Search
- Page Path
- HOME > Search
- Progastrin, annexin A2, and tumor-associated macrophages in gastric adenocarcinoma
- Konstantinos Christofidis, Rodanthi Fioretzaki, Stylianos Mavropoulos Papoudas, Nikolaos Charalampakis, Nikolaos Kavantzas, Dimitrios Schizas, Stratigoula Sakellariou
- J Pathol Transl Med. 2026;60(2):263-279. Published online March 10, 2026
- DOI: https://doi.org/10.4132/jptm.2025.12.20
- 1,544 View
- 144 Download
-
 Abstract
Abstract
 PDF
PDF Supplementary Material
Supplementary Material - Background
Gastric adenocarcinoma is a major cause of cancer mortality worldwide, and reliable biomarkers remain insufficient. This study investigates the immunohistochemical expression of progastrin (hPG) and annexin A2 (ANXA2) and the polarization of tumor-associated macrophages in gastric adenocarcinoma to explore their potential prognostic and biological significance. Methods: A retrospective analysis was conducted on formalin-fixed, paraffin-embedded tissue samples from 60 patients with gastric adenocarcinoma (primary tumors, lymph node metastases, and non-tumoral gastric mucosa) and gastric biopsies from 23 healthy controls. The expression of hPG and ANXA2 was quantified using the H-score, and the CD163/human leukocyte antigen–DR (HLA-DR) ratio was used to represent macrophage polarization (M2/M1). Statistical analyses included non-parametric tests, Spearman correlations, Kaplan-Meier survival curves, and Cox proportional-hazards models. Results: ANXA2 expression was significantly elevated in cancer cells from primary tumors and lymph node metastases, compared with the non-tumoral gastric mucosa tissues and gastric mucosa tissues from healthy controls. ANXA2 expression increased with the tumor grade. High ANXA2 levels were associated with shorter overall and disease-free survival, but they did not have independent prognostic value. Although hPG expression correlated positively with ANXA2, it showed no significant prognostic association. The CD163/HLA-DR ratio increased with tumor progression and negatively correlated with ANXA2, but it did not influence survival outcomes. Conclusions: This study is the first to demonstrate the adverse prognostic impact of ANXA2 overexpression in gastric adenocarcinoma tissues from Caucasian patients. Our results suggest that ANXA2 might have utility as a prognostic biomarker and therapeutic target, if further large-scale studies validate and expand our findings.
- Association study of TYMS gene expression with TYMS and ENOSF1 genetic variants in neoadjuvant chemotherapy response of gastric cancer
- Khadijeh Arjmandi, Iman Salahshourifar, Shiva Irani, Fereshteh Ameli, Mohsen Esfandbod
- J Pathol Transl Med. 2025;59(2):105-114. Published online February 25, 2025
- DOI: https://doi.org/10.4132/jptm.2024.11.05
- 4,612 View
- 150 Download
- 3 Web of Science
- 3 Crossref
-
 Abstract
Abstract
 PDF
PDF - Background
The present research was designed to study the associations between genetic variants of TYMS and ENOSF1 genes with TYMS and ENOSF1 gene expression in neoadjuvant chemotherapy response among patients with gastric cancer. Methods: Formalin-embedded and paraffin-fixed matched tumor and normal gastric cancer tissue samples from patients who received neoadjuvant 5-fluorouracil (5-FU) treatment were obtained. DNA and RNA were extracted for all samples. A 28-bp variable number tandem repeat (VNTR) at the 5' untranslated region of TYMS gene and rs2612091 and rs2741171 variants in the ENOSF1 gene were genotyped for normal tissue samples. The real-time polymerase chain reaction method was used to study the expression of ENOSF1 and TYMS genes in both normal and tumor tissues. Data were analyzed using REST 2000 and SPSS ver. 26.0 software programs. Results: A significant association between TYMS 2R3R VNTR genotypes and 5-FU therapy was found (p = .032). The 3R3R and 2R2R genotypes were significantly associated with increased and decreased survival time, respectively (p = .003). The 3R3R genotype was significantly associated with TYMS overexpression (p < .001). Moreover, a significant association was found between the rs2612091 genotype and treatment outcome (p = .017). Conclusions: This study highlights the impact of TYMS and ENOSF1 genes as predictive indicators for survival and response to 5-FU–based neoadjuvant chemotherapy in gastric cancer patients. -
Citations
Citations to this article as recorded by- Innovative biomaterial strategies for mitigating radiotherapy toxicity: multidimensional mechanistic interventions of nano-microscale materials and hydrogels
Yifan Liu, Fengdi Jiang, Jie Song, Huaijin Qiao, Junlong Dai, Hao Bai, Shuyu Zhang
Coordination Chemistry Reviews.2026; 549: 217313. CrossRef - Mebendazole impairs the expression and function of enzymes in nucleotide metabolism pathways, leading to Selective Cytotoxicity, Cell Cycle Arrest, and Damage to Cell Morphology in Gastric Cancer
Emerson Lucena da Silva, Felipe Pantoja Mesquita, Pedro Victor da Rocha Lima, José Hélio de Araújo Filho, Francisco Laio de Oliveira, Ana Beatriz da Lima, Pedro Filho Noronha Souza, Raquel Carvalho Montenegro
Chemico-Biological Interactions.2026; 430: 111973. CrossRef - Multi‐Omics Analysis of TYMS as a Prognostic Biomarker and Therapeutic Target for Lung Adenocarcinoma
Hansen Shi, Peijun Zhang, Jiayi Chen, Hua Li, Xiaohuai Zhang, Biyun zeng, Tiancai Liu, Tao Zeng
Cancer Medicine.2026;[Epub] CrossRef
- Innovative biomaterial strategies for mitigating radiotherapy toxicity: multidimensional mechanistic interventions of nano-microscale materials and hydrogels
- A standardized pathology report for gastric cancer: 2nd edition
- Young Soo Park, Myeong-Cherl Kook, Baek-hui Kim, Hye Seung Lee, Dong-Wook Kang, Mi-Jin Gu, Ok Ran Shin, Younghee Choi, Wonae Lee, Hyunki Kim, In Hye Song, Kyoung-Mee Kim, Hee Sung Kim, Guhyun Kang, Do Youn Park, So-Young Jin, Joon Mee Kim, Yoon Jung Choi, Hee Kyung Chang, Soomin Ahn, Mee Soo Chang, Song-Hee Han, Yoonjin Kwak, An Na Seo, Sung Hak Lee, Mee-Yon Cho
- J Pathol Transl Med. 2023;57(1):1-27. Published online January 15, 2023
- DOI: https://doi.org/10.4132/jptm.2022.12.23
- 42,247 View
- 1,615 Download
- 26 Web of Science
- 23 Crossref
-
 Abstract
Abstract
 PDF
PDF Supplementary Material
Supplementary Material - The first edition of ‘A Standardized Pathology Report for Gastric Cancer’ was initiated by the Gastrointestinal Pathology Study Group of the Korean Society of Pathologists and published 17 years ago. Since then, significant advances have been made in the pathologic diagnosis, molecular genetics, and management of gastric cancer (GC). To reflect those changes, a committee for publishing a second edition of the report was formed within the Gastrointestinal Pathology Study Group of the Korean Society of Pathologists. This second edition consists of two parts: standard data elements and conditional data elements. The standard data elements contain the basic pathologic findings and items necessary to predict the prognosis of GC patients, and they are adequate for routine surgical pathology service. Other diagnostic and prognostic factors relevant to adjuvant therapy, including molecular biomarkers, are classified as conditional data elements to allow each pathologist to selectively choose items appropriate to the environment in their institution. We trust that the standardized pathology report will be helpful for GC diagnosis and facilitate large-scale multidisciplinary collaborative studies.
-
Citations
Citations to this article as recorded by- GAST-NET: A multi-modal and multi-task deep learning framework for preoperative prediction of perineural invasion and prognostic risk in gastric cancer
Shidi Miao, Hexiang Dong, Jinyang Feng, Yuyang Jiang, Mengzhuo Sun, Zengyao Liu, Qiujun Wang, Xuemei Ding, Ruitao Wang
International Journal of Medical Informatics.2026; 212: 106348. CrossRef - Poorly cohesive carcinoma diffusely involving the whole gastrointestinal tract: a case report
Wei Gao, Yusheng Yang, Xinyi Hu, Yujuan Shi, Kai Liu, Minmin Gu, Jing Wang
Discover Oncology.2026;[Epub] CrossRef - Comprehensive Overview of Gastric Cancer Immunohistochemistry: Key Biomarkers, Advanced Detection Methods, and Perspectives
Bogdan Oprea
Medicina.2026; 62(4): 683. CrossRef - Tumor Budding in Gastric Carcinoma: Beyond Counting Cells at the Invasive Front—A Review of Current Evidence and Biological Perspectives
Catalin-Bogdan Satala, Gabriela Gurau, Alina-Mihaela Gurau, Gabriela Patrichi, Daniela Mihalache
International Journal of Molecular Sciences.2026; 27(9): 3787. CrossRef - Spatial and Temporal Tumor Heterogeneity in Gastric Cancer: Discordance of Predictive Biomarkers
Hye Seung Lee
Journal of Gastric Cancer.2025; 25(1): 192. CrossRef - PD-L1 as a Biomarker in Gastric Cancer Immunotherapy
Yunjoo Cho, Soomin Ahn, Kyoung-Mee Kim
Journal of Gastric Cancer.2025; 25(1): 177. CrossRef - Korean Gastric Cancer Association-Led Nationwide Survey on Surgically Treated Gastric Cancers in 2023
Dong Jin Kim, Jeong Ho Song, Ji-Hyeon Park, Sojung Kim, Sin Hye Park, Cheol Min Shin, Yoonjin Kwak, Kyunghye Bang, Chung-sik Gong, Sung Eun Oh, Yoo Min Kim, Young Suk Park, Jeesun Kim, Ji Eun Jung, Mi Ran Jung, Bang Wool Eom, Ki Bum Park, Jae Hun Chung, S
Journal of Gastric Cancer.2025; 25(1): 115. CrossRef - A Comprehensive and Comparative Review of Global Gastric Cancer Treatment Guidelines: 2024 Update
Sang Soo Eom, Keun Won Ryu, Hye Sook Han, Seong-Ho Kong
Journal of Gastric Cancer.2025; 25(1): 153. CrossRef - Korea, Japan, Europe, and the United States: Why are guidelines for gastric cancer different?
Emily E. Stroobant, Seong-Ho Kong, Maria Bencivenga, Takahiro Kinoshita, Tae-Han Kim, Takeshi Sano, Giovanni de Manzoni, Han-Kwang Yang, Yuko Kitagawa, Vivian E. Strong
Gastric Cancer.2025; 28(4): 559. CrossRef - Can the Japanese guidelines for endoscopic submucosal dissection be safely applied to Korean gastric cancer patients? A multicenter retrospective study based on the Korean Gastric Cancer Association nationwide survey
Hayemin Lee, Mi Ryeong Park, Junhyun Lee
Annals of Surgical Treatment and Research.2025; 109(2): 81. CrossRef - Double optimal transport for differential gene regulatory network inference with unpaired samples
Mengyu Li, Bencong Zhu, Cheng Meng, Xiaodan Fan, Laura Cantini
Bioinformatics.2025;[Epub] CrossRef - A Randomized Controlled Trial to Evaluate the Effect of Fibrin Glue on Bleeding after Gastric Endoscopic Submucosal Dissection
Tae-Se Kim, Tae-Jun Kim, Yang Won Min, Hyuk Lee, Byung-Hoon Min, Jun Haeng Lee, Poong-Lyul Rhee, Jae J. Kim
Gut and Liver.2025; 19(5): 677. CrossRef - Diagnostic accuracy of stereomicroscopy assessment of invasion depth in ex vivo specimens of early gastric cancer
Jing Wang, Lin Chang, Dong-Feng Niu, Yan Yan, Chang-Qi Cao, Shi-Jie Li, Qi Wu
World Journal of Gastroenterology.2025;[Epub] CrossRef - SMMILe enables accurate spatial quantification in digital pathology using multiple-instance learning
Zeyu Gao, Anyu Mao, Yuxing Dong, Hannah Clayton, Jialun Wu, Jiashuai Liu, ChunBao Wang, Kai He, Tieliang Gong, Chen Li, Mireia Crispin-Ortuzar
Nature Cancer.2025; 6(12): 2025. CrossRef - Genomic and Transcriptomic Characterization of Gastric Cancer with Bone Metastasis
Sujin Oh, Soo Kyung Nam, Keun-Wook Lee, Hye Seung Lee, Yujun Park, Yoonjin Kwak, Kyu Sang Lee, Ji-Won Kim, Jin Won Kim, Minsu Kang, Young Suk Park, Sang-Hoon Ahn, Yun-Suhk Suh, Do Joong Park, Hyung Ho Kim
Cancer Research and Treatment.2024; 56(1): 219. CrossRef - Microscopic tumor mapping of post-neoadjuvant therapy pancreatic cancer specimens to predict post-surgical recurrence: A prospective cohort study
Yeshong Park, Yeon Bi Han, Jinju Kim, MeeYoung Kang, Boram Lee, Eun Sung Ahn, Saemi Han, Haeryoung Kim, Hee-Young Na, Ho-Seong Han, Yoo-Seok Yoon
Pancreatology.2024; 24(4): 562. CrossRef - Effect of Neoadjuvant Chemotherapy on Tumor-Infiltrating Lymphocytes in Resectable Gastric Cancer: Analysis from a Western Academic Center
Elliott J. Yee, Danielle Gilbert, Jeffrey Kaplan, Sachin Wani, Sunnie S. Kim, Martin D. McCarter, Camille L. Stewart
Cancers.2024; 16(7): 1428. CrossRef - Interpretation of PD-L1 expression in gastric cancer: summary of a consensus meeting of Korean gastrointestinal pathologists
Soomin Ahn, Yoonjin Kwak, Gui Young Kwon, Kyoung-Mee Kim, Moonsik Kim, Hyunki Kim, Young Soo Park, Hyeon Jeong Oh, Kyoungyul Lee, Sung Hak Lee, Hye Seung Lee
Journal of Pathology and Translational Medicine.2024; 58(3): 103. CrossRef - Expression of claudin 18.2 in poorly cohesive carcinoma and its association with clinicopathologic parameters in East Asian patients
Moonsik Kim, Byung Woog Kang, Jihyun Park, Jin Ho Baek, Jong Gwang Kim
Pathology - Research and Practice.2024; 263: 155628. CrossRef - Clinicopathological analysis of claudin 18.2 focusing on intratumoral heterogeneity and survival in patients with metastatic or unresectable gastric cancer
T.-Y. Kim, Y. Kwak, S.K. Nam, D. Han, D.-Y. Oh, S.-A. Im, H.S. Lee
ESMO Open.2024; 9(12): 104000. CrossRef - Pathological Interpretation of Gastric Tumors in Endoscopic Submucosal Dissection
Jung Yeon Kim
Journal of Digestive Cancer Research.2023; 11(1): 15. CrossRef - Histopathology of Gastric Cancer
Baek-hui Kim, Sung Hak Lee
The Korean Journal of Helicobacter and Upper Gastrointestinal Research.2023; 23(2): 143. CrossRef - Endoscopic submucosal dissection hands-on training with artificial mucosal layer EndoGEL
Tae-Se Kim, Jun Haeng Lee
Journal of Innovative Medical Technology.2023; 1(1): 5. CrossRef
- GAST-NET: A multi-modal and multi-task deep learning framework for preoperative prediction of perineural invasion and prognostic risk in gastric cancer
- Extremely well-differentiated adenocarcinoma of the stomach: diagnostic pitfalls in endoscopic biopsy
- Jongwon Lee, In-Seob Lee, Ji Yong Ahn, Young Soo Park, Jihun Kim
- J Pathol Transl Med. 2022;56(2):63-72. Published online November 16, 2021
- DOI: https://doi.org/10.4132/jptm.2021.10.12
- 9,594 View
- 474 Download
- 4 Web of Science
- 5 Crossref
-
 Abstract
Abstract
 PDF
PDF Supplementary Material
Supplementary Material - Background
Extremely well-differentiated adenocarcinoma (EWDA) is a deceptively bland-looking adenocarcinoma of the stomach. It often causes diagnostic problems, especially in endoscopic biopsy samples. To better recognize this deceptively bland lesion, we carefully reviewed a series of EWDAs treated at our institution.
Methods
A total of 55 specimens from 19 patients were obtained. Endoscopic, gross and microscopic features defining EWDA were described and documented. For comparison, hyperplastic polyp specimens were randomly selected and analyzed.
Results
Most cases (18 of 19, 94.7%) were advanced gastric cancer (AGC) and primarily located in the body of the stomach (15 of 19, 79.0%). The majority of AGCs were non-ulcerated (11 of 18, 61.1%) with an undermining growth pattern and a relatively small mucosal involvement. Specific histologic features included an irregular glandular shape, an undulating apical cytoplasmic border, disproportionately large glands, a variably distended mucinous cytoplasm. Classical features, such as small infiltrating glands or desmoplastic reactions, were barely observed. Identification of irregularly spaced nuclei and disruption of the foveolar epithelial structure, along with atypical features described above were helpful in making a diagnosis especially in gastric forceps biopsies.
Conclusions
Awareness of the histomorphologic characteristics described in this report would lead to timely diagnosis and prevent repeated endoscopic procedures. -
Citations
Citations to this article as recorded by- Crawling-type Gastric Adenocarcinoma: Clinicopathology, Diagnosis, and Endoscopic Treatment Implications
Cong Bang Huynh, Jin Won Chang
Journal of Digestive Cancer Research.2026; 14(1): 1. CrossRef - Artificial intelligence-assisted diagnosis of early gastric cancer: present practice and future prospects
Changda Lei, Wenqiang Sun, Kun Wang, Ruixia Weng, Xiuji Kan, Rui Li
Annals of Medicine.2025;[Epub] CrossRef - Unusual or Uncommon Histology of Gastric Cancer
Jinho Shin, Young Soo Park
Journal of Gastric Cancer.2024; 24(1): 69. CrossRef - A case of gastric adenocarcinoma with pyloric gland-type infiltrating submucosa
Kaiho Hirata, Shusuke Yagi, Hideki Miyazaki, Kazuhiko Yamada, Naoki Akazawa, Naoki Enomoto, Kyoko Nohara, Chizu Yokoi, Toru Igari, Norihiro Kokudo
Surgical Case Reports.2024;[Epub] CrossRef - Gastric-type extremely well-differentiated adenocarcinoma of the stomach: A rare tumor with diagnostic difficulties and high inter-observer variation in endoscopic pinch biopsies
Soomin Ahn, Sujin Park, Hyun Hee Koh, Han Gyeol Kim, Hyunjin Kim, Jae Yeong Son, Boram Lee, Hyunwoo Lee, Soohyun Hwang, Junhun Cho, Yun Kyung Lee, Ryoji Kushima, Amitabh Srivastava, Kyoung-Mee Kim
Pathology - Research and Practice.2024; 263: 155599. CrossRef
- Crawling-type Gastric Adenocarcinoma: Clinicopathology, Diagnosis, and Endoscopic Treatment Implications
- Deep learning for computer-assisted diagnosis of hereditary diffuse gastric cancer
- Sean A. Rasmussen, Thomas Arnason, Weei-Yuarn Huang
- J Pathol Transl Med. 2021;55(2):118-124. Published online January 22, 2021
- DOI: https://doi.org/10.4132/jptm.2020.12.22
- 6,062 View
- 136 Download
- 9 Web of Science
- 9 Crossref
-
 Abstract
Abstract
 PDF
PDF - Background
Patients with hereditary diffuse gastric cancer often undergo prophylactic gastrectomy to minimize cancer risk. Because intramucosal poorly cohesive carcinomas in this setting are typically not grossly visible, many pathologists assess the entire gastrectomy specimen microscopically. With 150 or more slides per case, this is a major time burden for pathologists. This study utilizes deep learning methods to analyze digitized slides and detect regions of carcinoma.
Methods
Prophylactic gastrectomy specimens from seven patients with germline CDH1 mutations were analyzed (five for training/validation and two for testing, with a total of 133 tumor foci). All hematoxylin and eosin slides containing cancer foci were digitally scanned, and patches of size 256×256 pixels were randomly extracted from regions of cancer as well as from regions of normal background tissue, resulting in 15,851 images for training/validation and 970 images for testing. A model with DenseNet-169 architecture was trained for 150 epochs, then evaluated on images from the test set. External validation was conducted on 814 images scanned at an outside institution.
Results
On individual patches, the trained model achieved a receiver operating characteristic (ROC) area under the curve (AUC) of 0.9986. This enabled it to maintain a sensitivity of 90% with a false-positive rate of less than 0.1%. On the external validation dataset, the model achieved a similar ROC AUC of 0.9984. On whole slide images, the network detected 100% of tumor foci and correctly eliminated an average of 99.9% of the non-cancer slide area from consideration.
Conclusions
Overall, our model shows encouraging progress towards computer-assisted diagnosis of hereditary diffuse gastric cancer. -
Citations
Citations to this article as recorded by- Now and future of artificial intelligence-based signet ring cell diagnosis: A survey
Zhu Meng, Junhao Dong, Limei Guo, Fei Su, Jiaxuan Liu, Guangxi Wang, Zhicheng Zhao
Expert Systems with Applications.2026; 296: 129188. CrossRef - Development and application of deep learning-based diagnostics for pathologic diagnosis of gastric endoscopic submucosal dissection specimens
Soomin Ahn, Yiyu Hong, Sujin Park, Yunjoo Cho, Inwoo Hwang, Ji Min Na, Hyuk Lee, Byung-Hoon Min, Jun Haeng Lee, Jae J. Kim, Kyoung-Mee Kim
Gastric Cancer.2025; 28(4): 609. CrossRef - A Comprehensive Literature Review of the CDH1 Mutation and Its Role in Gastric Cancer
Malik Samardali, Jehad Samardaly, Ibrahim Shanti
Cureus.2025;[Epub] CrossRef - Deep learning for multiclass tumor cell detection in histopathology slides of hereditary diffuse gastric cancer
Robin Lomans, Valentina Angerilli, Joey Spronck, Liudmila L. Kodach, Irene Gullo, Fátima Carneiro, Rachel S. van der Post, Francesco Ciompi
iScience.2025; 28(8): 113064. CrossRef - Non-endoscopic Applications of Machine Learning in Gastric Cancer: A Systematic Review
Marianne Linley L. Sy-Janairo, Jose Isagani B. Janairo
Journal of Gastrointestinal Cancer.2024; 55(1): 47. CrossRef - Artificial intelligence applicated in gastric cancer: A bibliometric and visual analysis via CiteSpace
Guoyang Zhang, Jingjing Song, Zongfeng Feng, Wentao Zhao, Pan Huang, Li Liu, Yang Zhang, Xufeng Su, Yukang Wu, Yi Cao, Zhengrong Li, Zhigang Jie
Frontiers in Oncology.2023;[Epub] CrossRef - Deep learning of endoscopic features for the assessment of neoadjuvant therapy response in locally advanced rectal cancer
Anqi Wang, Jieli Zhou, Gang Wang, Beibei Zhang, Hongyi Xin, Haiyang Zhou
Asian Journal of Surgery.2023; 46(9): 3568. CrossRef - Preparing Data for Artificial Intelligence in Pathology with Clinical-Grade Performance
Yuanqing Yang, Kai Sun, Yanhua Gao, Kuansong Wang, Gang Yu
Diagnostics.2023; 13(19): 3115. CrossRef - Using Deep Learning to Predict Final HER2 Status in Invasive Breast Cancers That are Equivocal (2+) by Immunohistochemistry
Sean A. Rasmussen, Valerie J. Taylor, Alexi P. Surette, Penny J. Barnes, Gillian C. Bethune
Applied Immunohistochemistry & Molecular Morphology.2022; 30(10): 668. CrossRef
- Now and future of artificial intelligence-based signet ring cell diagnosis: A survey
- Tumor immune response and immunotherapy in gastric cancer
- Yoonjin Kwak, An Na Seo, Hee Eun Lee, Hye Seung Lee
- J Pathol Transl Med. 2020;54(1):20-33. Published online November 1, 2019
- DOI: https://doi.org/10.4132/jptm.2019.10.08
- 20,442 View
- 749 Download
- 74 Web of Science
- 68 Crossref
-
 Abstract
Abstract
 PDF
PDF - Remarkable developments in immuno-oncology have changed the landscape of gastric cancer (GC) treatment. Because immunotherapy intervenes with tumor immune response rather than directly targeting tumor cells, it is important to develop a greater understanding of tumor immunity. This review paper summarizes the tumor immune reaction and immune escape mechanisms while focusing on the role of T cells and their co-inhibitory signals, such as the immune checkpoint molecules programmed death-1 and programmed deathligand 1 (PD-L1). This paper also describes past clinical trials of immunotherapy for patients with GC and details their clinical implications. Strong predictive markers are essential to improve response to immunotherapy. Microsatellite instability, Epstein-Barr virus, PD-L1 expression, and tumor mutational burden are now regarded as potent predictive markers for immunotherapy in patients with GC. Novel immunotherapy and combination therapy targeting new immune checkpoint molecules such as lymphocyte-activation gene 3, T cell immunoglobulin, and mucin domain containing-3, and indoleamine 2,3-dioxygenase have been suggested, and trials are ongoing to evaluate their safety and efficacy. Immunotherapy is an important treatment option for patients with GC and has great potential for improving patient outcome, and further research in immuno-oncology should be carried out.
-
Citations
Citations to this article as recorded by- Using glucocorticoid receptor-related genes to create and validate a survival model predicting gastric cancer
Ke Guo, Ping Huang, Jiasheng Zhang, Bo Zhang, Linyang Li, Jiaxin Li
Computational Biology and Chemistry.2026; 120: 108726. CrossRef - Mechanisms of Metabolic Reprogramming Regulating Immunosuppression in the Gastric Cancer Tumor Microenvironment
Wenting Dong, Xuepeng Qian, Honglin Liu, Jinhai Huo, Weiming Wang
Biomolecules.2026; 16(1): 160. CrossRef - Perineural invasion in digestive tract tumors: Immune system interactions and therapeutic strategies
Jia-Yu Pang, Rong-Yao Jin, Han-Xiao Zhang, Ying-Hua Zhang, Xin-Yue Wei, Wen-Bo Cao, Yi-Xiao Chen, Jun-Ling Wang, Sai-Jun Mo
World Journal of Clinical Oncology.2026;[Epub] CrossRef - FOXP3+/CD8+ ratio associated with aggressive behavior in RUNX3‐methylated diffuse esophagogastric junction tumor
Suguru Maruyama, Yu Imamura, Tasuku Toihata, Ikumi Haraguchi, Manabu Takamatsu, Makiko Yamashita, Yuichiro Nakashima, Eiji Oki, Kenichi Taguchi, Manabu Yamamoto, Shinji Mine, Akihiko Okamura, Jun Kanamori, Souya Nunobe, Takeshi Sano, Shigehisa Kitano, Tet
Cancer Science.2025; 116(1): 178. CrossRef - Immune biomarkers and predictive signatures in gastric cancer: Optimizing immunotherapy responses
Sundaram Vickram, Shofia Saghya Infant, S. Manikandan, D. Jenila Rani, C.M. Mathan Muthu, Hitesh Chopra
Pathology - Research and Practice.2025; 265: 155743. CrossRef - Korean Practice Guidelines for Gastric Cancer 2024: An Evidence-based, Multidisciplinary Approach (Update of 2022 Guideline)
In-Ho Kim, Seung Joo Kang, Wonyoung Choi, An Na Seo, Bang Wool Eom, Beodeul Kang, Bum Jun Kim, Byung-Hoon Min, Chung Hyun Tae, Chang In Choi, Choong-kun Lee, Ho Jung An, Hwa Kyung Byun, Hyeon-Su Im, Hyung-Don Kim, Jang Ho Cho, Kyoungjune Pak, Jae-Joon Kim
Journal of Gastric Cancer.2025; 25(1): 5. CrossRef - Targeted therapy and immunotherapy for gastric cancer: rational strategies, novel advancements, challenges, and future perspectives
Dong Luo, Yunmei Liu, Zhengmao Lu, Lei Huang
Molecular Medicine.2025;[Epub] CrossRef - Prognostic value of the triglyceride-glucose index in gastric cancer
Tugce Eskazan, Suat Saribas, Bekir Kocazeybek
World Journal of Gastroenterology.2025;[Epub] CrossRef - Deciphering the dual role of autophagy in gastric cancer and gastroesophageal junction cancer: from tumor suppression to cancer progression
Lili Lei, Junling Zhang, Ran Wei, Bingqi Dong, Xin Wang, Ying Zhou
Discover Oncology.2025;[Epub] CrossRef - Comparison of clinicopathological parameters with the presence of Epstein–Barr virus and the absence of DNA mismatch repair proteins in gastric adenocarcinomas
Özge Eyeoğlu, Serra Kayaçetin
Asian Biomedicine.2025; 19(2): 86. CrossRef - The impact of combined immunotherapy on the cellular composition of the tumor microenvironment in patients with gastric carcinoma
L.A. Tashireva, A.Yu. Kalinchuk, D.M. Loos, E.A. Tsarenkova, A.V. Avgustinovich, S.G. Afanas’ev, S.V. Vtorushin
Russian Journal of Archive of Pathology.2025; 87(4): 24. CrossRef - Immunotherapy in gastric adenocarcinoma – a rapidly evolving treatment landscape
Yang Wang, Geoffrey Chong
Critical Reviews in Oncology/Hematology.2025; 216: 104941. CrossRef - Ubiquilin-4 induces immune escape in gastric cancer by activating the notch signaling pathway
Quan Jiang, Hao Chen, Shixin Zhou, Tao Zhu, Wenshuai Liu, Hao Wu, Yong Zhang, Fenglin Liu, Yihong Sun
Cellular Oncology.2024; 47(1): 303. CrossRef - Expression and prognostic value of APOBEC2 in gastric adenocarcinoma and its association with tumor-infiltrating immune cells
Lipan Wei, Xiuqian Wu, Lan Wang, Ling Chen, Xuejun Wu, Tiantian Song, Yuanyuan Wang, Wenjun Chang, Aizhen Guo, Yongdong Niu, Haihua Huang
BMC Cancer.2024;[Epub] CrossRef - Identification and characterization of CLEC11A and its derived immune signature in gastric cancer
Qing Zheng, Zhenqi Gong, Baizhi Li, Runzi Cheng, Weican Luo, Cong Huang, Huaiming Wang
Frontiers in Immunology.2024;[Epub] CrossRef - Cervical cancer subtype identification and model building based on lipid metabolism and post-infection microenvironment immune landscape
Yongzhi Chen, Rongjie Cui, Dun Xiong, Yuan Zhao, Jianyu Pang, Samina Gul, Qi Qi, Yuheng Tang, Xuhong Zhou, Wenru Tang
Heliyon.2024; 10(9): e30746. CrossRef - Systematic Analysis of Tumor Stem Cell-related Gene Characteristics
to Predict the PD-L1 Immunotherapy and Prognosis of Gastric
Cancer
Chenchen Wang, Ying Chen, Ru Zhou, Ya’nan Yang, Yantian Fang
Current Medicinal Chemistry.2024; 31(17): 2467. CrossRef - Comprehensive landscape of m6A regulator-related gene patterns and tumor microenvironment infiltration characterization in gastric cancer
Bin Peng, Yinglin Lin, Gao Yi, Mingzhen Lin, Yao Xiao, Yezhenghong Qiu, Wenxia Yao, Xinke Zhou, Zhaoyu Liu
Scientific Reports.2024;[Epub] CrossRef - Distinctive Phenotypic and Microenvironmental Characteristics of Neuroendocrine Carcinoma and Adenocarcinoma Components in Gastric Mixed Adenoneuroendocrine Carcinoma
Yoonjin Kwak, Soo Kyung Nam, Yujun Park, Yun-Suhk Suh, Sang-Hoon Ahn, Seong-Ho Kong, Do Joong Park, Hyuk-Joon Lee, Hyung-Ho Kim, Han-Kwang Yang, Hye Seung Lee
Modern Pathology.2024; 37(10): 100568. CrossRef - Computed tomography-detected extramural venous invasion-related gene signature: a potential negative biomarker of immune checkpoint inhibitor treatment in patients with gastric cancer
Hao Yang, Xinyi Gou, Caizhen Feng, Yinli Zhang, Fan Chai, Nan Hong, Yingjiang Ye, Yi Wang, Bo Gao, Jin Cheng
Journal of Translational Medicine.2023;[Epub] CrossRef - A standardized pathology report for gastric cancer: 2nd edition
Young Soo Park, Myeong-Cherl Kook, Baek-hui Kim, Hye Seung Lee, Dong-Wook Kang, Mi-Jin Gu, Ok Ran Shin, Younghee Choi, Wonae Lee, Hyunki Kim, In Hye Song, Kyoung-Mee Kim, Hee Sung Kim, Guhyun Kang, Do Youn Park, So-Young Jin, Joon Mee Kim, Yoon Jung Choi,
Journal of Pathology and Translational Medicine.2023; 57(1): 1. CrossRef - A Standardized Pathology Report for Gastric Cancer: 2nd Edition
Young Soo Park, Myeong-Cherl Kook, Baek-hui Kim, Hye Seung Lee, Dong-Wook Kang, Mi-Jin Gu, Ok Ran Shin, Younghee Choi, Wonae Lee, Hyunki Kim, In Hye Song, Kyoung-Mee Kim, Hee Sung Kim, Guhyun Kang, Do Youn Park, So-Young Jin, Joon Mee Kim, Yoon Jung Choi,
Journal of Gastric Cancer.2023; 23(1): 107. CrossRef - Korean Practice Guidelines for Gastric Cancer 2022: An Evidence-based, Multidisciplinary Approach
Tae-Han Kim, In-Ho Kim, Seung Joo Kang, Miyoung Choi, Baek-Hui Kim, Bang Wool Eom, Bum Jun Kim, Byung-Hoon Min, Chang In Choi, Cheol Min Shin, Chung Hyun Tae, Chung sik Gong, Dong Jin Kim, Arthur Eung-Hyuck Cho, Eun Jeong Gong, Geum Jong Song, Hyeon-Su Im
Journal of Gastric Cancer.2023; 23(1): 3. CrossRef - Could Toll-like Receptor 2 Serve as Biomarker to Detect Advanced Gastric Cancer?
Marek Majewski, Kamil Torres, Paulina Mertowska, Sebastian Mertowski, Izabela Korona-Głowniak, Jan Korulczyk, Witold Zgodziński, Ewelina Grywalska
International Journal of Molecular Sciences.2023; 24(6): 5824. CrossRef - Research Progress of Immunotherapy for Gastric Cancer
Zhipeng Zhang, Ningning Liu, Mingyu Sun
Technology in Cancer Research & Treatment.2023;[Epub] CrossRef - Case Report: A rare synchronous multiple gastric carcinoma achieved progression-free disease through NGS-guided serial treatment
Xinyi Shao, Jin Yin, Di Wang, Erjiong Huang, Yini Zhang, Jiani C. Yin, Chen Huang, Hao Wu, Xiaoli Wu
Frontiers in Oncology.2023;[Epub] CrossRef - Artificial Intelligence-Enabled Gastric Cancer Interpretations
Mustafa Yousif, Liron Pantanowitz
Surgical Pathology Clinics.2023; 16(4): 673. CrossRef - The Optimal Tumor Mutational Burden Cutoff Value as a Novel Marker for Predicting the Efficacy of Programmed Cell Death-1 Checkpoint Inhibitors in Advanced Gastric Cancer
Jae Yeon Jang, Youngkyung Jeon, Sun Young Jeong, Sung Hee Lim, Won Ki Kang, Jeeyun Lee, Seung Tae Kim
Journal of Gastric Cancer.2023; 23(3): 476. CrossRef - Biomarkers for Predicting Response to Personalized Immunotherapy in Gastric Cancer
Moonsik Kim, Ji Yun Jeong, An Na Seo
Diagnostics.2023; 13(17): 2782. CrossRef - The Prognostic Value of the Prognostic Nutritional Index in Patients with Advanced or Metastatic Gastric Cancer Treated with Immunotherapy
Yuting Pan, Yue Ma, Guanghai Dai
Nutrients.2023; 15(19): 4290. CrossRef - Molecular classification of gastric cancer predicts survival in patients undergoing radical gastrectomy based on project HOPE
Kenichiro Furukawa, Keiichi Hatakeyama, Masanori Terashima, Takeshi Nagashima, Kenichi Urakami, Keiichi Ohshima, Akifumi Notsu, Takashi Sugino, Taisuke Yagi, Keiichi Fujiya, Satoshi Kamiya, Makoto Hikage, Yutaka Tanizawa, Etsuro Bando, Yae Kanai, Yasuto A
Gastric Cancer.2022; 25(1): 138. CrossRef - Immunotherapy for Gastric Cancer: A 2021 Update
Christo Kole, Nikolaos Charalampakis, Sergios Tsakatikas, Nikolaos-Iasonas Kouris, George Papaxoinis, Michalis V Karamouzis, Anna Koumarianou, Dimitrios Schizas
Immunotherapy.2022; 14(1): 41. CrossRef - The immune microenvironment in gastric adenocarcinoma
Yana Zavros, Juanita L. Merchant
Nature Reviews Gastroenterology & Hepatology.2022; 19(7): 451. CrossRef - Immunomodulation by Gut Microbiome on Gastrointestinal Cancers: Focusing on Colorectal Cancer
Raghad Khalid AL-Ishaq, Lenka Koklesova, Peter Kubatka, Dietrich Büsselberg
Cancers.2022; 14(9): 2140. CrossRef - An Immunity-Associated lncRNA Signature for Predicting Prognosis in Gastric Adenocarcinoma
Xiaowen Zhao, Pingfan Wu, Dongling Liu, Changtian Li, Ling Xue, Zhe Liu, Meng Zhu, Jie Yang, Ziyi Chen, Yaling Li, Yali She, Kathiravan Srinivasan
Journal of Healthcare Engineering.2022; 2022: 1. CrossRef - RNA modification writers influence tumor microenvironment in gastric cancer and prospects of targeted drug therapy
Peng Song, Sheng Zhou, Xiaoyang Qi, Yuwen Jiao, Yu Gong, Jie Zhao, Haojun Yang, Zhifen Qian, Jun Qian, Liming Tang
Journal of Bioinformatics and Computational Biology.2022;[Epub] CrossRef - Identification of the three subtypes and the prognostic characteristics of stomach adenocarcinoma: analysis of the hypoxia-related long non-coding RNAs
Zehua Fan, Yanqun Wang, Rong Niu
Functional & Integrative Genomics.2022; 22(5): 919. CrossRef - Complete Response of High Microsatellite Instability Gastric Cancer and Synchronous Microsatellite Stability Rectal Cancer
Zachary E Hunzeker, Pooja Bhakta, Sindusha R Gudipally, Sri Bharathi Kavuri, Rohit Venkatesan, Chukwuyejulumafor Nwanze
Cureus.2022;[Epub] CrossRef - Immune Profiling in Gastric Cancer Reveals the Dynamic Landscape of Immune Signature Underlying Tumor Progression
Yuhan Wei, Jianwei Zhang, Xueke Fan, Zhi Zheng, Xiaoyue Jiang, Dexi Chen, Yuting Lu, Yingrui Li, Miao Wang, Min Hu, Qi Du, Liuting Yang, Hongzhong Li, Yi Xiao, Yongfu Li, Jiangtao Jin, Deying Wang, Xiangliang Yuan, Qin Li
Frontiers in Immunology.2022;[Epub] CrossRef - Tumor vessel normalization and immunotherapy in gastric cancer
Xianzhe Yu, Shan He, Jian Shen, Qiushi Huang, Peng Yang, Lin Huang, Dan Pu, Li Wang, Lu Li, Jinghua Liu, Zelong Liu, Lingling Zhu
Therapeutic Advances in Medical Oncology.2022;[Epub] CrossRef - FN1 is a prognostic biomarker and correlated with immune infiltrates in gastric cancers
Han Wang, Junchang Zhang, Huan Li, Hong Yu, Songyao Chen, Shuhao Liu, Changhua Zhang, Yulong He
Frontiers in Oncology.2022;[Epub] CrossRef - Molecular Pathology of Gastric Cancer
Moonsik Kim, An Na Seo
Journal of Gastric Cancer.2022; 22(4): 264. CrossRef - Bioinformatics Analysis and Structure of Gastric Cancer Prognosis Model Based on Lipid Metabolism and Immune Microenvironment
Yongzhi Chen, Hongjun Yuan, Qian Yu, Jianyu Pang, Miaomiao Sheng, Wenru Tang
Genes.2022; 13(9): 1581. CrossRef - Clinical implications of interleukins-31, 32, and 33 in gastric cancer
Qing-Hua Liu, Ji-Wei Zhang, Lei Xia, Steven G Wise, Brett David Hambly, Kun Tao, Shi-San Bao
World Journal of Gastrointestinal Oncology.2022; 14(9): 1808. CrossRef - Microbiota and the Immune System—Actors in the Gastric Cancer Story
Marek Majewski, Paulina Mertowska, Sebastian Mertowski, Konrad Smolak, Ewelina Grywalska, Kamil Torres
Cancers.2022; 14(15): 3832. CrossRef - Bioinformatics and Experimental Analyses Reveal MAP4K4 as a Potential Marker for Gastric Cancer
Junping Zhang, Xiaoping Cai, Weifeng Cui, Zheng Wei
Genes.2022; 13(10): 1786. CrossRef - Common strategies for effective immunotherapy of gastroesophageal cancers using immune checkpoint inhibitors
Shuang Ma, Fei Chen
Pathology - Research and Practice.2022; 238: 154110. CrossRef - High-level of intratumoral GITR+ CD4 T cells associate with poor prognosis in gastric cancer
Shouyu Ke, Feng Xie, Yixian Guo, Jieqiong Chen, Zeyu Wang, Yimeng Yu, Haigang Geng, Danhua Xu, Xu Liu, Xiang Xia, Fengrong Yu, Chunchao Zhu, Zizhen Zhang, Gang Zhao, Bin Li, Wenyi Zhao
iScience.2022; 25(12): 105529. CrossRef - Characteristics of Adenosine-to-Inosine RNA editing-based subtypes and novel risk score for the prognosis and drug sensitivity in stomach adenocarcinoma
Jingjing Pan, Xinyuan Gu, Jing Luo, Xinye Qian, Qiang Gao, Tianjie Li, Longying Ye, Chenlu Li
Frontiers in Cell and Developmental Biology.2022;[Epub] CrossRef - Inhibition of NF‐κB is required for oleanolic acid to downregulate PD‐L1 by promoting DNA demethylation in gastric cancer cells
Xirong Lu, Yuyi Li, Wei Yang, Minghao Tao, Yanmiao Dai, Jinkang Xu, Qianfei Xu
Journal of Biochemical and Molecular Toxicology.2021;[Epub] CrossRef - Prognostic Value of C-Reactive Protein to Albumin Ratio in Gastric Cancer: A Meta-Analysis
Liang Yue, Yi Lu, Yulin Li, Yilin Wang
Nutrition and Cancer.2021; 73(10): 1864. CrossRef - Immunogenic characteristics of microsatellite instability‐low esophagogastric junction adenocarcinoma based on clinicopathological, molecular, immunological and survival analyses
Yu Imamura, Tasuku Toihata, Ikumi Haraguchi, Yoko Ogata, Manabu Takamatsu, Aya Kuchiba, Norio Tanaka, Osamu Gotoh, Seiichi Mori, Yuichiro Nakashima, Eiji Oki, Masaki Mori, Yoshinao Oda, Kenichi Taguchi, Manabu Yamamoto, Masaru Morita, Naoya Yoshida, Hideo
International Journal of Cancer.2021; 148(5): 1260. CrossRef - Two Similar Signatures for Predicting the Prognosis and Immunotherapy Efficacy of Stomach Adenocarcinoma Patients
Taohua Yue, Shuai Zuo, Jing Zhu, Shihao Guo, Zhihao Huang, Jichang Li, Xin Wang, Yucun Liu, Shanwen Chen, Pengyuan Wang
Frontiers in Cell and Developmental Biology.2021;[Epub] CrossRef - Tumor microenvironment characterization in stage IV gastric cancer
Feng Yang, Zhenbao Wang, Xianxue Zhang
Bioscience Reports.2021;[Epub] CrossRef - E2F2 inhibition induces autophagy via the PI3K/Akt/mTOR pathway in gastric cancer
Hui Li, Shufen Zhao, Liwei Shen, Peige Wang, Shihai Liu, Yingji Ma, Zhiwei Liang, Gongjun Wang, Jing Lv, Wensheng Qiu
Aging.2021; 13(10): 13626. CrossRef - Chemoradiation induces upregulation of immunogenic cell death-related molecules together with increased expression of PD-L1 and galectin-9 in gastric cancer
S. H. Petersen, L. F. Kua, S. Nakajima, W. P. Yong, K. Kono
Scientific Reports.2021;[Epub] CrossRef - Establishment of an Immune Cell Infiltration Score to Help Predict the Prognosis and Chemotherapy Responsiveness of Gastric Cancer Patients
Quan Jiang, Jie Sun, Hao Chen, Chen Ding, Zhaoqing Tang, Yuanyuan Ruan, Fenglin Liu, Yihong Sun
Frontiers in Oncology.2021;[Epub] CrossRef - Microsatellite instability in Gastric Cancer: Between lights and shadows
Elisabetta Puliga, Simona Corso, Filippo Pietrantonio, Silvia Giordano
Cancer Treatment Reviews.2021; 95: 102175. CrossRef - Survival-associated alternative splicing events interact with the immune microenvironment in stomach adenocarcinoma
Zai-Sheng Ye, Miao Zheng, Qin-Ying Liu, Yi Zeng, Sheng-Hong Wei, Yi Wang, Zhi-Tao Lin, Chen Shu, Qiu-Hong Zheng, Lu-Chuan Chen
World Journal of Gastroenterology.2021; 27(21): 2871. CrossRef - Immunotherapy of gastric cancer: Past, future perspective and challenges
Jun Xie, Liping Fu, Li Jin
Pathology - Research and Practice.2021; 218: 153322. CrossRef - Clinicopathologic and Prognostic Association of GRP94 Expression in Colorectal Cancer with Synchronous and Metachronous Metastases
Sumi Yun, Sukmook Lee, Ho-Young Lee, Hyeon Jeong Oh, Yoonjin Kwak, Hye Seung Lee
International Journal of Molecular Sciences.2021; 22(13): 7042. CrossRef - Injectable shear-thinning polylysine hydrogels for localized immunotherapy of gastric cancer through repolarization of tumor-associated macrophages
Yan Yang, Yang Yang, Meili Chen, Jianquan Chen, Jinyan Wang, Yajun Ma, Hanqing Qian
Biomaterials Science.2021; 9(19): 6597. CrossRef - Correlation between LRP1B Mutations and Tumor Mutation Burden in Gastric Cancer
Sizhe Hu, Xiaokang Zhao, Feng Qian, Cancan Jin, Kaishun Hou, Tao Huang
Computational and Mathematical Methods in Medicine.2021; 2021: 1. CrossRef - Comprehensive Analysis to Identify MAGEA3 Expression Correlated With Immune Infiltrates and Lymph Node Metastasis in Gastric Cancer
Jinji Jin, Jianxin Tu, Jiahuan Ren, Yiqi Cai, Wenjing Chen, Lifang Zhang, Qiyu Zhang, Guanbao Zhu
Frontiers in Oncology.2021;[Epub] CrossRef - Effect of P2X7 receptor on tumorigenesis and its pharmacological properties
Wen-jun Zhang, Ce-gui Hu, Zheng-ming Zhu, Hong-liang Luo
Biomedicine & Pharmacotherapy.2020; 125: 109844. CrossRef - Current status and future potential of predictive biomarkers for immune checkpoint inhibitors in gastric cancer
Byung Woog Kang, Ian Chau
ESMO Open.2020; 5(4): e000791. CrossRef - Is Ramucirumab Still the Only Second-Line Treatment in Metastatic Gastric Cancer?
Khalil El Gharib, Hampig Raphael Kourie
Pharmacogenomics.2020; 21(17): 1203. CrossRef - Deep Learning Predicts Underlying Features on Pathology Images with Therapeutic Relevance for Breast and Gastric Cancer
Renan Valieris, Lucas Amaro, Cynthia Aparecida Bueno de Toledo Osório, Adriana Passos Bueno, Rafael Andres Rosales Mitrowsky, Dirce Maria Carraro, Diana Noronha Nunes, Emmanuel Dias-Neto, Israel Tojal da Silva
Cancers.2020; 12(12): 3687. CrossRef
- Using glucocorticoid receptor-related genes to create and validate a survival model predicting gastric cancer
- Multiple Neuroendocrine Tumors in Stomach and Duodenum in a Multiple Endocrine Neoplasia Type 1 Patient
- Bohyun Kim, Han-Kwang Yang, Woo Ho Kim
- J Pathol Transl Med. 2018;52(2):126-129. Published online December 21, 2017
- DOI: https://doi.org/10.4132/jptm.2017.09.16
- 9,752 View
- 145 Download
- 1 Web of Science
- 1 Crossref
-
 Abstract
Abstract
 PDF
PDF - A 67-year-old woman with a history of subtotal parathyroidectomy, distal pancreatectomy, and total splenectomy 23 years prior underwent surgical gastric resection for neuroendocrine tumors of the stomach and duodenum. Meticulous examination of the entire stomach and duodenum revealed multiple scattered, minute neuroendocrine tumors. To the best of our knowledge, this is the first case report of a patient diagnosed with gastroduodenal neuroendocrine tumors associated with multiple endocrine neoplasia type 1 (MEN 1) in whom complete histologic mapping of the whole gastrectomy specimen was performed. The presence of MEN 1–associated neuroendocrine tumors in the stomach is very rare, but should be considered in patients diagnosed with MEN 1 who present with a new tumor in the stomach.
-
Citations
Citations to this article as recorded by- A Case of Asymptomatic Multiple Endocrine Neoplasia Type I with Thymic Carcinoid
Suk Ki Park, Moon Won Lee, In Sub Han, Young Joo Park, Sung Yong Han, Joon Woo Park, Bong Eun Lee, Gwang Ha Kim, Sang Soo Kim
The Korean Journal of Helicobacter and Upper Gastrointestinal Research.2019; 19(1): 65. CrossRef
- A Case of Asymptomatic Multiple Endocrine Neoplasia Type I with Thymic Carcinoid
- Extramural Perineural Invasion in pT3 and pT4 Gastric Carcinomas
- Alejandro España-Ferrufino, Leonardo S. Lino-Silva, Rosa A. Salcedo-Hernández
- J Pathol Transl Med. 2018;52(2):79-84. Published online November 9, 2017
- DOI: https://doi.org/10.4132/jptm.2017.11.01
- 11,518 View
- 143 Download
- 15 Web of Science
- 13 Crossref
-
 Abstract
Abstract
 PDF
PDF - Background
Perineural invasion (PNI) is widely studied in malignant tumors, and its prognostic significance is well demonstrated. Most studies have focused on evaluating the mural PNI (mPNI); however, extramural PNI (ePNI) may influence the prognosis in gastric cancer. We evaluated the prognostic value of ePNI compared with mPNI in gastric cancer in this observational comparative cross-sectional study.
Methods
Seventy-three pT3 and pT4 gastric carcinomas with PNI were evaluated. Forty-eight (65.7%) were in the mPNI group and the remaining in the ePNI group.
Results
Clinicopathologic characteristics between the two groups were similar, except for the outcomes. The 5-year disease-specific survival (DSS) rate was 64% for the mPNI group and 50% for the ePNI group (p=.039), a difference that did not remain significant in multivariate analysis. The only independent adverse prognostic factor in multivariate analysis was the presence of lymph node metastasis (hazard ratio, 1.757; 95% confidence interval, 1.082 to 2.854; p=.023).
Conclusions
We demonstrated the prognostic effect of ePNI for DSS in surgically resected pT3–pT4 gastric cancer patients. ePNI could be considered in the staging and prognostic systems of gastric cancer to stratify patients with a high risk of recurrence. -
Citations
Citations to this article as recorded by- The neural niche in cancer: mechanistic insights into tumor–neuron–immune crosstalk and therapeutic opportunities
Nese Aysit, Esra Altintas, Fulya Koksalar Alkan, Gurkan Ozturk, Hasan Korkaya
Frontiers in Cell and Developmental Biology.2026;[Epub] CrossRef - Investigation of Conditioned Media-Mediated Communication between Pancreatic Cancer Cells and Neurons
Didem Karakaş
Acta Medica Nicomedia.2025; 8(1): 15. CrossRef - Innervating 3D in vitro models: bioengineering and design blueprints
Mariana-Tomás de Carvalho, Margarida Henriques-Pereira, Maria V. Monteiro, Meriem Lamghari, João F. Mano, Vítor M. Gaspar
Trends in Biotechnology.2025; 43(11): 2743. CrossRef - Development and validation of a preoperative CT-based risk score integrating morphological and body composition parameters to predict recurrence-free survival in gastric cancer patients following curative surgery
Ruochen Cong, Jialei Ming, Ruonan Xu, Liyu Zhu, Xinyue Wang, Zhengqi Zhu
European Journal of Radiology.2025; 191: 112318. CrossRef - Comparison of 2D and 3D measured iodine concentration of gastric adenocarcinoma on spectral-CT and their relationship with perineural invasion
Tianxia Bei, Xiaoqiang Yao, Xuejun Chen, Yue Wu, Jing Li, Jinrong Qu
BMC Medical Imaging.2025;[Epub] CrossRef - Neural control of tumor immunity
Burak Kizil, Francesco De Virgiliis, Christoph Scheiermann
The FEBS Journal.2024; 291(21): 4670. CrossRef - Spectral CT-based nomogram for preoperative prediction of perineural invasion in locally advanced gastric cancer: a prospective study
Jing Li, Shuning Xu, Yi Wang, Mengjie Fang, Fei Ma, Chunmiao Xu, Hailiang Li
European Radiology.2023; 33(7): 5172. CrossRef - Crosstalk between cancer cells and the nervous system
Meng Huang, Gu Gong, Yicheng Deng, Xinmiao Long, Wenyong Long, Qing Liu, Wei Zhao, Rufu Chen
Medicine Advances.2023; 1(3): 173. CrossRef - Targeting tumor innervation: premises, promises, and challenges
Xinyu Li, Xueqiang Peng, Shuo Yang, Shibo Wei, Qing Fan, Jingang Liu, Liang Yang, Hangyu Li
Cell Death Discovery.2022;[Epub] CrossRef - Cancer-Associated Neurogenesis and Nerve-Cancer Cross-talk
Deborah A. Silverman, Vena K. Martinez, Patrick M. Dougherty, Jeffrey N. Myers, George A. Calin, Moran Amit
Cancer Research.2021; 81(6): 1431. CrossRef - Perineural Invasion and Postoperative Adjuvant Chemotherapy Efficacy in Patients With Gastric Cancer
Qing Tao, Wen Zhu, Xiaohui Zhao, Mei Li, Yongqian Shu, Deqiang Wang, Xiaoqin Li
Frontiers in Oncology.2020;[Epub] CrossRef - Perineural invasion as a predictive factor for survival outcome in gastric cancer patients: a systematic review and meta-analysis
Bochao Zhao, Wu Lv, Di Mei, Rui Luo, Shiyang Bao, Baojun Huang, Jie Lin
Journal of Clinical Pathology.2020; 73(9): 544. CrossRef - Consensus-Expressed CXCL8 and MMP9 Identified by Meta-Analyzed Perineural Invasion Gene Signature in Gastric Cancer Microarray Data
Xiuzhi Jia, Minjia Lu, Chen Rui, Ying Xiao
Frontiers in Genetics.2019;[Epub] CrossRef
- The neural niche in cancer: mechanistic insights into tumor–neuron–immune crosstalk and therapeutic opportunities
- HER2 Status and Its Heterogeneity in Gastric Carcinoma of Vietnamese Patient
- Dang Anh Thu Phan, Vu Thien Nguyen, Thi Ngoc Ha Hua, Quoc Dat Ngo, Thi Phuong Thao Doan, Sao Trung Nguyen, Anh Tu Thai, Van Thanh Nguyen
- J Pathol Transl Med. 2017;51(4):396-402. Published online June 19, 2017
- DOI: https://doi.org/10.4132/jptm.2017.04.24
- 12,381 View
- 165 Download
- 13 Web of Science
- 11 Crossref
-
 Abstract
Abstract
 PDF
PDF - Background
Human epidermal growth factor receptor 2 (HER2) is related to the pathogenesis and poor outcome of numerous types of carcinomas, including gastric carcinoma. Gastric cancer patients with HER2 positivity have become potential candidates for targeted therapy with trastuzumab.
Methods
We investigated 208 gastric cancer specimens using immunohistochemistry (IHC), fluorescence in situ hybridization and dual in situ hybridization (ISH). We also investigated the concordance between IHC and ISH. The correlation between HER2 status and various clinicopathological findings was also investigated.
Results
In total, 15.9% (33/208) and 24.5% (51/208) of gastric cancers showed HER2 gene amplification and protein overexpression, respectively. A high level of concordance between ISH and IHC analyses (91.3%, κ = 0.76) was found. A significant correlation between HER2 status and intestinal-type (p < .05) and differentiated carcinomas (p < .05) was also noted. The HER2 heterogeneity was high in gastric cancers; we found 68.8% phenotypic heterogeneity and 57.6% genotypic heterogeneity. Heterogeneity in HER2 protein expression and gene amplification showed a close association with diffuse histologic type and IHC 2+.
Conclusions
HER2 protein overexpression and gene amplification were detected in 24.5% and 15.9% of gastric cancer specimens, respectively. Intestinal-type showed a higher level of HER2 protein overexpression and gene amplification than diffuse type. HER2 status also showed a significant relationship with well- and moderately-differentiated carcinomas. The ratio of phenotypic and genotypic heterogeneity of HER2 was high in gastric carcinomas and was associated with HER2 IHC 2+ and diffuse histologic type. -
Citations
Citations to this article as recorded by- Advancing human epidermal growth factor receptor 2-positive gastric cancer therapy: Toward targeted immunotherapy and antibody-drug conjugates
Jia-Ju Xu, Chun-Xiao Ni, Ping Wang, Li-Dong Qin, Jia-Ju Xu
World Journal of Gastrointestinal Oncology.2026;[Epub] CrossRef - Interpretable deep learning-based hierarchical multi-modal fusion model for predicting HER2 expression in gastric cancer
Chenxi Hu, Changfeng Feng, Ziyi Ye, Shuaihang Kong, Jing Han, Tingting Yang, Kai Zhong, Xingguang Zhong, Qijun Shen
Frontiers in Oncology.2026;[Epub] CrossRef - Human epidermal growth factor receptor 2 expression and socio-demographic, clinical, and histopathological characteristics in gastric carcinoma at St Francis Hospital Nsambya, Uganda
Steven Wanda, Gorretti Nassali, Brian Bbosa, Francis Basimbe, Joviah Akulu, Davis Nsamba, Praise Nimusiima, Joshua Muhumuza, Emmanuel Othieno, Maxwel Dancan Okuku
BMC Gastroenterology.2025;[Epub] CrossRef - Expression of Her-2 and Ki-67 in Gastric Cancer Formalin Fixed Paraffin Embedded Tissue Blocks and Their Correlation with Histological Grades at the Uganda Cancer Institute Pathology Laboratory
Hassan Wasswa, Abraham Birungi, Lawrence Amadile, Richard Kasadha, Saphurah Nabaasa, Jolly Ninsiima, Tonny Okecha, Frank Ssedyabane, Raymond Atwine, Lauben Tibenderana
Pathology and Laboratory Medicine International.2024; Volume 16: 23. CrossRef - Identifying HER2 from serum‐derived exosomes in advanced gastric cancer as a promising biomarker for assessing tissue HER2 status and predicting the efficacy of trastuzumab‐based therapy
Qian Li, Minzhi Lv, Lihua Lv, Nida Cao, Aiguang Zhao, Jiayan Chen, Xi Tang, Rongkui Luo, Shan Yu, Yan Zhou, Yuehong Cui, Wei Guo, Tianshu Liu
Cancer Medicine.2023; 12(4): 4110. CrossRef - A study of human epidermal growth factor receptor 2 overexpression by immunohistochemistry in patients with gastric adenocarcinoma
Rafid A. Abood, Saad Alomar, Sawsan S. Alharoon
Journal of Public Health in Africa.2023; 14(9): 4. CrossRef - Receptor Tyrosine Kinases Amplified in Diffuse-Type Gastric Carcinoma: Potential Targeted Therapies and Novel Downstream Effectors
Hideki Yamaguchi, Yuko Nagamura, Makoto Miyazaki
Cancers.2022; 14(15): 3750. CrossRef - Cell differentiation trajectory predicts patient potential immunotherapy response and prognosis in gastric cancer
Renshen Xiang, Yuping Rong, Yuhang Ge, Wei Song, Jun Ren, Tao Fu
Aging.2021; 13(4): 5928. CrossRef - Identification of stem cell-related subtypes and risk scoring for gastric cancer based on stem genomic profiling
Renshen Xiang, Wei Song, Jun Ren, Jing Wu, Jincheng Fu, Tao Fu
Stem Cell Research & Therapy.2021;[Epub] CrossRef - Oncogenic signaling pathways associated with immune evasion and resistance to immune checkpoint inhibitors in cancer
Yoshie Kobayashi, Seung-Oe Lim, Hirohito Yamaguchi
Seminars in Cancer Biology.2020; 65: 51. CrossRef - Blockade of ITGA2 Induces Apoptosis and Inhibits Cell Migration in Gastric Cancer
Yu-Chang Chuang, Hsin-Yi Wu, Yu-Ling Lin, Shey-Cherng Tzou, Cheng-Hsun Chuang, Ting-Yan Jian, Pin-Rong Chen, Yuan-Ching Chang, Chi-Hsin Lin, Tse-Hung Huang, Chao-Ching Wang, Yi-Lin Chan, Kuang-Wen Liao
Biological Procedures Online.2018;[Epub] CrossRef
- Advancing human epidermal growth factor receptor 2-positive gastric cancer therapy: Toward targeted immunotherapy and antibody-drug conjugates
- Perivascular Epithelioid Cell Tumor in the Stomach
- Sun Ah Shin, Jiwoon Choi, Kyung Chul Moon, Woo Ho Kim
- J Pathol Transl Med. 2017;51(4):428-432. Published online April 4, 2017
- DOI: https://doi.org/10.4132/jptm.2016.09.16
- 10,579 View
- 141 Download
- 6 Web of Science
- 7 Crossref
-
 Abstract
Abstract
 PDF
PDF - Perivascular epithelioid cell tumors or PEComas can arise in any location in the body. However, a limited number of cases of gastric PEComa have been reported. We present two cases of gastric PEComas. The first case involved a 62-year-old woman who presented with a 4.2 cm gastric subepithelial mass in the prepyloric antrum, and the second case involved a 67-year-old man with a 5.0 cm mass slightly below the gastroesophageal junction. Microscopic examination revealed that both tumors were composed of perivascular epithelioid cells that were immunoreactive for melanocytic and smooth muscle markers. Prior to surgery, the clinical impression of both tumors was gastrointestinal stromal tumor (GIST), and the second case was erroneously diagnosed as GIST even after microscopic examination. Although gastric PEComa is a very rare neoplasm, it should be considered in the differential diagnosis of gastric submucosal lesions.
-
Citations
Citations to this article as recorded by- Extra-uterine Endometrial Stromal Sarcoma of the Stomach Mimicking a Gastrointestinal Stromal Tumour: A Case Report
Adam Mylonakis, Eleandros Kyros, Maria Karakeke, Eleni Karakeke, Andreas Panagakis, Pagona Kastanaki, Lysandros Karydakis, Alexandros Pergaris, Stratigoula Sakellariou, Alexandros Papalampros
Cureus.2026;[Epub] CrossRef - A Perivascular Epithelioid Cell Tumor in the Ascending Colon: A Rare Case Involving a Patient With Tuberous Sclerosis
Kai Seharada, Masato Kitazawa, Satoshi Nakamura, Yuta Yamamoto, Yuji Soejima
Cureus.2025;[Epub] CrossRef - Malignant Perivascular Epithelioid Cell Tumor of Ovary: A Rare Case Report
Anuradha Sharma, Reetika Sharma, Jyoti Bala, Monika Sharma
Journal of Mid-life Health.2025; 16(1): 107. CrossRef - A Case of a Primary Perivascular Epithelial Tumor of the Stomach with Lymph Node Metastasis
Nobuyuki NISHIDA, Hideki HIDAKA, Roko HAMADA, Takanori MEI, Kazuki TAKEDA, Kosuke MARUTSUKA, Kazuya MAEKAWA, Jiro OHUCHIDA
Nihon Rinsho Geka Gakkai Zasshi (Journal of Japan Surgical Association).2025; 86(9): 1178. CrossRef - Unusual paediatric sigmoid perivascular epithelioid cell tumour with regional lymph node metastasis treated using gemcitabine and docetaxel: a case report and literature review
Hsiu-Chung Cheng, Chia-Yu Kuo, Ching-Wen Huang, Hsiang-Hung Shih, Chih-Hung Lin, Jaw-Yuan Wang
Journal of International Medical Research.2021;[Epub] CrossRef - Gastric Perivascular Epithelioid Cell Tumor (PEComa)
Jinghong Xu, Yu Yan, Xueping Xiang, Peter Jiang, Xiangrong Hu, Wenjun Yang
American Journal of Clinical Pathology.2019; 152(2): 221. CrossRef - Robotic wedge resection of a rare gastric perivascular epithelioid cell tumor: A case report
Alessandra Marano, Francesca Maione, Yanghee Woo, Luca Pellegrino, Paolo Geretto, Diego Sasia, Mirella Fortunato, Giulio Fraternali Orcioni, Roberto Priotto, Renato Fasoli, Felice Borghi
World Journal of Clinical Cases.2019; 7(23): 4011. CrossRef
- Extra-uterine Endometrial Stromal Sarcoma of the Stomach Mimicking a Gastrointestinal Stromal Tumour: A Case Report
- Molecular Dimensions of Gastric Cancer: Translational and Clinical Perspectives
- Yoon Young Choi, Sung Hoon Noh, Jae-Ho Cheong
- J Pathol Transl Med. 2016;50(1):1-9. Published online October 26, 2015
- DOI: https://doi.org/10.4132/jptm.2015.09.10
- 16,405 View
- 158 Download
- 21 Web of Science
- 22 Crossref
-
 Abstract
Abstract
 PDF
PDF - Gastric cancer is a global health burden and has the highest incidence in East Asia. This disease is complex in nature because it arises from multiple interactions of genetic, local environmental, and host factors, resulting in biological heterogeneity. This genetic intricacy converges on molecular characteristics reflecting the pathophysiology, tumor biology, and clinical outcome. Therefore, understanding the molecular characteristics at a genomic level is pivotal to improving the clinical care of patients with gastric cancer. A recent landmark study, The Cancer Genome Atlas (TCGA) project, showed the molecular landscape of gastric cancer through a comprehensive molecular evaluation of 295 primary gastric cancers. The proposed molecular classification divided gastric cancer into four subtypes: Epstein-Barr virus–positive, microsatellite unstable, genomic stable, and chromosomal instability. This information will be taken into account in future clinical trials and will be translated into clinical therapeutic decisions. To fully realize the clinical benefit, many challenges must be overcome. Rapid growth of high-throughput biology and functional validation of molecular targets will further deepen our knowledge of molecular dimensions of this cancer, allowing for personalized precision medicine.
-
Citations
Citations to this article as recorded by- Establishment and characterization of a new mouse gastric carcinoma cell line, MCC
Yushen Wang, Xianju Li, Yi Wang, Jun Qin
Cancer Cell International.2025;[Epub] CrossRef - Advancements and challenges in gastric cancer: epidemiology, biomarkers, and therapeutic strategies
Marina Nishimuni, Laura Carolina Lopez Claro, Maria Ignez Freitas Melro Braghiroli
Surgical and Experimental Pathology.2024;[Epub] CrossRef - Tissue Biomarkers in Gastric Cancer Treatment: Present and Future
Giulia Airò, Virginia Agnetti, Fabiana Pratticò, Marianna Peroni, Simona Bui, Giovanni Mura, Maria Urbanowicz-Nijaki, Eleonora Lai, Marco Puzzoni, Fabiana Contu, Nerina Denaro, Mario Scartozzi, Cinzia Solinas, Chiara Tommasi
International Journal of Translational Medicine.2024; 4(4): 640. CrossRef - Research Progress of MSI Gastric Cancer Subtypes
成菊 马
Advances in Clinical Medicine.2022; 12(07): 6719. CrossRef - OCT4-mediated transcription confers oncogenic advantage for a subset of gastric tumors with poor clinical outcome
Jaishree Pandian, Ponmathi Panneerpandian, Balaji T. Sekar, Karthikeyan Selvarasu, Kumaresan Ganesan
Functional & Integrative Genomics.2022; 22(6): 1345. CrossRef - Secondary Primary Cancer after Primary Gastric Cancer: Literature Review and Big Data Analysis Using the Health Insurance Review and Assessment Service (HIRA) Database of Republic of Korea
Jeong Ho Song, Yeonkyoung Lee, Jaesung Heo, Sang-Yong Son, Hoon Hur, Sang-Uk Han
Cancers.2022; 14(24): 6165. CrossRef - Microsatellite instability: a review of what the oncologist should know
Kai Li, Haiqing Luo, Lianfang Huang, Hui Luo, Xiao Zhu
Cancer Cell International.2020;[Epub] CrossRef - Single Patient Classifier Assay, Microsatellite Instability, and Epstein-Barr Virus Status Predict Clinical Outcomes in Stage II/III Gastric Cancer: Results from CLASSIC Trial
Chul Kyu Roh, Yoon Young Choi, Seohee Choi, Won Jun Seo, Minah Cho, Eunji Jang, Taeil Son, Hyoung-Il Kim, Hyeseon Kim, Woo Jin Hyung, Yong-Min Huh, Sung Hoon Noh, Jae-Ho Cheong
Yonsei Medical Journal.2019; 60(2): 132. CrossRef - Ten Thousand Consecutive Gastrectomies for Gastric Cancer: Perspectives of a Master Surgeon
Yoon Young Choi, Minah Cho, In Gyu Kwon, Taeil Son, Hyoung-Il Kim, Seung Ho Choi, Jae-Ho Cheong, Woo Jin Hyung
Yonsei Medical Journal.2019; 60(3): 235. CrossRef - Clinicopathologic significance of human leukocyte antigen class I expression in patients with stage II and III gastric cancer
Yujun Park, Jiwon Koh, Yoonjin Kwak, Sang-Hoon Ahn, Do Joong Park, Hyung-Ho Kim, Woo Ho Kim, Hye Seung Lee
Cancer Immunology, Immunotherapy.2019; 68(11): 1779. CrossRef - Phosphoproteomics Enables Molecular Subtyping and Nomination of Kinase Candidates for Individual Patients of Diffuse-Type Gastric Cancer
Mengsha Tong, Chunyu Yu, Jinwen Shi, Wenwen Huang, Sai Ge, Mingwei Liu, Lei Song, Dongdong Zhan, Xia Xia, Wanlin Liu, Jinwen Feng, Wenhao Shi, Jiafu Ji, Jing Gao, Tieliu Shi, Weimin Zhu, Chen Ding, Yi Wang, Fuchu He, Lin Shen, Tingting Li, Jun Qin
iScience.2019; 22: 44. CrossRef - Trastuzumab Specific Epitope Evaluation as a Predictive and Prognostic Biomarker in Gastric Cancer Patients
Jiwon Koh, Soo Kyung Nam, Youn Woo Lee, Jin Won Kim, Keun-Wook Lee, Chan-Young Ock, Do-Youn Oh, Sang-Hoon Ahn, Hyung-Ho Kim, Keon-Wook Kang, Woo Ho Kim, Ho-Young Lee, Hye Seung Lee
Biomolecules.2019; 9(12): 782. CrossRef - Targeted drug delivery of capecitabine to mice xenograft gastric cancer by PAMAM dendrimer nanocarrier
Sharareh Jafari, Fatemeh Nabavizadeh, Jalal Vahedian, Mehdi Shafie Ardestani, Hedayat Samandari, Ali Zare Mehrjerdi
african journal of gastroenterology and hepatology.2019; 2(1): 28. CrossRef - New therapeutic options opened by the molecular classification of gastric cancer
Mihaela Chivu-Economescu, Lilia Matei, Laura G Necula, Denisa L Dragu, Coralia Bleotu, Carmen C Diaconu
World Journal of Gastroenterology.2018; 24(18): 1942. CrossRef - Proposed Molecular and miRNA Classification of Gastric Cancer
Lara Alessandrini, Melissa Manchi, Valli De Re, Riccardo Dolcetti, Vincenzo Canzonieri
International Journal of Molecular Sciences.2018; 19(6): 1683. CrossRef - High serum MMP-14 predicts worse survival in gastric cancer
Aaro Kasurinen, Taina Tervahartiala, Alli Laitinen, Arto Kokkola, Timo Sorsa, Camilla Böckelman, Caj Haglund, Dajun Deng
PLOS ONE.2018; 13(12): e0208800. CrossRef - miR-30 functions as an oncomiR in gastric cancer cells through regulation of P53-mediated mitochondrial apoptotic pathway
Jianjun Wang, Yang Jiao, Lunmeng Cui, Lili Jiang
Bioscience, Biotechnology, and Biochemistry.2017; 81(1): 119. CrossRef - Clinicopathologic implication of meticulous pathologic examination of regional lymph nodes in gastric cancer patients
Jiwon Koh, Hee Eun Lee, Woo Ho Kim, Hye Seung Lee, Ju-Seog Lee
PLOS ONE.2017; 12(3): e0174814. CrossRef - Perioperative chemotherapy for resectable gastric cancer – what is the evidence?
Erling A Bringeland, Hans H Wasmuth, Jon E Grønbech
Scandinavian Journal of Gastroenterology.2017; 52(6-7): 647. CrossRef - Molecular classifications of gastric cancers: Novel insights and possible future applications
Silvio Ken Garattini, Debora Basile, Monica Cattaneo, Valentina Fanotto, Elena Ongaro, Marta Bonotto, Francesca V Negri, Rosa Berenato, Paola Ermacora, Giovanni Gerardo Cardellino, Mariella Giovannoni, Nicoletta Pella, Mario Scartozzi, Lorenzo Antonuzzo,
World Journal of Gastrointestinal Oncology.2017; 9(5): 194. CrossRef - GRAM domain-containing protein 1B (GRAMD1B), a novel component of the JAK/STAT signaling pathway, functions in gastric carcinogenesis
Puja Khanna, Pei Jou Chua, Belinda Shu Ee Wong, Changhong Yin, Aye Aye Thike, Wei Keat Wan, Puay Hoon Tan, Gyeong Hun Baeg
Oncotarget.2017; 8(70): 115370. CrossRef - Clinicopathologic implications of immune classification by PD-L1 expression and CD8-positive tumor-infiltrating lymphocytes in stage II and III gastric cancer patients
Jiwon Koh, Chan-Young Ock, Jin Won Kim, Soo Kyung Nam, Yoonjin Kwak, Sumi Yun, Sang-Hoon Ahn, Do Joong Park, Hyung-Ho Kim, Woo Ho Kim, Hye Seung Lee
Oncotarget.2017; 8(16): 26356. CrossRef
- Establishment and characterization of a new mouse gastric carcinoma cell line, MCC
- Gastric-Type Extremely Well-Differentiated Adenocarcinoma of the Stomach: A Challenge for Preoperative Diagnosis
- Mee Joo, Song Hee Han
- J Pathol Transl Med. 2016;50(1):71-74. Published online September 30, 2015
- DOI: https://doi.org/10.4132/jptm.2015.07.14
- 14,548 View
- 184 Download
- 10 Web of Science
- 11 Crossref
-
 Abstract
Abstract
 PDF
PDF - Gastric-type extremely well-differentiated adenocarcinoma (EWDA) is a rare type of gastric adenocarcinoma characterized by infiltration of well-formed mucinous glands with little or no nuclear atypia, which resemble foveolar epithelium or pyloric glands. Because of its high degree of differentiation, preoperative biopsy diagnosis of gastric-type EWDA is very difficult. We encountered a case of gastric-type EWDA, manifesting as a Borrmann type 4 lesion, in a 47-year-old man. Despite four repeated biopsies, the preoperative biopsy diagnosis was not conclusive due to the scarcity of diagnostic tumor cells and lack of knowledge regarding the unusual histologic findings of gastric-type EWDA. We herein describe the histologic findings of gastric-type EWDA in detail, with the aim of facilitating a preoperative biopsy diagnosis and understanding of this rare type of gastric adenocarcinoma.
-
Citations
Citations to this article as recorded by- Crawling-type Gastric Adenocarcinoma: Clinicopathology, Diagnosis, and Endoscopic Treatment Implications
Cong Bang Huynh, Jin Won Chang
Journal of Digestive Cancer Research.2026; 14(1): 1. CrossRef - Unusual or Uncommon Histology of Gastric Cancer
Jinho Shin, Young Soo Park
Journal of Gastric Cancer.2024; 24(1): 69. CrossRef - Clinical pathological characteristics of “crawling-type” gastric adenocarcinoma cancer: A case report
Yong-Wei Xu, Yan Song, Jun Tian, Ba-Cui Zhang, Yu-Sheng Yang, Jing Wang
World Journal of Gastrointestinal Oncology.2024; 16(4): 1660. CrossRef - Gastric-type extremely well-differentiated adenocarcinoma of the stomach: A rare tumor with diagnostic difficulties and high inter-observer variation in endoscopic pinch biopsies
Soomin Ahn, Sujin Park, Hyun Hee Koh, Han Gyeol Kim, Hyunjin Kim, Jae Yeong Son, Boram Lee, Hyunwoo Lee, Soohyun Hwang, Junhun Cho, Yun Kyung Lee, Ryoji Kushima, Amitabh Srivastava, Kyoung-Mee Kim
Pathology - Research and Practice.2024; 263: 155599. CrossRef - Clinicopathological features of gastric adenocarcinoma of fundic gland type
Bao-Zhen Guo, Zhen-Zhen Liu, Gao-Fei Shen, Fei Zhu, Hui-Fen Lian, Xin Li, Jun-Yi Zheng, Jin-Peng Li, Shui-Miao Deng, Rui Huang
World Chinese Journal of Digestology.2023; 31(6): 244. CrossRef - Characterization of pathological stomach tissue using polarization-sensitive second harmonic generation microscopy
Hwanhee Jeon, MacAulay Harvey, Richard Cisek, Elisha Bennett, Danielle Tokarz
Biomedical Optics Express.2023; 14(10): 5376. CrossRef - Extremely well-differentiated adenocarcinoma of the stomach: diagnostic pitfalls in endoscopic biopsy
Jongwon Lee, In-Seob Lee, Ji Yong Ahn, Young Soo Park, Jihun Kim
Journal of Pathology and Translational Medicine.2022; 56(2): 63. CrossRef - Helicobacter pylori-negative Gastric Cancer
Sun-Young Lee
The Korean Journal of Helicobacter and Upper Gastrointestinal Research.2021; 21(1): 10. CrossRef - Preoperative diagnosis of a gastric extremely well-differentiated adenocarcinoma
Katsushi Suenaga, Shiro Matsumoto, Alan Kawarai Lefor, Yoshimasa Miura, Yoshinori Hosoya, Daigo Kuboki, Hidenori Haruta, Kentaro Kurashina, Atsushi Kihara, Daisuke Matsubara, Yasunari Sakuma, Joji Kitayama, Naohiro Sata
International Journal of Surgery Case Reports.2020; 73(C): 319. CrossRef - Gastric adenocarcinoma of the fundic gland type: clinicopathological features of eight patients treated with endoscopic submucosal dissection
Chengfang Li, Xinglong Wu, Shuang Yang, Xiaorong Yang, Jin Yao, Hong Zheng
Diagnostic Pathology.2020;[Epub] CrossRef - Gastric Adenocarcinoma of the Fundic Gland Type
Mark A Benedict, Gregory Y Lauwers, Dhanpat Jain
American Journal of Clinical Pathology.2018; 149(6): 461. CrossRef
- Crawling-type Gastric Adenocarcinoma: Clinicopathology, Diagnosis, and Endoscopic Treatment Implications
- Endogenous Gastric-Resident Mesenchymal Stem Cells Contribute to Formation of Cancer Stroma and Progression of Gastric Cancer
- Eun-Kyung Kim, Hye-Jung Kim, Young-Il Yang, Jong Tae Kim, Min-Young Choi, Chang Soo Choi, Kwang-Hee Kim, Jeong-Han Lee, Won-Hee Jang, Soon-Ho Cheong
- Korean J Pathol. 2013;47(6):507-518. Published online December 24, 2013
- DOI: https://doi.org/10.4132/KoreanJPathol.2013.47.6.507
- 11,053 View
- 54 Download
- 18 Crossref
-
 Abstract
Abstract
 PDF
PDF Background Carcinoma-associated fibroblasts (CAFs) contribute to carcinogenesis and cancer progression, although their origin and role remain unclear. We recently identified and investigated the
in situ identity and implications of gastric submucosa-resident mesenchymal stem cells (GS-MSCs) in the progression of gastric carcinogenesis.Methods We isolated GS-MSCs from gastric submucosa using hydrogel-supported organ culture and defined their identity. Isolated cells were assessed
in vitro by immunophenotype and mesengenic multipotency. Reciprocal interactions between GS-MSCs and gastric cancer cells were evaluated. To determine the role of GS-MSCs, xenografts were constructed of gastric cancer cells admixed with or without GS-MSCs.Results Isolated cells fulfilled MSCs requirements in regard to plastic adherence, stromal cell immunophenotype, and multipotency. We demonstrated a paracrine loop that gastric cancer cells enhanced the migration, proliferation, and differentiation of GS-MSCs; additionally, GS-MSCs promoted the proliferation of gastric cancer cell
in vitro . Xenograft experiments showed that GS-MSCs significantly promoted cancer growth and angiogenesis. GS-MSCs that integrated into gastric cancer became not only CAFs but also rarely endothelial cells which contributed to the formation of cellular and vascular cancer stroma.Conclusions Endogenous GS-MSCs play an important role in gastric cancer progression.
-
Citations
Citations to this article as recorded by- Tumor microenvironment dynamics in gastric cancer pathogenesis and therapeutic resistance
Zhenhua Lu, Qinnan Zhang, Jing Han, Jiafu Ji, Xiaofang Xing
Molecular Cancer.2026;[Epub] CrossRef - The therapeutic potential of mesenchymal stem cells in intestinal diseases: from mechanisms to clinical translation
Jia-Zhi Yang, Li-Yan He, Xian-Zhou Lu
Stem Cell Research & Therapy.2025;[Epub] CrossRef - Gastric cancer and mesenchymal stem cell-derived exosomes: from pro-tumorigenic effects to anti-cancer vehicles
Maryam Dolatshahi, Ahmad Reza Bahrami, Qaiser Iftikhar Sheikh, Mohsen Ghanbari, Maryam M. Matin
Archives of Pharmacal Research.2024; 47(1): 1. CrossRef - Extracellular vesicles derived from SARS-CoV-2 M-protein-induced triple negative breast cancer cells promoted the ability of tissue stem cells supporting cancer progression
Hoai-Nga Thi Nguyen, Cat-Khanh Vuong, Mizuho Fukushige, Momoko Usuda, Liora Kaho Takagi, Toshiharu Yamashita, Mana Obata-Yasuoka, Hiromi Hamada, Motoo Osaka, Toru Tsukada, Yuji Hiramatsu, Osamu Ohneda
Frontiers in Oncology.2024;[Epub] CrossRef - Revisiting the role of mesenchymal stromal cells in cancer initiation, metastasis and immunosuppression
Yanyan Zhang, Charles Wang, Jian Jian Li
Experimental Hematology & Oncology.2024;[Epub] CrossRef - Mesenchymal stem cells and their potential therapeutic benefits and challenges in the treatment and pathogenesis of gastric cancer
Asma'a H. Mohamed, Alaa Shafie, Waleed Al Abdulmonem, Hassan Swed Alzahrani, Amal Adnan Ashour, Ahmed Hjazi, Azfar Jamal, Allolo D. Aldreiwish, Mohammad Azhar Kamal, Fuzail Ahmad, Nazia Khan
Pathology - Research and Practice.2024; 260: 155422. CrossRef - Cancer-associated fibroblasts promote proliferation, angiogenesis, metastasis and immunosuppression in gastric cancer
Peiyuan Li, Huan Zhang, Tao Chen, Yajing Zhou, Jiaoyang Yang, Jin Zhou
Matrix Biology.2024; 132: 59. CrossRef - Mesenchymal Stem Cell-Derived Exosomes Modulate Angiogenesis in Gastric Cancer
Fawzy Akad, Veronica Mocanu, Sorin Nicolae Peiu, Viorel Scripcariu, Bogdan Filip, Daniel Timofte, Florin Zugun-Eloae, Magdalena Cuciureanu, Monica Hancianu, Teodor Oboroceanu, Laura Condur, Radu Florin Popa
Biomedicines.2023; 11(4): 1031. CrossRef - LRRC15 expression indicates high level of stemness regulated by TWIST1 in mesenchymal stem cells
Kensuke Toriumi, Yuta Onodera, Toshiyuki Takehara, Tatsufumi Mori, Joe Hasei, Kanae Shigi, Natsumi Iwawaki, Toshifumi Ozaki, Masao Akagi, Mahito Nakanishi, Takeshi Teramura
iScience.2023; 26(7): 106946. CrossRef - Rat Adipose-Derived Stromal Cells (ADSCs) Increases the Glioblastoma Growth and Decreases the Animal Survival
Isabele Cristiana Iser, Liziane Raquel Beckenkamp, Juliana Hofstatter Azambuja, Francine Luciano Rahmeier, Paula Andreghetto Bracco, Ana Paula Santin Bertoni, Rita de Cássia Sant’Anna Alves, Elizandra Braganhol, Léder Leal Xavier, Marilda da Cruz Fernande
Stem Cell Reviews and Reports.2022; 18(4): 1495. CrossRef - Mesenchymal/stromal stem cells: necessary factors in tumour progression
Xinyu Li, Qing Fan, Xueqiang Peng, Shuo Yang, Shibo Wei, Jingang Liu, Liang Yang, Hangyu Li
Cell Death Discovery.2022;[Epub] CrossRef - Mesenchymal stromal cells in cancer: a review of their immunomodulatory functions and dual effects on tumor progression
Sabine Galland, Ivan Stamenkovic
The Journal of Pathology.2020; 250(5): 555. CrossRef - A highly efficient non-viral process for programming mesenchymal stem cells for gene directed enzyme prodrug cancer therapy
Yoon Khei Ho, Jun Yung Woo, Geraldine Xue En Tu, Lih-Wen Deng, Heng-Phon Too
Scientific Reports.2020;[Epub] CrossRef - Fruit-Derived Polysaccharides and Terpenoids: Recent Update on the Gastroprotective Effects and Mechanisms
Mohammed Safwan Ali Khan, Syeda Umme Kulsoom Khundmiri, Syeda Rukhaiya Khundmiri, Mohammad M. Al-Sanea, Pooi Ling Mok
Frontiers in Pharmacology.2018;[Epub] CrossRef - Gastric cancer mesenchymal stem cells derived IL-8 induces PD-L1 expression in gastric cancer cells via STAT3/mTOR-c-Myc signal axis
Li Sun, Qianqian Wang, Bin Chen, Yuanyuan Zhao, Bo Shen, Hua Wang, Jing Xu, Miaolin Zhu, Xiangdong Zhao, Changgen Xu, Zhihong Chen, Mei Wang, Wenrong Xu, Wei Zhu
Cell Death & Disease.2018;[Epub] CrossRef - Mesenchymal stem cells: key players in cancer progression
Sarah M. Ridge, Francis J. Sullivan, Sharon A. Glynn
Molecular Cancer.2017;[Epub] CrossRef - Gastric cancer-derived mesenchymal stem cells prompt gastric cancer progression through secretion of interleukin-8
Wei Li, Ying Zhou, Jin Yang, Xu Zhang, Huanhuan Zhang, Ting Zhang, Shaolin Zhao, Ping Zheng, Juan Huo, Huiyi Wu
Journal of Experimental & Clinical Cancer Research.2015;[Epub] CrossRef - Mesenchymal stem cells – A new hope for radiotherapy-induced tissue damage?
Nils H. Nicolay, Ramon Lopez Perez, Juergen Debus, Peter E. Huber
Cancer Letters.2015; 366(2): 133. CrossRef
- Tumor microenvironment dynamics in gastric cancer pathogenesis and therapeutic resistance
- Gastric Adenocarcinoma of Fundic Gland Type: Report of Three Cases
- Eun Su Park, Young Eun Kim, Cheol Keun Park, Takashi Yao, Ryoji Kushima, Kyoung-Mee Kim
- Korean J Pathol. 2012;46(3):287-291. Published online June 22, 2012
- DOI: https://doi.org/10.4132/KoreanJPathol.2012.46.3.287
- 13,451 View
- 118 Download
- 25 Crossref
-
 Abstract
Abstract
 PDF
PDF Recently, fundic gland type gastric adenocarcinoma (GA-FG) has been reported as a new entity. This report describes GA-FG among Koreans for the first time. From March 2008 to July 2010 we identified only three cases of GA-FG out of over 6,000 GAs resected by endoscopy or surgery. Cell differentiation by mucin proteins, pepsinogen-I, and H+/K+-ATPase was evaluated. All three cases were male patients and diagnosed as early stage GA. Histologically, GA-FGs were well-differentiated adenocarcinoma with pale gray-blue, basophilic columnar or cuboidal cells and mildly enlarged nuclei, resembling chief cells. All three cases were positive for pepsinogen-I and were classified as gastric mucin phenotype. Among three histologic subtypes of GA-FG, since tumors were mainly composed of chief cells, our three cases were classified as chief cell predominant type. In conclusion, GA-FG is very rare among Koreans and pepsinogen-I and MUC6 expression are typical immunohistochemical findings in GA-FG suggesting differentiation toward fundic glands.
-
Citations
Citations to this article as recorded by- Endoscopic Submucosal Dissection of Early Gastric Adenocarcinoma of Fundic Gland Type: A Case Report
Ming Zhong, Wei Wei, Huang Zhong, Hang Gong, Tingyu Wang
Revista Española de Enfermedades Digestivas.2025;[Epub] CrossRef - Oxyntic Gland Neoplasms - From Adenoma to Advanced Gastric Cancer: A Review of 29 Cases
Gi Hwan Kim, Jun Su Lee, Jeong Hoon Lee, Young Soo Park
Journal of Gastric Cancer.2024; 24(4): 378. CrossRef - Transcriptome analysis reveals the essential role of NK2 homeobox 1/thyroid transcription factor 1 (NKX2-1/TTF-1) in gastric adenocarcinoma of fundic-gland type
Kazushi Fukagawa, Yu Takahashi, Nobutake Yamamichi, Natsuko Kageyama-Yahara, Yoshiki Sakaguchi, Miho Obata, Rina Cho, Nobuyuki Sakuma, Sayaka Nagao, Yuko Miura, Naoki Tamura, Daisuke Ohki, Hiroya Mizutani, Seiichi Yakabi, Chihiro Minatsuki, Keiko Niimi, Y
Gastric Cancer.2023; 26(1): 44. CrossRef - Clinicopathological Features and the Prevalence of Oxyntic Gland Neoplasm: A Single-center Retrospective Study
Hikari Asahara, Toshitatsu Takao, Yumiko Asahara, Masakyo Asahara, Douglas Motomura, Hiroya Sakaguchi, Tetsuya Yoshizaki, Nobuaki Ikezawa, Madoka Takao, Yoshinori Morita, Takashi Toyonaga, Masato Komatsu, Ryoji Kushima, Yuzo Kodama
Internal Medicine.2023; 62(19): 2763. CrossRef - Clinicopathological features of gastric adenocarcinoma of fundic gland type
Bao-Zhen Guo, Zhen-Zhen Liu, Gao-Fei Shen, Fei Zhu, Hui-Fen Lian, Xin Li, Jun-Yi Zheng, Jin-Peng Li, Shui-Miao Deng, Rui Huang
World Chinese Journal of Digestology.2023; 31(6): 244. CrossRef - Endoscopic Resection for Gastric Adenocarcinoma of the Fundic Gland Type: A Case Series
Hwa Jin Lee, Gwang Ha Kim, Dong Chan Joo, Moon Won Lee, Bong Eun Lee, Kyungbin Kim
The Korean Journal of Gastroenterology.2023; 81(6): 259. CrossRef - Gastric adenocarcinoma of the fundic gland type: A review of the literature
Zhiyong Zhai, Wei Hu, Zhaoyu Huang, Zemin Chen, Sicun Lu, Wei Gong
JGH Open.2023; 7(12): 812. CrossRef - Clinicopathological features of early stage gastric adenocarcinoma of fundic gland type
Huan Zhang, Shuyan Wang, Yongping Zhang, Fusang Ye, Chunnian Wang
Medicine.2022; 101(2): e28469. CrossRef - Gastric Adenocarcinoma of Fundic Gland Type Treated by Endoscopic Submucosal Dissection
Yong Bo Park, Gwang Ha Kim, Kyungbin Kim, Tae Kyoung Ha, Guk Bin Park, Young Min Kwak
The Korean Journal of Helicobacter and Upper Gastrointestinal Research.2021; 21(1): 82. CrossRef - Gastric epithelial neoplasm of fundic-gland mucosa lineage: proposal for a new classification in association with gastric adenocarcinoma of fundic-gland type
Hiroya Ueyama, Takashi Yao, Yoichi Akazawa, Takuo Hayashi, Koichi Kurahara, Yumi Oshiro, Masayoshi Yamada, Ichiro Oda, Shin Fujioka, Chiaki Kusumoto, Masayoshi Fukuda, Kunihisa Uchita, Tomohiro Kadota, Yasuhiro Oono, Kazuhisa Okamoto, Kazunari Murakami, Y
Journal of Gastroenterology.2021; 56(9): 814. CrossRef - Endoscopic resection is a suitable initial treatment strategy for oxyntic gland adenoma or gastric adenocarcinoma of the fundic gland type
Masaya Iwamuro, Chiaki Kusumoto, Masahiro Nakagawa, Sayo Kobayashi, Masao Yoshioka, Tomoki Inaba, Tatsuya Toyokawa, Shinichiro Hori, Shouichi Tanaka, Kazuhiro Matsueda, Takehiro Tanaka, Hiroyuki Okada
Scientific Reports.2021;[Epub] CrossRef - A series of five patients with oxyntic gland adenoma: Deciphering the clinical and histological features of these rare gastric polyps
Jerry C. Nagaputra, Tracy Jie Zhen Loh, Sangeeta Mantoo, Rafay Azhar, Vikneswaran Namasivayam, Wei Qiang Leow
Human Pathology Reports.2021; 26: 300566. CrossRef - Gastric adenocarcinoma of the fundic gland: A review of clinicopathological characteristics, treatment and prognosis
Xiang-yu Meng, Guang Yang, Cheng-ji Dong, Ru-yi Zheng
Rare Tumors.2021;[Epub] CrossRef - Gastric adenocarcinoma of the fundic gland type: clinicopathological features of eight patients treated with endoscopic submucosal dissection
Chengfang Li, Xinglong Wu, Shuang Yang, Xiaorong Yang, Jin Yao, Hong Zheng
Diagnostic Pathology.2020;[Epub] CrossRef - Multiple gastric adenocarcinoma of fundic gland type: A case report
Ou Chen, Ze-Yong Shao, Xiong Qiu, Guang-Ping Zhang
World Journal of Clinical Cases.2019; 7(18): 2871. CrossRef - Gastric Adenocarcinoma of the Fundic Gland Type
Mark A Benedict, Gregory Y Lauwers, Dhanpat Jain
American Journal of Clinical Pathology.2018; 149(6): 461. CrossRef - Oxyntic Gland Adenoma Treated by Endoscopic Mucosal Resection
In Ji Song, Jin Woo Joo, Jun Chul Park, Sung Kwan Shin, Yong Chan Lee, Sang Kil Lee
The Korean Journal of Helicobacter and Upper Gastrointestinal Research.2017; 17(2): 94. CrossRef - Chief cell‐predominant gastric polyps: a series of 12 cases with literature review
Karen Chan, Ian S Brown, Trevor Kyle, Gregory Y Lauwers, Marian Priyanthi Kumarasinghe
Histopathology.2016; 68(6): 825. CrossRef - Twelve-year natural history of a gastric adenocarcinoma of fundic gland type
Yoshinori Sato, Takashi Fujino, Akira Kasagawa, Ryo Morita, Shun-ichiro Ozawa, Yasumasa Matsuo, Tadateru Maehata, Hiroshi Yasuda, Masayuki Takagi, Fumio Itoh
Clinical Journal of Gastroenterology.2016; 9(6): 345. CrossRef - Clinicopathological features of gastric adenocarcinoma of the fundic gland (chief cell predominant type) by retrospective and prospective analyses of endoscopic findings
Takashi Chiba, Katsuaki Kato, Takayuki Masuda, Shuichi Ohara, Noriyuki Iwama, Takenobu Shimada, Daisuke Shibuya
Digestive Endoscopy.2016; 28(7): 722. CrossRef - Gastric Adenocarcinoma of the Fundic Gland Type Treated by Endoscopic Mucosal Resection: A Case Report and Review of the Literature
Eleanor Lewin, Philip Daroca, Sanjay Sikka, Tong Wu, Yukihiro Nakanishi
Case Reports in Pathology.2016; 2016: 1. CrossRef - Gastric adenocarcinoma of the fundic gland (chief cell-predominant type): A review of endoscopic and clinicopathological features
Masaki Miyazawa, Mitsuru Matsuda, Masaaki Yano, Yasumasa Hara, Fumitaka Arihara, Yosuke Horita, Koichiro Matsuda, Akito Sakai, Yatsugi Noda
World Journal of Gastroenterology.2016; 22(48): 10523. CrossRef - Oxyntic gland adenoma endoscopically mimicking a gastric neuroendocrine tumor: A case report
Tae-In Lee
World Journal of Gastroenterology.2015; 21(16): 5099. CrossRef - Oxyntic gland polyp/adenoma
Rajkumar Vajpeyi, Jyoti Dekate
Diagnostic Histopathology.2014; 20(11): 446. CrossRef - Gastric adenocarcinoma of fundic gland type with unusual behavior
Tetsuya Ueo, Hirotoshi Yonemasu, Tetsuya Ishida
Digestive Endoscopy.2014; 26(2): 293. CrossRef
- Endoscopic Submucosal Dissection of Early Gastric Adenocarcinoma of Fundic Gland Type: A Case Report
- Genetic Analysis of Epstein-Barr Virus Latent Membrane Protein 1 and Immunohistochemical Expression of Transforming Growth Factor (TGF)-beta1, TGF-betaRII, p21, p16, E2F1, Thymidylate Synthase, and NF-kappaB in Epstein-Barr Virus Encoded RNA-positive Gastric Adenocarcinoma
- Mee Yon Cho, Minseob Eom, Kwang Hwa Park, Mee Dong Kim, Seung Hoon Sung, Myoung Soo Kim, Dae Sung Kim, Sun Ju Choi
- Korean J Pathol. 2006;40(3):176-184.
- 2,260 View
- 22 Download
-
 Abstract
Abstract
 PDF
PDF - BACKGROUND
:Although clinicopathologic differences have been described between Epstein-Barr virus (EBV)-positive and negative gastric adenocarcinomas, the pathogenetic basis for these differences remains unclear. In this study, efforts were made to confirm that expression of EBV-latent membrane protein (LMP1) and immunohistochemical characteristics of EBVpositive gastric adenocarcinomas.
METHODS
We investigated genomic deletion, and RNA & protein expression of the EBV-LMP1, as well as immunohistochemical protein expression of transforming growth factor (TGF)-beta1, TGF-bata RII, p21, p16, E2F1, thymidylate synthase, and NF-kappaB in relation to EBV positive gastric adenocarcinoma.
RESULTS
A total of 38 Epstein-Barr Virus Encoded RNA-positive and 80 negative gastric carcinomas were examined. A 30 bp DNA deletion in the EBV-LMP1 gene, initiating at codon 342, was detected in 94.4% of EBVpositive cases. By RT-PCR and western blotting, EBV-LMP1 mRNA and protein expressions were absent in all cases, re-gardless of DNA deletion. No significant differences in TGF-bata1, TGF-betaRII, p21, NF-kappaB, E2F1, or thymidylate synthase expression were identified. However, the decreased expression of p16 was found in 84.2% of EBV-positive carcinomas, relative to only 57.5% of EBV-negative tumors (p=0.024).
CONCLUSION
EBV-LMP1 DNA deletion, mRNA and protein losses are highly prevalent in EBV-positive gastric adenocarcinoma among Korean patients, along with decreased p16 expression.
- Molecular Biological Characteristics of Differentiated Early Gastric Cancer on the Basis of Mucin Expression.
- Nari Shin, Hye Yeon Kim, Woo Kyung Kim, Min Gyung Park, Kyung Bin Kim, Dong Hoon Shin, Kyung Un Choi, Jee Yeon Kim, Chang Hun Lee, Gi Young Huh, Mee Young Sol, Do Youn Park
- Korean J Pathol. 2011;45(1):69-78.
- DOI: https://doi.org/10.4132/KoreanJPathol.2011.45.1.69
- 5,066 View
- 24 Download
- 2 Crossref
-
 Abstract
Abstract
 PDF
PDF - BACKGROUND
It is clear that the biologic characteristics of gastric cancer are different on the basis of mucin phenotypes. However, there are unabated controversies on the exact biologic differences of mucin expression in gastric cancer.
METHODS
We analyzed various protein expressions and microsatellite instability (MSI) status based on mucin expression in 130 differentiated early gastric adenocarcinoma cases. Furthermore, we evaluated the genomic alternation in 10 selected differentiated early gastric adenocarcinoma cases using array based comparative genomic hybridization (aCGH).
RESULTS
Intestinal mucin predominant subtype showed significantly elevated p53 protein and caudal-related homeobox 2 expression, and delocalization of beta catenin expressions compared to the gastric mucin predominant subtype. On MSI status, the gastric mucin predominant subtype more frequently showed unstable status than the intestinal mucin predominant subtype. CGH study showed more frequent chromosomal gain and loss in the intestinal mucin predominant subtype than the gastric mucin predominant subtype, albeit without statistical significance. Interestingly, there were significant differences in chromosomal alternation between four mucin phenotypes.
CONCLUSIONS
Study results suggest possible different points of biologic behaviors in early differentiated gastric adenocarcinomas by mucin expression type. -
Citations
Citations to this article as recorded by- Mucin Expression in Gastric Cancer: Reappraisal of Its Clinicopathologic and Prognostic Significance
Dae Hwan Kim, Nari Shin, Gwang Ha Kim, Geum Am Song, Tae-Yong Jeon, Dong-Heon Kim, Gregory Y. Lauwers, Do Youn Park
Archives of Pathology & Laboratory Medicine.2013; 137(8): 1047. CrossRef - Microsatellite Instability Status in Gastric Cancer: A Reappraisal of Its Clinical Significance and Relationship with Mucin Phenotypes
Joo-Yeun Kim, Na Ri Shin, Ahrong Kim, Hyun-Jeong Lee, Won-young Park, Jee-Yeon Kim, Chang-Hun Lee, Gi-Young Huh, Do Youn Park
Korean Journal of Pathology.2013; 47(1): 28. CrossRef
- Mucin Expression in Gastric Cancer: Reappraisal of Its Clinicopathologic and Prognostic Significance
- HER2 Status in Gastric Adenocarcinomas Assessed by Immunohistochemistry, Automated Silver-Enhanced In Situ Hybridization and Fluorescence In Situ Hybridization.
- Aeri Kim, Jung Min Bae, Se Won Kim, Mi Jin Gu, Young Kyung Bae
- Korean J Pathol. 2010;44(5):493-501.
- DOI: https://doi.org/10.4132/KoreanJPathol.2010.44.5.493
- 5,087 View
- 35 Download
- 3 Crossref
-
 Abstract
Abstract
 PDF
PDF - BACKGROUND
Recently, many studies have focused on human epidermal growth factor receptor 2 (HER2) status in gastric cancer due to HER2-targeted therapy using trastuzumab. We investigated HER2 overexpression and amplification and their concordance rate in Korean gastric adenocarcinomas.
METHODS
Tissue microarrays were constructed with 232 gastric adenocarcinoma samples. We performed immunohistochemistry (IHC), silver-enhanced in situ hybridization (SISH) and fluorescence in situ hybridization (FISH) for HER2.
RESULTS
IHC was negative in 94.8% (218/232), equivocal in 1.7% (4/232) and positive in 3.5% (8/232) of cases. HER2 protein expression was heterogeneous in 75% (9/12) of IHC 2+/3+ cancers. Gene amplification was observed in 6.5% (15/230) by SISH and the same 15 cases were also FISH-positive. We observed HER2 amplification in 1.4%, 27.3%, 25%, and 100% of IHC 0, 1+, 2+, and 3+ gastric adenocarcinomas, respectively. The concordance rate between IHC and SISH results was 95.7%.
CONCLUSIONS
HER2 overexpression and amplification were less frequent in gastric adenocarcinomas than breast carcinomas. Compared to breast carcinoma, (1) there may be IHC-negative but gene amplification-positive cases for HER2 and (2) frequent intratumoral heterogeneity of IHC for HER2 in gastric adenocarcinomas. -
Citations
Citations to this article as recorded by- Epidemiologic Study of Human Epidermal Growth Factor Receptor 2 Expression in Advanced/Metastatic Gastric Cancer: an Assessment of Human Epidermal Growth Factor Receptor 2 Status in Tumor Tissue Samples of Gastric and Gastro-Esophageal Junction Cancer
Kyung Won Seo, Taeyong Jeon, Sewon Kim, Sung Soo Kim, Kwanghee Kim, Byoung-Jo Suh, Sunhwi Hwang, SeongHee Choi, Seungwan Ryu, Jae Seok Min, Young-Joon Lee, Ye Seob Jee, Hyeondong Chae, Doo Hyun Yang, Sang Ho Lee
Journal of Gastric Cancer.2017; 17(1): 52. CrossRef - Synopsis on Clinical Practice Guideline of Gastric Cancer in Korea: An Evidence-Based Approach
Jun Haeng Lee, Jae G. Kim, Hye-Kyung Jung, Jung Hoon Kim, Woo Kyoung Jeong, Tae Joo Jeon, Joon Mee Kim, Young Il Kim, Keun Won Ryu, Seong-Ho Kong, Hyoung Il Kim, Hwoon-Yong Jung, Yong Sik Kim, Dae Young Zang, Jae Yong Cho, Joon Oh Park, Do Hoon Lim, Eun S
The Korean Journal of Gastroenterology.2014; 63(2): 66. CrossRef - Clinical Practice Guidelines for Gastric Cancer in Korea: An Evidence-Based Approach
Jun Haeng Lee, Jae G. Kim, Hye-Kyung Jung, Jung Hoon Kim, Woo Kyoung Jeong, Tae Joo Jeon, Joon Mee Kim, Young Il Kim, Keun Won Ryu, Seong-Ho Kong, Hyoung-Il Kim, Hwoon-Yong Jung, Yong Sik Kim, Dae Young Zang, Jae Yong Cho, Joon Oh Park, Do Hoon Lim, Eun S
Journal of Gastric Cancer.2014; 14(2): 87. CrossRef
- Epidemiologic Study of Human Epidermal Growth Factor Receptor 2 Expression in Advanced/Metastatic Gastric Cancer: an Assessment of Human Epidermal Growth Factor Receptor 2 Status in Tumor Tissue Samples of Gastric and Gastro-Esophageal Junction Cancer
- Detection of JC Virus T-Ag in Early Gastric Cancer.
- Eun Jeong Jang, Jung Sik Jang, Jae Hoon Kim, Han Ik Bae, In Soo Suh
- Korean J Pathol. 2010;44(5):456-461.
- DOI: https://doi.org/10.4132/KoreanJPathol.2010.44.5.456
- 4,451 View
- 41 Download
- 1 Crossref
-
 Abstract
Abstract
 PDF
PDF - BACKGROUND
JC virus (JCV) is a polyomavirus that commonly infects humans and can cause progressive multifocal leukoencephalopathy in immunocompromised patients. Recently, many reports have documented detection of JCV in gastrointestinal tract cancers. We investigated the presence of JCV in gastric adenocarcinoma, adenoma, and non-neoplastic gastric mucosa.
METHODS
We selected paraffin-embedded tissue from endoscopic mucosal resections performed from January 2007 to September 2008. DNA was extracted from the paraffin-embedded specimens of 30 adenocarcinomas, 20 adenomas of the stomach, and 20 non-neoplastic gastric mucosa. Polymerase chain reaction amplifications were performed using gene-specific primers to detect the JCV gene sequences, and immunohistochemical staining was performed to detect the T-antigen (T-Ag) protein.
RESULTS
The T-Ag sequence was detected in nine of 30 gastric cancers (30%), two of 20 adenomas (10%), and eight of 20 non-neoplastic gastric mucosa specimens (40%). T-Ag protein expression was found in five of 30 gastric cancers (16.7%) and one of 20 non-neoplastic gastric mucosa specimens (5%), whereas no expression was observed in any of the adenomas.
CONCLUSIONS
Although we could not detect a correlation between JCV and gastric cancer, we demonstrated the presence of JCV T-Ag expression in human gastric cancers. These findings suggest a possible role for JCV in gastric carcinogenesis. -
Citations
Citations to this article as recorded by- Associations Between Gastric Cancer Risk and Virus Infection Other Than Epstein-Barr Virus: A Systematic Review and Meta-analysis Based on Epidemiological Studies
Hui Wang, Xiao-Long Chen, Kai Liu, Dan Bai, Wei-Han Zhang, Xin-Zu Chen, Jian-Kun Hu
Clinical and Translational Gastroenterology.2020; 11(7): e00201. CrossRef
- Associations Between Gastric Cancer Risk and Virus Infection Other Than Epstein-Barr Virus: A Systematic Review and Meta-analysis Based on Epidemiological Studies
- Fine Needle Aspiration Cytology of Gastric Glomus Tumor: A Case Report.
- Dong Geun Lee, Kyu Yun Jang, Myoung Ja Chung, Woo Sung Moon, Myoung Jae Kang, Ho Sung Park
- Korean J Pathol. 2010;44(4):448-452.
- DOI: https://doi.org/10.4132/KoreanJPathol.2010.44.4.448
- 4,483 View
- 49 Download
- 2 Crossref
-
 Abstract
Abstract
 PDF
PDF - Glomus tumors of the stomach are rare and are usually found as a solitary, intramural lesion. Here, we report a case of a gastric glomus tumor in a 60-year-old woman diagnosed by endoscopic ultrasound-guided fine-needle aspiration cytology. Endoscopic ultrasound revealed a 4 x 3 cm-sized, round, isoechoic mass at the fourth layer of the gastric wall. Smears revealed cohesive clusters of small, uniform, round to polygonal cells with scant cytoplasm and round, hyperchromatic nuclei with homogeneous chromatin. Immunocytochemistry by liquid-based cytology was positive for smooth muscle actin. The cytologic diagnosis of a glomus tumor was confirmed by a specimen from the laparoscopic resection. Although the cytologic features of glomus tumors are quite distinctive, an immunocytochemical stain from a liquid-based cytology preparation can further help to ascertain the diagnosis.
-
Citations
Citations to this article as recorded by- Glomus Tumor of the Stomach: A Systematic Review and Illustrative Case Report
Andrea Pansa, Laura Samà, Laura Ruspi, Federico Sicoli, Ferdinando Carlo Maria Cananzi, Vittorio Quagliuolo
Digestive Diseases.2023; 41(1): 17. CrossRef - Cytologic analysis of a glomus tumor in the left second toe: Case report
Jay Hwang, Susan McDowell, Bradley Cole, Aaron Huber, Maria Cecilia D. Reyes
Diagnostic Cytopathology.2022;[Epub] CrossRef
- Glomus Tumor of the Stomach: A Systematic Review and Illustrative Case Report
- Mutation and Expression of DNA2 Gene in Gastric and Colorectal Carcinomas.
- Sung Hak Lee, Yoo Ri Kim, Nam Jin Yoo, Sug Hyung Lee
- Korean J Pathol. 2010;44(4):354-359.
- DOI: https://doi.org/10.4132/KoreanJPathol.2010.44.4.354
- 5,155 View
- 36 Download
- 3 Crossref
-
 Abstract
Abstract
 PDF
PDF - BACKGROUND
Deregulation of DNA repair and replication are involved in cancer development. DNA2 is a nuclease/helicase that plays roles in DNA repair and replication. The aim of this study was to explore DNA2 mutation and DNA2 protein expression in gastric cancers (GCs) and colorectal cancers (CRCs).
METHODS
We analyzed two mononucleotide repeats in DNA2 in 27 GCs with high microsatellite instability (MSI-H), 34 GCs with stable MSI (MSS), 29 CRCs with MSI-H and 35 CRCs with MSS by single-strand conformation polymorphism. We also analyzed DNA2 expression in GCs and CRCs either with MSI-H or MSS.
RESULTS
We found DNA2 mutations in two GCs (7.1%) and two CRCs with MSI-H (6.9%), but not in cancers with MSS. The mutations consisted of three cases of a c.2593delT and one of a c.2592_2593delTT, which would result in premature stopping of amino acid synthesis (p.Ser865Hisfsx6 and p.Ser865Thrfsx20, respectively). DNA2 expression was observed in 16 (80%) of the GCs and 15 (75%) of the CRCs with MSI-H, but all of the cancers with DNA2 frameshift mutations were weak or negative for DNA2.
CONCLUSIONS
Our data indicate that DNA2 mutation and loss of DNA2 expression occur in GCs and CRCs, and suggest that these alterations may contribute to cancer pathogenesis by deregulating DNA repair and replication. -
Citations
Citations to this article as recorded by- Multiple roles of DNA2 nuclease/helicase in DNA metabolism, genome stability and human diseases
Li Zheng, Yuan Meng, Judith L Campbell, Binghui Shen
Nucleic Acids Research.2020; 48(1): 16. CrossRef - Integration of multiple networks and pathways identifies cancer driver genes in pan-cancer analysis
Claudia Cava, Gloria Bertoli, Antonio Colaprico, Catharina Olsen, Gianluca Bontempi, Isabella Castiglioni
BMC Genomics.2018;[Epub] CrossRef - Replication intermediates that escape Dna2 activity are processed by Holliday junction resolvase Yen1
Gizem Ölmezer, Maryna Levikova, Dominique Klein, Benoît Falquet, Gabriele Alessandro Fontana, Petr Cejka, Ulrich Rass
Nature Communications.2016;[Epub] CrossRef
- Multiple roles of DNA2 nuclease/helicase in DNA metabolism, genome stability and human diseases
- Prognostic Value of Phosphorylated Akt and Survivin Expression in Gastric Adenocarcinoma.
- Soong Lee, Yun Cheol Kim, Hyeon Min Lee, Ki Sang Lee, Byung Chul Shin, Hyung Seok Kim, Jae Hyuk Lee, Chang Soo Park, Kyung Hwa Lee
- Korean J Pathol. 2010;44(3):252-258.
- DOI: https://doi.org/10.4132/KoreanJPathol.2010.44.3.252
- 5,079 View
- 22 Download
- 3 Crossref
-
 Abstract
Abstract
 PDF
PDF - BACKGROUND
pAkt (the phosphorylated form of the proto-oncogene protein c-akt) and survivin (human BIRC5 protein) are candidate apoptosis-related molecules that may be responsible for cancer progression. The aim of this study was to determine the expression of pAkt and survivin in malignant stomach neoplasm, and their value as prognostic indicators of cancer.
METHODS
The expression of pAkt and survivin in 144 cases of gastric cancer was detected by immunohistochemistry and compared with established clinicopathological parameters and prognosis of this disease.
RESULTS
Expression of pAkt showed significant correlations with depth of invasion, lymph node and distant metastasis, as well as the stage (p < 0.05 for all three correlations), but not with the Lauren classification. Survivin expression closely correlated with histological type, Lauren classification, depth of invasion, metastasis, and stage (p < 0.05 for all). The overall survival of patients with pAkt/survivin expression was inferior to that of patients with loss of pAkt/survivin expression. Cox multivariate analysis demonstrated a significant correlation between stage (p = 0.04), survivin expression (p = 0.02), and prognosis.
CONCLUSIONS
Patients with pAkt/survivin expression in gastric cancer are at increased risk of cancer-related mortality via the apoptosis resistance pathway. Expression of pAkt and survivin could be used as a prognostic indicator for gastric cancer. -
Citations
Citations to this article as recorded by- Transcriptome analysis reveals GPNMB as a potential therapeutic target for gastric cancer
Feifei Ren, Qitai Zhao, Bin Liu, Xiangdong Sun, Youcai Tang, Huang Huang, Lu Mei, Yong Yu, Hui Mo, Haibin Dong, Pengyuan Zheng, Yang Mi
Journal of Cellular Physiology.2020; 235(3): 2738. CrossRef - The Relationship Between Estrogen/Progesterone Receptor and Expression of mTOR/pAkt Proteins and the Analysis of Prognosis in Breast Cancer
Sun Wook Han, Moon Soo Lee, Sung Yong Kim, Gil Ho Kang, Zi Sun Kim, Cheol Wan Lim, Ji Hyun Lee, Hyun Ju Lee, Mee-Hye Oh, Min Hyuk Lee
Journal of Breast Disease.2013; 1(1): 8. CrossRef - Clinicopathological correlations of mTOR and pAkt expression in non-small cell lung cancer
Mee-Hye Oh, Hyun Ju Lee, Seol Bong Yoo, Xianhua Xu, Jae Sung Choi, Yong Hoon Kim, Seok Yeol Lee, Choon-Taek Lee, Sanghoon Jheon, Jin-Haeng Chung
Virchows Archiv.2012; 460(6): 601. CrossRef
- Transcriptome analysis reveals GPNMB as a potential therapeutic target for gastric cancer
- The Prognostic Significance of the Tumor-Infiltrating FoxP3-Positive Regulatory T Cells in Gastric Carcinoma.
- Sang Jae Noh, Shin Young Park, Kyung Ryoul Kim, Chan Young Kim, Keun Sang Kwon, Ho Sung Park, Ho Lee, Myoung Ja Chung, Woo Sung Moon, Kyu Yun Jang
- Korean J Pathol. 2010;44(1):9-15.
- DOI: https://doi.org/10.4132/KoreanJPathol.2010.44.1.9
- 4,799 View
- 42 Download
- 2 Crossref
-
 Abstract
Abstract
 PDF
PDF - BACKGROUND
Regulatory T cells (Tregs) are known to be key regulators of immune responses in patients with autoimmune disease and infection and also for attenuating antitumor immunity by the host. It has been reported that high numbers of tumor-infiltrating Tregs might be associated with poor clinical outcomes for several malignant tumors. Therefore, this study aimed to examine the impact of tumor-infiltrating Tregs on the prognosis of gastric carcinoma patients.
METHODS
The immunohistochemical staining for anti-fork head Box P3 (FoxP3) antibody was performed by using a 3 mm core from the tumor specimens of each of the 173 gastric cancer patients for constructing a tissue microarray. FoxP3-positive Tregs were quantified by calculating the numbers of positive cells per 5 high-power fields on light microscopy. Thereafter, the 173 patients were subdivided into the low Tregs group (< or = 3/5 high power fields [HPF], n = 41) and the high Tregs group (> 3/5 HPF, n = 132).
RESULTS
The high Tregs group was significantly associated with a higher stage, more invasion depth and lymph node metastasis (p = 0.009, p = 0.036, p = 0.006, respectively). The high Tregs group showed significantly poorer overall survival and event-free survival (p = 0.004, p = 0.017, respectively) on the univariate analysis. The Tregs group and the tumor, node and metastasis stage were also independent prognostic factors that were significantly associated with overall survival (p = 0.025, p < 0.001, respectively) by multivariate analysis.
CONCLUSIONS
Our results indicated that a high number of tumor-infiltrating FoxP3-positive Tregs could be an indicator of poor long term survival for gastric carcinoma patients. -
Citations
Citations to this article as recorded by- Tumor-infiltrating PD1-Positive Lymphocytes and FoxP3-Positive Regulatory T Cells Predict Distant Metastatic Relapse and Survival of Clear Cell Renal Cell Carcinoma
Myoung Jae Kang, Kyoung Min Kim, Jun Sang Bae, Ho Sung Park, Ho Lee, Myoung Ja Chung, Woo Sung Moon, Dong Geun Lee, Kyu Yun Jang
Translational Oncology.2013; 6(3): 282. CrossRef - Significance of Foxp3 Positive Regulatory T Cell and Tumor Infiltrating T Lymphocyte in Triple Negative Breast Cancer
Hanna Kang, Harin Cheong, Min Sun Cho, Heasoo Koo, Woon Sup Han, Kyung Eun Lee, Byung In Moon, Sun Hee Sung
The Korean Journal of Pathology.2011; 45(1): 53. CrossRef
- Tumor-infiltrating PD1-Positive Lymphocytes and FoxP3-Positive Regulatory T Cells Predict Distant Metastatic Relapse and Survival of Clear Cell Renal Cell Carcinoma
- Expression of Survivin in Gastric Carcinoma and its Relation to Tumor Cell Proliferation and Apoptosis.
- Wan Sik Lee, Sung Bum Cho, Jong Sun Rew, Jae Hyuk Lee, Chang Soo Park, Young Eun Joo
- Korean J Pathol. 2009;43(4):329-334.
- DOI: https://doi.org/10.4132/KoreanJPathol.2009.43.4.329
- 4,486 View
- 43 Download
- 1 Crossref
-
 Abstract
Abstract
 PDF
PDF - BACKGROUND
Survivin, a novel antiapoptotic gene has been linked with tumor development and progression in various human carcinomas including gastric carcinomas. The aim of this study was to evaluate the expression of survivin in gastric carcinoma and its correlation with tumor cell proliferation and apoptosis. METHODS: Expression of survivin was evaluated immunohistochemically in 84 surgically resected gastric carcinomas. Tumor cell apoptosis was evaluated with terminal deoxynucleotidyl transferase (TdT) mediated deoxyuridine triphosphate (dUTP) nick-end labeling (TUNEL), and Ki-67 immunostaining was used for evaluation of tumor cell proliferation. RESULTS: Expression of survivin was noted in 53.6% of the gastric carcinomas, and was significantly associated with depth of invasion, status of lymph node metastasis or tumor stage (p=0.022, 0.034, 0.040, respectively). There was an inverse correlation between survivin expression and apoptotic index (p=0.015). But there was no significant correlation between survivin expression and Ki-67 labeling index (p=0.430). CONCLUSIONS: These results suggest that survivin expression may contribute to tumor development and progression by inhibiting apoptosis in human gastric carcinoma. -
Citations
Citations to this article as recorded by- Significance of intracellular localization of survivin in cervical squamous cell lesions: Correlation with disease progression
SOO-AH KIM, RAN HONG
Oncology Letters.2014; 7(5): 1589. CrossRef
- Significance of intracellular localization of survivin in cervical squamous cell lesions: Correlation with disease progression
- beta-Catenin Expression in Gastric Carcinogenesis.
- Haeyoun Kang, Yon Rak Choi, Hoguen Kim
- Korean J Pathol. 2001;35(5):376-382.
- 2,247 View
- 17 Download
-
 Abstract
Abstract
 PDF
PDF - BACKGROUND
The molecular pathogenesis of gastric carcinoma is not yet well characterized. The purpose of this study is to assess the role of beta-catenin in gastric carcinogenesis.
METHODS
We analyzed beta-catenin expression using immunohistochemistry on 68 gastric adenomas and 34 gastric adenocarcinomas, and compared the result with pathological and molecular types of tumors and E-cadherin expression.
RESULTS
Nuclear expression of beta-catenin was noted more frequently in gastric adenomas than in carcinomas (40% vs. 21%, 0.05< or = P<1). There was no significant relationship between nuclear beta-catenin expression and histologic degree of adenoma, histologic type of carcinoma or microsatellite instability. E-cadherin expression showed significantly more frequent decrease in the membrane stainability of carcinomas compared to adenomas (P<0.01).
CONCLUSIONS
The frequent nuclear beta-catenin expression in gastric adenomas suggests that the beta-catenin alteration might play an early role in gastric carcinogenesis.
- CD24 Expression in Gastric Adenocarcinoma Is Associated with Tumor Invasiveness.
- Kyeong Cheon Jung, Jae Nam Seo, Tae Woon Kim, Young Mi Choi, Kwon Ik Oh, Hun Ho Song, Hyung Sik Shin, Young Euy Park
- Korean J Pathol. 2004;38(6):388-393.
- 2,584 View
- 17 Download
-
 Abstract
Abstract
 PDF
PDF - BACKGROUND
CD24, also referred to as the heat stable antigen in mice, is a glycosyl phosphatidylinositol- linked glycoprotein expressed by thymocytes, B cells, neutrophils and immature neuronal cells. It has been recently observed in a variety of human malignancy. Here, we demonstrated the expression of CD24 in gastric adenocarcinomas.
METHODS
A total of 40 gastric adenocarcinomas and 20 tubular adenomas were immunohistochemically examined for the expression of CD24 and matrix metalloproteinase-2 (MMP-2) proteins. The immunoreactivity of CD24 was semiquantitatively scored (0, 1+, 2+) and compared with clinicopathologic variables and MMP-2 expression in tumor cells.
RESULTS
CD24 was rarely expressed in normal gastric tissue and not expressed in tubular adenoma. In contrast, a moderate/strong expression (2+) of CD24 was observed in 25% of gastric adenocarcinomas, and 30% cases showed a weak CD24 staining (1+). Moreover, CD24 expression was significantly correlated with the depth of tumor invasion and MMP-2 expression.
CONCLUSION
These results suggest that the aberrant expression of CD24 in gastric adenocarcinomas might be associated with tumor progression and invasiveness.
- Triple Synchronous Cancers of Stomach, Pancreas, and Kidney.
- Seung Koo Lee, Byung Ha Choi, Shin Kwang Khang, Byung Sik Kim, Jooryung Huh
- Korean J Pathol. 2001;35(6):547-550.
- 2,239 View
- 10 Download
-
 Abstract
Abstract
- Synchronous occurrence of triple distinct malignant tumors in the same patient is very rare. We report a unique case of a triple cancer occurring in a 70-year-old Korean woman with synchronous signet ring cell carcinoma of the stomach, renal cell carcinoma of the conventional type of the left kidney, and invasive ductal adenocarcinoma and intraductal papillary carcinoma of the pancreas. All three cancers were successfully resected simultaneously by total gastrectomy, nephrectomy, and partial pancreatectomy with corresponding lymphadenectomies. This patient tolerated these surgical procedures well and led a normal healthy life during the 18 months of follow-up. In summary, a successful resection of synchronous triple cancers which has never been previously reported in such combination, is described.
- A Standardized Pathology Report for Gastric Cancer.
- Woo Ho Kim, Cheol Keun Park, Young Bae Kim, Youn Wha Kim, Ho Guen Kim, Han Ik Bae, Kyu Sang Song, Hee Kyung Chang, Hee Jin Chang, Yang Seok Chae
- Korean J Pathol. 2005;39(2):106-113.
- 5,111 View
- 342 Download
-
 Abstract
Abstract
 PDF
PDF - BACKGROUND
AND METHODS: The Gastrointestinal Pathology Study Group of the Korean Society of Pathologists developed a standardized pathology reporting format for gastric cancer in collaboration with the Korean Gastric Cancer Association. RESULTS: The diagnostic parameters are divided into two part: the standard part and the optional part. The standard part contains most of the items listed in the Japanese classification, the TNM classification by UICC, the WHO classification, and the Korean Gastric Cancer Association classification. Therefore, the standard part is adequate for routine surgical pathology service. We included detailed descriptions on each item.
CONCLUSIONS
The authors anticipate that this standardization can improve the diagnostic accuracy and decrease the discrepancies that occur in the pathologic diagnosis of gastric cancer. Furthermore, the standard format can encourage large scale multi-institutional collaborative studies.
- Correlation Between Expression of p53, Bcl-2 Protein and Ki-67 Labelling Index and Lymph Node Metastasis in Early Gastric Cancer.
- Joon Hyuk Choi, Young Ran Shim
- Korean J Pathol. 2002;36(1):7-12.
- 2,533 View
- 41 Download
-
 Abstract
Abstract
 PDF
PDF - BACKGROUND
The purpose of this study was to investigate the difference of cliniopathological variables and p53, bcl-2, and Ki-67 labelling index between early gastric cancer with and without lymph node metastasis.
METHODS
The authors analyzed thirty patients who had early gastric cancer confined to submucosa (sm cancer) without lymph node metastasis and thirty patients who had sm cancer with lymph node metastasis. The expression of p53 protein, bcl-2 protein and Ki-67 labelling index were evaluated by immunohistochemistry.
RESULTS
No significant correlation was found between lymph node metastasis and age, sex, tumor size, Lauren classification, histologic grade, and venous invasion. But lymphatic invasion was significantly correlated to lymph node metastasis (p<0.01). The p53 positive rate was 73.3% (22/30) and 66.7% (20/30) in sm cancer with and without lymph node metastatsis, respectively. The bcl-2 positive rate was 40.0% (12/30) and 30.0% (9/30) in sm cancer with and without lymph node metastasis, respectively. The Ki-67 labelling index (%) was 63.9+/-15.3 and 61.4+/-12.8 in sm cancer with and without lymph node metastasis, respectively. The lymph node metastasis was not significantly correlated to expression of p53 protein, bcl-2 protein or Ki-67 labelling index.
CONCLUSIONS
Expression of p53, bcl-2 protein and proliferative activity of sm cancer may not influence lymph node metastasis. Lymphatic invasion is a significant predictor of lymph node metastasis.
- Primary Choriocarcinoma of the Stomach: A Case Report.
- Eun Joo Seo, Hi Jeong Kwon, Ki Ouk Min, Keun Woo Lim, Seong Lee, Byung Kee Kim
- Korean J Pathol. 2002;36(1):55-58.
- 2,280 View
- 14 Download
-
 Abstract
Abstract
 PDF
PDF - A case of primary gastric choriocarcinoma with multiple liver metastases is described. A 54-year-old man underwent gastrectomy for an advanced cancer. An ulcerofungating tumor with extensive hemorrhagic necrosis was found in the anterior wall of the prepyloric antrum. The histologic examination revealed a biphasic pattern composed of uninucleated cyto or intermediate trophoblasts and syncytiotrophoblasts. A small area of the adenocarcinoma forming glandular structures and poorly differentiated solid sheets was also noted on the superficial portion of the tumor. Immunohistochemical staining for the beta human chorionic gonadotropin (-hCG) and the human placental lactogen (hPL) showed strong immunoreactivity, particularly in the cytoplasms of the syncytiotrophoblasts and intermediate trophoblasts, respectively. A diagnosis of the choriocarcinoma was made. Multiple hepatic nodules consistent with metastases were noted on the abdominal sonogram. The serum -hCG level of the patient was significantly increased.
- Ras Gene Mutations and Expression of ERK1 and ERK2 Proteins in Stomach Cancer.
- Jinyoung Yoo, Seok Jin Kang, Byung Kee Kim, Chang Suk Kang
- Korean J Pathol. 2002;36(2):77-83.
- 2,255 View
- 14 Download
-
 Abstract
Abstract
 PDF
PDF - BACKGROUND
We investigated stomach cancers for ras abnormalities and expression of ERK1 and ERK2 to determine their significance in the tumor development and/or progression and to evaluate their potential correlation with clinicopathologic parameters.
METHODS
Seventy gastric adenocarcinomas were studied immunohistochemically in paraffin-embedded tissue sections for the expression of ERK1 and ERK2 proteins. All tumors were further analyzed with the use of a polymerase chain reaction technique and a direct sequence analysis procedure for the presence of the mutated ras gene.
RESULTS
ERK1 and/or ERK2 was expressed in 65.7% (46/70) of the tumors; overexpression of ERK1 was observed in 38 (54.3%) tumors, whereas ERK2 was detected in 29 (41.4%). Nine (12.8%) samples demonstrated multations in the ras gene: 4 in H-ras and 5 in K-ras. Seven of the 9 (77.8%) mutated tumors were of the intestinal type. No association was established between the ras abnormalities and the overexpression of ERK1 and/or ERK2. However, the correlation between ERK2 and progression (early vs. advanced) was statistically significant (p<0.05).
CONCLUSIONS
These data indicate that ras abnormalities are uncommon events in gastric adenocarcinomas. The majority of ras mutations, however, occurred in intestinal-type tumors, supporting the notion of different molecular mechanisms involved between the intestinal-and diffuse-type lesions. Enhanced ERK2 activity may provide assistance in the determination of tumor penetration in these tumors.
- Expression of Nitric Oxide Synthase Isotypes in Advanced Gastric Carcinoma.
- Kyong Mee Kwon, Young Chae Chu, Tae Sook Hwang
- Korean J Pathol. 2002;36(6):374-371.
- 2,067 View
- 11 Download
-
 Abstract
Abstract
 PDF
PDF - BACKGROUND
Increased expression of nitric oxide synthase (NOS) isotypes is present in human tumor cell lines and solid tumor tissues. Hypoxia upregulates NOS expression, and nitric oxide (NO) induces mitogenesis among endothelial cells. NO has been known to induce vascular endothelial growth factor (VEGF) expression in carcinoma cells and to induce neovascularization in tumors.
METHODS
The expression and cellular localization of 3 isotypes of NOS was detected by immunohistochemistry in 73 advanced gastric carcinoma tissues along with adjacent normal gastric mucosa; and the relationship to known clinicopathologic parameters, microvascular density, and VEGF expression was analysed.
RESULTS
Forty-four (60.3%), 56 (76.7%), and 52 (71.2%) of the 73 cases revealed eNOS, nNOS, and iNOS expression, respectively. Intestinal type adenocarcinomas tended to have higher activity of eNOS (p=0.000) and nNOS (p=0.001) activities than did the diffuse type adenocarcinomas. All isotypes of NOS (eNOS, p=0.001; nNOS, p=0.005; iNOS, p=0.044) tended to be highly expressed when the tumor was differentiated. There was no significant relationship between any of the 3 NOS isotypes and microvascular density, whereas VEGF was closely related with microvascular density (p=0.000). The expression of VEGF was not related to with any of the NOS isotype expressions.
CONCLUSIONS
From the above results, we speculated that NO may be implicated in the early stage of the gastric carcinogenesis rather than the growth and progression stages, and NO does not appear to affect angiogenesis or VEGF expression in the advanced gastric carcinoma.
- A High Thymidylate Synthase Expression is Related to Better Outcome for Advanced Gastric Cancer Patients Treated with 5-FU Chemotherapy after Curative Resection.
- Mee Yon Cho, Sang Yeop Yi, Min seob Eom, Shu Peng Zhang, Hwan Sik Kim, Jong In Lee, Dae Sung Kim
- Korean J Pathol. 2006;40(2):128-136.
- 2,215 View
- 21 Download
-
 Abstract
Abstract
 PDF
PDF - BACKGROUND
The expressions of thymidylate synthase (TS), E2F-1, pRb, and p53 are correlated with DNA synthesis. The significance of their expressions is still controversial for predicting the outcome of 5-fluorouracil (5-FU) therapy in the patients with advanced gastric carcinoma. Furthermore, their prognostic value in the metastatic lesions of gastric carcinoma has not yet been confirmed.
METHODS
To ascertain their prognostic value, we immunohistochemically analyzed the expressions of TS, E2F-1, pRb, and p53 in the primary tumors and the related metastatic lymph nodes, and we then compared the survival between the high and low expression group of each protein. Ninety four patients with advanced gastric carcinoma who were treated by complete resection and adjuvant 5-FU chemotherapy were analyzed.
RESULTS
The TS expression in primary tumors was significantly correlated with that of E2F-1. The expression of these genes showed no significant difference between the primary tumors and the metastatic lymph nodes except for E2F-1, which was significantly higher in the lymph node metastasis than in the primary tumors. After complete resection and 5-FU-based adjuvant chemotherapy, patients with a high TS expression in the primary tumors showed a longer survival than those patients having primary tumors with a low TS expression (p=0.0392).
CONCLUSION
A high TS expression in the primary tumors may be related to a better outcome for advanced gastric cancer patients who were treated with 5-FU chemotherapy after curative resection.

 E-submission
E-submission
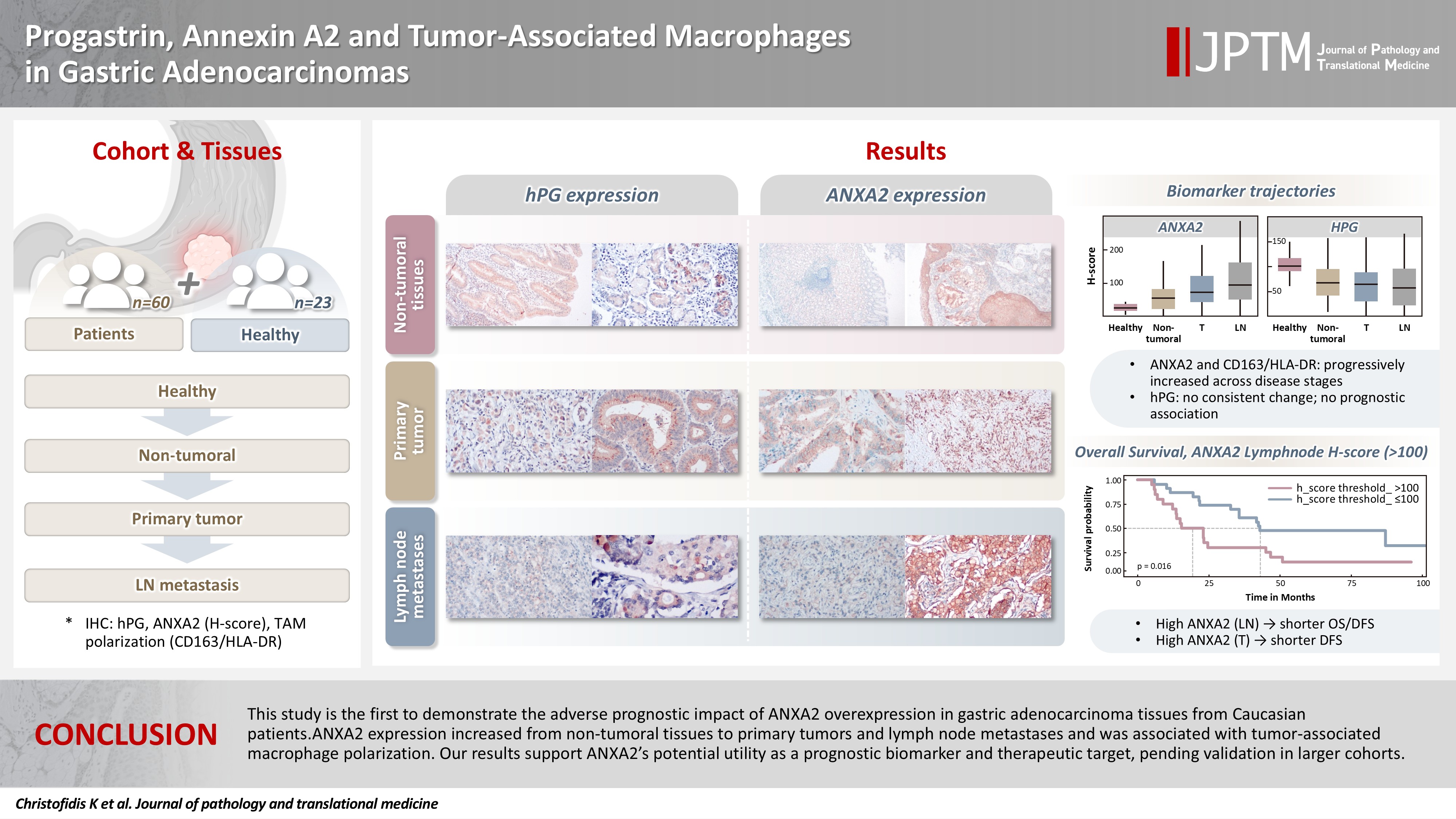






 First
First Prev
Prev



