obstruction of the transverse colon (Fig. 1A, B). Mechanical obstructive ileus was suspected, and a colonoscopy revealed luminal erosion and stricture of the transverse colon. Microscopic examination of multiple blind biopsies showed erosion and vascular congestion with no evidence of malignancy. A whole body scan to evaluate the presence of metastatic lesions did not show any abnormal fluorodeoxyglucose uptake. The patient was diagnosed with colonic malignancy, and right hemicolectomy was performed.
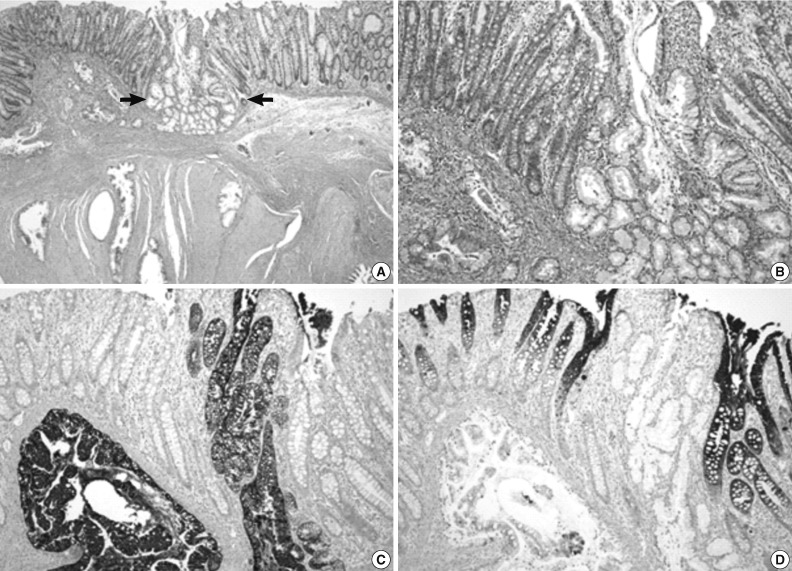
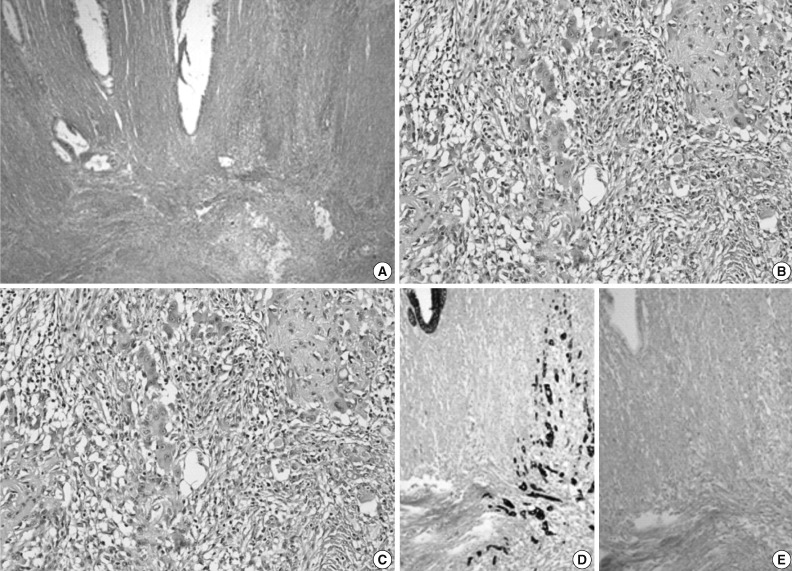
- On gross examination, the resected specimen displayed infiltrative submucosal lesions that involved the whole circumference and resulted in colonic stricture (Fig. 1C). Histologically, the colonic mucosa was intact except for focal erosion, and the muscular layer was markedly hypertrophied at the stricture site. Further microscopic examination revealed that a benign glandular structure of gastric origin invaded the erosive colonic mucosa. These glands showed a downward growth pattern toward the submucosa and the muscular layer, gastric differentiation, and focal cystic dilatation (Fig. 2A). Additionally, we identified an adenocarcinoma that originated in the heterotopic gastric glands in the submucosa and muscular layers. The adenocarcinoma foci were composed of glands as well as isolated tumor cells and clusters with focal mucin production and were moderately to poorly differentiated (Fig. 3A). The tumor was distributed through the colonic wall and pericolic soft tissue. Immunohistochemically, both the heterotopic glands and adenocarcinoma showed positive results for cytokeratin (CK) 7 and MUC5AC and were negative for CK20, CDX2, and MUC2 (Figs. 2, 3). On the basis of these findings, heterotopic gastric mucosa in the transverse colon and development of invasive adenocarcinoma were diagnosed and staged at pT3, pN1b (2/43), pMx according to the 7th edition of the Cancer Staging Manual of the American Joint Committee on Cancer.
CASE REPORT
- Heterotopic gastric mucosa has been identified throughout the gastrointestinal tract, more commonly in the cervical esophagus, duodenum, or Meckel's diverticulum.5,6 Although the etiology of gastric heterotopia is still unknown, it has been hypothesized that it could be either developmental or acquired. Depending on the location, three mechanisms have been suggested for the development of heterotopic gastric mucosa. One possibility is that remnant tissue could remain in the distal esophageal portion of the gut during the developmental descent of the stomach. Although this mechanism could explain gastric heterotopia of the esophagus, it cannot explain heterotopic gastric mucosa in more distal intestinal tracts.1 It has also been proposed that the condition may be acquired and is the result of an abnormal regenerative process following the destruction of normal intestinal mucosa. However, this theory is not supported by any descriptions of gastric heterotopia following destruction of gastrointestinal mucosa due to gastroenteritis or other inflammatory conditions.4 The third and most plausible hypothesis is that heterotopic gastric mucosa is the result of abnormal differentiation of local tissues. Pluripotent primitive endoderm stem cells have the ability to differentiate into all cell types of the gastrointestinal epithelium. An error in differentiation could lead to the gastric mucosa being present anywhere throughout the gastrointestinal tract.1,4
- Gastric heterotopia in the large bowel is fairy infrequent, and most cases have been reported in the rectum. Review of the literature has revealed only eight such cases, including the present one, involving the colon proximal to the rectum.1-3
- The risk and possibility of malignant transformation of heterotopic gastric mucosa have not been properly evaluated, although malignant change of heterotopic gastric mucosa has been reported for the esophagus, gallbladder, and adenomyosis of the stomach.4,6 We found only one report describing an association between gastric heterotopia in the colon and a premalignant tubulovillous adenoma.2 Moreover, two case reports of adenocarcinoma in the small bowel arising from heterotopic gas tric mucosa were published.6,7 One case described multifocal gastric heterotopia with gastritis cystic profunda in the ileum and an adenocarcinoma in one of the lesions.6 The other case documented jejunal adenocarcinoma in heterotopic gastric mucosa with variable grades of dysplasia.7
- In our case, features of both benign glandular structure with cystic dilatation and adenocarcinoma in the colonic wall were observed, raising the question of whether the adenocarcinoma was of gastric or intestinal origin. The observation that the cystically dilated glands and adenocarcinoma were CK7+/CK20-/CDX2-/MUC5AC+/MUC2- whereas the adjacent colonic mucosa was CK7-/CK20+/CDX2+/MUC5AC-/MUC2+ indicated that the heterotopic glands and carcinoma were of gastric origin.
- As mentioned previously, the risk of malignant transformation of heterotopic gastric mucosa is unknown. Moreover, no case reports have described the malignant transformation of heterotopic gastric mucosa of the colon. Sauer et al.4 proposed that the number of cases of gastric heterotopia that undergo malignant change may be underestimated because once the tumor grows, it obliterates the minute focus of heterotopic gastric mucosa.
- In conclusion, we report the first case of colonic adenocarcinoma arising from gastric heterotopia. Our findings suggest that heterotopic gastric mucosa of the colon has the potential of undergoing malignant transformation.
DISCUSSION
- 1. Murray FE, Lombard M, Dervan P, Fitzgerald RJ, Crowe J. Bleeding from multifocal heterotopic gastric mucosa in the colon controlled by an H2 antagonist. Gut 1988; 29: 848-851. ArticlePubMedPMC
- 2. Cappell MS. Colonic tubulovillous adenoma associated with gastric heterotopia. N J Med 1995; 92: 512-515. PubMed
- 3. Taylor FM 3rd, Swank RI 2nd. Epithelial heterotopia in the colon of a child: a case presentation and review of the literature. J Fla Med Assoc 1982; 69: 788-791. PubMed
- 4. Sauer CG, Bickston SJ, Borowitz SM. Gastric heterotopia of the rectum. J Pediatr Gastroenterol Nutr 2010; 50: 329-333. ArticlePubMed
- 5. Simon E, Petke D, Böger C, et al. The spatial distribution of LGR5+ cells correlates with gastric cancer progression. PLoS One 2012; 7: e35486.ArticlePubMedPMC
- 6. Schaefer IM, Schüler P, Enders C, et al. High chromosomal instability in adenocarcinoma of the ileum arising from multifocal gastric heterotopia with gastritis cystica profunda. Med Oncol 2011; 28: 1023-1026. ArticlePubMedPDF
- 7. Caruso ML, Marzullo F. Jejunal adenocarcinoma in congenital heterotopic gastric mucosa. J Clin Gastroenterol 1988; 10: 92-94. ArticlePubMed
REFERENCES
Fig. 1Preoperative abdominal computed tomography and gross specimen. (A, B) Focal narrowing of the transverse colon (arrows) with proximal intestinal loop dilatation is detected. (C) Gross examination of the resected specimen shows focal erosion and stricture of the transverse colon (arrows).
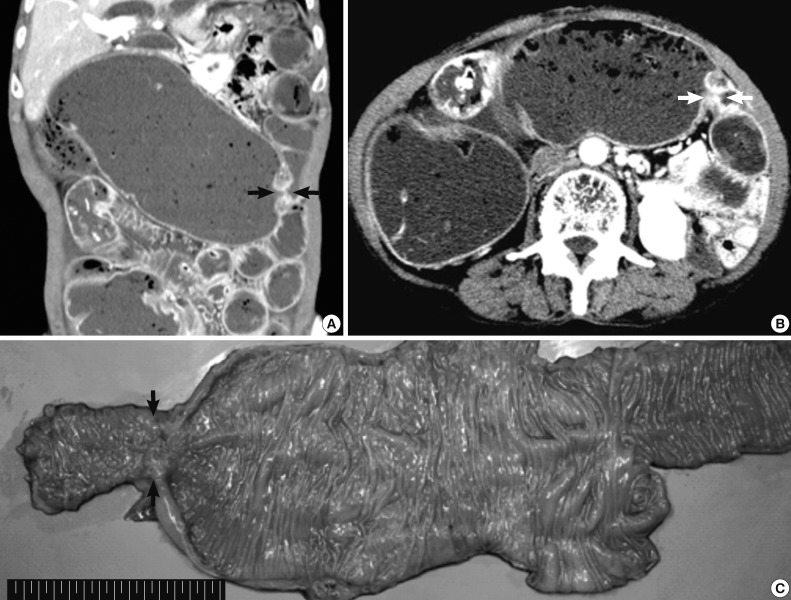

Fig. 2Histopathologic findings of the resected specimen. (A) Hematoxylin and eosin staining shows normal colonic mucosa adjacent to heterotopic gastric mucosa with cystic dilatation. (B) High magnification view of the tissue in Fig. 2A (arrows). Heterotopic gastric mucosa is immunopositive for cytokeratin (CK) 7 (C) but negative for CK20 (D).


Figure & Data
References
Citations
Citations to this article as recorded by 

- Anorectal gastric heterotopia as a rare cause of constipation: Case report and review of pediatric literature
Kathryn M. Stephenson, Raj P. Kapur, Jeffrey R. Avansino, Lusine Ambartsumyan
JPGN Reports.2025; 6(4): 407. CrossRef - Intussusception of Heterotopic Gastric Mucosa in the Transverse Colon: A Rare Cause of Perforation and Bleeding
Sho Fujiwara, Ryuichi Nishimura, Nozomi Koyamada
Cureus.2024;[Epub] CrossRef - Gastric heterotopia of colon found cancer workup in liver abscess: A case report
Jun Gi Park, Jeong Ill Suh, Yeo Un Kim
World Journal of Clinical Cases.2022; 10(15): 5012. CrossRef - Gastric heterotopia in the ileum mimicking Meckel's diverticulum
Reza Shojaeian, Negar Nekooei, Paria Dehghanian
Journal of Pediatric Surgery Case Reports.2022; 84: 102361. CrossRef - Sometimes Things Are Not Where They Are Supposed to Be: A Case Report of Gastric Heterotopia in the Rectum
Asher Lippe, Scott Lippe
Physician's Journal of Medicine.2022;[Epub] CrossRef - Gastric heterotopia of the rectum
Eduardo Dantas, Diva Yamaguti, Kendi Yamazaki
Gastroenterología y Hepatología.2021; 44(8): 579. CrossRef - Bleeding Gastric Heterotopia of Cecal Diverticulum in an Adolescent: A Case Report
Hyun-Il Seo, Jae-Young Kwak
Advances in Pediatric Surgery.2021; 27(1): 32. CrossRef - Gastric heterotopia of the rectum
Eduardo Dantas, Diva Yamaguti, Kendi Yamazaki
Gastroenterología y Hepatología (English Edition).2021; 44(8): 579. CrossRef - Polypoid Gastric Heterotopia of Colon
Marcela Adriana Duran Alvarez, Carla Noemi Tafur Sanchez
GE - Portuguese Journal of Gastroenterology.2020; 27(1): 65. CrossRef - Heterotopic Respiratory Mucosa in the Rectum: An Unusual Type and Site of Heterotopia in the Gastrointestinal Tract
Caroline Bsirini, Pratyusha Tirumanisetty, Joseph N. Dytoc, Diana Agostini-Vulaj, Christopher Steevens, Asad Ullah, Aaron R. Huber
International Journal of Surgical Pathology.2019; 27(2): 221. CrossRef - Perforation of Heterotopic Gastric Mucosa in ileal duplication in an adult: A case report
Vaanathi Paulvannan, Seshukumar Bylapudi, Mithun Kumar Ramesh Kumar, Mahesh Nachimuthu, Paulvannan Subramanian
Journal of Surgical Case Reports.2019;[Epub] CrossRef - Mixed adenoneuroendocrine carcinoma of the tongue arising within a congenital enteric cyst
Louis J. Ligthelm, Belinda K. Bunn, Erich J. Raubenheimer, Willie F. P. van Heerden
Head & Neck.2018;[Epub] CrossRef - The outlet patch: gastric heterotopia of the colorectum and anus
Abul A S R Mannan, Michael Vieth, Armen Khararjian, Binny Khandakar, Dora Lam‐Himlin, David Heydt, Feriyl Bhaijee, Henry J Venbrux, Kathleen Byrnes, Lysandra Voltaggio, Norman Barker, Songyang Yuan, Elizabeth A Montgomery
Histopathology.2018; 73(2): 220. CrossRef - Large heterotopic gastric mucosa and a concomitant diverticulum in the rectum: Clinical experience and endoscopic management
Wen-Guo Chen, Hua-Tuo Zhu, Ming Yang, Guo-Qiang Xu, Li-Hua Chen, Hong-Tan Chen
World Journal of Gastroenterology.2018; 24(30): 3462. CrossRef - Gastric heterotopia in the rectum. A rare cause of ectopic gastric tissue
George A. Salem, Javid Fazili, Tauseef Ali
Arab Journal of Gastroenterology.2017; 18(1): 42. CrossRef - Gastric heterotopia in rectum: A literature review and its diagnostic pitfall
Peyman Dinarvand, Ashley A. Vareedayah, Nancy J Phillips, Christine Hachem, Jinping Lai
SAGE Open Medical Case Reports.2017;[Epub] CrossRef - Heterotopic gastric mucosa in the anus and rectum: first case report of endoscopic submucosal dissection and systematic review
Federico Iacopini, Takuji Gotoda, Walter Elisei, Patrizia Rigato, Fabrizio Montagnese, Yutaka Saito, Guido Costamagna, Giampaolo Iacopini
Gastroenterology Report.2016; 4(3): 196. CrossRef
 PubReader
PubReader ePub Link
ePub Link-
 Cite this Article
Cite this Article
- Cite this Article
-
- Close
- Download Citation
- Close
- Figure
Colonic Adenocarcinoma Arising from Gastric Heterotopia: A Case Study



Fig. 1 Preoperative abdominal computed tomography and gross specimen. (A, B) Focal narrowing of the transverse colon (arrows) with proximal intestinal loop dilatation is detected. (C) Gross examination of the resected specimen shows focal erosion and stricture of the transverse colon (arrows).
Fig. 2 Histopathologic findings of the resected specimen. (A) Hematoxylin and eosin staining shows normal colonic mucosa adjacent to heterotopic gastric mucosa with cystic dilatation. (B) High magnification view of the tissue in Fig. 2A (arrows). Heterotopic gastric mucosa is immunopositive for cytokeratin (CK) 7 (C) but negative for CK20 (D).
Fig. 3 Microscopic appearance of the adenocarcinoma component. (A-C) Invasive adenocarcinoma is located in an area of gastric heterotopia. Adenocarcinoma is immunopositive for cytokeratin (CK) 7 (D) but negative for CK20 (E).
Fig. 1
Fig. 2
Fig. 3
Colonic Adenocarcinoma Arising from Gastric Heterotopia: A Case Study

 E-submission
E-submission