Search
- Page Path
- HOME > Search
- Cutaneous soft tissue tumors in the 5th edition of the World Health Organization classification of skin tumors: key updates and new entities
- Joon Hyuk Choi
- J Pathol Transl Med. 2026;60(2):144-183. Published online March 13, 2026
- DOI: https://doi.org/10.4132/jptm.2026.01.09
- 1,565 View
- 78 Download
-
 Abstract
Abstract
 PDF
PDF - The 5th edition of the World Health Organization (WHO) classification of skin tumors introduces a dedicated chapter on cutaneous soft tissue tumors, providing a comprehensive, standardized reference with updated diagnostic criteria that directly inform routine dermatopathology practice and molecular diagnostics. This edition incorporates several key changes, including newly recognized entities such as EWSR1::SMAD3-rearranged fibroblastic tumor, neurotrophic tyrosine receptor kinase (NTRK)–rearranged spindle cell neoplasm, superficial CD34-positive fibroblastic tumor, and CRTC1::TRIM11 cutaneous tumor. Diagnostic terminology has also been refined; for example, the term ‘atypical intradermal smooth muscle neoplasm’ replaces ‘cutaneous leiomyosarcoma’ for lesions confined to the dermis, whereas the designation leiomyosarcoma is reserved for tumors with overt subcutaneous infiltration. In addition, epithelioid fibrous histiocytoma has been reassigned to the family of tumors of uncertain differentiation. This review summarizes the key updates and newly recognized entities in the chapter on cutaneous soft tissue tumors in the 5th edition of the WHO classification of skin tumors, emphasizing their clinicopathological and molecular implications.
- A comprehensive review of ossifying fibromyxoid tumor: insights into its clinical, pathological, and molecular landscape
- Kyriakos Chatzopoulos, Antonia Syrnioti, Mohamed Yakoub, Konstantinos Linos
- J Pathol Transl Med. 2026;60(1):6-19. Published online January 14, 2026
- DOI: https://doi.org/10.4132/jptm.2025.10.02
- 2,975 View
- 149 Download
-
 Abstract
Abstract
 PDF
PDF - Ossifying fibromyxoid tumor (OFMT) is a rare mesenchymal neoplasm first described in 1989. It typically arises in the superficial soft tissues of the extremities as a slow-growing, painless mass. Histologically, it is commonly characterized by a multilobular architecture composed of uniform epithelioid cells embedded in a fibromyxoid matrix, often surrounded by a rim of metaplastic bone. While classic cases are readily identifiable, the tumor's histopathological heterogeneity can mimic a range of benign and malignant neoplasms, posing significant diagnostic challenges. Molecularly, most OFMTs harbor PHF1 rearrangements, commonly involving fusion partners such as EP400, MEAF6, or TFE3. This review underscores the importance of an integrated diagnostic approach- incorporating histopathological, immunohistochemical, and molecular data- to accurately classify OFMT and distinguish it from its mimics. Expanding awareness of its morphologic and molecular spectrum is essential for precise diagnosis, optimal patient management, and a deeper understanding of this enigmatic neoplasm.
- Recent topics on thyroid cytopathology: reporting systems and ancillary studies
- Mitsuyoshi Hirokawa, Ayana Suzuki
- J Pathol Transl Med. 2025;59(4):214-224. Published online June 30, 2025
- DOI: https://doi.org/10.4132/jptm.2025.04.18
- 4,876 View
- 312 Download
-
 Abstract
Abstract
 PDF
PDF - As fine-needle aspiration techniques and diagnostic methodologies for thyroid nodules have continued to evolve and reporting systems have been updated accordingly, we need to be up to date with the latest information to achieve accurate diagnoses. However, the diagnostic approaches and therapeutic strategies for thyroid nodules vary across laboratories and institutions. Several differences exist between Western and Eastern practices regarding thyroid fine-needle aspiration. This review describes the reporting systems for thyroid cytopathology and ancillary studies. Updated reporting systems enhance the accuracy, consistency, and clarity of cytology reporting, leading to improved patient outcomes and management strategies. Although a single global reporting system is optimal, reporting systems tailored to each country is acceptable. In such cases, compatibility must be ensured to facilitate data sharing. Ancillary methods include liquid-based cytology, immunocytochemistry, biochemical measurements, flow cytometry, molecular testing, and artificial intelligence, all of which improve diagnostic accuracy. These methods continue to evolve, and cytopathologists should actively adopt the latest methods and information to achieve more accurate diagnoses. We believe this review will be useful to practitioners of routine thyroid cytology.
- Next step of molecular pathology: next-generation sequencing in cytology
- Ricella Souza da Silva, Fernando Schmitt
- J Pathol Transl Med. 2024;58(6):291-298. Published online November 7, 2024
- DOI: https://doi.org/10.4132/jptm.2024.10.22
- 7,479 View
- 404 Download
- 4 Web of Science
- 4 Crossref
-
 Abstract
Abstract
 PDF
PDF - The evolving landscape of precision oncology underscores the pivotal shift from morphological diagnosis to treatment decisions driven by molecular profiling. Recent guidelines from the European Society for Medical Oncology recomend the use of next-generation sequencing (NGS) across a broader range of cancers, reflecting its superior efficiency and clinical value. NGS not only updates oncology testing by offering quicker, sample-friendly, and sensitive analysis but also reduces the need for multiple individual tests. Cytology samples, often obtained through less invasive methods, can yield high-quality genetic material suitable for molecular analysis. This article focuses on optimizing the use of cytology samples in NGS, and outlines their potential benefits in identifying actionable molecular alterations for targeted therapies across various solid tumors. It also addresses the need for validation studies and the strategies to incorporate or combine different types of samples into routine clinical practice. Integrating cytological and liquid biopsies into routine clinical practice, alongside conventional tissue biopsies, offers a comprehensive approach to tumor genotyping, early disease detection, and monitoring of therapeutic responses across various solid tumor types. For comprehensive biomarker characterization, all patient specimens, although limited, is always valuable.
-
Citations
Citations to this article as recorded by- Unraveling the nexus: Tumor mutational burden, PD‐L1 expression, and oncogenic alterations in non–small cell lung cancer cytology specimens
Min Dai, Francis Anthony San Lucas, Hector Alvarez, Leomar Ballester, Hui Chen, Keyur P. Patel, Asif Rashid, Shun Rao, Mark J. Routbort, Gloria Sura, Keith Sweeney, Gokce Toruner, Peng Wei, Richard Yang, Hyvan Dang, Rajyalakshmi Luthra, Sinchita Roy‐Chowd
Cancer Cytopathology.2026;[Epub] CrossRef - The World Health Organization Reporting System for Lymph Node, Spleen, and Thymus Cytopathology: Part 1 – Lymph Node
Immacolata Cozzolino, Mats Ehinger, Maria Calaminici, Andrea Ronchi, Mousa A. Al-Abbadi, Helena Barroca, Beata Bode-Lesniewska, David F. Chhieng, Ruth L. Katz, Oscar Lin, L. Jeffrey Medeiros, Martha Bishop Pitman, Arvind Rajwanshi, Fernando C. Schmitt, Ph
Acta Cytologica.2025; 70(2): 185. CrossRef - The impact of cytological preparation techniques on RNA quality: A comparative study on smear samples
Cisel Aydin Mericoz, Gulsum Caylak, Elif Sevin Sanioglu, Zeynep Seçil Satilmis, Ayse Humeyra Dur Karasayar, Ibrahim Kulac
Cancer Cytopathology.2025;[Epub] CrossRef - Reimagining cytopathology in the molecular era: Integration or fragmentation?
Sumanta Das, R. Naveen Kumar, Biswajit Dey, Pranjal Kalita
Cytojournal.2025; 22: 94. CrossRef
- Unraveling the nexus: Tumor mutational burden, PD‐L1 expression, and oncogenic alterations in non–small cell lung cancer cytology specimens
- Establishing molecular pathology curriculum for pathology trainees and continued medical education: a collaborative work from the Molecular Pathology Study Group of the Korean Society of Pathologists
- Jiwon Koh, Ha Young Park, Jeong Mo Bae, Jun Kang, Uiju Cho, Seung Eun Lee, Haeyoun Kang, Min Eui Hong, Jae Kyung Won, Youn-La Choi, Wan-Seop Kim, Ahwon Lee
- J Pathol Transl Med. 2023;57(5):265-272. Published online September 15, 2023
- DOI: https://doi.org/10.4132/jptm.2023.08.26
- 6,049 View
- 209 Download
- 1 Web of Science
- 1 Crossref
-
 Abstract
Abstract
 PDF
PDF - Background
The importance of molecular pathology tests has increased during the last decade, and there is a great need for efficient training of molecular pathology for pathology trainees and as continued medical education.
Methods
The Molecular Pathology Study Group of the Korean Society of Pathologists appointed a task force composed of experienced molecular pathologists to develop a refined educational curriculum of molecular pathology. A 3-day online educational session was held based on the newly established structure of learning objectives; the audience were asked to score their understanding of 22 selected learning objectives before and after the session to assess the effect of structured education.
Results
The structured objectives and goals of molecular pathology was established and posted as a web-based interface which can serve as a knowledge bank of molecular pathology. A total of 201 pathologists participated in the educational session. For all 22 learning objectives, the scores of self-reported understanding increased after educational session by 9.9 points on average (range, 6.6 to 17.0). The most effectively improved items were objectives from next-generation sequencing (NGS) section: ‘NGS library preparation and quality control’ (score increased from 51.8 to 68.8), ‘NGS interpretation of variants and reference database’ (score increased from 54.1 to 68.0), and ‘whole genome, whole exome, and targeted gene sequencing’ (score increased from 58.2 to 71.2). Qualitative responses regarding the adequacy of refined educational curriculum were collected, where favorable comments dominated.
Conclusions
Approach toward the education of molecular pathology was refined, which would greatly benefit the future trainees. -
Citations
Citations to this article as recorded by- Presence of RB1 or Absence of LRP1B Mutation Predicts Poor Overall Survival in Patients with Gastric Neuroendocrine Carcinoma and Mixed Adenoneuroendocrine Carcinoma
In Hye Song, Bokyung Ahn, Young Soo Park, Deok Hoon Kim, Seung-Mo Hong
Cancer Research and Treatment.2025; 57(2): 492. CrossRef
- Presence of RB1 or Absence of LRP1B Mutation Predicts Poor Overall Survival in Patients with Gastric Neuroendocrine Carcinoma and Mixed Adenoneuroendocrine Carcinoma
- Single-center study on clinicopathological and typical molecular pathologic features of metastatic brain tumor
- Su Hwa Kim, Young Suk Lee, Sung Hak Lee, Yeoun Eun Sung, Ahwon Lee, Jun Kang, Jae-Sung Park, Sin Soo Jeun, Youn Soo Lee
- J Pathol Transl Med. 2023;57(4):217-231. Published online July 11, 2023
- DOI: https://doi.org/10.4132/jptm.2023.06.10
- 6,321 View
- 175 Download
- 1 Crossref
-
 Abstract
Abstract
 PDF
PDF - Background
The metastatic brain tumor is the most common brain tumor. The aim of this study was to demonstrate the clinicopathological and molecular pathologic features of brain metastases (BM).
Methods
A total of 269 patients were diagnosed with BM through surgical resection at Seoul St. Mary’s Hospital from January 2010 to March 2020. We reviewed the clinicopathological features and molecular status of primary and metastatic brain tissues using immunohistochemistry and molecular pathology results.
Results
Among 269 patients, 139 males and 130 females were included. The median age of primary tumor was 58 years (range, 13 to 87 years) and 86 patients (32.0%) had BM at initial presentation. Median BM free interval was 28.0 months (range, 1 to 286 months). The most frequent primary site was lung 46.5% (125/269), and followed by breast 15.6% (42/269), colorectum 10.0% (27/269). Epidermal growth factor receptor (EGFR) mutation was found in 50.8% (32/63) and 58.0% (40/69) of lung primary and BM, respectively. In both breast primary and breast cancer with BM, luminal B was the most frequent subtype at 37.9% (11/29) and 42.9% (18/42), respectively, followed by human epidermal growth factor receptor 2 with 31.0% (9/29) and 33.3% (14/42). Triple-negative was 20.7% (6/29) and 16.7% (7/42), and luminal A was 10.3% (3/29) and 7.1% (3/42) of breast primary and BM, respectively. In colorectal primary and colorectal cancer with BM, KRAS mutation was found in 76.9% (10/13) and 66.7% (2/3), respectively.
Conclusions
We report the clinicopathological and molecular pathologic features of BM that can provide useful information for understanding the pathogenesis of metastasis and for clinical trials based on the tumor’s molecular pathology. -
Citations
Citations to this article as recorded by- Colorectal cancer metastasis to the brain: A scoping review of incidence, treatment, and outcomes
Hunter J Hutchinson, Melanie Gonzalez, Diana Feier, Colin E Welch, Brandon Lucke-Wold
World Journal of Gastrointestinal Pathophysiology.2025;[Epub] CrossRef
- Colorectal cancer metastasis to the brain: A scoping review of incidence, treatment, and outcomes
- A standardized pathology report for gastric cancer: 2nd edition
- Young Soo Park, Myeong-Cherl Kook, Baek-hui Kim, Hye Seung Lee, Dong-Wook Kang, Mi-Jin Gu, Ok Ran Shin, Younghee Choi, Wonae Lee, Hyunki Kim, In Hye Song, Kyoung-Mee Kim, Hee Sung Kim, Guhyun Kang, Do Youn Park, So-Young Jin, Joon Mee Kim, Yoon Jung Choi, Hee Kyung Chang, Soomin Ahn, Mee Soo Chang, Song-Hee Han, Yoonjin Kwak, An Na Seo, Sung Hak Lee, Mee-Yon Cho
- J Pathol Transl Med. 2023;57(1):1-27. Published online January 15, 2023
- DOI: https://doi.org/10.4132/jptm.2022.12.23
- 39,938 View
- 1,572 Download
- 23 Web of Science
- 22 Crossref
-
 Abstract
Abstract
 PDF
PDF Supplementary Material
Supplementary Material - The first edition of ‘A Standardized Pathology Report for Gastric Cancer’ was initiated by the Gastrointestinal Pathology Study Group of the Korean Society of Pathologists and published 17 years ago. Since then, significant advances have been made in the pathologic diagnosis, molecular genetics, and management of gastric cancer (GC). To reflect those changes, a committee for publishing a second edition of the report was formed within the Gastrointestinal Pathology Study Group of the Korean Society of Pathologists. This second edition consists of two parts: standard data elements and conditional data elements. The standard data elements contain the basic pathologic findings and items necessary to predict the prognosis of GC patients, and they are adequate for routine surgical pathology service. Other diagnostic and prognostic factors relevant to adjuvant therapy, including molecular biomarkers, are classified as conditional data elements to allow each pathologist to selectively choose items appropriate to the environment in their institution. We trust that the standardized pathology report will be helpful for GC diagnosis and facilitate large-scale multidisciplinary collaborative studies.
-
Citations
Citations to this article as recorded by- GAST-NET: A multi-modal and multi-task deep learning framework for preoperative prediction of perineural invasion and prognostic risk in gastric cancer
Shidi Miao, Hexiang Dong, Jinyang Feng, Yuyang Jiang, Mengzhuo Sun, Zengyao Liu, Qiujun Wang, Xuemei Ding, Ruitao Wang
International Journal of Medical Informatics.2026; 212: 106348. CrossRef - Poorly cohesive carcinoma diffusely involving the whole gastrointestinal tract: a case report
Wei Gao, Yusheng Yang, Xinyi Hu, Yujuan Shi, Kai Liu, Minmin Gu, Jing Wang
Discover Oncology.2026;[Epub] CrossRef - Comprehensive Overview of Gastric Cancer Immunohistochemistry: Key Biomarkers, Advanced Detection Methods, and Perspectives
Bogdan Oprea
Medicina.2026; 62(4): 683. CrossRef - Spatial and Temporal Tumor Heterogeneity in Gastric Cancer: Discordance of Predictive Biomarkers
Hye Seung Lee
Journal of Gastric Cancer.2025; 25(1): 192. CrossRef - PD-L1 as a Biomarker in Gastric Cancer Immunotherapy
Yunjoo Cho, Soomin Ahn, Kyoung-Mee Kim
Journal of Gastric Cancer.2025; 25(1): 177. CrossRef - Korean Gastric Cancer Association-Led Nationwide Survey on Surgically Treated Gastric Cancers in 2023
Dong Jin Kim, Jeong Ho Song, Ji-Hyeon Park, Sojung Kim, Sin Hye Park, Cheol Min Shin, Yoonjin Kwak, Kyunghye Bang, Chung-sik Gong, Sung Eun Oh, Yoo Min Kim, Young Suk Park, Jeesun Kim, Ji Eun Jung, Mi Ran Jung, Bang Wool Eom, Ki Bum Park, Jae Hun Chung, S
Journal of Gastric Cancer.2025; 25(1): 115. CrossRef - A Comprehensive and Comparative Review of Global Gastric Cancer Treatment Guidelines: 2024 Update
Sang Soo Eom, Keun Won Ryu, Hye Sook Han, Seong-Ho Kong
Journal of Gastric Cancer.2025; 25(1): 153. CrossRef - Korea, Japan, Europe, and the United States: Why are guidelines for gastric cancer different?
Emily E. Stroobant, Seong-Ho Kong, Maria Bencivenga, Takahiro Kinoshita, Tae-Han Kim, Takeshi Sano, Giovanni de Manzoni, Han-Kwang Yang, Yuko Kitagawa, Vivian E. Strong
Gastric Cancer.2025; 28(4): 559. CrossRef - Can the Japanese guidelines for endoscopic submucosal dissection be safely applied to Korean gastric cancer patients? A multicenter retrospective study based on the Korean Gastric Cancer Association nationwide survey
Hayemin Lee, Mi Ryeong Park, Junhyun Lee
Annals of Surgical Treatment and Research.2025; 109(2): 81. CrossRef - Double optimal transport for differential gene regulatory network inference with unpaired samples
Mengyu Li, Bencong Zhu, Cheng Meng, Xiaodan Fan, Laura Cantini
Bioinformatics.2025;[Epub] CrossRef - A Randomized Controlled Trial to Evaluate the Effect of Fibrin Glue on Bleeding after Gastric Endoscopic Submucosal Dissection
Tae-Se Kim, Tae-Jun Kim, Yang Won Min, Hyuk Lee, Byung-Hoon Min, Jun Haeng Lee, Poong-Lyul Rhee, Jae J. Kim
Gut and Liver.2025; 19(5): 677. CrossRef - Diagnostic accuracy of stereomicroscopy assessment of invasion depth in ex vivo specimens of early gastric cancer
Jing Wang, Lin Chang, Dong-Feng Niu, Yan Yan, Chang-Qi Cao, Shi-Jie Li, Qi Wu
World Journal of Gastroenterology.2025;[Epub] CrossRef - SMMILe enables accurate spatial quantification in digital pathology using multiple-instance learning
Zeyu Gao, Anyu Mao, Yuxing Dong, Hannah Clayton, Jialun Wu, Jiashuai Liu, ChunBao Wang, Kai He, Tieliang Gong, Chen Li, Mireia Crispin-Ortuzar
Nature Cancer.2025; 6(12): 2025. CrossRef - Genomic and Transcriptomic Characterization of Gastric Cancer with Bone Metastasis
Sujin Oh, Soo Kyung Nam, Keun-Wook Lee, Hye Seung Lee, Yujun Park, Yoonjin Kwak, Kyu Sang Lee, Ji-Won Kim, Jin Won Kim, Minsu Kang, Young Suk Park, Sang-Hoon Ahn, Yun-Suhk Suh, Do Joong Park, Hyung Ho Kim
Cancer Research and Treatment.2024; 56(1): 219. CrossRef - Microscopic tumor mapping of post-neoadjuvant therapy pancreatic cancer specimens to predict post-surgical recurrence: A prospective cohort study
Yeshong Park, Yeon Bi Han, Jinju Kim, MeeYoung Kang, Boram Lee, Eun Sung Ahn, Saemi Han, Haeryoung Kim, Hee-Young Na, Ho-Seong Han, Yoo-Seok Yoon
Pancreatology.2024; 24(4): 562. CrossRef - Effect of Neoadjuvant Chemotherapy on Tumor-Infiltrating Lymphocytes in Resectable Gastric Cancer: Analysis from a Western Academic Center
Elliott J. Yee, Danielle Gilbert, Jeffrey Kaplan, Sachin Wani, Sunnie S. Kim, Martin D. McCarter, Camille L. Stewart
Cancers.2024; 16(7): 1428. CrossRef - Interpretation of PD-L1 expression in gastric cancer: summary of a consensus meeting of Korean gastrointestinal pathologists
Soomin Ahn, Yoonjin Kwak, Gui Young Kwon, Kyoung-Mee Kim, Moonsik Kim, Hyunki Kim, Young Soo Park, Hyeon Jeong Oh, Kyoungyul Lee, Sung Hak Lee, Hye Seung Lee
Journal of Pathology and Translational Medicine.2024; 58(3): 103. CrossRef - Expression of claudin 18.2 in poorly cohesive carcinoma and its association with clinicopathologic parameters in East Asian patients
Moonsik Kim, Byung Woog Kang, Jihyun Park, Jin Ho Baek, Jong Gwang Kim
Pathology - Research and Practice.2024; 263: 155628. CrossRef - Clinicopathological analysis of claudin 18.2 focusing on intratumoral heterogeneity and survival in patients with metastatic or unresectable gastric cancer
T.-Y. Kim, Y. Kwak, S.K. Nam, D. Han, D.-Y. Oh, S.-A. Im, H.S. Lee
ESMO Open.2024; 9(12): 104000. CrossRef - Pathological Interpretation of Gastric Tumors in Endoscopic Submucosal Dissection
Jung Yeon Kim
Journal of Digestive Cancer Research.2023; 11(1): 15. CrossRef - Histopathology of Gastric Cancer
Baek-hui Kim, Sung Hak Lee
The Korean Journal of Helicobacter and Upper Gastrointestinal Research.2023; 23(2): 143. CrossRef - Endoscopic submucosal dissection hands-on training with artificial mucosal layer EndoGEL
Tae-Se Kim, Jun Haeng Lee
Journal of Innovative Medical Technology.2023; 1(1): 5. CrossRef
- GAST-NET: A multi-modal and multi-task deep learning framework for preoperative prediction of perineural invasion and prognostic risk in gastric cancer
- Biomarker testing of cytology specimens in personalized medicine for lung cancer patients
- Hyojin Kim, Jin-Haeng Chung
- J Pathol Transl Med. 2022;56(6):326-333. Published online November 9, 2022
- DOI: https://doi.org/10.4132/jptm.2022.10.17
- 7,361 View
- 179 Download
- 10 Web of Science
- 8 Crossref
-
 Abstract
Abstract
 PDF
PDF - Every patient with advanced non–small cell lung cancer (NSCLC) should be tested for targetable driver mutations and gene arrangements that may open avenues for targeted therapy. As most patients with NSCLC in the advanced stage of the disease are not candidates for surgery, these tests have to be performed on small biopsies or cytology samples. A growing number of other genetic changes with targetable mutations may be treatable in the near future. To identify patients who might benefit from novel targeted therapy, relevant markers should be tested in an appropriate context. In addition, immunotherapy of lung cancer is guided by the status of programmed death-ligand 1 expression in tumor cells. The variety and versatility of cytological specimen preparations offer significant advantages for molecular testing; however, they frequently remain underused. Therefore, evaluating the utility and adequacy of cytologic specimens is important, not only from a lung cancer diagnosis, but also for the large number of ancillary studies that are necessary to provide appropriate clinical management. A large proportion of lung cancers is diagnosed by aspiration or exfoliative cytology specimens; thus, optimizing strategies to triage and best use the tissue for diagnosis and biomarker studies forms a critical component of lung cancer management. In this review, we discuss the opportunities and challenges of using cytologic specimens for biomarker testing of lung cancer and the role of cytopathology in the molecular era.
-
Citations
Citations to this article as recorded by- Companion Diagnostics in Clinical Therapy: Current Applications and Future Directions
Yuesong Wu, Rou Xue, Xiangwen Luo, Jiangnan Liao, Zongbo Zhang, Jinhai Deng, Teng Liu, Xin Li, Zhe‐Sheng Chen, Mingzhu Yin
MedComm.2026;[Epub] CrossRef - Multidisciplinary international expert consensus recommendations on tissue acquisition in non-small cell lung cancer
Pyng Lee, Karim Abdelhamid, Rachel Butler, Wendy Cooper, Misako Nagasaka, Solange Peters, Luis Seijo, Gerard Silvestri, Bernard Wee, Kazuhiro Yasufuku, Neal Navani
eBioMedicine.2026; 126: 106223. CrossRef - Proposal of real-world solutions for the implementation of predictive biomarker testing in patients with operable non-small cell lung cancer
Paul Hofman, Petros Christopoulos, Nicky D’Haene, John Gosney, Nicola Normanno, Ed Schuuring, Ming-Sound Tsao, Christine Quinn, Jayne Russell, Katherine E Keating, Fernando López-Ríos
Lung Cancer.2025; 201: 108107. CrossRef - Validation of ancillary procedures on formalin liquid fixed aspiration cytologic samples: from minimum to maximum
Orsolya Rideg, Tímea Dergez, Arnold Tóth, Tamás Tornóczky, Gábor Pavlovics, Endre Kálmán
American Journal of Clinical Pathology.2025; 164(6): 924. CrossRef - Molecular testing of cytology specimens: Issues in specimen adequacy and clinical utility
Ghulam Ghous, Komal Ijaz, Magda Esebua, Lester J. Layfield
Diagnostic Cytopathology.2024; 52(2): 123. CrossRef - The updated College of American Pathologists principles of analytic validation of immunohistochemical assays: A step forward for cytopathology
Sinchita Roy‐Chowdhuri
Cancer Cytopathology.2024; 132(9): 547. CrossRef - Best-Practice Biomarker Testing of Oesophago-Gastric Cancer in the UK: Expert Consensus Recommendations Developed Using a Modified Delphi
N.P. West, W. Mansoor, P. Taniere, E. Smyth, M. Rodriguez-Justo, A. Oniscu, P. Carter
Clinical Oncology.2024; 36(11): 701. CrossRef - Next step of molecular pathology: next-generation sequencing in cytology
Ricella Souza da Silva, Fernando Schmitt
Journal of Pathology and Translational Medicine.2024; 58(6): 291. CrossRef
- Companion Diagnostics in Clinical Therapy: Current Applications and Future Directions
- Follicular lymphoma: updates for pathologists
- Mahsa Khanlari, Jennifer R. Chapman
- J Pathol Transl Med. 2022;56(1):1-15. Published online December 27, 2021
- DOI: https://doi.org/10.4132/jptm.2021.09.29
- 34,114 View
- 1,066 Download
- 22 Web of Science
- 21 Crossref
-
 Abstract
Abstract
 PDF
PDF - Follicular lymphoma (FL) is the most common indolent B-cell lymphoma and originates from germinal center B-cells (centrocytes and centroblasts) of the lymphoid follicle. Tumorigenesis is believed to initiate early in precursor B-cells in the bone marrow (BM) that acquire the t(14;18)(q32;q21). These cells later migrate to lymph nodes to continue their maturation through the germinal center reaction, at which time they acquire additional genetic and epigeneticabnormalities that promote lymphomagenesis. FLs are heterogeneous in terms of their clinicopathologic features. Most FLs are indolent and clinically characterized by peripheral lymphadenopathy with involvement of the spleen, BM, and peripheral blood in a substantial subset of patients, sometimes accompanied by constitutional symptoms and laboratory abnormalities. Diagnosis is established by the histopathologic identification of a B-cell proliferation usually distributed in an at least partially follicular pattern, typically, but not always, in a lymph node biopsy. The B-cell proliferation is biologically of germinal center cell origin, thus shows an expression of germinal center-associated antigens as detected by immunophenotyping. Although many cases of FLs are typical and histopathologic features are straightforward, the biologic and histopathologic variability of FL is wide, and an accurate diagnosis of FL over this disease spectrum requires knowledge of morphologic variants that can mimic other lymphomas, and rarely non-hematologic malignancies, clinically unique variants, and pitfalls in the interpretation of ancillary studies. The overall survival for most patients is prolonged, but relapses are frequent. The treatment landscape in FL now includes the application of immunotherapy and targeted therapy in addition to chemotherapy.
-
Citations
Citations to this article as recorded by- Follicular Cholecystitis: A Case Report Highlighting the Diagnostic Challenges and Management Implications
Ativitch Asavachaisuvikom, Burana Khiankaew, Narongsak Rungsakulkij
Gastro Hep Advances.2026; 5(2): 100833. CrossRef - Follicular lymphoma with signet ring cell morphology: Clinicopathologic analysis of 31 cases
Xenia Parisi, L. Jeffrey Medeiros
Human Pathology.2026; 171: 106071. CrossRef - PRIMARY SPLENIC DIFFUSE LARGE B-CELL LYMPHOMA WITH CD30 EXPRESSION: A RARE CASE REPORT
Beyza Öztürk, Hüseyin Buğra Kutlu, Meltem Ayyıldız Mercan, Dicle Tamer Türk, Yusuf Emre Aytin, Funda Üstün, Fulya Öz Puyan
TURKISH MEDICAL STUDENT JOURNAL.2026;[Epub] CrossRef - Relapsed/Refractory Follicular Lymphoma: Current Advances and Emerging Perspectives
Giulio Caridà, Enrica Antonia Martino, Antonella Bruzzese, Daniele Caracciolo, Caterina Labanca, Francesco Mendicino, Eugenio Lucia, Virginia Olivito, Teresa Rossi, Antonino Neri, Ernesto Vigna, Pierfrancesco Tassone, Pierosandro Tagliaferri, Fortunato Mo
European Journal of Haematology.2025; 114(5): 775. CrossRef - Frequency and Distribution of Lymphomas in Northwestern India: A Retrospective Analysis of 923 Cases Using the Latest World Health Organization Classification 5th Edition
Immanuel Paul Thayakaran, Biren Parikh
Indian Journal of Hematology and Blood Transfusion.2025;[Epub] CrossRef - IGH/IGK gene rearrangement in the diagnosis of B-cell non-Hodgkin lymphoma: experience from three centers
Ke Yang, Zhizhong Wang, Beibei Xin, Yunhang Li, Jiuzhou Zhao, Rui Sun, Weizhen Wang, Dongxu Chen, Chengzhi Zhao, Yongjun Guo, Jie Ma, Bing Wei
Annals of Hematology.2025; 104(7): 3779. CrossRef - Imaging Evaluation of Periarticular Soft Tissue Masses in the Appendicular Skeleton: A Pictorial Review
Francesco Pucciarelli, Maria Carla Faugno, Daniela Valanzuolo, Edoardo Massaro, Lorenzo Maria De Sanctis, Elisa Zaccaria, Marta Zerunian, Domenico De Santis, Michela Polici, Tiziano Polidori, Andrea Laghi, Damiano Caruso
Journal of Imaging.2025; 11(7): 217. CrossRef - Understanding the clinical approach to “pathologically ambiguous follicular lymphoma” through a Real-World cohort
Sarah Matarasso Greenfeld, Svetlana Dmitrienko, Ian Shrier, Jean Luc Deschenes, Sarit Assouline
Leukemia & Lymphoma.2025; 66(12): 2332. CrossRef - Deciphering and targeting oncogenic pathways through integrated approaches and amino acid metabolism in hematologic malignancies
Farhan Ikhtiar, Adil Jamal, Syed M. Safeer Mehdi Bokhari
Discover Oncology.2025;[Epub] CrossRef - Transformation of low-grade follicular lymphoma to a high-grade follicular lymphoma with the histopathological diagnosis from oral biopsy: a case report
Gabriela Silveira de Araujo, Leandro Dorigan de Macedo, Alfredo Ribeiro-Silva, Hilton Marcos Alves Ricz, Lara Maria Alencar Ramos Innocentini
Hematology, Transfusion and Cell Therapy.2024; 46: S380. CrossRef - The follicular lymphoma tumor microenvironment at single-cell and spatial resolution
Andrea J. Radtke, Mark Roschewski
Blood.2024; 143(12): 1069. CrossRef - Chronic pancreatitis for the clinician: complications and special forms of the disease. Interdisciplinary position paper of the Catalan Society of Digestology (SCD) and the Catalan Pancreatic Society (SCPanc)
Xavier MOLERO, Juan R. AYUSO, Joaquim BALSELLS, Jaume BOADAS, Juli BUSQUETS, Anna CASTERÀS, Mar CONCEPCIÓN, Míriam CUATRECASAS, Gloria FERNÀNDEZ ESPARRACH, Esther FORT, Francisco GARCIA BOROBIA, Àngels GINÈS, Lucas ILZARBE, Carme LORAS, Miquel MASACHS, Xa
Minerva Gastroenterology.2024;[Epub] CrossRef - Concurrent identification of follicular lymphoma and papillary thyroid carcinoma
Lama A. Alzelfawi, Norah ALhumaidan, Abrar H. Alageel, Buthaina J. Yahya, Saud D. Alrasheedi, Adel S. Alqahtani
International Journal of Surgery Case Reports.2024;[Epub] CrossRef - Impact of Primary Disease Site of Involvement by Early-Stage Follicular Lymphoma on Patient Outcomes
Olivia Davis, Carmen Lessani, Rana Kasht, Andrew Cohoon, Sami Ibrahimi, Adam Asch, Silas Day, Taha Al-Juhaishi
Clinical Lymphoma Myeloma and Leukemia.2024; 24(12): 837. CrossRef - Recent developments in CD19-targeted therapies for follicular lymphoma
Aditi Saha, Julio C. Chavez
Expert Opinion on Biological Therapy.2024; 24(10): 1049. CrossRef - Unraveling the complexity of follicular lymphoma: insights and innovations
Xijing Li
American Journal of Cancer Research.2024; 14(12): 5573. CrossRef - Clinical features and prognostic factors in 49 patients with follicular lymphoma at a single center: A retrospective analysis
Hao Wu, Hui-Cong Sun, Gui-Fang Ouyang
World Journal of Clinical Cases.2023; 11(14): 3176. CrossRef - A rare case of follicular lymphoma of the bladder
Matthew DeSanto, Robert Strait, Jared Zopp, Kevin Brown, Samuel Deem
Urology Case Reports.2023; 51: 102542. CrossRef - Analysis of immunophenotypic features in hyaline vascular type Castleman disease
Yu Chang, Yu Ma, Chen Chang, Wensheng Li
Diagnostic Pathology.2023;[Epub] CrossRef - Leg Edema Unveiled: The Uncommon Culprit of Follicular Lymphoma
Syed Muhammad IbnE Ali Jaffari, Samaha Nisar, Narjis Malik, Syed Muhammad Aun Ali Jaffari, Omar Nisar
Journal of Shalamar Medical & Dental College - JSHMDC.2023; 4(2): 125. CrossRef - A Review of the Totality of Evidence in the Development of ABP 798, A Rituximab Biosimilar
Patrick Cobb, Dietger Niederwieser, Stanley Cohen, Caroline Hamm, Gerd Burmester, Neungseon Seo, Sonya G Lehto, Vladimir Hanes
Immunotherapy.2022; 14(9): 727. CrossRef
- Follicular Cholecystitis: A Case Report Highlighting the Diagnostic Challenges and Management Implications
- Distinctive morphological and molecular features of urothelial carcinoma with an inverted growth pattern
- Francesca Sanguedolce, Beppe Calò, Marco Chirico, Ugo Falagario, Gian Maria Busetto, Magda Zanelli, Alessandra Bisagni, Maurizio Zizzo, Stefano Ascani, Giuseppe Carrieri, Luigi Cormio
- J Pathol Transl Med. 2021;55(4):239-246. Published online June 14, 2021
- DOI: https://doi.org/10.4132/jptm.2021.04.20
- 8,536 View
- 211 Download
- 2 Web of Science
- 2 Crossref
-
 Abstract
Abstract
 PDF
PDF - Urothelial carcinoma with an inverted growth pattern (UC-IGP) is a peculiar entity within the spectrum of urothelial lesions. While efforts have been made over the last few decades to unravel its carcinogenesis and relationship with conventional urothelial carcinoma, the exact classification of inverted urothelial lesions is a matter of debate. The morphological features of UC-IGP pose several issues in differential diagnosis with other mostly benign lesions. Various techniques, including immunohistochemistry, UroVysion, and many molecular methods, have been employed to study the exact nature of this lesion. The aim of this review is to provide a comprehensive overview of the morphological and immunophenotypical aspects of UC-IGP. Moreover, we present and discuss the immunohistochemical and molecular markers involved in diagnosis and prognosis of UC-IGP lesions.
-
Citations
Citations to this article as recorded by- HER2 Expression in Bladder Cancer: A Focused View on Its Diagnostic, Prognostic, and Predictive Role
Francesca Sanguedolce, Magda Zanelli, Andrea Palicelli, Alessandra Bisagni, Maurizio Zizzo, Stefano Ascani, Maria Carmela Pedicillo, Angelo Cormio, Ugo Giovanni Falagario, Giuseppe Carrieri, Luigi Cormio
International Journal of Molecular Sciences.2023; 24(4): 3720. CrossRef - Proteomic-Based Machine Learning Analysis Reveals PYGB as a Novel Immunohistochemical Biomarker to Distinguish Inverted Urothelial Papilloma From Low-Grade Papillary Urothelial Carcinoma With Inverted Growth
Minsun Jung, Cheol Lee, Dohyun Han, Kwangsoo Kim, Sunah Yang, Ilias P. Nikas, Kyung Chul Moon, Hyeyoon Kim, Min Ji Song, Bohyun Kim, Hyebin Lee, Han Suk Ryu
Frontiers in Oncology.2022;[Epub] CrossRef
- HER2 Expression in Bladder Cancer: A Focused View on Its Diagnostic, Prognostic, and Predictive Role
- Highly prevalent BRAF V600E and low-frequency TERT promoter mutations underlie papillary thyroid carcinoma in Koreans
- Sue Youn Kim, Taeeun Kim, Kwangsoon Kim, Ja Seong Bae, Jeong Soo Kim, Chan Kwon Jung
- J Pathol Transl Med. 2020;54(4):310-317. Published online June 15, 2020
- DOI: https://doi.org/10.4132/jptm.2020.05.12
- 12,085 View
- 199 Download
- 30 Web of Science
- 32 Crossref
-
 Abstract
Abstract
 PDF
PDF - Background
The presence of telomerase reverse transcriptase (TERT) promoter mutations have been associated with a poor prognosis in patients with papillary thyroid carcinomas (PTC). The frequency of TERT promoter mutations varies widely depending on the population and the nature of the study.
Methods
Data were prospectively collected in 724 consecutive patients who underwent thyroidectomy for PTC from 2018 to 2019. Molecular testing for BRAF V600E and TERT promoter mutations was performed in all cases.
Results
TERT promoter alterations in two hotspots (C228T and C250T) and C216T were found in 16 (2.2%) and 4 (0.6%) of all PTCs, respectively. The hotspot mutations were significantly associated with older age at diagnosis, larger tumor size, extrathyroidal extension, higher pathologic T category, lateral lymph node metastasis, and higher American Thyroid Association recurrence risk. The patients with C216T variant were younger and had a lower American Thyroid Association recurrence risk than those with hotspot mutations. Concurrent BRAF V600E was found in 19 of 20 cases with TERT promoter mutations. Of 518 microcarcinomas measuring ≤1.0 cm in size, hotspot mutations and C216T variants were detected in five (1.0%) and three (0.6%) cases, respectively.
Conclusions
Our study indicates a low frequency of TERT promoter mutations in Korean patients with PTC and supports previous findings that TERT promoter mutations are more common in older patients with unfavorable clinicopathologic features and BRAF V600E. TERT promoter mutations in patients with microcarcinoma are uncommon and may have a limited role in risk stratification. The C216T variant seems to have no clinicopathologic effect on PTC. -
Citations
Citations to this article as recorded by- Summary and Analysis of Molecular Biological Changes, PD-L1 Immune Status and Clinicopathological Features of 78 Cases of Papillary Thyroid Carcinoma (<1 cm in Diameter) Combined With Lateral Cervical Lymph Node Metastasis
Xiaoteng Sun, Zhengyan He, Weijie Yu, Baoyuan Li, Xinmiao Xu, Xiaoqin Zhang, Minglong Yin
Applied Immunohistochemistry & Molecular Morphology.2026; 34(2): 95. CrossRef - The impact of C216T and hot spot mutations of the TERT promoter on the clinicopathologic characteristics and S100A10 expression in papillary thyroid carcinoma: a comparative study
Ping Li, Chuqiang Huang, Xiaoling Liu, Huihui Gui, Jian Li
Diagnostic Pathology.2025;[Epub] CrossRef - Refining NTRK Fusion Detection in Papillary Thyroid Carcinoma Through Pan-TRK Immunohistochemistry and Histopathologic Features
Hyun Lee, Sue Youn Kim, Ji Min Park, Seung-Hyun Jung, Ozgur Mete, Chan Kwon Jung
Endocrine Pathology.2025;[Epub] CrossRef - Validation of Diagnostic Utility of Washout CYFRA 21-1 in Lymph Node Metastasis of Thyroid Cancer
Jeongmin Lee, Yuri Shin, Jeongun Kwak, Hye Lim Park, Sohee Lee, Mee Kyung Kim, Ja Seong Bae, Chan Kwon Jung, So Lyung Jung, Jung-Min Lee, Sang-Ah Chang, Dong-Jun Lim
Clinical Cancer Research.2025; 31(10): 1922. CrossRef - 2025 Korean Thyroid Association Clinical Management Guideline on Active Surveillance for Low-Risk Papillary Thyroid Carcinoma
Eun Kyung Lee, Min Joo Kim, Seung Heon Kang, Bon Seok Koo, Kyungsik Kim, Mijin Kim, Bo Hyun Kim, Ji-hoon Kim, Shin Je Moon, Kyorim Back, Young Shin Song, Jong-hyuk Ahn, Hwa Young Ahn, Ho-Ryun Won, Won Sang Yoo, Min Kyoung Lee, Jeongmin Lee, Ji Ye Lee, Kyo
International Journal of Thyroidology.2025; 18(1): 30. CrossRef - 2025 Korean Thyroid Association Clinical Management Guideline on Active Surveillance for Low-Risk Papillary Thyroid Carcinoma
Eun Kyung Lee, Min Joo Kim, Seung Heon Kang, Bon Seok Koo, Kyungsik Kim, Mijin Kim, Bo Hyun Kim, Ji-hoon Kim, Shinje Moon, Kyorim Back, Young Shin Song, Jong-hyuk Ahn, Hwa Young Ahn, Ho-Ryun Won, Won Sang Yoo, Min Kyoung Lee, Jeongmin Lee, Ji Ye Lee, Kyon
Endocrinology and Metabolism.2025; 40(3): 307. CrossRef - Evaluation of BRAF V600E and TERT mutation analysis in differential thyroid cancers
Nigar Aktash, Ahmet Cem Dural, Husnu Aydin, Nuri Alper Sahbaz, Deniz Guzey, Serdar Altınay, Cevher Akarsu, Yasir Musa Kesgin, Sezer Bulut, Mehmet Karabulut
Updates in Surgery.2025;[Epub] CrossRef - Active surveillance for adult low-risk papillary thyroid microcarcinoma—a review focused on the 30-year experience of Kuma Hospital—
Yasuhiro Ito, Akira Miyauchi, Makoto Fujishima, Masashi Yamamoto, Takahiro Sasaki
Endocrine Journal.2024; 71(1): 7. CrossRef - Diagnostic utilities of washout CYFRA 21-1 combined with washout thyroglobulin for metastatic lymph nodes in thyroid cancer: a prospective study
Joonseon Park, Solji An, Kwangsoon Kim, Jeong Soo Kim, Chan Kwon Jung, Ja Seong Bae
Scientific Reports.2024;[Epub] CrossRef - Korean Thyroid Association Guidelines on the Management of Differentiated Thyroid Cancers; Part I. Initial Management of Differentiated Thyroid Cancers - Chapter 5. Evaluation of Recurrence Risk Postoperatively and Initial Risk Stratification in Different
Eun Kyung Lee, Young Shin Song, Ho-Cheol Kang, Sun Wook Kim, Dong Gyu Na, Shin Je Moon, Dong-Jun Lim, Kyong Yeun Jung, Yun Jae Chung, Chan Kwon Jung, Young Joo Park
International Journal of Thyroidology.2024; 17(1): 68. CrossRef - Korean Thyroid Association Management Guidelines for Patients with Thyroid Nodules 2024
Young Joo Park, Eun Kyung Lee, Young Shin Song, Su Hwan Kang, Bon Seok Koo, Sun Wook Kim, Dong Gyu Na, Seung-Kuk Baek, So Won Oh, Min Kyoung Lee, Sang-Woo Lee, Young Ah Lee, Yong Sang Lee, Ji Ye Lee, Dong-Jun Lim, Leehi Joo, Yuh-Seog Jung, Chan Kwon Jung,
International Journal of Thyroidology.2024; 17(1): 208. CrossRef - PD-L1 Expression and Its Modulating Factors in Anaplastic Thyroid Carcinoma
Shipra Agarwal, Chan Kwon Jung, Pranitha Gaddam, Mitsuyoshi Hirokawa, Takuya Higashiyama, Jen-Fan Hang, Wei-An Lai, Somboon Keelawat, Zhiyan Liu, Hee Young Na, So Yeon Park, Junya Fukuoka, Shinya Satoh, Zhanna Mussazhanova, Masahiro Nakashima, Kennichi Ka
American Journal of Surgical Pathology.2024; 48(10): 1233. CrossRef - 2023 Korean Thyroid Association Management Guidelines for Patients with Thyroid Nodules
Young Joo Park, Eun Kyung Lee, Young Shin Song, Soo Hwan Kang, Bon Seok Koo, Sun Wook Kim, Dong Gyu Na, Seung-Kuk Baek, So Won Oh, Min Kyoung Lee, Sang-Woo Lee, Young Ah Lee, Yong Sang Lee, Ji Ye Lee, Dong-Jun Lim, Leehi Joo, Yuh-Seog Jung, Chan Kwon Jung
International Journal of Thyroidology.2023; 16(1): 1. CrossRef - Incidence and risk factors for occult lesions in low-risk papillary thyroid microcarcinoma patients with tumor characteristics appropriate for thermal ablation: A retrospective study
Langping Jin, Kaijun Zhu, Changliang Xu, Jiaying Lu, Liming Huang
Medicine.2023; 102(38): e34938. CrossRef - Identification of NIFTP-Specific mRNA Markers for Reliable Molecular Diagnosis of Thyroid Tumors
So-Yeon Lee, Jong-Lyul Park, Kwangsoon Kim, Ja Seong Bae, Jae-Yoon Kim, Seon-Young Kim, Chan Kwon Jung
Endocrine Pathology.2023; 34(3): 311. CrossRef - Risk factors and predictive model for recurrence in papillary thyroid carcinoma: a single-center retrospective cohort study based on 955 cases
Yin Li, Jiahe Tian, Ke Jiang, Zhongyu Wang, Songbo Gao, Keyang Wei, Ankui Yang, Qiuli Li
Frontiers in Endocrinology.2023;[Epub] CrossRef - BRAFV600E Positivity-Dependent Effect of Age on Papillary Thyroid Cancer Recurrence Risk
Joonseon Park, Solji An, Kwangsoon Kim, Ja Seong Bae, Jeong Soo Kim
Cancers.2023; 15(22): 5395. CrossRef - BRAFV600E mutation test on fine‐needle aspiration specimens of thyroid nodules: Clinical correlations for 4600 patients
Huang Chen, Aiping Song, Ye Wang, Yifan He, Jie Tong, Jinxi Di, Chun Li, Zhongren Zhou, Xiaopin Cai, Dingrong Zhong, Jiping Da
Cancer Medicine.2022; 11(1): 40. CrossRef - Clinicopathological indicators for TERT promoter mutation in papillary thyroid carcinoma
Hee Young Na, Hyeong Won Yu, Woochul Kim, Jae Hoon Moon, Chang Ho Ahn, Sang Il Choi, Yeo Koon Kim, June Young Choi, So Yeon Park
Clinical Endocrinology.2022; 97(1): 106. CrossRef - A Systematic Review and Meta‐analysis on the Occurrence of Biomarker Mutation in Colorectal Cancer among the Asian Population
Hafeez Afolabi, Salzihan Md Salleh, Zaidi Zakaria, Ch’ng Ewe Seng, Siti Norasikin Binti Mohd Nafil, Ahmad Aizat Bin Abdul Aziz, Yusuf Wada, Ahmad Irekeola, Syed Sameer Aga
BioMed Research International.2022;[Epub] CrossRef -
A Significance of Concomitant BRAF
V600E
and TERT Mutations in Polish Patients with Papillary Thyroid Microcarcinoma: A Retrospective Cohort Study Based on 430 Cases
Artur Kuchareczko, Janusz Kopczyński, Artur Kowalik, Kinga Hińcza-Nowak, Agnieszka Walczyk, Iwona Pałyga, Tomasz Trybek, Monika Szymonek, Danuta Gąsior-Perczak, Klaudia Gadawska-Juszczyk, Estera Mikina, Izabela Płachta, Agnieszka Suligowska, Agnieszka Płu
Thyroid®.2022; 32(11): 1372. CrossRef - Machine learning for identifying benign and malignant of thyroid tumors: A retrospective study of 2,423 patients
Yuan-yuan Guo, Zhi-jie Li, Chao Du, Jun Gong, Pu Liao, Jia-xing Zhang, Cong Shao
Frontiers in Public Health.2022;[Epub] CrossRef - TERT Promoter and BRAF V600E Mutations in Papillary Thyroid Cancer: A Single-Institution Experience in Korea
Min Jhi Kim, Jin Kyong Kim, Gi Jeong Kim, Sang-Wook Kang, Jandee Lee, Jong Ju Jeong, Woong Youn Chung, Daham Kim, Kee-Hyun Nam
Cancers.2022; 14(19): 4928. CrossRef - Frequency of TERT Promoter Mutations in Real-World Analysis of 2,092 Thyroid Carcinoma Patients (Endocrinol Metab 2022;37:652-63, Heera Yang et al.)
Sue Youn Kim, Chan Kwon Jung
Endocrinology and Metabolism.2022; 37(6): 947. CrossRef - Frequency of TERT Promoter Mutations in Real-World Analysis of 2,092 Thyroid Carcinoma Patients (Endocrinol Metab 2022;37:652-63, Heera Yang et al.)
Hyunju Park, Jae Hoon Chung
Endocrinology and Metabolism.2022; 37(6): 949. CrossRef - Molecular Pathology of Non-familial Follicular Epithelial–Derived Thyroid Cancer in Adults: From RAS/BRAF-like Tumor Designations to Molecular Risk Stratification
Paula Soares, Antónia Afonso Póvoa, Miguel Melo, João Vinagre, Valdemar Máximo, Catarina Eloy, José Manuel Cameselle-Teijeiro, Manuel Sobrinho-Simões
Endocrine Pathology.2021; 32(1): 44. CrossRef - Clinicopathological Characteristics and Recurrence-Free Survival of Rare Variants of Papillary Thyroid Carcinomas in Korea: A Retrospective Study
Mijin Kim, Sun Wook Cho, Young Joo Park, Hwa Young Ahn, Hee Sung Kim, Yong Joon Suh, Dughyun Choi, Bu Kyung Kim, Go Eun Yang, Il-Seok Park, Ka Hee Yi, Chan Kwon Jung, Bo Hyun Kim
Endocrinology and Metabolism.2021; 36(3): 619. CrossRef - Clinical Application of TERT Promoter Mutations in Urothelial Carcinoma
Yujiro Hayashi, Kazutoshi Fujita, George J. Netto, Norio Nonomura
Frontiers in Oncology.2021;[Epub] CrossRef - MicroRNA Profile for Diagnostic and Prognostic Biomarkers in Thyroid Cancer
Jong-Lyul Park, Seon-Kyu Kim, Sora Jeon, Chan-Kwon Jung, Yong-Sung Kim
Cancers.2021; 13(4): 632. CrossRef - Prospective Analysis of TERT Promoter Mutations in Papillary Thyroid Carcinoma at a Single Institution
Yun-Suk Choi, Seong-Woon Choi, Jin-Wook Yi
Journal of Clinical Medicine.2021; 10(10): 2179. CrossRef - Significance of telomerase reverse-transcriptase promoter mutations in differentiated thyroid cancer
Hung-Fei Lai, Chi-Yu Kuo, Shih-Ping Cheng
Formosan Journal of Surgery.2021; 54(5): 171. CrossRef - Early Diagnosis of Low-Risk Papillary Thyroid Cancer Results Rather in Overtreatment Than a Better Survival
Jolanta Krajewska, Aleksandra Kukulska, Malgorzata Oczko-Wojciechowska, Agnieszka Kotecka-Blicharz, Katarzyna Drosik-Rutowicz, Malgorzata Haras-Gil, Barbara Jarzab, Daria Handkiewicz-Junak
Frontiers in Endocrinology.2020;[Epub] CrossRef
- Summary and Analysis of Molecular Biological Changes, PD-L1 Immune Status and Clinicopathological Features of 78 Cases of Papillary Thyroid Carcinoma (<1 cm in Diameter) Combined With Lateral Cervical Lymph Node Metastasis
- Adjunctive markers for classification and diagnosis of central nervous system tumors: results of a multi-center neuropathological survey in Korea
- Yoon Jin Cha, Se Hoon Kim, Na Rae Kim
- J Pathol Transl Med. 2020;54(2):165-170. Published online February 20, 2020
- DOI: https://doi.org/10.4132/jptm.2020.02.04
- 8,689 View
- 225 Download
- 1 Web of Science
- 1 Crossref
-
 Abstract
Abstract
 PDF
PDF Supplementary Material
Supplementary Material - Background
The revised 4th 2016 World Health Organization (WHO) classification of tumors of the central nervous system (CNS) classification has adopted integrated diagnosis encompassing the histology and molecular features of CNS tumors. We aimed to investigate the immunohistochemistry, molecular testing, and testing methods for diagnosis of CNS tumors in pathological labs of tertiary centers in Korea, and evaluate the adequacy of tests for proper diagnosis in daily practice.
Methods
A survey, composed of eight questions concerning molecular testing for diagnosis of CNS tumors, was sent to 10 neuropathologists working in tertiary centers in Korea.
Results
For diagnosis of astrocytic and oligodendroglial tumors, all 10 centers performed isocitrate dehydrogenase mutations testing and 1p/19q loss of heterozygosity. For glioneuronal tumors, immunohistochemistry (IHC) assays for synaptophysin (n = 9), CD34 (n = 7), BRAF(VE1) (n = 5) were used. For embryonal tumors, particularly in medulloblastoma, four respondents used IHC panel (growth factor receptor bound protein 2-associated protein 1, filamin A, and yes-associated protein 1) for molecular subclassification. Regarding meningioma, all respondents performed Ki-67 IHC and five performed telomerase reverse transcriptase promoter mutation.
Conclusions
Most tertiary centers made proper diagnosis in line with 2016 WHO classification. As classification of CNS tumors has evolved to be more complex and more ancillary tests are required, these should be performed considering the effect of necessity and justification. -
Citations
Citations to this article as recorded by- Exploring the role of epidermal growth factor receptor variant III in meningeal tumors
Rashmi Rana, Vaishnavi Rathi, Kirti Chauhan, Kriti Jain, Satnam Singh Chhabra, Rajesh Acharya, Samir Kumar Kalra, Anshul Gupta, Sunila Jain, Nirmal Kumar Ganguly, Dharmendra Kumar Yadav, Timir Tripathi
PLOS ONE.2021; 16(9): e0255133. CrossRef
- Exploring the role of epidermal growth factor receptor variant III in meningeal tumors
- Double cocktail immunostains with high molecular weight cytokeratin and GATA-3: useful stain to discriminate in situ involvement of prostatic ducts or acini from stromal invasion by urothelial carcinoma in the prostate
- Junghye Lee, Youngeun Yoo, Sanghui Park, Min-Sun Cho, Sun Hee Sung, Jae Y. Ro
- J Pathol Transl Med. 2020;54(2):146-153. Published online February 10, 2020
- DOI: https://doi.org/10.4132/jptm.2019.11.12
- 8,957 View
- 138 Download
- 2 Web of Science
- 2 Crossref
-
 Abstract
Abstract
 PDF
PDF - Background
Distinguishing prostatic stromal invasion (PSI) by urothelial carcinoma (UC) from in situ UC involving prostatic ducts or acini with no stromal invasion (in situ involvement) may be challenging on hematoxylin and eosin stained sections. However, the distinction between them is important because cases with PSI show worse prognosis. This study was performed to assess the utility of double cocktail immunostains with high molecular weight cytokeratin (HMWCK) and GATA-3 to discriminate PSI by UC from in situ UC involvement of prostatic ducts or acini in the prostate.
Methods
Among 117 radical cystoprostatectomy specimens for bladder UCs, 25 cases showed secondary involvement of bladder UC in prostatic ducts/acini only or associated stromal invasion and of these 25 cases, seven cases revealed equivocal PSI. In these seven cases with equivocal PSI, HMWCK, and GATA-3 double immunohistochemical stains were performed to identify whether this cocktail stain is useful to identify the stromal invasion.
Results
In all cases, basal cells of prostate glands showed strong cytoplasmic staining for HMWCK and UC cells showed strong nuclear staining for GATA-3. In cases with stromal invasion of UC, GATA-3-positive tumor cells in the prostatic stroma without surrounding HMWCK-positive basal cells were highlighted and easily recognized. Among seven equivocal cases, two cases showed PSI and five in situ UC in the prostate. In two cases, the original diagnoses were revised.
Conclusions
Our study suggested that HMWCK and GATA-3 double stains could be utilized as an adjunct method in the distinction between PSI by UC from in situ UC involving prostatic ducts or acini. -
Citations
Citations to this article as recorded by- Aberrant expression of GATA3 in metastatic adenocarcinoma of the prostate: an important pitfall
João Lobo, Nazario P Tenace, Sofia Cañete‐Portillo, Isa Carneiro, Rui Henrique, Roberta Lucianò, Lara R Harik, Cristina Magi‐Galluzzi
Histopathology.2024; 84(3): 507. CrossRef - Utility of D2-40, Cytokeratin 5/6, and High–Molecular-weight Cytokeratin (Clone 34βE12) in Distinguishing Intraductal Spread of Urothelial Carcinoma From Prostatic Stromal Invasion
Oleksii A. Iakymenko, Laurence M. Briski, Katiana S. Delma, Merce Jorda, Oleksandr N. Kryvenko
American Journal of Surgical Pathology.2022; 46(4): 454. CrossRef
- Aberrant expression of GATA3 in metastatic adenocarcinoma of the prostate: an important pitfall
- Molecular characteristics of meningiomas
- Young Suk Lee, Youn Soo Lee
- J Pathol Transl Med. 2020;54(1):45-63. Published online January 15, 2020
- DOI: https://doi.org/10.4132/jptm.2019.11.05
- 23,557 View
- 692 Download
- 51 Web of Science
- 57 Crossref
-
 Abstract
Abstract
 PDF
PDF - Meningioma is the most common primary intracranial tumor in adults. The grading of meningioma is based on World Health Organization criteria, which rely on histopathological features alone. This grading system is unable to conclusively predict the clinical behavior of these tumors (i.e., recurrence or prognosis in benign or atypical grades). Advances in molecular techniques over the last decade that include genomic and epigenomic data associated with meningiomas have been used to identify genetic biomarkers that can predict tumor behavior. This review summarizes the molecular characteristics of meningioma using genetic and epigenetic biomarkers. Molecular alterations that can predict meningioma behavior may be integrated into the upcoming World Health Organization grading system.
-
Citations
Citations to this article as recorded by- Multi-Institutional Modified Delphi For Genomics in Expert Consensus Survey of Genomic Testing for Anterior Skull Base Malignancies
Anirudh Saraswathula, Shreya Sriram, Corinna Levine, Nyall R. London, Shirley Y. Su, Mathew Geltzeiler, Sanjeet V. Rangarajan, Ian Witterick, Brian Thorp, Kathleen Kelly Gallagher, Kenneth Byrd, Ricardo Carrau, Waleed Abuzeid, Eric Wang, Carl Snyderman, E
Journal of Neurological Surgery Part B: Skull Base.2026; 87(01): 014. CrossRef - Long-Term Prognostic Impact of the Dural Tail in the Local Control of Intracranial Meningiomas
Franco Rubino, Romulo A. de Almeida, Komal Shah, Shaan M. Raza, Franco DeMonte
Journal of Neurological Surgery Part B: Skull Base.2026;[Epub] CrossRef - Recurrence of Resected Skull Base Meningiomas during Long-term Follow-up: Incidence and Predisposing Factors
Joshua Ian Macarthur, Cathal John Hannan, Callum Howard, Jane Halliday, Omar Nathan Pathmanaban, Charlotte Hammerbeck-Ward, Scott A. Rutherford, Andrew T. King
Journal of Neurological Surgery Part B: Skull Base.2025; 86(03): 245. CrossRef - Role of H3K27me3 and Ki-67 Labeling Index in Assessing the Biological Behavior of Meningiomas
Shalaka Deshpande, Bhavna Nayal, Rajesh Nair, Deepak Nayak, Padmapriya J, Geetha V
World Neurosurgery.2025; 194: 123514. CrossRef - Context aware machine learning techniques for brain tumor classification and detection – A review
Usman Amjad, Asif Raza, Muhammad Fahad, Doaa Farid, Adnan Akhunzada, Muhammad Abubakar, Hira Beenish
Heliyon.2025; 11(2): e41835. CrossRef - Case report: Clonal evolution analysis of a rare case of meningioma lung metastases identifies actionable alterations in matched longitudinal tumour samples
Nicola Cosgrove, Orla M. Fitzpatrick, Liam Grogan, Bryan T. Hennessy, Simon J. Furney, Sinead Toomey
Frontiers in Oncology.2025;[Epub] CrossRef - Post-operative Hemorrhage After Tumor Removal of Multiple Meningiomas
Bob Irfan Syahputra, Akhmad Imron, Dhany Febriantara, Helza Efriani
International Journal of Recent Surgical and Medical Sciences.2025; 11: e005. CrossRef - Diarylpentanoid, a curcumin analog, inhibits malignant meningioma growth in both in vitro and in vivo models
Anna Terasawa, Kazuhiro Shimazu, Hiroshi Nanjo, Masatomo Miura, Hiroyuki Shibata
World Journal of Experimental Medicine.2025;[Epub] CrossRef - Tumour-associated macrophages in human meningiomas
Rahmina Meta, Henrik Sahlin Pettersen, Sofie Eline Tollefsen, Borgny Ytterhus, Øyvind Olav Salvesen, Wenche Sjursen, Sverre Helge Torp, Jianhong Zhou
PLOS One.2025; 20(5): e0319960. CrossRef - Binary Classification of Meningioma Grades Using CNN and VGG16 + XGBoost Deep Learning Models
L. Priya, D. Saraswathi, M. Bhuvaneshwari, K. Dhanya, K. Krishna Kousalya, Deepali
Applied Computational Intelligence and Soft Computing.2025;[Epub] CrossRef - Unusual adrenal metastasis of anaplastic meningioma: A case report
M. Inouss, C. Rhoul, A. Kharkhach, T. Bouhout, B. Serji
International Journal of Surgery Case Reports.2025;[Epub] CrossRef - TERT promoter mutations in meningiomas: associations with clinicopathological features and insights into spatial and temporal heterogeneity in a 165-case cohort
Ahmet Boduroğlu, Mualla Özcan, Güzide Ayşe Ocak
Pathology - Research and Practice.2025; 274: 156191. CrossRef - The Gut–Brain Axis in Brain Tumors: Insights into Tumor Development, Progression, and Therapy
Sarah Adriana Scuderi, Alessio Ardizzone, Elsa Calcaterra, Nicoletta Palermo, Fabiola De Luca, Antonio Catalfamo, Emanuela Esposito, Anna Paola Capra
Biomedicines.2025; 13(9): 2172. CrossRef - The Natural History and Treatment of Meningiomas: An Update
Arsene Daniel Nyalundja, Fabrice Mugisha, Claire Karekezi
Seminars in Neurology.2024; 44(01): 001. CrossRef - Epidemiology, Genetics, and DNA Methylation Grouping of Hyperostotic Meningiomas
Gray Umbach, Edwina B. Tran, Charlotte D. Eaton, Abrar Choudhury, Ramin Morshed, Javier E. Villanueva-Meyer, Philip V. Theodosopoulos, Stephen T. Magill, Michael W. McDermott, David R. Raleigh, Ezequiel Goldschmidt
Operative Neurosurgery.2024; 26(6): 662. CrossRef - The Evolving Classification of Meningiomas: Integration of Molecular Discoveries to Inform Patient Care
S. Joy Trybula, Mark W. Youngblood, Constantine L. Karras, Nikhil K. Murthy, Amy B. Heimberger, Rimas V. Lukas, Sean Sachdev, John A. Kalapurakal, James P. Chandler, Daniel J. Brat, Craig M. Horbinski, Stephen T. Magill
Cancers.2024; 16(9): 1753. CrossRef - Minimally Invasive Approaches in the Surgical Treatment of Intracranial Meningiomas: An Analysis of 54 Cases
Guenther C. Feigl, Daniel Staribacher, Gavin Britz, Dzmitry Kuzmin
Brain Tumor Research and Treatment.2024; 12(2): 93. CrossRef - Related mechanisms, current treatments, and new perspectives in meningioma
Gizem Inetas‐Yengin, Omer Faruk Bayrak
Genes, Chromosomes and Cancer.2024;[Epub] CrossRef - Clinical application of intraoperative ultrasound superb microvascular imaging in brain tumors resections: contributing to the achievement of total tumoral resection
Siman Cai, Hao Xing, Yuekun Wang, Yu Wang, Wenbin Ma, Yuxin Jiang, Jianchu Li, Hongyan Wang
BMC Medical Imaging.2024;[Epub] CrossRef - WHO CNS 5 and meningiomas: What’s new?
Indranil Chakrabarti, Sujaya Mazumder
IP Archives of Cytology and Histopathology Research.2024; 9(2): 67. CrossRef - Molecular Developments in Parasellar Tumors and Potential Therapeutic Implications
Paraskevi Xekouki, Vasiliki Venetsanaki, Georgios Kyriakopoulos, Krystallenia Alexandraki, Anna Angelousi, Gregory Kaltsas
Endocrine Reviews.2024; 45(6): 880. CrossRef - The Impact of Molecular and Genetic Analysis on the Treatment of Patients with Atypical Meningiomas
Janez Ravnik, Hojka Rowbottom
Diagnostics.2024; 14(16): 1782. CrossRef - Differential Expression of Proteins and Genes at the Tumor‐Brain Interface in Invasive Meningioma
Kornwika Senglek, Chinachote Teerapakpinyo, Nutchawan Jittapiromsak, Pakrit Jittapiromsak, Irin Lertparinyaphorn, Paul Scott Thorner, Shanop Shuangshoti
Genes, Chromosomes and Cancer.2024;[Epub] CrossRef - Protein expression of CD44 in patients with meningioma tumors: association with clinicopathological parameters and survival
Trupti Trivedi, Neha Bhalala, Kirti Dialani, Priti Trivedi
Journal of the Egyptian National Cancer Institute.2024;[Epub] CrossRef - DNA methylation profiling of meningiomas highlights clinically distinct molecular subgroups
Jyotsna Singh, Ravi Sharma, Nidhi Shukla, Priya Narwal, Amit Katiyar, Swati Mahajan, Saumya Sahu, Ajay Garg, Mehar C. Sharma, Ashish Suri, Chitra sarkar, Vaishali Suri
Journal of Neuro-Oncology.2023; 161(2): 339. CrossRef - Spinal meningiomas, from biology to management - A literature review
Nicolas Serratrice, Imène Lameche, Christian Attieh, Moussa A Chalah, Joe Faddoul, Bilal Tarabay, Rabih Bou-Nassif, Youssef Ali, Joseph G Mattar, François Nataf, Samar S Ayache, Georges N Abi Lahoud
Frontiers in Oncology.2023;[Epub] CrossRef - Somatic mutation landscape in a cohort of meningiomas that have undergone grade progression
Sarah A Cain, Bernard Pope, Stefano Mangiola, Theo Mantamadiotis, Katharine J Drummond
BMC Cancer.2023;[Epub] CrossRef - Actualización sobre el meningioma: correlación clínico-radiológica y radio-patológica
A. Navarro-Ballester, M. Aleixandre-Barrachina, S.F. Marco-Doménech
Radiología.2023; 65(5): 458. CrossRef - SMARCE1-related meningiomas: A clear example of cancer predisposing syndrome
Erika Fiorentini, Laura Giunti, Andrea Di Rita, Simone Peraio, Carla Fonte, Chiara Caporalini, Anna Maria Buccoliero, Maria Luigia Censullo, Giulia Gori, Alice Noris, Rosa Pasquariello, Roberta Battini, Rossana Pavone, Flavio Giordano, Sabrina Giglio, Ber
European Journal of Medical Genetics.2023; 66(7): 104784. CrossRef - Grade scoring system reveals distinct molecular subtypes and identifies KIF20A as a novel biomarker for predicting temozolomide treatment efficiency in gliomas
Liguo Ye, Shi’ao Tong, Yaning Wang, Yu Wang, Wenbin Ma
Journal of Cancer Research and Clinical Oncology.2023; 149(12): 9857. CrossRef - Integrated clinical genomic analysis reveals xenobiotic metabolic genes are downregulated in meningiomas of current smokers
A. Basit Khan, Rajan Patel, Malcolm F. McDonald, Eric Goethe, Collin English, Ron Gadot, Arya Shetty, Shervin Hosseingholi Nouri, Arif O. Harmanci, Akdes S. Harmanci, Tiemo J. Klisch, Akash J. Patel
Journal of Neuro-Oncology.2023; 163(2): 397. CrossRef - DNA methylation meningioma biomarkers: attributes and limitations
Zhaohui Li, Yufei Gao, Jinnan Zhang, Liang Han, Hang Zhao
Frontiers in Molecular Neuroscience.2023;[Epub] CrossRef - Meningioma: A Biography—Tumor Forever Tied to the Origins and “Soul of Neurosurgery”
Nolan J. Brown, Zach Pennington, Cathleen C. Kuo, Julian Gendreau, Sachiv Chakravarti, Rohin Singh, Dontré M. Douse, Jamie J. Van Gompel
World Neurosurgery.2023; 178: 191. CrossRef - Molecular genetic features of meningiomas
E.S. Makashova, N.V. Lasunin, M.V. Galkin, S.V. Zolotova, K.O. Karandasheva, A.V. Golanov
Burdenko's Journal of Neurosurgery.2023; 87(4): 101. CrossRef - Novel Advances in Treatment of Meningiomas: Prognostic and Therapeutic Implications
Gerardo Caruso, Rosamaria Ferrarotto, Antonello Curcio, Luisa Metro, Francesco Pasqualetti, Paola Gaviani, Valeria Barresi, Filippo Flavio Angileri, Maria Caffo
Cancers.2023; 15(18): 4521. CrossRef - Update on meningioma: Clinical-radiological and radio-pathological correlation
A. Navarro-Ballester, M. Aleixandre-Barrachina, S.F. Marco-Doménech
Radiología (English Edition).2023; 65(5): 458. CrossRef - Early Preventive Strategies and CNS Meningioma – Is This Feasible? A Comprehensive Review of the Literature
Daniel Sescu, Aminta Chansiriwongs, Katarzyna Julia Minta, Jyothi Vasudevan, Chandrasekaran Kaliaperumal
World Neurosurgery.2023; 180: 123. CrossRef - Domestic Animal Models of Central Nervous System Tumors: Focus on Meningiomas
Michele Tomanelli, Tullio Florio, Gabriela Vargas, Aldo Pagano, Paola Modesto
Life.2023; 13(12): 2284. CrossRef - Assessment of parameters of the acid-base state among patients with meningiomas and gliomas in the postoperative period
E. S. Orlova, I. O. Ishchenko, K. K. Kukanov, N. E. Voinov, A. P. Gerasimov, N. E. Ivanova
Russian Neurosurgical Journal named after Professor A. L. Polenov.2023; 15(2): 21. CrossRef - The integrated multiomic diagnosis of sporadic meningiomas: a review of its clinical implications
Stephanie M. Robert, Shaurey Vetsa, Arushii Nadar, Sagar Vasandani, Mark W. Youngblood, Evan Gorelick, Lan Jin, Neelan Marianayagam, E Zeynep Erson-Omay, Murat Günel, Jennifer Moliterno
Journal of Neuro-Oncology.2022; 156(2): 205. CrossRef - Clinical presentation, diagnostic findings and outcome of dogs undergoing surgical resection for intracranial meningioma: 101 dogs
Alexander K. Forward, Holger Andreas Volk, Giunio Bruto Cherubini, Tom Harcourt-Brown, Ioannis N. Plessas, Laurent Garosi, Steven De Decker
BMC Veterinary Research.2022;[Epub] CrossRef - Sphenoid wing meningiomas: peritumoral brain edema as a prognostic factor in surgical outcome
Abdalrahman Nassar, Volodymyr Smolanka, Andriy Smolanka, Dipak Chaulagain, Oleg Devinyak
Neurosurgical Review.2022; 45(4): 2951. CrossRef - Potential Molecular Mechanisms of Recurrent and Progressive Meningiomas: A Review of the Latest Literature
Wenjie Peng, Pei Wu, Minghao Yuan, Bo Yuan, Lian Zhu, Jiesong Zhou, Qian Li
Frontiers in Oncology.2022;[Epub] CrossRef - Case Report: Upper Thoracic Purely Extradural Spinal Meningioma With Nerve Root Attachment: A Case Report and Literature Review
Zhao-Lin Wang, Jian-Hui Mou, Dong Sun, Peng Liu
Frontiers in Surgery.2022;[Epub] CrossRef - Molecular diagnosis and treatment of meningiomas: an expert consensus (2022)
Jiaojiao Deng, Lingyang Hua, Liuguan Bian, Hong Chen, Ligang Chen, Hongwei Cheng, Changwu Dou, Dangmurenjiapu Geng, Tao Hong, Hongming Ji, Yugang Jiang, Qing Lan, Gang Li, Zhixiong Liu, Songtao Qi, Yan Qu, Songsheng Shi, Xiaochuan Sun, Haijun Wang, Yongpi
Chinese Medical Journal.2022; 135(16): 1894. CrossRef - Оновлена інформація про менінгіоми крила клиноподібної кістки
Abdalrahman Nassar, Volodymyr Smolanka
INTERNATIONAL NEUROLOGICAL JOURNAL.2022; 18(1): 43. CrossRef - The Prognostic Value of Methylation Signatures and NF2 Mutations in Atypical Meningiomas
Rahmina Meta, Henning B. Boldt, Bjarne W. Kristensen, Felix Sahm, Wenche Sjursen, Sverre H. Torp
Cancers.2021; 13(6): 1262. CrossRef - Neurofibromatosis Type 2 (NF2) and the Implications for Vestibular Schwannoma and Meningioma Pathogenesis
Suha Bachir, Sanjit Shah, Scott Shapiro, Abigail Koehler, Abdelkader Mahammedi, Ravi N. Samy, Mario Zuccarello, Elizabeth Schorry, Soma Sengupta
International Journal of Molecular Sciences.2021; 22(2): 690. CrossRef - Meningioma: A Review of Epidemiology, Pathology, Diagnosis, Treatment, and Future Directions
Christian Ogasawara, Brandon D. Philbrick, D. Cory Adamson
Biomedicines.2021; 9(3): 319. CrossRef - The substantial loss of H3K27me3 can stratify risk in grade 2, but not in grade 3 meningioma
Minsun Jung, Seong-Ik Kim, Ka Young Lim, Jeongmo Bae, Chul-Kee Park, Seung Hong Choi, Sung-Hye Park, Jae-Kyung Won
Human Pathology.2021; 115: 96. CrossRef - Papillary Meningioma: Case Presentation with Emphasis on Surgical and Medical Therapy of a Rare Variant of Meningioma
Gerardo Cazzato, Valeria Internò, Antonietta Cimmino, Raffaella Messina, Marco Tucci, Teresa Lettini, Leonardo Resta, Giuseppe Ingravallo
Diseases.2021; 9(3): 63. CrossRef - An Overview of Managements in Meningiomas
Lianhua Zhao, Wei Zhao, Yanwei Hou, Cuixia Wen, Jing Wang, Pei Wu, Zaiyu Guo
Frontiers in Oncology.2020;[Epub] CrossRef - Multi-Omics Analysis in Initiation and Progression of Meningiomas: From Pathogenesis to Diagnosis
Jiachen Liu, Congcong Xia, Gaiqing Wang
Frontiers in Oncology.2020;[Epub] CrossRef - Molecular Mechanism and Approach in Progression of Meningioma
Zhiwei Shao, Lihong Liu, Yanghao Zheng, Sheng Tu, Yuanbo Pan, Sheng Yan, Qichun Wei, Anwen Shao, Jianmin Zhang
Frontiers in Oncology.2020;[Epub] CrossRef - Multiple meningiomas: does quantity matter? a population-based survival analysis with underlined age and sex differences
Andres Ramos-Fresnedo, Ricardo A. Domingo, Tito Vivas-Buitrago, Larry Lundy, Daniel M. Trifiletti, Mark E. Jentoft, Amit B. Desai, Alfredo Quiñones-Hinojosa
Journal of Neuro-Oncology.2020; 149(3): 413. CrossRef - Meningioma: A Review of Clinicopathological and Molecular Aspects
Kristin Huntoon, Angus Martin Shaw Toland, Sonika Dahiya
Frontiers in Oncology.2020;[Epub] CrossRef - Neues zur Einteilung und Therapie von Meningeomen
Corinna Seliger, Wolfgang Wick
Neurologie up2date.2020; 3(04): 343. CrossRef
- Multi-Institutional Modified Delphi For Genomics in Expert Consensus Survey of Genomic Testing for Anterior Skull Base Malignancies
- The Prognostic Impact of Synchronous Ipsilateral Multiple Breast Cancer: Survival Outcomes according to the Eighth American Joint Committee on Cancer Staging and Molecular Subtype
- Jinah Chu, Hyunsik Bae, Youjeong Seo, Soo Youn Cho, Seok-Hyung Kim, Eun Yoon Cho
- J Pathol Transl Med. 2018;52(6):396-403. Published online October 23, 2018
- DOI: https://doi.org/10.4132/jptm.2018.10.03
- 8,790 View
- 103 Download
- 8 Web of Science
- 10 Crossref
-
 Abstract
Abstract
 PDF
PDF - Background
In the current American Joint Committee on Cancer staging system of breast cancer, only tumor size determines T-category regardless of whether the tumor is single or multiple. This study evaluated if tumor multiplicity has prognostic value and can be used to subclassify breast cancer.
Methods
We included 5,758 patients with invasive breast cancer who underwent surgery at Samsung Medical Center, Seoul, Korea, from 1995 to 2012.
Results
Patients were divided into two groups according to multiplicity (single, n = 4,744; multiple, n = 1,014). Statistically significant differences in lymph node involvement and lymphatic invasion were found between the two groups (p < .001). Patients with multiple masses tended to have luminal A molecular subtype (p < .001). On Kaplan-Meier survival analysis, patients with multiple masses had significantly poorer disease-free survival (DFS) (p = .016). The prognostic significance of multiplicity was seen in patients with anatomic staging group I and prognostic staging group IA (p = .019 and p = .032, respectively). When targeting patients with T1-2 N0 M0, hormone receptor–positive, and human epidermal growth factor receptor 2 (HER2)–negative cancer, Kaplan-Meier survival analysis also revealed significantly reduced DFS with multiple cancer (p = .031). The multivariate analysis indicated that multiplicity was independently correlated with worse DFS (hazard ratio, 1.23; 95% confidence interval, 1.03 to 1.47; p = .025). The results of this study indicate that tumor multiplicity is frequently found in luminal A subtype, is associated with frequent lymph node metastasis, and is correlated with worse DFS.
Conclusions
Tumor multiplicity has prognostic value and could be used to subclassify invasive breast cancer at early stages. Adjuvant chemotherapy would be necessary for multiple masses of T1–2 N0 M0, hormone-receptor-positive, and HER2-negative cancer. -
Citations
Citations to this article as recorded by- Beyond Pathology: MRI-Derived Metrics Unlock Preoperative Prognostic Risk Stratification in Breast Cancer
Mehrad Zare Gholinejad, Alisa Mohebbi, Afshin Mohammadi, Seyed Mohammad Tavangar
Clinical Breast Cancer.2026;[Epub] CrossRef - The Role of Serum Beta-Human Chorionic Gonadotropin (β-hCG) in Differentiating Benign and Malignant Breast Lesions at a Tertiary Care Center in Jharkhand
Neyaz Ahmad, Khushboo Rani, Zenith Kerketta, Krishna Murari, Anish Baxla, Ujala Murmu, Amit Nishant, Shreya .
Cureus.2025;[Epub] CrossRef - Role of Large Format Histology in Diagnosis of Breast Carcinoma
Hari Shankar Pandey, Sanya Bhasin, Suman Kumari Pandey
NMO Journal.2025; 19(2): 189. CrossRef - Prognostic Impact of Multiple Synchronous T1 Breast Cancer
Hongki Gwak, Sung Hoo Jung, Young Jin Suh, Seok Jin Nam, Jai Hong Han, Se Jeong Oh, Eun Hwa Park, Seong Hwan Kim
Cancers.2024; 16(23): 4019. CrossRef - Deep learning-based system for automatic prediction of triple-negative breast cancer from ultrasound images
Alexandre Boulenger, Yanwen Luo, Chenhui Zhang, Chenyang Zhao, Yuanjing Gao, Mengsu Xiao, Qingli Zhu, Jie Tang
Medical & Biological Engineering & Computing.2023; 61(2): 567. CrossRef - Multicentre prospective cohort study of unmet supportive care needs among patients with breast cancer throughout their cancer treatment trajectory in Penang: a PenBCNeeds Study protocol
Noorsuzana Mohd Shariff, Nizuwan Azman, Rohayu Hami, Noor Mastura Mohd Mujar, Mohammad Farris Iman Leong Bin Abdullah
BMJ Open.2021; 11(3): e044746. CrossRef - The subgross morphology of breast carcinomas: a single-institution series of 2033 consecutive cases documented in large-format histology slides
Tibor Tot, Maria Gere, Syster Hofmeyer, Annette Bauer, Ulrika Pellas
Virchows Archiv.2020; 476(3): 373. CrossRef - Editorial for “Synchronous Breast Cancer: Phenotypic Similarities on MRI”
Uma Sharma
Journal of Magnetic Resonance Imaging.2020; 52(1): 309. CrossRef - Synchronous Multiple Breast Cancers—Do We Need to Reshape Staging?
Minodora Onisâi, Adrian Dumitru, Iuliana Iordan, Cătălin Aliuș, Oana Teodor, Adrian Alexandru, Daniela Gheorghiță, Iulian Antoniac, Adriana Nica, Alexandra-Ana Mihăilescu, Sebastian Grădinaru
Medicina.2020; 56(5): 230. CrossRef - Molecular mechanism of triple‑negative breast cancer‑associated BRCA1 and the identification of signaling pathways
Feng Qi, Wen‑Xing Qin, Yuan‑Sheng Zang
Oncology Letters.2019;[Epub] CrossRef
- Beyond Pathology: MRI-Derived Metrics Unlock Preoperative Prognostic Risk Stratification in Breast Cancer
- Protein Phosphatase Magnesium-Dependent 1δ (PPM1D) Expression as a Prognostic Marker in Adult Supratentorial Diffuse Astrocytic and Oligodendroglial Tumors
- Hui Jeong Jeong, Chang Gok Woo, Bora Lee, Shin Kwang Khang, Soo Jeong Nam, Jene Choi
- J Pathol Transl Med. 2018;52(2):71-78. Published online October 18, 2017
- DOI: https://doi.org/10.4132/jptm.2017.10.21
- 9,413 View
- 201 Download
- 2 Web of Science
- 2 Crossref
-
 Abstract
Abstract
 PDF
PDF Supplementary Material
Supplementary Material - Background
Protein phosphatase magnesium-dependent 1δ (PPM1D) is a p53-induced serine/ threonine phosphatase, which is overexpressed in various human cancers. A recent study reported that a mutation in the PPM1D gene is associated with poor prognosis in brainstem gliomas. In this study, we evaluated the utility of PPM1D as a prognostic biomarker of adult supratentorial diffuse astrocytic and oligodendroglial tumors.
Methods
To investigate PPM1D protein expression, mRNA expression, and copy number changes, immunohistochemistry, RNAscope in situ hybridization, and fluorescence in situ hybridization were performed in 84 adult supratentorial diffuse gliomas. We further analyzed clinical characteristics and overall survival (OS) according to PPM1D protein expression, and examined its correlation with other glioma biomarkers such as isocitrate dehydrogenase (IDH) mutation, and p53 expression.
Results
Forty-six cases (54.8%) were PPM1D-positive. PPM1D expression levels were significantly correlated with PPM1D transcript levels (p= .035), but marginally with PPM1D gene amplification (p=.079). Patients with high-grade gliomas showed a higher frequency of PPM1D expression than those with low-grade gliomas (p <.001). Multivariate analysis demonstrated that PPM1D expression (hazard ratio [HR], 2.58; p=.032), age over 60 years (HR, 2.55; p=.018), and IDH1 mutation (HR, 0.18; p=.002) were significantly independent prognostic factors; p53 expression had no prognostic significance (p=.986). The patients with tumor expressing PPM1D showed a shorter OS (p=.003). Moreover, patients with tumor harboring wild-type IDH1 and PPM1D expression had the worst OS (p<.001).
Conclusions
Our data suggest that a subset of gliomas express PPM1D; PPM1D expression is a significant marker of poor prognosis in adult supratentorial diffuse astrocytic and oligodendroglial tumors. -
Citations
Citations to this article as recorded by- Characteristic analysis and identification of novel molecular biomarkers in elderly glioblastoma patients using the 2021 WHO Classification of Central Nervous System Tumors
Yaning Wang, Junlin Li, Yaning Cao, Wenlin Chen, Hao Xing, Xiaopeng Guo, Yixin Shi, Yuekun Wang, Tingyu Liang, Liguo Ye, Delin Liu, Tianrui Yang, Yu Wang, Wenbin Ma
Frontiers in Neuroscience.2023;[Epub] CrossRef - Metal-dependent Ser/Thr protein phosphatase PPM family: Evolution, structures, diseases and inhibitors
Rui Kamada, Fuki Kudoh, Shogo Ito, Itsumi Tani, Jose Isagani B. Janairo, James G. Omichinski, Kazuyasu Sakaguchi
Pharmacology & Therapeutics.2020; 215: 107622. CrossRef
- Characteristic analysis and identification of novel molecular biomarkers in elderly glioblastoma patients using the 2021 WHO Classification of Central Nervous System Tumors
- Molecular Testing of Brain Tumor
- Sung-Hye Park, Jaekyung Won, Seong-Ik Kim, Yujin Lee, Chul-Kee Park, Seung-Ki Kim, Seung-Hong Choi
- J Pathol Transl Med. 2017;51(3):205-223. Published online May 12, 2017
- DOI: https://doi.org/10.4132/jptm.2017.03.08
- 37,222 View
- 1,179 Download
- 40 Web of Science
- 48 Crossref
-
 Abstract
Abstract
 PDF
PDF - The World Health Organization (WHO) classification of central nervous system (CNS) tumors was revised in 2016 with a basis on the integrated diagnosis of molecular genetics. We herein provide the guidelines for using molecular genetic tests in routine pathological practice for an accurate diagnosis and appropriate management. While astrocytomas and IDH-mutant (secondary) glioblastomas are characterized by the mutational status of IDH, TP53, and ATRX, oligodendrogliomas have a 1p/19q codeletion and mutations in IDH, CIC, FUBP1, and the promoter region of telomerase reverse transcriptase (TERTp). IDH-wildtype (primary) glioblastomas typically lack mutations in IDH, but are characterized by copy number variations of EGFR, PTEN, CDKN2A/B, PDGFRA, and NF1 as well as mutations of TERTp. High-grade pediatric gliomas differ from those of adult gliomas, consisting of mutations in H3F3A, ATRX, and DAXX, but not in IDH genes. In contrast, well-circumscribed low-grade neuroepithelial tumors in children, such as pilocytic astrocytoma, pleomorphic xanthoastrocytoma, and ganglioglioma, often have mutations or activating rearrangements in the BRAF, FGFR1, and MYB genes. Other CNS tumors, such as ependymomas, neuronal and glioneuronal tumors, embryonal tumors, meningothelial, and other mesenchymal tumors have important genetic alterations, many of which are diagnostic, prognostic, and predictive markers and therapeutic targets. Therefore, the neuropathological evaluation of brain tumors is increasingly dependent on molecular genetic tests for proper classification, prediction of biological behavior and patient management. Identifying these gene abnormalities requires cost-effective and high-throughput testing, such as next-generation sequencing. Overall, this paper reviews the global guidelines and diagnostic algorithms for molecular genetic testing of brain tumors.
-
Citations
Citations to this article as recorded by- Low-grade Epilepsy-associated Tumors and Other Acquired Causes of Epilepsy
Horst Urbach, Soroush Doostkam
Neuroimaging Clinics of North America.2026; 36(2): 273. CrossRef - Barriers to care to pediatric brain tumors in low- and middle-income countries: A proposal for a critical framework
Savi Agarwal, Jorge Lazareff
Surgical Neurology International.2026; 17: 154. CrossRef - Navigating rare vascular brain tumors: A retrospective observational study
Sana Ahuja, Dipanker S Mankotia, Naveen Kumar, Vyomika Teckchandani, Sufian Zaheer
Cancer Research, Statistics, and Treatment.2025; 8(2): 92. CrossRef - Author’s reply to Dagar and Mallick
Sana Ahuja, Sufian Zaheer
Cancer Research, Statistics, and Treatment.2025; 8(3): 245. CrossRef - The Clinical Utility of Cell-Free DNA in Brain Tumor Management: A Comprehensive Review
Qama Abuhassan, Hanan Hassan Ahmed, Radhwan Abdul Kareem, Soumya V. Menon, Priya Priyadarshini Nayak, J. Bethanney Janney, Vimal Arora, Aashna Sinha, Saif Aldeen Jaber, Hayder Naji Sameer, Ahmed Yaseen, Zainab H. Athab, Mohaned Adil
Journal of Molecular Neuroscience.2025;[Epub] CrossRef - PTEN regulates expression of its pseudogene in glioblastoma cells in DNA methylation-dependent manner
Tatyana F. Kovalenko, Bhupender Yadav, Ksenia S. Anufrieva, Tatyana D. Larionova, Tatiana E. Aksinina, Yaroslav A. Latyshev, Soniya Bastola, Michail I. Shakhparonov, Amit Kumar Pandey, Marat S. Pavlyukov
Biochimie.2024; 219: 74. CrossRef - CDKN2A/B deletion in IDH-mutant astrocytomas: An evaluation by Fluorescence in-situ hybridization
Manali Ranade, Sridhar Epari, Omshree Shetty, Sandeep Dhanavade, Sheetal Chavan, Ayushi Sahay, Arpita Sahu, Prakash Shetty, Aliasgar Moiyadi, Vikash Singh, Archya Dasgupta, Abhishek Chatterjee, Sadhana Kannan, Tejpal Gupta
Journal of Neuro-Oncology.2024; 167(1): 189. CrossRef - Central neurocytoma exhibits radial glial cell signatures with FGFR3 hypomethylation and overexpression
Yeajina Lee, Tamrin Chowdhury, Sojin Kim, Hyeon Jong Yu, Kyung-Min Kim, Ho Kang, Min-Sung Kim, Jin Wook Kim, Yong-Hwy Kim, So Young Ji, Kihwan Hwang, Jung Ho Han, Jinha Hwang, Seong-Keun Yoo, Kyu Sang Lee, Gheeyoung Choe, Jae-Kyung Won, Sung-Hye Park, Yon
Experimental & Molecular Medicine.2024; 56(4): 975. CrossRef - Liquid biopsy in brain tumors: Potential for impactful clinical applications
Tania Eid, Lina Ghandour, Joseph Abi Ghanem, Hazem Assi, Rami Mahfouz
Human Gene.2024; 42: 201333. CrossRef - Diagnostic challenges in complicated case of glioblastoma
Tatiana Aghova, Halka Lhotska, Libuse Lizcova, Karla Svobodova, Lucie Hodanova, Karolina Janeckova, Kim Vucinic, Martin Gregor, Dora Konecna, Filip Kramar, Jiri Soukup, David Netuka, Zuzana Zemanova
Pathology and Oncology Research.2024;[Epub] CrossRef - Drugging Hijacked Kinase Pathways in Pediatric Oncology: Opportunities and Current Scenario
Marina Ferreira Candido, Mariana Medeiros, Luciana Chain Veronez, David Bastos, Karla Laissa Oliveira, Julia Alejandra Pezuk, Elvis Terci Valera, María Sol Brassesco
Pharmaceutics.2023; 15(2): 664. CrossRef - BrainBase: a curated knowledgebase for brain diseases
Lin Liu, Yang Zhang, Guangyi Niu, Qianpeng Li, Zhao Li, Tongtong Zhu, Changrui Feng, Xiaonan Liu, Yuansheng Zhang, Tianyi Xu, Ruru Chen, Xufei Teng, Rongqin Zhang, Dong Zou, Lina Ma, Zhang Zhang
Nucleic Acids Research.2022; 50(D1): D1131. CrossRef - A heat shock protein 90 inhibitor reduces oncoprotein expression and induces cell death in heterogeneous glioblastoma cells with EGFR, PDGFRA, CDK4, and NF1 aberrations
Kuan-Ta Ho, Pei-Fan Chen, Jian-Ying Chuang, Po-Wu Gean, Yuan-Shuo Hsueh
Life Sciences.2022; 288: 120176. CrossRef - Bicentric validation of the navigated transcranial magnetic stimulation motor risk stratification model
Tizian Rosenstock, Levin Häni, Ulrike Grittner, Nicolas Schlinkmann, Meltem Ivren, Heike Schneider, Andreas Raabe, Peter Vajkoczy, Kathleen Seidel, Thomas Picht
Journal of Neurosurgery.2022; 136(4): 1194. CrossRef - Foramen magnum meningioma presented as cervical myelopathy in a pregnant COVID-19 patient: A case report
Olivia Josephine Wijaya, Djohan Ardiansyah
Annals of Medicine and Surgery.2022; 77: 103647. CrossRef - The telomere maintenance mechanism spectrum and its dynamics in gliomas
Sojin Kim, Tamrin Chowdhury, Hyeon Jong Yu, Jee Ye Kahng, Chae Eun Lee, Seung Ah. Choi, Kyung-Min Kim, Ho Kang, Joo Ho Lee, Soon-Tae Lee, Jae-Kyung Won, Kyung Hyun Kim, Min-Sung Kim, Ji Yeoun Lee, Jin Wook Kim, Yong-Hwy Kim, Tae Min Kim, Seung Hong Choi,
Genome Medicine.2022;[Epub] CrossRef - A review of predictive, prognostic and diagnostic biomarkers for brain tumours: towards personalised and targeted cancer therapy
Ernest Osei, Pascale Walters, Olivia Masella, Quinton Tennant, Amber Fishwick, Eugenia Dadzie, Anmol Bhangu, Johnson Darko
Journal of Radiotherapy in Practice.2021; 20(1): 83. CrossRef - Use of a novel navigable tubular retractor system in 1826 minimally invasive parafascicular surgery (MIPS) cases involving deep-seated brain tumors, hemorrhages and malformations
Martina M. Cartwright, Penny Sekerak, Joseph Mark, Julian Bailes
Interdisciplinary Neurosurgery.2021; 23: 100919. CrossRef - Disentangling the therapeutic tactics in GBM: From bench to bedside and beyond
S. Daisy Precilla, Shreyas S. Kuduvalli, Anitha Thirugnanasambandhar Sivasubramanian
Cell Biology International.2021; 45(1): 18. CrossRef - Sequencing of a central nervous system tumor demonstrates cancer transmission in an organ transplant
Marie-Claude Gingras, Aniko Sabo, Maria Cardenas, Abbas Rana, Sadhna Dhingra, Qingchang Meng, Jianhong Hu, Donna M Muzny, Harshavardhan Doddapaneni, Lesette Perez, Viktoriya Korchina, Caitlin Nessner, Xiuping Liu, Hsu Chao, John Goss, Richard A Gibbs
Life Science Alliance.2021; 4(9): e202000941. CrossRef - Radiogenomics of Gliomas
Chaitra Badve, Sangam Kanekar
Radiologic Clinics of North America.2021; 59(3): 441. CrossRef - Chemical analysis of the human brain by imaging mass spectrometry
Akhila Ajith, Yeswanth Sthanikam, Shibdas Banerjee
The Analyst.2021; 146(18): 5451. CrossRef - Correlation between Stage and Histopathological Features and Clinical Outcomes in Patients with Glioma Tumors
Andre Lona, Alfansuri Kadri, Irina Kemala Nasution
Open Access Macedonian Journal of Medical Sciences.2021; 9(T3): 262. CrossRef - Pediatric Glioma: An Update of Diagnosis, Biology, and Treatment
Yusuke Funakoshi, Nobuhiro Hata, Daisuke Kuga, Ryusuke Hatae, Yuhei Sangatsuda, Yutaka Fujioka, Kosuke Takigawa, Masahiro Mizoguchi
Cancers.2021; 13(4): 758. CrossRef - Discovery of clinically relevant fusions in pediatric cancer
Stephanie LaHaye, James R. Fitch, Kyle J. Voytovich, Adam C. Herman, Benjamin J. Kelly, Grant E. Lammi, Jeremy A. Arbesfeld, Saranga Wijeratne, Samuel J. Franklin, Kathleen M. Schieffer, Natalie Bir, Sean D. McGrath, Anthony R. Miller, Amy Wetzel, Katheri
BMC Genomics.2021;[Epub] CrossRef - Clinicopathological correlation of glioma patients with respect to immunohistochemistry markers: A prospective study of 115 patients in a Tertiary Care Hospital in North India
Gitanshu Dahuja, Ashok Gupta, Arpita Jindal, Gaurav Jain, Santosh Sharma, Arvind Kumar
Asian Journal of Neurosurgery.2021; 16(04): 732. CrossRef - Role of Extracellular Vesicles in Glioma Progression: Deciphering Cellular Biological Processes to Clinical Applications
Rashmi Rana, Shikha Joon, Kirti Chauhan, Vaishnavi Rathi, Nirmal Kumar Ganguly, Chandni Kumari, Dharmendra Kumar Yadav
Current Topics in Medicinal Chemistry.2021; 21(8): 696. CrossRef - A comprehensive overview on the molecular biology of human glioma: what the clinician needs to know
P. D. Delgado-López, P. Saiz-López, R. Gargini, E. Sola-Vendrell, S. Tejada
Clinical and Translational Oncology.2020; 22(11): 1909. CrossRef - Use of Fluorescence In Situ Hybridization (FISH) in Diagnosis and Tailored Therapies in Solid Tumors
Natalia Magdalena Chrzanowska, Janusz Kowalewski, Marzena Anna Lewandowska
Molecules.2020; 25(8): 1864. CrossRef - From Banding to BAM Files
Adrian M. Dubuc
Surgical Pathology Clinics.2020; 13(2): 343. CrossRef - Mutation profiling of anaplastic ependymoma grade III by Ion Proton next generation DNA sequencing
Ejaz Butt, Sabra Alyami, Tahani Nageeti, Muhammad Saeed, Khalid AlQuthami, Abdellatif Bouazzaoui, Mohammad Athar, Zainularifeen Abduljaleel, Faisal Al-Allaf, Mohiuddin Taher
F1000Research.2020; 8: 613. CrossRef - Assessment of the non-linear optical behavior of cells for discrimination between normal and malignant glial cells
Soraya Emamgholizadeh Minaei, Alireza Ghader, Ali Abbasian Ardakani, Samideh Khoei, Mohammad Hosein Majles Ara
Laser Physics.2020; 30(12): 125601. CrossRef - Brain Tumor Classification in Magnetic Resonance Images Using Deep Learning and Wavelet Transform
Ahmad M. Sarhan
Journal of Biomedical Science and Engineering.2020; 13(06): 102. CrossRef - Mutation profiling of anaplastic ependymoma grade III by Ion Proton next generation DNA sequencing
Muhammad Butt, Sabra Alyami, Tahani Nageeti, Muhammad Saeed, Khalid AlQuthami, Abdellatif Bouazzaoui, Mohammad Athar, Zainularifeen Abduljaleel, Faisal Al-Allaf, Mohiuddin Taher
F1000Research.2019; 8: 613. CrossRef - A Multiplex Quantitative Reverse Transcription Polymerase Chain Reaction Assay for the Detection of KIAA1549–BRAF Fusion Transcripts in Formalin-Fixed Paraffin-Embedded Pilocytic Astrocytomas
David Bret, Valentin Chappuis, Delphine Poncet, François Ducray, Karen Silva, Fabrice Mion, Alexandre Vasiljevic, Carole Ferraro-Peyret, Carmine Mottolese, Pierre Leblond, Mathieu Gabut, Didier Frappaz, Nathalie Streichenberger, David Meyronet, Pierre-Pau
Molecular Diagnosis & Therapy.2019; 23(4): 537. CrossRef - A PTPmu Biomarker is Associated with Increased Survival in Gliomas
Mette L. Johansen, Jason Vincent, Haley Gittleman, Sonya E. L. Craig, Marta Couce, Andrew E. Sloan, Jill S. Barnholtz-Sloan, Susann M. Brady-Kalnay
International Journal of Molecular Sciences.2019; 20(10): 2372. CrossRef -
ATRX Mutations in Pineal Parenchymal Tumors of Intermediate Differentiation
Haydee Martínez, Michelle Nagurney, Zi-Xuan Wang, Charles G Eberhart, Christopher M Heaphy, Mark T Curtis, Fausto J Rodriguez
Journal of Neuropathology & Experimental Neurology.2019; 78(8): 703. CrossRef - The role of fibroblast growth factors and their receptors in gliomas: the mutations involved
Vasiliki Georgiou, Vasiliki Gkretsi
Reviews in the Neurosciences.2019; 30(5): 543. CrossRef - The Role of Next Generation Sequencing in Diagnosis of Brain Tumors: A Review Study
Sadegh Shirian, Yahya Daneshbod, Saranaz Jangjoo, Amir Ghaemi, Arash Goodarzi, Maryam Ghavideldarestani, Ahmad Emadi, Arman Ai, Akbar Ahmadi, Jafar Ai
Archives of Neuroscience.2019;[Epub] CrossRef - Applied Precision Cancer Medicine in Neuro-Oncology
H. Taghizadeh, L. Müllauer, J. Furtner, J. A. Hainfellner, C. Marosi, M. Preusser, G. W. Prager
Scientific Reports.2019;[Epub] CrossRef - Comparative genomic analysis of driver mutations in matched primary and recurrent meningiomas
Joshua Loewenstern, John Rutland, Corey Gill, Hanane Arib, Margaret Pain, Melissa Umphlett, Yayoi Kinoshita, Russell McBride, Michael Donovan, Robert Sebra, Joshua Bederson, Mary Fowkes, Raj Shrivastava
Oncotarget.2019; 10(37): 3506. CrossRef - Next generation DNA sequencing of atypical choroid plexus papilloma of brain: Identification of novel mutations in a female patient by Ion Proton
Mohiuddin Taher, Amal Hassan, Muhammad Saeed, Raid Jastania, Tahani Nageeti, Hisham Alkhalidi, Ghida Dairi, Zainularifeen Abduljaleel, Mohammad Athar, Abdellatif Bouazzaoui, Wafa El‑Bjeirami, Faisal Al‑Allaf
Oncology Letters.2019;[Epub] CrossRef - Pure mechanistic analysis of additive neuroprotective effects between baicalin and jasminoidin in ischemic stroke mice
Peng-qian Wang, Qiong Liu, Wen-juan Xu, Ya-nan Yu, Ying-ying Zhang, Bing Li, Jun Liu, Zhong Wang
Acta Pharmacologica Sinica.2018; 39(6): 961. CrossRef - The Smad4/PTEN Expression Pattern Predicts Clinical Outcomes in Colorectal Adenocarcinoma
Yumin Chung, Young Chan Wi, Yeseul Kim, Seong Sik Bang, Jung-Ho Yang, Kiseok Jang, Kyueng-Whan Min, Seung Sam Paik
Journal of Pathology and Translational Medicine.2018; 52(1): 37. CrossRef - Primary brain tumours in adults
Sarah Lapointe, Arie Perry, Nicholas A Butowski
The Lancet.2018; 392(10145): 432. CrossRef - Epidemiology and Overview of Gliomas
Mary Elizabeth Davis
Seminars in Oncology Nursing.2018; 34(5): 420. CrossRef - Toward Precision Medicine: Promising Areas of Research in Glioma
Mary Elizabeth Davis, Dana Bossert, Malbora Manne
Seminars in Oncology Nursing.2018; 34(5): 569. CrossRef - Molecular Basis of Pediatric Brain Tumors
Alexia Klonou, Christina Piperi, Antonios N. Gargalionis, Athanasios G. Papavassiliou
NeuroMolecular Medicine.2017; 19(2-3): 256. CrossRef
- Low-grade Epilepsy-associated Tumors and Other Acquired Causes of Epilepsy
- Molecular Testing of Lymphoproliferative Disorders: Current Status and Perspectives
- Yoon Kyung Jeon, Sun Och Yoon, Jin Ho Paik, Young A Kim, Bong Kyung Shin, Hyun-Jung Kim, Hee Jeong Cha, Ji Eun Kim, Jooryung Huh, Young-Hyeh Ko
- J Pathol Transl Med. 2017;51(3):224-241. Published online May 10, 2017
- DOI: https://doi.org/10.4132/jptm.2017.04.09
- 23,236 View
- 713 Download
- 14 Web of Science
- 16 Crossref
-
 Abstract
Abstract
 PDF
PDF - Molecular pathologic testing plays an important role for the diagnosis, prognostication and decision of treatment strategy in lymphoproliferative disease. Here, we briefly review the molecular tests currently used for lymphoproliferative disease and those which will be implicated in clinical practice in the near future. Specifically, this guideline addresses the clonality test for B- and T-cell proliferative lesions, molecular cytogenetic tests for malignant lymphoma, determination of cell-of-origin in diffuse large B-cell lymphoma, and molecular genetic alterations incorporated in the 2016 revision of the World Health Organization classification of lymphoid neoplasms. Finally, a new perspective on the next-generation sequencing for diagnostic, prognostic, and therapeutic purpose in malignant lymphoma will be summarized.
-
Citations
Citations to this article as recorded by- Pediatric lymphoproliferative disorders – Emerging insights and management: A narrative review
Emmanuel Ifeanyi Obeagu
Medicine.2026; 105(4): e47367. CrossRef - Presence of minimal residual disease determined by next-generation sequencing is not a reliable prognostic biomarker in children with acute lymphoblastic leukemia
Elizabeta Krstevska Bozhinovikj, Nadica Matevska-Geshkovska, Marija Staninova Stojovska, Emilija Gjorgievska, Aleksandra Jovanovska, Nevenka Ridova, Irina Panovska Stavridis, Svetlana Kocheva, Aleksandar Dimovski
Leukemia & Lymphoma.2025; 66(6): 1121. CrossRef - Haematogenous seeding in mycosis fungoides and Sézary syndrome: current evidence and clinical implications
Robert Gniadecki, Emmanuella Guenova, Christiane Querfeld, Jan P Nicolay, Julia Scarisbrick, Lubomir Sokol
British Journal of Dermatology.2025; 192(3): 381. CrossRef - Exploring External Quality Control Methods for PCR–Polyacrylamide Gel Electrophoresis–Based Lymphocyte Receptor Gene Rearrangement Assays in Korea
Jieun Kim, Ho Hyun Song, Soobin Chae, GeonWoo Choi, Jeong Won Shin
Journal of Laboratory Medicine and Quality Assurance.2025; 47(2): 43. CrossRef - Laboratory analysis of 182 cases of B-cell lymphoproliferative disorders other than typical chronic lymphocytic leukemia: Single-center study
Shams Salah Mahdi, Nuha Abd Ali Al-Sarai
Iraqi Journal of Hematology.2025; 14(2): 218. CrossRef - Assessment of Bone Marrow Involvement in B‐Cell non‐Hodgkin Lymphoma Using Immunoglobulin Gene Rearrangement Analysis with Next‐Generation Sequencing
Min Ji Jeon, Eun Sang Yu, Dae Sik Kim, Chul Won Choi, Ha Nui Kim, Jung Ah Kwon, Soo‐Young Yoon, Jung Yoon
Journal of Clinical Laboratory Analysis.2024;[Epub] CrossRef - Thymus and lung mucosa-associated lymphoid tissue lymphoma with adenocarcinoma of the lung: a case report and literature review
Yu Pang, Daosheng Li, Yiqian Chen, Qinqin Liu, Yuheng Wu, Qingliang Teng, Yuyu Liu
World Journal of Surgical Oncology.2023;[Epub] CrossRef - Development and implementation of an automated and highly accurate reporting process for NGS-based clonality testing
Sean T. Glenn, Phillip M. Galbo, Jesse D. Luce, Kiersten Marie Miles, Prashant K. Singh, Manuel J. Glynias, Carl Morrison
Oncotarget.2023; 14(1): 450. CrossRef - A comparison of capillary electrophoresis and next-generation sequencing in the detection of immunoglobulin heavy chain H and light chain κ gene rearrangements in the diagnosis of classic hodgkin’s lymphoma
Juan-Juan Zhang, Yu-Xin Xie, Li-Lin Luo, Xuan-Tao Yang, Yi-Xing Wang, Yue Cao, Zheng-Bo Long, Wan-Pu Wang
Bioengineered.2022; 13(3): 5868. CrossRef - Lymphoproliferative disorder involving body fluid: diagnostic approaches and roles of ancillary studies
Jiwon Koh, Sun Ah Shin, Ji Ae Lee, Yoon Kyung Jeon
Journal of Pathology and Translational Medicine.2022; 56(4): 173. CrossRef - Diagnostic Workup of Primary Cutaneous B Cell Lymphomas: A Clinician's Approach
Giulia Tadiotto Cicogna, Martina Ferranti, Mauro Alaibac
Frontiers in Oncology.2020;[Epub] CrossRef - Kappa and lambda immunohistochemistry and in situ hybridization in the evaluation of atypical cutaneous lymphoid infiltrates
Alexandra C. Hristov, Nneka I. Comfere, Claudia I. Vidal, Uma Sundram
Journal of Cutaneous Pathology.2020; 47(11): 1103. CrossRef - Primary lung mucosa-associated lymphoid tissue lymphoma accompanied by multiple sclerosis
Ke-Ke Yu, Lei Zhu, Ji-Kai Zhao, Rui-Ying Zhao, Yu-Chen Han
Chinese Medical Journal.2019; 132(13): 1625. CrossRef - Diagnostic accuracy of SOX11 immunohistochemistry in mantle cell lymphoma: A meta-analysis
Woojoo Lee, Eun Shin, Bo-Hyung Kim, Hyunchul Kim, Riccardo Dolcetti
PLOS ONE.2019; 14(11): e0225096. CrossRef - Views of dermatopathologists about clonality assays in the diagnosis of cutaneous T‐cell and B‐cell lymphoproliferative disorders
Nneka Comfere, Uma Sundram, Maria Yadira Hurley, Brian Swick
Journal of Cutaneous Pathology.2018; 45(1): 39. CrossRef - A Next-Generation Sequencing Primer—How Does It Work and What Can It Do?
Yuriy O. Alekseyev, Roghayeh Fazeli, Shi Yang, Raveen Basran, Thomas Maher, Nancy S. Miller, Daniel Remick
Academic Pathology.2018; 5: 2374289518766521. CrossRef
- Pediatric lymphoproliferative disorders – Emerging insights and management: A narrative review
- Good Laboratory Standards for Clinical Next-Generation Sequencing Cancer Panel Tests
- Jihun Kim, Woong-Yang Park, Nayoung K. D. Kim, Se Jin Jang, Sung-Min Chun, Chang-Ohk Sung, Jene Choi, Young-Hyeh Ko, Yoon-La Choi, Hyo Sup Shim, Jae-Kyung Won
- J Pathol Transl Med. 2017;51(3):191-204. Published online May 10, 2017
- DOI: https://doi.org/10.4132/jptm.2017.03.14
- 30,062 View
- 1,122 Download
- 36 Web of Science
- 38 Crossref
-
 Abstract
Abstract
 PDF
PDF - Next-generation sequencing (NGS) has recently emerged as an essential component of personalized cancer medicine due to its high throughput and low per-base cost. However, no sufficient guidelines for implementing NGS as a clinical molecular pathology test are established in Korea. To ensure clinical grade quality without inhibiting adoption of NGS, a taskforce team assembled by the Korean Society of Pathologists developed laboratory guidelines for NGS cancer panel testing procedures and requirements for clinical implementation of NGS. This consensus standard proposal consists of two parts: laboratory guidelines and requirements for clinical NGS laboratories. The laboratory guidelines part addressed several important issues across multistep NGS cancer panel tests including choice of gene panel and platform, sample handling, nucleic acid management, sample identity tracking, library preparation, sequencing, analysis and reporting. Requirements for clinical NGS tests were summarized in terms of documentation, validation, quality management, and other required written policies. Together with appropriate pathologist training and international laboratory standards, these laboratory standards would help molecular pathology laboratories to successfully implement NGS cancer panel tests in clinic. In this way, the oncology community would be able to help patients to benefit more from personalized cancer medicine.
-
Citations
Citations to this article as recorded by- Report of the Korean Association for External Quality Assessment of Next-Generation Sequencing Analysis for Somatic Variants in Hematologic Malignancies (2021–2025)
Dongju Won, Yu Jin Park, JoungEun Lee, Mijung Kwon, Saeam Shin, Seung-Tae Lee, Jong Rak Choi
Journal of Laboratory Medicine and Quality Assurance.2026; 48(1): 10. CrossRef - Tumour purity assessment with deep learning in colorectal cancer and impact on molecular analysis
Lydia A Schoenpflug, Aikaterini Chatzipli, Korsuk Sirinukunwattana, Susan Richman, Andrew Blake, James Robineau, Kirsten D Mertz, Clare Verrill, Simon J Leedham, Claire Hardy, Celina Whalley, Keara Redmond, Philip Dunne, Steven Walker, Andrew D Beggs, Ult
The Journal of Pathology.2025; 265(2): 184. CrossRef - Clinical Validation of Local Versus Commercial Genomic Testing in Cancer: A Comparison of Tissue and Plasma Concordance
Lucy G. Faulkner, Lynne Howells, Susann Lehman, Caroline Cowley, Zahirah Sidat, Jacqui Shaw, Anne L. Thomas
Cancer Investigation.2025; 43(2): 119. CrossRef - Diagnostic Implications of NGS-Based Molecular Profiling in Mature B-Cell Lymphomas with Potential Bone Marrow Involvement
Bernhard Strasser, Sebastian Mustafa, Josef Seier, Erich Wimmer, Josef Tomasits
Diagnostics.2025; 15(6): 727. CrossRef - Pragmatic nationwide master observational trial based on genomic alterations in advanced solid tumors: KOrean Precision Medicine Networking Group Study of MOlecular profiling guided therapy based on genomic alterations in advanced Solid tumors (KOSMOS)-II
Sun Young Kim, Jee Hyun Kim, Tae-Yong Kim, Sook Ryun Park, Shinkyo Yoon, Soohyeon Lee, Se-Hoon Lee, Tae Min Kim, Sae-Won Han, Hye Ryun Kim, Hongseok Yun, Sejoon Lee, Jihun Kim, Yoon-La Choi, Kui Son Choi, Heejung Chae, Hyewon Ryu, Gyeong-Won Lee, Dae Youn
BMC Cancer.2024;[Epub] CrossRef - Reporting of somatic variants in clinical cancer care: recommendations of the Swiss Society of Molecular Pathology
Yann Christinat, Baptiste Hamelin, Ilaria Alborelli, Paolo Angelino, Valérie Barbié, Bettina Bisig, Heather Dawson, Milo Frattini, Tobias Grob, Wolfram Jochum, Ronny Nienhold, Thomas McKee, Matthias Matter, Edoardo Missiaglia, Francesca Molinari, Sacha Ro
Virchows Archiv.2024; 485(6): 1033. CrossRef - Acute myeloid leukemia and myelodysplastic neoplasms: clinical implications of myelodysplasia-related genes mutations and TP53 aberrations
Hyunwoo Kim, Ja Young Lee, Shinae Yu, Eunkyoung Yoo, Hye Ran Kim, Sang Min Lee, Won Sik Lee
Blood Research.2024;[Epub] CrossRef - Validation and Clinical Application of ONCOaccuPanel for Targeted Next-Generation Sequencing of Solid Tumors
Moonsik Kim, Changseon Lee, Juyeon Hong, Juhee Kim, Ji Yun Jeong, Nora Jee-Young Park, Ji-Eun Kim, Ji Young Park
Cancer Research and Treatment.2023; 55(2): 429. CrossRef - Establishing molecular pathology curriculum for pathology trainees and continued medical education: a collaborative work from the Molecular Pathology Study Group of the Korean Society of Pathologists
Jiwon Koh, Ha Young Park, Jeong Mo Bae, Jun Kang, Uiju Cho, Seung Eun Lee, Haeyoun Kang, Min Eui Hong, Jae Kyung Won, Youn-La Choi, Wan-Seop Kim, Ahwon Lee
Journal of Pathology and Translational Medicine.2023; 57(5): 265. CrossRef - Clinical applications of next-generation sequencing in the diagnosis of genetic disorders in Korea: a narrative review
Jihoon G. Yoon, Man Jin Kim, Yong Jin Kwon, Jong-Hee Chae
Journal of the Korean Medical Association.2023; 66(10): 613. CrossRef - Obtaining spatially resolved tumor purity maps using deep multiple instance learning in a pan-cancer study
Mustafa Umit Oner, Jianbin Chen, Egor Revkov, Anne James, Seow Ye Heng, Arife Neslihan Kaya, Jacob Josiah Santiago Alvarez, Angela Takano, Xin Min Cheng, Tony Kiat Hon Lim, Daniel Shao Weng Tan, Weiwei Zhai, Anders Jacobsen Skanderup, Wing-Kin Sung, Hwee
Patterns.2022; 3(2): 100399. CrossRef - Update on Molecular Diagnosis in Extranodal NK/T-Cell Lymphoma and Its Role in the Era of Personalized Medicine
Ka-Hei (Murphy) Sun, Yin-Ting (Heylie) Wong, Ka-Man (Carmen) Cheung, Carmen (Michelle) Yuen, Yun-Tat (Ted) Chan, Wing-Yan (Jennifer) Lai, Chun (David) Chao, Wing-Sum (Katie) Fan, Yuen-Kiu (Karen) Chow, Man-Fai Law, Ho-Chi (Tommy) Tam
Diagnostics.2022; 12(2): 409. CrossRef - Defining Novel DNA Virus-Tumor Associations and Genomic Correlates Using Prospective Clinical Tumor/Normal Matched Sequencing Data
Chad M. Vanderbilt, Anita S. Bowman, Sumit Middha, Kseniya Petrova-Drus, Yi-Wei Tang, Xin Chen, Youxiang Wang, Jason Chang, Natasha Rekhtman, Klaus J. Busam, Sounak Gupta, Meera Hameed, Maria E. Arcila, Marc Ladanyi, Michael F. Berger, Snjezana Dogan, Ahm
The Journal of Molecular Diagnostics.2022; 24(5): 515. CrossRef - Performance Evaluation of Three DNA Sample Tracking Tools in a Whole Exome Sequencing Workflow
Gertjan Wils, Céline Helsmoortel, Pieter-Jan Volders, Inge Vereecke, Mauro Milazzo, Jo Vandesompele, Frauke Coppieters, Kim De Leeneer, Steve Lefever
Molecular Diagnosis & Therapy.2022; 26(4): 411. CrossRef - Clinical Quality Considerations when Using Next-Generation Sequencing (NGS) in Clinical Drug Development
Timothé Ménard, Alaina Barros, Christopher Ganter
Therapeutic Innovation & Regulatory Science.2021; 55(5): 1066. CrossRef - Fast Healthcare Interoperability Resources (FHIR)–Based Quality Information Exchange for Clinical Next-Generation Sequencing Genomic Testing: Implementation Study
Donghyeong Seong, Sungwon Jung, Sungchul Bae, Jongsuk Chung, Dae-Soon Son, Byoung-Kee Yi
Journal of Medical Internet Research.2021; 23(4): e26261. CrossRef - Status of Next-Generation Sequencing-Based Genetic Diagnosis in Hematologic Malignancies in Korea (2017-2018)
JinJu Kim, Ja Young Lee, Jungwon Huh, Myung-Hyun Nam, Myungshin Kim, Young-Uk Cho, Sun-Young Kong, Seung-Tae Lee, In-Suk Kim
Laboratory Medicine Online.2021; 11(1): 25. CrossRef - MSI-Testung
Josef Rüschoff, Gustavo Baretton, Hendrik Bläker, Wolfgang Dietmaier, Manfred Dietel, Arndt Hartmann, Lars-Christian Horn, Korinna Jöhrens, Thomas Kirchner, Ruth Knüchel, Doris Mayr, Sabine Merkelbach-Bruse, Hans-Ulrich Schildhaus, Peter Schirmacher, Mark
Der Pathologe.2021; 42(4): 414. CrossRef - Molecular biomarker testing for non–small cell lung cancer: consensus statement of the Korean Cardiopulmonary Pathology Study Group
Sunhee Chang, Hyo Sup Shim, Tae Jung Kim, Yoon-La Choi, Wan Seop Kim, Dong Hoon Shin, Lucia Kim, Heae Surng Park, Geon Kook Lee, Chang Hun Lee
Journal of Pathology and Translational Medicine.2021; 55(3): 181. CrossRef - MSI testing
Josef Rüschoff, Gustavo Baretton, Hendrik Bläker, Wolfgang Dietmaier, Manfred Dietel, Arndt Hartmann, Lars-Christian Horn, Korinna Jöhrens, Thomas Kirchner, Ruth Knüchel, Doris Mayr, Sabine Merkelbach-Bruse, Hans-Ulrich Schildhaus, Peter Schirmacher, Mark
Der Pathologe.2021; 42(S1): 110. CrossRef - 16S rDNA microbiome composition pattern analysis as a diagnostic biomarker for biliary tract cancer
Huisong Lee, Hyeon Kook Lee, Seog Ki Min, Won Hee Lee
World Journal of Surgical Oncology.2020;[Epub] CrossRef -
Risk Stratification Using a Novel Genetic Classifier Including
PLEKHS1
Promoter Mutations for Differentiated Thyroid Cancer with Distant Metastasis
Chan Kwon Jung, Seung-Hyun Jung, Sora Jeon, Young Mun Jeong, Yourha Kim, Sohee Lee, Ja-Seong Bae, Yeun-Jun Chung
Thyroid®.2020; 30(11): 1589. CrossRef - Biomarker testing for advanced lung cancer by next-generation sequencing; a valid method to achieve a comprehensive glimpse at mutational landscape
Anurag Mehta, Smreti Vasudevan, Sanjeev Kumar Sharma, Manoj Panigrahi, Moushumi Suryavanshi, Mumtaz Saifi, Ullas Batra
Applied Cancer Research.2020;[Epub] CrossRef - Application Areas of Traditional Molecular Genetic Methods and NGS in relation to Hereditary Urological Cancer Diagnosis
Dmitry S. Mikhaylenko, Alexander S. Tanas, Dmitry V. Zaletaev, Marina V. Nemtsova
Journal of Oncology.2020; 2020: 1. CrossRef - Assembling and Validating Bioinformatic Pipelines for Next-Generation Sequencing Clinical Assays
Jeffrey A SoRelle, Megan Wachsmann, Brandi L. Cantarel
Archives of Pathology & Laboratory Medicine.2020; 144(9): 1118. CrossRef - Standard operating procedure for somatic variant refinement of sequencing data with paired tumor and normal samples
Erica K. Barnell, Peter Ronning, Katie M. Campbell, Kilannin Krysiak, Benjamin J. Ainscough, Lana M. Sheta, Shahil P. Pema, Alina D. Schmidt, Megan Richters, Kelsy C. Cotto, Arpad M. Danos, Cody Ramirez, Zachary L. Skidmore, Nicholas C. Spies, Jasreet Hun
Genetics in Medicine.2019; 21(4): 972. CrossRef - A DNA pool of FLT3-ITD positive DNA samples can be used efficiently for analytical evaluation of NGS-based FLT3-ITD quantitation - Testing several different ITD sequences and rates, simultaneously
Zoltán A. Mezei, Dávid Tornai, Róza Földesi, László Madar, Andrea Sümegi, Mária Papp, Péter Antal-Szalmás
Journal of Biotechnology.2019; 303: 25. CrossRef - Pharmacogenomic Testing: Clinical Evidence and Implementation Challenges
Catriona Hippman, Corey Nislow
Journal of Personalized Medicine.2019; 9(3): 40. CrossRef - Cancer Panel Assay for Precision Oncology Clinic: Results from a 1-Year Study
Dohee Kwon, Binnari Kim, Hyeong Chan Shin, Eun Ji Kim, Sang Yun Ha, Kee-Taek Jang, Seung Tae Kim, Jeeyun Lee, Won Ki Kang, Joon Oh Park, Kyoung-Mee Kim
Translational Oncology.2019; 12(11): 1488. CrossRef - Analytical Evaluation of an NGS Testing Method for Routine Molecular Diagnostics on Melanoma Formalin-Fixed, Paraffin-Embedded Tumor-Derived DNA
Irene Mancini, Lisa Simi, Francesca Salvianti, Francesca Castiglione, Gemma Sonnati, Pamela Pinzani
Diagnostics.2019; 9(3): 117. CrossRef - Benchmark Database for Process Optimization and Quality Control of Clinical Cancer Panel Sequencing
Donghyeong Seong, Jongsuk Chung, Ki-Wook Lee, Sook-Young Kim, Byung-Suk Kim, Jung-Keun Song, Sungwon Jung, Taeseob Lee, Donghyun Park, Byoung-Kee Yi, Woong-Yang Park, Dae-Soon Son
Biotechnology and Bioprocess Engineering.2019; 24(5): 793. CrossRef - Use of the Ion PGM and the GeneReader NGS Systems in Daily Routine Practice for Advanced Lung Adenocarcinoma Patients: A Practical Point of View Reporting a Comparative Study and Assessment of 90 Patients
Simon Heeke, Véronique Hofman, Elodie Long-Mira, Virginie Lespinet, Salomé Lalvée, Olivier Bordone, Camille Ribeyre, Virginie Tanga, Jonathan Benzaquen, Sylvie Leroy, Charlotte Cohen, Jérôme Mouroux, Charles Marquette, Marius Ilié, Paul Hofman
Cancers.2018; 10(4): 88. CrossRef - Use of the Ion AmpliSeq Cancer Hotspot Panel in clinical molecular pathology laboratories for analysis of solid tumours: With emphasis on validation with relevant single molecular pathology tests and the Oncomine Focus Assay
Ahwon Lee, Sung-Hak Lee, Chan Kwon Jung, Gyungsin Park, Kyo Young Lee, Hyun Joo Choi, Ki Ouk Min, Tae Jung Kim, Eun Jung Lee, Youn Soo Lee
Pathology - Research and Practice.2018; 214(5): 713. CrossRef - Recent Advancement of the Molecular Diagnosis in Pediatric Brain Tumor
Jeong-Mo Bae, Jae-Kyung Won, Sung-Hye Park
Journal of Korean Neurosurgical Society.2018; 61(3): 376. CrossRef - The long tail of molecular alterations in non-small cell lung cancer: a single-institution experience of next-generation sequencing in clinical molecular diagnostics
Caterina Fumagalli, Davide Vacirca, Alessandra Rappa, Antonio Passaro, Juliana Guarize, Paola Rafaniello Raviele, Filippo de Marinis, Lorenzo Spaggiari, Chiara Casadio, Giuseppe Viale, Massimo Barberis, Elena Guerini-Rocco
Journal of Clinical Pathology.2018; 71(9): 767. CrossRef - Clinical laboratory utilization management and improved healthcare performance
Christopher Naugler, Deirdre L. Church
Critical Reviews in Clinical Laboratory Sciences.2018; 55(8): 535. CrossRef - Development of HLA-A, -B and -DR Typing Method Using Next-Generation Sequencing
Dong Hee Seo, Jeong Min Lee, Mi Ok Park, Hyun Ju Lee, Seo Yoon Moon, Mijin Oh, So Young Kim, Sang-Heon Lee, Ki-Eun Hyeong, Hae-Jin Hu, Dae-Yeon Cho
The Korean Journal of Blood Transfusion.2018; 29(3): 310. CrossRef - Value-based genomics
Jun Gong, Kathy Pan, Marwan Fakih, Sumanta Pal, Ravi Salgia
Oncotarget.2018; 9(21): 15792. CrossRef
- Report of the Korean Association for External Quality Assessment of Next-Generation Sequencing Analysis for Somatic Variants in Hematologic Malignancies (2021–2025)
- Molecular Testing of Lung Cancers
- Hyo Sup Shim, Yoon-La Choi, Lucia Kim, Sunhee Chang, Wan-Seop Kim, Mee Sook Roh, Tae-Jung Kim, Seung Yeon Ha, Jin-Haeng Chung, Se Jin Jang, Geon Kook Lee
- J Pathol Transl Med. 2017;51(3):242-254. Published online April 21, 2017
- DOI: https://doi.org/10.4132/jptm.2017.04.10
- 18,685 View
- 618 Download
- 28 Web of Science
- 28 Crossref
-
 Abstract
Abstract
 PDF
PDF - Targeted therapies guided by molecular diagnostics have become a standard treatment of lung cancer. Epidermal growth factor receptor (EGFR) mutations and anaplastic lymphoma kinase (ALK) rearrangements are currently used as the best predictive biomarkers for EGFR tyrosine kinase inhibitors and ALK inhibitors, respectively. Besides EGFR and ALK, the list of druggable genetic alterations has been growing, including ROS1 rearrangements, RET rearrangements, and MET alterations. In this situation, pathologists should carefully manage clinical samples for molecular testing and should do their best to quickly and accurately identify patients who will benefit from precision therapeutics. Here, we grouped molecular biomarkers of lung cancers into three categories—mutations, gene rearrangements, and amplifications—and propose expanded guidelines on molecular testing of lung cancers.
-
Citations
Citations to this article as recorded by- Association between PD-L1 expression with EGFR, ALK, and ROS1 driver oncogene mutations in non-small cell lung cancer
Dülger Onur, Yaylım İlhan, Öz Büge
Indian Journal of Pathology and Microbiology.2025; 68(1): 36. CrossRef - Evidence-based Approach to Transthoracic Needle Biopsy: Procedural Techniques, Risks, and Controversies
Shravan Sridhar, Hannah G. Ahn, Sayedomid Ebrahimzadeh, Felicia Tang, Brett Elicker
RadioGraphics.2025;[Epub] CrossRef - Enhancing Lung Cancer Care in Portugal: Bridging Gaps for Improved Patient Outcomes
Raquel Ramos, Conceição Souto Moura, Mariana Costa, Nuno Jorge Lamas, Renato Correia, Diogo Garcez, José Miguel Pereira, Carlos Sousa, Nuno Vale
Journal of Personalized Medicine.2024; 14(5): 446. CrossRef - Evolution of therapy for ALK-positive lung carcinomas: Application of third-generation ALK inhibitors in real clinical practice
A. F. Nasretdinov, A. V. Sultanbaev, Sh. I. Musin, K. V. Menshikov, R. T. Ayupov, A. A. Izmailov, G. A. Serebrennikov, V. E. Askarov, D. V. Feoktistov
Meditsinskiy sovet = Medical Council.2024; (10): 74. CrossRef - Cost-effectiveness of next-generation sequencing for advanced EGFR/ALK-negative non-small cell lung cancer
Dong-Won Kang, Sun-Kyeong Park, Sokbom Kang, Eui-Kyung Lee
Lung Cancer.2024; 197: 107970. CrossRef - miR-92a-3p regulates cisplatin-induced cancer cell death
Romain Larrue, Sandy Fellah, Nihad Boukrout, Corentin De Sousa, Julie Lemaire, Carolane Leboeuf, Marine Goujon, Michael Perrais, Bernard Mari, Christelle Cauffiez, Nicolas Pottier, Cynthia Van der Hauwaert
Cell Death & Disease.2023;[Epub] CrossRef - Diagnostic Approach of Lung Cancer: A Literature Review
Jesi Hana, Novia Nurul Faizah
Jurnal Respirasi.2023; 9(2): 141. CrossRef - Molecular Pathology of Lung Cancer
James J. Saller, Theresa A. Boyle
Cold Spring Harbor Perspectives in Medicine.2022; 12(3): a037812. CrossRef - Landscape of EGFR mutations in lung adenocarcinoma: a single institute experience with comparison of PANAMutyper testing and targeted next-generation sequencing
Jeonghyo Lee, Yeon Bi Han, Hyun Jung Kwon, Song Kook Lee, Hyojin Kim, Jin-Haeng Chung
Journal of Pathology and Translational Medicine.2022; 56(5): 249. CrossRef - Molecular biomarker testing for non–small cell lung cancer: consensus statement of the Korean Cardiopulmonary Pathology Study Group
Sunhee Chang, Hyo Sup Shim, Tae Jung Kim, Yoon-La Choi, Wan Seop Kim, Dong Hoon Shin, Lucia Kim, Heae Surng Park, Geon Kook Lee, Chang Hun Lee
Journal of Pathology and Translational Medicine.2021; 55(3): 181. CrossRef - TM4SF4 and LRRK2 Are Potential Therapeutic Targets in Lung and Breast Cancers through Outlier Analysis
Kyungsoo Jung, Joon-Seok Choi, Beom-Mo Koo, Yu Jin Kim, Ji-Young Song, Minjung Sung, Eun Sol Chang, Ka-Won Noh, Sungbin An, Mi-Sook Lee, Kyoung Song, Hannah Lee, Ryong Nam Kim, Young Kee Shin, Doo-Yi Oh, Yoon-La Choi
Cancer Research and Treatment.2021; 53(1): 9. CrossRef - The promises and challenges of early non‐small cell lung cancer detection: patient perceptions, low‐dose CT screening, bronchoscopy and biomarkers
Lukas Kalinke, Ricky Thakrar, Sam M. Janes
Molecular Oncology.2021; 15(10): 2544. CrossRef - Cost-effectiveness analyses of targeted therapy and immunotherapy for advanced non-small cell lung cancer in the United States: a systematic review
Anthony Yu, Eva Huang, Momoka Abe, Kang An, Sun-Kyeong Park, Chanhyun Park
Expert Review of Pharmacoeconomics & Outcomes Research.2021; 21(3): 381. CrossRef - The expanding capability and clinical relevance of molecular diagnostic technology to identify and evaluate EGFR mutations in advanced/metastatic NSCLC
Parth Shah, Jacob Sands, Nicola Normanno
Lung Cancer.2021; 160: 118. CrossRef - Testing for EGFR Mutations and ALK Rearrangements in Advanced Non-Small-Cell Lung Cancer: Considerations for Countries in Emerging Markets
Mercedes L Dalurzo, Alejandro Avilés-Salas, Fernando Augusto Soares, Yingyong Hou, Yuan Li, Anna Stroganova, Büge Öz, Arif Abdillah, Hui Wan, Yoon-La Choi
OncoTargets and Therapy.2021; Volume 14: 4671. CrossRef - Treatment of Patients With Non–Small-Cell Lung Cancer Harboring Rare Oncogenic Mutations
Melina E. Marmarelis, Corey J. Langer
Clinical Lung Cancer.2020; 21(5): 395. CrossRef - Detection of Targetable Genetic Alterations in Korean Lung Cancer Patients: A Comparison Study of Single-Gene Assays and Targeted Next-Generation Sequencing
Eunhyang Park, Hyo Sup Shim
Cancer Research and Treatment.2020; 52(2): 543. CrossRef - High prevalence of ROS1 gene rearrangement detected by FISH in EGFR and ALK negative lung adenocarcinoma
Yuyin Xu, Heng Chang, Lijing Wu, Xin Zhang, Ling Zhang, Jing Zhang, Yuan Li, Lei Shen, Xiaoli Zhu, Xiaoyan Zhou, Qianming Bai
Experimental and Molecular Pathology.2020; 117: 104548. CrossRef - An All-In-One Transcriptome-Based Assay to Identify Therapy-Guiding Genomic Aberrations in Nonsmall Cell Lung Cancer Patients
Jiacong Wei, Anna A. Rybczynska, Pei Meng, Martijn Terpstra, Ali Saber, Jantine Sietzema, Wim Timens, Ed Schuuring, T. Jeroen N. Hiltermann, Harry. J.M. Groen, Anthonie van der Wekken, Anke van den Berg, Klaas Kok
Cancers.2020; 12(10): 2843. CrossRef - Immunotherapy in EGFR-Mutant and ALK-Positive Lung Cancer
Alexander Gavralidis, Justin F. Gainor
The Cancer Journal.2020; 26(6): 517. CrossRef - Role of Immunocytochemistry in the Cytological Diagnosis of Pulmonary Tumors
Jasna Metovic, Luisella Righi, Luisa Delsedime, Marco Volante, Mauro Papotti
Acta Cytologica.2020; 64(1-2): 16. CrossRef - Molecular Diagnostic Assays and Clinicopathologic Implications of MET Exon 14 Skipping Mutation in Non–small-cell Lung Cancer
Eun Kyung Kim, Kyung A. Kim, Chang Young Lee, Sangwoo Kim, Sunhee Chang, Byoung Chul Cho, Hyo Sup Shim
Clinical Lung Cancer.2019; 20(1): e123. CrossRef - PD‐L1 expression in ROS1‐rearranged non‐small cell lung cancer: A study using simultaneous genotypic screening of EGFR, ALK, and ROS1
Jongmin Lee, Chan Kwon Park, Hyoung‐Kyu Yoon, Young Jo Sa, In Sook Woo, Hyo Rim Kim, Sue Youn Kim, Tae‐Jung Kim
Thoracic Cancer.2019; 10(1): 103. CrossRef - Human Leukocyte Antigen Class I and Programmed Death-Ligand 1 Coexpression Is an Independent Poor Prognostic Factor in Adenocarcinoma of the Lung
Yeon Bi Han, Hyun Jung Kwon, Soo Young Park, Eun-Sun Kim, Hyojin Kim, Jin-Haeng Chung
Journal of Pathology and Translational Medicine.2019; 53(2): 86. CrossRef - Molecular testing for advanced non-small cell lung cancer in Malaysia: Consensus statement from the College of Pathologists, Academy of Medicine Malaysia, the Malaysian Thoracic Society, and the Malaysian Oncological Society
Pathmanathan Rajadurai, Phaik Leng Cheah, Soon Hin How, Chong Kin Liam, Muhammad Azrif Ahmad Annuar, Norhayati Omar, Noriah Othman, Nurhayati Mohd Marzuki, Yong Kek Pang, Ros Suzanna Ahmad Bustamam, Lye Mun Tho
Lung Cancer.2019; 136: 65. CrossRef - Somatic mutations and immune checkpoint biomarkers
Brielle A. Parris, Eloise Shaw, Brendan Pang, Richie Soong, Kwun Fong, Ross A. Soo
Respirology.2019; 24(3): 215. CrossRef - Adverse Event Management in Patients with BRAF V600E-Mutant Non-Small Cell Lung Cancer Treated with Dabrafenib plus Trametinib
Anna Chalmers, Laura Cannon, Wallace Akerley
The Oncologist.2019; 24(7): 963. CrossRef - Genetic and clinicopathologic characteristics of lung adenocarcinoma with tumor spread through air spaces
Jae Seok Lee, Eun Kyung Kim, Moonsik Kim, Hyo Sup Shim
Lung Cancer.2018; 123: 121. CrossRef
- Association between PD-L1 expression with EGFR, ALK, and ROS1 driver oncogene mutations in non-small cell lung cancer
- Molecular Testing for Gastrointestinal Cancer
- Hye Seung Lee, Woo Ho Kim, Yoonjin Kwak, Jiwon Koh, Jeong Mo Bae, Kyoung-Mee Kim, Mee Soo Chang, Hye Seung Han, Joon Mee Kim, Hwal Woong Kim, Hee Kyung Chang, Young Hee Choi, Ji Y. Park, Mi Jin Gu, Min Jin Lhee, Jung Yeon Kim, Hee Sung Kim, Mee-Yon Cho
- J Pathol Transl Med. 2017;51(2):103-121. Published online February 19, 2017
- DOI: https://doi.org/10.4132/jptm.2017.01.24
- 24,256 View
- 910 Download
- 62 Web of Science
- 54 Crossref
-
 Abstract
Abstract
 PDF
PDF - With recent advances in molecular diagnostic methods and targeted cancer therapies, several molecular tests have been recommended for gastric cancer (GC) and colorectal cancer (CRC). Microsatellite instability analysis of gastrointestinal cancers is performed to screen for Lynch syndrome, predict favorable prognosis, and screen patients for immunotherapy. The epidermal growth factor receptor (EGFR) tyrosine kinase inhibitor has been approved in metastatic CRCs with wildtype RAS (KRAS and NRAS exon 2–4). A BRAF mutation is required for predicting poor prognosis. Additionally, amplification of human epidermal growth factor receptor 2 (HER2) and MET is also associated with resistance to EGFR inhibitor in metastatic CRC patients. The BRAF V600E mutation is found in sporadic microsatellite unstable CRCs, and thus is helpful for ruling out Lynch syndrome. In addition, the KRAS mutation is a prognostic biomarker and the PIK3CA mutation is a molecular biomarker predicting response to phosphoinositide 3-kinase/AKT/mammalian target of rapamycin inhibitors and response to aspirin therapy in CRC patients. Additionally, HER2 testing should be performed in all recurrent or metastatic GCs. If the results of HER2 immunohistochemistry are equivocal, HER2 silver or fluorescence in situ hybridization testing are essential for confirmative determination of HER2 status. Epstein-Barr virus–positive GCs have distinct characteristics, including heavy lymphoid stroma, hypermethylation phenotype, and high expression of immune modulators. Recent advances in next-generation sequencing technologies enable us to examine various genetic alterations using a single test. Pathologists play a crucial role in ensuring reliable molecular testing and they should also take an integral role between molecular laboratories and clinicians.
-
Citations
Citations to this article as recorded by- Spatial and Temporal Tumor Heterogeneity in Gastric Cancer: Discordance of Predictive Biomarkers
Hye Seung Lee
Journal of Gastric Cancer.2025; 25(1): 192. CrossRef - Korean Practice Guidelines for Gastric Cancer 2024: An Evidence-based, Multidisciplinary Approach (Update of 2022 Guideline)
In-Ho Kim, Seung Joo Kang, Wonyoung Choi, An Na Seo, Bang Wool Eom, Beodeul Kang, Bum Jun Kim, Byung-Hoon Min, Chung Hyun Tae, Chang In Choi, Choong-kun Lee, Ho Jung An, Hwa Kyung Byun, Hyeon-Su Im, Hyung-Don Kim, Jang Ho Cho, Kyoungjune Pak, Jae-Joon Kim
Journal of Gastric Cancer.2025; 25(1): 5. CrossRef - Elucidating the Role of KRAS, NRAS, and BRAF Mutations and Microsatellite Instability in Colorectal Cancer via Next-Generation Sequencing
Marta Rada Rodríguez, Bárbara Angulo Biedma, Irene Rodríguez Pérez, Javier Azúa Romeo
Cancers.2025; 17(13): 2071. CrossRef - Chitosan and Its Derivative‐Based Nanoparticles in Gastrointestinal Cancers: Molecular Mechanisms of Action and Promising Anticancer Strategies
Zahra Shokati Eshkiki, Fatemeh Mansouri, Amir Reza Karamzadeh, Abolfazl Namazi, Hafez Heydari, Javad Akhtari, Seidamir Pasha Tabaeian, Abolfazl Akbari, Hongda Liu
Journal of Clinical Pharmacy and Therapeutics.2024;[Epub] CrossRef - Colorectal Cancer: Genetic Underpinning and Molecular Therapeutics for Precision Medicine
Gideon T. Dosunmu, Ardaman Shergill
Genes.2024; 15(5): 538. CrossRef - Effector Function Characteristics of Exhausted CD8+ T-Cell in Microsatellite Stable and Unstable Gastric Cancer
Dong-Seok Han, Yoonjin Kwak, Seungho Lee, Soo Kyung Nam, Seong-Ho Kong, Do Joong Park, Hyuk-Joon Lee, Nak-Jung Kwon, Hye Seung Lee, Han-Kwang Yang
Cancer Research and Treatment.2024; 56(4): 1146. CrossRef - A Standardized Pathology Report for Gastric Cancer: 2nd Edition
Young Soo Park, Myeong-Cherl Kook, Baek-hui Kim, Hye Seung Lee, Dong-Wook Kang, Mi-Jin Gu, Ok Ran Shin, Younghee Choi, Wonae Lee, Hyunki Kim, In Hye Song, Kyoung-Mee Kim, Hee Sung Kim, Guhyun Kang, Do Youn Park, So-Young Jin, Joon Mee Kim, Yoon Jung Choi,
Journal of Gastric Cancer.2023; 23(1): 107. CrossRef - A standardized pathology report for gastric cancer: 2nd edition
Young Soo Park, Myeong-Cherl Kook, Baek-hui Kim, Hye Seung Lee, Dong-Wook Kang, Mi-Jin Gu, Ok Ran Shin, Younghee Choi, Wonae Lee, Hyunki Kim, In Hye Song, Kyoung-Mee Kim, Hee Sung Kim, Guhyun Kang, Do Youn Park, So-Young Jin, Joon Mee Kim, Yoon Jung Choi,
Journal of Pathology and Translational Medicine.2023; 57(1): 1. CrossRef - Korean Practice Guidelines for Gastric Cancer 2022: An Evidence-based, Multidisciplinary Approach
Tae-Han Kim, In-Ho Kim, Seung Joo Kang, Miyoung Choi, Baek-Hui Kim, Bang Wool Eom, Bum Jun Kim, Byung-Hoon Min, Chang In Choi, Cheol Min Shin, Chung Hyun Tae, Chung sik Gong, Dong Jin Kim, Arthur Eung-Hyuck Cho, Eun Jeong Gong, Geum Jong Song, Hyeon-Su Im
Journal of Gastric Cancer.2023; 23(1): 3. CrossRef - Influence of location-dependent sex difference on PD-L1, MMR/MSI, and EGFR in colorectal carcinogenesis
Jina Choi, Nayoung Kim, Ryoung Hee Nam, Jin Won Kim, Chin-Hee Song, Hee Young Na, Gyeong Hoon Kang, Alvaro Galli
PLOS ONE.2023; 18(2): e0282017. CrossRef - Comprehensive Analysis of Epigenetic Associated Genes with Differential
Gene Expression and Prognosis in Gastric Cancer
Songlin An, Xinbao Li, Bing Li, Yan Li
Combinatorial Chemistry & High Throughput Screening.2023; 26(3): 527. CrossRef - Liquid Biopsy in Advanced Colorectal Cancer: Clinical Applications of Different Analytes
Marco Donatello Delcuratolo, Andrea Modrego-Sánchez, Maristella Bungaro, Beatriz Antón-Pascual, Santiago Teran, Valentina Dipace, Silvia Novello, Rocio Garcia-Carbonero, Francesco Passiglia, Cristina Graválos-Castro
Journal of Molecular Pathology.2023; 4(3): 128. CrossRef - Exosomal circ_0001190 Regulates the Progression of Gastric Cancer via miR-586/SOSTDC1 Axis
Chao Liu, Jing Yang, Fengchi Zhu, Zhiying Zhao, Lixue Gao
Biochemical Genetics.2022; 60(6): 1895. CrossRef - Optimization of pre‐analytical and analytical steps for DNA and RNA analysis of fresh cytology samples
Ana Dolinar, Gašper Grubelnik, Irena Srebotnik‐Kirbiš, Margareta Strojan Fležar, Margareta Žlajpah
Cancer Medicine.2022; 11(21): 4021. CrossRef - Retracted: Connexin 43 upregulation by dioscin‐inhibited gastric cancer metastasis by suppressing PI3K/Akt pathway
Food Science & Nutrition.2022;[Epub] CrossRef - Molecular Pathology of Gastric Cancer
Moonsik Kim, An Na Seo
Journal of Gastric Cancer.2022; 22(4): 264. CrossRef - Case report: Undifferentiated sarcoma with multiple tumors involved in Lynch syndrome: Unexpected favorable outcome to sintilimab combined with chemotherapy
Jiaying Liu, Xiaona Chang, Guixiang Xiao, Jingmin Zhong, Bo Huang, Jiwei Zhang, Beibei Gao, Gang Peng, Xiu Nie
Frontiers in Oncology.2022;[Epub] CrossRef - The SUMO E3 ligase CBX4 is identified as a poor prognostic marker of gastric cancer through multipronged OMIC analyses
Yi Pan, Qingshang Li, Zhijun Cao, Shuliang Zhao
Genes & Diseases.2021; 8(6): 827. CrossRef - Worldwide variation in lynch syndrome screening: case for universal screening in low colorectal cancer prevalence areas
George Kunnackal John, Vipin Das Villgran, Christine Caufield-Noll, Francis Giardiello
Familial Cancer.2021; 20(2): 145. CrossRef - Tamoxifen Downregulates the Expression of Notch1 and DLL1 Genes in MKN-45 Gastric Cancer Cells
Faranak Khanipouyani, Hassan Akrami
Journal of Gastrointestinal Cancer.2021; 52(3): 922. CrossRef - Kallikrein-11, in Association with Coiled-Coil Domain Containing 25, as a Potential Prognostic Marker for Cholangiocarcinoma with Lymph Node Metastasis
Saeranee Siriphak, Ravinnipa Chanakankun, Tanakorn Proungvitaya, Sittiruk Roytrakul, Doungdean Tummanatsakun, Wunchana Seubwai, Molin Wongwattanakul, Siriporn Proungvitaya
Molecules.2021; 26(11): 3105. CrossRef - ISH-based HER2 diagnostics
Josef Rüschoff, Iris Nagelmeier, Bharat Jasani, Oliver Stoss
Der Pathologe.2021; 42(S1): 62. CrossRef - Identification and Analysis of Key Genes Driving Gastric Cancer Through Bioinformatics
Zhao Liu, Shihai Liu, Jing Guo, Libin Sun, Shasha Wang, Yixuan Wang, Wensheng Qiu, Jing Lv
Genetic Testing and Molecular Biomarkers.2021; 25(1): 1. CrossRef - Microsatellite Instability in Colorectal Cancer Liquid Biopsy—Current Updates on Its Potential in Non-Invasive Detection, Prognosis and as a Predictive Marker
Francis Yew Fu Tieng, Nadiah Abu, Learn-Han Lee, Nurul-Syakima Ab Mutalib
Diagnostics.2021; 11(3): 544. CrossRef - Metformin attenuates synergic effect of diabetes mellitus and Helicobacter pylori infection on gastric cancer cells proliferation by suppressing PTEN expression
Huibin Lu, Xinwei Han, Jianzhuang Ren, Kewei Ren, Zongming Li, Quanhui Zhang
Journal of Cellular and Molecular Medicine.2021; 25(10): 4534. CrossRef - Recent Advances in the Diagnosis, Staging, Treatment, and Prognosis of Advanced Gastric Cancer: A Literature Review
Zhi-da Chen, Peng-fei Zhang, Hong-qing Xi, Bo Wei, Lin Chen, Yun Tang
Frontiers in Medicine.2021;[Epub] CrossRef - Tumor immune response and immunotherapy in gastric cancer
Yoonjin Kwak, An Na Seo, Hee Eun Lee, Hye Seung Lee
Journal of Pathology and Translational Medicine.2020; 54(1): 20. CrossRef - Comparative analysis of HER2 copy number between plasma and tissue samples in gastric cancer using droplet digital PCR
Boram Kim, Soo Kyung Nam, Soo Hyun Seo, Kyoung Un Park, Sang-Hoon Ahn, Do Joong Park, Hyung-Ho Kim, Woo Ho Kim, Hye Seung Lee
Scientific Reports.2020;[Epub] CrossRef - Differential prognostic impact of CD8+ T cells based on human leucocyte antigen I and PD-L1 expression in microsatellite-unstable gastric cancer
Yoonjin Kwak, Jiwon Koh, Yujun Park, Yun Ji Hong, Kyoung Un Park, Hyung-Ho Kim, Do Joong Park, Sang-Hoon Ahn, Woo Ho Kim, Hye Seung Lee
British Journal of Cancer.2020; 122(9): 1399. CrossRef - High-Accuracy Determination of Microsatellite Instability Compatible with Liquid Biopsies
Amanda Bortolini Silveira, François-Clément Bidard, Amélie Kasperek, Samia Melaabi, Marie-Laure Tanguy, Manuel Rodrigues, Guillaume Bataillon, Luc Cabel, Bruno Buecher, Jean-Yves Pierga, Charlotte Proudhon, Marc-Henri Stern
Clinical Chemistry.2020; 66(4): 606. CrossRef - Chitosan: A compound for drug delivery system in gastric cancer-a review
Rana Shafabakhsh, Bahman Yousefi, Zatollah Asemi, Banafsheh Nikfar, Mohammad Ali Mansournia, Jamal Hallajzadeh
Carbohydrate Polymers.2020; 242: 116403. CrossRef - MSI and EBV Positive Gastric Cancer’s Subgroups and Their Link with Novel Immunotherapy
Maria Grazia Rodriquenz, Giandomenico Roviello, Alberto D’Angelo, Daniele Lavacchi, Franco Roviello, Karol Polom
Journal of Clinical Medicine.2020; 9(5): 1427. CrossRef - Theoretical calculations of molecular descriptors for anticancer activities of 1, 2, 3-triazole-pyrimidine derivatives against gastric cancer cell line (MGC-803): DFT, QSAR and docking approaches
Rhoda Oyeladun Oyewole, Abel Kolawole Oyebamiji, Banjo Semire
Heliyon.2020; 6(5): e03926. CrossRef - Identification of a Clinical Cutoff Value for Multiplex KRASG12/G13 Mutation Detection in Colorectal Adenocarcinoma Patients Using Digital Droplet PCR, and Comparison with Sanger Sequencing and PNA Clamping Assay
Kyung Ha Lee, Tae Hee Lee, Min Kyung Choi, In Sun Kwon, Go Eun Bae, Min-Kyung Yeo
Journal of Clinical Medicine.2020; 9(7): 2283. CrossRef - PD-L1 Testing in Gastric Cancer by the Combined Positive Score of the 22C3 PharmDx and SP263 Assay with Clinically Relevant Cut-offs
Yujun Park, Jiwon Koh, Hee Young Na, Yoonjin Kwak, Keun-Wook Lee, Sang-Hoon Ahn, Do Joong Park, Hyung-Ho Kim, Hye Seung Lee
Cancer Research and Treatment.2020; 52(3): 661. CrossRef - Clinical and Molecular Assessment of Patients with Lynch Syndrome and Sarcomas Underpinning the Association with MSH2 Germline Pathogenic Variants
Nathália de Angelis de Carvalho, Bianca Naomi Niitsuma, Vanessa Nascimento Kozak, Felipe D’almeida Costa, Mariana Petaccia de Macedo, Bruna Elisa Catin Kupper, Maria Letícia Gobo Silva, Maria Nirvana Formiga, Sahlua Miguel Volc, Samuel Aguiar Junior, Eden
Cancers.2020; 12(7): 1848. CrossRef - Farnesoid X receptor antagonizes Wnt/β-catenin signaling in colorectal tumorigenesis
Junhui Yu, Shan Li, Jing Guo, Zhengshui Xu, Jianbao Zheng, Xuejun Sun
Cell Death & Disease.2020;[Epub] CrossRef - YAP promotes self-renewal of gastric cancer cells by inhibiting expression of L-PTGDS and PTGDR2
Qingli Bie, Xiaozhe Li, Shiqi Liu, Xiao Yang, Zhenwen Qian, Rou Zhao, Xiaobei Zhang, Bin Zhang
International Journal of Clinical Oncology.2020; 25(12): 2055. CrossRef - ISH-basierte HER2-Diagnostik
Josef Rüschoff, Iris Nagelmeier, Bharat Jasani, Oliver Stoss
Der Pathologe.2020; 41(6): 606. CrossRef - Histone Deacetylase Inhibitor Trichostatin A Suppresses Cell Proliferation and Induces Apoptosis by Regulating the PI3K/AKT Signalling Pathway in Gastric Cancer Cells
Xinli An, Zekun Wei, Botian Ran, Hao Tian, Hongyu Gu, Yan Liu, Hongjuan Cui, Shunqin Zhu
Anti-Cancer Agents in Medicinal Chemistry.2020; 20(17): 2114. CrossRef - Role of Her-2 in Gastrointestinal Tumours beyond Gastric Cancer: A Tool for Precision Medicine
Csongor G. Lengyel, Baker Habeeb, Shah Z. Khan, Khalid El Bairi, Sara C. Altuna, Sadaqat Hussain, Syed Ayub Mazher, Dario Trapani, Angelica Petrillo
Gastrointestinal Disorders.2020; 3(1): 1. CrossRef - Next-generation Sequencing in the Management of Gastric and Esophageal Cancers
Jill C. Rubinstein, Norman G. Nicolson, Nita Ahuja
Surgical Clinics of North America.2019; 99(3): 511. CrossRef - Molecular profile in Paraguayan colorectal cancer patients, towards to a precision medicine strategy
Tania Fleitas-Kanonnikoff, Carolina Martinez‐Ciarpaglini, Josefina Ayala, Cinthia Gauna, Rita Denis, Ita Yoffe, Silvia Sforza, María Teresa Martínez, Alicia Pomata, Maider Ibarrola‐Villava, Sipan Arevshatyan, Verónica Burriel, Diego Boscá, Oscar Pastor, A
Cancer Medicine.2019; 8(6): 3120. CrossRef - Human epidermal growth factor receptor 2-positive digestive tumors
Anna D. Wagner, Berna C. Özdemir, Josef Rüschoff
Current Opinion in Oncology.2019; 31(4): 354. CrossRef - Assessing molecular subtypes of gastric cancer: microsatellite unstable and Epstein-Barr virus subtypes. Methods for detection and clinical and pathological implications
Carolina Martinez-Ciarpaglini, Tania Fleitas-Kanonnikoff, Valentina Gambardella, Marta Llorca, Cristina Mongort, Regina Mengual, Gema Nieto, Lara Navarro, Marisol Huerta, Susana Rosello, Desamparados Roda, Noelia Tarazona, Samuel Navarro, Gloria Ribas, An
ESMO Open.2019; 4(3): e000470. CrossRef - Current and future molecular diagnostics of gastric cancer
Rachel Sin-Yu Choi, Wing Yin Xenia Lai, Lok Ting Claire Lee, Wing Lam Christa Wong, Xiao Meng Pei, Hin Fung Tsang, Joel Johnson Leung, William Chi Shing Cho, Man Kee Maggie Chu, Elaine Yue Ling Wong, Sze Chuen Cesar Wong
Expert Review of Molecular Diagnostics.2019; 19(10): 863. CrossRef - Clinicopathologic significance of human leukocyte antigen class I expression in patients with stage II and III gastric cancer
Yujun Park, Jiwon Koh, Yoonjin Kwak, Sang-Hoon Ahn, Do Joong Park, Hyung-Ho Kim, Woo Ho Kim, Hye Seung Lee
Cancer Immunology, Immunotherapy.2019; 68(11): 1779. CrossRef - Development and Validation of an Easy-to-Implement, Practical Algorithm for the Identification of Molecular Subtypes of Gastric Cancer: Prognostic and Therapeutic Implications
Jiwon Koh, Keun-Wook Lee, Soo Kyung Nam, An Na Seo, Ji-Won Kim, Jin Won Kim, Do Joong Park, Hyung-Ho Kim, Woo Ho Kim, Hye Seung Lee
The Oncologist.2019; 24(12): e1321. CrossRef - Mechanisms and Therapy for Cancer Metastasis to the Brain
Federica Franchino, Roberta Rudà, Riccardo Soffietti
Frontiers in Oncology.2018;[Epub] CrossRef - Status of programmed death-ligand 1 expression in sarcomas
Hyung Kyu Park, Mingi Kim, Minjung Sung, Seung Eun Lee, Yu Jin Kim, Yoon-La Choi
Journal of Translational Medicine.2018;[Epub] CrossRef - Design and synthesis of near-infrared fluorescence-enhancement probes for the cancer-specific enzyme hNQO1
Changyu Zhang, Bei-Bei Zhai, Tao Peng, Zelin Zhong, Lianbin Xu, Qiang-Zhe Zhang, Lu-Yuan Li, Long Yi, Zhen Xi
Dyes and Pigments.2017; 143: 245. CrossRef - Progress in the treatment of advanced gastric cancer
Zheyu Song, Yuanyu Wu, Jiebing Yang, Dingquan Yang, Xuedong Fang
Tumor Biology.2017; 39(7): 101042831771462. CrossRef - Pathologische Einteilung und Diagnostik des Ösophagus- und Magenkarzinoms
S. Förster, A. Tannapfel
Der Gastroenterologe.2017; 12(5): 394. CrossRef - NR4A1-induced increase in the sensitivity of a human gastric cancer line to TNFα-mediated apoptosis is associated with the inhibition of JNK/Parkin-dependent mitophagy
Hongzhu Yan, Feng Xiao, Jue Zou, Chengmin Qiu, Weiwei Sun, Minmin Gu, Li Zhang
International Journal of Oncology.2017;[Epub] CrossRef
- Spatial and Temporal Tumor Heterogeneity in Gastric Cancer: Discordance of Predictive Biomarkers
- The Utilization of Cytologic Fine-Needle Aspirates of Lung Cancer for Molecular Diagnostic Testing
- Michael H. Roh
- J Pathol Transl Med. 2015;49(4):300-309. Published online June 16, 2015
- DOI: https://doi.org/10.4132/jptm.2015.06.16
- 17,759 View
- 173 Download
- 35 Web of Science
- 34 Crossref
-
 Abstract
Abstract
 PDF
PDF - In this era of precision medicine, our understanding and knowledge of the molecular landscape associated with lung cancer pathogenesis continues to evolve. This information is being increasingly exploited to treat advanced stage lung cancer patients with tailored, targeted therapy. During the management of these patients, minimally invasive procedures to obtain samples for tissue diagnoses are desirable. Cytologic fine-needle aspirates are often utilized for this purpose and are important not only for rendering diagnoses to subtype patients’ lung cancers, but also for ascertaining molecular diagnostic information for treatment purposes. Thus, cytologic fine-needle aspirates must be utilized and triaged judiciously to achieve both objectives. In this review, strategies in utilizing fine-needle aspirates will be discussed in the context of our current understanding of the clinically actionable molecular aberrations underlying non-small cell lung cancer and the molecular assays applied to these samples in order to obtain treatment-relevant molecular diagnostic information.
-
Citations
Citations to this article as recorded by- The Bridge: Supernatant Derived From Cytological Sample Preparations
Sinchita Roy‐Chowdhuri
Cytopathology.2025; 36(3): 222. CrossRef - Optimizing cytology and small biopsy specimen processing for ancillary studies: recommendations from the American Society of Cytopathology taskforce
Sinchita Roy-Chowdhuri, Christine N. Booth, Jonas J. Heymann, Elizabeth Jenkins, Joshua R. Menke, Sara E. Monaco, Ritu Nayar, Michiya Nishino, Roberto Ruiz-Cordero, Donna K. Russell, Anjali Saqi, Kaitlin E. Sundling, Michael J. Thrall, Vanda F. Torous, Ch
Journal of the American Society of Cytopathology.2025; 14(5): 285. CrossRef - Utility of Cell Block Preparation From CT-Guided FNAC with Immunohistochemistry in Diagnosing Primary Lung Carcinoma: A Prospective Study in A Tertiary Care Hospital
Subhargha Mandal, Abhishek Chowdhury, Riju Bhattacharyya, Palas Bhattacharyya
National Journal of Medical Research.2025; 15(04): 286. CrossRef - The American Cancer Society National Lung Cancer Roundtable strategic plan: Methods for improving turnaround time of comprehensive biomarker testing in non–small cell lung cancer
Sinchita Roy‐Chowdhuri, Haresh Mani, Adam H. Fox, Anne Tsao, Lynette M. Sholl, Farhood Farjah, Bruce E. Johnson, Raymond U. Osarogiagbon, M. Patricia Rivera, Gerard A. Silvestri, Robert A. Smith, Ignacio I. Wistuba
Cancer.2024; 130(24): 4200. CrossRef - Endobronchial ultrasound bronchoscopy Franseen fine needle biopsy tool versus standard fine needle aspiration needle: Impact on diagnosis and tissue adequacy
Matthew C. Aboudara, Timothy Saettele, Ossama Tawfik
Respiratory Medicine.2023; 208: 107131. CrossRef - Adequacy of small biopsy and cytology specimens for comprehensive genomic profiling of patients with non-small-cell lung cancer to determine eligibility for immune checkpoint inhibitor and targeted therapy
Erin Faber, Horiana Grosu, Sharjeel Sabir, Francis Anthony San Lucas, Bedia A Barkoh, Roland L Bassett, Rajyalakshmi Luthra, John Stewart, Sinchita Roy-Chowdhuri
Journal of Clinical Pathology.2022; 75(9): 612. CrossRef - Biomarker testing of cytology specimens in personalized medicine for lung cancer patients
Hyojin Kim, Jin-Haeng Chung
Journal of Pathology and Translational Medicine.2022; 56(6): 326. CrossRef - Formalin fixation for optimal concordance of programmed death‐ligand 1 immunostaining between cytologic and histologic specimens from patients with non–small cell lung cancer
Bregje M. Koomen, Jose van der Starre‐Gaal, Judith M. Vonk, Jan H. von der Thüsen, Jacqueline J. C. van der Meij, Kim Monkhorst, Stefan M. Willems, Wim Timens, Nils A. ’t Hart
Cancer Cytopathology.2021; 129(4): 304. CrossRef - Collection and Handling of Thoracic Small Biopsy and Cytology Specimens for Ancillary Studies: Guidelines from the College of American Pathologists (CAP)
Sinchita Roy-Chowdhuri
Journal of Molecular Pathology.2021; 2(1): 23. CrossRef - A decade of change: Trends in the practice of cytopathology at a tertiary care cancer centre
Peyman Dinarvand, Chinhua Liu, Sinchita Roy‐Chowdhuri
Cytopathology.2021; 32(5): 604. CrossRef - False‐negative programmed death‐ligand 1 immunostaining in ethanol‐fixed endobronchial ultrasound‐guided transbronchial needle aspiration specimens of non‐small‐cell lung cancer patients
Bregje M Koomen, Willem Vreuls, Mirthe de Boer, Emma J de Ruiter, Juergen Hoelters, Aryan Vink, Stefan M Willems
Histopathology.2021; 79(4): 480. CrossRef - Influence of preanalytical variables on performance of delta-like protein 3 (DLL3) predictive immunohistochemistry
Teodora Radonic, S. Duin, W. Vos, P. Kortman, Aeilko H. Zwinderman, Erik Thunnissen
Virchows Archiv.2021; 478(2): 293. CrossRef - Comparison of the SuperARMS and ARMS for detecting EGFR mutations in liquid-based cytology specimens from NSCLC patients
Wei Wu, Ziyang Cao, Wei Zhang, Liping Zhang, Likun Hou, Chunyan Wu
Diagnostic Pathology.2020;[Epub] CrossRef - Tumor cell representation by an improvised technique of fine-needle aspiration specimen acquisition and cell block preparation: Our experience in lung cancer cases in a peripheral center of eastern India
AnupKr Boler, Shreosee Roy, Arghya Bandyopadhyay, Abhishek Bandyopadhyay, MrinalKanti Ghosh
Journal of Cytology.2020; 37(2): 87. CrossRef - A new guideline from the College of American Pathologists to improve the adequacy of thoracic small specimens for ancillary studies
Sinchita Roy‐Chowdhuri
Cancer Cytopathology.2020; 128(10): 690. CrossRef - The utilization of cytologic and small biopsy samples for ancillary molecular testing
Michael H. Roh
Modern Pathology.2019; 32: 77. CrossRef - Diff-Quik Cytology Smears from Endobronchial Ultrasound Transbronchial Needle Aspiration Lymph Node Specimens as a Source of DNA for Next-Generation Sequencing Instead of Cell Blocks
David Fielding, Andrew J. Dalley, Farzad Bashirzadeh, Mahendra Singh, Lakshmy Nandakumar, Amy E. McCart Reed, Debra Black, Stephen Kazakoff, John V. Pearson, Katia Nones, Nicola Waddell, Sunil R. Lakhani, Peter T. Simpson
Respiration.2019; 97(6): 525. CrossRef - Molecular testing on endobronchial ultrasound (EBUS) fine needle aspirates (FNA): Impact of triage
Simon Sung, John P. Crapanzano, David DiBardino, David Swinarski, William A. Bulman, Anjali Saqi
Diagnostic Cytopathology.2018; 46(2): 122. CrossRef - Salvaging the supernatant: next generation cytopathology for solid tumor mutation profiling
Sinchita Roy-Chowdhuri, Meenakshi Mehrotra, Ana Maria Bolivar, Rashmi Kanagal-Shamanna, Bedia A. Barkoh, Brette Hannigan, Stephanie Zalles, Wenrui Ye, Dzifa Duose, Russell Broaddus, Gregg Staerkel, Ignacio Wistuba, L. Jeffrey Medeiros, Rajyalakshmi Luthra
Modern Pathology.2018; 31(7): 1036. CrossRef - Fluorescence in situ hybridization analysis on cytologic smears: An accurate and efficient method in the diagnosis of melanotic Xp11 translocation renal cancer
Alia Gupta, Mark Micale, Kurt D. Bernacki
Diagnostic Cytopathology.2018; 46(9): 786. CrossRef - Comprehensive Validation of Cytology Specimens for Next-Generation Sequencing and Clinical Practice Experience
Agnes Balla, Ken J. Hampel, Mukesh K. Sharma, Catherine E. Cottrell, Nikoletta Sidiropoulos
The Journal of Molecular Diagnostics.2018; 20(6): 812. CrossRef - Advances in Molecular Testing Techniques in Cytologic Specimens
Sinchita Roy-Chowdhuri
Surgical Pathology Clinics.2018; 11(3): 669. CrossRef - Next-generation molecular diagnosis: single-cell sequencing from bench to bedside
Wanjun Zhu, Xiao-Yan Zhang, Sadie L. Marjani, Jialing Zhang, Wengeng Zhang, Shixiu Wu, Xinghua Pan
Cellular and Molecular Life Sciences.2017; 74(5): 869. CrossRef - Big data from small samples: Informatics of next‐generation sequencing in cytopathology
Sinchita Roy‐Chowdhuri, Somak Roy, Sara E. Monaco, Mark J. Routbort, Liron Pantanowitz
Cancer Cytopathology.2017; 125(4): 236. CrossRef - Next‐generation sequencing of liquid‐based cytology non–small cell lung cancer samples
Jordan P. Reynolds, Yaolin Zhou, Maureen A. Jakubowski, Zhen Wang, Jennifer A. Brainard, Roger D. Klein, Carol F. Farver, Francisco A. Almeida, Yu‐Wei Cheng
Cancer Cytopathology.2017; 125(3): 178. CrossRef - Concurrent fine needle aspirations and core needle biopsies: a comparative study of substrates for next-generation sequencing in solid organ malignancies
Sinchita Roy-Chowdhuri, Hui Chen, Rajesh R Singh, Savitri Krishnamurthy, Keyur P Patel, Mark J Routbort, Jawad Manekia, Bedia A Barkoh, Hui Yao, Sharjeel Sabir, Russell R Broaddus, L Jeffrey Medeiros, Gregg Staerkel, John Stewart, Rajyalakshmi Luthra
Modern Pathology.2017; 30(4): 499. CrossRef - Diagnosis of anaplastic lymphoma kinase rearrangement in cytological samples through a fluorescence in situ hybridization–based assay: Cytological smears versus cell blocks
Federica Zito Marino, Giulio Rossi, Matteo Brunelli, Maria Gabriella Malzone, Giuseppina Liguori, Giuseppe Bogina, Alessandro Morabito, Gaetano Rocco, Renato Franco, Gerardo Botti
Cancer Cytopathology.2017; 125(5): 303. CrossRef - Next‐generation sequencing of cytologic preparations: An analysis of quality metrics
David H. Hwang, Elizabeth P. Garcia, Matthew D. Ducar, Edmund S. Cibas, Lynette M. Sholl
Cancer Cytopathology.2017; 125(10): 786. CrossRef - Recent advances in the pathology and molecular genetics of lung cancer: A practical review for cytopathologists
Erika F. Rodriguez, Sara E. Monaco
Journal of the American Society of Cytopathology.2016; 5(5): 252. CrossRef - Utilization of ancillary studies in the cytologic diagnosis of respiratory lesions: The papanicolaou society of cytopathology consensus recommendations for respiratory cytology
Lester J. Layfield, Sinchita Roy‐Chowdhuri, Zubair Baloch, Hormoz Ehya, Kim Geisinger, Susan J. Hsiao, Oscar Lin, Neal I. Lindeman, Michael Roh, Fernando Schmitt, Nikoletta Sidiropoulos, Paul A. VanderLaan
Diagnostic Cytopathology.2016; 44(12): 1000. CrossRef - Inflammatory myofibroblastic tumour
Michael McDermott
Seminars in Diagnostic Pathology.2016; 33(6): 358. CrossRef - Preanalytic Variables in Cytology: Lessons Learned From Next-Generation Sequencing—The MD Anderson Experience
Sinchita Roy-Chowdhuri, John Stewart
Archives of Pathology & Laboratory Medicine.2016; 140(11): 1191. CrossRef - Biomarker Testing in Lung Carcinoma Cytology Specimens: A Perspective From Members of the Pulmonary Pathology Society
Sinchita Roy-Chowdhuri, Dara L. Aisner, Timothy Craig Allen, Mary Beth Beasley, Alain Borczuk, Philip T. Cagle, Vera Capelozzi, Sanja Dacic, Gilda da Cunha Santos, Lida P. Hariri, Keith M. Kerr, Sylvie Lantuejoul, Mari Mino-Kenudson, Andre Moreira, Kirtee
Archives of Pathology & Laboratory Medicine.2016; 140(11): 1267. CrossRef - Identification of a novel partner gene, KIAA1217, fused to RET: Functional characterization and inhibitor sensitivity of two isoforms in lung adenocarcinoma
Mi-Sook Lee, Ryong Nam Kim, Hoseok I, Doo-Yi Oh, Ji-Young Song, Ka-Won Noh, Yu-Jin Kim, Jung Wook Yang, Maruja E. Lira, Chang Hun Lee, Min Ki Lee, Yeoung Dae Kim, Mao Mao, Joungho Han, Jhingook Kim, Yoon-La Choi
Oncotarget.2016; 7(24): 36101. CrossRef
- The Bridge: Supernatant Derived From Cytological Sample Preparations
- Molecular Imaging in the Era of Personalized Medicine
- Kyung-Ho Jung, Kyung-Han Lee
- J Pathol Transl Med. 2015;49(1):5-12. Published online January 15, 2015
- DOI: https://doi.org/10.4132/jptm.2014.10.24
- 16,113 View
- 215 Download
- 32 Web of Science
- 27 Crossref
-
 Abstract
Abstract
 PDF
PDF - Clinical imaging creates visual representations of the body interior for disease assessment. The role of clinical imaging significantly overlaps with that of pathology, and diagnostic workflows largely depend on both fields. The field of clinical imaging is presently undergoing a radical change through the emergence of a new field called molecular imaging. This new technology, which lies at the intersection between imaging and molecular biology, enables noninvasive visualization of biochemical processes at the molecular level within living bodies. Molecular imaging differs from traditional anatomical imaging in that biomarkers known as imaging probes are used to visualize target molecules-of-interest. This ability opens up exciting new possibilities for applications in oncologic, neurological and cardiovascular diseases. Molecular imaging is expected to make major contributions to personalized medicine by allowing earlier diagnosis and predicting treatment response. The technique is also making a huge impact on pharmaceutical development by optimizing preclinical and clinical tests for new drug candidates. This review will describe the basic principles of molecular imaging and will briefly touch on three examples (from an immense list of new techniques) that may contribute to personalized medicine: receptor imaging, angiogenesis imaging, and apoptosis imaging.
-
Citations
Citations to this article as recorded by- Ionic Cell Microscopy: A new modality for visualizing cells using microfluidic impedance cytometry and generative artificial intelligence
Mahtab Kokabi, Gulam M. Rather, Mehdi Javanmard
Biosensors and Bioelectronics: X.2025; 24: 100619. CrossRef - Multimodal imaging in cancer detection: the role of SPIONs and USPIONs as contrast agents for MRI, SPECT, and PET
Zahra Shaghaghi, Ramin Mansouri, Sahar Nosrati, Maryam Alvandi
Future Oncology.2025; 21(18): 2367. CrossRef - Supramolecular fluorescence biosensing based on macrocycles
Jia-Hong Tian, Haiqi Xu, Xin-Yue Hu, Dong-Sheng Guo
Supramolecular Materials.2024; 3: 100063. CrossRef - A non-invasive osteopontin-targeted phase changeable fluorescent nanoprobe for molecular imaging of myocardial fibrosis
Xueli Zhao, Yuze Qin, Bo Wang, Jiao Liu, Yueyue Wang, Kun Chen, Jia Zhao, Lanlan Zhang, Yuanming Wu, Liwen Liu
Nanoscale Advances.2024; 6(14): 3590. CrossRef - The Role of Molecular Imaging in Personalized Medicine
Suliman Salih, Aisyah Elliyanti, Ajnas Alkatheeri, Fatima AlYafei, Bashayer Almarri, Hasina Khan
Journal of Personalized Medicine.2023; 13(2): 369. CrossRef - Development of a multifunctional platform for near-infrared imaging and targeted radionuclide therapy for tumors
Huihui He, Ke Li, Hang Li, Shiliang Zhu, Shuai Qin, Yong Mao, Jianguo Lin, Ling Qiu, Chunjing Yu
European Journal of Pharmaceutics and Biopharmaceutics.2023; 185: 107. CrossRef - Quantum Biotechnology
Nicolas P. Mauranyapin, Alex Terrasson, Warwick P. Bowen
Advanced Quantum Technologies.2022;[Epub] CrossRef - Preparation Fe3O4@chitosan-graphene quantum dots nanocomposites for fluorescence and magnetic resonance imaging
Kai Wang, Xiaoguang Xu, Yan Li, Mayifei Rong, Lifeng Wang, Liying Lu, Jian Wang, Fengwen Zhao, Bowen Sun, Yong Jiang
Chemical Physics Letters.2021; 783: 139060. CrossRef - Network Medicine: A Clinical Approach for Precision Medicine and Personalized Therapy in Coronary Heart Disease
Teresa Infante, Luca Del Viscovo, Maria Luisa De Rimini, Sergio Padula, Pio Caso, Claudio Napoli
Journal of Atherosclerosis and Thrombosis.2020; 27(4): 279. CrossRef - Nanodrug Delivery Systems for the Treatment of Ovarian Cancer
Jonathan M. Pantshwa, Pierre P. D. Kondiah, Yahya E. Choonara, Thashree Marimuthu, Viness Pillay
Cancers.2020; 12(1): 213. CrossRef - Molecular imaging of the urokinase plasminogen activator receptor: opportunities beyond cancer
V. M. Baart, R. D. Houvast, L. F. de Geus-Oei, P. H. A. Quax, P. J. K. Kuppen, A. L. Vahrmeijer, C. F. M. Sier
EJNMMI Research.2020;[Epub] CrossRef - In vivo SPECT imaging of an 131I-labeled PM 2.5 mimic substitute
Dong-Hui Pan, Jie Sheng, Xin-Yu Wang, Qian-Huan Huang, Jun-Jie Yan, Li-Zhen Wang, Run-Ling Yang, Dong-Jian Shi, Yu-Ping Xu, Ming-Qing Chen
Nuclear Science and Techniques.2020;[Epub] CrossRef - Optofluidics in bio-imaging applications
Sihui Chen, Rui Hao, Yi Zhang, Hui Yang
Photonics Research.2019; 7(5): 532. CrossRef - Nitrogen-vacancy centers in diamond for nanoscale magnetic resonance imaging applications
Alberto Boretti, Lorenzo Rosa, Jonathan Blackledge, Stefania Castelletto
Beilstein Journal of Nanotechnology.2019; 10: 2128. CrossRef - Online molecular image repository and analysis system: A multicenter collaborative open-source infrastructure for molecular imaging research and application
Mahabubur Rahman, Hiroshi Watabe
Computers in Biology and Medicine.2018; 96: 233. CrossRef - Nε-Acryloyllysine Piperazides as Irreversible Inhibitors of Transglutaminase 2: Synthesis, Structure–Activity Relationships, and Pharmacokinetic Profiling
Robert Wodtke, Christoph Hauser, Gloria Ruiz-Gómez, Elisabeth Jäckel, David Bauer, Martin Lohse, Alan Wong, Johanna Pufe, Friedrich-Alexander Ludwig, Steffen Fischer, Sandra Hauser, Dieter Greif, M. Teresa Pisabarro, Jens Pietzsch, Markus Pietsch, Reik Lö
Journal of Medicinal Chemistry.2018; 61(10): 4528. CrossRef - Genomic Interventions in Medicine
Oluwadurotimi S Aworunse, Oluwatomiwa Adeniji, Olusola L Oyesola, Itunuoluwa Isewon, Jelili Oyelade, Olawole O Obembe
Bioinformatics and Biology Insights.2018;[Epub] CrossRef - Restriction spectrum imaging: An evolving imaging biomarker in prostate MRI
Ryan L. Brunsing, Natalie M. Schenker-Ahmed, Nathan S. White, J. Kellogg Parsons, Christopher Kane, Joshua Kuperman, Hauke Bartsch, Andrew Karim Kader, Rebecca Rakow-Penner, Tyler M. Seibert, Daniel Margolis, Steven S. Raman, Carrie R. McDonald, Nikdokht
Journal of Magnetic Resonance Imaging.2017; 45(2): 323. CrossRef - Personalized medicine: a new option for nuclear medicine and molecular imaging in the third millennium
Orazio Schillaci, Nicoletta Urbano
European Journal of Nuclear Medicine and Molecular Imaging.2017; 44(4): 563. CrossRef - Nano-Magnetic Resonance Imaging (Nano-MRI) Gives Personalized Medicine a New Perspective
Lorenzo Rosa, Jonathan Blackledge, Albert Boretti
Biomedicines.2017; 5(1): 7. CrossRef - Optical nanoprobes for biomedical applications: shining a light on upconverting and near-infrared emitting nanoparticles for imaging, thermal sensing, and photodynamic therapy
E. Hemmer, P. Acosta-Mora, J. Méndez-Ramos, S. Fischer
Journal of Materials Chemistry B.2017; 5(23): 4365. CrossRef - Drug Discovery by Molecular Imaging and Monitoring Therapy Response in Lymphoma
Senthilkumar Kalimuthu, Ju Hye Jeong, Ji Min Oh, Byeong-Cheol Ahn
International Journal of Molecular Sciences.2017; 18(8): 1639. CrossRef - Chemistry and engineering of cyclodextrins for molecular imaging
Wing-Fu Lai, Andrey L. Rogach, Wing-Tak Wong
Chemical Society Reviews.2017; 46(20): 6379. CrossRef - Prototypes of Lanthanide(III) Agents Responsive to Enzymatic Activities in Three Complementary Imaging Modalities: Visible/Near-Infrared Luminescence, PARACEST-, and T1-MRI
Jiefang He, Célia S. Bonnet, Svetlana V. Eliseeva, Sara Lacerda, Thomas Chauvin, Pascal Retailleau, Frederic Szeremeta, Bernard Badet, Stéphane Petoud, Éva Tóth, Philippe Durand
Journal of the American Chemical Society.2016; 138(9): 2913. CrossRef - Nanoparticles in practice for molecular-imaging applications: An overview
Parasuraman Padmanabhan, Ajay Kumar, Sundramurthy Kumar, Ravi Kumar Chaudhary, Balázs Gulyás
Acta Biomaterialia.2016; 41: 1. CrossRef - A new neuroinformatics approach to personalized medicine in neurology: The Virtual Brain
Maria I. Falcon, Viktor Jirsa, Ana Solodkin
Current Opinion in Neurology.2016; 29(4): 429. CrossRef - Targeted multimodal nano-reporters for pre-procedural MRI and intra-operative image-guidance
Joonseok Lee, Andrew C. Gordon, Hacksung Kim, Wooram Park, Soojeong Cho, Byeongdu Lee, Andrew C. Larson, Elena A. Rozhkova, Dong-Hyun Kim
Biomaterials.2016; 109: 69. CrossRef
- Ionic Cell Microscopy: A new modality for visualizing cells using microfluidic impedance cytometry and generative artificial intelligence
- The Clinicopathologic Features of Molecular Apocrine Breast Cancer
- Yoon Jin Cha, Woo-Hee Jung, Ja Seung Koo
- Korean J Pathol. 2012;46(2):169-176. Published online April 25, 2012
- DOI: https://doi.org/10.4132/KoreanJPathol.2012.46.2.169
- 8,923 View
- 52 Download
- 12 Crossref
-
 Abstract
Abstract
 PDF
PDF Background To elucidate the clinicopathologic features and their implications on the immunohistochemistry in cases of molecular apocrine breast cancer (MABC).
Methods Immunohistochemical (IHC) staining for estrogen receptor (ER), human epidermal growth factor receptor 2 (HER-2), cytokeratin (CK) 5/6, epidermal growth factor receptor (EGFR), androgen receptor (AR), gamma-glutamyltrasferase 1 (GGT1) and Ki-67 was performed on tissue microarray breast cancer samples from 204 patients. Phenotypes of breast cancer were divided based on the IHC status of ER, AR and GGT1 into the following: luminal type, ER positive and AR and/or GGT1 positive; basal type, ER, AR, and GGT1 negative; non-basal type, ER positive and AR and GGT1 negative; and MABC type, ER negative and AR and/or GGT1 positive.
Results In our series of patients (n=204), there were 26 cases of MABC. Besides, there were 18, 60, and 100 cases of luminal type, basal type and non-basal type, respectively. The MABC demonstrated apocrine histology and a higher prevalence of HER-2 positivity than other phenotypes. With the basal type, the MABC manifested a more frequent expression of CK5/6 and EGFR and a higher Ki-67 index than other phenotypes (p<0.001). There were no significant differences in patient prognosis between the phenotypes of breast cancer.
Conclusions MABC are distinguishable from other phenotypes based on the apocrine histology and a higher expression rate of HER-2.
-
Citations
Citations to this article as recorded by- The role of histopathologic testing on apocrine carcinoma of the breast
Burak Ilhan, Selman Emiroğlu, Rustu Türkay, Rıdvan Ilhan
Current Problems in Cancer.2020; 44(2): 100501. CrossRef - Cancer du sein triple négatif exprimant le récepteur aux androgènes : de la biologie à la thérapeutique
Thomas Grellety
Bulletin du Cancer.2020; 107(4): 506. CrossRef - Triple‐negative apocrine carcinoma: A rare pathologic subtype with a better prognosis than other triple‐negative breast cancers
Cletus A. Arciero, Albert H. Diehl, Yuan Liu, Qin Sun, Theresa Gillespie, Xiaoxian Li, Preeti Subhedar
Journal of Surgical Oncology.2020; 122(6): 1232. CrossRef - Apocrine lesions of the breast: part 2 of a two-part review. Invasive apocrine carcinoma, the molecular apocrine signature and utility of immunohistochemistry in the diagnosis of apocrine lesions of the breast
Clare D'Arcy, Cecily M Quinn
Journal of Clinical Pathology.2019; 72(1): 7. CrossRef - Enhancing Abiraterone Acetate Efficacy in Androgen Receptor–positive Triple-negative Breast Cancer: Chk1 as a Potential Target
Thomas Grellety, Celine Callens, Elodie Richard, Adrien Briaux, Valérie Vélasco, Marina Pulido, Anthony Gonçalves, Pierre Gestraud, Gaetan MacGrogan, Hervé Bonnefoi, Bruno Cardinaud
Clinical Cancer Research.2019; 25(2): 856. CrossRef - A clinicopathologic study of invasive apocrine carcinoma of the breast: A single-center experience
Denira Imamovic, Nurija Bilalovic, Faruk Skenderi, Vanesa Beslagic, Timur Ceric, Berisa Hasanbegovic, Semir Beslija, Semir Vranic
The Breast Journal.2018; 24(6): 1105. CrossRef - Dose invasive apocrine adenocarcinoma has worse prognosis than invasive ductal carcinoma of breast: evidence from SEER database
Ning Zhang, Hanwen Zhang, Tong Chen, Qifeng Yang
Oncotarget.2017; 8(15): 24579. CrossRef - Pure Apocrine Carcinomas Represent a Clinicopathologically Distinct Androgen Receptor–Positive Subset of Triple-Negative Breast Cancers
Anne M. Mills, Chelsea E. Gottlieb, Scott M. Wendroth, Christiana M. Brenin, Kristen A. Atkins
American Journal of Surgical Pathology.2016; 40(8): 1109. CrossRef - Androgen Receptor: A Complex Therapeutic Target for Breast Cancer
Ramesh Narayanan, James Dalton
Cancers.2016; 8(12): 108. CrossRef - Early versus late distant metastasis and adjuvant chemotherapy alone versus both radiotherapy and chemotherapy in molecular apocrine breast cancer
Xiaozhen Liu, Yang Yang, Xiaolong Feng, Honghong Shen, Jian Liu, Xia Liu, Yun Niu
Oncotarget.2016; 7(31): 48905. CrossRef - Molecular and diagnostic features of apocrine breast lesions
Pavel Gromov, Jaime A Espinoza, Irina Gromova
Expert Review of Molecular Diagnostics.2015; 15(8): 1011. CrossRef - Prevalencia de receptores androgénicos en el cáncer de mama
Claudia Janeth Rodríguez-Silva, José Luis González-Vela, Ascary Alcides Velázquez-Pacheco
Gaceta Mexicana de Oncología.2015; 14(3): 135. CrossRef
- The role of histopathologic testing on apocrine carcinoma of the breast
- Comparison of the DNA Preservation in Neutral-Buffered Formalin Fixed Paraffin-Embedded Tissue and in Non-Buffered Formalin Fixed Paraffin-Embedded Tissue.
- An Na Seo, Jae Hoon Kim, Dakeun Lee, Ji Yun Jeong, Ji Young Park
- Korean J Pathol. 2011;45(6):549-556.
- DOI: https://doi.org/10.4132/KoreanJPathol.2011.45.6.549
- 5,427 View
- 64 Download
- 2 Crossref
-
 Abstract
Abstract
 PDF
PDF - BACKGROUND
The preservation of optimized DNA and its extraction from formalin-fixed, paraffin-embedded (FFPE) tissues are important issues. There has been some doubt over whether 10% neutral-buffered formalin is an ideal fixation solution for DNA preservation over non-buffered formalin, as conventionally recommended. In this study, the correlation between the efficiency of DNA extraction from FFPE tissues and buffered formalin was evaluated.
METHODS
Several tissues with same conditions except fixatives were fixed in four different formalin solution groups and were routinely processed as paraffin-embedding protocols. DNAs were extracted from four different FFPE tissues that were stored for over 3 months and over 9 months. The quantity and quality of the DNAs were assessed with a NanoDrop ND-1000 spectrophotometer, and the polymerase chain reaction (PCR) amplification and degradation were analyzed via microchip electrophoresis. KRAS mutation analysis and microsatellite instability (BAT25) PCR were performed with each sample.
RESULTS
The results showed no remarkable difference in the four groups.
CONCLUSIONS
The study findings demonstrate that DNA preservation is fairly unaffected by a neutral buffer where there is short formalin manufacture period and an adequate formalin fixation time before embedding in paraffin. -
Citations
Citations to this article as recorded by- Forensic analysis of formalin-fixed non-paraffin-embedded tissues: a comparative study of STR and mtDNA profiling
Kangana Aggarwal, Nidhi Sharma, Braja Kishore Mohapatra, Kamal Chauhan, Harpreet Singh, Chittaranjan Behera
Forensic Science, Medicine and Pathology.2025;[Epub] CrossRef - Comparison of Direct Sequencing, PNA Clamping-Real Time Polymerase Chain Reaction, and Pyrosequencing Methods for the Detection ofEGFRMutations in Non-small Cell Lung Carcinoma and the Correlation with Clinical Responses to EGFR Tyrosine Kinase Inhibitor
Hyun Ju Lee, Xianhua Xu, Hyojin Kim, Yan Jin, Pingli Sun, Ji Eun Kim, Jin-Haeng Chung
Korean Journal of Pathology.2013; 47(1): 52. CrossRef
- Forensic analysis of formalin-fixed non-paraffin-embedded tissues: a comparative study of STR and mtDNA profiling
- CD56 and High Molecular Weight Cytokeratin as Diagnostic Markers of Papillary Thyroid Carcinoma.
- Mi Kyung Shin, Jeong Won Kim, Young Su Ju
- Korean J Pathol. 2011;45(5):477-484.
- DOI: https://doi.org/10.4132/KoreanJPathol.2011.45.5.477
- 6,425 View
- 38 Download
- 10 Crossref
-
 Abstract
Abstract
 PDF
PDF - BACKGROUND
The incidence of papillary thyroid carcinoma (PTC) has been increasing recently and a precise diagnosis is essential for optimal treatment. Ancillary immunohistochemical stains are important for diagnosing some difficult cases.
METHODS
The dignostic value of CD56, high molecular weight cytokeratin (HMCK), galectin-3 (GAL3), and cytokeratin 19 (CK19) were evaluated to distinguish PTC from other benign thyroid lesions (BTL). We studied 23 cases of papillary thyroid overt carcinomas, 57 papillary thyroid microcarcinomas, five follicular adenomas, five cases of Hashimoto's thyroiditis, and 12 nodular hyperplasias.
RESULTS
The statistical analysis showed significantly different expressions of CD56, HMCK, GAL3, and CK19 in PTC vs other BTL. The diagnostic specificity of HMCK and CD56 (90.9% and 72.7%, respectively) was higher than that of GAL3 and CK19 (50.0% and 36.4%, respectively). However, the sensitivity of HMCK and CD56 detection (92.5% and 95.0%, respectively) was lower than that of GAL3 and CK19 (98.8% and 100.0%, respectively). The combined use of CD56, HMCK, GAL3, and CK19 showed 87.5% sensitivity, 100.0% specificity, and 100.0% positive predictive value in differentiating PTC from other BTL.
CONCLUSIONS
Although the differential diagnosis of thyroid follicular lesions are based on histological and cytomorphological criteria, CD56 and HMCK might be useful markers for diagnosing PTC. -
Citations
Citations to this article as recorded by- Diagnostic role of immunohistochemical markers CK19 and CD56 in thyroid neoplasms
Pallavi Priyadarshini, Manoj Kumar Patro, Prasanta Kumar Das
MGM Journal of Medical Sciences.2023; 10(2): 176. CrossRef - CD56 Expression in Papillary Thyroid Carcinoma Is Highly Dependent on the Histologic Subtype: A Potential Diagnostic Pitfall
Uiju Cho, Yourha Kim, Sora Jeon, Chan Kwon Jung
Applied Immunohistochemistry & Molecular Morphology.2022; 30(5): 389. CrossRef - CD-56 IMMUNOREACTIVITY IN FOLLICULAR CELL DERIVED LESIONS OF THYROID
Elvin Merin Cherian, Priya P. V, Sankar S
Journal of Evolution of Medical and Dental Sciences.2018; 7(17): 2066. CrossRef - Diagnostic utility of CK19 and CD56 in the differentiation of thyroid papillary carcinoma from its mimics
Nehal S. Abouhashem, Suzan M. Talaat
Pathology - Research and Practice.2017; 213(5): 509. CrossRef - Use of CD56 and cyclin D1 in differentiating thyroid hyperplasia from papillary thyroid carcinoma
Maha E. Salama, Wael S. Ibrahim
Egyptian Journal of Pathology.2016; 36(1): 39. CrossRef - CD56, HBME-1 and cytokeratin 19 expressions in papillary thyroid carcinoma and nodular thyroid lesions
Senay Erdogan-Durmus, Deniz Ozcan, Enver Yarikkaya, Ali Kurt, Aynur Arslan
Journal of Research in Medical Sciences.2016;[Epub] CrossRef - Defining the value of CD56, CK19, Galectin 3 and HBME-1 in diagnosis of follicular cell derived lesions of thyroid with systematic review of literature
Duško Dunđerović, Jasmina Marković Lipkovski, Ivan Boričic, Ivan Soldatović, Vesna Božic, Dubravka Cvejić, Svetislav Tatić
Diagnostic Pathology.2015;[Epub] CrossRef - Utility of immunohistochemical markers in differential diagnosis of follicular cell-derived thyroid lesions
HananAlSaeid Alshenawy
Journal of Microscopy and Ultrastructure.2014; 2(3): 127. CrossRef - Utility of Immunohistochemical Markers in Diagnosis of Follicular Cell Derived Thyroid Lesions
Hanan AlSaeid Alshenawy
Pathology & Oncology Research.2014; 20(4): 819. CrossRef - Potential diagnostic utility of CD56 and claudin-1 in papillary thyroid carcinoma and solitary follicular thyroid nodules
Rasha M. Abd El Atti, Lobna S. Shash
Journal of the Egyptian National Cancer Institute.2012; 24(4): 175. CrossRef
- Diagnostic role of immunohistochemical markers CK19 and CD56 in thyroid neoplasms
- Molecular Biologic Techniques in Cytopathologic Diagnosis.
- Tae Sook Hwang
- Korean J Pathol. 2009;43(5):387-392.
- DOI: https://doi.org/10.4132/KoreanJPathol.2009.43.5.387
- 5,659 View
- 50 Download
- 9 Crossref
-
 Abstract
Abstract
 PDF
PDF - Molecular diagnosis is an application of the knowledge on molecular mechanisms of disease to diagnosis, therapeutic decision-making, and prognostication. Basically any molecular diagnostic technique could be used in molecular diagnostic cytopathology. Currently applicable molecular techniques in the cytopathology field include PCR based molecular techniques (SSCP, DHPLC, RFLP, LOH, MSI, RT-PCR, QRT-PCR, allele-specific PCR, sequencing, and methylation analysis), FISH, cDNA microarray, aCGH, and reverse-phase protein microarray, etc. Exfoliative cytology as well as fine needle aspiration cytology specimen can be used for analysis. In order to obtain a successful result, collection of target cells without contamination of the blood cells, inflammatory cells including histiocytes, and stromal cells, and a good DNA yield are most important. Molecular diagosis finds its full meaning when interpreted by those who can combine the clinical background of the disease with morphological, immunocytochemical, and molecular diagnostic results. Therefore, these assays would fulfill their full potential when interpreted by the cytopathologists.
-
Citations
Citations to this article as recorded by- Clinical significance of EGFR mutation types in lung adenocarcinoma: A multi-centre Korean study
Hee-Young Yoon, Jeong-Seon Ryu, Yun Su Sim, Dojin Kim, Sung Yong Lee, Juwhan Choi, Sojung Park, Yon Ju Ryu, Jin Hwa Lee, Jung Hyun Chang, Jung Weon Lee
PLOS ONE.2020; 15(2): e0228925. CrossRef - Efficiency of EGFR mutation analysis for small microdissected cytological specimens using multitech DNA extraction solution
Seo Young Oh, Hoon Taek Lee
Cancer Cytopathology.2015; 123(7): 401. CrossRef - EGFR Analysis in Cytologic Samples of Lung Adenocarcinoma by Microdissection
Jeong Yeon Han, Hoon Taek Lee, Seo Young Oh
Korean Journal of Clinical Laboratory Science.2015; 47(3): 125. CrossRef - Improved DNA Extraction Method for Molecular Diagnosis from Smaller numbers of Cells
Seo Young Oh, Jeong Yeon Han, So Ra Lee, Hoon Taek Lee
Korean Journal of Clinical Laboratory Science.2014; 46(3): 99. CrossRef - Development of an Ammonium Sulfate DNA Extraction Method for Obtaining Amplifiable DNA in a Small Number of Cells and Its Application to Clinical Specimens
Seo Young Oh, Wook Youn Kim, Tae Sook Hwang, Hye Seung Han, So Dug Lim, Wan Seop Kim
BioMed Research International.2013; 2013: 1. CrossRef - Detection and comparison of peptide nucleic acid-mediated real-time polymerase chain reaction clamping and direct gene sequencing for epidermal growth factor receptor mutations in patients with non-small cell lung cancer
Hee Joung Kim, Kye Young Lee, Young-Chul Kim, Kyu-Sik Kim, Sung Yong Lee, Tae Won Jang, Min Ki Lee, Kyeong-Cheol Shin, Gwan Ho Lee, Jae Chol Lee, Jeong Eun Lee, Sun Young Kim
Lung Cancer.2012; 75(3): 321. CrossRef - Possible use and role of molecular techniques in fine-needle aspiration cytology (FNAC) practice
Fernando Schmitt, Helena Barroca
Diagnostic Histopathology.2011; 17(7): 286. CrossRef - Comparative Analysis of Peptide Nucleic Acid (PNA)-Mediated Real-Time PCR Clamping and DNA Direct Sequencing for EGFR Mutation Detection
Hee Joung Kim, Wan Seop Kim, Kyeong-Cheol Shin, Gwan Ho Lee, Mi-Jin Kim, Jeong Eun Lee, Kyu Sang Song, Sun Young Kim, Kye Young Lee
Tuberculosis and Respiratory Diseases.2011; 70(1): 21. CrossRef - PNA-Mediated PCR Clamping for the Detection of EGFR Mutations in Non-Small Cell Lung Cancer
Kye Young Lee, Hee Joung Kim, Sun Jong Kim, Gwang Ha Yoo, Won Dong Kim, Seo Young Oh, Wan Seop Kim
Tuberculosis and Respiratory Diseases.2010; 69(4): 271. CrossRef
- Clinical significance of EGFR mutation types in lung adenocarcinoma: A multi-centre Korean study
- The Utility of HMW-CK and CK5/6 Immunohistochemical Stains for Differentiating Ductal Proliferative Lesions and Ducal Carcinoma of the Breast.
- Sung Hee Son, Ju Yeon Song, Hye Kyoung Yoon
- Korean J Pathol. 2008;42(1):21-26.
- 4,415 View
- 119 Download
-
 Abstract
Abstract
 PDF
PDF - BACKGROUND
Basal-type cytokeratins may help to distinguish benign from malignant intraductal proliferative lesions. The basal-type cytokeratins expression is markedly decreased or absent in atypical ductal hyperplasia (ADH), ductal carcinoma in situ (DCIS) and invasive ductal carcinomas (IDC). However, the expression patterns vary according to the antibodies that are used for staining.
METHODS
HMW-CK (clone 34 E12) was applied to 175 lesions, and CK5/6 (clone D5/16B4) was applied to 145 lesions. The specimens were IDC (n=165), DCIS (n=35), ADH (n=37), florid ductal hyperplasia (FDH) (n=38) and columnar cell lesion (CCL) (n=45). The expression patterns of HMW-CK and CK5/6 were categorized as negative, focal positive and positive.
RESULTS
Loss of the HMW-CK expression was noted in 76% (66/87) of the IDC, 78% (21/27) of the DCIS, 78% (21/28) of the ADH, and 55% (10/18) of the FDH. Loss of the CK5/6 expression was found in 96% (75/78) of the IDC, in all the DCIS (n=8) and ADH (n=9), and in none of the FDH (n=20). Loss of the CK5/6 expression is more reliable than that of the HMW-CK expression for differentiating FDH, ADH and malignant intraductal proliferatve lesions. Eleven (73%) of 15 CCLs revealed the loss of the HMW-CK expression, but all the CCLs (n=30) were negative for CK5/6 (p=0.0161).
CONCLUSION
CK5/6 antibody is more reliable than HMW-CK antibody for differentiating FDH from ADH or DCIS, and for discriminating CCL.

 E-submission
E-submission

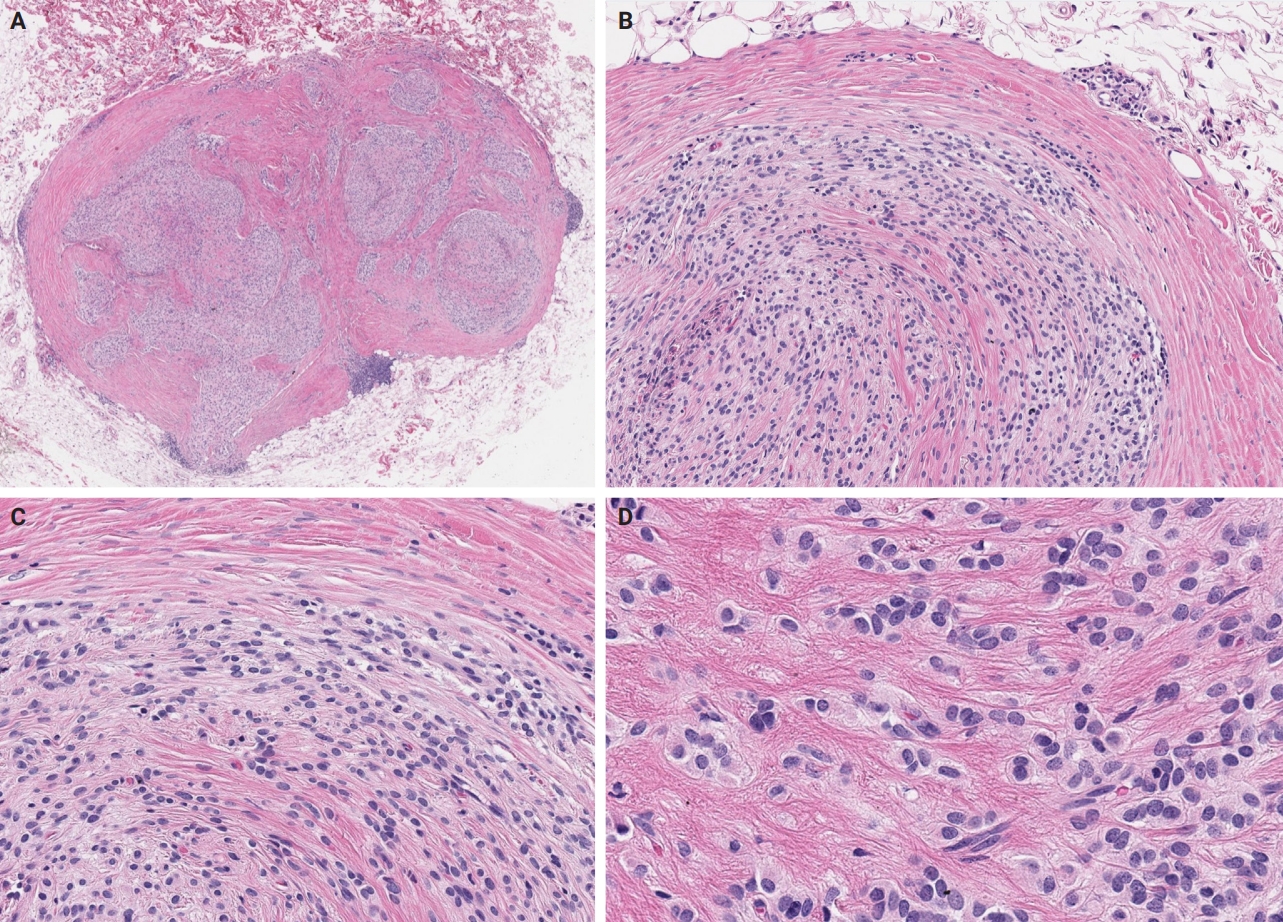
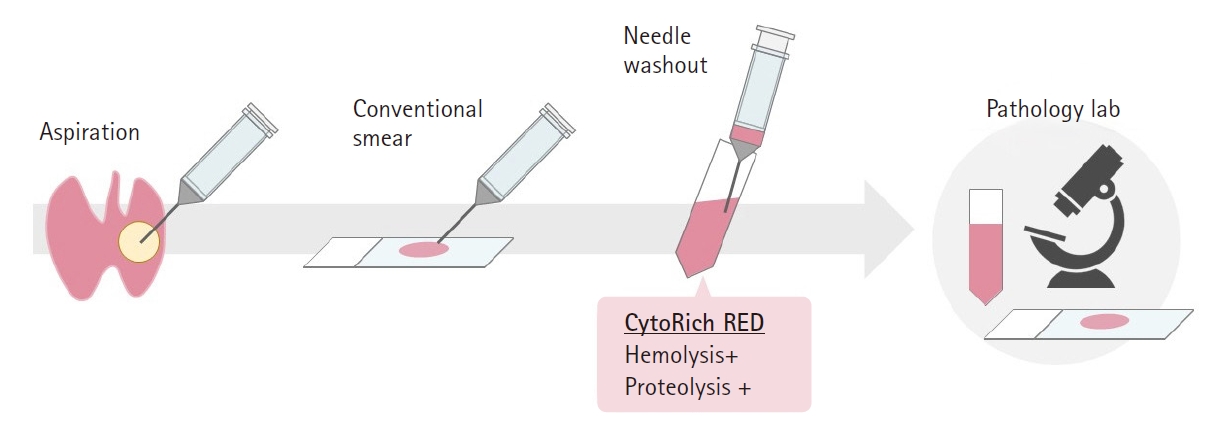











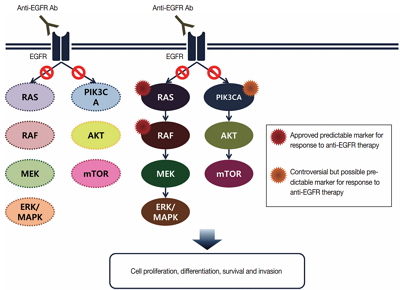
 First
First Prev
Prev



