Articles
- Page Path
- HOME > J Pathol Transl Med > Volume 47(6); 2013 > Article
-
Case Study
Cystic Benign Phyllodes Tumor in the Inguinal Region - Jai Hyang Go
-
Korean Journal of Pathology 2013;47(6):583-586.
DOI: https://doi.org/10.4132/KoreanJPathol.2013.47.6.583
Published online: December 24, 2013
Department of Pathology, Dankook University College of Medicine, Cheonan, Korea.
- Corresponding Author: Jai Hyang Go, M.D. Department of Pathology, Dankook University College of Medicine, 119 Dandae-ro, Dongnam-gu, Cheonan 330-997, Korea. Tel: +82-41-550-6972, Fax: +82-41-561-9127, cyjy555@hanmail.net
• Received: November 13, 2012 • Revised: January 21, 2013 • Accepted: January 22, 2013
© 2013 The Korean Society of Pathologists/The Korean Society for Cytopathology
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/3.0/) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
Abstract
- The present lesion was the first reported case of a benign intracystic phyllodes tumor in the inguinal region. We report the case of a 51-year-old female patient who presented with an inguinal mass. A clinical diagnosis of malignant lymphoma was considered in this case. The resected tumor was well-circumscribed and showed numerous papillary nodular protrusions into a central cystic cavity (3.5×2.5 cm). The microscopic findings showed hyperplastic epithelium-lined cysts with leaf-like intraluminal epithelium-lined bland stromal projections. The epithelial cell linings were strongly positive for estrogen and progesterone receptors.
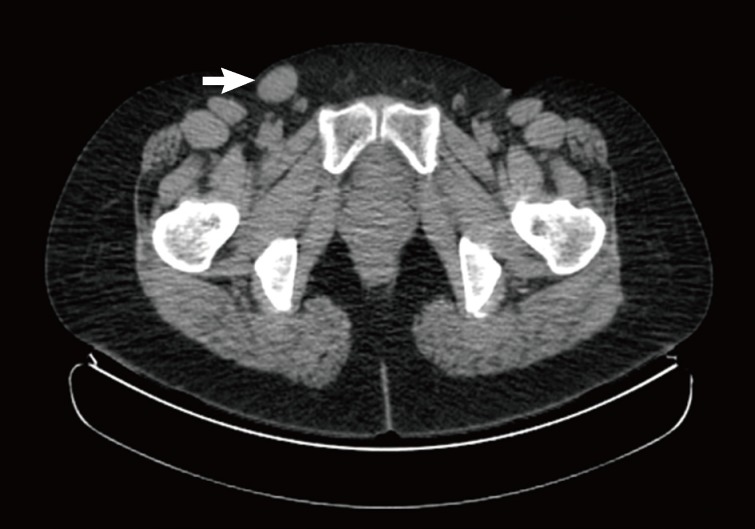
- A 51-year-old female was admitted to our hospital for the evaluation of a mass in the right groin area that had been present for two years. She had no specific clinical history. A mass lesion was identified with positron emission tomography-comcomputed tomography. The mass was 27 mm in the lmgest diameter with F-18 fluoro-deoxyglucose uptake (SUVmax 1.4), and it was located on the superolateral side of the inguinal area (Fig. 1). The mass was excised under local anesthesia. A differential diagnosis of lymphoma is the major concern in this case.
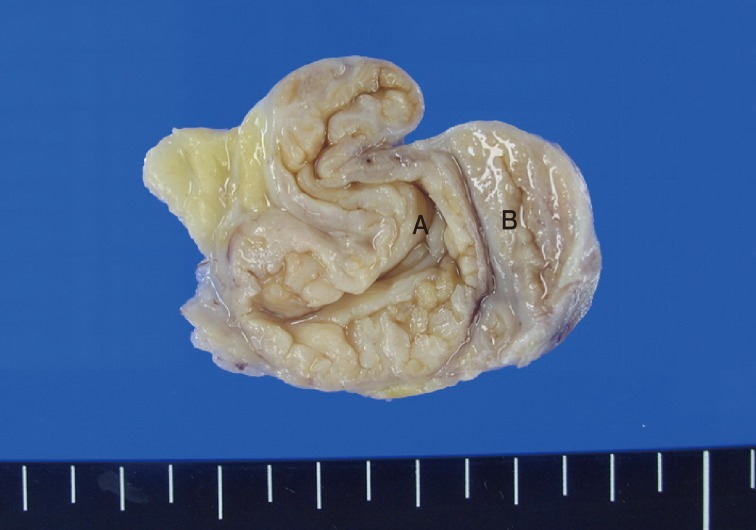
- The resected specimen was 4×3 cm in size and the tumor was well circumscribed. The mass showed numerous papillary nodular protrusions into a central elongated cystic cavity (3.5×2.5 cm). No necrosis or hemorrhage was observed within the mass (Fig. 2). Eccentrically, a whitish gray, solid soft tissue area (1.2 cm in diameter) was present.
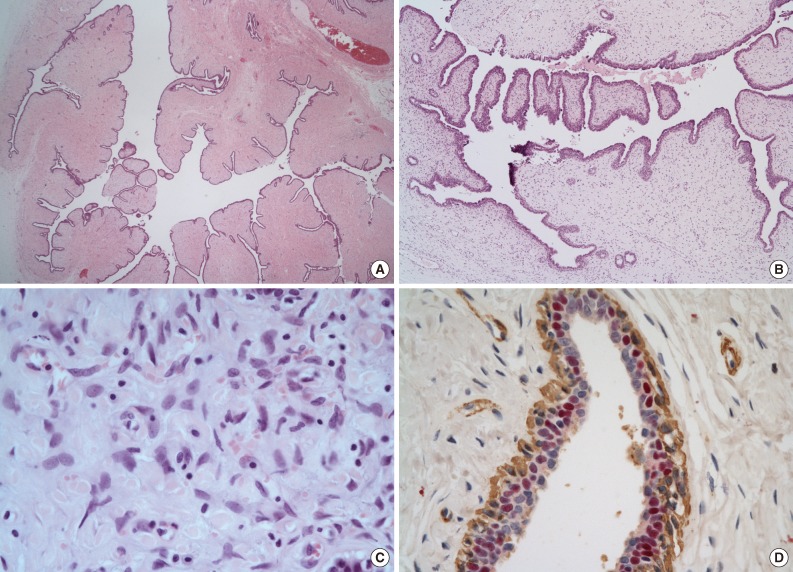
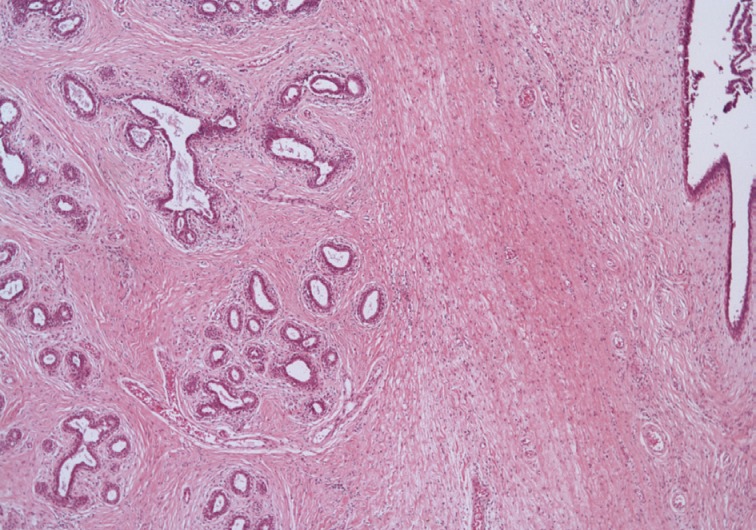
- The microscopic findings showed a central elongated cyst with intraluminal projections (Fig. 3A), which were composed of hyperplastic epithelium-lined stroma (Fig. 3B). Stromal cells had no mitosis, low-to-moderate cellularity, and mild cytologic atypia (Fig. 3C). The lining epithelial cells were strongly positive for estrogen and progesterone receptors (Fig. 3D). In the periphery of the mass, normal breast tissue was identified (Fig. 4). The final pathological results were consistent with a benign PT with prominent intracystic growth, derived from an ectopic breast. Three months after the procedure, the patient was doing well without any recurrence.
CASE REPORT
- Ectopic breast tissue occurs along the milk lines, which extend bilaterally from the mid-axillae through the normal breasts and then inferiorly to the medial groins. The inferior extensions of the milk lines traverse the vulva bilaterally. The milk ridge, the embryological anlage, later undergoes complete atrophy. Ectopic breasts develop from the milk ridges that fail to atrophy.1 Most patients with clinically apparent ectopic breast tissue have unilateral or bilateral axillary involvement. Thoracic, abdominal, inguinal, and vulvar ectopic breasts are much less common,1 and only one case has been presented as an inguinal mass.2
- Ectopic breast tissue can involve benign and malignant pathologic changes similar to those that occur in normally located breast tissue,3 such as fibroadenoma, fibrocystic changes, sarcomatous changes, and carcinoma.8
- Anogenital mammary-like glands are mainly located in the sulcus between the labia minora and majora and are often found in pathologic specimens originating from the anogenital area. Their normal histology varies, but closely imitates breast tissue. Lesions involving anogenital mammary-like glands manifest a striking similarity to their mammary counterparts. Therefore, in this specific location, mammary-like lesions might originate from ectopic breast or anogenital mammary-like glands.4
- PTs in an ectopic location are rare, and they mostly have been reported in the vulva and axilla.3,5 A review of the literature revealed only four cases in the vulva and two cases in the axilla (Table 1).3,9-12 All cases had normal mammary tissues surrounding the mass.
- PTs represent fibroepithelial proliferations of the breast tissue,6 and they are circumscribed biphasic tumors, analogous to fibroadenomas.7 The external surface of a PT is well circumscribed but not encapsulated.1 The bisected tumor is composed of firm, bulging gray to tan tissue.1 The size of the tumors vary from 1 to 40 cm. Due to a rapid increase in the size of the mass, degenerative changes might occur from hemorrhagic fluid flowing into the cystic cavity. Larger tumors may show hemorrhage and necrosis,7 and these alterations are more common in malignant PTs but can occur in large benign lesions as well.1 As a result, they frequently contain gross clefts or cystic cavities.7 Histologically, the tumor is characterized by a double-layered benign epithelial component arranged in clefts and surrounded by an overgrowing hypercellular mesenchymal component typically organized in leaf-like structures. They usually exhibit an enhanced intracanalicular growth pattern with leaf-like projections into the dilated lumina.7 Depending on the bland or sarcomatous characteristics of their mesenchymal (stromal) components, they display a morphological spectrum lying somewhere in between fibroadenomas and pure stromal sarcomas.7
- Although larger lesions are prone to cystic degeneration,6 it is uncommon to observe a PT growing entirely within a cystically dilated ductal structure in the breast; the tumor rarely shows intracystic growth.7 An unusual variant of PT has an exaggerated cystic component resulting in a gross appearance that is difficult to distinguish from a cystic papilloma.1 Only five similar cases have been reported in the literature so far in breasts. Two separate reports of an intracystic growth pattern of PT, both borderline cases, were described in 1998 in Japan,13,14 and an intracystic PT that grew along the lumen of the lactiferous duct and caused nipple discharge was described in a Malay woman.15 Recently, two additional cases of benign intracystic PTs of the breast were reported.6,7 Because an intraductal growth pattern may mimic an intraductal papilloma on ultrasonography, which can lead to microdochectomy rather than the referred wide excision for PT in the breast,15 a preoperative diagnosis is important in these tumors of the breast. However, a PT with a predominant cystic component has not been reported yet in an ectopic breast. To our knowledge, the present lesion was the first reported case of a benign intracystic PT in the inguinal region. It is worthy to recognize this entity to rule out malignant lymphoma, because a clinically inguinal mass raises the possibility of lymph node enlargement.
DISCUSSION
- 1. Rosen PP. Rosen's breast pathology. 3rd ed. Philadelphia: Wolters Kluwer Lippincott Williams & Wilkins, 2009; 28-31.
- 2. Dworak O, Reck T, Greskötter KR, Köckerling F. Hamartoma of an ectopic breast arising in the inguinal region. Histopathology 1994; 24: 169-171. ArticlePubMed
- 3. Oshida K, Miyauchi M, Yamamoto N, et al. Phyllodes tumor arising in ectopic breast tissue of the axilla. Breast Cancer 2003; 10: 82-84. ArticlePubMedPDF
- 4. Kazakov DV, Spagnolo DV, Kacerovska D, Michal M. Lesions of anogenital mammary-like glands: an update. Adv Anat Pathol 2011; 18: 1-28. PubMed
- 5. Mannan AA, Kahvic M, Aziz AH. Phyllodes tumor of the vulva: report of a rare case and review of the literature. Am J Dermatopathol 2010; 32: 384-386. ArticlePubMed
- 6. Santosh KV, Sumana BS. Benign intracystic phyllodes tumor of breast. Indian J Pathol Microbiol 2010; 53: 385-386. ArticlePubMed
- 7. Maimoon SA, Wilkinson AR. Benign cystic papillary phyllodes tumor: an alarming gross appearance. Indian J Pathol Microbiol 2011; 54: 191-192. ArticlePubMed
- 8. Hong JH, Oh MJ, Hur JY, Lee JK. Accessory breast tissue presenting as a vulvar mass in an adolescent girl. Arch Gynecol Obstet 2009; 280: 317-320. ArticlePubMedPDF
- 9. Tbakhi A, Cowan DF, Kumar D, Kyle D. Recurring phyllodes tumor in aberrant breast tissue of the vulva. Am J Surg Pathol 1993; 17: 946-950. ArticlePubMed
- 10. Tresserra F, Grases PJ, Izquierdo M, Cararach M, Fernandez-Cid A. Fibroadenoma phyllodes arising in vulvar supernumerary breast tissue: report of two cases. Int J Gynecol Pathol 1998; 17: 171-173. PubMed
- 11. Chulia MT, Paya A, Niveiro M, Ceballos S, Aranda FI. Phyllodes tumor in ectopic breast tissue of the vulva. Int J Surg Pathol 2001; 9: 81-83. ArticlePubMedPDF
- 12. Saleh HA, Klein LH. Cystosarcoma phyllodes arising synchronously in right breast and bilateral axillary ectopic breast tissue. Arch Pathol Lab Med 1990; 114: 624-626. PubMed
- 13. Horiguchi J, Iino Y, Aiba S, et al. Phyllodes tumor showing intracystic growth: a case report. Jpn J Clin Oncol 1998; 28: 705-708. ArticlePubMed
- 14. Takao S, Sakamoto G, Akiyama F, et al. Phyllodes tumor with features of intracystic tumor. Jpn J Breast Cancer 1998; 13: 813-816.
- 15. Lian D, Cheah E, Tan PH, Thng CH, Tan SM. Phyllodes tumour with intraductal growth: a rare cause of nipple discharge. Histopathology 2007; 50: 666-669. ArticlePubMed
REFERENCES
Fig. 1Positron emission tomography-computed tomography shows a right inguinal mass (27 mm) with low metabolism (arrow).


Fig. 2The tumor is well-circumscribed and showed numerous papillary nodular protrusions into a central cystic cavity (3.5×2.5 cm) (A). Eccentrically, a normal breast tissue (B) is present.


Fig. 3The microscopic findings show a central elongated cyst with intraluminal projections (A), which is composed of hyperplastic epithelium lined stroma (B). Stromal cells have no mitosis, low-to-moderated cellularity, and mild cytoplasm atypia (C). The lining epithelial cells are diffusely and strongly positive for estrogen receptor. Myoepithelial cells are nonreactive (immunohistochemistry for estrogen receptor [red] and smooth muscle actin [brown]) (D).


Table 1.Summary of the reported cases of ectopic phyllodes tumor
| Case No. | Age (yr) | Location | Size (cm) | Histology | Treatment | Reference |
|---|---|---|---|---|---|---|
| 1 | 20 | Labium majora | 3.0 × 4.0 | Benign | Excision | Tbakhi et al. [9] |
| 2 | 39 | Labium majus | 1.4 × 2.0 | Benign | Excision | Tresserra et al. [10] |
| 3 | 17 | Labium majus and minus | 1.6 × 1.2 | Benign | Excision | Tresserra et al. [10] |
| 4 | 34 | Labium majus | 6.0 × 4.0 | Benign | Excision | Chulia et al. [11] |
| 5 | 31 | Axilla | 1.7 × 1.6 | Benign | Excision | Oshida et al. [3] |
| 6 | 45 | Axilla | 3 × 2 | Low-grade | Wide excision | Saleh and Klein [12] |
Figure & Data
References
Citations
Citations to this article as recorded by 

- BENIGN PHYLLODES TUMOR ARISING IN AXILLARY BREAST TISSUE; A RARE CASE WITH AVAILABLE REVIEW OF LITERATURE
MONICA DASH, PRAGNYA PARAMITA MISHRA, PREMANAND PANDA
International Journal of Current Pharmaceutical Research.2025; : 124. CrossRef - Benign phyllodes tumor arising from accessory breast tissue of the axilla: An inquisitive rarity
Sonam Sharma
Saudi Surgical Journal.2024; 12(1): 54. CrossRef - Computed tomography and magnetic resonance imaging in diagnosis of metastatic pleural lesion with pleural effusion in patients with breast carcinoma
P. M. Kotlyarov, I. D. Lagkueva, N. I. Sergeev
Russian Pulmonology.2019; 29(1): 112. CrossRef - Mama ectópica en la región inguinal
V.Y. Presas, L.M. Mastronardi, S. Saucedo, E. Rojas Bilbao
Clínica e Investigación en Ginecología y Obstetricia.2017; 44(2): 89. CrossRef
 PubReader
PubReader ePub Link
ePub Link-
 Cite this Article
Cite this Article
- Cite this Article
-
- Close
- Download Citation
- Close
- Figure
Cystic Benign Phyllodes Tumor in the Inguinal Region




Fig. 1 Positron emission tomography-computed tomography shows a right inguinal mass (27 mm) with low metabolism (arrow).
Fig. 2 The tumor is well-circumscribed and showed numerous papillary nodular protrusions into a central cystic cavity (3.5×2.5 cm) (A). Eccentrically, a normal breast tissue (B) is present.
Fig. 3 The microscopic findings show a central elongated cyst with intraluminal projections (A), which is composed of hyperplastic epithelium lined stroma (B). Stromal cells have no mitosis, low-to-moderated cellularity, and mild cytoplasm atypia (C). The lining epithelial cells are diffusely and strongly positive for estrogen receptor. Myoepithelial cells are nonreactive (immunohistochemistry for estrogen receptor [red] and smooth muscle actin [brown]) (D).
Fig. 4 In the periphery of the mass, normal breast tissue is identified.
Fig. 1
Fig. 2
Fig. 3
Fig. 4
Cystic Benign Phyllodes Tumor in the Inguinal Region
| Case No. | Age (yr) | Location | Size (cm) | Histology | Treatment | Reference |
|---|---|---|---|---|---|---|
| 1 | 20 | Labium majora | 3.0 × 4.0 | Benign | Excision | Tbakhi et al. [9] |
| 2 | 39 | Labium majus | 1.4 × 2.0 | Benign | Excision | Tresserra et al. [10] |
| 3 | 17 | Labium majus and minus | 1.6 × 1.2 | Benign | Excision | Tresserra et al. [10] |
| 4 | 34 | Labium majus | 6.0 × 4.0 | Benign | Excision | Chulia et al. [11] |
| 5 | 31 | Axilla | 1.7 × 1.6 | Benign | Excision | Oshida et al. [3] |
| 6 | 45 | Axilla | 3 × 2 | Low-grade | Wide excision | Saleh and Klein [12] |
Table 1. Summary of the reported cases of ectopic phyllodes tumor

 E-submission
E-submission