Articles
- Page Path
- HOME > J Pathol Transl Med > Volume 56(3); 2022 > Article
-
Newsletter
What’s new in breast pathology 2022: WHO 5th edition and biomarker updates -
Kristen Muller1
 , Julie M. Jorns2
, Julie M. Jorns2 , Gary Tozbikian3
, Gary Tozbikian3
-
Journal of Pathology and Translational Medicine 2022;56(3):170-171.
DOI: https://doi.org/10.4132/jptm.2022.04.25
Published online: May 15, 2022
1Department of Pathology and Laboratory Medicine, Dartmouth-Hitchcock Medical Center, Lebanon, NH, USA
2Department of Pathology, Medical College of Wisconsin, Milwaukee, WI, USA
3Department of Pathology, The Ohio State University Wexner Medical Center, Columbus, OH, USA
- Corresponding Author: Kristen Muller, DO Department of Pathology and Laboratory Medicine, Dartmouth-Hitchcock Medical Center, Lebanon, NH, USA E-mail: kristen.e.muller@hitchcock.org
- This article has been published jointly, with consent, in both Journal of Pathology and Translational Medicine and PathologyOutlines.com.
• Received: April 23, 2022 • Accepted: April 28, 2022
© 2022 The Korean Society of Pathologists/The Korean Society for Cytopathology
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/4.0) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
- Abstract
- RARE VARIANTS OF INVASIVE BREAST CARCINOMA OF NO SPECIAL TYPE (IBC-NST)
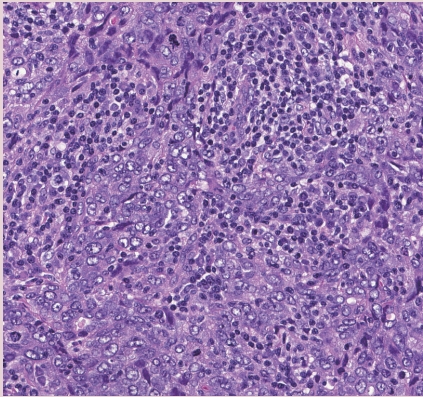
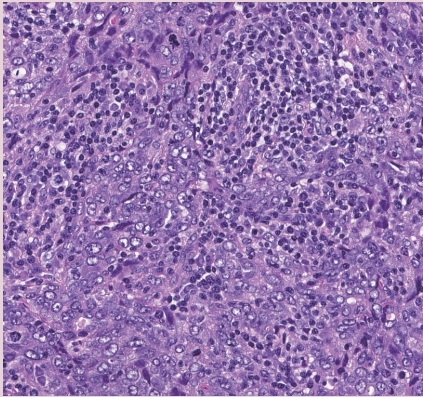
- INVASIVE BREAST CARCINOMA WITH MEDULLARY PATTERN
- NEUROENDOCRINE TUMORS
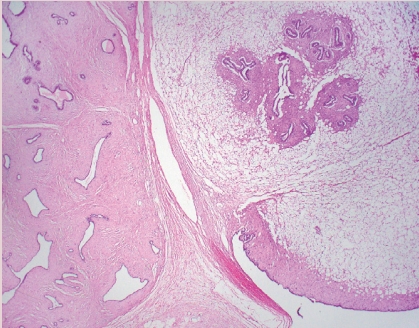
- WELL-DIFFERENTIATED LIPOSARCOMA IN PHYLLODES TUMOR
- MUCINOUS CYSTADENOCARCINOMA
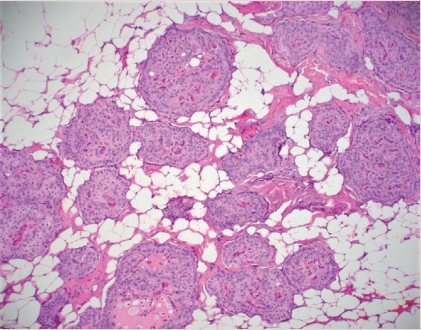
- TALL CELL CARCINOMA WITH REVERSE POLARITY (TCCRP)
- PROGNOSTIC AND PREDICTIVE BIOMARKERS UPDATE
- Meet the Authors
- REFERENCES
Figure & Data
References
Citations
Citations to this article as recorded by 

- Comparative Evaluation of Machine Learning-Based Radiomics and Deep Learning for Breast Lesion Classification in Mammography
Alessandro Stefano, Fabiano Bini, Eleonora Giovagnoli, Mariangela Dimarco, Nicolò Lauciello, Daniela Narbonese, Giovanni Pasini, Franco Marinozzi, Giorgio Russo, Ildebrando D’Angelo
Diagnostics.2025; 15(8): 953. CrossRef - Cannabinol improves exemestane efficacy in estrogen receptor-positive breast cancer models: a comparative study with cannabidiol
Cristina Ferreira Almeida, Maria João Valente, Natércia Teixeira, Susana Rocha, Ana Paula Ribeiro, Anne Marie Vinggaard, Georgina Correia-da-Silva, Cristina Amaral
European Journal of Pharmacology.2025; 1000: 177712. CrossRef - Impact of minor cannabinoids on key pharmacological targets of estrogen receptor-positive breast cancer
Cristina Ferreira Almeida, Georgina Correia-da-Silva, Ana Paula Ribeiro, Natércia Teixeira, Cristina Amaral
Biochimica et Biophysica Acta (BBA) - Molecular and Cell Biology of Lipids.2025; 1870(6): 159658. CrossRef - Reopening Pandora’s box: Is there a role for HDL in breast cancer?
Maria Isabela Bloise Alves Caldas Sawada, Monique de Fatima de Mello Santana, Milena Gomes Vancini, Marisa Passarelli
Seminars in Cancer Biology.2025; 114: 227. CrossRef - Breast cancer metastasizing to Jaw bones as the sole primary source: Systematic review
Sonia Gupta, Nausheen Aga, Aruna Vanka, Ruchira Shreevats, Muna Eisa Mohamed Hassan, Fatema Matcheswala
National Journal of Maxillofacial Surgery.2025; 16(2): 220. CrossRef - Correlation of Histopathology and Radiological Findings Among the Diverse Breast Lesions in a Tertiary Care Centre
Ranjani Mohan, Sathish Selvakumar A, Ragupathy S, Meenakshisundaram K, Shanmugapriya S, Rajeswari Kathiah, Rajeswari T, Priavadhana Rajan Prasaad, Dinesh Kumar S, Sarika K
Cureus.2024;[Epub] CrossRef - Diagnostic Challenge in Veterinary Pathology: Metastatic Mammary Tumor in a Female Tiger (Panthera Tigris)
Charisha Fraser, Mun Keong Kok, Intan Shameha Abdul Razak, Yulianna Puspitasari, Annas Salleh
Veterinary Pathology.2024; 61(4): 508. CrossRef - Expression of cell surface zinc transporter LIV1 in triple negative breast cancer is an indicator of poor prognosis and therapy failure
Roshni Saravanan, Vaishnavi Balasubramanian, Sandhya Sundaram, Bhawna Dev, Pavithra Vittalraj, Ravi Shankar Pitani, Gouthaman Shanmugasundaram, Suresh Kumar Rayala, Ganesh Venkatraman
Journal of Cellular Physiology.2024;[Epub] CrossRef - Oral Soft Tissue Metastasis from Breast Cancer as the Only Primary Source: Systematic Review
Nausheen Aga, Ruchira Shreevats, Sonia Gupta, Harman Sandhu, Muna E.M. Hassan, Harnisha V. Prajapati
Avicenna Journal of Medicine.2024; 14(01): 022. CrossRef - Influence of tumor microenvironment on the different breast cancer subtypes and applied therapies
Cristina Ferreira Almeida, Georgina Correia-da-Silva, Natércia Teixeira, Cristina Amaral
Biochemical Pharmacology.2024; 223: 116178. CrossRef - Specific feature recognition on group specific networks (SFR-GSN): a biomarker identification model for cancer stages
Bolin Chen, Yuxin Wang, Jinlei Zhang, Yourui Han, Hamza Benhammouda, Jun Bian, Ruiming Kang, Xuequn Shang
Frontiers in Genetics.2024;[Epub] CrossRef - BREAST CANCER IN THE POLTAVA REGION: CLINICAL AND MORPHOLOGICAL ASPECTS
K. R. Novykov, L. P. Lytvynenko, B. M. Fylenko, N. V. Roiko, O. K. Prylutskyi, S. A. Proskurnia
Bulletin of Problems Biology and Medicine.2024; 1(2): 9. CrossRef - Breast cancer metastasizing to salivary glands: Systematic review
Sonia Gupta, Mayur Manoharrao Shingade, Manasi Pangarkar, Annie Evangelin Nithiakumar, Pallavi Sharma, Nausheen Aga, Kinza Qureshi, Muna Eisa Mohamed Hassan, Achla Bharti Yadav
National Journal of Maxillofacial Surgery.2024; 15(2): 199. CrossRef - Granular cell tumour of the breast
Nicole Ellen James, Yue Guan, Fawaz Musa, Giulio Cuffolo
BMJ Case Reports.2024; 17(8): e258326. CrossRef - Primary extra – nodal DLBCL at rare sites: A case series
Shruti Vijayakumar, Shalini Kuruvilla, Kavitha Kanjirakkattu Mana Parameswaran, Shahin Hameed
Indian Journal of Pathology and Oncology.2024; 11(3): 289. CrossRef - Molecular Targets of Minor Cannabinoids in Breast Cancer: In Silico and In Vitro Studies
Cristina Ferreira Almeida, Andreia Palmeira, Maria João Valente, Georgina Correia-da-Silva, Anne Marie Vinggaard, Maria Emília Sousa, Natércia Teixeira, Cristina Amaral
Pharmaceuticals.2024; 17(9): 1245. CrossRef - Immune environment of high-TIL breast cancer: triple negative and hormone receptor positive HER2 negative
Su-Jin Shin, Inho Park, Heounjeong Go, Jiwon Ko, Yangkyu Lee, Jee Hung Kim, Sung Gwe Ahn, Joon Jeong, Soong June Bae, Yoon Jin Cha
npj Breast Cancer.2024;[Epub] CrossRef - Precision medicine in cancer treatment: Revolutionizing care through proteomics, genomics, and personalized therapies
Riddhi Jawdekar, Vaishnavi Mishra, Kajal Hatgoankar, Yugeshwari R. Tiwade, Nandkishor J. Bankar
Journal of Cancer Research and Therapeutics.2024; 20(6): 1687. CrossRef - Immune checkpoint inhibitor resistance in hepatocellular carcinoma
Zhijie Wang, Yichuan Wang, Peng Gao, Jin Ding
Cancer Letters.2023; 555: 216038. CrossRef - Demographic and Clinical Features of Patients with Metastatic Breast Cancer: A Retrospective Multicenter Registry Study of the Turkish Oncology Group
Izzet Dogan, Sercan Aksoy, Burcu Cakar, Gul Basaran, Ozlem Ercelep, Nil Molinas Mandel, Taner Korkmaz, Erhan Gokmen, Cem Sener, Adnan Aydiner, Pinar Saip, Yesim Eralp
Cancers.2023; 15(6): 1667. CrossRef - The role of tumor microenvironment in drug resistance: emerging technologies to unravel breast cancer heterogeneity
Vincenzo Salemme, Giorgia Centonze, Lidia Avalle, Dora Natalini, Alessio Piccolantonio, Pietro Arina, Alessandro Morellato, Ugo Ala, Daniela Taverna, Emilia Turco, Paola Defilippi
Frontiers in Oncology.2023;[Epub] CrossRef - Unraveling the Role of Adiponectin Receptors in Obesity-Related Breast Cancer
Giuseppina Daniela Naimo, Alessandro Paolì, Francesca Giordano, Martina Forestiero, Maria Luisa Panno, Sebastiano Andò, Loredana Mauro
International Journal of Molecular Sciences.2023; 24(10): 8907. CrossRef - Novel Molecular Targets for Immune Surveillance of Hepatocellular Carcinoma
Pietro Guerra, Andrea Martini, Patrizia Pontisso, Paolo Angeli
Cancers.2023; 15(14): 3629. CrossRef - Breast metastasis as the first presentation of an anorectal melanoma diagnosed on fine needle aspiration cytology: a case report
Adil Aziz Khan, Shaivy Malik, Sana Ahuja, Mukul Singh
Surgical and Experimental Pathology.2023;[Epub] CrossRef - Meme Kanserinde Ki67 İndeks Ölçümlerinin Manuel ve Dijital Yöntemler Açısından Kıyaslanması
Zuhal SİLAV
İstanbul Gelişim Üniversitesi Sağlık Bilimleri Dergisi.2023; (20): 397. CrossRef
 PubReader
PubReader ePub Link
ePub Link-
 Cite this Article
Cite this Article
- Cite this Article
-
- Close
- Download Citation
- Close
- Figure
- Related articles
-
- What's new in molecular genetic pathology 2026: emerging biomarkers for personalized cancer therapies
- What’s new in hematopathology 2025: myeloid neoplasms in the WHO 5th edition and ICC
- What’s new in neuropathology 2024: CNS WHO 5th edition updates
- What’s new in adrenal gland pathology: WHO 5th edition for adrenal cortex
- What’s new in thyroid pathology 2024: updates from the new WHO classification and Bethesda system
What’s new in breast pathology 2022: WHO 5th edition and biomarker updates



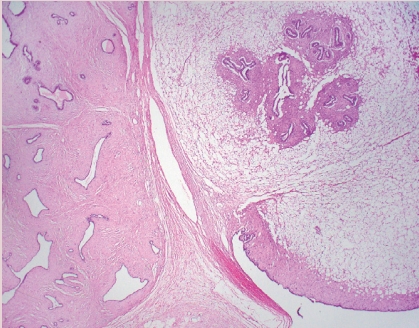
Fig. 1. IBC-NST with medullary pattern. High-grade tumor with a syncytial growth pattern and prominent TILs.
Fig. 2. Phyllodes tumor with liposarcomatous differentiation. Other features (stromal atypia, mitotic activity) in this tumor supported the designation as malignant phyllodes tumor.
Fig. 3. Tall cell carcinoma with reverse polarity (TCCRP). The tumor is characterized by infiltration of nests of tumor cells with fibrovascular cores and bland columnar cells with apically-located nuclei and abundant eosinophilic cytoplasm.
Fig. 1.
Fig. 2.
Fig. 3.
What’s new in breast pathology 2022: WHO 5th edition and biomarker updates

 E-submission
E-submission